Immune-Modulating Lipid Nanomaterials for the Delivery of Biopharmaceuticals
Abstract
1. Introduction
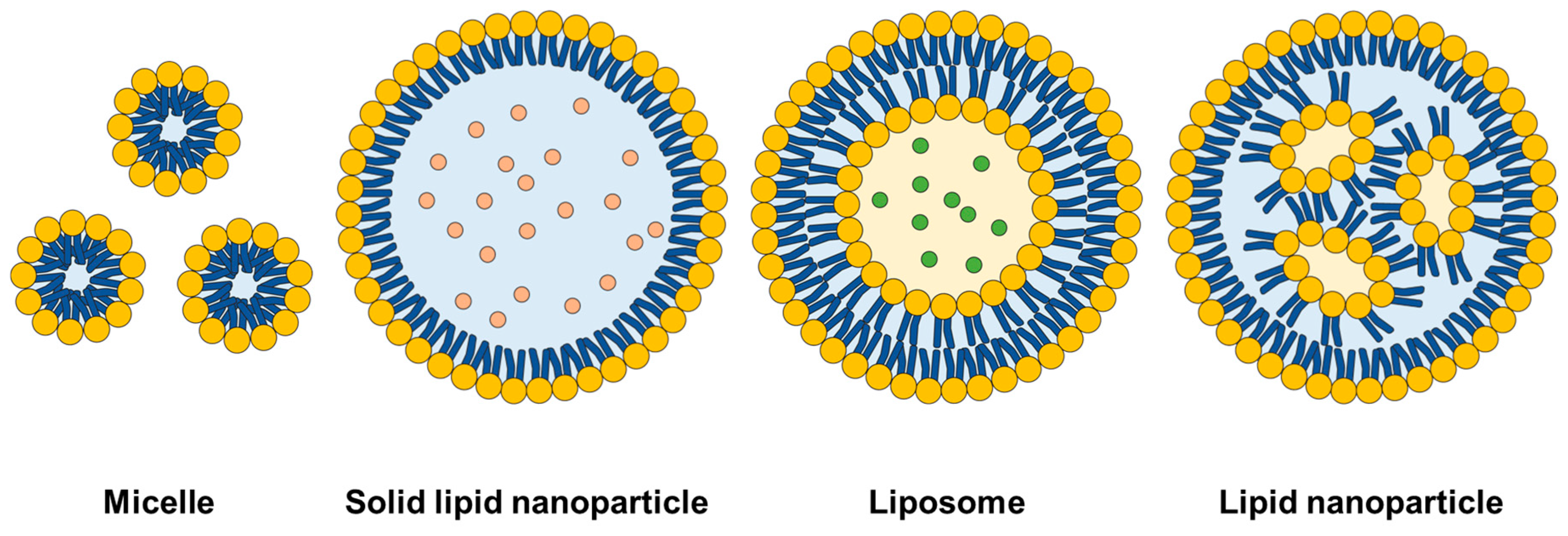
2. Design of Lipid Nanoparticles
2.1. Typical Composition
2.2. Composition for Ionizable LNP
3. Lipid Nanoparticles for Biopharmaceutical Delivery
4. Target Organs of Lipid Nanoparticles
5. Immune-Modulating Lipid Nanoparticles
5.1. siRNA-LNP
5.2. Gene-Editing LNP
5.3. mRNA-LNP
5.4. Cyclic Di-Nucleotide-LNP
5.5. Peptide-LNP
6. Challenges and Perspectives
7. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Gupta, U.; Saren, B.N.; Khaparkhuntikar, K.; Madan, J.; Singh, P.K. Applications of lipid-engineered nanoplatforms in the delivery of various cancer therapeutics to surmount breast cancer. J. Control. Release 2022, 348, 1089–1115. [Google Scholar] [CrossRef] [PubMed]
- Pinto, I.S.; Cordeiro, R.A.; Faneca, H. Polymer- and lipid-based gene delivery technology for CAR T cell therapy. J. Control. Release 2023, 353, 196–215. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Wang, J.; Wu, W.; Tony To, S.S.; Zhao, H.; Wang, J. Advances in lipid-based drug delivery: Enhancing efficiency for hydrophobic drugs. Expert Opin. Drug Deliv. 2015, 12, 1475–1499. [Google Scholar] [CrossRef]
- Paliwal, R.; Paliwal, S.R.; Kenwat, R.; Kurmi, B.D.; Sahu, M.K. Solid lipid nanoparticles: A review on recent perspectives and patents. Expert Opin. Ther. Pat. 2020, 30, 179–194. [Google Scholar] [CrossRef] [PubMed]
- Mirchandani, Y.; Patravale, V.B.; Brijesh, S. Solid lipid nanoparticles for hydrophilic drugs. J. Control. Release 2021, 335, 457–464. [Google Scholar] [CrossRef]
- Shah, S.; Dhawan, V.; Holm, R.; Nagarsenker, M.S.; Perrie, Y. Liposomes: Advancements and innovation in the manufacturing process. Adv. Drug Deliv. Rev. 2020, 154–155, 102–122. [Google Scholar] [CrossRef]
- Tenchov, R.; Bird, R.; Curtze, A.E.; Zhou, Q. Lipid Nanoparticles—From Liposomes to mRNA Vaccine Delivery, a Landscape of Research Diversity and Advancement. ACS Nano 2021, 15, 16982–17015. [Google Scholar] [CrossRef]
- Eygeris, Y.; Patel, S.; Jozic, A.; Sahay, G. Deconvoluting Lipid Nanoparticle Structure for Messenger RNA Delivery. Nano Lett. 2020, 20, 4543–4549. [Google Scholar] [CrossRef]
- Hou, X.; Zaks, T.; Langer, R.; Dong, Y. Lipid nanoparticles for mRNA delivery. Nat. Rev. Mater. 2021, 6, 1078–1094. [Google Scholar] [CrossRef]
- Xu, L.; Wang, X.; Liu, Y.; Yang, G.; Falconer, R.J.; Zhao, C.-X. Lipid Nanoparticles for Drug Delivery. Adv. NanoBiomed Res. 2022, 2, 2100109. [Google Scholar] [CrossRef]
