Race, Ethnicity, and Pharmacogenomic Variation in the United States and the United Kingdom
Abstract
1. Introduction
2. Materials and Methods
2.1. Biobank Volunteer Participants
2.2. Biobank Participant Data
2.3. Pharmacogenomic Variants
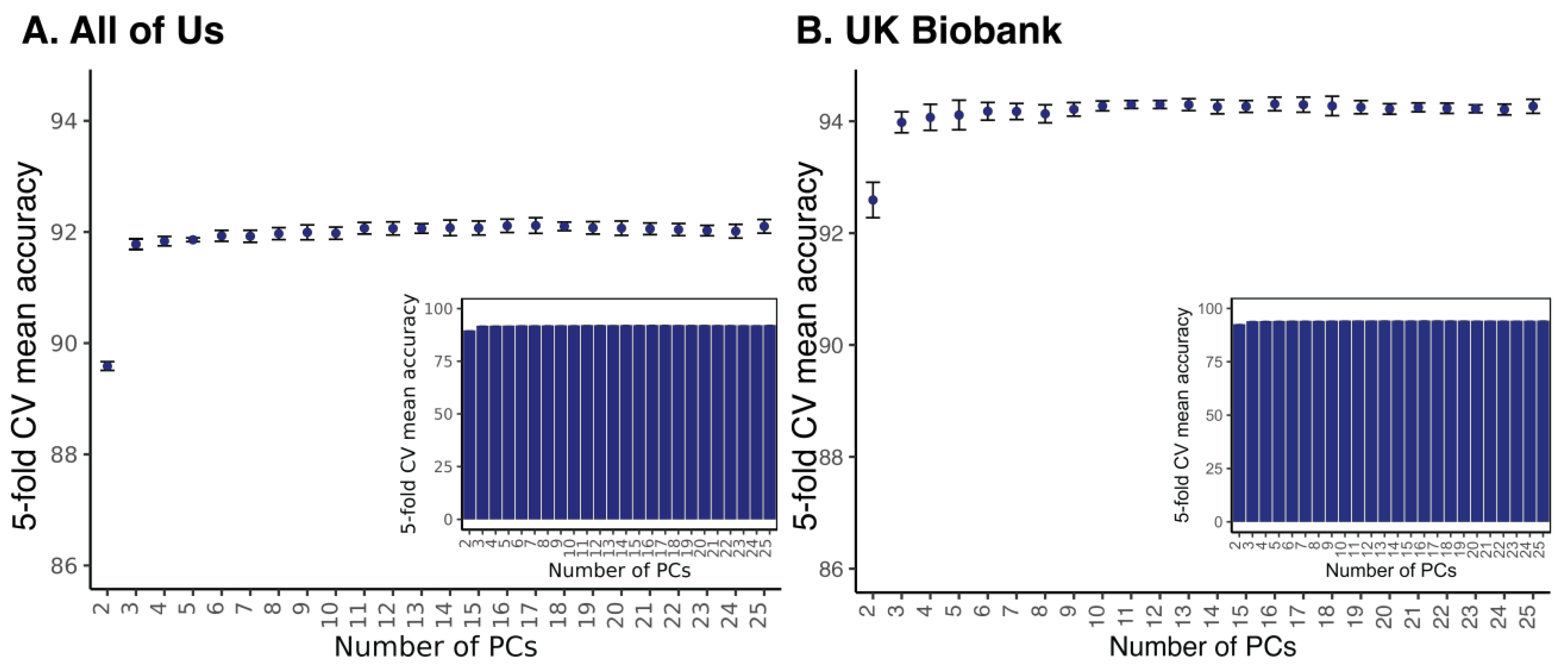
2.4. Machine Learning Prediction
2.5. Predicted Adverse Drug Reactions
3. Results
3.1. Race and Ethnicity in the All of Us and UKB Cohorts
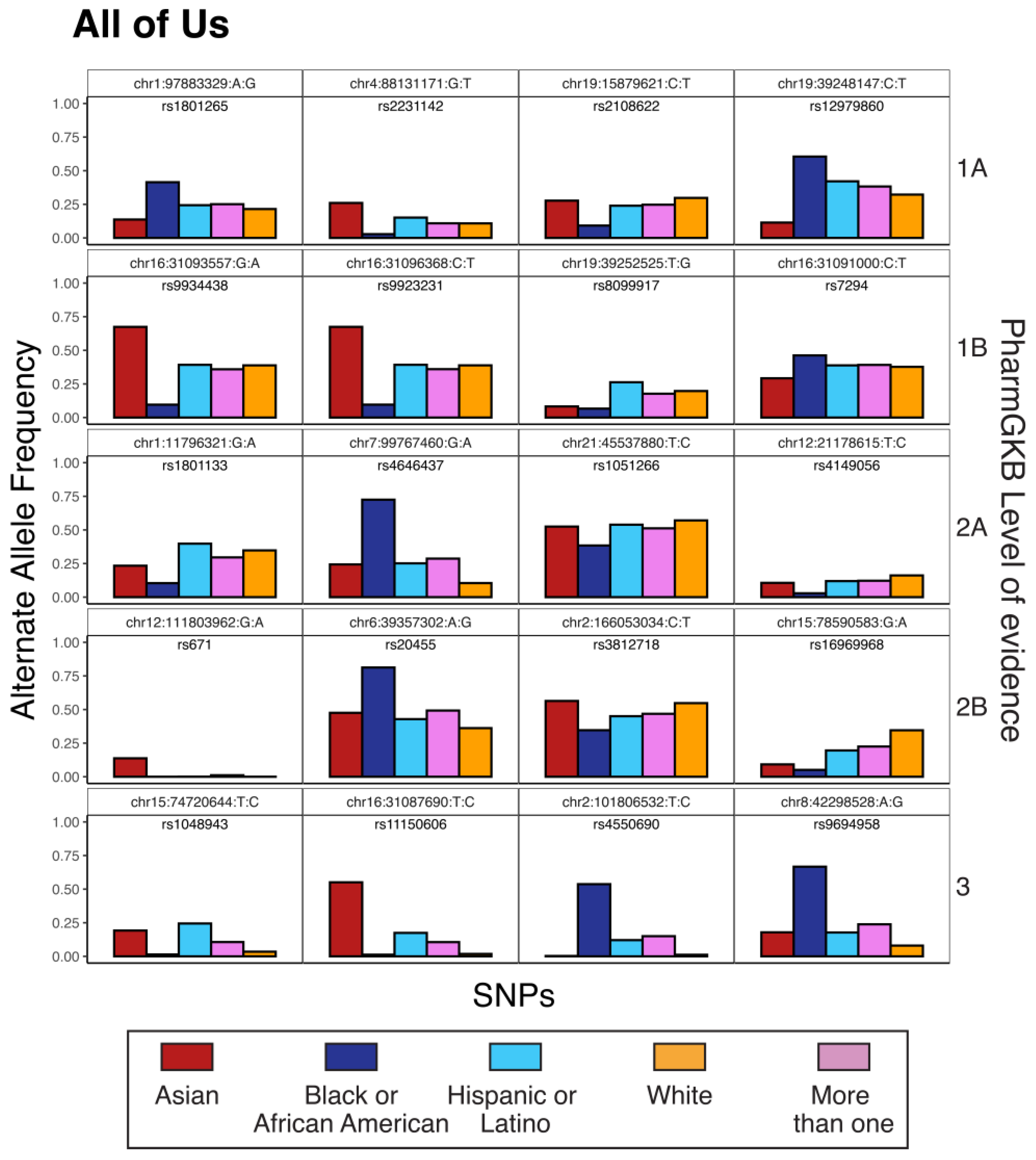
3.2. Pharmacogenomic Variation, Race, and Ethnicity
3.3. Adverse Drug Reactions
4. Discussion
Caveats and Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Eichelbaum, M.; Ingelman-Sundberg, M.; Evans, W.E. Pharmacogenomics and individualized drug therapy. Annu. Rev. Med. 2006, 57, 119–137. [Google Scholar] [CrossRef] [PubMed]
- Evans, W.E.; Relling, M.V. Pharmacogenomics: Translating functional genomics into rational therapeutics. Science 1999, 286, 487–491. [Google Scholar] [CrossRef] [PubMed]
- Relling, M.V.; Evans, W.E. Pharmacogenomics in the clinic. Nature 2015, 526, 343–350. [Google Scholar] [CrossRef] [PubMed]
- Pritchard, D.; Patel, J.N.; Stephens, L.E.; McLeod, H.L. Comparison of FDA Table of Pharmacogenetic Associations and Clinical Pharmacogenetics Implementation Consortium guidelines. Am. J. Health Syst. Pharm. 2022, 79, 993–1005. [Google Scholar] [CrossRef] [PubMed]
- Abdullah-Koolmees, H.; van Keulen, A.M.; Nijenhuis, M.; Deneer, V.H.M. Pharmacogenetics Guidelines: Overview and Comparison of the DPWG, CPIC, CPNDS, and RNPGx Guidelines. Front. Pharmacol. 2020, 11, 595219. [Google Scholar] [CrossRef] [PubMed]
- Yasuda, S.U.; Zhang, L.; Huang, S.M. The role of ethnicity in variability in response to drugs: Focus on clinical pharmacology studies. Clin. Pharmacol. Ther. 2008, 84, 417–423. [Google Scholar] [CrossRef]
- Nagar, S.D.; Moreno, A.M.; Norris, E.T.; Rishishwar, L.; Conley, A.B.; O’Neal, K.L.; Velez-Gomez, S.; Montes-Rodriguez, C.; Jaraba-Alvarez, W.V.; Torres, I.; et al. Population Pharmacogenomics for Precision Public Health in Colombia. Front. Genet. 2019, 10, 241. [Google Scholar] [CrossRef]
