Abstract
The charging safety of electric vehicles is an area of focus in the electric automobile industry. For the purpose of ensuring safety, charging electric vehicles as soon as possible is a goal pursued by the public. In order to ensure the safety of electric vehicles during fast charging and to reduce the cycle life decay of the battery, a simplified multi-particle lithium-ion battery model is proposed, based on the pseudo two-dimensional (P2D) model. The model was developed by considering heterogeneous electrochemical reactions in the negative electrode area. The Butler–Volmer (BV) kinetic equation and the distribution of the pore wall flux in the negative electrode is approximated by the quasi-linear approximation method. Furthermore, this paper also analyzes the conditions of lithium precipitation from the negative electrode of a lithium-ion battery in the case of high charging rates, which has a certain reference significance for fast-charging control applications. The experimental and simulation results show that the model has a high simulation accuracy and can reflect the heterogeneity of electrochemical reactions in the negative electrode of the battery. The model can be adapted to fast-charging control applications.
1. Introduction
In order to deal with the global energy crisis, countries all over the world began to seek cleaner and more sustainable new energy sources. As a clean and sustainable energy, electric energy has been widely studied in the field of science and technology. At the same time, the electric vehicle industry also accelerated development. The battery is undoubtedly an important part of an electric vehicle. In order to connect the micro-changes inside the battery with the macro-changes outside the battery, it is necessary to build a battery model from the physical structure and the physical and electrochemical characteristics of the battery according to the micro-behavior of the battery during the charging and discharging process. Compared with the battery equivalent circuit model, the construction of battery physical model does not depend on a large number of experiments, and the physical meaning of battery model parameters is clear [1]. In recent years, more and more attention has been paid to battery models, which have been applied in battery state estimations and the in analysis of battery aging mechanisms.
The physical battery model is the P2D model studied by Doyle and Newman [2], which is based on porous electrode theory and concentrated solution theory. The model includes one dimension of electrode thickness direction and a pseudo second dimension of the electrode particle sphere radius direction. The P2D model offers a high simulation accuracy and can describe battery characteristics very accurately, but its calculation process is complex and its simulation time is long. Therefore, the P2D model is not directly used in practical engineering. Instead, a simplified model based on the P2D model is used. These simplified models can not only ensure simulation accuracy, but also greatly reduce the amount of calculation. Among the simplified models, the simplest is the single-particle (SP) battery model proposed by Haran [3]. In this model, a single particle is used to replace the whole electrode, and it is thought that all the electrochemical reactions and changes of liquid concentration in the cell are uniform. The SP model ignores the influence of the micro- and liquid dynamics process of the battery, which greatly improves the simulation speed of the model compared to that of the P2D model. However, the simulation accuracy of the SP model is slightly insufficient, and it can only be applied in particular contexts, such as low rate current and thin electrode. Beyond the SP model, there are the extended single-particle (ESP) battery model [4] and the multi-particle (MP) battery model [5]. Compared with the SP model, the ESP model takes into account the influence of liquid dynamics on the battery. Compared with SP model and ESP model, MP model considers the heterogeneity of electrochemical reaction in the battery.
In order to analyze the safety of the battery during fast charging and the cycle life decay, a simplified multi-particle (SMP) battery model is proposed based on the P2D battery model. The SMP model is used to analyze the lithium precipitation and aging of the negative electrode during fast charging. In the SMP model, the negative electrode is equivalent to four electrode characteristic particles in the direction of electrode thickness to reflect the inhomogeneity of the electrode. Because the SMP model focuses on the heterogeneity of the electrochemical reaction and the lithium precipitation of the battery’s negative electrode, In SMP model, all active particles in the electrode are approximately represented by one particle to reduce the complexity of the model. Compared with other simplified models, such as the SP model and the MP model, the SMP model can not only characterize the heterogeneity of the battery’s negative electrode electrochemical reaction, but also offers the advantage of high simulation accuracy. The SMP model is more suitable for use in on-board BMS to ensure safe fast charging without lithium precipitation in electric vehicles.
The nomenclature of the symbols used in this paper is shown in Table 1.

Table 1.
Nomenclature of symbols used in modeling.
The core aim of this study was to solve the heterogeneous hole wall flow of the negative electrode. Secondly, the LiCoO2 battery was used as the object to simulate the model. At the same time, conditions for the control of the precipitation of negative lithium in a lithium-ion battery in case of a high rate of charging are proposed. The simulation results show that a high simulation accuracy can be obtained from the SMP model when charging at a high rate.
