Clinical and Neuroimaging Features in a Patient with Non-Ketotic Hyperglycemia
Abstract
1. Introduction
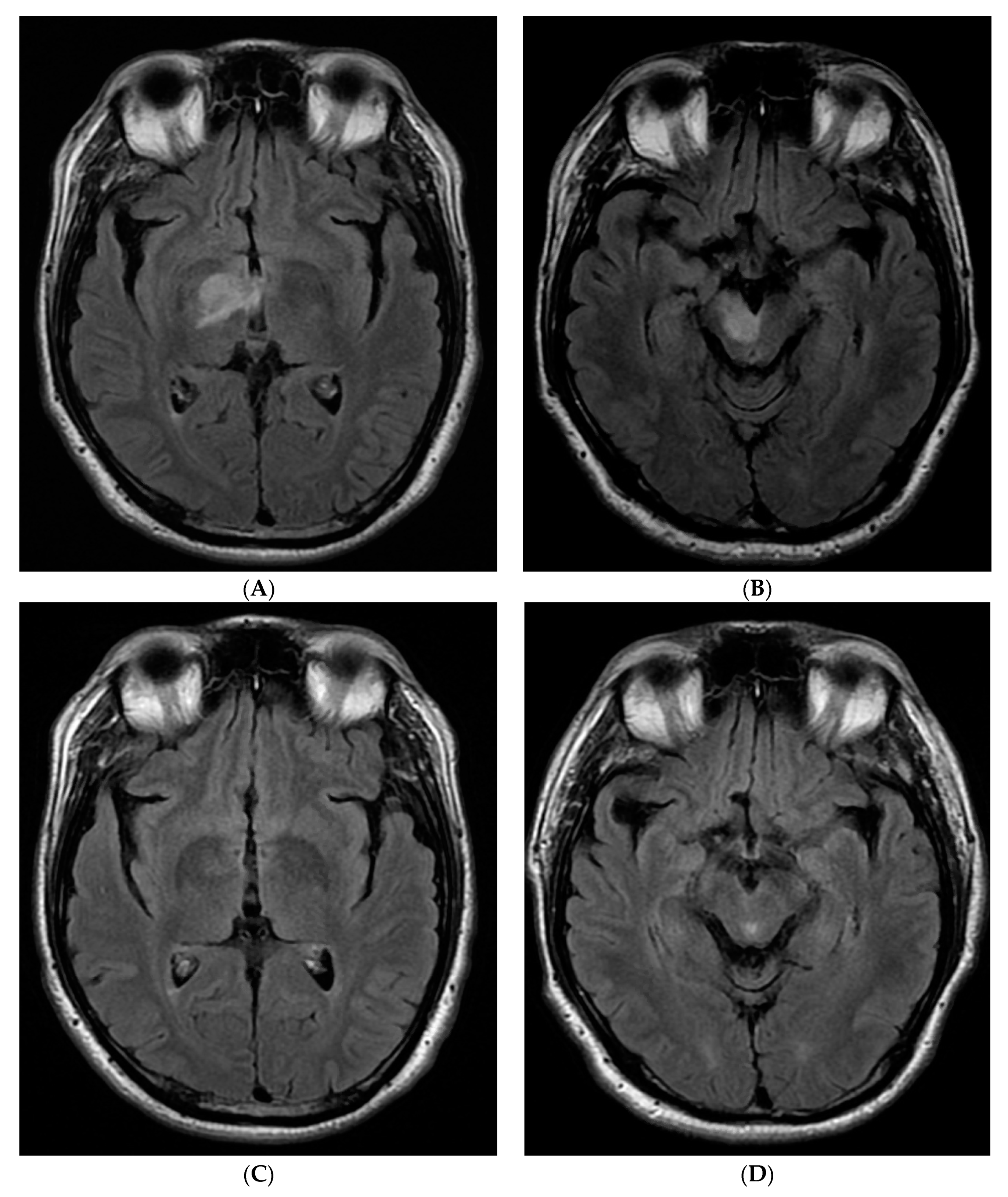
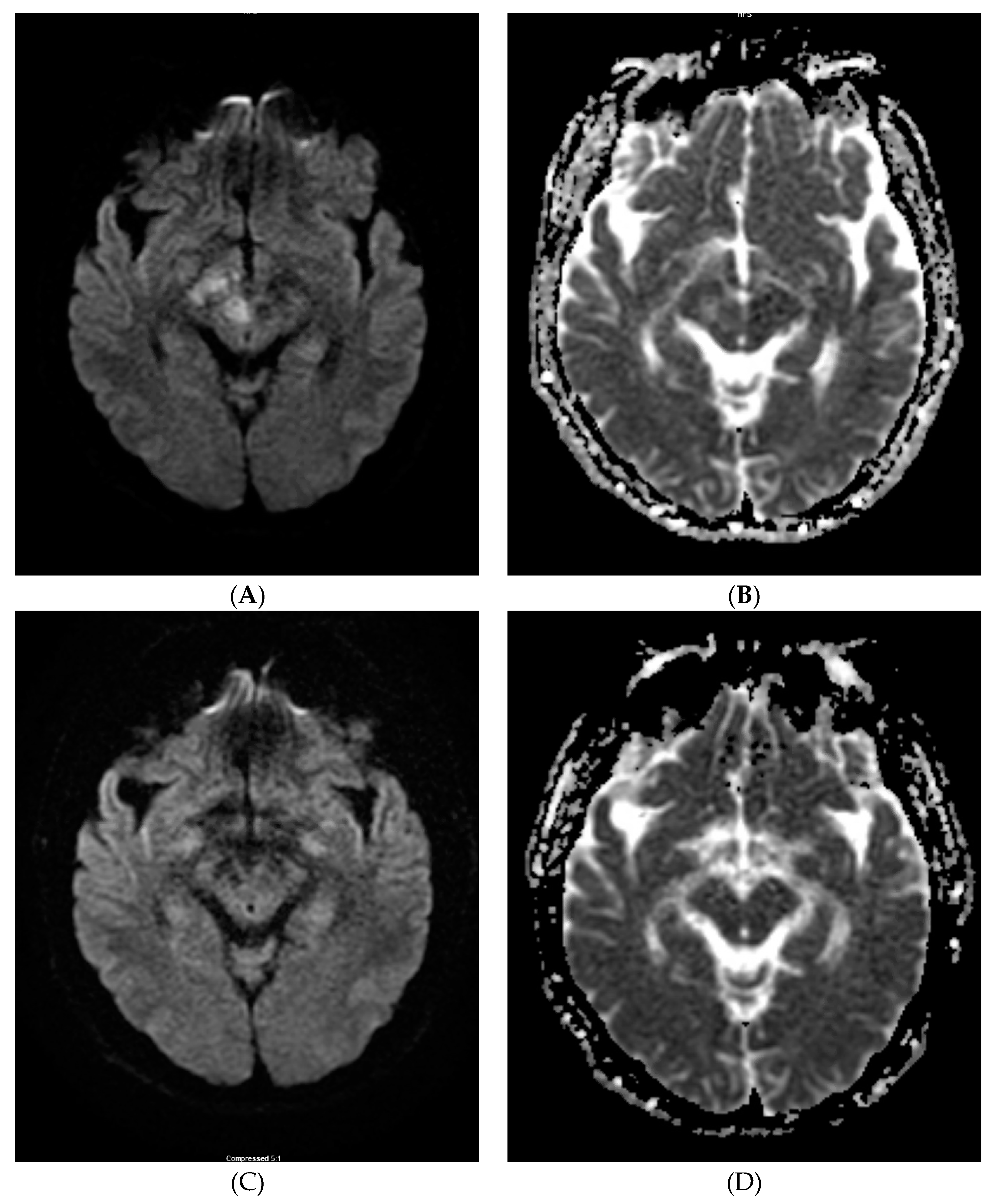
2. Case Report
3. Discussion
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| NKH | non-ketotic hyperglycemic |
| HC–HB | hemichorea and hemiballism |
| CSF | cerebrospinal fluid |
References
- Hernandez, G.T.; Cooper, C.J.; Teleb, M.; Quansah, R.; Bizet, J.; Rodriguez, E. Chorea, Hyperglycemia, Basal Ganglia Syndrome (C-H-BG) in an uncontrolled diabetic patient with normal glucose levels on presentation. Am. J. Case Rep. 2014, 15, 143–146. [Google Scholar] [CrossRef] [PubMed]
- Damani, A.; Ghoshal, A.; Salins, N.; Deodhar, J.; Muckaden, M. Management of hemichorea hemiballismus syndrome in an acute palliative care setting. Indian J. Palliat. Care 2015, 21, 72. [Google Scholar] [CrossRef] [PubMed]
- Hansford, B.G.; Albert, D.; Yang, E. Classic neuroimaging findings of nonketotic hyperglycemia on computed tomography and magnetic resonance imaging with absence of typical movement disorder symptoms (hemichorea-hemiballism). J. Radiol. Case Rep. 2013, 7, 1–9. [Google Scholar] [CrossRef]
- Qi, X.; Yan, Y.; Gao, Y.; Zheng, Z.; Chang, Y. Hemichorea associated with non-ketotic hyperglycaemia: A case report. Diabetes Res. Clin. Pract. 2012, 95, e1–e3. [Google Scholar] [CrossRef]
- Oh, S.-H.; Lee, K.-Y.; Im, J.-H.; Lee, M.-S. Chorea associated with non-ketotic hyperglycemia and hyperintensity basal ganglia lesion on T1-weighted brain MRI study. J. Neurol. Sci. 2002, 200, 57–62. [Google Scholar] [CrossRef]
- Cheema, H.; Federman, D.; Kam, A. Hemichorea–hemiballismus in non-ketotic hyperglycaemia. J. Clin. Neurosci. 2011, 18, 293–294. [Google Scholar] [CrossRef] [PubMed]