- Nagar, S.D.; Conley, A.B.; Jordan, I.K. Population structure and pharmacogenomic risk stratification in the United States. BMC Biol. 2020, 18, 140. [Google Scholar] [CrossRef]
- Huang, S.M.; Temple, R. Is this the drug or dose for you? Impact and consideration of ethnic factors in global drug development, regulatory review, and clinical practice. Clin. Pharmacol. Ther. 2008, 84, 287–294. [Google Scholar] [CrossRef]
- Chen, M.L. Ethnic or racial differences revisited: Impact of dosage regimen and dosage form on pharmacokinetics and pharmacodynamics. Clin. Pharmacokinet. 2006, 45, 957–964. [Google Scholar] [CrossRef]
- Bjornsson, T.D.; Wagner, J.A.; Donahue, S.R.; Harper, D.; Karim, A.; Khouri, M.S.; Murphy, W.R.; Roman, K.; Schneck, D.; Sonnichsen, D.S.; et al. A review and assessment of potential sources of ethnic differences in drug responsiveness. J. Clin. Pharmacol. 2003, 43, 943–967. [Google Scholar] [CrossRef] [PubMed]
- Bachtiar, M.; Lee, C.G. Genetics of population differences in drug response. Curr. Genet. Med. Rep. 2013, 1, 162–170. [Google Scholar] [CrossRef]
- Ramamoorthy, A.; Pacanowski, M.A.; Bull, J.; Zhang, L. Racial/ethnic differences in drug disposition and response: Review of recently approved drugs. Clin. Pharmacol. Ther. 2015, 97, 263–273. [Google Scholar] [CrossRef] [PubMed]
- Chauhan, D.; Onatade, R.; Clarke, H. We cannot ignore race when it comes to pharmacogenomics. Pharm. J. 2022, 308, 7691. [Google Scholar] [CrossRef]
- Rothstein, M.A.; Epps, P.G. Pharmacogenomics and the (ir)relevance of race. Pharmacogenomics J. 2001, 1, 104–108. [Google Scholar] [CrossRef]
- Ortega, V.E.; Meyers, D.A. Pharmacogenetics: Implications of race and ethnicity on defining genetic profiles for personalized medicine. J. Allergy Clin. Immunol. 2014, 133, 16–26. [Google Scholar] [CrossRef]
- Magavern, E.F.; Gurdasani, D.; Ng, F.L.; Lee, S.S. Health equality, race and pharmacogenomics. Br. J. Clin. Pharmacol. 2022, 88, 27–33. [Google Scholar] [CrossRef]