- Thapa, R.K.; Kim, J.O. Nanomedicine-based commercial formulations: Current developments and future prospects. J. Pharm. Investig. 2023, 53, 19–33. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Tang, L.; Tian, Y.; Ji, X.; Hu, Q.; Zhou, B.; Ding, Z.; Xu, H.; Yang, L. DP7-C-modified liposomes enhance immune responses and the antitumor effect of a neoantigen-based mRNA vaccine. J. Control. Release 2020, 328, 210–221. [Google Scholar] [CrossRef]
- Karmacharya, P.; Patil, B.R.; Kim, J.O. Recent advancements in lipid–mRNA nanoparticles as a treatment option for cancer immunotherapy. J. Pharm. Investig. 2022, 52, 415–426. [Google Scholar] [CrossRef] [PubMed]
- Shim, G.; Jeong, S.; Oh, J.L.; Kang, Y. Lipid-based nanoparticles for photosensitive drug delivery systems. J. Pharm. Investig. 2022, 52, 151–160. [Google Scholar] [CrossRef] [PubMed]
- Krzysztoń, R.; Salem, B.; Lee, D.J.; Schwake, G.; Wagner, E.; Rädler, J.O. Microfluidic self-assembly of folate-targeted monomolecular siRNA-lipid nanoparticles. Nanoscale 2017, 9, 7442–7453. [Google Scholar] [CrossRef]
- Baran-Rachwalska, P.; Torabi-Pour, N.; Sutera, F.M.; Ahmed, M.; Thomas, K.; Nesbit, M.A.; Welsh, M.; Moore, C.B.T.; Saffie-Siebert, S.R. Topical siRNA delivery to the cornea and anterior eye by hybrid silicon-lipid nanoparticles. J. Control. Release 2020, 326, 192–202. [Google Scholar] [CrossRef]
- Wang, M.-Z.; Gu, T.-W.; Xu, Y.; Yang, L.; Jiang, Z.-H.; Peng, L.-H. Mechanical stretching of cells and lipid nanoparticles for nucleic acid delivery. J. Control. Release 2021, 339, 208–219. [Google Scholar] [CrossRef]
- Chen, S.; Tam, Y.Y.C.; Lin, P.J.C.; Sung, M.M.H.; Tam, Y.K.; Cullis, P.R. Influence of particle size on the in vivo potency of lipid nanoparticle formulations of siRNA. J. Control. Release 2016, 235, 236–244. [Google Scholar] [CrossRef]
- Saunders, N.R.M.; Paolini, M.S.; Fenton, O.S.; Poul, L.; Devalliere, J.; Mpambani, F.; Darmon, A.; Bergère, M.; Jibault, O.; Germain, M. A Nanoprimer To Improve the Systemic Delivery of siRNA and mRNA. Nano Lett. 2020, 20, 4264–4269. [Google Scholar] [CrossRef]
- Sato, Y.; Note, Y.; Maeki, M.; Kaji, N.; Baba, Y.; Tokeshi, M.; Harashima, H. Elucidation of the physicochemical properties and potency of siRNA-loaded small-sized lipid nanoparticles for siRNA delivery. J. Control. Release 2016, 229, 48–57. [Google Scholar] [CrossRef]
- He, Z.; Hu, Y.; Nie, T.; Tang, H.; Zhu, J.; Chen, K.; Liu, L.; Leong, K.W.; Chen, Y.; Mao, H.Q. Size-controlled lipid nanoparticle production using turbulent mixing to enhance oral DNA delivery. Acta Biomater. 2018, 81, 195–207. [Google Scholar] [CrossRef] [PubMed]
