Prediction of Unwanted Crystallization of Freeze-Dried Protein Formulations Using α-Relaxation Measurements
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparations of Formulations
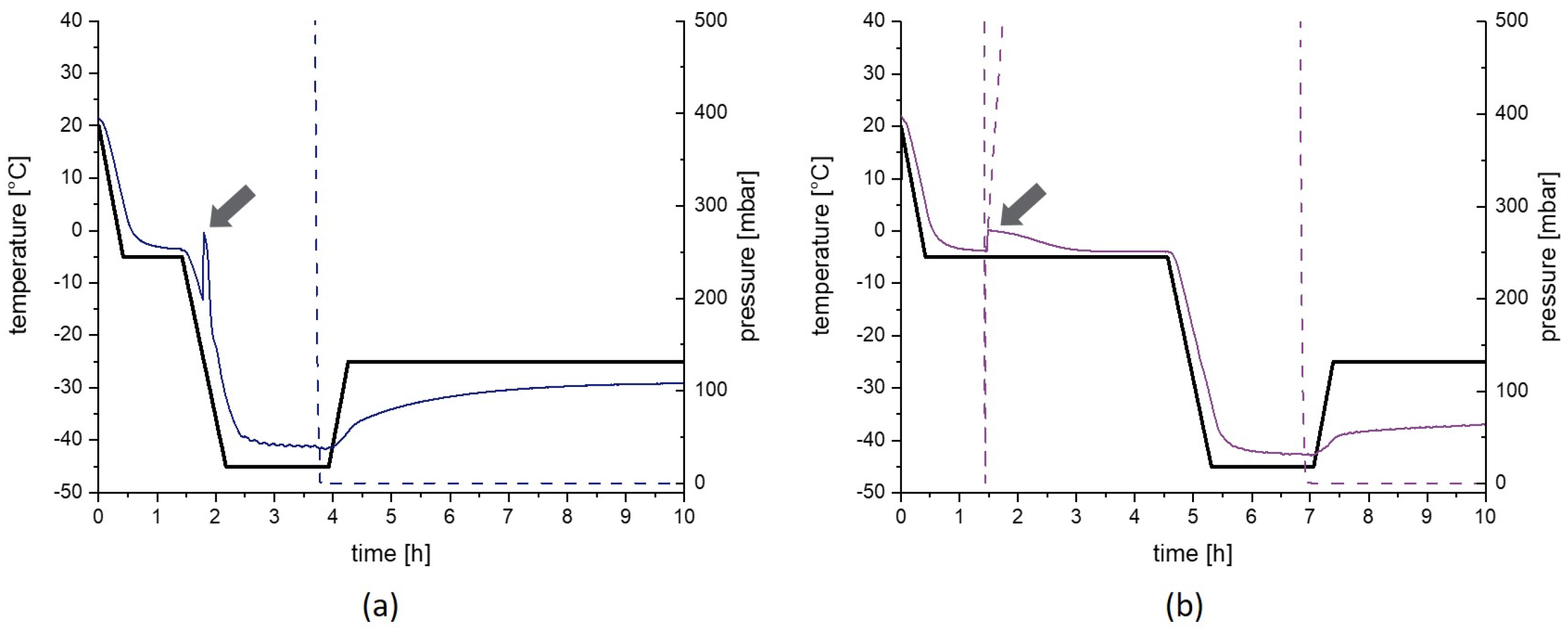