- Lee, S.S. Racializing drug design: Implications of pharmacogenomics for health disparities. Am. J. Public Health 2005, 95, 2133–2138. [Google Scholar] [CrossRef]
- Yudell, M.; Roberts, D.; DeSalle, R.; Tishkoff, S.; signatories. NIH must confront the use of race in science. Science 2020, 369, 1313–1314. [Google Scholar] [CrossRef]
- Yudell, M.; Roberts, D.; DeSalle, R.; Tishkoff, S. SCIENCE AND SOCIETY. Taking race out of human genetics. Science 2016, 351, 564–565. [Google Scholar] [CrossRef]
- Flanagin, A.; Frey, T.; Christiansen, S.L.; Committee, A.M.A.M.o.S. Updated Guidance on the Reporting of Race and Ethnicity in Medical and Science Journals. JAMA 2021, 326, 621–627. [Google Scholar] [CrossRef] [PubMed]
- Gannett, L. The biological reification of race. Br. J. Philos. Sci. 2004, 55, 323–345. [Google Scholar] [CrossRef]
- Braun, L. Reifying human difference: The debate on genetics, race, and health. Int. J. Health Serv. 2006, 36, 557–573. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.S.; Mountain, J.; Koenig, B.A. The meanings of “race” in the new genomics: Implications for health disparities research. Yale J. Health Policy Law Ethics 2001, 1, 33–75. [Google Scholar] [PubMed]
- Yuan, J.; Hu, Z.; Mahal, B.A.; Zhao, S.D.; Kensler, K.H.; Pi, J.; Hu, X.; Zhang, Y.; Wang, Y.; Jiang, J.; et al. Integrated Analysis of Genetic Ancestry and Genomic Alterations across Cancers. Cancer Cell 2018, 34, 549–560.e9. [Google Scholar] [CrossRef]
- Witherspoon, D.J.; Wooding, S.; Rogers, A.R.; Marchani, E.E.; Watkins, W.S.; Batzer, M.A.; Jorde, L.B. Genetic similarities within and between human populations. Genetics 2007, 176, 351–359. [Google Scholar] [CrossRef] [PubMed]
- Tang, H.; Quertermous, T.; Rodriguez, B.; Kardia, S.L.; Zhu, X.; Brown, A.; Pankow, J.S.; Province, M.A.; Hunt, S.C.; Boerwinkle, E.; et al. Genetic structure, self-identified race/ethnicity, and confounding in case-control association studies. Am. J. Hum. Genet. 2005, 76, 268–275. [Google Scholar] [CrossRef]
