Liposome-Based Drug Delivery Systems in Cancer Research: An Analysis of Global Landscape Efforts and Achievements
Abstract
1. Introduction
2. Materials and Methods
2.1. Search Plan and Refining the Retrieved Documents
2.2. Data Export
2.3. Bibliometric Analyses and Visualization
3. Results
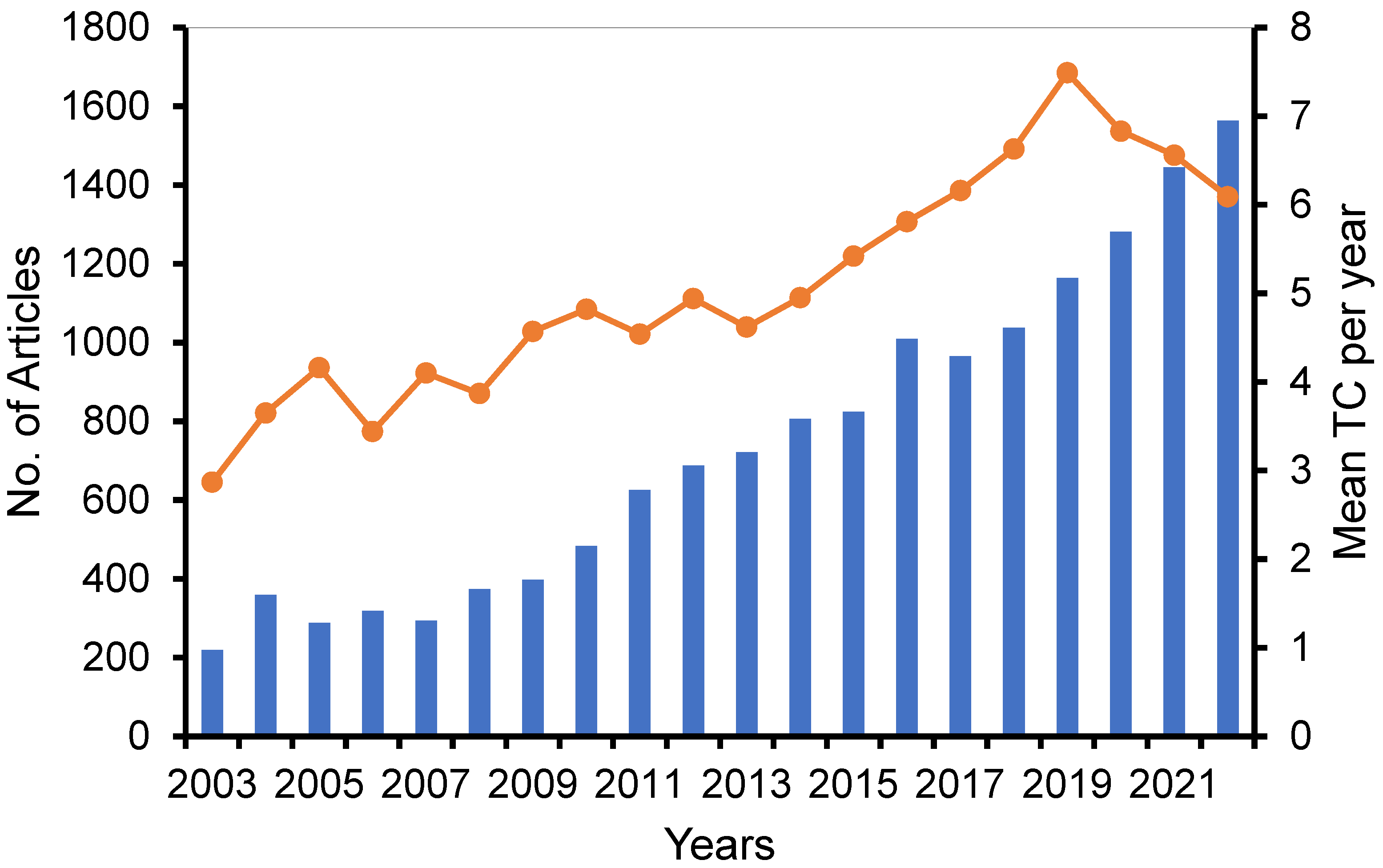
3.1. Analysis of Publications by Year
3.2. Analysis of Contributing Journals
3.3. Analysis of Articles
3.4. Analysis of Authors
3.5. Active Countries
3.6. Bibliometric Mapping
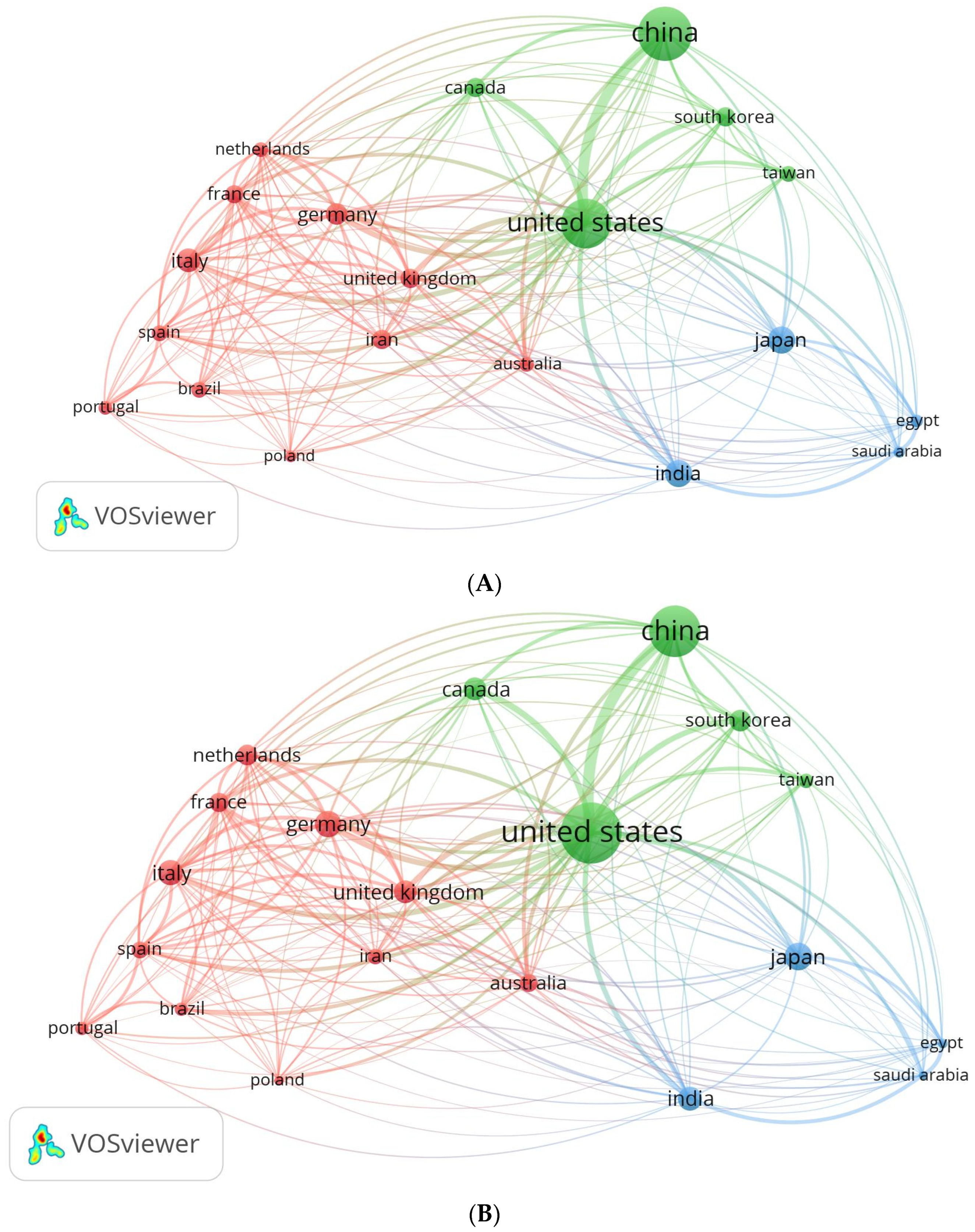
3.6.1. International Collaboration
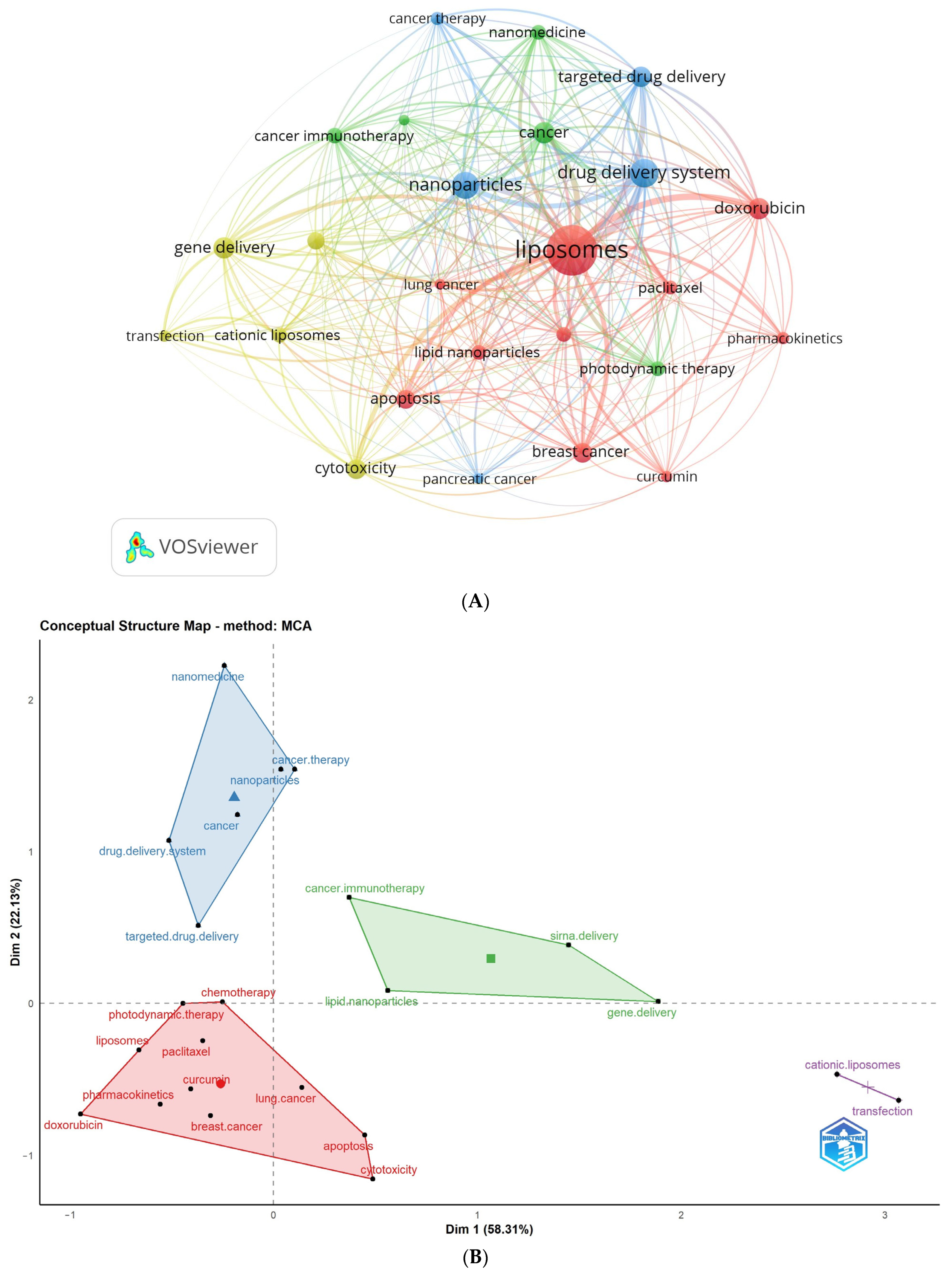
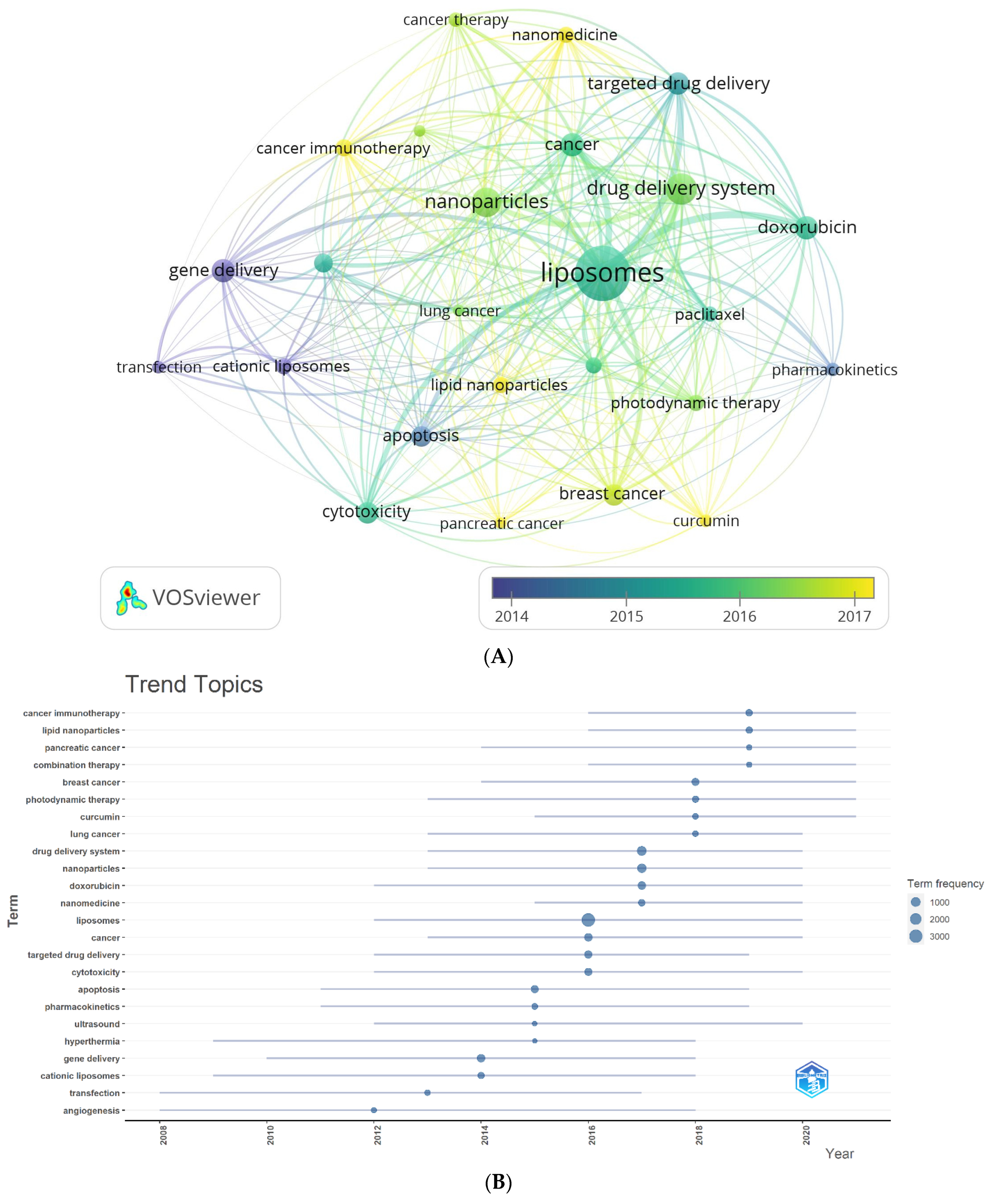
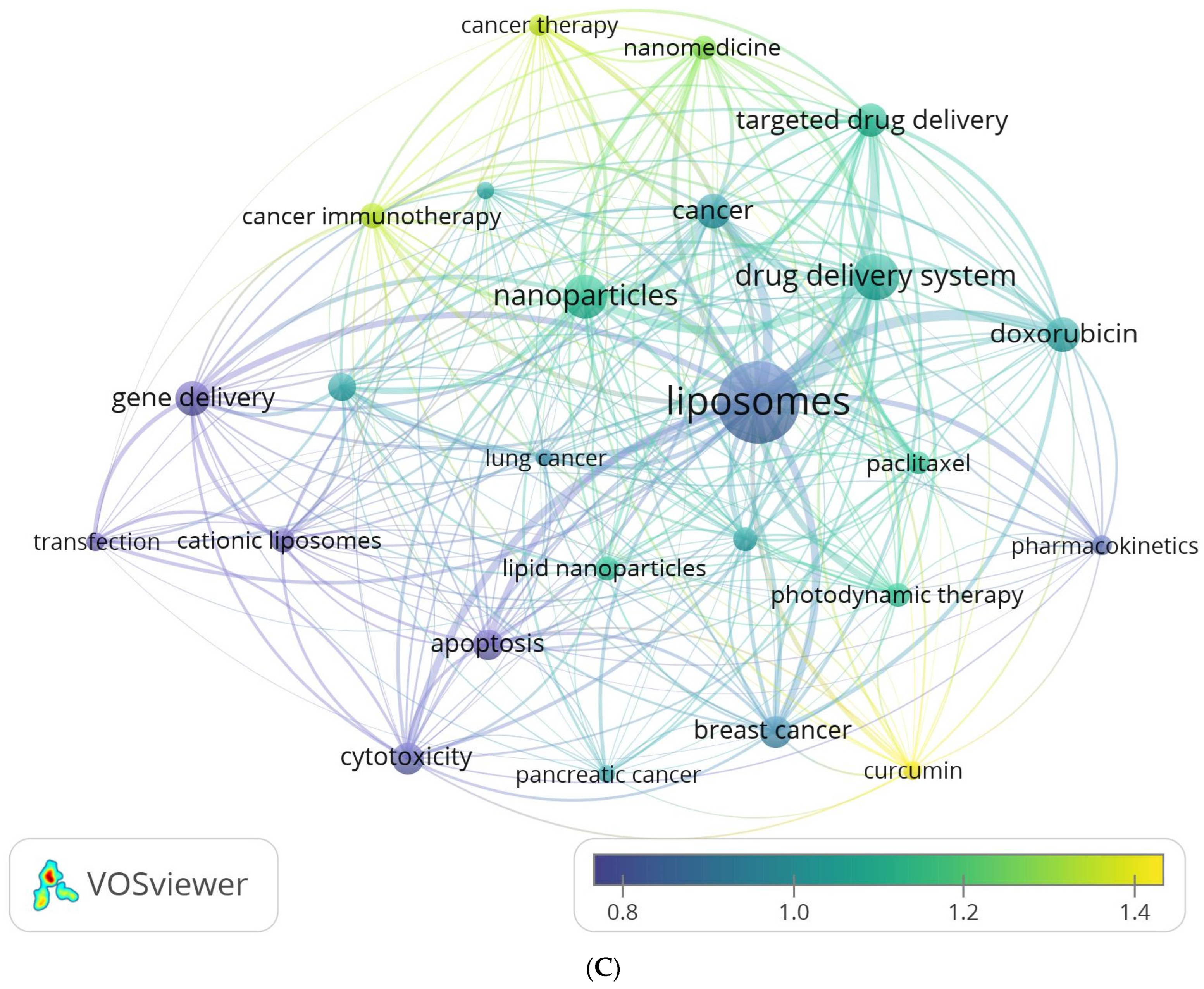
3.6.2. Analysis of Author Keywords and Hotspots Forecasting
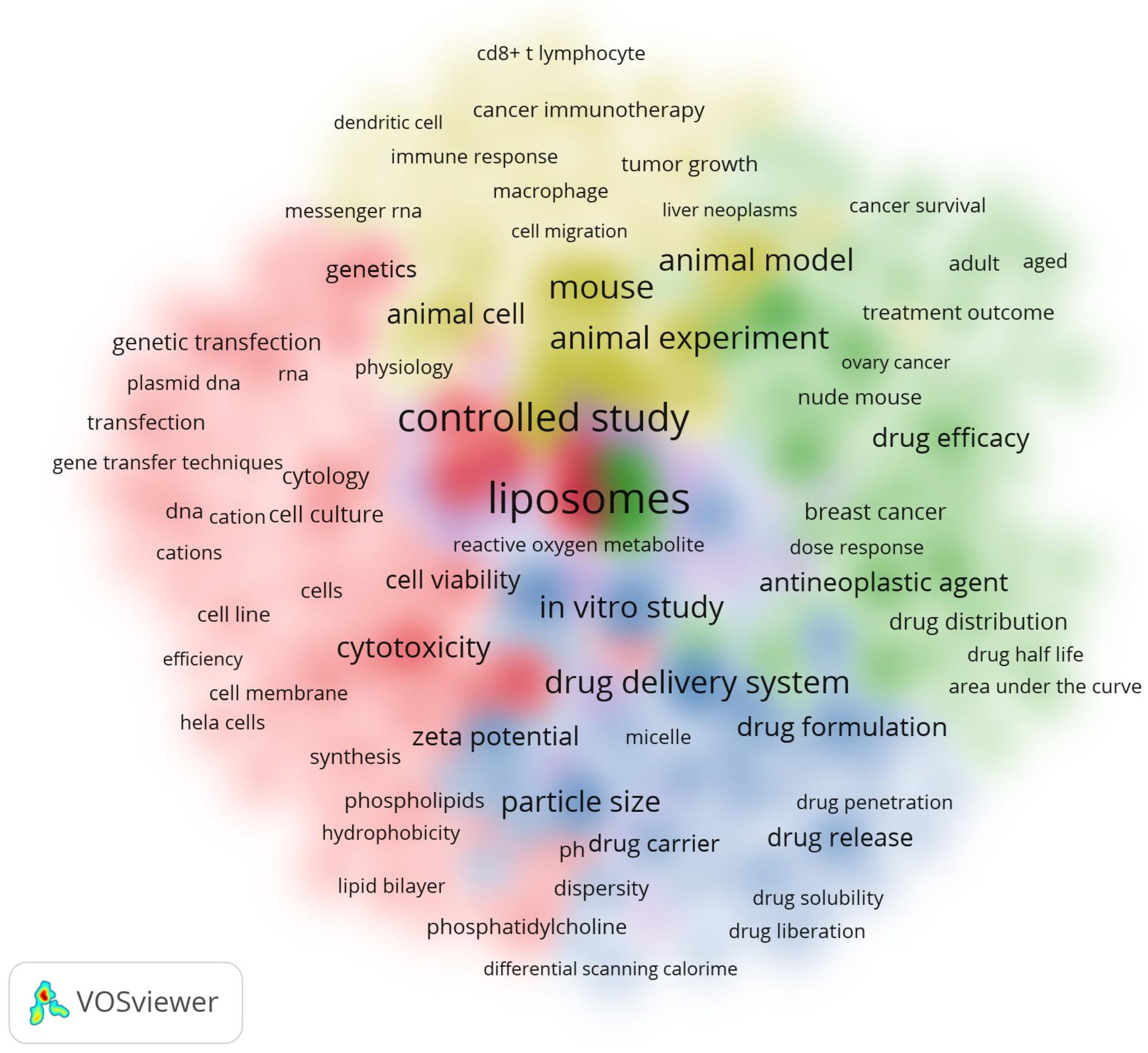
3.6.3. Analysis of All Keywords
4. Discussion
4.1. Cluster 1: Liposomal Advancements in Cancer Therapy: Targeting Apoptosis, Breast, and Lung Cancers with Doxorubicin and Paclitaxel, Integrating Curcumin, and Optimizing Pharmacokinetics with Lipid Nanoparticles
4.2. Cluster 2: Liposomal Advancements: Propelling Cancer Immunotherapy, Nanomedicine Precision, and Photodynamic Therapy across Oncology
4.3. Cluster 3: Enhancing Targeted Cancer Therapy: The Role of Liposomes in Precision Drug Delivery
4.4. Cluster 4: Liposomal progresses: Unraveling the Potential in Cationic Liposomes for Enhanced Gene and siRNA Delivery with Minimized Cytotoxicity and Optimal Transfection Efficiency
5. Study Limitations
6. Conclusions and Future Directions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Nsairat, H.; Khater, D.; Sayed, U.; Odeh, F.; Al Bawab, A.; Alshaer, W. Liposomes: Structure, composition, types, and clinical applications. Heliyon 2022, 8, e09394. [Google Scholar] [CrossRef]
- Alshaer, W.; Zraikat, M.; Amer, A.; Nsairat, H.; Lafi, Z.; Alqudah, D.A.; Al Qadi, E.; Alsheleh, T.; Odeh, F.; Alkaraki, A.; et al. Encapsulation of echinomycin in cyclodextrin inclusion complexes into liposomes: In vitro anti-proliferative and anti-invasive activity in glioblastoma. RSC Adv. 2019, 9, 30976–30988. [Google Scholar] [CrossRef]
- Matalqah, S.M.; Aiedeh, K.; Mhaidat, N.M.; Alzoubi, K.H.; Bustanji, Y.; Hamad, I. Chitosan nanoparticles as a novel drug delivery system: A review article. Curr. Drug Targets 2020, 21, 1613–1624. [Google Scholar] [CrossRef]
- Fernandes, D.A. Liposomes for Cancer Theranostics. Pharmaceutics 2023, 15, 2448. [Google Scholar] [CrossRef]
- Gao, Y.; Liu, X.; Chen, N.; Yang, X.; Tang, F. Recent Advance of Liposome Nanoparticles for Nucleic Acid Therapy. Pharmaceutics 2023, 15, 178. [Google Scholar] [CrossRef]
- Nikolova, M.P.; Kumar, E.M.; Chavali, M.S. Updates on Responsive Drug Delivery Based on Liposome Vehicles for Cancer Treatment. Pharmaceutics 2022, 14, 2195. [Google Scholar] [CrossRef]
- Gu, Z.; Da Silva, C.G.; van der Maaden, K.; Ossendorp, F.; Cruz, L.J. Liposome-based drug delivery systems in cancer immunotherapy. Pharmaceutics 2020, 12, 1054. [Google Scholar] [CrossRef]
- Chen, J.; Hu, S.; Sun, M.; Shi, J.; Zhang, H.; Yu, H.; Yang, Z. Recent advances and clinical translation of liposomal delivery systems in cancer therapy. Eur. J. Pharm. Sci. 2024, 193, 106688. [Google Scholar] [CrossRef]
- Moghimi, S.M.; Hamad, I. Liposome-mediated triggering of complement cascade. J. Liposome Res. 2008, 18, 195–209. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Xie, G.; Xiao, X.; Cheng, C. Characterization of PDL1 enhanced siRNA/albumin liposome for effective therapeutic function in lung cancer. J. Cancer Res. Clin. Oncol. 2023, 149, 3835–3846. [Google Scholar] [CrossRef] [PubMed]
- Zhu, W.; Yu, H.; Jia, M.; Lin, C.; Yuan, Z.; Tan, X.; Yan, P. Multi-targeting liposomal codelivery of cisplatin and rapamycin inhibits pancreatic cancer growth and metastasis through stromal modulation. Int. J. Pharm. 2023, 644, 123316. [Google Scholar] [CrossRef] [PubMed]
- Tan, C.; Wang, J.; Sun, B. Biopolymer-liposome hybrid systems for controlled delivery of bioactive compounds: Recent advances. Biotechnol. Adv. 2021, 48, 107727. [Google Scholar] [CrossRef] [PubMed]
