Quercetin and Its Nano-Formulations for Brain Tumor Therapy—Current Developments and Future Perspectives for Paediatric Studies
Abstract
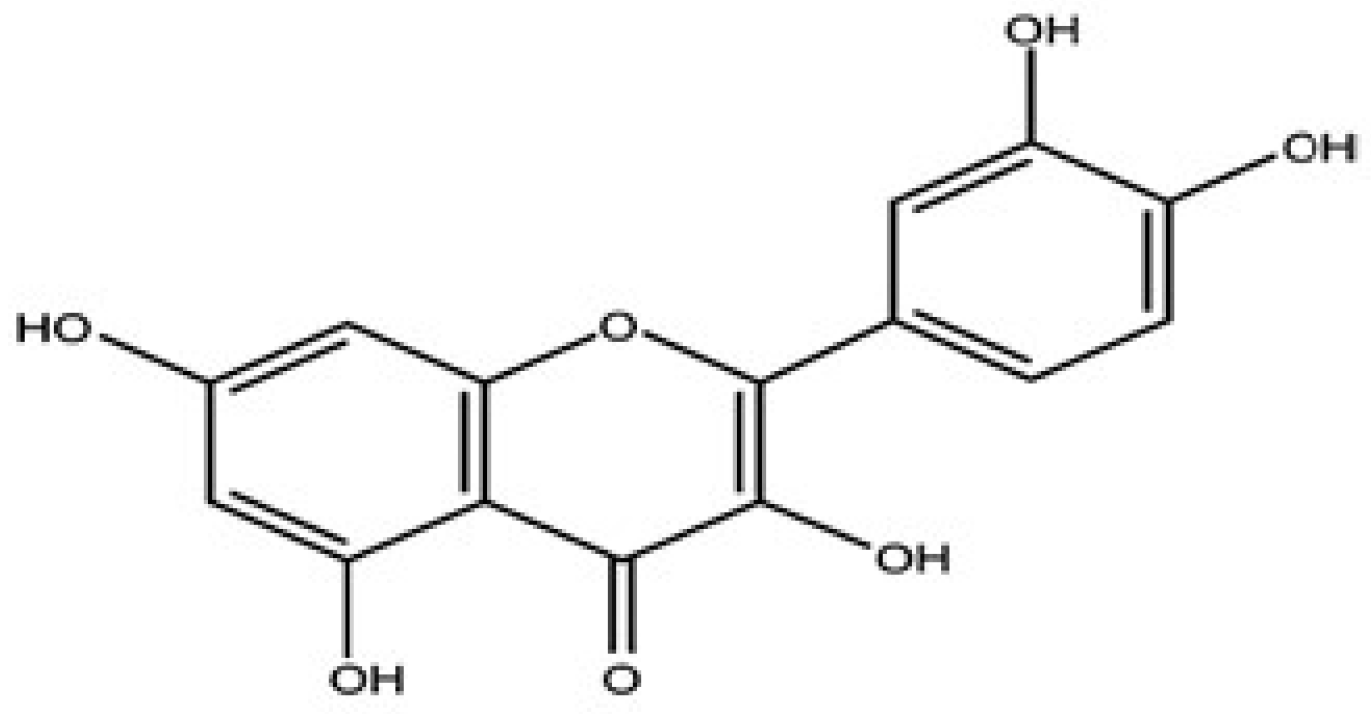
1. Introduction
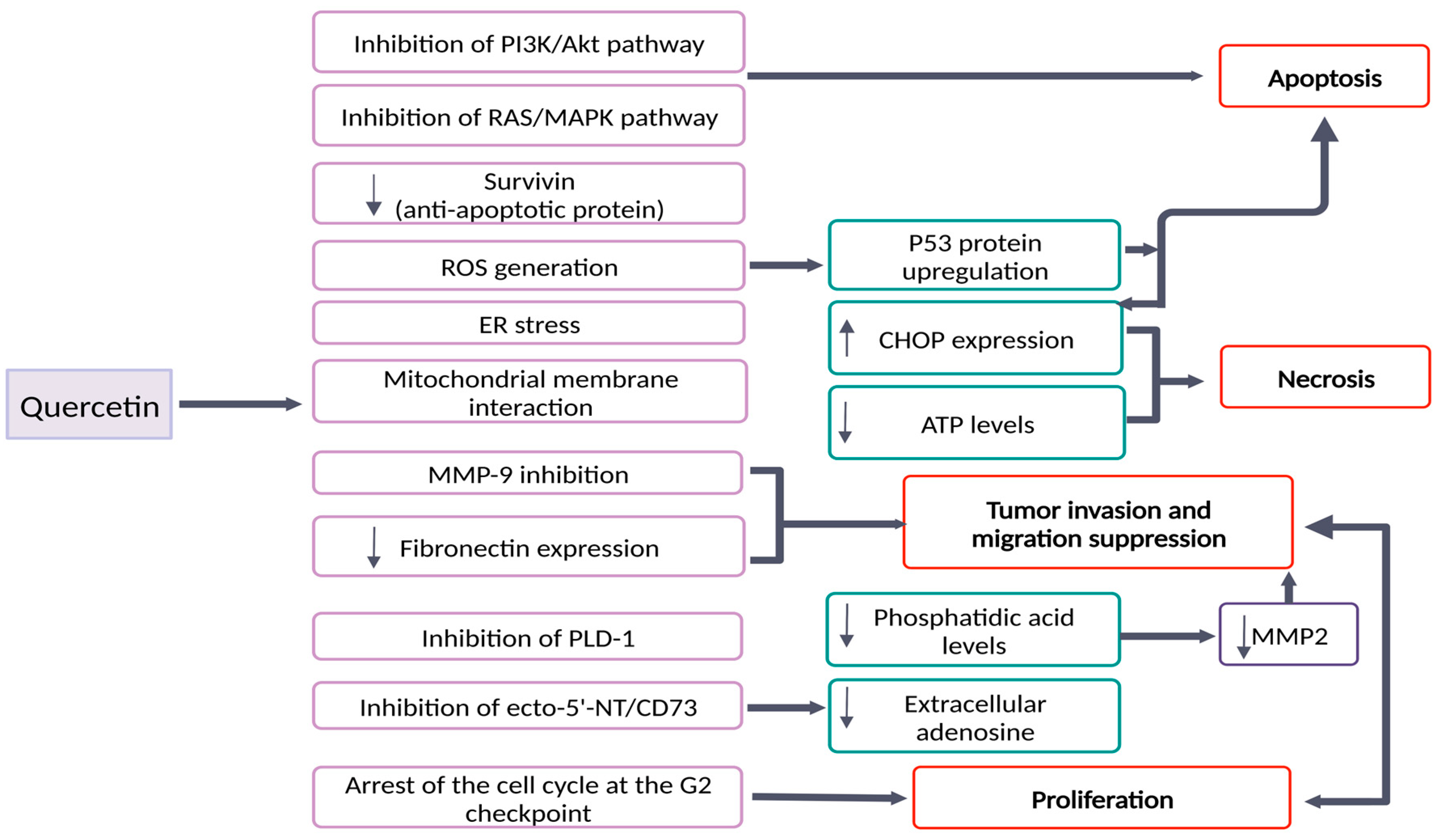
2. Quercetin’s Effects in Brain Tumors: Molecular Mechanisms and Signaling Pathways
| Type of Que | Cell Line | Results | Conclusions | References |
|---|---|---|---|---|
| Que | U87-MG U251 SHG44 | ↓ P-Akt ↓ P-ERK ↓ Bcl-2 ↓ MMP-9 ↓ Fibronectin | - inhibition of viability and migration of glioma cells - Apoptosis induction - Suppression of RAS/MAPK/ERK and PI3K/Akt pathways | Pan et al., 2015 [24] |
| PEG2000-DPSE-coated quercetin nano-particles | C6 | ↑ ROS production ↑ Cytotoxicity ↑ P53 expression ↓ Membrane potential - Cytochrome c release - Caspase activation | PEG2000-DPSE-coated quercetin nano-particles showed: - dose dependent toxicity on C6 cells - apoptosis induction through ROS accumulation - Enhancement of the anticancer effect of quercetin on C6 cells | Wang et al., 2013 [37] |
| Que | A172 LBC3 | ↑ ROS generation ↑ Caspase 3/7/9 activity ↑ CHOP - Deregulated SOD1 and SOD2 expression ↓ ATP | - induction of apoptosis via intrinsic pathway on A172 cell line - apoptosis/necrosis shift after 48 h of treatment on LBC3 cell line | Kusaczuk et al., 2022 [27] |
| Que | U138MG | ↓ cell membrane permeability ↑ Caspase 3/7 activity after 48–72 h of treatment - No induction of caspase activation at 30 µmol/L - accumulation of cells at G2 phase ↓ mitotic index - QUE (100 µmol/L) did not cause hippocampal organotypic culture damage - the neuronal injury was reduced to 35% after QUE treatment | ↓ Cell proliferation - Necrotic and apoptotic cell death - Arrest at the G2 checkpoint of the cell cycle - Neuroprotective effects | Braganhol et al., 2006 [39] |
| Que | A172 | ↓ Cell viability - No ROS generation - down-regulation of ERK and Akt ↓ mitochondrial membrane potential ↑ caspase 3-activity ↓ survivin expression | - Apoptosis induced by QUE through caspase-dependent mechanisms - Involvement of Akt, ERK and survivin downregulation - ROS generation is not the cause of apoptosis | Kim et al., 2008 [40] |
| Que | U373MG | ↑ cells in the sub-G1 phase ↓mitochondrial membrane potential ↑ Caspase 3/7/9 activation and activity - Activation of JNK ↑ p53 expression ↑ AVO | - QUE induces apoptosis through increase p53 expression - QUE induces protective autophagy - QUE combined with an autophagy inhibitor can be an effective anticancer agent | Kim et al., 2013 [31] |
| Que | U87 | ↓ PLD1 expression - inhibition of PLD1 activity by QUE - inhibition of NFκB transactivation ↓ MMP-2 activity | - Through PLD-1 suppression by QUE, glioma cell proliferation is achieved - QUE suppressed PLD1 expression through inhibition of NFkB transactivation - QUE inhibited tumor cell invasion by reducing MMP-2 activity. | Park et al., 2011 [42] |
| Que | U138MG | ↑ concentrations of adenine nucleotide/ nucleoside in the extracellular medium - ecto-5′-nucleotidase/CD73 converts AMP to adenosine - QUE inhibits ecto-5′-nucleotidase/CD73 - U138MG proliferation was inhibited by an ecto-5′-nucleotidase/CD73 inhibitor (APCP) | - Inhibition of ecto-5′-NT/CD73 may slow the tumor proliferation by reducing the synthesis of extracellular adenosine. | Braganhol et al., 2007 [41] |
3. Bioavailability and Metabolism of Que
4. Nano-Delivery Systems Entrapping Que—New Approaches for Bioavailability Improvement
| Formulation | Cell Line/Animal Model | Main Findings on Que Bioavailability/Brain Targeting | References |
|---|---|---|---|
| Que-loaded with plasma exosomes | In vivo: AD Rats; I.V admin 12 mg/kg | (a) Improvement of pharmacokinetic parameters; (b) Que concentration increased by 2.5-fold in cerebrum and 1.5-fold in cerebellum (c) Biocompatible and safe | Qi et al., 2020 [123] |
| RVG29-Functionalized Lipid Nanoparticles for Que | In vitro: hCMEC/D3 cell line | (a) Increase 1.5-fold in the permeability across the in vitro model of the BBB; (b) With the highest concentration the cytotoxicity is lower than 15% | Pinheiro et al., in 2020 [89] |
| Transferrin- functionalized Lipid Nanoparticles for Que | In vitro: hCMEC/D3 cell line | (a) Increment in the permeability across the in vitro model of blood-brain-barrier; (b) No significant cytotoxicity in tested concentrations | Pinheiro et al., in 2020 [97] |
| Que-SPION | In vivo: healthy rat models | (a) Que concentration in plasma and brain are significantly higher (b) SPION does not cross BBB | Najafabadi et al., 2018 & Amanzadeh et al., 2019 [99,101] |
| Que SLN | In vivo: Wistar rat model | (a) Increased antioxidant activity (b) Enhanced brain delivery | Dhawan et al., 2011 [107] |
| Que NLC | Ex vivo: nasal tissue from the sheep; in vivo: Wistar rat models | Que NLC revealed sustained delivery of drug and significant brain distribution | Patil et al., 2017 [124] |
| (a) Que loaded nano lipidic carriers (NLC) composition of phospholipids, vitamin E acetate and glyceryl (b) Que loaded solid lipid nanoparticles (SLN) | In vitro: Caco-2 cell line—cellular uptake In vivo: Rat animal model—pharmacokinetic, biodistribution and brain delivery studies. | (a) Increased Que intestinal permeability; increased antioxidant activity (b) Enhanced bioavailability and brain penetration | Kumar et al., 2016 [110] |
| Que PBCA NP and Que PBCA NP coated with polysorbate-80 | In vitro: (a) Drug stability (b) Release studies In vivo: Wistar rat model (a) pharmacokinetic studies (b) biodistribution | (a) Sustained release of Que (b) Relative bioavailability increased by 2.38- and 4.93-fold; (c) Increased concentration of Que in the brain with the Que PBCA NP coated with P-80 | Bagad et al., 2015 [115] |
| SPION@APTES@FAPEG@ CQ; SPION functionalized with APTES ((3-Aminopropyl) triethoxysilane) and polyethylene glycol (PEG); conjugated with folic acid and loaded with Carboxylated Que | In vitro: Cytotoxicity- L929 cell line and U87 cells with MTT assay | SPION@APTES@FA- PEG did not show a cytotoxic effect; good ability for targeting cancer cells | Akal et al., 2016 [102] |