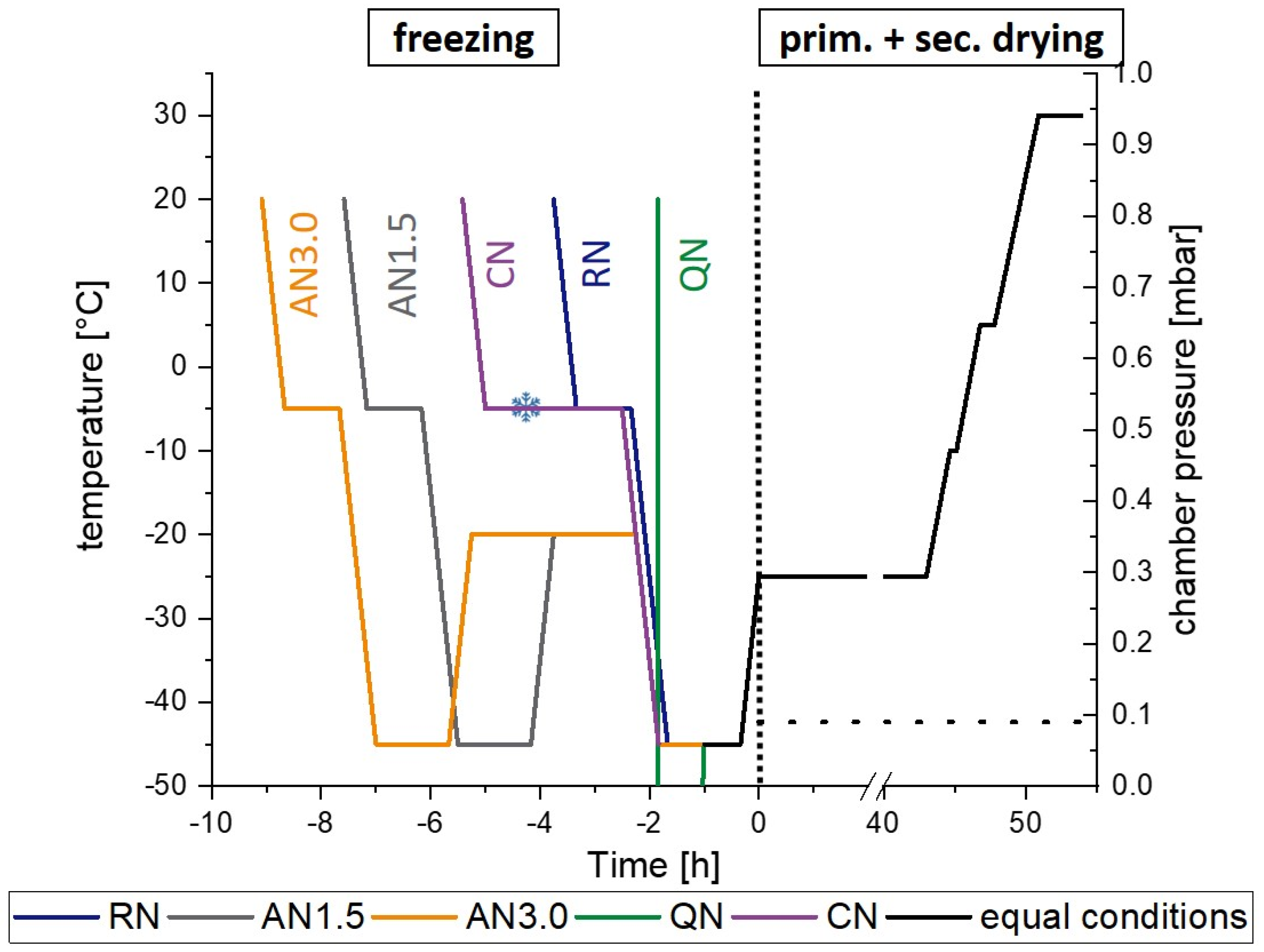
2.2. Freeze-Drying Protocols
2.3. Residual Moisture Content
2.4. Differential Scanning Calorimetry
2.5. Isothermal Microcalorimetry (To Determine τβ)