- Bishani, A.; Makarova, D.M.; Shmendel, E.V.; Maslov, M.A.; Sen’kova, A.V.; Savin, I.A.; Gladkikh, D.V.; Zenkova, M.A.; Chernolovskaya, E.L. Influence of the Composition of Cationic Liposomes on the Performance of Cargo Immunostimulatory RNA. Pharmaceutics 2023, 15, 2184. [Google Scholar] [CrossRef] [PubMed]
- Duché, G.; Heu, C.; Thordarson, P. Development and Characterization of Nanoscale Gel-Core Liposomes Using a Short Self-Assembled Peptide Hydrogel: Implications for Drug Delivery. ACS Appl. Nano Mater. 2023, 6, 14745–14755. [Google Scholar] [CrossRef]
- Wan, H.; Wang, S.; Li, C.; Zeng, B.; Wu, H.; Liu, C.; Chen, L.; Jin, M.; Huang, W.; Zang, Y.; et al. LA67 Liposome-Loaded Thermo-Sensitive Hydrogel with Active Targeting for Efficient Treatment of Keloid via Peritumoral Injection. Pharmaceutics 2023, 15, 2157. [Google Scholar] [CrossRef] [PubMed]
- Tang, J.S.J.; Smaczniak, A.D.; Tepper, L.; Rosencrantz, S.; Aleksanyan, M.; Dähne, L.; Rosencrantz, R.R. Glycopolymer Based LbL Multilayer Thin Films with Embedded Liposomes. Macromol. Biosci. 2022, 22, 2100461. [Google Scholar] [CrossRef]
- Hasanbegloo, K.; Banihashem, S.; Faraji Dizaji, B.; Bybordi, S.; Farrokh-Eslamlou, N.; Abadi, P.G.S.; Jazi, F.S.; Irani, M. Paclitaxel-loaded liposome-incorporated chitosan (core)/poly(ε-caprolactone)/chitosan (shell) nanofibers for the treatment of breast cancer. Int. J. Biol. Macromol. 2023, 230, 123380. [Google Scholar] [CrossRef]
- Lafi, Z.; Alshaer, W.; Hatmal, M.M.; Zihlif, M.; Alqudah, D.A.; Nsairat, H.; Azzam, H.; Aburjai, T.; Bustanji, Y.; Awidi, A. Aptamer-functionalized pH-sensitive liposomes for a selective delivery of echinomycin into cancer cells. RSC Adv. 2021, 11, 29164–29177. [Google Scholar] [CrossRef]
- Gharaibeh, L.; Alshaer, W.; Wehaibi, S.; Al Buqain, R.; Alqudah, D.A.; Al-Kadash, A.; Al-Azzawi, H.; Awidi, A.; Bustanji, Y. Fabrication of aptamer-guided siRNA loaded lipopolyplexes for gene silencing of notch 1 in MDA-mb-231 triple negative breast cancer cell line. J. Drug Deliv. Sci. Technol. 2021, 65, 102733. [Google Scholar] [CrossRef]
- Caritá, A.C.; Resende de Azevedo, J.; Chevalier, Y.; Arquier, D.; Buri, M.V.; Riske, K.A.; Ricci Leonardi, G.; Bolzinger, M.A. Elastic cationic liposomes for vitamin C delivery: Development, characterization and skin absorption study. Int. J. Pharm. 2023, 638, 122897. [Google Scholar] [CrossRef] [PubMed]
- Dzhumashev, D.; Anton-Joseph, S.; Morel, V.J.; Timpanaro, A.; Bordon, G.; Piccand, C.; Aleandri, S.; Luciani, P.; Rössler, J.; Bernasconi, M. Rapid liposomal formulation for nucleolin targeting to rhabdomyosarcoma cells. Eur. J. Pharm. Biopharm. 2024, 194, 49–61. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Fan, M.; Huang, D.; Li, B.; Xu, R.; Gao, F.; Chen, Y. Clodronate-loaded liposomal and fibroblast-derived exosomal hybrid system for enhanced drug delivery to pulmonary fibrosis. Biomaterials 2021, 271, 120761. [Google Scholar] [CrossRef] [PubMed]
- Cevenini, A.; Celia, C.; Orrù, S.; Sarnataro, D.; Raia, M.; Mollo, V.; Locatelli, M.; Imperlini, E.; Peluso, N.; Peltrini, R.; et al. Liposome-embedding silicon microparticle for oxaliplatin delivery in tumor chemotherapy. Pharmaceutics 2020, 12, 559. [Google Scholar] [CrossRef]
- Pardhi, E.; Yadav, R.; Chaurasiya, A.; Madan, J.; Guru, S.K.; Singh, S.B.; Mehra, N.K. Multifunctional targetable liposomal drug delivery system in the management of leukemia: Potential, opportunities, and emerging strategies. Life Sci. 2023, 325, 121771. [Google Scholar] [CrossRef]
- Alwattar, J.K.; Mneimneh, A.T.; Abla, K.K.; Mehanna, M.M.; Allam, A.N. Smart stimuli-responsive liposomal nanohybrid systems: A critical review of theranostic behavior in cancer. Pharmaceutics 2021, 13, 355. [Google Scholar] [CrossRef] [PubMed]
- Ning, S.; Zhang, X.; Suo, M.; Lyu, M.; Pan, Y.; Jiang, Y.; Yang, H.; Yip Lam, J.W.; Zhang, T.; Pan, L.; et al. Platelet-derived exosomes hybrid liposomes facilitate uninterrupted singlet oxygen generation to enhance breast cancer immunotherapy. Cell Rep. Phys. Sci. 2023, 4, 101505. [Google Scholar] [CrossRef]
- Bang, C.; Park, M.G.; Cho, I.K.; Lee, D.E.; Kim, G.L.; Jang, E.H.; Shim, M.K.; Yoon, H.Y.; Lee, S.; Kim, J.H. Liposomes targeting the cancer cell-exposed receptor, claudin-4, for pancreatic cancer chemotherapy. Biomater. Res. 2023, 27, 53. [Google Scholar] [CrossRef] [PubMed]