- Paschou, P.; Lewis, J.; Javed, A.; Drineas, P. Ancestry informative markers for fine-scale individual assignment to worldwide populations. J. Med. Genet. 2010, 47, 835–847. [Google Scholar] [CrossRef]
- Jin, Y.; Schaffer, A.A.; Feolo, M.; Holmes, J.B.; Kattman, B.L. GRAF-pop: A Fast Distance-Based Method To Infer Subject Ancestry from Multiple Genotype Datasets without Principal Components Analysis. G3 2019, 9, 2447–2461. [Google Scholar] [CrossRef] [PubMed]
- Fang, H.; Hui, Q.; Lynch, J.; Honerlaw, J.; Assimes, T.L.; Huang, J.; Vujkovic, M.; Damrauer, S.M.; Pyarajan, S.; Gaziano, J.M.; et al. Harmonizing Genetic Ancestry and Self-identified Race/Ethnicity in Genome-wide Association Studies. Am. J. Hum. Genet. 2019, 105, 763–772. [Google Scholar] [CrossRef]
- Edwards, A.W. Human genetic diversity: Lewontin’s fallacy. Bioessays 2003, 25, 798–801. [Google Scholar] [CrossRef] [PubMed]
- National Academies of Sciences, Engineering, and Medicine. Using Population Descriptors in Genetics and Genomics Research: A New Framework for an Evolving Field; The National Academies Press: Washington, DC, USA, 2023. [Google Scholar]
- Vyas, D.A.; Eisenstein, L.G.; Jones, D.S. Hidden in Plain Sight—Reconsidering the Use of Race Correction in Clinical Algorithms. N. Engl. J. Med. 2020, 383, 874–882. [Google Scholar] [CrossRef] [PubMed]
- Marino-Ramirez, L.; Sharma, S.; Rishishwar, L.; Conley, A.B.; Nagar, S.D.; Jordan, I.K. Effects of genetic ancestry and socioeconomic deprivation on ethnic differences in serum creatinine. Gene 2022, 837, 146709. [Google Scholar] [CrossRef] [PubMed]
- Hsu, C.Y.; Yang, W.; Parikh, R.V.; Anderson, A.H.; Chen, T.K.; Cohen, D.L.; He, J.; Mohanty, M.J.; Lash, J.P.; Mills, K.T.; et al. Race, Genetic Ancestry, and Estimating Kidney Function in CKD. N. Engl. J. Med. 2021, 385, 1750–1760. [Google Scholar] [CrossRef]
- Bycroft, C.; Freeman, C.; Petkova, D.; Band, G.; Elliott, L.T.; Sharp, K.; Motyer, A.; Vukcevic, D.; Delaneau, O.; O’Connell, J.; et al. The UK Biobank resource with deep phenotyping and genomic data. Nature 2018, 562, 203–209. [Google Scholar] [CrossRef]