| Que-loaded freeze-dried nanomicelles | In vitro: C6 glioma cells and In vivo: BALB/c nude mice | Que-loaded freeze-dried nanomicelles had efficient sustained release profile, increased intracellular uptake with low cytotoxicity, efficient penetration of BBB, and powerful cytotoxicity on C6 glioma cells. | Wang et al., 2016 [116] |
| Que liposomes | In vitro: Rat animal model | Intranasal Que liposomes enhance brain delivery | Priprem et al., 2008 [118] |
| P80-Que@Se Que-loaded selenium nano-particles coated with acacia and P80; | In vitro: (a) cytotoxicity studies: PC12 cell line; (b) Cell Counting Kit (CCK) (c) DPPH radical scavenging assay –antioxidant activity | (a) Low cytotoxicity (b) Protection from H2O2-induced cell death (c) High antioxidant activity (d) Enhanced brain delivery | Yi et al., 2020 [103] |
| Nano-emulsion—nose-to-brain delivery Que and curcumin (2:1) | In vitro: (a) Cytotoxic effect—human glioblastoma U373-MG cell (b) Nose to Brain delivery -nasal mucosa of sheep In vivo: (c) Allograft mouse tumor model | (a) Significant inhibition of the human glioblastoma U373MG cell growth (b) Effective CNS targeting (c) Higher anticancer activity as compared to doxorubicin | Mahajan et al.,2021 [119] |
5. Anticancer Activity of Que Nano-Delivery Systems: Evidence from In Vitro and In Vivo Studies
6. Synergism of Que with Other Therapeutic Agents
7. Que in Glioblastoma Multiforme—Case Study
8. Safety Aspects of Que Use
9. Conclusions and Remarks
10. Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Conflicts of Interest
References
- Qi, X.; Jha, S.K.; Jha, N.K.; Dewanjee, S.; Dey, A.; Deka, R.; Pritam, P.; Ramgopal, K.; Liu, W.; Hou, K. Antioxidants in brain tumors: Current therapeutic significance and future prospects. Mol. Cancer 2022, 21, 204. [Google Scholar] [CrossRef]
- Shah, V.; Kochar, P. Brain Cancer: Implication to Disease, Therapeutic Strategies and Tumor Targeted Drug Delivery Approaches. Recent Pat. Anticancer Drug Discov. 2018, 13, 70–85. [Google Scholar] [CrossRef]
- Udaka, Y.T.; Packer, R.J. Pediatric Brain Tumors. Neurol. Clin. 2018, 36, 533–556. [Google Scholar] [CrossRef] [PubMed]
- Ostrom, Q.T.; Francis, S.S.; Barnholtz-Sloan, J.S. Epidemiology of Brain and Other CNS Tumors. Curr. Neurol. Neurosci. Rep. 2021, 21, 68. [Google Scholar] [CrossRef] [PubMed]
- Melcher, V.; Kerl, K. The growing relevance of immunoregulation in pediatric brain tumors. Cancers 2021, 13, 5601. [Google Scholar] [CrossRef] [PubMed]
- Blionas, A.; Giakoumettis, D.; Klonou, A.; Neromyliotis, E.; Karydakis, P.; Themistocleous, M.S. Paediatric gliomas: Diagnosis, molecular biology and management. Ann. Transl. Med. 2018, 6, 251. [Google Scholar] [CrossRef]
- Stucklin, A.S.G.; Ramaswamy, V.; Daniels, C.; Taylor, M.D. Review of molecular classification and treatment implications of pediatric brain tumors. Curr. Opin. Pediatr. 2018, 30, 3–9. [Google Scholar] [CrossRef]
- Minturn, J.E.; Fisher, M.J. Gliomas in children. Curr. Treat. Options Neurol. 2013, 15, 316–327. [Google Scholar] [CrossRef]
- Tan, A.C.; Ashley, D.M.; López, G.Y.; Malinzak, M.; Friedman, H.S.; Khasraw, M. Management of glioblastoma: State of the art and future directions. CA Cancer J. Clin. 2020, 70, 299–312. [Google Scholar] [CrossRef]
- Aldape, K.; Brindle, K.M.; Chesler, L.; Chopra, R.; Gajjar, A.; Gilbert, M.R.; Gottardo, N.; Gutmann, D.H.; Hargrave, D.; Holland, E.C.; et al. Challenges to curing primary brain tumours. Nat. Rev. Clin. Oncol. 2019, 16, 509–520. [Google Scholar] [CrossRef]
- Laquintana, V.; Trapani, A.; Denora, N.; Wang, F.; Gallo, J.; Trapani, G. New strategies to deliver anticancer drugs to brain tumors. Expert Opin. Drug Deliv. 2009, 6, 1017–1032. [Google Scholar] [CrossRef] [PubMed]
- Atanasov, A.G.; Zotchev, S.B.; Dirsch, V.M.; Orhan, I.E.; Banach, M.; Rollinger, J.M.; Barreca, D.; Weckwerth, W.; Bauer, R.; Bayer, E.A.; et al. Natural products in drug discovery: Advances and opportunities. Nat. Rev. Drug Discov. 2021, 20, 200–216. [Google Scholar] [CrossRef] [PubMed]
- Sang, D.P.; Li, R.J.; Lan, Q. Quercetin sensitizes human glioblastoma cells to temozolomide in vitro via inhibition of Hsp27. Acta Pharmacol. Sin. 2014, 35, 832–838. [Google Scholar] [CrossRef]
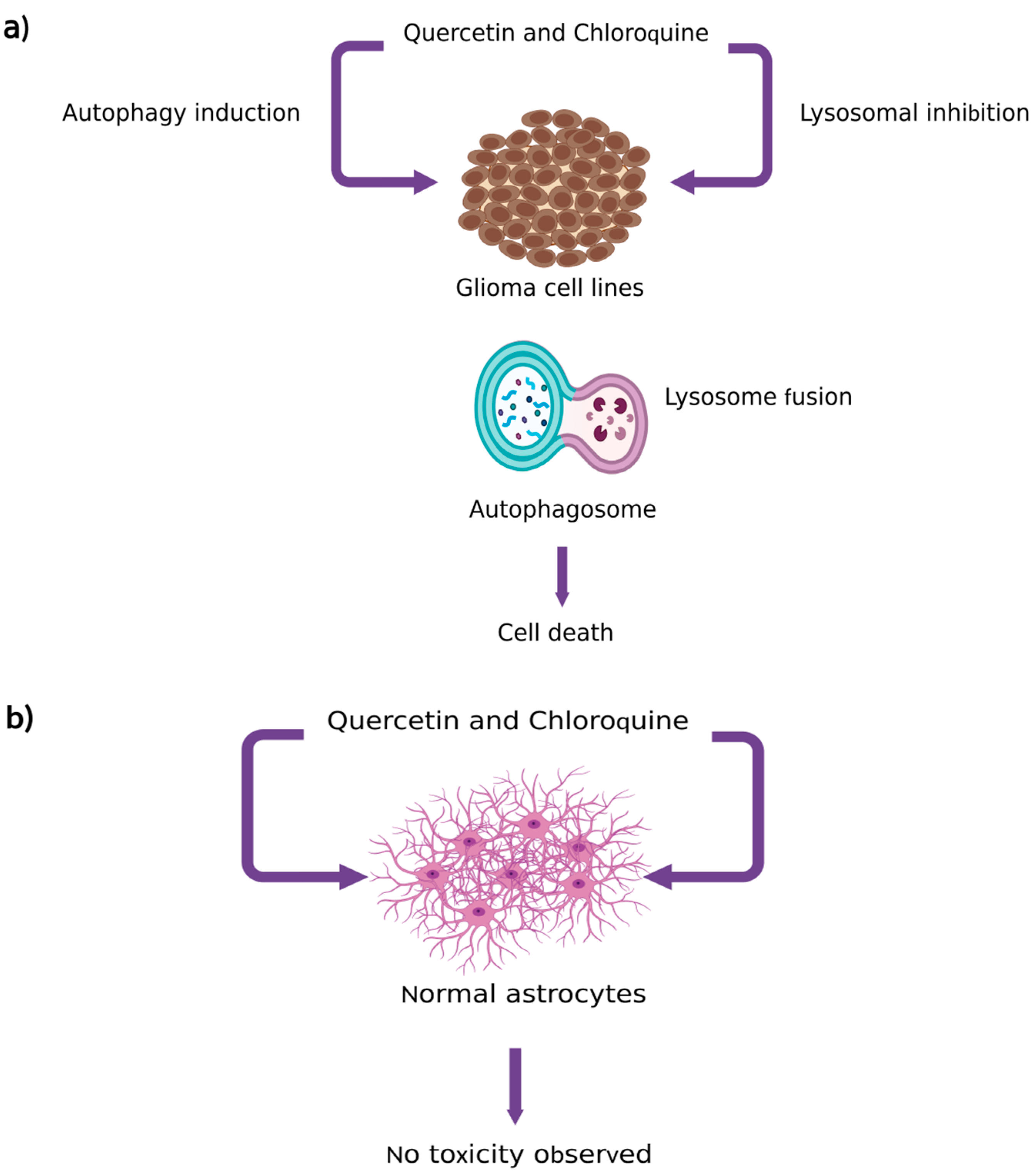
- Jang, E.; Kim, I.Y.; Kim, H.; Lee, D.M.; Seo, D.Y.; Lee, J.A.; Choi, K.S.; Kim, E. Quercetin and chloroquine synergistically kill glioma cells by inducing organelle stress and disrupting Ca2+ homeostasis. Biochem. Pharmacol. 2020, 178, 114098. [Google Scholar] [CrossRef] [PubMed]
- Saeed, N.; Khan, M.R.; Shabbir, M. Antioxidant activity, total phenolic and total flavonoid contents of whole plant extracts Torilis leptophylla L. BMC Complement. Altern. Med. 2012, 12, 221. [Google Scholar] [CrossRef] [PubMed]
- Maleki, S.J.; Crespo, J.F.; Cabanillas, B. Anti-inflammatory effects of flavonoids. Food Chem. 2019, 299, 125124. [Google Scholar] [CrossRef]
- Singh, P.; Arif, Y.; Bajguz, A.; Hayat, S. The role of quercetin in plants. Plant Physiol. Biochem. 2021, 166, 10–19. [Google Scholar] [CrossRef]
- Kumar, S.; Pandey, A.K. Chemistry and Biological Activities of Flavonoids: An Overview. Sci. World J. 2013, 2013, 162750. [Google Scholar] [CrossRef]
- Lakhanpal, P.; Rai, D.K. Quercetin: A Versatile Flavonoid. Internet J. Med. Updat. EJOURNAL 2007, 2, 22–37. [Google Scholar] [CrossRef]
- Babaei, F.; Mirzababaei, M.; Nassiri-Asl, M. Quercetin in Food: Possible Mechanisms of Its Effect on Memory. J. Food Sci. 2018, 83, 2280–2287. [Google Scholar] [CrossRef]
- Azeem, M.; Hanif, M.; Mahmood, K.; Ameer, N.; Chughtai, F.R.S.; Abid, U. An insight into anticancer, antioxidant, antimicrobial, antidiabetic and anti-inflammatory effects of quercetin: A review. Polym. Bull. 2022, 80, 241–262. [Google Scholar] [CrossRef]