2. Multi-Particle Battery Model
2.1. P2D Model
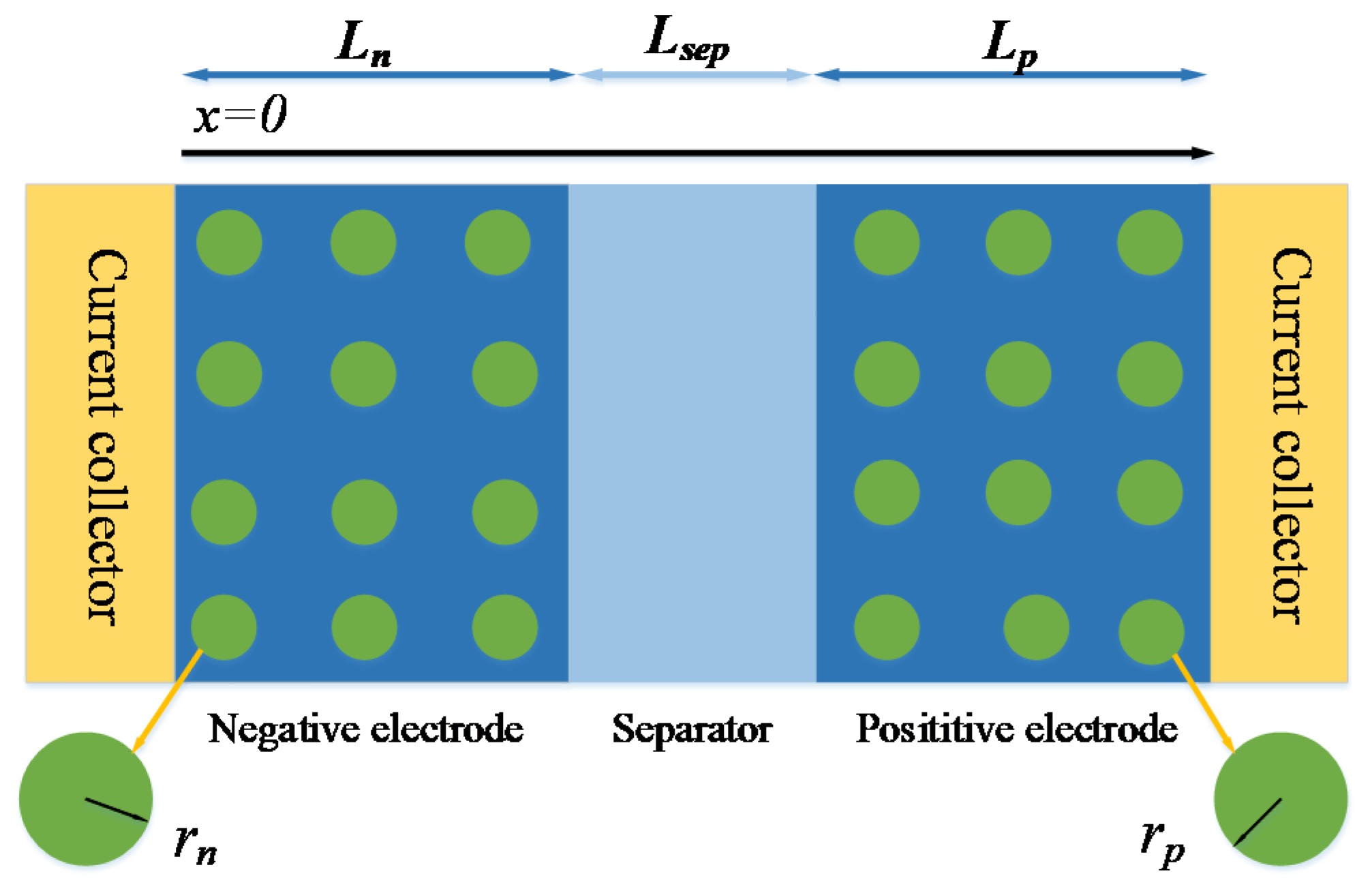
As shown in Figure 1, the P2D model divides the battery into three parts: positive, separator and negative. The positive and negative regions of the battery are represented by a series of electrode particles. The solid phase diffusion process of the battery is described by the diffusion process along the particle radius r. The liquid phase diffusion process is described by x along the thickness direction of the battery [6,7]. The mathematical expression of the P2D model is shown in Table 2.
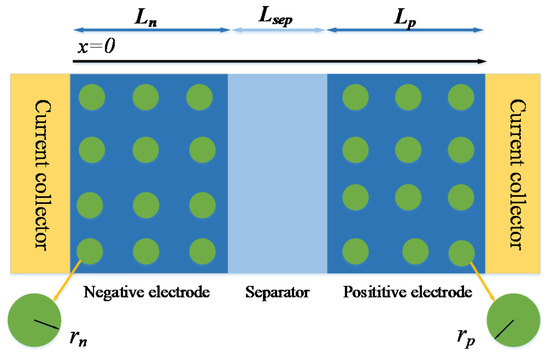
Figure 1.
Physical model of P2D.

Table 2.
The original P2D model equation.