- All of Us Research Program, I.; Denny, J.C.; Rutter, J.L.; Goldstein, D.B.; Philippakis, A.; Smoller, J.W.; Jenkins, G.; Dishman, E. The “All of Us” Research Program. N. Engl. J. Med. 2019, 381, 668–676. [Google Scholar] [CrossRef]
- Whirl-Carrillo, M.; McDonagh, E.M.; Hebert, J.M.; Gong, L.; Sangkuhl, K.; Thorn, C.F.; Altman, R.B.; Klein, T.E. Pharmacogenomics knowledge for personalized medicine. Clin. Pharmacol. Ther. 2012, 92, 414–417. [Google Scholar] [CrossRef]
- Purcell, S.; Neale, B.; Todd-Brown, K.; Thomas, L.; Ferreira, M.A.; Bender, D.; Maller, J.; Sklar, P.; de Bakker, P.I.; Daly, M.J.; et al. PLINK: A tool set for whole-genome association and population-based linkage analyses. Am. J. Hum. Genet. 2007, 81, 559–575. [Google Scholar] [CrossRef]
- Galinsky, K.J.; Bhatia, G.; Loh, P.R.; Georgiev, S.; Mukherjee, S.; Patterson, N.J.; Price, A.L. Fast Principal-Component Analysis Reveals Convergent Evolution of ADH1B in Europe and East Asia. Am. J. Hum. Genet. 2016, 98, 456–472. [Google Scholar] [CrossRef]
- Chang, C.C.; Chow, C.C.; Tellier, L.C.; Vattikuti, S.; Purcell, S.M.; Lee, J.J. Second-generation PLINK: Rising to the challenge of larger and richer datasets. Gigascience 2015, 4, 7. [Google Scholar] [CrossRef]
- Pedregosa, F.; Varoquaux, G.; Gramfort, A.; Michel, V.; Thirion, B.; Grisel, O.; Blondel, M.; Prettenhofer, P.; Weiss, R.; Dubourg, V. Scikit-learn: Machine learning in Python. J. Mach. Learn. Res. 2011, 12, 2825–2830. [Google Scholar]
- Chen, C.H.; Wang, S.C.; Tsou, H.H.; Ho, I.K.; Tian, J.N.; Yu, C.J.; Hsiao, C.F.; Chou, S.Y.; Lin, Y.F.; Fang, K.C.; et al. Genetic polymorphisms in CYP3A4 are associated with withdrawal symptoms and adverse reactions in methadone maintenance patients. Pharmacogenomics 2011, 12, 1397–1406. [Google Scholar] [CrossRef]
- Araoz, H.V.; D’Aloi, K.; Foncuberta, M.E.; Sanchez La Rosa, C.G.; Alonso, C.N.; Chertkoff, L.; Felice, M. Pharmacogenetic studies in children with acute lymphoblastic leukemia in Argentina. Leuk. Lymphoma 2015, 56, 1370–1378. [Google Scholar] [CrossRef]