- Anand David, A.V.; Arulmoli, R.; Parasuraman, S. Overviews of biological importance of quercetin: A bioactive flavonoid. Pharmacogn. Rev. 2016, 10, 84–89. [Google Scholar] [CrossRef] [PubMed]
- Almatroodi, S.A.; Alsahli, M.A.; Aljohani, A.S.M.; Alhumaydhi, F.A.; Babiker, A.Y.; Khan, A.A.; Rahmani, A.H. Potential Therapeutic Targets of Resveratrol, a Plant Polyphenol, and Its Role in the Therapy of Various Types of Cancer. Molecules 2022, 27, 2665. [Google Scholar] [CrossRef] [PubMed]
- Pan, H.C.; Jiang, Q.; Yu, Y.; Mei, J.P.; Cui, Y.K.; Zhao, W.J. Quercetin promotes cell apoptosis and inhibits the expression of MMP-9 and fibronectin via the AKT and ERK signalling pathways in human glioma cells. Neurochem. Int. 2015, 80, 60–71. [Google Scholar] [CrossRef]
- Hu, J.U.N.; Wang, J.; Wang, G.; Yao, Z.; Dang, X. Pharmacokinetics and antitumor efficacy of DSPE-PEG2000 polymeric liposomes loaded with quercetin and temozolomide: Analysis of their effectiveness in enhancing the chemosensitization of drug-resistant glioma cells. Int. J. Mol. Med. 2016, 37, 690–702. [Google Scholar] [CrossRef]
- Menon, P.K.; Sharma, A.; Lafuente, J.V.; Muresanu, D.F.; Aguilar, Z.P.; Wang, Y.A.; Patnaik, R.; Mössler, H.; Sharma, H.S. Intravenous Administration of Functionalized Magnetic Iron Oxide Nanoparticles Does Not Induce CNS Injury in the Rat: Influence of Spinal Cord Trauma and Cerebrolysin Treatment. Int. Rev. Neurobiol. 2017, 137, 47–63. [Google Scholar] [CrossRef] [PubMed]
- Kusaczuk, M.; Krętowski, R.; Naumowicz, M.; Stypułkowska, A.; Cechowska-Pasko, M. A Preliminary Study of the Effect of Quercetin on Cytotoxicity, Apoptosis, and Stress Responses in Glioblastoma Cell Lines. Int. J. Mol. Sci. 2022, 23, 1345. [Google Scholar] [CrossRef]
- Lou, M.; Zhang, L.; Ji, P.; Feng, F.; Liu, J.; Yang, C.; Li, B.; Wang, L. Quercetin nanoparticles induced autophagy and apoptosis through AKT / ERK / Caspase-3 signaling pathway in human neuroglioma cells: In vitro and in vivo. Biomed. Pharmacother. 2016, 84, 1–9. [Google Scholar] [CrossRef]
- Tavana, E.; Mollazadeh, H.; Mohtashami, E.; Modaresi, S.M.S.; Hosseini, A.; Sabri, H.; Soltani, A.; Javid, H.; Afshari, A.R.; Sahebkar, A. Quercetin: A promising phytochemical for the treatment of glioblastoma multiforme. BioFactors 2020, 46, 356–366. [Google Scholar] [CrossRef]
- Jakubowicz-Gil, J.; Langner, E.; Bądziul, D.; Wertel, I.; Rzeski, W. Apoptosis induction in human glioblastoma multiforme T98G cells upon temozolomide and quercetin treatment. Tumor Biol. 2013, 34, 2367–2378. [Google Scholar] [CrossRef]
- Kim, H.; Moon, J.Y.; Ahn, K.S.; Cho, S.K. Quercetin induces mitochondrial mediated apoptosis and protective autophagy in human glioblastoma U373MG Cells. Oxidative Med. Cell. Longev. 2013, 2013, 596496. [Google Scholar] [CrossRef] [PubMed]
- Shi, H.; Li, X.Y.; Chen, Y.; Zhang, X.; Wu, Y.; Wang, Z.X.; Chen, P.H.; Dai, H.Q.; Feng, J.; Chatterjee, S.; et al. Quercetin Induces Apoptosis via Downregulation of Vascular Endothelial Growth Factor/Akt Signaling Pathway in Acute Myeloid Leukemia Cells. Front. Pharmacol. 2020, 11, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Jakubowicz-Gil, J.; Langner, E.; Wertel, I.; Piersiak, T.; Rzeski, W. Temozolomide, quercetin and cell death in the MOGGCCM astrocytoma cell line. Chem. Biol. Interact. 2010, 188, 190–203. [Google Scholar] [CrossRef]
- Tamtaji, O.R.; Razavi, Z.S.; Razzaghi, N.; Aschner, M.; Barati, E.; Mirzaei, H. Quercetin and Glioma: Which signaling pathways are involved? Curr. Mol. Pharmacol. 2022, 15, 962–968. [Google Scholar] [CrossRef] [PubMed]
- Pistritto, G.; Trisciuoglio, D.; Ceci, C.; Garufi, A.; D’Orazi, G. Apoptosis as anticancer mechanism: Function and dysfunction of its modulators and targeted therapeutic strategies. Aging 2016, 8, 603–619. [Google Scholar] [CrossRef] [PubMed]
- Goldar, S.; Khaniani, M.S.; Derakhshan, S.M.; Baradaran, B. Molecular mechanisms of apoptosis and roles in cancer development and treatment. Asian Pac. J. Cancer Prev. 2015, 16, 2129–2144. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Wang, J.; Luo, J.; Wang, L.; Chen, X.; Zhang, L.; Jiang, S. PEG2000-DPSE-coated quercetin nanoparticles remarkably enhanced anticancer effects through induced programed cell death on C6 glioma cells. J. Biomed. Mater. Res. Part A 2013, 101, 3076–3085. [Google Scholar] [CrossRef]
- Robaszkiewicz, A.; Balcerczyk, A.; Bartosz, G. Antioxidative and prooxidative effects of quercetin on A549 cells. Cell Biol. Int. 2007, 31, 1245–1250. [Google Scholar] [CrossRef]
- Braganhol, E.; Zamin, L.L.; Delgado Canedo, A.; Horn, F.; Tamajusuku, A.S.K.; Wink, M.R.; Salbego, C.; Battastini, A.M.O. Antiproliferative effect of quercetin in the human U138MG glioma cell line. Anticancer Drugs 2006, 17, 663–671. [Google Scholar] [CrossRef]
- Kim, E.J.; Choi, C.H.; Park, J.Y.; Kang, S.K.; Kim, Y.K. Underlying mechanism of quercetin-induced cell death in human glioma cells. Neurochem. Res. 2008, 33, 971–979. [Google Scholar] [CrossRef]
- Braganhol, E.; Tamajusuku, A.S.K.; Bernardi, A.; Wink, M.R.; Battastini, A.M.O. Ecto-5′-nucleotidase/CD73 inhibition by quercetin in the human U138MG glioma cell line. Biochim. Biophys. Acta Gen. Subj. 2007, 1770, 1352–1359. [Google Scholar] [CrossRef]
- Park, M.H.; Min, D.S. Quercetin-induced downregulation of phospholipase D1 inhibits proliferation and invasion in U87 glioma cells. Biochem. Biophys. Res. Commun. 2011, 412, 710–715. [Google Scholar] [CrossRef] [PubMed]
- Currie, G.M. Pharmacology, part 2: Introduction to pharmacokinetics. J. Nucl. Med. Technol. 2018, 46, 221–230. [Google Scholar] [CrossRef] [PubMed]
- Toutain, P.L.; Bousquet-Mélou, A. Bioavailability and its assessment. J. Vet. Pharmacol. Ther. 2004, 27, 455–466. [Google Scholar] [CrossRef]