2.2. Battery Physical Modeling
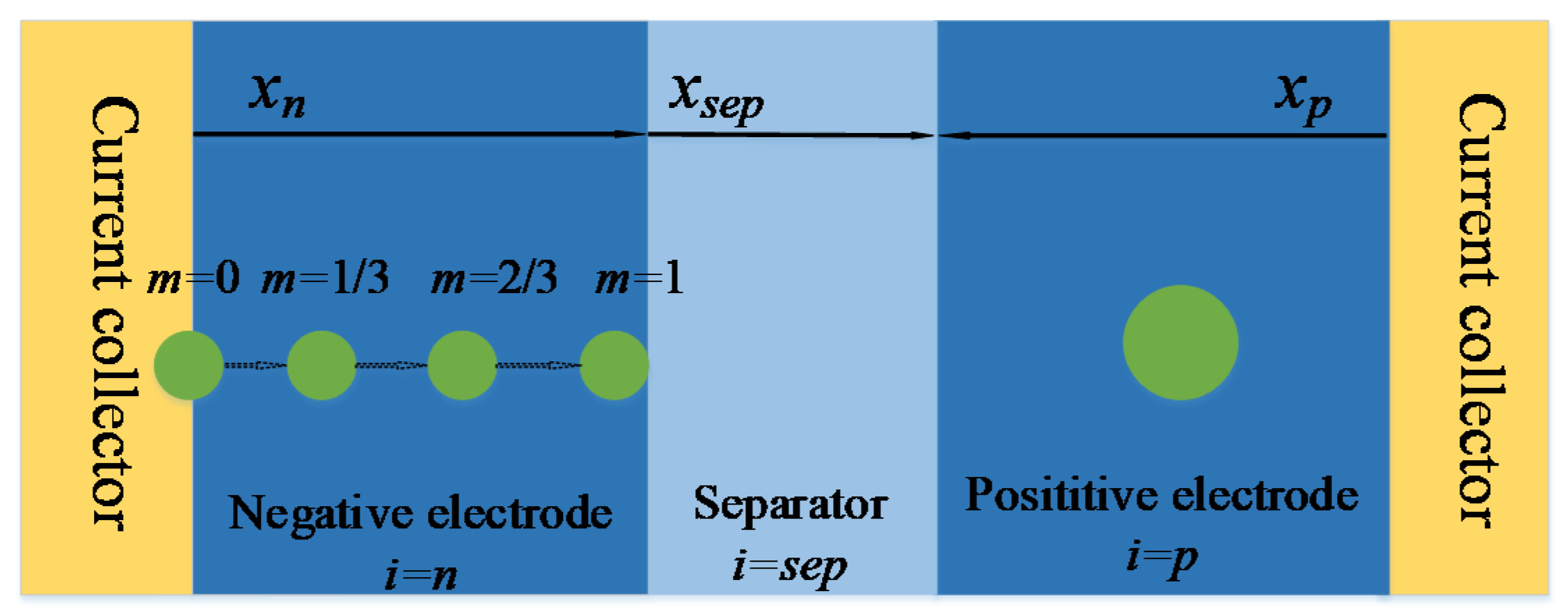
As shown in Figure 2, four characteristic points were selected in the x axis negative electrode thickness direction, and based on these four characteristic points, the heterogeneous of electrochemical reaction in the negative electrode was reflected. The positive electrode was directly modeled by a single-particle model. In order to distinguish the characteristic points of the different electrodes, subscripts i and m were used to represent the positions of the electrodes and characteristic particles on xi, respectively. i = n is negative, i = p is positive and i = sep is separator. m is 0, 1/3, 2/3 and 1, respectively [8]. Next, taking the solution of negative electrode parameters in the model as an example, the order of P2D model will be gradually reduced.
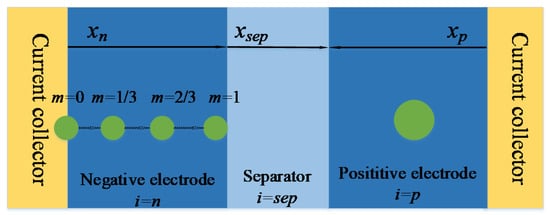
Figure 2.
Physical structure of simplified multi-particle model.
2.3. Quasi-Linearization Approximation
In the P2D model, the nonlinear equation between the electrochemical reaction potential and the flux of the pore wall is solved by using a nonlinear solver, which greatly increases the computational complexity of the model. In order to improve the speed of the model, the SMP model in this paper used the quasi-linearization approximation to approximate the BV equation. The approximate references for the BV dynamic equation and the pore wall flux are given in [3,4]. There is a nonlinear relationship between the electrochemical overpotential and the Li ion pore wall flux in the battery model is described by BV kinetic equation:
where , is the electrochemical reaction constant.
According to Taylor’s law, the BV dynamic equation is Taylor expanded at the average pore wall flux [9], and the linearized BV dynamic equation is obtained:
Taking into account the quadratic integral relationship between the electric potential and the pore wall flux, the details of the negative pore wall flux distribution have little effect on the electric potential distribution. Therefore, a piecewise linearization method is selected to measure the distribution of the pore wall flux in each electrode area. Approximate treatment:
The relationship between the average pore wall flux and the pore wall flux at each negative characteristic point can be calculated by the equation:
The equation can be expressed as a matrix equation:
where . In addition, the average pore wall flux can be obtained by an approximate calculation:
In the positive electrode, there is only one characteristic particle, so the pore wall flux of the positive electrode can be directly replaced by the average pore wall flux.
In order to find the negative pore wall flux at each characteristic particle, the potential balance equation should be introduced:
In order to solve the potential balance equation, the parameters in the potential balance equation should be calculated.
3. Potential Balance Equation Calculation
3.1. Solid and Liquid Ohmic Potential
The solution of solid and liquid potentials is universal for both positive and negative electrodes. Ignoring the influence of the electrolyte lithium-ion concentration on the liquid phase potential distribution, the liquid phase ohmic potential distribution conforms to:
And:
Integrate (9) once to obtain:
Substitute (10) into (8), and the result is written as:
Integrate (11) once to get the distribution of liquid phase potential:
According to Equation (12), the liquid phase ohmic potential at each characteristic point can be obtained, and the liquid phase potential in the three regions can also be obtained. Next, the liquid phase potential of the entire negative electrode region is:
The liquid phase potential of the dissepiment area is:
The liquid phase potential in the anode region is:
The sum of Equations (13)–(15) is the total liquid phase potential inside the battery, which can be expressed as:
Similar to the solution of liquid phase potential, the solid phase potential can also be obtained by the approximate solution method, and the solid phase potential can be expressed as:
3.2. Electromotive Force and Liquid Overpotential
The open circuit potential function of the LiCoO2 battery is obtained by reference [10].
Positive electrode:
Negative electrode:
In the SMP model, the lithium insertion rate at each characteristic particle is:
Next, the surface electromotive force of the characteristic particle can be expressed as:
Reference [10] obtained the calculation equation of electrolyte concentration difference overpotential as:
3.3. Calculation of the Heterogeneous Pore Wall Flux
The parameters and equations of the negative electrode are expressed by the following matrix:
where , are the coefficient matrices of the linearized BV equation, and is the solid-phase matrix and the liquid-phase potential matrix, respectively. Here and are given, and the solid phase matrix is similar to the liquid phase, so they are not given here.