- Ning, S.; Suo, M.; Huang, Q.; Gao, S.; Qiao, K.; Lyu, M.; Huang, Q.; Zhang, T.; Tang, B.Z. Biomimetic fusion liposomes boosting antitumor immunity and promote memory T cell differentiation to inhibit postoperative recurrence of breast cancer. Nano Today 2024, 54, 102106. [Google Scholar] [CrossRef]
- Zhao, Z.; Wang, W.; Wang, G.; Huang, Z.; Zhou, L.; Lin, L.; Ou, Y.; Huang, W.; Zhang, X.; Wu, C.; et al. Dual peptides-modified cationic liposomes for enhanced Lung cancer gene therapy by a gap junction regulating strategy. J. Nanobiotechnol. 2023, 21, 473. [Google Scholar] [CrossRef]
- Alimu, G.; Yan, T.; Zhu, L.; Du, Z.; Ma, R.; Fan, H.; Chen, S.; Alifu, N.; Zhang, X. Liposomes loaded with dual clinical photosensitizers for enhanced photodynamic therapy of cervical cancer. RSC Adv. 2023, 13, 3459–3467. [Google Scholar] [CrossRef]
- Fulton, M.D.; Najahi-Missaoui, W. Liposomes in Cancer Therapy: How Did We Start and Where Are We Now. Int. J. Mol. Sci. 2023, 24, 6615. [Google Scholar] [CrossRef]
- Fidan, Y.; Muçaj, S.; Timur, S.S.; Gürsoy, R.N. Recent advances in liposome-based targeted cancer therapy. J. Liposome Res. 2024, in press. [Google Scholar] [CrossRef]
- Bustanji, Y.; Taneera, J.; Semreen, M.H.; Abu-Gharbieh, E.; El-Huneidi, W.; Faris, M.A.I.E.; Alzoubi, K.H.; Soares, N.C.; Albustanji, B.; Abuhelwa, A.Y.; et al. Gold nanoparticles and breast cancer: A bibliometric analysis of the current state of research and future directions. OpenNano 2023, 12, 100164. [Google Scholar] [CrossRef]
- Aria, M.; Cuccurullo, C. bibliometrix: An R-tool for comprehensive science mapping analysis. J. Informetr. 2017, 11, 959–975. [Google Scholar] [CrossRef]
- Bustanji, Y.; Shihab, K.H.A.; El-Huneidi, W.; Semreen, M.H.; Abu-Gharbieh, E.; Alzoubi, K.H.; Alqudah, M.A.Y.; Abuhelwa, A.Y.; Abu-Rish, E.Y.; Bajes, H.; et al. Analysis and mapping of global scientific research on human monkeypox over the past 20 years. Vet. World 2023, 16, 693–703. [Google Scholar] [CrossRef]
- Moral-Munoz, J.; Herrera-Viedma, E.; Espejo, A.; Cobo, M. Software tools for conducting bibliometric analysis in science: An up-to-date review. Prof. Inf. 2020, 29, e290103. [Google Scholar] [CrossRef]
- van Eck, N.J.; Waltman, L. Software survey: VOSviewer, a computer program for bibliometric mapping. In Proceedings of the 12th International Conference on Scientometrics and Informetrics, Rio de Janeiro, Brazil, 14–17 July 2009; International Society for Scientometrics and Informetrics: Leuven, Belgium, 2009; pp. 886–897. [Google Scholar]
- Wallin, J.A. Bibliometric methods: Pitfalls and possibilities. Basic Clin. Pharmacol. Toxicol. 2005, 97, 261–275. [Google Scholar] [CrossRef] [PubMed]
- Salmerón-Manzano, E.; Manzano-Agugliaro, F. Bibliometric studies and worldwide research trends on global health. Int. J. Environ. Res. Public Health 2020, 17, 5748. [Google Scholar] [CrossRef] [PubMed]
- Bustanji, Y.; Taneera, J.; Bargooth, A.; Abuhelwa, A.; Issa, A.; El-Huneidi, W.; Abu-Gharbieh, E.; H. Alzoubi, K.; Alqudah, M.A.Y.; Alhusban, A.; et al. Exploring the global landscape of self-medication among students: Trends, risks, and recommendations for safe and responsible practices. Pharm. Pract. 2024, 22, 2928. [Google Scholar]
- Van Eck, N.J.; Waltman, L. Software survey: VOSviewer, a computer program for bibliometric mapping. Scientometrics 2010, 84, 523–538. [Google Scholar] [CrossRef] [PubMed]
- Olive, K.P.; Jacobetz, M.A.; Davidson, C.J.; Gopinathan, A.; McIntyre, D.; Honess, D.; Madhu, B.; Goldgraben, M.A.; Caldwell, M.E.; Allard, D.; et al. Inhibition of Hedgehog signaling enhances delivery of chemotherapy in a mouse model of pancreatic cancer. Science 2009, 324, 1457–1461. [Google Scholar] [CrossRef]
- Cabral, H.; Matsumoto, Y.; Mizuno, K.; Chen, Q.; Murakami, M.; Kimura, M.; Terada, Y.; Kano, M.R.; Miyazono, K.; Uesaka, M.; et al. Accumulation of sub-100 nm polymeric micelles in poorly permeable tumours depends on size. Nat. Nanotechnol. 2011, 6, 815–823. [Google Scholar] [CrossRef]
- Kamerkar, S.; Lebleu, V.S.; Sugimoto, H.; Yang, S.; Ruivo, C.F.; Melo, S.A.; Lee, J.J.; Kalluri, R. Exosomes facilitate therapeutic targeting of oncogenic KRAS in pancreatic cancer. Nature 2017, 546, 498–503. [Google Scholar] [CrossRef]
