Artificial Intelligence (AI) in Pharmaceutical Formulation and Dosage Calculations
Abstract
1. Introduction
2. AI in Pharmaceutical Sciences
2.1. AI in Pharmaceutics
2.2. Data Requirements and Preprocessing in AI Models
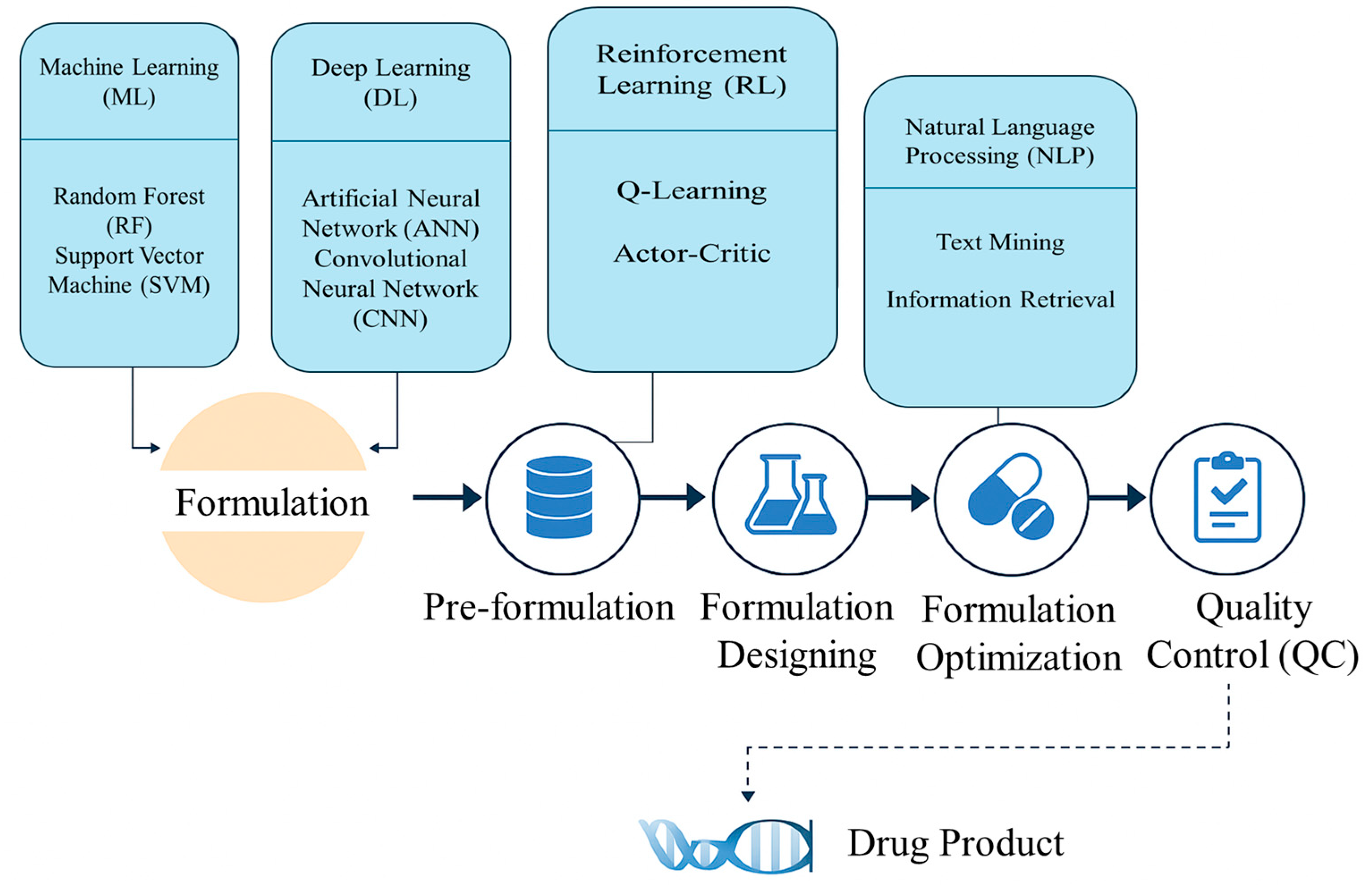
3. AI in Formulation Development
3.1. Predictive Modeling of Drug Properties
3.2. Excipient Selection and Optimization
3.3. High-Throughput Screening and Formulation Design
3.4. AI in Novel Drug Delivery Systems
4. Dose Determination and Precision Dosing
4.1. AI Systems to Personalized Dosage Based on Patient Data
4.2. Adaptive Dosing Using Real-Time Monitoring
4.3. AI in Pediatric and Geriatric Dosage Calculations
4.4. AI-Driven Dose Optimization in Clinical Trials
4.5. Drug–Drug and Drug–Disease Interaction Predictions
5. AI in Quality Control and Regulatory Compliance
5.1. AI to Verify Automated Calculation
5.2. AI in Documentation and Audit Trails
5.3. AI Monitoring Real-Time Release Testing
6. Challenges and Limitations in AI Usage
6.1. Data Quality and Availability
6.2. Interpretability and Transparency
6.3. Regulatory and Ethical Issues
6.4. Infrastructure and Skill Gaps
7. Prospects and Innovations
7.1. AI with Emerging Technologies
7.2. 3D-Printed and Personalized Pharmaceuticals
7.3. AI in Clinical Trials and Drug Repurposing
7.4. Predictive Toxicology and Safety Assessment
7.5. Quantum Computing and AI-Enhanced Molecular Simulation
8. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| AI | Artificial Intelligence |
| ANN | Artificial Neural Network |
| API | Active Pharmaceutical Ingredient |
| BO | Bayesian Optimal Interval |
| CDSS | Clinical Decision Support System |
| CNN | Convolutional Neural Network |
| CQAs | Critical Quality Attributes |
| CRM | Continual Reassessment Method |
| DDIs | Drug–Drug Interactions |
| DDzIs | Drug–Disease Interactions |
| DL | Deep Learning |
| DoE | Design of Experiment |
| EHRs | Electronic Health Records |
| GMP | Good Manufacturing Practices |
| GNN | Graph Neural Network |
| HTE | High-Throughput Experimentation |
| HTS | High-Throughput Screening |
| IoT | Internet of Things |
| LIMS | Laboratory Information Management System |
| ML | Machine Learning |
| MTD | Maximum Tolerated Dose |
| NLP | Natural Language Processing |
| OBD | Optimal Biologic Dose |
| PAT | Process Analytical Technology |
| PD | Pharmacodynamics |
| PDI | Polydispersity Index |
| PK | Pharmacokinetics |
| QSAR | Quantitative Structure–Activity Relationship |
| QbD | Quality by Design |
| RTRT | Real-Time Release Testing |
| SVM | Support Vector Machine |
| TDM | Therapeutic Drug Monitoring |
| XAI | Explainable Artificial Intelligence |
References
- Chen, H.; Engkvist, O.; Wang, Y. The rise of deep learning in drug discovery. Drug Discov. Today 2018, 23, 1241–1250. [Google Scholar] [CrossRef]
- Gupta, R.; Srivastava, D.; Sahu, M.; Tiwari, S.; Ambasta, R.K.; Kumar, P. Artificial intelligence to deep learning: Machine intelligence approach for drug discovery. Mol. Divers. 2021, 25, 1315–1360. [Google Scholar] [CrossRef]
- Wang, S.; Di, J.; Wang, D.; Dai, X.; Hua, Y.; Gao, X.; Zheng, A.; Gao, J. State-of-the-Art Review of Artificial Neural Networks to Predict, Characterize and Optimize Pharmaceutical Formulation. Pharmaceutics 2022, 14, 183. [Google Scholar] [CrossRef]
- Mak, K.K.; Pichika, M.R. Artificial intelligence in drug development: Present status and future prospects. Drug Discov. Today 2019, 24, 773–780. [Google Scholar] [CrossRef] [PubMed]
- Shen, L.; Hu, M.; Xu, X.; Zhou, Y.; Wu, W.; Ge, X.; Wang, G.; Wang, Y.; Li, Z. Precision dosing of voriconazole in immunocompromised children under 2 years: Integrated machine learning and population pharmacokinetic modeling. Front. Pharmacol. 2025, 16, 1671652. [Google Scholar] [CrossRef] [PubMed]
- Topol, E.J. The Creative Destruction of Medicine: How the Digital Revolution Will Create Better Health Care; Basic Books: New York, NY, USA, 2012. [Google Scholar]
- Ramezanpour, M.; Leung, S.S.Y.; Delgado-Magnero, K.H.; Basheti, I.A.; Chung, R. Artificial intelligence in liposome formulation: Support vector machine modeling of encapsulation efficiency. J. Control Release 2016, 226, 332–341. [Google Scholar]
- Han, R.; Ye, Z.; Zhang, Y.; Cheng, Y.; Zheng, Y.; Ouyang, D. Predicting liposome formulations by the integrated machine learning and molecular modeling approaches. Asian J. Pharm. Sci. 2023, 18, 100811. [Google Scholar] [CrossRef]