- Guo, Z.J.; Zhang, Y.; Tang, X.; Li, H.; Sun, Q.S. Pharmacokinetic interaction between tanshinones and polyphenolic extracts of Salvia miltinorrhiza Bunge after intravenous administration in rats. Biol. Pharm. Bull. 2008, 31, 1469–1474. [Google Scholar] [CrossRef] [PubMed]
- Krogholm, K.S.; Bredsdorff, L.; Knuthsen, P.; Haraldsdóttir, J.; Rasmussen, S.E. Relative bioavailability of the flavonoids quercetin, hesperetin and naringenin given simultaneously through diet. Eur. J. Clin. Nutr. 2010, 64, 432–435. [Google Scholar] [CrossRef]
- Hollman, P.C.H. Absorption, bioavailability, and metabolism of flavonoids. Pharm. Biol. 2004, 42, 74–83. [Google Scholar] [CrossRef]
- Williamson, G.; Kay, C.D.; Crozier, A. The Bioavailability, Transport, and Bioactivity of Dietary Flavonoids: A Review from a Historical Perspective. Compr. Rev. Food Sci. Food Saf. 2018, 17, 1054–1112. [Google Scholar] [CrossRef]
- Li, H.; Li, M.; Fu, J.; Ao, H.; Wang, W.; Wang, X. Enhancement of oral bioavailability of quercetin by metabolic inhibitory nanosuspensions compared to conventional nanosuspensions. Drug Deliv. 2021, 28, 1226–1236. [Google Scholar] [CrossRef]
- Wein, S.; Beyer, B.; Zimmermann, B.F.; Blank, R.H.; Wolffram, S. Bioavailability of Quercetin from Onion Extracts after Intraruminal Application in Cows. J. Agric. Food Chem. 2018, 66, 10188–10192. [Google Scholar] [CrossRef]
- Graefe, E.U.; Wittig, J.; Mueller, S.; Riethling, A.K.; Uehleke, B.; Drewelow, B.; Pforte, H.; Jacobasch, G.; Derendorf, H.; Veit, M. Pharmacokinetics and bioavailability of quercetin glycosides in humans. J. Clin. Pharmacol. 2001, 41, 492–499. [Google Scholar] [CrossRef] [PubMed]
- Kasikci, M.B.; Bagdatlioglu, N. Bioavailability of quercetin. Curr. Res. Nutr. Food Sci. 2016, 4, 146–151. [Google Scholar] [CrossRef]
- Almeida, A.F.; Borge, G.I.A.; Piskula, M.; Tudose, A.; Tudoreanu, L.; Valentová, K.; Williamson, G.; Santos, C.N. Bioavailability of Quercetin in Humans with a Focus on Interindividual Variation. Compr. Rev. Food Sci. Food Saf. 2018, 17, 714–731. [Google Scholar] [CrossRef]
- Carbonaro, M.; Grant, G. Absorption of quercetin and rutin in rat small intestine. Ann. Nutr. Metab. 2005, 49, 178–182. [Google Scholar] [CrossRef]
- Crespy, V.; Morand, C.; Besson, C.; Manach, C.; Demigne, C.; Remesy, C. Quercetin, but not its glycosides, is absorbed from the rat stomach. J. Agric. Food Chem. 2002, 50, 618–621. [Google Scholar] [CrossRef] [PubMed]
- Ikeno, A.; Hashimoto, K.; Shimizu, M.; Terao, J.; Kumazawa, S.; Nakayama, T. Absorption and Metabolism of Quercetin in Caco-2 Cells. Food Sci. Technol. Res. 1999, 5, 90–92. [Google Scholar] [CrossRef]
- Lodi, F.; Jimenez, R.; Moreno, L.; Kroon, P.A.; Needs, P.W.; Hughes, D.A.; Santos-Buelga, C.; Gonzalez-Paramas, A.; Cogolludo, A.; Lopez-Sepulveda, R.; et al. Glucuronidated and sulfated metabolites of the flavonoid quercetin prevent endothelial dysfunction but lack direct vasorelaxant effects in rat aorta. Atherosclerosis 2009, 204, 34–39. [Google Scholar] [CrossRef]
- Boots, A.W.; Haenen, G.R.M.M.; Bast, A. Health effects of quercetin: From antioxidant to nutraceutical. Eur. J. Pharmacol. 2008, 585, 325–337. [Google Scholar] [CrossRef]
- Graf, B.A.; Mullen, W.; Caldwell, S.T.; Hartley, R.C.; Duthie, G.G.; Lean, M.E.J.; Crozier, A.; Edwards, C.A. Disposition and metabolism of [2-14C]quercetin-4′ glucoside in rats. Drug Metab. Dispos. 2005, 33, 1036–1043. [Google Scholar] [CrossRef]
- Vrba, J.; Kren, V.; Vacek, J.; Papouskova, B.; Ulrichova, J. Quercetin, quercetin glycosides and taxifolin differ in their ability to induce AhR activation and cyp1a1 expression in HepG2 cells. Phyther. Res. 2012, 26, 1746–1752. [Google Scholar] [CrossRef]
- Williamson, G.; Manach, C. Bioavailability and bioefficacy of polyphenols in humans. II. Review of 93 intervention studies. Am. J. Clin. Nutr. 2015, 81, 243S–255S. [Google Scholar] [CrossRef]
- Dabeek, W.M.; Marra, M.V. Dietary quercetin and kaempferol: Bioavailability in humans. Nutrients 2019, 11, 3–19. [Google Scholar] [CrossRef]
- Araújo, K.C.F.; Eula, E.M.; Pazini, F.; Valadares, M.C.; De Oliveira, V. Bioconversion of quercetin and rutin and the cytotoxicity activities of the transformed products. Food Chem. Toxicol. 2013, 51, 93–96. [Google Scholar] [CrossRef]
- Beekmann, K.; Actis-Goretta, L.; Van Bladeren, P.J.; Dionisi, F.; Destaillats, F.; Rietjens, I.M.C.M. A state-of-the-art overview of the effect of metabolic conjugation on the biological activity of flavonoids. Food Funct. 2012, 3, 1008–1018. [Google Scholar] [CrossRef] [PubMed]
- Lodi, F.; Jiménez, R.; Menendez, C.; Needs, P.W.; Duarte, J.; Perez-Vizcaino, F. Glucuronidated metabolites of the flavonoid quercetin do not auto-oxidise, do not generate free radicals and do not decrease nitric oxide bioavailability. Planta Med. 2008, 74, 741–746. [Google Scholar] [CrossRef] [PubMed]
- Williamson, G.; Barron, D.; Shimoi, K.; Terao, J. In vitro biological properties of flavonoid conjugates found in vivo. Free. Radic. Res. 2005, 39, 457–469. [Google Scholar] [CrossRef]
- Walgren, R.A.; Lin, J.T.; Kinne, R.K.H.; Walle, T. Cellular uptake of dietary flavonoid quercetin 4’-β-glucoside by sodium-dependent glucose transporter SGLT11. J. Pharmacol. Exp. Ther. 2000, 294, 837–843. [Google Scholar]
- Wróbel-Biedrawa, D.; Grabowska, K.; Galanty, A.; Sobolewska, D.; Podolak, I. A Flavonoid on the Brain: Quercetin as a Potential Therapeutic Agent in Central Nervous System Disorders. Life 2022, 12, 591. [Google Scholar] [CrossRef] [PubMed]
- Manach, C.; Regerat, F.; Texier, O.; Agullo, G.; Demigne, C.; Remesy, C. Bioavailability, metabolism and physiological impact of 4-oxo-flavonoids. Nutr. Res. 1996, 16, 517–544. [Google Scholar] [CrossRef]
- O’Leary, K.A.; Day, A.J.; Needs, P.W.; Mellon, F.A.; O’Brien, N.M.; Williamson, G. Metabolism of quercetin-7- and quercetin-3-glucuronides by an in vitro hepatic model: The role of human β-glucuronidase, sulfotransferase, catechol-O-methyltransferase and multi-resistant protein 2 (MRP2) in flavonoid metabolism. Biochem. Pharmacol. 2003, 65, 479–491. [Google Scholar] [CrossRef]