- Xin, S.; Zhao, Y.; Wang, C.; Huang, Y.; Zhuang, W.; Ma, Y.; Huang, M.; Xu, X.; Wang, X.; Zhang, L. Polymorphisms of NF-kappaB pathway genes influence adverse drug reactions of gefitinib in NSCLC patients. Pharmacogenomics J. 2020, 20, 285–293. [Google Scholar] [CrossRef] [PubMed]
- Genomes Project, C.; Auton, A.; Brooks, L.D.; Durbin, R.M.; Garrison, E.P.; Kang, H.M.; Korbel, J.O.; Marchini, J.L.; McCarthy, S.; McVean, G.A.; et al. A global reference for human genetic variation. Nature 2015, 526, 68–74. [Google Scholar] [CrossRef]
- Bergstrom, A.; McCarthy, S.A.; Hui, R.; Almarri, M.A.; Ayub, Q.; Danecek, P.; Chen, Y.; Felkel, S.; Hallast, P.; Kamm, J.; et al. Insights into human genetic variation and population history from 929 diverse genomes. Science 2020, 367, eaay5012. [Google Scholar] [CrossRef] [PubMed]
- Schlebusch, C.M.; Malmstrom, H.; Gunther, T.; Sjodin, P.; Coutinho, A.; Edlund, H.; Munters, A.R.; Vicente, M.; Steyn, M.; Soodyall, H.; et al. Southern African ancient genomes estimate modern human divergence to 350,000 to 260,000 years ago. Science 2017, 358, 652–655. [Google Scholar] [CrossRef]
- Lewis, A.C.F.; Molina, S.J.; Appelbaum, P.S.; Dauda, B.; Di Rienzo, A.; Fuentes, A.; Fullerton, S.M.; Garrison, N.A.; Ghosh, N.; Hammonds, E.M.; et al. Getting genetic ancestry right for science and society. Science 2022, 376, 250–252. [Google Scholar] [CrossRef]
- Serre, D.; Paabo, S. Evidence for gradients of human genetic diversity within and among continents. Genome Res. 2004, 14, 1679–1685. [Google Scholar] [CrossRef]
- Nobles, M. History counts: A comparative analysis of racial/color categorization in US and Brazilian censuses. Am. J. Public Health 2000, 90, 1738–1745. [Google Scholar] [CrossRef]
- Huddart, R.; Fohner, A.E.; Whirl-Carrillo, M.; Wojcik, G.L.; Gignoux, C.R.; Popejoy, A.B.; Bustamante, C.D.; Altman, R.B.; Klein, T.E. Standardized Biogeographic Grouping System for Annotating Populations in Pharmacogenetic Research. Clin. Pharmacol. Ther. 2019, 105, 1256–1262. [Google Scholar] [CrossRef] [PubMed]