- Ding, J.; Wang, K.; Liu, W.; She, Y.; Sun, Q.; Shi, J.; Sun, H.; Wang, D.C.; Shao, F. Pore-forming activity and structural autoinhibition of the gasdermin family. Nature 2016, 535, 111–116. [Google Scholar] [CrossRef] [PubMed]
- Arruebo, M.; Fernández-Pacheco, R.; Ibarra, M.R.; Santamaría, J. Magnetic nanoparticles for drug delivery. Nano Today 2007, 2, 22–32. [Google Scholar] [CrossRef]
- O’Brien, M.E.R.; Wigler, N.; Inbar, M.; Rosso, R.; Grischke, E.; Santoro, A.; Catane, R.; Kieback, D.G.; Tomczak, P.; Ackland, S.P.; et al. Reduced cardiotoxicity and comparable efficacy in a phase III trial of pegylated liposomal doxorubicin HCl (CAELYX™/Doxil®) versus conventional doxorubicin for first-line treatment of metastatic breast cancer. Ann. Oncol. 2004, 15, 440–449. [Google Scholar] [CrossRef] [PubMed]
- Ito, A.; Shinkai, M.; Honda, H.; Kobayashi, T. Medical application of functionalized magnetic nanoparticles. J. Biosci. Bioeng. 2005, 100, 1–11. [Google Scholar] [CrossRef]
- van der Pol, E.; Böing, A.N.; Harrison, P.; Sturk, A.; Nieuwland, R. Classification, functions, and clinical relevance of extracellular vesicles. Pharmacol. Rev. 2012, 64, 676–705. [Google Scholar] [CrossRef]
- Alkilany, A.M.; Murphy, C.J. Toxicity and cellular uptake of gold nanoparticles: What we have learned so far? J. Nanopart. Res. 2010, 12, 2313–2333. [Google Scholar] [CrossRef] [PubMed]
- Sun, D.; Zhuang, X.; Xiang, X.; Liu, Y.; Zhang, S.; Liu, C.; Barnes, S.; Grizzle, W.; Miller, D.; Zhang, H.G. A novel nanoparticle drug delivery system: The anti-inflammatory activity of curcumin is enhanced when encapsulated in exosomes. Mol. Ther. 2010, 18, 1606–1614. [Google Scholar] [CrossRef]
- Aiedeh, K.M.; Taha, M.O.; Al-Hiari, Y.; Bustanji, Y.; Alkhatib, H.S. Effect of ionic crosslinking on the drug release properties of chitosan diacetate matrices. J. Pharm. Sci. 2007, 96, 38–43. [Google Scholar] [CrossRef] [PubMed]
- Khdair, A.; Hamad, I.; Alkhatib, H.; Bustanji, Y.; Mohammad, M.; Tayem, R.; Aiedeh, K. Modified-chitosan nanoparticles: Novel drug delivery systems improve oral bioavailability of doxorubicin. Eur. J. Pharm. Sci. 2016, 93, 38–44. [Google Scholar] [CrossRef]
- Yeh, P.Y.; Chen, J.Y.; Shen, M.Y.; Che, T.F.; Lim, S.C.; Wang, J.; Tsai, W.S.; Frank, C.W.; Huang, C.J.; Chang, Y.C. Liposome-tethered supported lipid bilayer platform for capture and release of heterogeneous populations of circulating tumor cells. J. Mater. Chem. B 2023, 11, 8159–8169. [Google Scholar] [CrossRef] [PubMed]
- Askarizadeh, A.; Mashreghi, M.; Mirhadi, E.; Mirzavi, F.; Shargh, V.H.; Badiee, A.; Alavizadeh, S.H.; Arabi, L.; Jaafari, M.R. Doxorubicin-loaded liposomes surface engineered with the matrix metalloproteinase-2 cleavable polyethylene glycol conjugate for cancer therapy. Cancer Nanotechnol. 2023, 14, 18. [Google Scholar] [CrossRef]
- Fu, S.; Chang, L.; Liu, S.; Gao, T.; Sang, X.; Zhang, Z.; Mu, W.; Liu, X.; Liang, S.; Yang, H.; et al. Temperature sensitive liposome based cancer nanomedicine enables tumour lymph node immune microenvironment remodelling. Nat. Commun. 2023, 14, 2248. [Google Scholar] [CrossRef]
- Zou, J. Site-specific delivery of cisplatin and paclitaxel mediated by liposomes: A promising approach in cancer chemotherapy. Environ. Res. 2023, 238, 117111. [Google Scholar] [CrossRef] [PubMed]
- AlKhatib, H.S.; Taha, M.O.; Aiedeh, K.M.; Bustanji, Y.; Sweileh, B. Synthesis and in vitro behavior of iron-crosslinked N-methyl and N-benzyl hydroxamated derivatives of alginic acid as controlled release carriers. Eur. Polym. J. 2006, 42, 2464–2474. [Google Scholar] [CrossRef]
- Cho, E.; Mun, S.J.; Jeon, M.; Kim, H.K.; Baek, H.; Ham, Y.S.; Gil, W.J.; Kim, J.W.; Yang, C.S. Tumor-targeted liposomes with platycodin D2 promote apoptosis in colorectal cancer. Mater. Today Bio 2023, 22, 100745. [Google Scholar] [CrossRef]
- Saraf, S.; Jain, S.K. pH-sensitive liposomes bearing a chemotherapeutic agent and a natural apoptosis modulator for effective intracellular delivery to the solid tumor. Drug Deliv. Transl. Res. 2023, 13, 2961–2981. [Google Scholar] [CrossRef]
- El-Hamid, E.S.A.; Gamal-Eldeen, A.M.; Sharaf Eldeen, A.M. Liposome-coated nano doxorubicin induces apoptosis on oral squamous cell carcinoma CAL-27 cells. Arch. Oral Biol. 2019, 103, 47–54. [Google Scholar] [CrossRef]