- El Otmani, H.; Moutaouakil, F.; Fadel, H.; El Ouafi, N.; Rafai, M.A.; El Moutaouakil, B.; Slassi, I. Chorea-ballismus in acute non-ketotic hyperglycaemia. Funct. Neurol. 2009, 24, 129–132. [Google Scholar] [PubMed]
- Lee, J.-H.; Park, S.-H.; Hamm, I.-S. Hemichorea-Hemiballism with Non-ketotic Hyperglycemia in a Chronic Renal Failure Patient. J. Korean Neurosurg. Soc. 2004, 36, 328–330. [Google Scholar]
- Bathla, G.; Policeni, B.; Agarwal, A. Neuroimaging in Patients with Abnormal Blood Glucose Levels. Am. J. Neuroradiol. 2014, 35, 833–840. [Google Scholar] [CrossRef] [PubMed]
- Chu, K.; Kang, D.-W.; Kim, D.-E.; Park, S.-H.; Roh, J.-K. Diffusion-weighted and gradient echo magnetic resonance findings of hemichorea-hemiballismus associated with diabetic hyperglycemia: A hyperviscosity syndrome? Arch. Neurol. 2002, 59, 448–452. [Google Scholar] [CrossRef] [PubMed]
- Jagota, P.; Bhidayasiri, R.; Lang, A.E. Movement disorders in patients with diabetes mellitus. J. Neurol. Sci. 2012, 314, 5–11. [Google Scholar] [CrossRef] [PubMed]
- Cherian, A.; Thomas, B.; Baheti, N.N.; Chemmanam, T.; Kesavadas, C. Concepts and controversies in nonketotic hyperglycemia-induced hemichorea: Further evidence from susceptibility-weighted MR imaging. J. Magn. Reson. Imaging JMRI 2009, 29, 699–703. [Google Scholar] [CrossRef]
- Chang, K.-H.; Tsou, J.-C.; Chen, S.-T.; Ro, L.-S.; Lyu, R.-K.; Chang, H.-S.; Hsu, W.-C.; Chen, C.-M.; Wu, Y.-R. Temporal features of magnetic resonance imaging and spectroscopy in non-ketotic hyperglycemic chorea-ballism patients: MRI and MRS features of non-ketotic hyperglycemic chorea-ballism. Eur. J. Neurol. 2010, 17, 589–593. [Google Scholar] [CrossRef] [PubMed]
- Lai, P.H.; Chen, P.C.; Chang, M.H.; Pan, H.; Yang, C.F.; Wu, M.T.; Li, J.Y.; Chen, C.; Liang, H.L.; Chen, W.L. In vivo proton MR spectroscopy of chorea-ballismus in diabetes mellitus. Neuroradiology 2001, 43, 525–531. [Google Scholar] [CrossRef] [PubMed]