- Asgarpour, Z.; Rezaei, M.; Akbarzadeh, A. Artificial intelligence in drug delivery: Applications and challenges. Drug Discov. Today 2021, 26, 1218–1232. [Google Scholar]
- Mughal, H.; Wang, H.; Zimmerman, M.; Paradis, M.d.; Freundlich, J.S. Random Forest Model Prediction of Compound Oral Exposure in the Mouse. ACS Pharmacol. Transl. Sci. 2021, 4, 338–343. [Google Scholar] [CrossRef]
- Bohr, A.; Kristensen, J.; Dyas, M.; Edirisinghe, M. Predicting size and PDI of liposomes using deep learning approaches. Int. J. Pharm. 2020, 588, 119735. [Google Scholar]
- von Korff, M.; Sander, T. Limits of Prediction for Machine Learning in Drug Discovery. Front. Pharmacol. 2022, 13, 832120. [Google Scholar] [CrossRef]
- Frazier, P.I. A tutorial on Bayesian optimization. arXiv 2018, arXiv:1807.02811. [Google Scholar] [CrossRef]
- Waibel, I.; Schneider, T.N.; Fischer, F.J.; Dumnoenchanvanit, P.; Kulakova, A.; Nguyen, T.D.; Egebjerg, T.; Bertelsen, S.; Lorenzen, N.; Arosio, P. Bayesian Optimization for Efficient Multiobjective Formulation Development of Biologics. Mol. Pharm. 2025, 22, 6636–6645. [Google Scholar] [CrossRef]
- Zohar, S.; Chevret, S. The continual reassessment method: Comparison of Bayesian stopping rules for dose-ranging studies. Stat. Med. 2001, 20, 2827–2843. [Google Scholar] [CrossRef]
- Yang, J.; Soltan, A.A.S.; Clifton, D.A. Machine learning generalizability across healthcare settings: Insights from multi-site COVID-19 screening. NPJ Digit. Med. 2022, 5, 69. [Google Scholar] [CrossRef]
- Jain, J.; Dhanasekaran, N.; Diab, M. From Complexity to Clarity: AI/NLP’s Role in Regulatory Compliance. In Proceedings of the Findings of the Association for Computational Linguistics, ACL 2025, Vienna, Austria, 27 July–1 August 2025; pp. 26629–26641. [Google Scholar]
- Liu, X.; Song, C.; Huang, F.; Fu, H.; Xiao, W.; Zhang, W. GraphCDR: A graph neural network method with contrastive learning for cancer drug response prediction. Brief. Bioinform. 2022, 23, bbab457. [Google Scholar] [CrossRef] [PubMed]
- Rudin, C. Stop explaining black box machine learning models for high stakes decisions and use interpretable models instead. Nat. Mach. Intell. 2019, 1, 206–215. [Google Scholar] [CrossRef] [PubMed]
- Zitnik, M.; Agrawal, M.; Leskovec, J. Modeling polypharmacy side effects with graph convolutional networks. Bioinformatics 2018, 34, i457–i466. [Google Scholar] [CrossRef] [PubMed]
- Luo, H.; Yin, W.; Wang, J.; Zhang, G.; Liang, W.; Luo, J.; Yan, C. Drug-drug interactions prediction based on deep learning and knowledge graph: A review. iScience 2024, 27, 109148. [Google Scholar] [CrossRef]
- Wang, R.; Zhuang, C. Graph neural networks driven acceleration in drug discovery. Acta Pharm. Sin. B 2025. [Google Scholar] [CrossRef]
- Lim, H.; Joo, Y.; Ha, E.; Song, Y.; Yoon, S.; Shin, T. Brain Age Prediction Using Multi-Hop Graph Attention Combined with Convolutional Neural Network. Bioengineering 2024, 11, 265. [Google Scholar] [CrossRef]
- Lee, Y.J.; Kahng, H.; Kim, S.B. Generative Adversarial Networks for De Novo Molecular Design. Mol. Inform. 2021, 40, e2100045. [Google Scholar] [CrossRef]
- Gao, R.; Wen, M.; Park, J.; Cho, K. Human Mesh Reconstruction with Generative Adversarial Networks from Single RGB Images. Sensors 2021, 21, 1350. [Google Scholar] [CrossRef]
- Altae-Tran, H.; Ramsundar, B.; Pappu, A.S.; Pande, V. Low data drug discovery with one-shot learning. ACS Cent. Sci. 2017, 3, 283–293. [Google Scholar] [CrossRef]
- Wang, Y.; Jiang, C.; Wu, Y.; Lv, T.; Sun, H.; Liu, Y.; Li, L.; Pan, X. Semantic-Powered Explainable Model-Free Few-Shot Learning Scheme of Diagnosing COVID-19 on Chest X-Ray. IEEE J. Biomed. Health Inform. 2022, 26, 5870–5882. [Google Scholar] [CrossRef]
- Gautam, A.; Panwar, M.; Biswas, D.; Acharyya, A. MyoNet: A Transfer-Learning-Based LRCN for Lower Limb Movement Recognition and Knee Joint Angle Prediction for Remote Monitoring of Rehabilitation Progress From sEMG. IEEE J. Transl. Eng. Health Med. 2020, 8, 2100310. [Google Scholar] [CrossRef]
- Panwar, P.; Chaurasia, S.; Gangrade, J.; Bilandi, A.; Pruthviraja, D. Optimizing knee osteoarthritis severity prediction on MRI images using deep stacking ensemble technique. Sci. Rep. 2024, 14, 26835. [Google Scholar] [CrossRef]