- Kimura, N.; Maeki, M.; Sato, Y.; Ishida, A.; Tani, H.; Harashima, H.; Tokeshi, M. Development of a microfluidic-based post-treatment process for size-controlled lipid nanoparticles and application to SiRNA delivery. ACS Appl. Mater. Interfaces 2020, 12, 34011–34020. [Google Scholar] [CrossRef] [PubMed]
- Knudson, C.J.; Alves-Peixoto, P.; Muramatsu, H.; Stotesbury, C.; Tang, L.; Lin, P.J.C.; Tam, Y.K.; Weissman, D.; Pardi, N.; Sigal, L.J. Lipid-nanoparticle-encapsulated mRNA vaccines induce protective memory CD8 T cells against a lethal viral infection. Mol. Ther. 2021, 29, 2769–2781. [Google Scholar] [CrossRef] [PubMed]
- Rojas, L.A.; Sethna, Z.; Soares, K.C.; Olcese, C.; Pang, N.; Patterson, E.; Lihm, J.; Ceglia, N.; Guasp, P.; Chu, A. Personalized RNA neoantigen vaccines stimulate T cells in pancreatic cancer. Nature 2023, 618, 144–150. [Google Scholar] [CrossRef]
- Blakney, A.K.; McKay, P.F.; Hu, K.; Samnuan, K.; Jain, N.; Brown, A.; Thomas, A.; Rogers, P.; Polra, K.; Sallah, H. Polymeric and lipid nanoparticles for delivery of self-amplifying RNA vaccines. J. Control. Release 2021, 338, 201–210. [Google Scholar] [CrossRef]
- Kauffman, K.J.; Dorkin, J.R.; Yang, J.H.; Heartlein, M.W.; DeRosa, F.; Mir, F.F.; Fenton, O.S.; Anderson, D.G. Optimization of lipid nanoparticle formulations for mRNA Delivery in vivo with fractional factorial and definitive screening designs. Nano Lett. 2015, 15, 7300–7306. [Google Scholar] [CrossRef]
- Shim, G.; Miao, W.; Ko, S.; Park, G.T.; Kim, J.Y.; Kim, M.G.; Kim, Y.B.; OH, Y.K. Immune-camouflaged graphene oxide nanosheets for negative regulation of phagocytosis by macrophages. J. Mater. Chem. B 2017, 5, 6666–6675. [Google Scholar] [CrossRef]
- Ibrahim, M.; Ramadan, E.; Elsadek, N.E.; Emam, S.E.; Shimizu, T.; Ando, H.; Ishima, Y.; Elgarhy, O.H.; Sarhan, H.A.; Hussein, A.K.; et al. Polyethylene glycol (PEG): The nature, immunogenicity, and role in the hypersensitivity of PEGylated products. J. Control. Release 2022, 351, 215–230. [Google Scholar] [CrossRef]
- Suzuki, Y.; Hyodo, K.; Tanaka, Y.; Ishihara, H. siRNA-lipid nanoparticles with long-term storage stability facilitate potent gene-silencing in vivo. J. Control. Release 2015, 220, 44–50. [Google Scholar] [CrossRef]
- Ramishetti, S.; Hazan-Halevy, I.; Palakuri, R.; Chatterjee, S.; Naidu Gonna, S.; Dammes, N.; Freilich, I.; Shmuel, L.K.; Danino, D.; Peer, D. A combinatorial library of lipid nanoparticles for RNA delivery to leukocytes. Adv. Mater. 2020, 32, 1906128. [Google Scholar] [CrossRef] [PubMed]