- Marín, L.; Miguélez, E.M.; Villar, C.J.; Lombó, F. Bioavailability of dietary polyphenols and gut microbiota metabolism: Antimicrobial properties. Biomed. Res. Int. 2015, 2015, 905215. [Google Scholar] [CrossRef]
- Khaled, K.A.; El-Sayed, Y.M.; Al-Hadiya, B.M. Disposition of the flavonoid quercetin in rats after single intravenous and oral doses. Drug Dev. Ind. Pharm. 2003, 29, 397–403. [Google Scholar] [CrossRef] [PubMed]
- Pool, H.; Mendoza, S.; Xiao, H.; McClements, D.J. Encapsulation and release of hydrophobic bioactive components in nanoemulsion-based delivery systems: Impact of physical form on quercetin bioaccessibility. Food Funct. 2013, 4, 162–174. [Google Scholar] [CrossRef]
- Simioni, C.; Zauli, G.; Martelli, A.M.; Vitale, M.; Sacchetti, G.; Gonelli, A.; Neri, L.M. Oxidative stress: Role of physical exercise and antioxidant nutraceuticals in adulthood and aging. Oncotarget 2018, 9, 17181–17198. [Google Scholar] [CrossRef]
- Chen, X.; Yin, O.Q.P.; Zuo, Z.; Chow, M.S.S. Pharmacokinetics and modeling of quercetin and metabolites. Pharm. Res. 2005, 22, 892–901. [Google Scholar] [CrossRef] [PubMed]
- Justino, G.C.; Santos, M.R.; Canário, S.; Borges, C.; Florêncio, M.H.; Mira, L. Plasma quercetin metabolites: Structure-antioxidant activity relationships. Arch. Biochem. Biophys. 2004, 432, 109–121. [Google Scholar] [CrossRef]
- Ishisaka, A.; Ichikawa, S.; Sakakibara, H.; Piskula, M.K.; Nakamura, T.; Kato, Y.; Ito, M.; Miyamoto, K.I.; Tsuji, A.; Kawai, Y.; et al. Accumulation of orally administered quercetin in brain tissue and its antioxidative effects in rats. Free Radic. Biol. Med. 2011, 51, 1329–1336. [Google Scholar] [CrossRef] [PubMed]
- Gelperina, S.; Kisich, K.; Iseman, M.D.; Heifets, L. Pulmonary Perspective The Potential Advantages of Nanoparticle Drug Delivery Systems in Chemotherapy of Tuberculosis. Am. J. Respir. Crit. Care Med. 2005, 172, 1487–1490. [Google Scholar] [CrossRef]
- Reddy, S.; Tatiparti, K.; Sau, S.; Iyer, A.K. Recent advances in nano delivery systems for blood-brain barrier (BBB) penetration and targeting of brain tumors. Drug Discov. Today 2021, 26, 1944–1952. [Google Scholar] [CrossRef] [PubMed]
- Sommonte, F.; Arduino, I.; Racaniello, G.F.; Lopalco, A.; Lopedota, A.A.; Denora, N. The Complexity of the Blood-Brain Barrier and the Concept of Age-Related Brain Targeting: Challenges and Potential of Novel Solid Lipid-Based Formulations. J. Pharm. Sci. 2022, 111, 577–592. [Google Scholar] [CrossRef]
- Bhatti, J.S.; Vijayvergiya, R.; Singh, B.; Bhatti, G.K. Exosome Nanocarriers, 1st ed.; Grumezescu, A.M., Ed.; Elsevier Inc.: Amsterdam, The Netherlands, 2019; ISBN 9780128162002. [Google Scholar]
- Heidarzadeh, M.; Gürsoy-Özdemir, Y.; Kaya, M.; Eslami Abriz, A.; Zarebkohan, A.; Rahbarghazi, R.; Sokullu, E. Exosomal delivery of therapeutic modulators through the blood–brain barrier; promise and pitfalls. Cell Biosci. 2021, 11, 142. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zhang, Y.; Cai, G.; Li, Q. Exosomes as actively targeted nanocarriers for cancer therapy. Int. J. Nanomed. 2020, 15, 4257–4273. [Google Scholar] [CrossRef] [PubMed]
- Arduino, I.; Liu, Z.; Rahikkala, A.; Figueiredo, P.; Correia, A.; Cutrignelli, A.; Denora, N.; Santos, H.A. Preparation of cetyl palmitate-based PEGylated solid lipid nanoparticles by microfluidic technique. Acta Biomater. 2021, 121, 566–578. [Google Scholar] [CrossRef]
- Arduino, I.; Depalo, N.; Re, F.; Dal Magro, R.; Panniello, A.; Margiotta, N.; Fanizza, E.; Lopalco, A.; Laquintana, V.; Cutrignelli, A.; et al. PEGylated solid lipid nanoparticles for brain delivery of lipophilic kiteplatin Pt(IV) prodrugs: An in vitro study. Int. J. Pharm. 2020, 583, 119351. [Google Scholar] [CrossRef] [PubMed]
- Arduino, I.; Liu, Z.; Iacobazzi, R.M.; Lopedota, A.A.; Lopalco, A.; Cutrignelli, A.; Laquintana, V.; Porcelli, L.; Azzariti, A.; Franco, M.; et al. Microfluidic preparation and in vitro evaluation of iRGD-functionalized solid lipid nanoparticles for targeted delivery of paclitaxel to tumor cells. Int. J. Pharm. 2021, 610, 121246. [Google Scholar] [CrossRef]
- Arduino, I.; Iacobazzi, R.M.; Riganti, C.; Lopedota, A.A.; Perrone, M.G.; Lopalco, A.; Cutrignelli, A.; Cantore, M.; Laquintana, V.; Franco, M.; et al. Induced expression of P-gp and BCRP transporters on brain endothelial cells using transferrin functionalized nanostructured lipid carriers: A first step of a potential strategy for the treatment of Alzheimer’s disease. Int. J. Pharm. 2020, 591, 120011. [Google Scholar] [CrossRef]
- Iacobazzi, R.M.; Arduino, I.; Di Fonte, R.; Lopedota, A.A.; Serratì, S.; Racaniello, G.; Bruno, V.; Laquintana, V.; Lee, B.C.; Silvestris, N.; et al. Microfluidic-assisted preparation of targeted ph-responsive polymeric micelles improves gemcitabine effectiveness in pdac: In vitro insights. Cancers 2022, 14, 5. [Google Scholar] [CrossRef]
- Pinheiro, R.G.R.; Granja, A.; Loureiro, J.A.; Pereira, M.C.; Pinheiro, M.; Neves, A.R.; Reis, S. RVG29-Functionalized Lipid Nanoparticles for Quercetin Brain Delivery and Alzheimer’s Disease. Pharm. Res. 2020, 37, 139. [Google Scholar] [CrossRef]
- Soni, S.; Ruhela, R.K.; Medhi, B. Nanomedicine in central nervous system (CNS) disorders: A present and future prospective. Adv. Pharm. Bull. 2016, 6, 319–335. [Google Scholar] [CrossRef]
- Chowdhury, A.; Kunjiappan, S.; Panneerselvam, T.; Somasundaram, B.; Bhattacharjee, C. Nanotechnology and nanocarrier-based approaches on treatment of degenerative diseases. Int. Nano Lett. 2017, 7, 91–122. [Google Scholar] [CrossRef]
- Le Novère, N.; Corringer, P.J.; Changeux, J.P. The diversity of subunit composition in nAChRs: Evolutionary origins, physiologic and pharmacologic consequences. J. Neurobiol. 2002, 53, 447–456. [Google Scholar] [CrossRef]