, . After obtaining the above five equations, the pore wall flux can be derived:
where , is the reference potential.
Combining Equations (5) and (24) together, the value of the reference potential can be obtained:
Finally, the pore wall flux can be expressed as:
4. The Update of Ion Concentration
After obtaining the pore wall flux at each characteristic particle, the solid phase concentration and the liquid phase concentration are updated. This article refers to [8,9] for the update of the concentration, which are not repeated here. Because there is only one particle in the positive electrode, the non-uniformity of the positive electrode reaction can be ignored, and the average concentration can be directly used in the renewal of the concentration.
In terms of the liquid concentration, the polynomial is directly used to approximate the solution, as in [1,9]. The approximate equation can be expressed as:
where the parameters , and are solved by the boundary conditions of the liquid concentration.
In terms of the solid concentration, in order to ensure the validity and accuracy of the model under a high rate of charging, the fourth-order polynomial function is used to approximate the solid concentration [1,8]. The approximate equation can be expressed as:
Subsequently, the average solid concentration and the average concentration flux are introduced to form the state space equation to update the solid concentration. Herein, the sampling interval is set to 1s. Then, the state space equation can be expressed as:
5. Battery Terminal Voltage
After solving all the above state quantities, the terminal voltage of the battery can be obtained. The terminal voltage of the battery is determined by the solid phase potential at the current collectors on both sides of the battery and the ohmic potential of the current collector, which can be expressed as:
Taking the above-mentioned parameters of the article into the expression of the battery’s terminal voltage, the terminal voltage can be expressed as:
Substituting the calculation results of each parameter into Equation (28), the terminal voltage can be expressed as:
6. Model Simulation and Battery Experiment
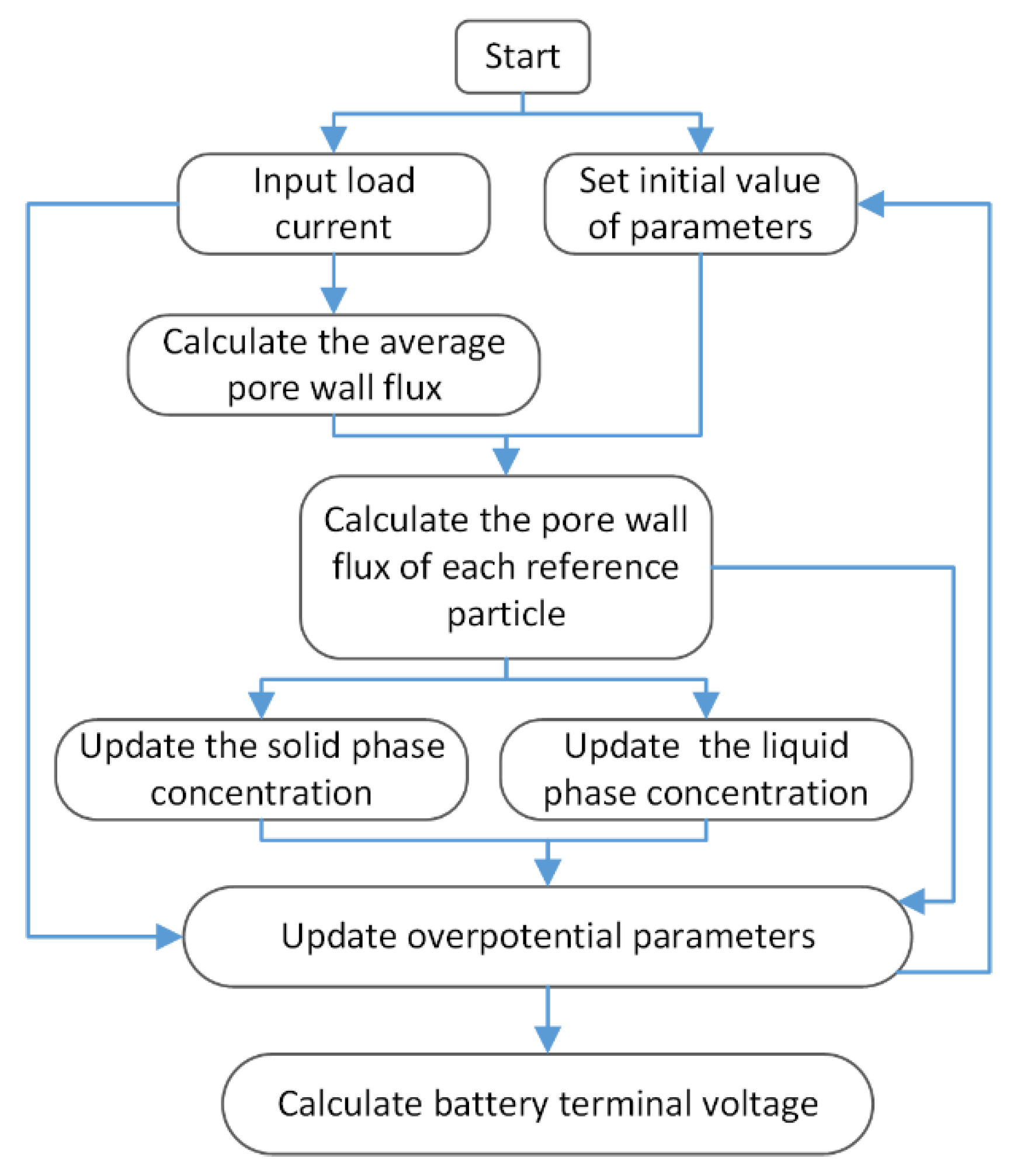
The battery experiment was carried out on a Neware battery test system at 25 °C. The experimental sample was a button-shaped LiCoO2 battery (40 mAh). The negative electrode material of the battery was graphite. All the simulation experiments in this paper were programmed on MATLAB. The calculation flowchart of the SMP model is shown in Figure 3. The simulation parameters of the model are given in Table 3.