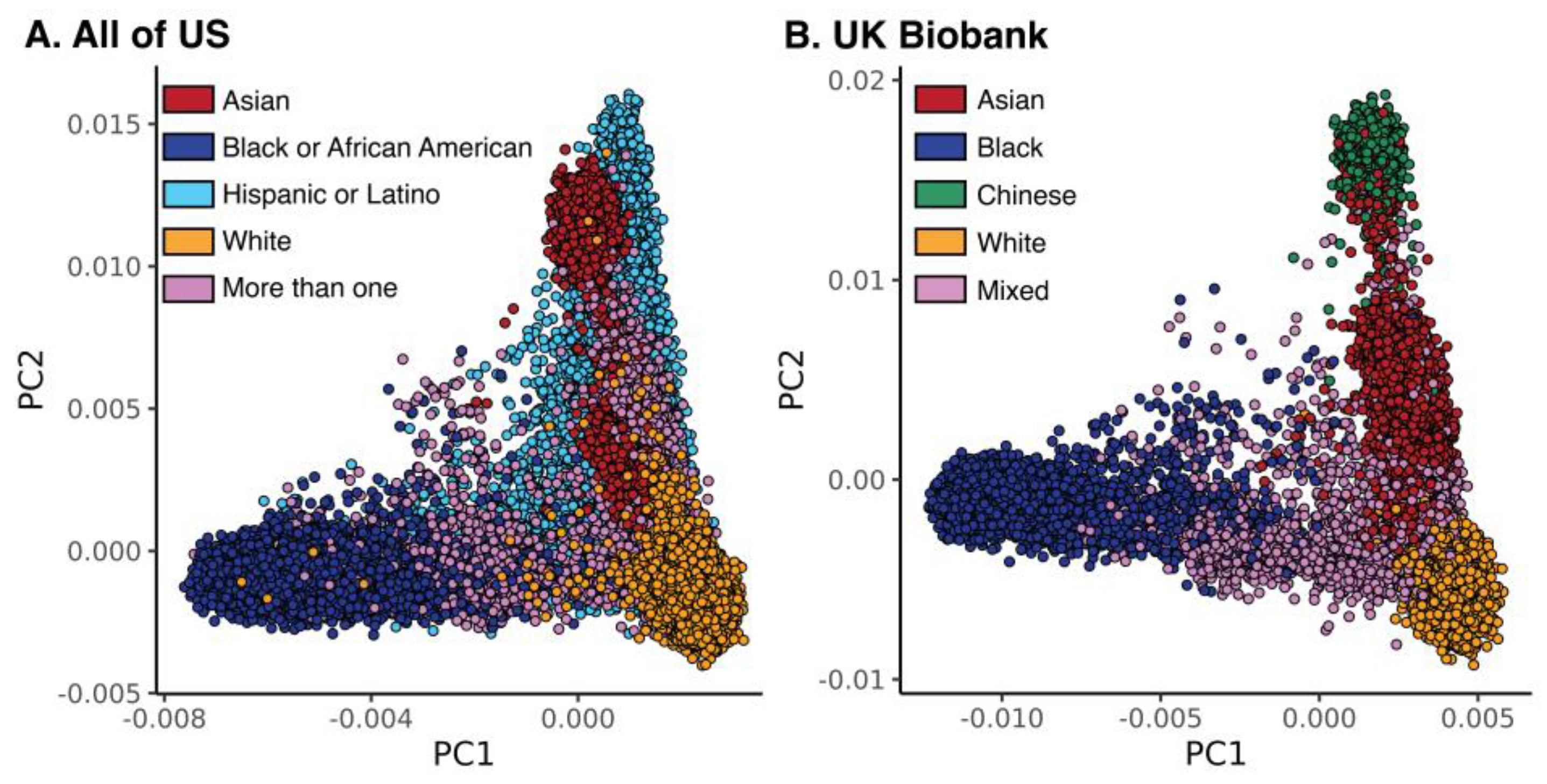
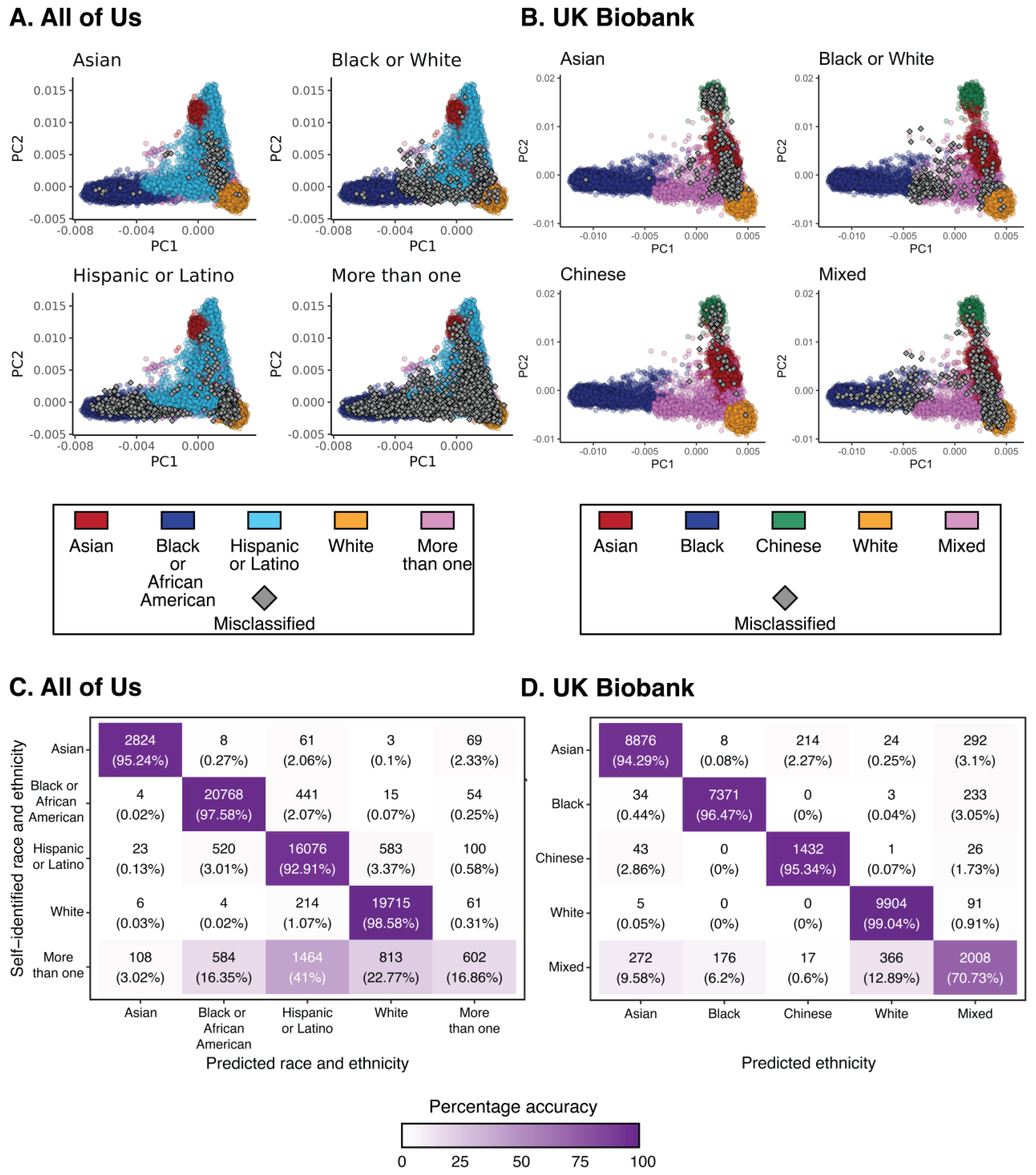
| All of Us | ||||||
| All | Asian | Black or African American | Hispanic or Latino | More than One | White | |
| N | 65,120 | 2965 | 21,282 | 17,302 | 3571 | 20,000 |
| Age (sd) | 53.63 (16.55) | 48.02 (16.89) | 53.00 (14.74) | 49.78 (15.79) | 46.04 (16.29) | 59.83 (17.00) |
| Female % | 60.79 | 59.53 | 55.26 | 68.55 | 64.72 | 59.45 |
| Male % | 38.21 | 40.00 | 43.26 | 30.64 | 34.42 | 39.81 |
| UKB | ||||||
| All | Asian | Chinese | Black | Mixed | White | |
| N | 31,396 | 9414 | 1502 | 7641 | 2839 | 10,000 |
| Age (sd) | 53.85 (8.41) | 53.30 (8.45) | 52.44 (7.67) | 51.91 (8.06) | 51.81 (8.13) | 56.65 (8.05) |
| Female % | 53.38 | 45.97 | 62.52 | 57.02 | 62.31 | 53.65 |
| Male % | 46.62 | 54.03 | 37.48 | 42.98 | 37.69 | 46.35 |
| Model | Hyperparameter a | Parameter Value | PCs Included | Mean Accuracy | Std. Dev. |
|---|---|---|---|---|---|
| K-Nearest Neighbors | K | 10 | 10 | 91.6% | 0.3% |
| Support Vector Machine | Kernel | Radial Basis Function | 15 | 94.0% | 0.1% |
| Regularization (C) | 10,000 | ||||
| Gamma | 10 | ||||
| Random Forest | Number of trees | 400 | 16 | 94.3% | 0.1% |
| Maximum depth of trees | 110 | ||||
| Minimum samples for leaf node | 2 | ||||
| Minimum samples to split node | 3 |
| Variant ID a | dbSNP ID b | Allele Weight (PC1, PC2) c | Level of Evidence d | Gene | Drug |
|---|---|---|---|---|---|
| chr1:97883329:A:G | rs1801265 | 1.2656, 1.039 | 1A | DPYD | capecitabine, fluorouracil |
| chr4:88131171:G:T | rs2231142 | 0.6817, 1.92 | 1A | ABCG2 | rosuvastatin |
| chr19:15879621:C:T | rs2108622 | 1.1024, 0.278 | 1A | CYP4F2 | warfarin |
| chr19:39248147:C:T | rs12979860 | 1.3562, 0.431 | 1A | IFNL3, IFNL4 | peginterferon alfa-2a, peginterferon alfa-2b, ribavirin, telaprevir, boceprevir |
| chr16:31093557:G:A | rs9934438 | 1.5132, 1.881 | 1B | VKORC1 | warfarin |
| chr16:31096368:C:T | rs9923231 | 1.5097, 1.881 | 1B | VKORC1 | warfarin |
| chr19:39252525:T:G | rs8099917 | 0.8534, 0.778 | 1B | IFNL3 | interferons, peginterferon alfa-2a, peginterferon alfa-2b, ribavirin |