- Roy, A.K.; Raghunandan, N.; Sarma, G.R.K.; Mathew, T. A Case Series of Hemichorea due to Non Ketotic Hyperglycemia with Unique MRI Brain Finding. J. Neurol. Neurophysiol. 2013, 2013, 11. [Google Scholar] [CrossRef]
- Wintermark, M.; Fischbein, N.J.; Mukherjee, P.; Yuh, E.L.; Dillon, W.P. Unilateral putaminal CT, MR, and diffusion abnormalities secondary to nonketotic hyperglycemia in the setting of acute neurologic symptoms mimicking stroke. AJNR Am. J. Neuroradiol. 2004, 25, 975–976. [Google Scholar] [PubMed]
- Lin, J.-J.; Lin, G.-Y.; Shih, C.; Shen, W.-C. Presentation of striatal hyperintensity on T1-weighted MRI in patients with hemiballism-hemichorea caused by non-ketotic hyperglycemia: Report of seven new cases and a review of literature. J. Neurol. 2001, 248, 750–755. [Google Scholar] [CrossRef] [PubMed]
- Yacoub, H.A. Abnormal Magnetic Resonance Imaging and Hemichorea Associated with Non-Ketotic Hyperglycemia. J. Neurol. Res. 2013, 3, 146–149. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, Y.; Parikh, A. Clinical and Neuroimaging Features in a Patient with Non-Ketotic Hyperglycemia. Neurol. Int. 2020, 12, 130-135. https://doi.org/10.3390/neurolint12030018
Zhang Y, Parikh A. Clinical and Neuroimaging Features in a Patient with Non-Ketotic Hyperglycemia. Neurology International. 2020; 12(3):130-135. https://doi.org/10.3390/neurolint12030018
Chicago/Turabian StyleZhang, Yonghua, and Aasheeta Parikh. 2020. "Clinical and Neuroimaging Features in a Patient with Non-Ketotic Hyperglycemia" Neurology International 12, no. 3: 130-135. https://doi.org/10.3390/neurolint12030018
APA StyleZhang, Y., & Parikh, A. (2020). Clinical and Neuroimaging Features in a Patient with Non-Ketotic Hyperglycemia. Neurology International, 12(3), 130-135. https://doi.org/10.3390/neurolint12030018