- Li, X.Q.; Wang, R.Q.; Wu, L.Q.; Chen, D.M. Transfer learning-enabled outcome prediction for guiding CRRT treatment of the pediatric patients with sepsis. BMC Med. Inform. Decis. Mak. 2024, 24, 266. [Google Scholar] [CrossRef]
- Mazumdar, H.; Khondakar, K.R.; Das, S.; Halder, A.; Kaushik, A. Artificial intelligence for personalized nanomedicine; from material selection to patient outcomes. Expert. Opin. Drug Deliv. 2025, 22, 85–108. [Google Scholar] [CrossRef]
- Xu, J.; Glicksberg, B.S.; Su, C.; Walker, P.; Bian, J.; Wang, F. Federated Learning for Healthcare Informatics. J. Health Inform. Res. 2021, 5, 1–19. [Google Scholar] [CrossRef]
- Yang, S.; Hu, X.; Zhu, J.; Zheng, B.; Bi, W.; Wang, X.; Wu, J.; Mi, Z.; Wu, Y. Aspects and Implementation of Pharmaceutical Quality by Design from Conceptual Frameworks to Industrial Applications. Pharmaceutics 2025, 17, 623. [Google Scholar] [CrossRef]
- Zhang, G.; Wang, Z.; Tong, Z.; Qin, Z.; Su, C.; Li, D.; Xu, S.; Li, K.; Zhou, Z.; Xu, Y.; et al. AI hybrid survival assessment for advanced heart failure patients with renal dysfunction. Nat. Commun. 2024, 15, 6756. [Google Scholar] [CrossRef]
- Choi, Y.; Oh, S.; Huh, J.W.; Joo, H.T.; Lee, H.; You, W.; Bae, C.M.; Choi, J.H.; Kim, K.J. Deep reinforcement learning extracts the optimal sepsis treatment policy from treatment records. Commun. Med. 2024, 4, 245. [Google Scholar] [CrossRef]
- Lim, D.; Lee, W.; Kim, W.T.; Joe, I. DRL-OS: A Deep Reinforcement Learning-Based Offloading Scheduler in Mobile Edge Computing. Sensors 2022, 22, 9212. [Google Scholar] [CrossRef]
- Li, Q.; Yang, Z.; Chen, K.; Zhao, M.; Long, H.; Deng, Y.; Hu, H.; Jia, C.; Wu, M.; Zhao, Z.; et al. Human-multimodal deep learning collaboration in ‘precise’ diagnosis of lupus erythematosus subtypes and similar skin diseases. J. Eur. Acad. Dermatol. Venereol. 2024, 38, 2268–2279. [Google Scholar] [CrossRef]
- Han, Y.; Deng, C.; Huang, G.B. Editorial: Brain-inspired cognition and understanding for next-generation AI: Computational models, architectures and learning algorithms. Front. Neurosci. 2023, 17, 1169027. [Google Scholar] [CrossRef]
- He, X.; Zhao, K.; Chu, X. Automated machine learning: Methods, systems, and challenges. Nat. Rev. Methods Primers 2021, 1, 29. [Google Scholar]
- Luo, J.; Luo, Y.; Cheng, X.; Liu, X.; Wang, F.; Fang, F.; Cao, J.; Liu, W.; Xu, R. Prediction of biological nutrients removal in full-scale wastewater treatment plants using H2O automated machine learning and back propagation artificial neural network model: Optimization and comparison. Bioresour. Technol. 2023, 390, 129842. [Google Scholar] [CrossRef]
- Bruynseels, K.; Santoni de Sio, F.; van den Hoven, J. Digital Twins in Health Care: Ethical Implications of an Emerging Engineering Paradigm. Front. Genet. 2018, 9, 31. [Google Scholar] [CrossRef]
- Corral-Acero, J.; Margara, F.; Marciniak, M.; Rodero, C.; Loncaric, F.; Feng, Y.; Gilbert, A.; Fernandes, J.F.; Bukhari, H.A.; Wajdan, A.; et al. The ‘Digital Twin’ to enable the vision of precision cardiology. Eur. Heart J. 2020, 41, 4556–4564. [Google Scholar] [CrossRef]
- LeCun, Y.; Bengio, Y.; Hinton, G. Deep learning. Nature 2015, 521, 436–444. [Google Scholar] [CrossRef]
- Rajkomar, A.; Dean, J.; Kohane, I. Machine learning in medicine. N. Engl. J. Med. 2019, 380, 1347–1358. [Google Scholar] [CrossRef]
- Stokes, J.M.; Yang, K.; Swanson, K.; Jin, W.; Cubillos-Ruiz, A.; Donghia, N.M.; MacNair, C.R.; French, S.; Carfrae, L.A.; Bloom-Ackermann, Z.; et al. A Deep Learning Approach to Antibiotic Discovery. Cell 2020, 180, 688–702.e613. [Google Scholar] [CrossRef]
- Ching, T.; Himmelstein, D.S.; Beaulieu-Jones, B.K. Opportunities and obstacles for deep learning in biology and medicine. J. R. Soc. Interface 2018, 15, 20170387. [Google Scholar] [CrossRef]
- Liu, X.; Faes, L.; Kale, A.U. A comparison of deep learning performance against health-care professionals in detecting diseases from medical imaging: A systematic review and meta-analysis. Lancet Digit. Health 2019, 1, e271–e297. [Google Scholar] [CrossRef]
- Rieke, N.; Hancox, J.; Li, W. The future of digital health with federated learning. npj Digit. Med. 2020, 3, 119. [Google Scholar] [CrossRef]
- Schwaller, P.; Vaucher, A.C.; Laplaza, R.; Bunne, C.; Krause, A.; Corminboeuf, C.; Laino, T. Machine intelligence for chemical reaction space. WIREs Comput. Mol. Sci. 2022, 12, e1604. [Google Scholar] [CrossRef]