- Feuser, P.E.; De Pieri, E.; Oliveira, M.E.; Cordeiro, A.P.; Cercena, R.; Hermes de Araújo, P.H.; Dal Bó, A.G.; Machado-de-Ávila, R.A. Cisplatin and paclitaxel-loaded liposomes induced cervical cancer (HeLa) cell death with multiple copies of human papillomavirus by apoptosis and decreased their cytotoxic effect on non-tumor cells. J. Drug Deliv. Sci. Technol. 2022, 73, 103457. [Google Scholar] [CrossRef]
- Wan, S.; Fan, Q.; Wu, Y.; Zhang, J.; Qiao, G.; Jiang, N.; Yang, J.; Liu, Y.; Li, J.; Chiampanichayakul, S.; et al. Curcumin-Loaded Platelet Membrane Bioinspired Chitosan-Modified Liposome for Effective Cancer Therapy. Pharmaceutics 2023, 15, 631. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Huang, Y.; Xu, J.; Zhao, R.; Xiong, C.; Habu, J.; Wang, Y.; Luo, X. Global publication trends and research hotspots of curcumin application in tumor: A 20-year bibliometric approach. Front. Oncol. 2022, 12, 1033683. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Wang, A.; Zhang, S.; Kim, J.; Xia, J.; Zhang, F.; Wang, D.; Wang, Q.; Wang, J. Paclitaxel-loaded ginsenoside Rg3 liposomes for drug-resistant cancer therapy by dual targeting of the tumor microenvironment and cancer cells. J. Adv. Res. 2023, 49, 159–173. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Liang, H.; Li, J.; Shao, Z.; Yang, D.; Bao, J.; Wang, K.; Xi, W.; Gao, Z.; Guo, R.; et al. Paclitaxel liposome (Lipusu) based chemotherapy combined with immunotherapy for advanced non-small cell lung cancer: A multicenter, retrospective real-world study. BMC Cancer 2024, 24, 107. [Google Scholar] [CrossRef] [PubMed]
- Rak, J.; Kabesova, M.; Benes, J.; Pouckova, P.; Vetvicka, D. Advances in Liposome-Encapsulated Phthalocyanines for Photodynamic Therapy. Life 2023, 13, 305. [Google Scholar] [CrossRef] [PubMed]
- Shi, C.; Li, M.; Zhang, Z.; Yao, Q.; Shao, K.; Xu, F.; Xu, N.; Li, H.; Fan, J.; Sun, W.; et al. Catalase-based liposomal for reversing immunosuppressive tumor microenvironment and enhanced cancer chemo-photodynamic therapy. Biomaterials 2020, 233, 119755. [Google Scholar] [CrossRef] [PubMed]
- Fahmy, S.A.; Azzazy, H.M.E.S.; Schaefer, J. Liposome photosensitizer formulations for effective cancer photodynamic therapy. Pharmaceutics 2021, 13, 1345. [Google Scholar] [CrossRef]
- Sesarman, A.; Tefas, L.; Sylvester, B.; Licarete, E.; Rauca, V.; Luput, L.; Patras, L.; Porav, S.; Banciu, M.; Porfire, A. Co-delivery of curcumin and doxorubicin in PEGylated liposomes favored the antineoplastic C26 murine colon carcinoma microenvironment. Drug Deliv. Transl. Res. 2019, 9, 260–272. [Google Scholar] [CrossRef]
- Feng, X.; Pi, C.; Fu, S.; Yang, H.; Zheng, X.; Hou, Y.; Wang, Y.; Zhang, X.; Zhao, L.; Wei, Y. Combination of Curcumin and Paclitaxel Liposomes Exhibits Enhanced Cytotoxicity Towards A549/A549-T Cells and Unaltered Pharmacokinetics. J. Biomed. Nanotechnol. 2020, 16, 1304–1313. [Google Scholar] [CrossRef]
- Alanazi, A.; Fadda, L.; Alhusaini, A.; Ahmad, R. Antioxidant, antiapoptotic, and antifibrotic effects of the combination of liposomal resveratrol and carvedilol against doxorubicin-induced cardiomyopathy in rats. J. Biochem. Mol. Toxicol. 2020, 34, e22492. [Google Scholar] [CrossRef] [PubMed]
- Duan, H.; Liu, C.; Hou, Y.; Liu, Y.; Zhang, Z.; Zhao, H.; Xin, X.; Liu, W.; Zhang, X.; Chen, L.; et al. Sequential Delivery of Quercetin and Paclitaxel for the Fibrotic Tumor Microenvironment Remodeling and Chemotherapy Potentiation via a Dual-Targeting Hybrid Micelle-in-Liposome System. ACS Appl. Mater. Interfaces 2022, 14, 10102–10116. [Google Scholar] [CrossRef]
- Zhang, Q.; Liu, C.; Wang, J. Synergistic Effect of Quercetin and Vitamin C in Reducing Acute Toxicity and Improving Antitumor Activity of Liposomal Doxorubicin. Curr. Top. Nutraceutical Res. 2019, 17, 223–228. [Google Scholar] [CrossRef]
- Mureşan, M.; Olteanu, D.; Filip, G.A.; Clichici, S.; Baldea, I.; Jurca, T.; Pallag, A.; Marian, E.; Frum, A.; Gligor, F.G.; et al. Comparative study of the pharmacological properties and biological effects of polygonum aviculare l. Herba extract-entrapped liposomes versus quercetin-entrapped liposomes on doxorubicin-induced toxicity on huvecs. Pharmaceutics 2021, 13, 1418. [Google Scholar] [CrossRef] [PubMed]