- Gotti, C.; Clementi, F.; Fornari, A.; Gaimarri, A.; Guiducci, S.; Manfredi, I.; Moretti, M.; Pedrazzi, P.; Pucci, L.; Zoli, M. Structural and functional diversity of native brain neuronal nicotinic receptors. Biochem. Pharmacol. 2009, 78, 703–711. [Google Scholar] [CrossRef]
- Hawkins, B.T.; Egleton, R.D.; Davis, T.P. Modulation of cerebral microvascular permeability by endothelial nicotinic acetylcholine receptors. Am. J. Physiol. Heart Circ. Physiol. 2005, 289, 212–219. [Google Scholar] [CrossRef] [PubMed]
- Lafon, M. Rabies virus receptors. J. Neurovirology 2005, 11, 82–87. [Google Scholar] [CrossRef]
- Liu, Y.; Huang, R.; Han, L.; Ke, W.; Shao, K.; Ye, L.; Lou, J.; Jiang, C. Brain-targeting gene delivery and cellular internalization mechanisms for modified rabies virus glycoprotein RVG29 nanoparticles. Biomaterials 2009, 30, 4195–4202. [Google Scholar] [CrossRef]
- Pinheiro, R.G.R.; Granja, A.; Loureiro, J.A.; Pereira, M.C.; Pinheiro, M.; Neves, A.R.; Reis, S. Quercetin lipid nanoparticles functionalized with transferrin for Alzheimer’s disease. Eur. J. Pharm. Sci. 2020, 148, 105314. [Google Scholar] [CrossRef] [PubMed]
- Mahmoudi, M.; Sant, S.; Wang, B.; Laurent, S.; Sen, T. Superparamagnetic iron oxide nanoparticles (SPIONs): Development, surface modification and applications in chemotherapy. Adv. Drug Deliv. Rev. 2011, 63, 24–46. [Google Scholar] [CrossRef] [PubMed]
- Enteshari Najafabadi, R.; Kazemipour, N.; Esmaeili, A.; Beheshti, S.; Nazifi, S. Using superparamagnetic iron oxide nanoparticles to enhance bioavailability of quercetin in the intact rat brain. BMC Pharmacol. Toxicol. 2018, 19, 59. [Google Scholar] [CrossRef]
- Huang, Y.; Zhang, B.; Xie, S.; Yang, B.; Xu, Q.; Tan, J. Superparamagnetic Iron Oxide Nanoparticles Modified with Tween 80 Pass through the Intact Blood-Brain Barrier in Rats under Magnetic Field. ACS Appl. Mater. Interfaces 2016, 8, 11336–11341. [Google Scholar] [CrossRef]
- Amanzadeh, E.; Esmaeili, A.; Abadi, R.E.N.; Kazemipour, N.; Pahlevanneshan, Z.; Beheshti, S. Quercetin conjugated with superparamagnetic iron oxide nanoparticles improves learning and memory better than free quercetin via interacting with proteins involved in LTP. Sci. Rep. 2019, 9, 6876. [Google Scholar] [CrossRef]
- Akal, Z.Ü.; Alpsoy, L.; Baykal, A. Superparamagnetic iron oxide conjugated with folic acid and carboxylated quercetin for chemotherapy applications. Ceram. Int. 2016, 42, 9065–9072. [Google Scholar] [CrossRef]
- Qi, Y.; Yi, P.; He, T.; Song, X.; Liu, Y.; Li, Q.; Zheng, J.; Song, R.; Liu, C.; Zhang, Z.; et al. Quercetin-loaded selenium nanoparticles inhibit amyloid-β aggregation and exhibit antioxidant activity. Colloids Surf. A Physicochem. Eng. Asp. 2020, 602, 125058. [Google Scholar] [CrossRef]
- Hariharan, S.; Dharmaraj, S. Selenium and selenoproteins: It’s role in regulation of inflammation. Inflammopharmacology 2020, 28, 667. [Google Scholar] [CrossRef] [PubMed]
- Ravichandran, V.; Lee, M.; Cao, T.G.N.; Shim, M.S. Polysorbate-based drug formulations for brain-targeted drug delivery and anticancer therapy. Appl. Sci. 2021, 11, 9336. [Google Scholar] [CrossRef]
- Werawatganone, P.; Muangsiri, W.; Chaivanon, T.; Kungwanpongpun, P.; Rattanawong, P. Solubilization of fluocinolone acetonide by cosolvents and surfactants for buccal solution preparation. Thai J. Pharm. Sci. 2018, 42, 84–89. [Google Scholar]
- Dhawan, S.; Kapil, R.; Singh, B. Formulation development and systematic optimization of solid lipid nanoparticles of quercetin for improved brain delivery. J. Pharm. Pharmacol. 2011, 63, 342–351. [Google Scholar] [CrossRef] [PubMed]
- Trevino, J.T.; Quispe, R.C.; Khan, F.; Novak, V. Non-Invasive Strategies for Nose-to-Brain Drug Delivery. J. Clin. Trials 2020, 10, 439. [Google Scholar]
- Lombardo, S.M.; Schneider, M.; Türeli, A.E.; Türeli, N.G. Key for crossing the BBB with nanoparticles: The rational design. Beilstein J. Nanotechnol. 2020, 11, 866–883. [Google Scholar] [CrossRef]
- Kumar, P.; Sharma, G.; Kumar, R.; Singh, B.; Malik, R.; Katare, O.P.; Raza, K. Promises of a biocompatible nanocarrier in improved brain delivery of quercetin: Biochemical, pharmacokinetic and biodistribution evidences. Int. J. Pharm. 2016, 515, 307–314. [Google Scholar] [CrossRef]
- Behan, N.; Birkinshaw, C.; Clarke, N. Poly n-butyl cyanoacrylate nanoparticles: A mechanistic study of polymerisation and particle formation. Biomaterials 2001, 22, 1335–1344. [Google Scholar] [CrossRef]
- Xu, Y.; Lu, S.; Liu, Q.; Hong, Y.; Xu, B.; Ping, Q.; Jin, X.; Shen, Y.; Webster, T.J.; Rao, Y. Preparation, intestinal segment stability, and mucoadhesion properties of novel thymopentin-loaded chitosan derivatives coated with poly (n-butyl) cyanoacrylate nanoparticles. Int. J. Nanomed. 2019, 14, 1659–1668. [Google Scholar] [CrossRef]
- Vauthier, C.; Labarre, D.; Ponchel, G. Design aspects of poly(alkylcyanoacrylate) nanoparticles for drug delivery. J. Drug Target. 2007, 15, 641–663. [Google Scholar] [CrossRef]
- Rempe, R.; Cramer, S.; Hüwel, S.; Galla, H.J. Transport of Poly(n-butylcyano-acrylate) nanoparticles across the blood-brain barrier in vitro and their influence on barrier integrity. Biochem. Biophys. Res. Commun. 2011, 406, 64–69. [Google Scholar] [CrossRef]
- Bagad, M.; Khan, Z.A. Poly(n-butylcyanoacrylate) nanoparticles for oral delivery of quercetin: Preparation, characterization, and pharmacokinetics and biodistribution studies in Wistar rats. Int. J. Nanomed. 2015, 10, 3921–3935. [Google Scholar] [CrossRef]
- Wang, G.; Wang, J.; Chen, X.; Du, L.; Li, F. Quercetin-loaded freeze-dried nanomicelles: Improving absorption and anti-glioma efficiency in vitro and in vivo. J. Control. Release 2016, 235, 276–290. [Google Scholar] [CrossRef] [PubMed]
- Juhairiyah, F.; de Lange, E.C.M. Understanding Drug Delivery to the Brain Using Liposome-Based Strategies: Studies that Provide Mechanistic Insights Are Essential. AAPS J. 2021, 23, 114. [Google Scholar] [CrossRef] [PubMed]