- Macarron, R.; Banks, M.N.; Bojanic, D.; Burns, D.J.; Cirovic, D.A.; Garyantes, T.; Green, D.V.S.; Hertzberg, R.P.; Janzen, W.P.; Paslay, J.W.; et al. Impact of high-throughput screening in biomedical research. Nat. Rev. Drug Discov. 2011, 10, 188–195. [Google Scholar] [CrossRef]
- Yu, L.X.; Amidon, G.; Khan, M.A.; Hoag, S.W.; Polli, J.; Raju, G.K.; Woodcock, J. Understanding pharmaceutical quality by design. Aaps J. 2014, 16, 771–783. [Google Scholar] [CrossRef]
- Li, M.; Zhang, R.; Zhang, Y. Application of machine learning in liposomal drug delivery systems. Drug Deliv. 2020, 27, 1591–1604. [Google Scholar]
- Matalqah, S.; Lafi, Z.; Mhaidat, Q.; Asha, N.; Yousef Asha, S. Applications of machine learning in liposomal formulation and development. Pharm. Dev. Technol. 2025, 30, 126–136. [Google Scholar] [CrossRef]
- Bhhatarai, B.; Walters, W.P. AI approaches for solubility prediction. J. Chem. Inf. Model. 2015, 55, 866–874. [Google Scholar]
- Bhhatarai, B.; Walters, W.P.; Hop, C.; Lanza, G.; Ekins, S. Opportunities and challenges using artificial intelligence in ADME/Tox. Nat. Mater. 2019, 18, 418–422. [Google Scholar] [CrossRef]
- Tawari, A.; Scott, J. Machine learning models for predicting solubility of drug-like molecules. J. Cheminformatics 2020, 12, 72. [Google Scholar]
- Walters, W.P.; Murcko, M.A. Assessing the impact of generative AI on medicinal chemistry. Nat. Biotechnol. 2020, 38, 143–145. [Google Scholar] [CrossRef]
- Zhang, Q.; Yang, G.; Wang, L. An AI-driven approach for predicting drug dissolution profiles in biorelevant media. Eur. J. Pharm. Sci. 2020, 152, 105421. [Google Scholar]
- Zhang, L.; Tan, J.; Han, D.; Zhu, H. From machine learning to deep learning: Progress in machine intelligence for rational drug discovery. Drug Discov. Today 2017, 22, 1680–1685. [Google Scholar] [CrossRef]
- Momeni, M.; Afkanpour, M.; Rakhshani, S.; Mehrabian, A.; Tabesh, H. A prediction model based on artificial intelligence techniques for disintegration time and hardness of fast disintegrating tablets in pre-formulation tests. BMC Med. Inform. Decis. Mak. 2024, 24, 88. [Google Scholar] [CrossRef]
- Dong, J.; Wu, Z.; Xu, H.; Ouyang, D. FormulationAI: A novel web-based platform for drug formulation design driven by artificial intelligence. Brief. Bioinform. 2023, 25, bbad419. [Google Scholar] [CrossRef]
- Karmalkar, P.; Gurulingappa, H.; Spies, E.; Flynn, J.A. Artificial intelligence-driven approach for patient-focused drug development. Front. Artif. Intell. 2023, 6, 1237124. [Google Scholar] [CrossRef]
- Goh, G.B.; Siegel, C.; Vishnu, A.; Hodas, N.O.; Baker, N. Chemception: A deep neural network with minimal chemistry knowledge matches the performance of expert-developed QSAR/QSPR models. ACS Cent. Sci. 2017, 3, 852–859. [Google Scholar]
- Schneider, G. Automating drug discovery. Nat. Rev. Drug Discov. 2018, 17, 97–113. [Google Scholar] [CrossRef]
- Chou, K.C.; Shen, H.B. Review: Recent advances in developing web-servers for predicting protein attributes. Nat. Sci. 2009, 1, 63–92. [Google Scholar] [CrossRef]
- Burger, B.; Maffettone, P.M.; Gusev, V.V.; Aitchison, C.M.; Bai, Y.; Wang, X.; Li, X.; Alston, B.M.; Li, B.; Clowes, R.; et al. A mobile robotic chemist. Nature 2020, 583, 237–241. [Google Scholar] [CrossRef]
- Joshi, S.; Bawage, S.; Tiwari, P.; Kirby, D.; Perrie, Y.; Dennis, V.; Singh, S.R. Liposomes: A promising carrier for respiratory syncytial virus therapeutics. Expert. Opin. Drug Deliv. 2019, 16, 969–980. [Google Scholar] [CrossRef]
- Marrink, S.J.; Corradi, V.; Souza, P.C.T.; Ingólfsson, H.I.; Tieleman, D.P.; Sansom, M.S.P. Computational modeling of realistic cell membranes. Chem. Rev. 2019, 119, 6184–6226. [Google Scholar] [CrossRef]
- Korotcov, A.; Tkachenko, V.; Russo, D.P.; Ekins, S. Comparison of deep learning with multiple machine learning methods and metrics using diverse drug discovery data sets. Mol. Pharm. 2017, 14, 4462–4475. [Google Scholar] [CrossRef]
- Hood, R.R.; DeVoe, D.L. High-throughput continuous flow production of nanoscale liposomes by microfluidics. Lab. A Chip 2015, 15, 3758–3767. [Google Scholar]
- Joshi, S.; Hussain, M.T.; Roces, C.B.; Anderluzzi, G.; Kastner, E.; Salmaso, S.; Kirby, D.J.; Perrie, Y. Microfluidics based manufacture of liposomes simultaneously entrapping hydrophilic and lipophilic drugs. Int. J. Pharm. 2016, 514, 160–168. [Google Scholar] [CrossRef] [PubMed]
