M Mode Ultrasound and Tissue Doppler Imaging to Assess Diaphragm Feature in Late Onset Pompe Disease
Abstract
:1. Introduction
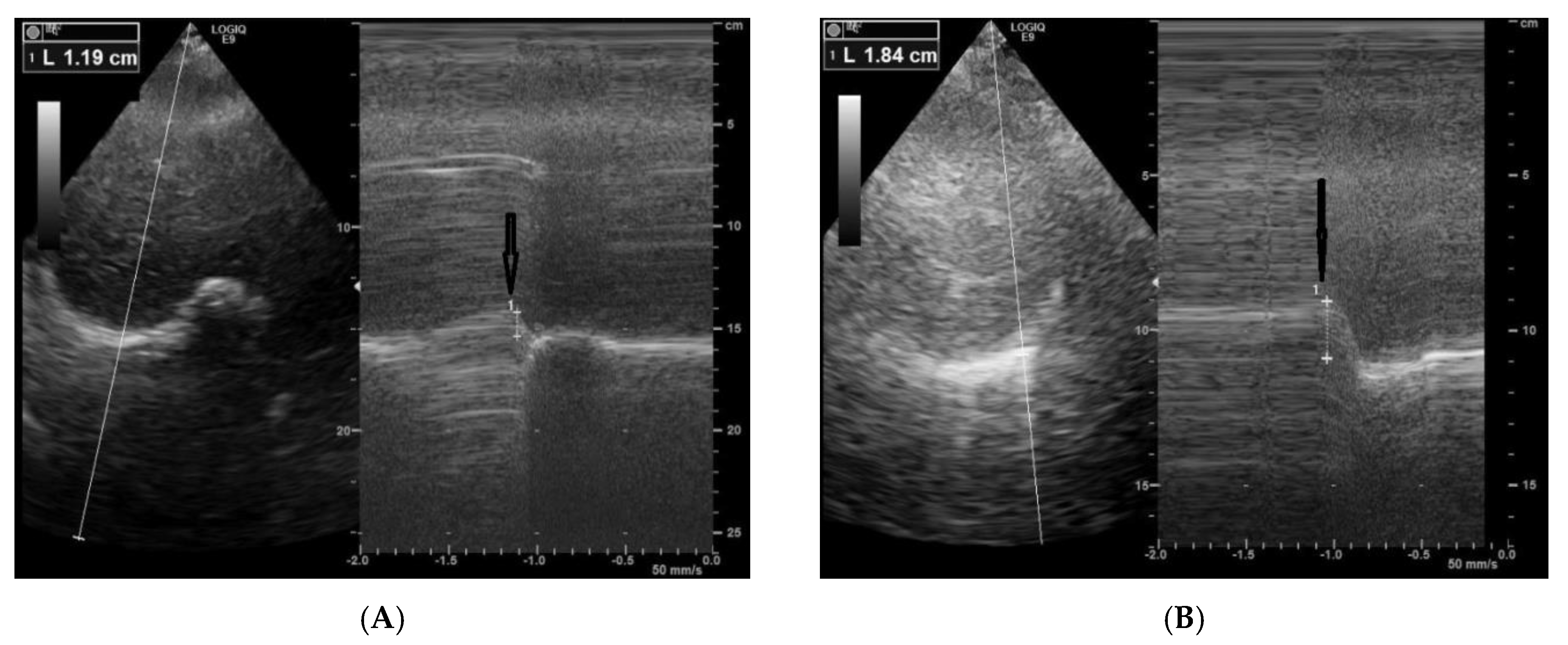
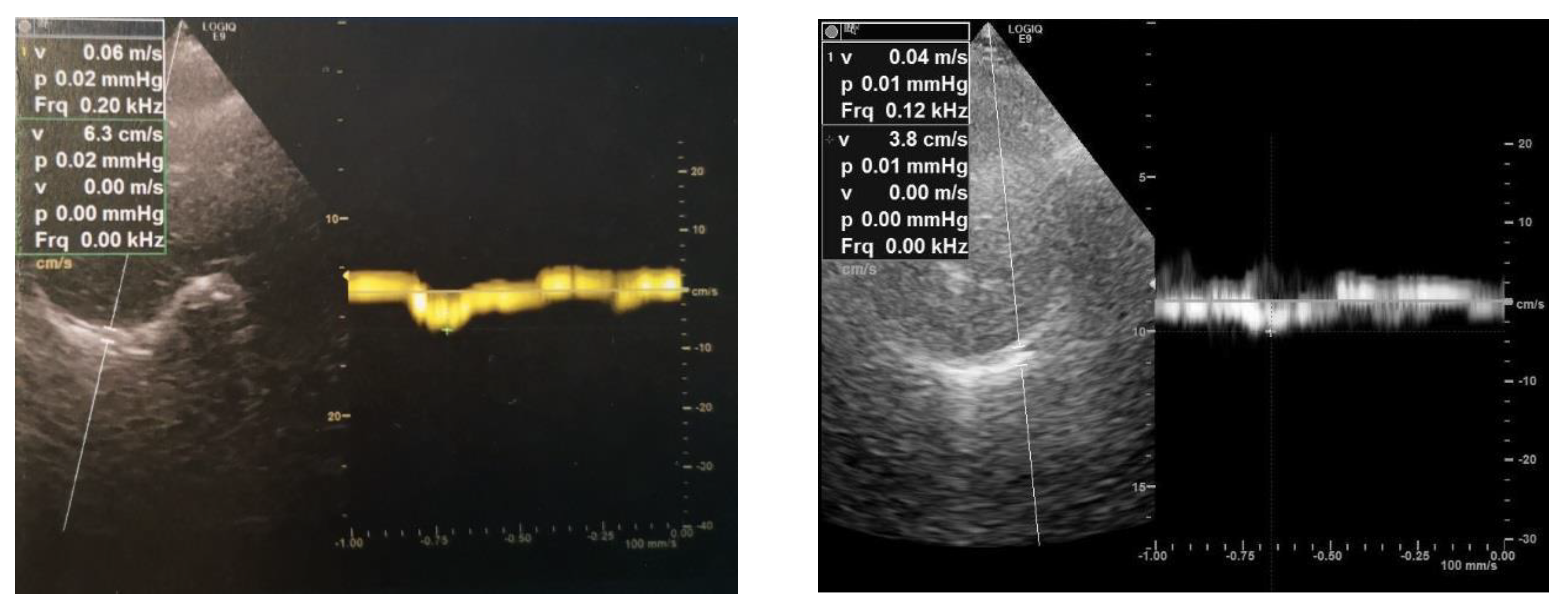
2. Case Report
3. Discussions
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Mellies, U.; Lofaso, F. Pompe disease: A neuromuscular disease with respiratory muscle involvement. Respir. Med. 2009, 103, 477–484. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Boentert, M.; Dräger, B.; Glatz, C.; Young, P. Sleep-Disordered Breathing and Effects of Noninvasive Ventilation in Patients with Late-Onset Pompe Disease. J. Clin. Sleep Med. 2016, 12, 1623–1632. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Boentert, M.; Karabul, N.; Wenninger, S.; Stubbe-Dräger, B.; Mengel, E.; Schoser, B.; Young, P. Sleep-related symptoms and sleep-disordered breathing in adult Pompe disease. Eur. J. Neurol. 2015, 22, 369–376. [Google Scholar] [CrossRef] [PubMed]
- Spiesshoefer, J.; Henke, C.; Kabitz, H.J.; Brix, T.; Görlich, D.; Herkenrath, S.; Randerath, W.; Young, P.; Boentert, M. The nature of respiratory muscle weakness in patients with late-onset Pompe disease. Neuromuscul. Disord. 2019, 29, 618–627. [Google Scholar] [CrossRef] [PubMed]
- Fayssoil, A.; Nguyen, L.S.; Ogna, A.; Stojkovic, T.; Meng, P.; Mompoint, D.; Carlier, R.; Prigent, H.; Clair, B.; Behin, A.; et al. Diaphragm sniff ultrasound: Normal values, relationship with sniff nasal pressure and accuracy for predicting respiratory involvement in patients with neuromuscular disorders. PLoS ONE 2019, 14, e0214288. [Google Scholar] [CrossRef] [PubMed]
- Van der Beek, N.A.M.E.; van Capelle, C.I.; van der Velden-van Etten, K.I.; Hop, W.C.J.; van den Berg, B.; Reuser, A.J.J.; van Doorn, P.A.; van der Ploeg, A.T.; Stam, H. Rate of progression and predictive factors for pulmonary outcome in children and adults with Pompe disease. Mol. Genet. Metab. 2011, 104, 129–136. [Google Scholar] [CrossRef] [PubMed]