- Davies, N.; Hovdal, D.; Edmunds, N.; Nordberg, P.; Dahlén, A.; Dabkowska, A.; Yanez Arteta, M.; Radulescu, A.; Kjellman, T.; Höijer, A.; et al. Functionalized lipid nanoparticles for subcutaneous administration of mRNA to achieve systemic exposures of a therapeutic protein. Mol. Ther. Nucleic Acids 2021, 24, 369–384. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Kong, N.; Zhang, X.; Cao, Y.; Langer, R.; Tao, W. The landscape of mRNA nanomedicine. Nat. Med. 2022, 28, 2273–2287. [Google Scholar] [CrossRef]
- Lou, G.; Anderluzzi, G.; Schmidt, S.T.; Woods, S.; Gallorini, S.; Brazzoli, M.; Giusti, F.; Ferlenghi, I.; Johnson, R.N.; Roberts, C.W.; et al. Delivery of self-amplifying mRNA vaccines by cationic lipid nanoparticles, The impact of cationic lipid selection. J. Control. Release 2020, 325, 370–379. [Google Scholar] [CrossRef] [PubMed]
- Rybakova, Y.; Kowalski, P.S.; Huang, Y.; Gonzalez, J.T.; Heartlein, M.W.; DeRosa, F.; Delcassian, D.; Anderson, D.G. mRNA delivery for therapeutic anti-HER2 antibody expression in vivo. Mol. Ther. 2019, 27, 1415–1423. [Google Scholar] [CrossRef] [PubMed]
- Rizvi, F.; Everton, E.; Smith, A.R.; Liu, H.; Osota, E.; Beattie, M.; Tam, Y.; Pardi, N.; Weissman, D.; Evans, V.G. Murine liver repair via transient activation of regenerative pathways in hepatocytes using lipid nanoparticle-complexed nucleoside-modified mRNA. Nat. Commun. 2021, 12, 613. [Google Scholar] [CrossRef]
- Suzuki, Y.; Onuma, H.; Sato, R.; Sato, Y.; Hashiba, A.; Maeki, M.; Tokeshi, M.; Kayesh, M.E.H.; Kohara, M.; Tsukiyama-Kohara, K.; et al. Lipid nanoparticles loaded with ribonucleoprotein–oligonucleotide complexes synthesized using a microfluidic device exhibit robust genome editing and hepatitis B virus inhibition. J. Control. Release 2021, 330, 61–71. [Google Scholar] [CrossRef]
- Kaczmarek, J.C.; Patel, A.K.; Rhym, L.H.; Palmiero, U.C.; Bhat, B.; Heartlein, M.W.; DeRosa, F.; Anderson, D.G. Systemic delivery of mRNA and DNA to the lung using polymer-lipid nanoparticles. Biomaterials 2021, 275, 120966. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Q.; Wei, T.; Farbiak, L.; Johnson, L.T.; Dilliard, S.A.; Siegwart, D.J. Selective organ targeting (SORT) nanoparticles for tissue-specific mRNA delivery and CRISPR–Cas gene editing. Nat. Nanotechnol. 2020, 15, 313–320. [Google Scholar] [CrossRef]
- Sago, C.D.; Lokugamage, M.P.; Islam, F.Z.; Krupczak, B.R.; Sato, M.; Dahlman, J.E. Nanoparticles that deliver RNA to bone marrow identified by in vivo directed evolution. J. Am. Chem. Soc. 2018, 140, 17095–17105. [Google Scholar] [CrossRef]
- Katakowski, J.A.; Mukherjee, G.; Wilner, S.E.; Maier, K.E.; Harrison, M.T.; DiLorenzo, T.P.; Levy, M.; Palliser, D. Delivery of siRNAs to dendritic cells using DEC205-targeted lipid nanoparticles to inhibit immune responses. Mol. Ther. 2016, 24, 146–155. [Google Scholar] [CrossRef]