- Barenholz, Y. Doxil®—The first FDA-approved nano-drug: Lessons learned. J. Control. Release 2012, 160, 117–134. [Google Scholar] [CrossRef]
- Kourou, K.; Exarchos, T.P.; Exarchos, K.P.; Karamouzis, M.V.; Fotiadis, D.I. Machine learning applications in cancer prognosis and prediction. Comput. Struct. Biotechnol. J. 2015, 13, 8–17. [Google Scholar] [CrossRef]
- Obermeyer, Z.; Emanuel, E.J. Predicting the future—Big data, machine learning, and clinical medicine. N. Engl. J. Med. 2016, 375, 1216–1219. [Google Scholar] [CrossRef]
- Choi, H.; Kang, H.J.; Ahn, I.; Gwon, H.; Kim, Y.; Seo, H.; Cho, H.N.; Han, J.; Kim, M.; Kee, G.; et al. Machine learning models to predict the warfarin discharge dosage using clinical information of inpatients from South Korea. Sci. Rep. 2023, 13, 22461. [Google Scholar] [CrossRef]
- Wiljer, D.; Charow, R.; Zhang, M.; Lo, B.; Sequeira, L.; Shen, N.; Sockalingam, S.; Rossos, P.; Crawford, A.; Strudwick, G. Enabling Digital Compassion in Digital Health Environments: Modified eDelphi Study to Identify Interprofessional Competencies and Technology Attributes. J. Med. Internet Res. 2025, 27, e66547. [Google Scholar] [CrossRef]
- Boughton, C.K.; Hovorka, R. Is an artificial pancreas (closed-loop system) for type 1 diabetes effective? Diabet. Med. 2019, 36, 279–286. [Google Scholar] [CrossRef]
- Germovsek, E.; Barker, C.I.; Sharland, M. What do I need to know about aminoglycoside dosing in neonates? Arch. Dis. Child. Educ. Pract. Ed. 2017, 102, 89–93. [Google Scholar] [CrossRef]
- Roberts, J.A.; Abdul-Aziz, M.H.; Lipman, J.; Mouton, J.W.; Vinks, A.A.; Felton, T.W. Individualised antibiotic dosing for patients who are critically ill: Challenges and potential solutions. Lancet Infect. Dis. 2014, 14, 498–509. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.H.; Kim, M.G. Precision dosing in pediatric patients using machine learning. Pharmaceutics 2021, 13, 83. [Google Scholar]
- Curtin, D.; Gallagher, P.F.; O’Mahony, D. Deprescribing in older people approaching end-of-life: Development and validation of STOPPFrail (Screening Tool of Older Persons Prescriptions in Frail adults with limited life expectancy). Age Ageing 2017, 46, 600–607. [Google Scholar]
- Tosca, E.M.; Green, B.; Hu, L.; Upton, R.N. Model-informed reinforcement learning for enabling precision dosing. Clin. Pharmacol. Ther. 2024, 115, 1421–1432. [Google Scholar] [CrossRef]
- Qian, L.; Cao, Y.; Chen, X.; Xu, Y. Application of physiologically based pharmacokinetic modeling to inform dosing decisions for geriatric patients. CPT Pharmacomet. Syst. Pharmacol. 2024, 13, 289–302. [Google Scholar] [CrossRef]
- Basu, S.; Legault, M.A.; Romero-Soriano, A.; Precup, D. On the challenges of using reinforcement learning in precision drug dosing: Delay and prolongedness of action effects. arXiv 2023, arXiv:2301.00512. [Google Scholar] [CrossRef]
- Polster, S.P. Machine learning for precision dosing: A review. Clin. Pharmacol. Ther. 2022, 112, 57–71. [Google Scholar]
- Yuan, Y.; Lin, R.; Li, D. Dose-finding designs for early-phase clinical trials using Bayesian model averaging. Stat. Med. 2016, 35, 4349–4360. [Google Scholar]
- Pai Mangalore, R.; Peel, T.N.; Udy, A.A.; Peleg, A.Y. The clinical application of beta-lactam antibiotic therapeutic drug monitoring in the critical care setting. J. Antimicrob. Chemother. 2023, 78, 2395–2405. [Google Scholar] [CrossRef] [PubMed]
- Riaz, I.B.; Khan, M.A.; Osterman, T.J. Artificial intelligence across the cancer care continuum. Cancer 2025, 131, e70050. [Google Scholar] [CrossRef] [PubMed]
- Giri, R.; Firdhos, S.H.; Vida, T.A. Artificial Intelligence in Anesthesia: Enhancing Precision, Safety, and Global Access Through Data-Driven Systems. J. Clin. Med. 2025, 14, 6900. [Google Scholar] [CrossRef]
- Zhang, Q.; Tian, X.; Chen, G.; Yu, Z.; Zhang, X.; Lu, J.; Zhang, J.; Wang, P.; Hao, X.; Huang, Y.; et al. A Prediction Model for Tacrolimus Daily Dose in Kidney Transplant Recipients With Machine Learning and Deep Learning Techniques. Front. Med. 2022, 9, 813117. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Sun, B.; Yi, Y.; Velkov, T.; Shen, J.; Dai, C.; Jiang, H. Progress of AI-Driven Drug-Target Interaction Prediction and Lead Optimization. Int. J. Mol. Sci. 2025, 26, 37. [Google Scholar] [CrossRef]