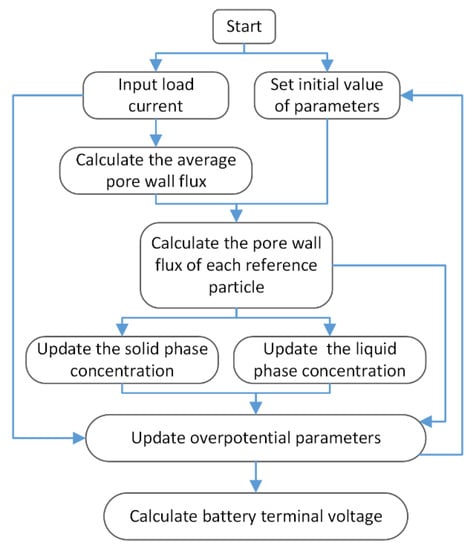
Figure 3.
Simulation flow chart of the SMP model.

Table 3.
Battery model simulation parameters and their values.
7. Results and Discussion
7.1. Terminal Voltage Results
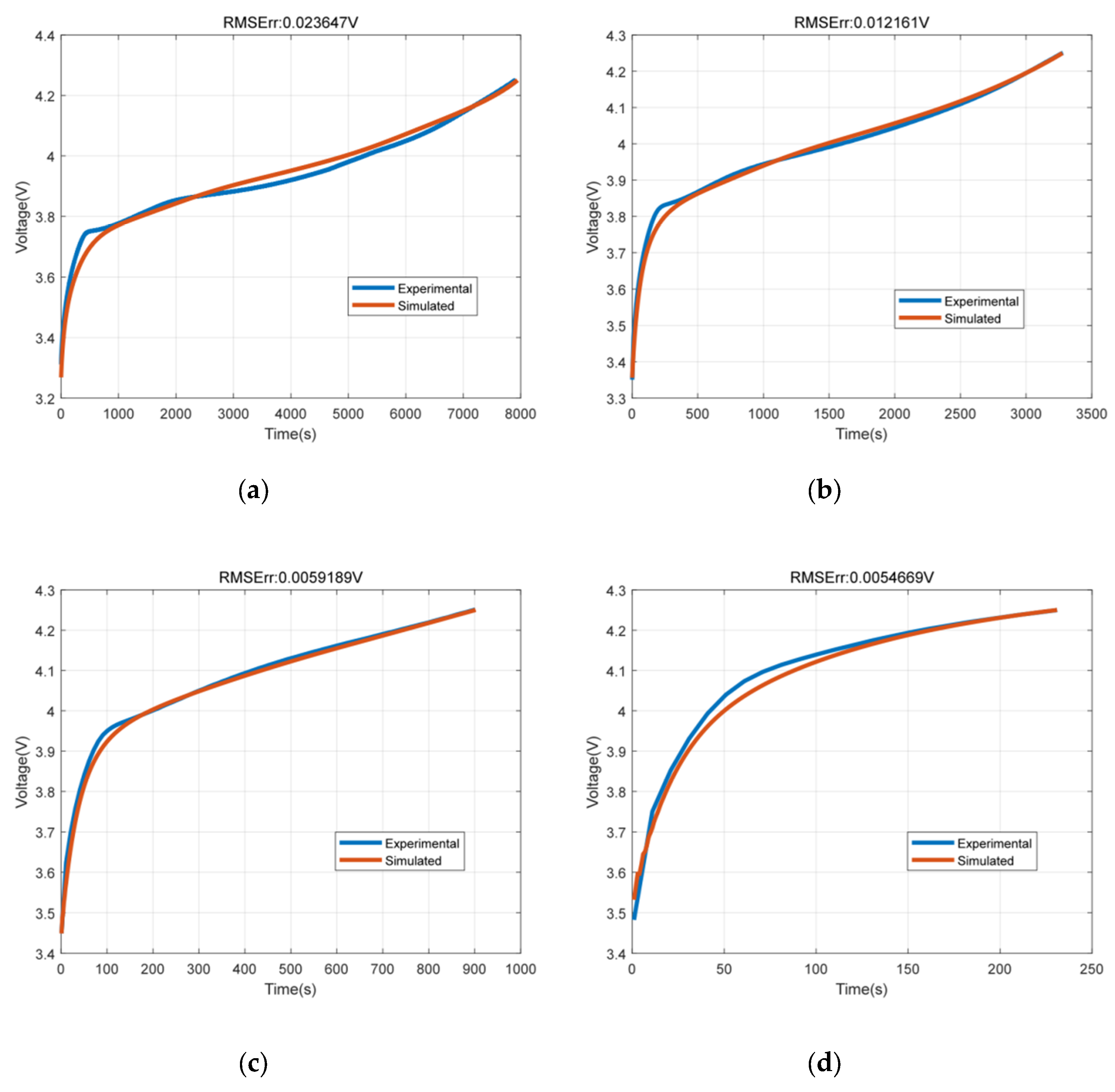
The SMP model was simulated with 0.5C, 1C, 2C and 3C current rates. The terminal voltage fitting results under the four charging currents are shown in Figure 4. It can be seen from the simulation results that the simulation results of SMP model can fit the experimental results well compared with the experimental values under four current conditions. The root mean square error (RMSE) was within 23.9 mV.