- Sato, Y.; Matsui, H.; Yamamoto, N.; Sato, R.; Munakata, T.; Kohara, M.; Harashima, H. Highly specific delivery of siRNA to hepatocytes circumvents endothelial cell-mediated lipid nanoparticle-associated toxicity leading to the safe and efficacious decrease in the hepatitis B virus. J. Control. Release 2017, 266, 216–225. [Google Scholar] [CrossRef] [PubMed]
- Patel, S.; Ashwanikumar, N.; Robinson, E.; Xia, Y.; Mihai, C.; Griffith, J.P.; Hou, S.; Esposito, A.A.; Ketova, T.; Welsher, K.; et al. Naturally-occurring cholesterol analogues in lipid nanoparticles induce polymorphic shape and enhance intracellular delivery of mRNA. Nat. Commun. 2020, 11, 983. [Google Scholar] [CrossRef]
- Yong, S.-B.; Ramishetti, S.; Goldsmith, M.; Diesendruck, Y.; Hazan-Halevy, I.; Chatterjee, S.; Naidu, G.S.; Ezra, A.; Peer, D. Dual-targeted lipid nanotherapeutic boost for chemo-immunotherapy of cancer. Adv. Mater. 2022, 34, 2106350. [Google Scholar] [CrossRef] [PubMed]
- Jung, M.; Lee, S.; Park, S.; Hong, J.; Kim, C.; Cho, I.; Sohn, H.S.; Kim, K.; Park, I.W.; Yoon, S.; et al. A therapeutic nanovaccine that generates anti-amyloid antibodies and amyloid-specific regulatory T cells for Alzheimer’s disease. Adv. Mater. 2023, 35, 2207719. [Google Scholar] [CrossRef] [PubMed]
- Bevers, S.; Kooijmans, S.A.A.; van de Velde, E.; Evers, M.J.W.; Seghers, S.; Gitz-Francois, J.J.J.M.; van Kronenburg, N.C.H.; Fens, M.H.A.M.; Mastrobattista, E.; Hassler, L.; et al. mRNA-LNP vaccines tuned for systemic immunization induce strong antitumor immunity by engaging splenic immune cells. Mol. Ther. 2022, 30, 3078–3094. [Google Scholar] [CrossRef]
- Liu, S.; Liu, J.; Li, H.; Mao, K.; Wang, H.; Meng, X.; Wang, J.; Wu, C.; Chen, H.; Wang, X.; et al. An optimized ionizable cationic lipid for brain tumor-targeted siRNA delivery and glioblastoma immunotherapy. Biomaterials 2022, 287, 121645. [Google Scholar] [CrossRef]
- Nakamura, T.; Kawakami, K.; Nomura, M.; Sato, Y.; Hyodo, M.; Hatakeyama, H.; Hayakawa, Y.; Harashima, H. Combined nano cancer immunotherapy based on immune status in a tumor microenvironment. J. Control. Release 2022, 345, 200–213. [Google Scholar] [CrossRef]
- Liu, Y.; Wang, L.; Song, Q.; Ali, M.; Crowe, W.N.; Kucera, G.L.; Hawkins, G.A.; Soker, S.; Thomas, K.W.; Miller, L.D.; et al. Intrapleural nano-immunotherapy promotes innate and adaptive immune responses to enhance anti-PD-L1 therapy for malignant pleural effusion. Nat. Nanotechnol. 2022, 17, 206–216. [Google Scholar] [CrossRef]