- Wu, P.; Nelson, S.D.; Zhao, J.; Stone, C.A., Jr.; Feng, Q.; Chen, Q.; Larson, E.A.; Li, B.; Cox, N.J.; Stein, C.M.; et al. DDIWAS: High-throughput electronic health record-based screening of drug-drug interactions. J. Am. Med. Inform. Assoc. 2021, 28, 1421–1430. [Google Scholar] [CrossRef]
- Luo, Y.; Zhao, X.; Zhou, J.; Yang, J.; Zhang, Y.; Kuang, W.; Peng, J.; Chen, L.; Zeng, J. A network integration approach for drug-target interaction prediction and computational drug repositioning from heterogeneous information. Nat. Commun. 2017, 8, 573. [Google Scholar] [CrossRef]
- Vilar, S.; Friedman, C.; Hripcsak, G. Detection of drug–drug interactions through data mining studies using clinical sources, scientific literature and social media. Brief. Bioinform. 2018, 19, 863–877. [Google Scholar] [CrossRef]
- US Food and Administration. FDA Proposes Framework to Advance Credibility of AI Models Used in Drug and Biological Product Submissions; US Food and Drug Administration: Silver Spring, MD, USA, 2023.
- US Food and Administration. Artificial Intelligence and Machine Learning in Drug Development: Guidance for Industry; US Food and Drug Administration: Silver Spring, MD, USA, 2023.
- Doshi, P. Role of AI in pharmaceutical quality assurance. J. Pharm. Sci. 2021, 110, 1648–1658. [Google Scholar]
- Lee, S. FDA perspective on continuous manufacturing. Consort. Contin. Pharm. Manuf. 2018, 103. [Google Scholar]
- Lee, S.L.; O’Connor, T.F.; Yang, X.; Cruz, C.N.; Chatterjee, S.; Madurawe, R.D.; Moore, C.M.V.; Yu, L.X.; Woodcock, J. Modernizing Pharmaceutical Manufacturing: From Batch to Continuous Production. J. Pharm. Innov. 2015, 10, 191–199. [Google Scholar] [CrossRef]
- Miner, G.; Delen, D.; Elder, J. Practical Text Mining and Statistical Analysis for Non-Structured Text Data Applications; Academic Press: Cambridge, MA, USA, 2012. [Google Scholar]
- Dakhole, M.R.; Thombre, K.R.; Gupta, K.R.; Umekar, M.J. Ensuring data integrity in the pharmaceutical lifecycle: Challenges, principles, and global implications. Ann. Pharm. Fr. 2025. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.X. Real-time release testing and AI integration. Pharm. Technol. 2022, 46, 24–33. [Google Scholar]
- Sultan, T.; Rozin, E.H.; Paul, S.; Tseng, Y.C.; Dave, V.S.; Cetinkaya, C. Machine learning modeling for ultrasonic quality attribute assessment of pharmaceutical tablets for continuous manufacturing and real-time release testing. Int. J. Pharm. 2024, 655, 124049. [Google Scholar] [CrossRef] [PubMed]
- European Medicines Agency. Guideline on Real-Time Release Testing and Parametric Release; European Medicines Agency: Amsterdam, The Netherlands, 2012. [Google Scholar]
- Fu, C.; Chen, Q. The future of pharmaceuticals: Artificial intelligence in drug discovery and development. J. Pharm. Anal. 2025, 15, 101248. [Google Scholar] [CrossRef]
- Beam, A.L.; Kohane, I.S. Big data and machine learning in health care. JAMA 2018, 319, 1317–1318. [Google Scholar] [CrossRef] [PubMed]
- Miotto, R.; Wang, F.; Wang, S.; Jiang, X.; Dudley, J.T. Deep learning for healthcare: Review, opportunities and challenges. Brief. Bioinform. 2018, 19, 1236–1246. [Google Scholar] [CrossRef] [PubMed]
- Ball, R.; Robb, M.; Anderson, S.; Pan, G.D. The FDA’s Sentinel Initiative—A comprehensive approach to medical product surveillance. Clin. Pharmacol. Ther. 2016, 99, 265–268. [Google Scholar] [CrossRef]
- Ribeiro, M.T.; Singh, S.; Guestrin, C. Why should I trust you?: Explaining the predictions of any classifier. In Proceedings of the ACM SIGKDD International Conference on Knowledge Discovery and Data Mining, San Francisco, CA, USA, 13–17 August 2016; pp. 1135–1144. [Google Scholar]
- Lundberg, S.M.; Lee, S.I. A unified approach to interpreting model predictions. Adv. Neural Inf. Process. Syst. 2017, 30, 4765–4774. [Google Scholar]
- Holzinger, A.; Biemann, C.; Pattichis, C.S.; Kell, D.B. What do we need to build explainable AI systems for the medical domain? arXiv 2017, arXiv:1712.09923. [Google Scholar]
- European Medicines Agency. Qualification Opinion for PROCOVA™: An AI-Based Approach for Prognostic Covariate Adjustment in Clinical Trials; European Medicines Agency: Amsterdam, The Netherlands, 2022. [Google Scholar]
- US Food and Administration. Artificial Intelligence and Machine Learning Software as a Medical Device (SaMD); US Food and Drug Administration: Silver Spring, MD, USA, 2021.