- Dorostkar, H.; Haghiralsadat, B.F.; Hemati, M.; Safari, F.; Hassanpour, A.; Naghib, S.M.; Roozbahani, M.H.; Mozafari, M.R.; Moradi, A. Reduction of Doxorubicin-Induced Cardiotoxicity by Co-Administration of Smart Liposomal Doxorubicin and Free Quercetin: In Vitro and In Vivo Studies. Pharmaceutics 2023, 15, 1920. [Google Scholar] [CrossRef] [PubMed]
- Ocaña-Arakachi, K.; Martínez-Herculano, J.; Jurado, R.; Llaguno-Munive, M.; Garcia-Lopez, P. Pharmacokinetics and Anti-Tumor Efficacy of PEGylated Liposomes Co-Loaded with Cisplatin and Mifepristone. Pharmaceuticals 2023, 16, 1337. [Google Scholar] [CrossRef]
- Tseu, G.Y.W.; Kamaruzaman, K.A. A Review of Different Types of Liposomes and Their Advancements as a Form of Gene Therapy Treatment for Breast Cancer. Molecules 2023, 28, 1498. [Google Scholar] [CrossRef]
- Alhamhoom, Y.; Kakinani, G.; Rahamathulla, M.; Ali, M.; Osmani, R.; Hani, U.; Yoonus Thajudeen, K.; Kiran Raj, G.; Gowda, D.V. Recent advances in the liposomal nanovesicles based immunotherapy in the treatment of cancer: A review. Saudi Pharm. J. 2023, 31, 279–294. [Google Scholar] [CrossRef]
- Chang, R.; Chu, X.; Zhang, J.; Fu, R.; Feng, C.; Jia, D.; Wang, R.; Yan, H.; Li, G.; Li, J. Liposome-Based Co-Immunotherapy with TLR Agonist and CD47-SIRPα Checkpoint Blockade for Efficient Treatment of Colon Cancer. Molecules 2023, 28, 3147. [Google Scholar] [CrossRef]
- Liu, X.; Yi, X.; Gu, J.; Ji, Z.; Zhu, M.; Shen, M.; Ren, Y.; Guo, L.; Liu, T.; Ding, N.; et al. Immunoregulatory liposomes hitchhiking on neutrophils for enhanced carbon ion radiotherapy-assisted immunotherapy of glioblastoma. Nano Today 2023, 53, 102037. [Google Scholar] [CrossRef]
- Lee, E.K.; Xiong, N.; Cheng, S.C.; Barry, W.T.; Penson, R.T.; Konstantinopoulos, P.A.; Hoffman, M.A.; Horowitz, N.; Dizon, D.S.; Stover, E.H.; et al. Combined pembrolizumab and pegylated liposomal doxorubicin in platinum resistant ovarian cancer: A phase 2 clinical trial. Gynecol. Oncol. 2020, 159, 72–78. [Google Scholar] [CrossRef] [PubMed]
- Ma, G.L.; Lin, W.F. Immune checkpoint inhibition mediated with liposomal nanomedicine for cancer therapy. Mil. Med. Res. 2023, 10, 20. [Google Scholar] [CrossRef] [PubMed]
- Huang, Z.; Wei, G.; Zeng, Z.; Huang, Y.; Huang, L.; Shen, Y.; Sun, X.; Xu, C.; Zhao, C. Enhanced cancer therapy through synergetic photodynamic/immune checkpoint blockade mediated by a liposomal conjugate comprised of porphyrin and IDO inhibitor. Theranostics 2019, 9, 5542–5557. [Google Scholar] [CrossRef]
- Moghassemi, S.; Dadashzadeh, A.; Azevedo, R.B.; Feron, O.; Amorim, C.A. Photodynamic cancer therapy using liposomes as an advanced vesicular photosensitizer delivery system. J. Control. Release 2021, 339, 75–90. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, S.; Carter, K.A.; Lovell, J.F. Liposomal formulations of photosensitizers. Biomaterials 2019, 218, 119341. [Google Scholar] [CrossRef]
- Xiao, Z.; Zhuang, B.; Zhang, G.; Li, M.; Jin, Y. Pulmonary delivery of cationic liposomal hydroxycamptothecin and 5-aminolevulinic acid for chemo-sonodynamic therapy of metastatic lung cancer. Int. J. Pharm. 2021, 601, 120572. [Google Scholar] [CrossRef]
- Peng, P.C.; Hong, R.L.; Tsai, T.; Chen, C.T. Co-encapsulation of chlorin e6 and chemotherapeutic drugs in a pegylated liposome enhance the efficacy of tumor treatment: Pharmacokinetics and therapeutic efficacy. Pharmaceutics 2019, 11, 617. [Google Scholar] [CrossRef]
- Zhu, Y.X.; Jia, H.R.; Duan, Q.Y.; Liu, X.; Yang, J.; Liu, Y.; Wu, F.G. Photosensitizer-Doped and Plasma Membrane-Responsive Liposomes for Nuclear Drug Delivery and Multidrug Resistance Reversal. ACS Appl. Mater. Interfaces 2020, 12, 36882–36894. [Google Scholar] [CrossRef]
- Nsairat, H.; Mahmoud, I.S.; Odeh, F.; Abuarqoub, D.; Al-Azzawi, H.; Zaza, R.; Qadri, M.I.; Ismail, S.; Al Bawab, A.; Awidi, A.; et al. Grafting of anti-nucleolin aptamer into preformed and remotely loaded liposomes through aptamer-cholesterol post-insertion. RSC Adv. 2020, 10, 36219–36229. [Google Scholar] [CrossRef]
- Honari, A.; Merillat, D.A.; Bellary, A.; Ghaderi, M.; Sirsi, S.R. Improving release of liposome-encapsulated drugs with focused ultrasound and vaporizable droplet-liposome nanoclusters. Pharmaceutics 2021, 13, 609. [Google Scholar] [CrossRef]