- Dammes, N.; Goldsmith, M.; Ramishetti, S.; Dearling, J.L.J.; Veiga, N.; Packard, A.B.; Peer, D. Conformation-sensitive targeting of lipid nanoparticles for RNA therapeutics. Nat. Nanotechnol. 2021, 16, 1030–1038. [Google Scholar] [CrossRef]
- Billingsley, M.M.; Singh, N.; Ravikumar, P.; Zhang, R.; June, C.H.; Mitchell, M.J. Ionizable lipid nanoparticle-mediated mRNA delivery for human CAR T cell engineering. Nano Lett. 2020, 20, 1578–1589. [Google Scholar] [CrossRef]
- Kim, D.; Wu, Y.; Shim, G.; Oh, Y.-K. Genome-editing-mediated restructuring of tumor immune microenvironment for prevention of metastasis. ACS Nano 2021, 15, 17635–17656. [Google Scholar] [CrossRef]
- Nakamura, T.; Sato, T.; Endo, R.; Sasaki, S.; Takahashi, N.; Sato, Y.; Hyodo, M.; Hayakawa, Y.; Harashima, H. STING agonist loaded lipid nanoparticles overcome anti-PD-1 resistance in melanoma lung metastasis via NK cell activation. J. Immunother. Cancer 2021, 9, e002852. [Google Scholar] [CrossRef]
- Akinc, A.; Zumbuehl, A.; Goldberg, M.; Leshchiner, E.S.; Busini, V.; Hossain, N.; Bacallado, S.A.; Nguyen, D.N.; Fuller, J.; Alvarez, R.; et al. A combinatorial library of lipid-like materials for delivery of RNAi therapeutics. Nat. Biotechnol. 2008, 26, 561–569. [Google Scholar] [CrossRef] [PubMed]
- Sabnis, S.; Kumarasinghe, E.S.; Salerno, T.; Mihai, C.; Ketova, T.; Senn, J.J.; Lynn, A.; Bulychev, A.; McFadyen, I.; Chan, J.; et al. A novel amino lipid series for mRNA delivery, improved endosomal escape and sustained pharmacology and safety in non-human primates. Mol. Ther. 2018, 26, 1509–1519. [Google Scholar] [CrossRef] [PubMed]
- Erasmus, J.H.; Khandhar, A.P.; Guderian, J.; Granger, B.; Archer, J.; Archer, M.; Gage, E.; Fuerte-Stone, J.; Larson, E.; Lin, S.; et al. A nanostructured lipid carrier for delivery of a replicating viral RNA provides single, low-dose protection against Zika. Mol. Ther. 2018, 26, 2507–2522. [Google Scholar] [CrossRef] [PubMed]
- Sago, C.D.; Lokugamage, M.P.; Paunovska, K.; Vanover, D.A.; Monaco, C.M.; Shah, N.N.; Gamboa Castro, M.; Anderson, S.E.; Rudoltz, T.G.; Lando, G.N.; et al. High-throughput in vivo screen of functional mRNA delivery identifies nanoparticles for endothelial cell gene editing. Proc. Natl. Acad. Sci. USA 2018, 115, E9944–E9952. [Google Scholar] [CrossRef]
- Malcolm, D.W.; Varghese, J.J.; Sorrells, J.E.; Ovitt, C.E.; Benoit, D.S.W. The effects of biological fluids on colloidal stability and siRNA delivery of a pH-responsive micellar nanoparticle delivery system. ACS Nano 2018, 12, 187–197. [Google Scholar] [CrossRef]