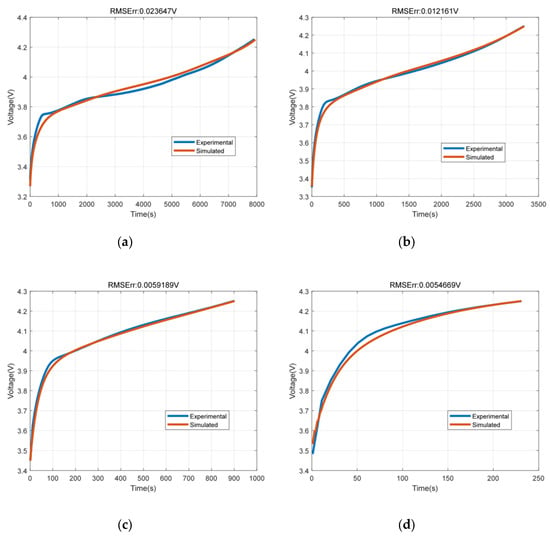
Figure 4.
The terminal voltage fitting results under the four charging currents. (a) Terminal voltage at 0.5C; (b) Terminal voltage at 1C; (c) Terminal voltage at 2C; (d) Terminal voltage at 3C.
7.2. Simulation Analysis of Lithium Precipitation
It can be seen from the simulation results that the SMP model can fit the terminal voltage value within a small margin of error compared with the experimental value under the four current conditions. In fast-charging applications, side reaction in the negative electrode lithium precipitation should be avoided to ensure safety. The side reaction overpotential is controlled by:
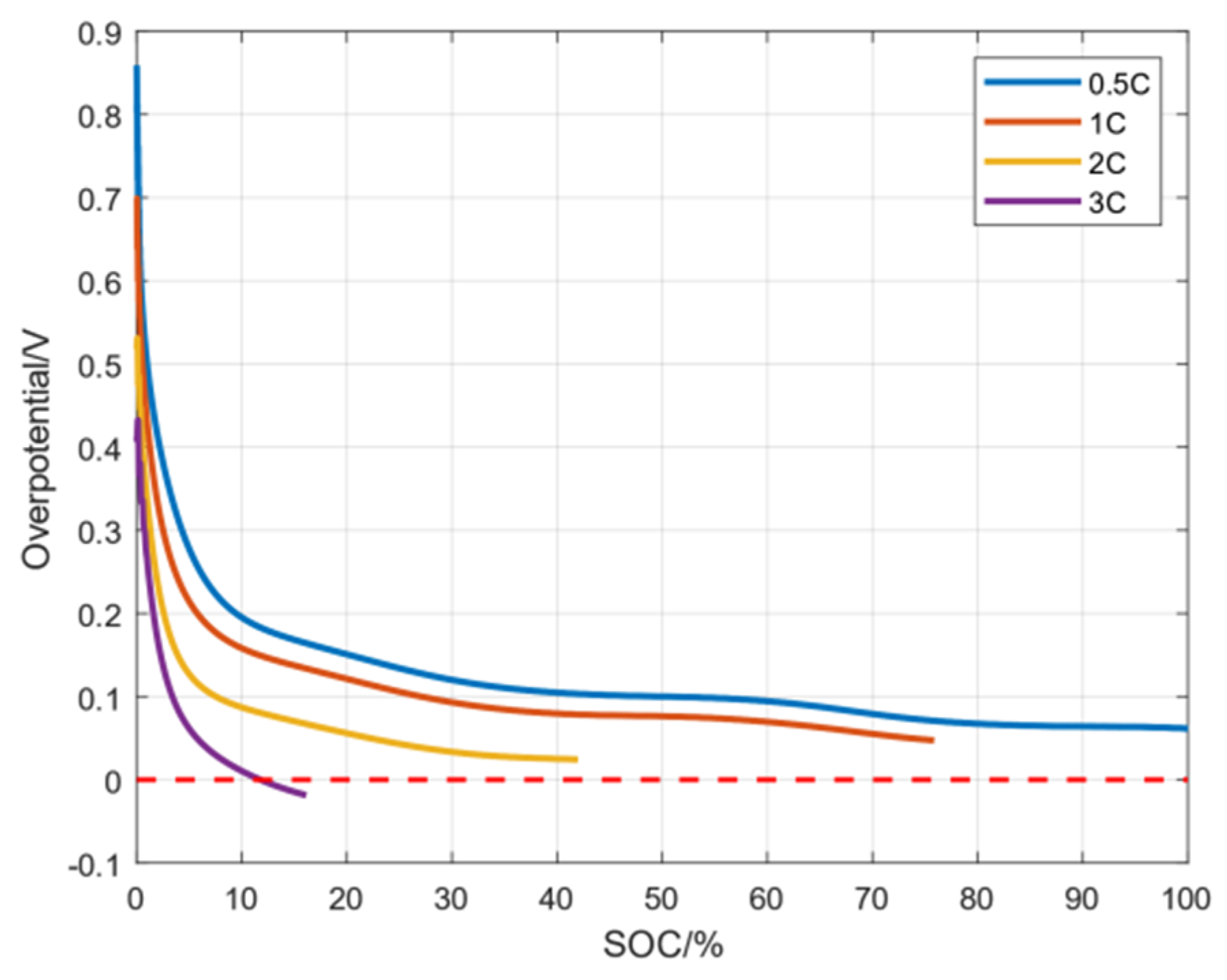
When , a negative electrode lithium precipitation side reaction does not occur. Only when does the negative electrode lithium precipitation side reaction occurs. Through simulation experiments, the average overpotential of the negative electrode lithium precipitation side reaction under three magnifications is shown in Figure 4. It can be seen from the figure that is a gradually decreasing function and eventually tends to 0 V. Moreover, the greater the charging current rate, the more easily the overpotential reaches a value below 0 V. In order to avoid a side reaction of lithium precipitation in the negative electrode, needs to be controlled above 0 V, and is expected to be as large as possible without causing damage to the battery.