- Yazdian-Robati, R.; Amiri, E.; Kamali, H.; Khosravi, A.; Taghdisi, S.M.; Jaafari, M.R.; Mashreghi, M.; Moosavian, S.A. CD44-specific short peptide A6 boosts cellular uptake and anticancer efficacy of PEGylated liposomal doxorubicin in vitro and in vivo. Cancer Nanotechnol. 2023, 14, 84. [Google Scholar] [CrossRef]
- Silli, E.K.; Li, M.; Shao, Y.; Zhang, Y.; Hou, G.; Du, J.; Liang, J.; Wang, Y. Liposomal nanostructures for Gemcitabine and Paclitaxel delivery in pancreatic cancer. Eur. J. Pharm. Biopharm. 2023, 192, 13–24. [Google Scholar] [CrossRef]
- Raza, F.; Evans, L.; Motallebi, M.; Zafar, H.; Pereira-Silva, M.; Saleem, K.; Peixoto, D.; Rahdar, A.; Sharifi, E.; Veiga, F.; et al. Liposome-based diagnostic and therapeutic applications for pancreatic cancer. Acta Biomater. 2023, 157, 1–23. [Google Scholar] [CrossRef]
- Aloss, K.; Hamar, P. Recent Preclinical and Clinical Progress in Liposomal Doxorubicin. Pharmaceutics 2023, 15, 893. [Google Scholar] [CrossRef]
- Mirzavi, F.; Barati, M.; Soleimani, A.; Vakili-Ghartavol, R.; Jaafari, M.R.; Soukhtanloo, M. A review on liposome-based therapeutic approaches against malignant melanoma. Int. J. Pharm. 2021, 599, 120413. [Google Scholar] [CrossRef] [PubMed]
- Alavi, M.; Varma, R.S. Overview of novel strategies for the delivery of anthracyclines to cancer cells by liposomal and polymeric nanoformulations. Int. J. Biol. Macromol. 2020, 164, 2197–2203. [Google Scholar] [CrossRef] [PubMed]
- Nsairat, H.; Alshaer, W.; Odeh, F.; Esawi, E.; Khater, D.; Bawab, A.A.; El-Tanani, M.; Awidi, A.; Mubarak, M.S. Recent advances in using liposomes for delivery of nucleic acid-based therapeutics. OpenNano 2023, 11, 100132. [Google Scholar] [CrossRef]
- Sharma, M.; Sudha Ambadipudi, S.S.S.S.; Kumar Chouhan, N.; Lakshma Nayak, V.; Pabbaraja, S.; Balaji Andugulapati, S.; Sistla, R. Design, synthesis and biological evaluation of novel cationic liposomes loaded with melphalan for the treatment of cancer. Bioorg. Med. Chem. Lett. 2023, 97, 129549. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Li, S.; Li, Y.; Li, X.; Yang, G.; Li, M.; Xie, Y.; Su, W.; Wu, J.; Jia, L.; et al. Cationic liposomes co-deliver chemotherapeutics and siRNA for the treatment of breast cancer. Eur. J. Med. Chem. 2022, 233, 114198. [Google Scholar] [CrossRef] [PubMed]
- Abu Lila, A.S.; Ishida, T.; Kiwada, H. Targeting anticancer drugs to tumor vasculature using cationic liposomes. Pharm. Res. 2010, 27, 1171–1183. [Google Scholar] [CrossRef] [PubMed]
- Majzoub, R.N.; Ewert, K.K.; Safinya, C.R. Cationic liposome-nucleic acid nanoparticle assemblies with applications in gene delivery and gene silencing. Philos. Trans. R. Soc. A Math. Phys. Eng. Sci. 2016, 374, 20150129. [Google Scholar] [CrossRef] [PubMed]
- Gladkikh, D.V.; Sen′ Kova, A.V.; Chernikov, I.V.; Kabilova, T.O.; Popova, N.A.; Nikolin, V.P.; Shmendel, E.V.; Maslov, M.A.; Vlassov, V.V.; Zenkova, M.A.; et al. Folate-equipped cationic liposomes deliver anti-mdr1-sirna to the tumor and increase the efficiency of chemotherapy. Pharmaceutics 2021, 13, 1252. [Google Scholar] [CrossRef]
- Sousa, D.A.; Gaspar, R.; Ferreira, C.J.O.; Baltazar, F.; Rodrigues, L.R.; Silva, B.F.B. In Vitro CRISPR/Cas9 Transfection and Gene-Editing Mediated by Multivalent Cationic Liposome–DNA Complexes. Pharmaceutics 2022, 14, 1087. [Google Scholar] [CrossRef]
- Zhang, J.; Guan, M.; Ma, C.; Liu, Y.; Lv, M.; Zhang, Z.; Gao, H.; Zhang, K. Highly Effective Detection of Exosomal miRNAs in Plasma Using Liposome-Mediated Transfection CRISPR/Cas13a. ACS Sens. 2023, 8, 565–575. [Google Scholar] [CrossRef]
- Desai, D.; Gaud, R.S.; Shende, P. Potential of Chimeric Antigen Receptor T-Cells in Cancer Therapy. In Advances in Experimental Medicine and Biology; Springer: Berlin/Heidelberg, Germany, 2021; Volume 1326, pp. 39–46. [Google Scholar]
- Huang, C.H.; Dong, T.; Phung, A.T.; Shah, J.R.; Larson, C.; Sanchez, A.B.; Blair, S.L.; Oronsky, B.; Trogler, W.C.; Reid, T.; et al. Full Remission of CAR-Deficient Tumors by DOTAP-Folate Liposome Encapsulation of Adenovirus. ACS Biomater. Sci. Eng. 2022, 8, 5199–5209. [Google Scholar] [CrossRef] [PubMed]
- Ding, Y.; Wang, L.; Li, H.; Miao, F.; Zhang, Z.; Hu, C.; Yu, W.; Tang, Q.; Shao, G. Application of lipid nanovesicle drug delivery system in cancer immunotherapy. J. Nanobiotechnol. 2022, 20, 214. [Google Scholar] [CrossRef] [PubMed]