- Gerke, S.; Minssen, T.; Cohen, I.G. Ethical and legal challenges of AI in healthcare. Sci. Transl. Med. 2020, 12, 295–336. [Google Scholar]
- US Food and Administration. Proposed Regulatory Framework for Modifications to AI/ML-Based Software as a Medical Device; US Food and Drug Administration: Silver Spring, MD, USA, 2019.
- Mittelstadt, B.D. Ethics of the health-related internet of things: A narrative review. Ethics Inf. Technol. 2017, 19, 157–175. [Google Scholar] [CrossRef]
- Topol, E.J. High-performance medicine: The convergence of human and artificial intelligence. Nat. Med. 2019, 25, 44–56. [Google Scholar] [CrossRef]
- Shah, P.; Kendall, F.; Khozin, S.; Goosen, R.; Hu, J.; Laramie, J.; Ringel, M.; Schork, N. Artificial intelligence and machine learning in clinical development: A translational perspective. NPJ Digit. Med. 2019, 2, 69. [Google Scholar] [CrossRef]
- Rajpurkar, P.; Chen, E.; Banerjee, O.; Topol, E.J. AI in health and medicine. Nat. Med. 2022, 28, 31–38. [Google Scholar] [CrossRef]
- Char, D.S.; Abràmoff, M.D.; Feudtner, C. Identifying ethical considerations for machine learning in health care. Nat. Med. 2020, 26, 1609–1612. [Google Scholar]
- Rajkomar, A.; Hardt, M.; Howell, M.D.; Corrado, G.; Chin, M.H. Ensuring fairness in machine learning to advance health equity. Ann. Intern. Med. 2018, 169, 866–872. [Google Scholar] [CrossRef]
- Xu, J.; Glicksberg, B.S.; Su, C.; Walker, P.; Bian, J.; Wang, F. Federated learning for healthcare applications: Opportunities and challenges. Nat. Mach. Intell. 2021, 3, 737–743. [Google Scholar]
- Salah, K. Blockchain for AI: Review and open research challenges. IEEE Access 2019, 7, 10127–10149. [Google Scholar] [CrossRef]
- Mamoshina, P.; Ojomoko, L.; Yanovich, Y.; Ostrovski, A.; Botezatu, A.; Prikhodko, P.; Izumchenko, E.; Aliper, A.; Romantsov, K.; Zhebrak, A.; et al. Converging blockchain and next-generation artificial intelligence technologies to decentralize and accelerate biomedical research and healthcare. Oncotarget 2018, 9, 5665–5690. [Google Scholar] [CrossRef]
- Islam, S.M.R.; Kwak, D.; Kabir, M.H.; Hossain, M.; Kwak, K.-S. The Internet of Things for health care: A comprehensive survey. IEEE Access 2015, 3, 678–708. [Google Scholar] [CrossRef]
- Biamonte, J.; Wittek, P.; Pancotti, N.; Rebentrost, P.; Wiebe, N.; Lloyd, S. Quantum machine learning. Nature 2017, 549, 195–202. [Google Scholar] [CrossRef]
- Sadia, M.; Arafat, B.; Ahmed, W. AI-enabled 3D printing in personalized medicine. Pharmaceutics 2022, 14, 491. [Google Scholar]
- Khaled, S.A.; Burley, J.C.; Alexander, M.R.; Yang, J.; Roberts, C.J. 3D printing of five-in-one dose combination polypill with defined immediate and sustained release profiles. J. Control Release 2015, 217, 308–314. [Google Scholar] [CrossRef] [PubMed]
- Trenfield, S.J.; Awad, A.; Goyanes, A.; Gaisford, S.; Basit, A.W. 3D printing pharmaceuticals: Drug development to frontline care. Trends Pharmacol. Sci. 2018, 39, 440–451. [Google Scholar] [CrossRef]
- Konta, A.A.; García-Piña, M.; Serrano, D.R. Personalised 3D Printed Medicines: Which Techniques and Polymers Are More Successful? Bioengineering 2017, 4, 79. [Google Scholar] [CrossRef]
- US Food and Administration. Technical Considerations for Additive Manufactured Medical Products; US Food and Drug Administration: Silver Spring, MD, USA, 2017.