- Whitehead, K.A.; Matthews, J.; Chang, P.H.; Niroui, F.; Dorkin, J.R.; Severgnini, M.; Anderson, D.G. In Vitro–In Vivo Translation of Lipid Nanoparticles for Hepatocellular siRNA Delivery. ACS Nano 2012, 6, 6922–6929. [Google Scholar] [CrossRef]
- Tay, B.Q.; Wright, Q.; Ladwa, R.; Perry, C.; Leggatt, G.; Simpson, F.; Wells, J.W.; Panizza, B.J.; Frazer, I.H.; Cruz, J.L.G. Evolution of Cancer Vaccines-Challenges, Achievements, and Future Directions. Vaccines 2021, 9, 535. [Google Scholar] [CrossRef]
- Blass, E.; Ott, P.A. Advances in the development of personalized neoantigen-based therapeutic cancer vaccines. Nat. Rev. Clin. Oncol. 2021, 18, 215–229. [Google Scholar] [CrossRef]
- Miao, L.; Zhang, Y.; Huang, L. mRNA vaccine for cancer immunotherapy. Mol. Cancer 2021, 20, 41. [Google Scholar] [CrossRef] [PubMed]
| Category | Cargo | Disease | Mode of Action | LNP Type | Ref. |
|---|---|---|---|---|---|
| Immune cell activation | Heme-oxygenase-1-targeting siRNA | Metastatic melanoma | Heme oxygenase-1 silencing PD-L1 blockade | iLNP | [43] |
| Amyloid-β (Aβ) peptide, rapamycin | Alzheimer’s Disease | Generation of Aβ-specific Th cells and Treg cells | Cationic liposomes | [44] | |
| E7, CD70, CD40L, TLR4 mRNA | Cervical cancer | Antigen-specific CD8 T cell response | iLNP | [45] | |
| CD47- and PD-L1-targeting siRNA | Glioblastoma multiforme | Simultaneous silencing of CD47 and PD-L1 | Cationic liposomes | [46] | |
| Cyclic di-GMP | Lymphoma, breast cancer | STING pathway activation CTLA4 blockade | Cationic liposomes | [47] | |
| Cyclic GMP-AMP | Malignant pleural effusion in lung cancer | STING pathway activation PD-L1 blockade | Anionic liposomes | [48] | |
| Interferon-gamma-targeting siRNA | Inflammatory bowel disease | Interferon gamma silencing | iLNP | [49] | |
| mRNA-encoding human fibroblast growth factor 21, steroid prodrug | - | Anti-inflammatory response | iLNP | [31] | |
| Polo-like kinase 1 and CD45 siRNA | - | Leukocyte-selective targeting | iLNP | [30] | |
| mRNA-encoding CD19-targeting CAR bearing the CD3ζ and 4–1BB costimulatory domains | Leukemia | CD19-targeting CAR expression in T cells | iLNP | [50] | |
| Vaccination | EVM158 mRNA | Mousepox | Antigen-specific CD8 T cell response | cLNP | [23] |
| plasmid DNA encoding TGF-β single guide RNA and Cas9 protein | Melanoma | Transforming growth factor-β editing In situ vaccination of tumor-associated antigens | Cationic liposomes | [51] | |
| Self-amplifying RNA (saRNA) encoding the influenza hemagglutinin glycoprotein, SARS-CoV-2 spike protein | Influenza | Antigen-specific humoral and cellular response | iLNP | [25] | |