As shown in Figure 5, since danger may occur during high charging rates, the battery is not fully charged under other high-rate conditions except at 0.5C. It can clearly be seen that when the charging rate is 3C, the overpotential of the electrode particles close to the diaphragm decreases to 0 V after 70% SOC, indicating that lithium precipitation has occurred on the negative electrode.
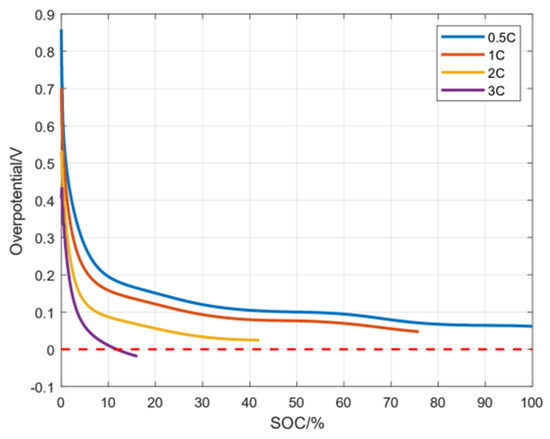
Figure 5.
The overpotential of electrode particles with m = 1 under different charging rates.
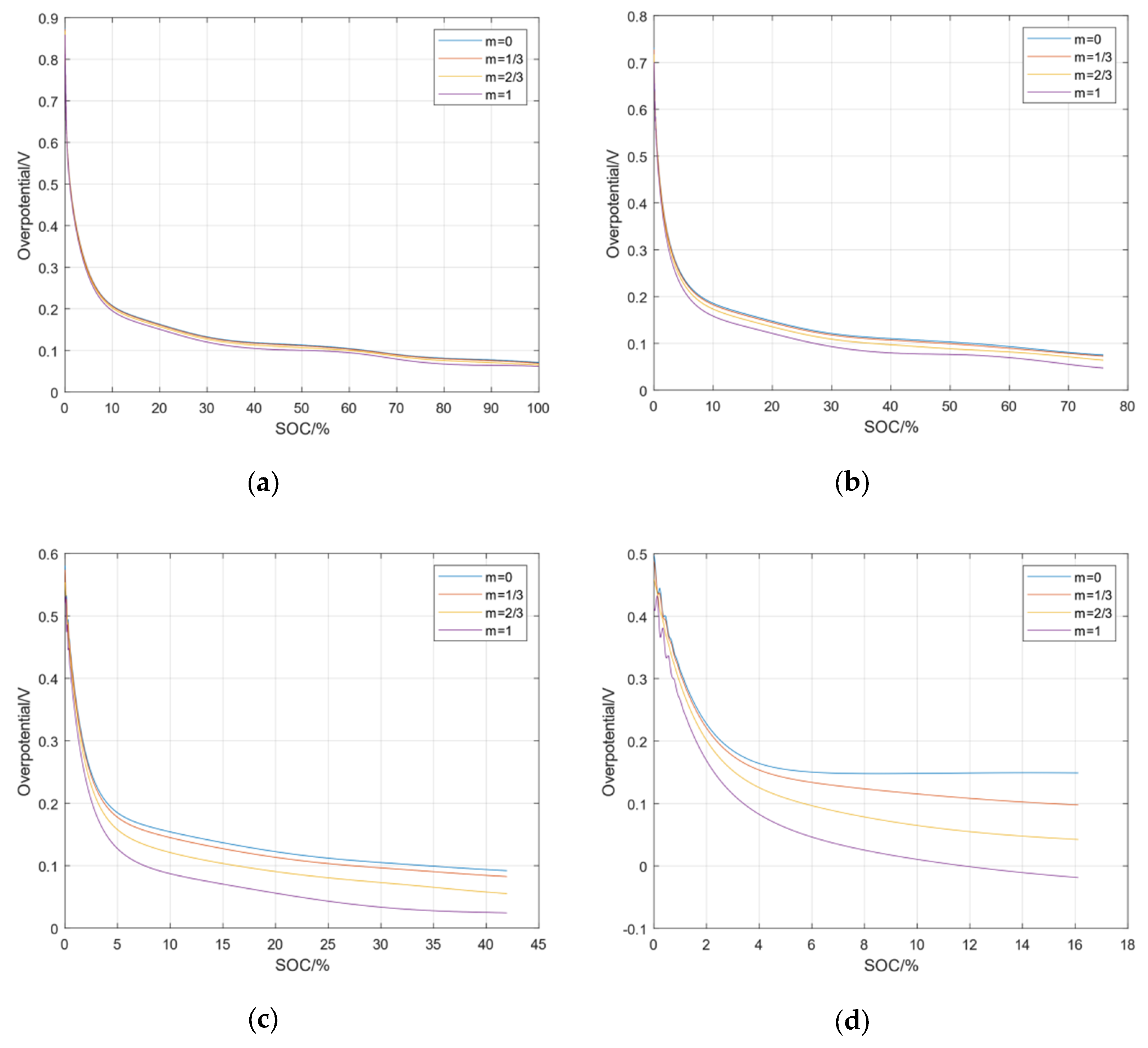
In addition to analyzing the overpotential of the negative electrode at different rates, it is also necessary to analyze the heterogeneity between the four negative particles at the same rate. Figure 6 shows the overpotentials of lithium precipitation side reactions at each particle of the negative electrode under four kinds of charging rates. It can be seen from the figure that the greater the rate of the charging current, the greater the heterogeneity of the electrochemical reaction inside the battery. When the battery is fast-charging, the reactive overpotential of each particle in the negative electrode can be used to control the lithium precipitation of different particles in the negative electrode. After analyzing the side effects of lithium precipitation, it can be applied to the fast-charging control of new energy vehicles to ensure a safe, fast charging without lithium precipitation.