- Zhang, R. Artificial intelligence in clinical trials: A disruptive innovation. Drug Discov. Today 2021, 26, 1212–1219. [Google Scholar]
- Harrer, S.; Shah, P.; Antony, B.; Hu, J. Artificial Intelligence for Clinical Trial Design. Trends Pharmacol. Sci. 2019, 40, 577–591. [Google Scholar] [CrossRef]
- Zhou, Y.; Hou, Y.; Shen, J.; Huang, Y.; Martin, W.; Cheng, F. Network-based drug repurposing for novel coronavirus 2019-nCoV/SARS-CoV-2. Cell Discov. 2020, 6, 14. [Google Scholar] [CrossRef] [PubMed]
- Zhavoronkov, A.; Ivanenkov, Y.A.; Aliper, A.; Veselov, M.S.; Aladinskiy, V.A.; Aladinskaya, A.V.; Terentiev, V.A.; Polykovskiy, D.A.; Kuznetsov, M.D.; Asadulaev, A.; et al. Deep learning enables rapid identification of potent DDR1 kinase inhibitors. Nat. Biotechnol. 2019, 37, 1038–1040. [Google Scholar] [CrossRef] [PubMed]
- Romano, J.D.; Hao, Y.; Moore, J.H.; Penning, T.M. Automating Predictive Toxicology Using ComptoxAI. Chem. Res. Toxicol. 2022, 35, 1370–1382. [Google Scholar] [CrossRef]
- Yang, Q.; Zhang, S.; Li, Y. Deep Learning Algorithm Based on Molecular Fingerprint for Prediction of Drug-Induced Liver Injury. Toxicology 2024, 502, 153736. [Google Scholar] [CrossRef] [PubMed]
- Beaulieu, D.; Kornjača, M.; Krunic, Z.; Stivaktakis, M.; Chen, J.; Ehmer, T.; Wang, S.-T.; Pham, A. Robust Quantum Reservoir Learning for Molecular Property Prediction. J. Chem. Inf. Model. 2025, 65, 8475–8485. [Google Scholar] [CrossRef]
- Smaldone, A.M.; Shee, Y.; Kyro, G.W.; Xu, C.; Vu, N.P.; Dutta, R.; Farag, M.H.; Galda, A.; Kumar, S.; Kyoseva, E.; et al. Quantum Machine Learning in Drug Discovery: Applications in Academia and Pharmaceutical Industries. Chem. Rev. 2025, 125, 5436–5460. [Google Scholar] [CrossRef]
| AI Technique | Use in Drug Formulation/Dosage Calculations | Reference |
|---|---|---|
| Artificial Neural Networks (ANNs) | Prediction of solubility, dissolution rates, encapsulation efficiency, drug-to-excipient ratios, and estimation of pharmacokinetic parameters. | [2,3] |
| Support Vector Machines (SVMs) | Classify excipients by compatibility, predict encapsulation efficiency in novel drug delivery systems such as liposomes and nanoparticle systems. | [7,8] |
| Random Forest (RF) | Selection for formulation parameters; prediction of optimal excipient concentrations for stability and bioavailability. | [9,10] |
| Deep Learning (DL, CNN/GNN) | Prediction of particle size, PDI, and drug release;, modeling complex nonlinear relationships in formulation datasets. | [11,12] |
| Bayesian Optimization | Optimization of dose, excipient concentration refinement, and efficient design of experiments (DoE). | [13,14] |
| Reinforcement Learning (RL) | Dosing strategies (e.g., insulin pumps), optimizing trial design for dose-escalation studies. | [15,16] |
| Natural Language Processing (NLP) | Extracts dosage guidelines, stability data, and drug interactions from literature. | [3,17,18] |
| Explainable AI (XAI) | Improves interpretability of dose predictions; regulatory acceptance. | [1,19] |
| Knowledge Graphs (KGs) | Predicts DDIs and DDzIs; supports polypharmacy dosage adjustments. | [20,21] |
| Graph Neural Networks (GNNs) | Drug–excipient compatibility; formulation stability prediction. | [22,23] |
| Generative Adversarial Networks (GANs) | Generate novel molecules; simulate formulation outcomes. | [24,25] |
| One-Shot/Few-Shot Learning | Dose–response prediction from limited data. | [26,27] |
| Transfer Learning | Improves model performance with small pharmaceutical datasets. | [28,29,30] |
| Federated Learning | Enables multi-institutional modeling without data sharing. | [31,32] |
| Hybrid ML–QbD Models | Combines AI with QbD for robust formulations. | [1,33,34] |
| Deep Reinforcement Learning | Optimizes trial designs and adaptive dosing. | [1,35,36] |
| Multimodal Learning | Combines chemical, imaging, omics, and text data for prediction. | [37,38] |
| AutoML (Automated ML) | Selects the best algorithms/features for dosage models automatically. | [39,40] |
| Digital Twins | Patient-specific simulation of drug response for precision dosing. | [41,42] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Joshi, S.; Sheth, S. Artificial Intelligence (AI) in Pharmaceutical Formulation and Dosage Calculations. Pharmaceutics 2025, 17, 1440. https://doi.org/10.3390/pharmaceutics17111440
Joshi S, Sheth S. Artificial Intelligence (AI) in Pharmaceutical Formulation and Dosage Calculations. Pharmaceutics. 2025; 17(11):1440. https://doi.org/10.3390/pharmaceutics17111440
Chicago/Turabian StyleJoshi, Sameer, and Sandeep Sheth. 2025. "Artificial Intelligence (AI) in Pharmaceutical Formulation and Dosage Calculations" Pharmaceutics 17, no. 11: 1440. https://doi.org/10.3390/pharmaceutics17111440
APA StyleJoshi, S., & Sheth, S. (2025). Artificial Intelligence (AI) in Pharmaceutical Formulation and Dosage Calculations. Pharmaceutics, 17(11), 1440. https://doi.org/10.3390/pharmaceutics17111440