| chr16:31091000:C:T | rs7294 | 0.4052, 0.466 | 1B | VKORC1 | warfarin |
| chr1:11796321:G:A | rs1801133 | 1.1024, 0.278 | 2A | MTHFR | methotrexate |
| chr7:99767460:G:A | rs4646437 | 3.2463, 0.22 | 2A | CYP3A4 | tacrolimus |
| chr21:45537880:T:C | rs1051266 | 0.8781, 0.147 | 2A | SLC19A1 | methotrexate |
| chr12:21178615:T:C | rs4149056 | 0.9113, 0.169 | 2A | SLCO1B1 | hmg coa reductase inhibitors |
| chr12:111803962:G:A | rs671 | 0.0322, 2.559 | 2B | ALDH2 | ethanol |
| chr6:39357302:A:G | rs20455 | 2.136, 0.483 | 2B | KIF6 | pravastatin |
| chr2:166053034:C:T | rs3812718 | 0.8992, 0.115 | 2B | SCN1A | carbamazepine |
| chr15:78590583:G:A | rs16969968 | 1.5382, 1.389 | 2B | CHRNA5 | nicotine |
| chr15:74720644:T:C | rs1048943 | 0.3912, 4.219 | 3 | CYP1A1 | capecitabine, docetaxel |
| chr16:31087690:T:C | rs11150606 | 0.2794, 5.708 | 3 | VKORC1 | warfarin |
| chr2:101806532:T:C | rs4550690 | 3.4072, 0.786 | 3 | MAP4K4 | anastrozole, exemestane |
| chr8:42298528:A:G | rs9694958 | 3.2572, 0.772 | 3 | IKBKB | gefitinib |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sharma, S.; Mariño-Ramírez, L.; Jordan, I.K. Race, Ethnicity, and Pharmacogenomic Variation in the United States and the United Kingdom. Pharmaceutics 2023, 15, 1923. https://doi.org/10.3390/pharmaceutics15071923
Sharma S, Mariño-Ramírez L, Jordan IK. Race, Ethnicity, and Pharmacogenomic Variation in the United States and the United Kingdom. Pharmaceutics. 2023; 15(7):1923. https://doi.org/10.3390/pharmaceutics15071923
Chicago/Turabian StyleSharma, Shivam, Leonardo Mariño-Ramírez, and I. King Jordan. 2023. "Race, Ethnicity, and Pharmacogenomic Variation in the United States and the United Kingdom" Pharmaceutics 15, no. 7: 1923. https://doi.org/10.3390/pharmaceutics15071923
APA StyleSharma, S., Mariño-Ramírez, L., & Jordan, I. K. (2023). Race, Ethnicity, and Pharmacogenomic Variation in the United States and the United Kingdom. Pharmaceutics, 15(7), 1923. https://doi.org/10.3390/pharmaceutics15071923