| Source | No of Publication | % | Citations | Scopus Percentile (Q) |
|---|---|---|---|---|
| Journal of controlled release | 617 | 4.15 | 39,071 | 98 (Q1) |
| International journal of pharmaceutics | 531 | 3.57 | 20,496 | 93 (Q1) |
| International journal of nanomedicine | 387 | 2.60 | 12,300 | 96 (Q1) |
| Biomaterials | 341 | 2.29 | 27,800 | 98 (Q1) |
| Colloids and surfaces B: biointerfaces | 241 | 1.62 | 7532 | 90 (Q1) |
| Molecular pharmaceutics | 239 | 1.61 | 9832 | 86 (Q1) |
| Pharmaceutics | 234 | 1.57 | 2940 | 76 (Q1) |
| Plos ONE | 167 | 1.12 | 5417 | 87 (Q1) |
| Pharmaceutical research | 161 | 1.08 | 5560 | 81 (Q1) |
| Drug delivery | 160 | 1.08 | 4546 | 85 (Q1) |
| Paper | Total Citations | TC per Year | Normalized TC |
|---|---|---|---|
| OLIVE KP, 2009, SCIENCE ([42]) | 2516 | 167.73 | 39.31 |
| CABRAL H, 2011, NAT NANOTECHNOL ([43]) | 1965 | 151.15 | 36.08 |
| KAMERKAR S, 2017, NATURE ([44]) | 1507 | 215.29 | 40.74 |
| DING J, 2016, NATURE ([45]) | 1503 | 187.88 | 36.92 |
| ARRUEBO M, 2007, NANO TODAY([46]) | 1408 | 82.82 | 21.48 |
| O’BRIEN MER, 2004, ANN ONCOL ([47]) | 1357 | 67.85 | 19.57 |
| ITO A, 2005, J BIOSCI BIOENG ([48]) | 1331 | 70.05 | 17.79 |
| VAN DER POL E, 2012, PHARMACOL REV ([49]) | 1301 | 108.42 | 23.94 |
| ALKILANY AM, 2010, J NANOPART RES ([50]) | 1219 | 87.07 | 19.43 |
| SUN D, 2010, MOL THER ([51]) | 1167 | 83.36 | 18.61 |
| Rank | Author | Documents | Total Citation for Author |
|---|---|---|---|
| 1st | Jaafari, M.R. | 92 | 10,717 |
| 2nd | Harashima, H. | 90 | 24,792 |
| 3rd | Lee, R.J. | 81 | 19,053 |
| 4th | Torchilin, V.P. | 76 | 61,532 |
| 5th | Zhang, Z. | 63 | 19,235 |
| 6th | Zhang, Q. | 62 | 21,758 |
| 7th | Storm, G. | 59 | 40,212 |
| 8th | He, Q. | 54 | 9270 |
| 9th | Oku, N. | 45 | 12,583 |
| 10th | Lopez-Berestein, G. | 44 | 36,331 |
| Rank | Country | Number of Publications | Total Citations | % of Total Documents | Citation/Document |
|---|---|---|---|---|---|
| 1st | China | 4205 | 136,047 | 28.3 | 32.4 |
| 2nd | United States | 3452 | 192,391 | 23.2 | 55.7 |
| 3rd | Japan | 1075 | 40,995 | 7.2 | 38.1 |
| 4th | India | 1030 | 30,699 | 6.9 | 29.8 |
| 5th | Italy | 779 | 31,782 | 5.2 | 40.8 |
| 6th | Germany | 697 | 36,029 | 4.7 | 51.7 |
| 7th | South Korea | 559 | 23,652 | 3.8 | 42.3 |
| 8th | United Kingdom | 537 | 27,788 | 3.6 | 51.7 |
| 9th | Iran | 517 | 12,301 | 3.5 | 23.8 |
| 10th | Canada | 517 | 27,227 | 3.5 | 52.7 |
| Author Keyword | Frequency | Cluster |
|---|---|---|
| apoptosis | 457 | 1 |
| breast cancer | 485 | 1 |
| chemotherapy | 272 | 1 |
| curcumin | 181 | 1 |
| doxorubicin | 591 | 1 |
| lipid nanoparticles | 282 | 1 |
| liposomes | 3305 | 1 |
| lung cancer | 162 | 1 |
| paclitaxel | 269 | 1 |
| pharmacokinetics | 200 | 1 |
| cancer | 593 | 2 |
| cancer immunotherapy | 319 | 2 |
| melanoma | 151 | 2 |
| nanomedicine | 299 | 2 |
| photodynamic therapy | 282 | 2 |
| cancer therapy | 220 | 3 |
| drug delivery system | 1029 | 3 |
| nanoparticles | 917 | 3 |
| pancreatic cancer | 151 | 3 |
| targeted drug delivery | 553 | 3 |
| cationic liposomes | 282 | 4 |
| cytotoxicity | 498 | 4 |
| gene delivery | 576 | 4 |
| siRNA delivery | 399 | 4 |
| transfection | 182 | 4 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hamad, I.; Harb, A.A.; Bustanji, Y. Liposome-Based Drug Delivery Systems in Cancer Research: An Analysis of Global Landscape Efforts and Achievements. Pharmaceutics 2024, 16, 400. https://doi.org/10.3390/pharmaceutics16030400
Hamad I, Harb AA, Bustanji Y. Liposome-Based Drug Delivery Systems in Cancer Research: An Analysis of Global Landscape Efforts and Achievements. Pharmaceutics. 2024; 16(3):400. https://doi.org/10.3390/pharmaceutics16030400
Chicago/Turabian StyleHamad, Islam, Amani A. Harb, and Yasser Bustanji. 2024. "Liposome-Based Drug Delivery Systems in Cancer Research: An Analysis of Global Landscape Efforts and Achievements" Pharmaceutics 16, no. 3: 400. https://doi.org/10.3390/pharmaceutics16030400
APA StyleHamad, I., Harb, A. A., & Bustanji, Y. (2024). Liposome-Based Drug Delivery Systems in Cancer Research: An Analysis of Global Landscape Efforts and Achievements. Pharmaceutics, 16(3), 400. https://doi.org/10.3390/pharmaceutics16030400