| mRNA encoding neoantigen | Lewis lung carcinoma | Antigen-specific cellular response | Cationic liposomes | [40] | |
| siRNA encoding the rabies virus glycoprotein | Rabies | Antigen-specific humoral and cellular response | cLNP | [33] |
| Brand Name | Cargo | Indication | Year | Company |
|---|---|---|---|---|
| Spikevax | mRNA | COVID-19 | 2020 | Moderna (Cambridge, MA, USA) |
| Comirnaty | mRNA | COVID-20 | 2020 | Pfizer-BioNTech (New York, NY, USA) |
| ONPATTRO | siRNA | Hereditary transthyretin-mediated amyloidosis | 2018 | Alnylam (Cambridge, MA, USA) |
| VYXEOS | Cytarabine/ daunorubicin | Acute myeloid leukemia | 2017 | Jazz Pharmaceuticals (Dublin, Ireland) |
| Onivyde | Irinotecan | Metastatic pancreatic cancer | 2015 | Merrimack (North Andover, MA, USA) |
| Marqibo | Vincristine | Philadelphia chromosome-negative acute lymphoblastic leukemia | 2012 | Spectrum (Reno, NV, USA) |
| Definity | Perflutren | Ultrasound enhancement for patients with suboptimal echocardiograms | 2001 | Lantheus Medical Imaging (North Billerica, MA, USA) |
| Visudyne | Verteporfin | Predominantly classic subfoveal choroidal neovascularization in patients with age-related macular degeneration (AMD), pathologic myopia, presumed ocular histoplasmosis | 2000 | Xediton Pharmaceuticals (Mississauga, ON, Canada) |
| AmBisome | Amphotericin B | A variety of serious fungal infections | 1997 | Gilead Sciences (Foster City, CA, USA) |
| DaunoXome | Daunorubicin | First-line therapy against advanced Kaposi’s sarcoma associated with HIV | 1996 | Galen (Craigavon, UK) |
| Doxil | Doxorubicin | Ovarian cancer, AIDS-related Kaposi sarcoma, and multiple myeloma | 1995 | Janssen (Beerse, Belgium) |
| Diprivan | Propofol | A sedative–hypnotic agent | 1989 | Fresenius Kabi (vor der Höhe, Germany) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, S.; Choi, B.; Kim, Y.; Shim, G. Immune-Modulating Lipid Nanomaterials for the Delivery of Biopharmaceuticals. Pharmaceutics 2023, 15, 1760. https://doi.org/10.3390/pharmaceutics15061760
Kim S, Choi B, Kim Y, Shim G. Immune-Modulating Lipid Nanomaterials for the Delivery of Biopharmaceuticals. Pharmaceutics. 2023; 15(6):1760. https://doi.org/10.3390/pharmaceutics15061760
Chicago/Turabian StyleKim, Songhee, Boseung Choi, Yoojin Kim, and Gayong Shim. 2023. "Immune-Modulating Lipid Nanomaterials for the Delivery of Biopharmaceuticals" Pharmaceutics 15, no. 6: 1760. https://doi.org/10.3390/pharmaceutics15061760
APA StyleKim, S., Choi, B., Kim, Y., & Shim, G. (2023). Immune-Modulating Lipid Nanomaterials for the Delivery of Biopharmaceuticals. Pharmaceutics, 15(6), 1760. https://doi.org/10.3390/pharmaceutics15061760