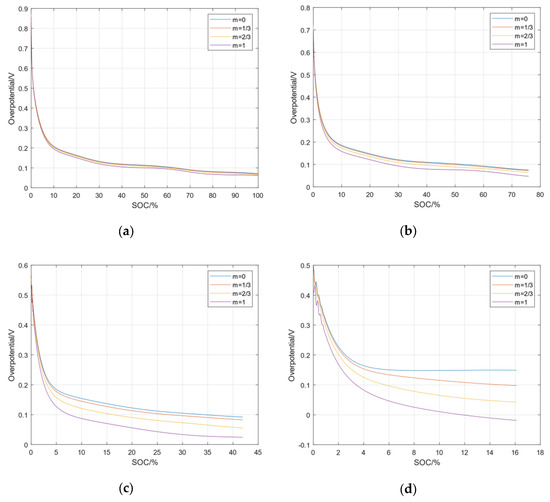
Figure 6.
The heterogeneous side reaction overpotentials in the negative electrode. (a) Overpotential at 0.5C; (b) Overpotential at 1C; (c) Overpotential at 2C; (d) Overpotential at 3C.
8. Conclusions
In this paper, in order to ensure the safety of electric vehicles during fast charging and to reduce the cycle life decay of the battery, a simplified multi-particle lithium-ion battery model is proposed based on the pseudo two-dimensional (P2D) model. The model was developed by considering the heterogeneous electrochemical reaction in the negative electrode area. The Butler–Volmer (BV) kinetic equation and the distribution of the pore wall flux in the negative electrode was approximated by the quasi-linear approximation method. The experimental and simulation results show that the model has a high simulation accuracy and can reflect the non-uniformity of electrochemical reactions in the negative electrode of the battery. The root mean square error (RMSE) of the model is within 28.2 mV for 1C to 3C current rates charging conditions. The model can be adapted to fast-charging control applications.
Author Contributions
Conceptualization, X.L.; methodology, software, data curation, and validation, X.L. and W.H.; formal analysis, X.L., J.T. and Y.T.; investigation, X.L.; resources, J.T., Y.T. and X.L.; All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by the National Natural Science Foundation of China (No. 51807121) the Natural Science Foundation of Guangdong Province (2021A1515010525), and the National Natural Science Foundation of China (No. 51807121).
Conflicts of Interest
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
References
- Subramanian, V.R.; Diwakar, V.D.; Tapriyal, D. Efficient macro-micro scale coupled modeling of batteries. J. Electrochem. Soc. 2005, 152, A2002–A2008. [Google Scholar] [CrossRef]
- Doyle, M. Modeling of galvanostatic charge and discharge of the lithium/polymer/insertion cell. J. Electrochem. Soc. 1993, 140, 1526. [Google Scholar] [CrossRef]
- Haran, B.S.; Popov, B.N.; White, R.E. Determination of the hydrogen diffusion coefficient in metal hydrides by impedance spectroscopy. J. Power Sources 1998, 75, 56–63. [Google Scholar] [CrossRef]
- Hui, P. Lithium ion battery parameter identification strategy based on extended single particle model. Acta Phys. Sin. 2018, 67, 259–269. [Google Scholar] [CrossRef]
- Majdabadi, M.M.; Farhad, S.; Farkhondeh, M.; Fraser, R.A.; Fowler, M. Simplified electrochemical multi-particle model for LiFePO4 cathodes in lithium-ion batteries. J. Power Sources 2015, 275, 633–643. [Google Scholar] [CrossRef]
- Kemper, P.; Li, S.E.; Kum, D. Simplification of pseudo two dimensional battery model using dynamic profile of lithium concentration. J. Power Sources 2015, 286, 510–525. [Google Scholar] [CrossRef]
- Jokar, A.; Rajabloo, B.; Désilets, M.; Lacroix, M. Review of simplified pseudo-two-dimensional models of lithium-ion batteries. J. Power Sources 2016, 327, 44–55. [Google Scholar] [CrossRef]
- Li, X.Y. Physical Model Simplification and Parameter Identification for State Estimation of the Retired Lithium Ion Battery; Harbin Institute of Technology: Harbin, China, 2018. [Google Scholar]
- Forman, J.C.; Bashash, S.; Stein, J.L.; Fathy, H.K. Reduction of an electrochemistry-based li-ion battery model via quasi-linearization and padé approximation. J. Electrochem. Soc. 2011, 158, A93–A101. [Google Scholar] [CrossRef]
- Han, X. Study on li-ion Battery Mechanism Model and State Estimation for Electric Vehicles; Tsinghua University: Beijing, China, 2014. [Google Scholar]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).