- Mellies, U.; Stehling, F.; Dohna-Schwake, C.; Ragette, R.; Teschler, H.; Voit, T. Respiratory failure in Pompe disease: Treatment with noninvasive ventilation. Neurology 2005, 64, 1465–1467. [Google Scholar] [CrossRef] [PubMed]
- Fromageot, C.; Lofaso, F.; Annane, D.; Falaize, L.; Lejaille, M.; Clair, B.; Gajdos, P.; Raphaël, J.C. Supine fall in lung volumes in the assessment of diaphragmatic weakness in neuromuscular disorders. Arch. Phys. Med. Rehabil. 2001, 82, 123–128. [Google Scholar] [CrossRef] [PubMed]
- Iandelli, I.; Gorini, M.; Misuri, G.; Gigliotti, F.; Rosi, E.; Duranti, R.; Scano, G. Assessing inspiratory muscle strength in patients with neurologic and neuromuscular diseases: Comparative evaluation of two noninvasive techniques. Chest 2001, 119, 1108–1113. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kishnani, P.S.; Steiner, R.D.; Bali, D.; Berger, K.; Byrne, B.J.; Case, L.E.; Crowley, J.F.; Downs, S.; Howell, R.R.; Kravitz, R.M.; et al. Pompe disease diagnosis and management guideline. Genet. Med. 2006, 8, 267–288. [Google Scholar] [CrossRef] [Green Version]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Meng, P.; Ogna, A.; Fayssoil, A. M Mode Ultrasound and Tissue Doppler Imaging to Assess Diaphragm Feature in Late Onset Pompe Disease. Neurol. Int. 2020, 12, 55-58. https://doi.org/10.3390/neurolint12030012
Meng P, Ogna A, Fayssoil A. M Mode Ultrasound and Tissue Doppler Imaging to Assess Diaphragm Feature in Late Onset Pompe Disease. Neurology International. 2020; 12(3):55-58. https://doi.org/10.3390/neurolint12030012
Chicago/Turabian StyleMeng, Paris, Adam Ogna, and Abdallah Fayssoil. 2020. "M Mode Ultrasound and Tissue Doppler Imaging to Assess Diaphragm Feature in Late Onset Pompe Disease" Neurology International 12, no. 3: 55-58. https://doi.org/10.3390/neurolint12030012
APA StyleMeng, P., Ogna, A., & Fayssoil, A. (2020). M Mode Ultrasound and Tissue Doppler Imaging to Assess Diaphragm Feature in Late Onset Pompe Disease. Neurology International, 12(3), 55-58. https://doi.org/10.3390/neurolint12030012