- Priprem, A.; Watanatorn, J.; Sutthiparinyanont, S.; Phachonpai, W.; Muchimapura, S. Anxiety and cognitive effects of quercetin liposomes in rats. Nanomed. Nanotechnol. Biol. Med. 2008, 4, 70–78. [Google Scholar] [CrossRef] [PubMed]
- Mahajan, H.; Patil, N. Nanoemulsion containing a synergistic combination of curcumin and quercetin for nose-To-brain delivery: In vitro and in vivo studies. Asian Pac. J. Trop. Biomed. 2021, 11, 510–518. [Google Scholar] [CrossRef]
- Karthikeyan, A.; Young, K.N.; Moniruzzaman, M.; Beyene, A.M.; Do, K.; Kalaiselvi, S.; Min, T. Curcumin and its modified formulations on inflammatory bowel disease (Ibd): The story so far and future outlook. Pharmaceutics 2021, 13, 484. [Google Scholar] [CrossRef]
- Hewlings, S.J.; Kalman, D.S. Curcumin: A review of its effects on human health. Foods 2017, 6, 92. [Google Scholar] [CrossRef]
- Mahajan, H.S.; Mahajan, M.S.; Nerkar, P.P.; Agrawal, A. Nanoemulsion-based intranasal drug delivery system of saquinavir mesylate for brain targeting. Drug Deliv. 2014, 21, 148–154. [Google Scholar] [CrossRef]
- Qi, Y.; Guo, L.; Jiang, Y.; Shi, Y.; Sui, H.; Zhao, L. Brain delivery of quercetin-loaded exosomes improved cognitive function in AD mice by inhibiting phosphorylated tau-mediated neurofibrillary tangles. Drug Deliv. 2020, 27, 745–755. [Google Scholar] [CrossRef] [PubMed]
- Patil, N.L.; Mahajan, H.S. Quercetin Loaded Nanostructured Lipid Carriers for Nose to Brain Delivery: In Vitro and In Vivo Studies. Am. J. Adv. Drug Deliv. 2018, 06, 09–20. [Google Scholar] [CrossRef]
- Giakoumettis, D.; Kritis, A.; Foroglou, N. C6 cell line: The gold standard in glioma research. Hippokratia 2018, 22, 105–112. [Google Scholar] [PubMed]
- Xie, Y.; Bergström, T.; Jiang, Y.; Johansson, P.; Dana, V.; Lindberg, N.; Segerman, A.; Wicher, G.; Niklasson, M.; Baskaran, S.; et al. The Human Glioblastoma Cell Culture Resource: Validated Cell Models Representing All Molecular Subtypes. EBioMedicine 2015, 2, 1351–1363. [Google Scholar] [CrossRef] [PubMed]
- Sibenaller, Z.; Etame, A.; Ali, M.; Barua, M.; Braun, T.; Casavant, T.; Ryken, T. Genetic characterization of commonly used glioma cell lines in the rat animal model system. Neurosurg. Focus 2005, 19, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Ersoz, M.; Erdemir, A.; Derman, S.; Arasoglu, T.; Mansuroglu, B. Quercetin-loaded nanoparticles enhance cytotoxicity and antioxidant activity on C6 glioma cells. Pharm. Dev. Technol. 2020, 25, 757–766. [Google Scholar] [CrossRef]
- Bhandarkar, S.; Prabhakar, B.; Pravin, S. Quercetin-loaded platelets as a potential targeted therapy for glioblastoma multiforme cell line U373-MG. Biotechnol. J. 2021, 16, 2100271. [Google Scholar] [CrossRef]
- Wang, G.; Wang, J.J.; Du, S.M.; Zeng, N.; Li, D.S.; Li, R.M.; Chen, J.Y.; Feng, J.B.; Yuan, S.H.; Ye, F. Effects of quercetin nanoliposomes on C6 glioma cells through induction of type III programmed cell death. Int. J. Nanomed. 2012, 7, 271–280. [Google Scholar]
- Liu, F.; Peng, B.; Li, M.; Ma, J.; Deng, G.; Zhang, S.; Sheu, W.C.; Zou, P.; Wu, H.; Liu, J.; et al. Targeted disruption of tumor vasculature via polyphenol nanoparticles to improve brain cancer treatment. Cell Rep. Phys. Sci. 2022, 3, 100691. [Google Scholar] [CrossRef]
- Tee, J.K.; Yip, L.X.; Tan, E.S.; Santitewagun, S.; Prasath, A.; Ke, P.C.; Ho, H.K.; Leong, D.T. Nanoparticles’ interactions with vasculature in diseases. Chem. Soc. Rev. 2019, 48, 5381–5407. [Google Scholar] [CrossRef] [PubMed]
- Setyawati, M.I.; Tay, C.Y.; Chia, S.L.; Goh, S.L.; Fang, W.; Neo, M.J.; Chong, H.C.; Tan, S.M.; Loo, S.C.J.; Ng, K.W.; et al. Titanium dioxide nanomaterials cause endothelial cell leakiness by disrupting the homophilic interaction of VE-cadherin. Nat. Commun. 2013, 4, 1673. [Google Scholar] [CrossRef] [PubMed]
- Vidyasagar, A.; Wilson, N.a.; Djamali, A. Heat shock protein 27 (HSP27): Biomarker of disease and therapeutic target. Fibrogenesis Tissue Repair 2012, 5, 7. [Google Scholar] [CrossRef]
- Iglesia, R.P.; Fernandes, C.F.d.L.; Coelho, B.P.; Prado, M.B.; Escobar, M.I.M.; Almeida, G.H.D.R.; Lopes, M.H. Heat shock proteins in glioblastoma biology: Where do we stand? Int. J. Mol. Sci. 2019, 20, 5794. [Google Scholar] [CrossRef]
- Engelhard, H.H.; Duncan, H.A.; Kim, S.; Criswell, P.S.; Van Eldik, L. Therapeutic effects of sodium butyrate on glioma cells in vitro and in the rat C6 glioma model. Neurosurgery 2001, 48, 616–625. [Google Scholar] [CrossRef]
- Taylor, M.A.; Khathayer, F.; Ray, S.K. Quercetin and Sodium Butyrate Synergistically Increase Apoptosis in Rat C6 and Human T98G Glioblastoma Cells Through Inhibition of Autophagy. Neurochem. Res. 2019, 44, 1715–1725. [Google Scholar] [CrossRef]
- Ko, J.H.; Sethi, G.; Um, J.Y.; Shanmugam, M.K.; Arfuso, F.; Kumar, A.P.; Bishayee, A.; Ahn, K.S. The role of resveratrol in cancer therapy. Int. J. Mol. Sci. 2017, 18, 2589. [Google Scholar] [CrossRef]
- Salehi, B.; Machin, L.; Monzote, L.; Sharifi-Rad, J.; Ezzat, S.M.; Salem, M.A.; Merghany, R.M.; El Mahdy, N.M.; Klllç, C.S.; Sytar, O.; et al. Therapeutic Potential of Quercetin: New Insights and Perspectives for Human Health. ACS Omega 2020, 5, 11849–11872. [Google Scholar] [CrossRef]
- Islam, M.S.; Quispe, C.; Hossain, R.; Islam, M.T.; Al-Harrasi, A.; Al-Rawahi, A.; Martorell, M.; Mamurova, A.; Seilkhan, A.; Altybaeva, N.; et al. Neuropharmacological Effects of Quercetin: A Literature-Based Review. Front. Pharmacol. 2021, 12, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-Arellano, L.; Salazar-García, M.; Corona, J.C. Neuroprotective effects of Quercetin in pediatric neurological diseases. Molecules 2020, 25, 5597. [Google Scholar] [CrossRef] [PubMed]
- Zamin, L.L.; Filippi-Chiela, E.C.; Dillenburg-Pilla, P.; Horn, F.; Salbego, C.; Lenz, G. Resveratrol and quercetin cooperate to induce senescence-like growth arrest in C6 rat glioma cells. Cancer Sci. 2009, 100, 1655–1662. [Google Scholar] [CrossRef] [PubMed]
- Nezami, M.A.; Duma, C. Proof of Concept in a Case Study of Glioblastoma Multiforme Successfully Treated with IV Quercetin in Combination with Leading Edge Gamma Knife and Standard Treatments. J. Cancer Ther. 2018, 9, 522–528. [Google Scholar] [CrossRef]
- Andres, S.; Pevny, S.; Ziegenhagen, R.; Bakhiya, N.; Schäfer, B.; Hirsch-Ernst, K.I.; Lampen, A. Safety Aspects of the Use of Quercetin as a Dietary Supplement. Mol. Nutr. Food Res. 2018, 62, 1700447. [Google Scholar] [CrossRef] [PubMed]
- Heeba, G.; Magda, M. Dual Effects of Quercetin in Doxorubicin-Induced Nephrotoxicity in Rats and its Modulation of the Cytotoxic Activity of Doxorubicin on Human Carcinoma Cells. Environ. Toxicol. 2016, 165, 624–636. [Google Scholar] [CrossRef] [PubMed]
- Hard, G.C.; Seely, J.C.; Betz, L.J.; Hayashi, S.m. Re-evaluation of the kidney tumors and renal histopathology occurring in a 2-year rat carcinogenicity bioassay of quercetin. Food Chem. Toxicol. 2007, 45, 600–608. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, C.L.; Peng, C.C.; Cheng, Y.M.; Lin, L.Y.; Ker, Y.B.; Chang, C.H.; Chen, K.C.; Peng, R.Y. Quercetin and ferulic acid aggravate renal carcinoma in long-term diabetic victims. J. Agric. Food Chem. 2010, 58, 9273–9280. [Google Scholar] [CrossRef]
- Singh, B.; Mense, S.M.; Bhat, N.K.; Putty, S.; Guthiel, W.A.; Remotti, F.; Bhat, H.K. Dietary Quercetin Exacerbates the Development of Estrogen- Induced Breast Tumors in Female ACI Rats. Toxicol. Appl. Pharmacol. 2010, 247, 83–90. [Google Scholar] [CrossRef] [PubMed]
- Sosnik, A.; Carcaboso, A.M. Nanomedicines in the future of pediatric therapy. Adv. Drug Deliv. Rev. 2014, 73, 140–161. [Google Scholar] [CrossRef] [PubMed]
- McCabe, E.R.B. Nanopediatrics: Enabling personalized medicine for children. Pediatr. Res. 2010, 67, 453–457. [Google Scholar] [CrossRef]
- Nanopediatrics. Available online: https://www.uclahealth.org/news/mattel-childrens-hospital-ucla-launches-new-nanopediatrics-program (accessed on 22 August 2022).
- Center for Pediatric Nanomedicine. Available online: https://pedsresearch.org/research/centers/center-for-pediatric-nanomedicine/overview/ (accessed on 22 August 2022).
- Stoyanova-Beninska, V.V.; Wohlfarth, T.; Isaac, M.; Kalverdijk, L.J.; van den Berg, H.; Gispen-de Wied, C. The EU paediatric regulation:. Effects on paediatric psychopharmacology in Europe. Eur. Neuropsychopharmacol. 2011, 21, 565–570. [Google Scholar] [CrossRef]
- Zwaan, C.M.; Kearns, P.; Caron, H.; Verschuur, A.; Riccardi, R.; Boos, J.; Doz, F.; Geoerger, B.; Morland, B.; Vassal, G. The role of the “innovative therapies for children with cancer” (ITCC) European consortium. Cancer Treat. Rev. 2010, 36, 328–334. [Google Scholar] [CrossRef] [PubMed]
- Di Pietro, M.L.; Cutrera, R.; Teleman, A.A.; Barbaccia, M.L. Placebo-controlled trials in pediatrics and the child’s best interest. Ital. J. Pediatr. 2015, 41, 11. [Google Scholar] [CrossRef] [PubMed]
- Milne, C.P.; Bruss, J.B. The economics of pediatric formulation development for off-patent drugs. Clin. Ther. 2008, 30, 2133–2145. [Google Scholar] [CrossRef] [PubMed]
- European Paediatric Formulation Initiative (EuPFI). Available online: http://www.eupfi.org/about-eupfi/ (accessed on 25 August 2022).





| Modified Que Formulations/Que with Other Therapeutic Molecules | Cell Line/Animal Model | Main Findings | References |
|---|---|---|---|
| Que-loaded nano-particles with poly (lactic-co-glycolic) acid (PLGA) | C6 glioma cells | Que-loaded nano-particles with poly (lactic-co-glycolic) acid (PLGA) effectively inhibited the cell proliferation. The smallest size of nano-particles revealed the better Que cellular uptake and antioxidant activity. | Ersoz et al., 2020 [128] |
| Que coated with PEG2000-DPSE | C6 glioma cells | Que coated with PEG2000-DPSE enhanced anticancer effect on C6 glioma cell lines | Wang et al., 2013 [37] |
| Que-platelet (Natural human platelets) | U373-MG cell line | Que obtained threefold enhancement of solubility, and increasement of cytotoxic effect. | Bhandarkar et al., 2021 [129] |
| Que nano-particles prepared with gold and loaded into PLGA | Human neuroglioma cells U8 and BALB/c-nude mice | Induction of cell autophagy and apoptosis in human neuroglioma cells as well as suppressed tumor growth through activation LC3/ERK/Caspase 3 and suppression AKT/mTOR signaling | Lou et al., 2016 [28] |
| Que-nano-liposomes | C6 glioma cells | Que-loaded liposomes has enhanced cytotoxic effects inducing type III programmed cell death (necrosis) in C6 glioma cells. | Wang et al., 2012 [130] |
| Que-nano-particles | Mouse model | Que nano-particles via anti-angiogenic have significant effect on reducing tumor development and enhancing drug delivery. | Liu et al., 2022 [131] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shala, A.L.; Arduino, I.; Salihu, M.B.; Denora, N. Quercetin and Its Nano-Formulations for Brain Tumor Therapy—Current Developments and Future Perspectives for Paediatric Studies. Pharmaceutics 2023, 15, 963. https://doi.org/10.3390/pharmaceutics15030963
Shala AL, Arduino I, Salihu MB, Denora N. Quercetin and Its Nano-Formulations for Brain Tumor Therapy—Current Developments and Future Perspectives for Paediatric Studies. Pharmaceutics. 2023; 15(3):963. https://doi.org/10.3390/pharmaceutics15030963
Chicago/Turabian StyleShala, Aida Loshaj, Ilaria Arduino, Mimoza Basholli Salihu, and Nunzio Denora. 2023. "Quercetin and Its Nano-Formulations for Brain Tumor Therapy—Current Developments and Future Perspectives for Paediatric Studies" Pharmaceutics 15, no. 3: 963. https://doi.org/10.3390/pharmaceutics15030963
APA StyleShala, A. L., Arduino, I., Salihu, M. B., & Denora, N. (2023). Quercetin and Its Nano-Formulations for Brain Tumor Therapy—Current Developments and Future Perspectives for Paediatric Studies. Pharmaceutics, 15(3), 963. https://doi.org/10.3390/pharmaceutics15030963