2.6. Specific Surface Area
2.7. X-ray Powder Diffraction
2.8. Procedure to Correlate Crystallization with Relaxation Time τβ
3. Results
3.1. Macroscopic and Microscopic Appearance
3.2. Residual Moisture
3.3. Specific Surface Area (SSA)
3.4. Differential Scanning Calorimetry Results
3.5. X-ray Powder Diffraction
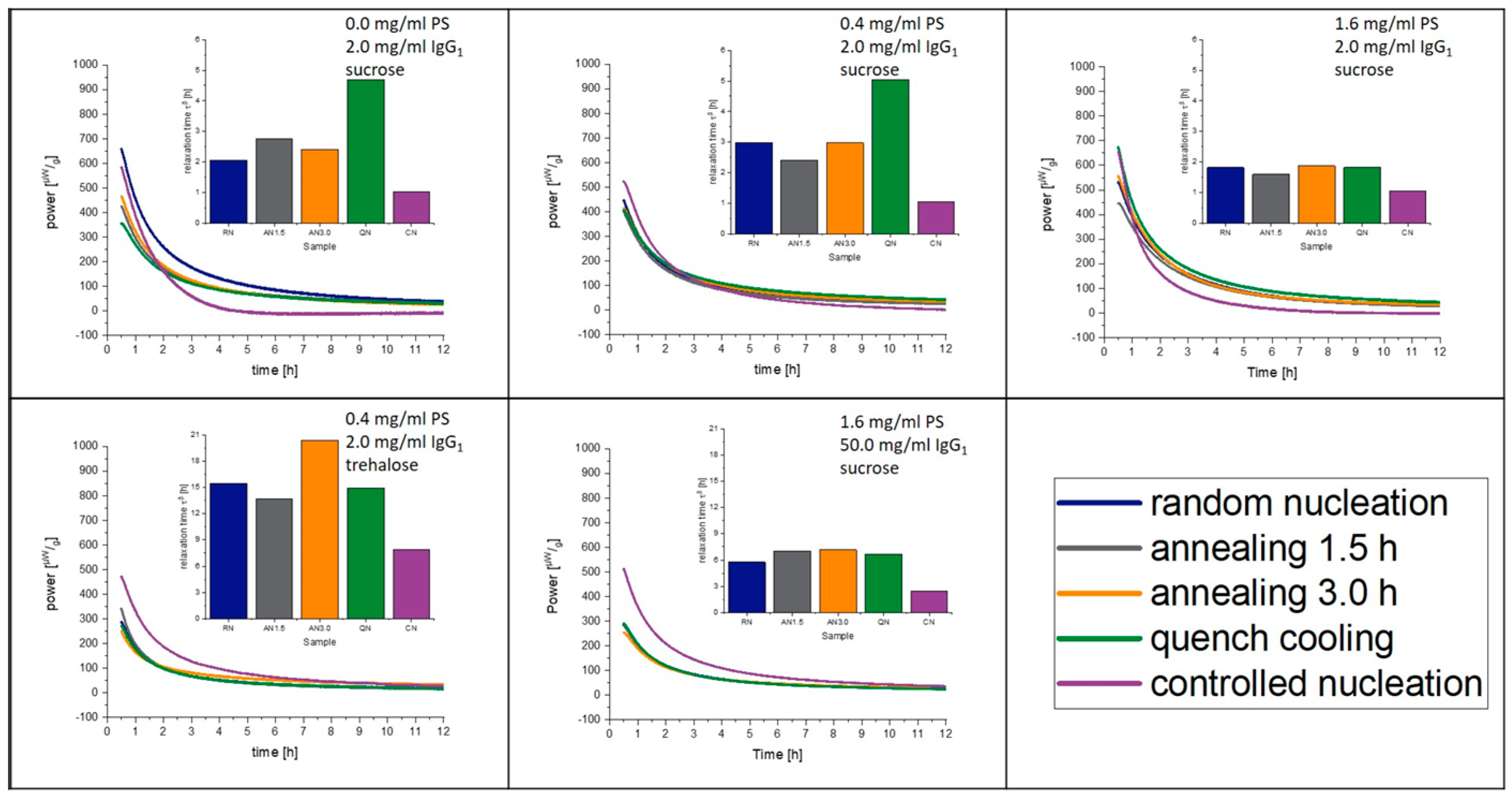
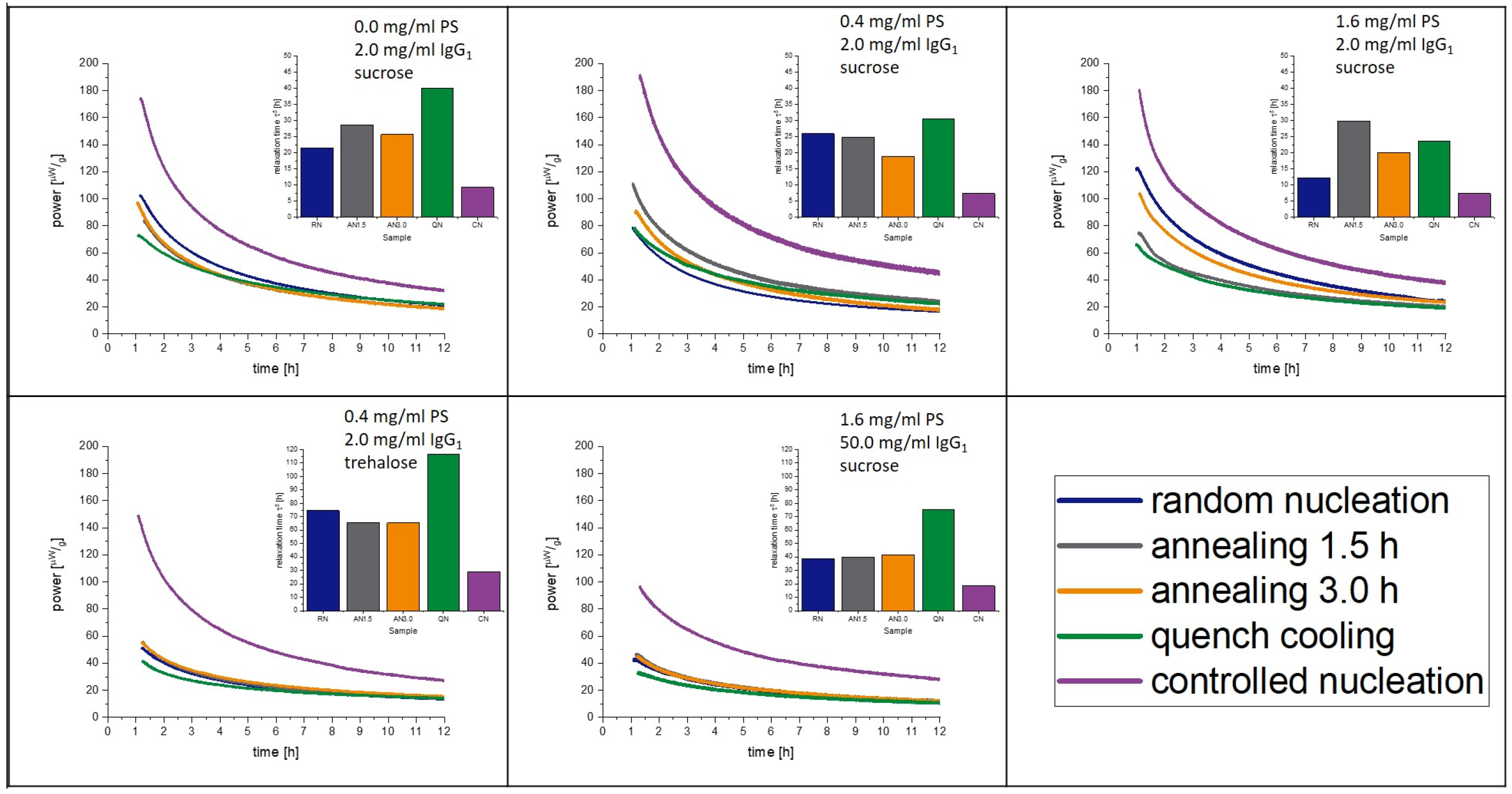
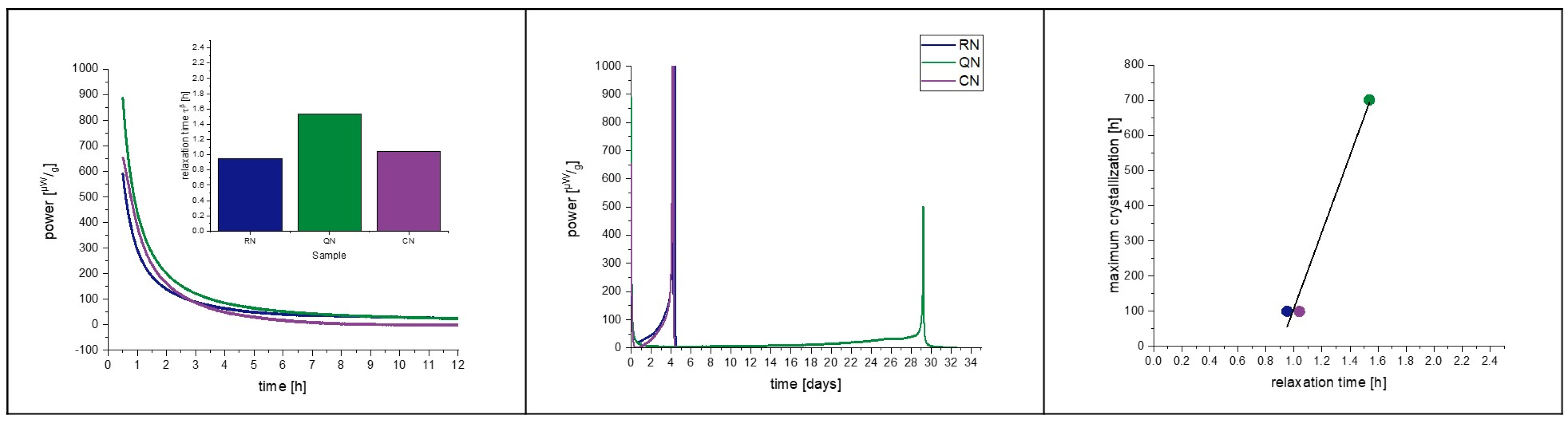
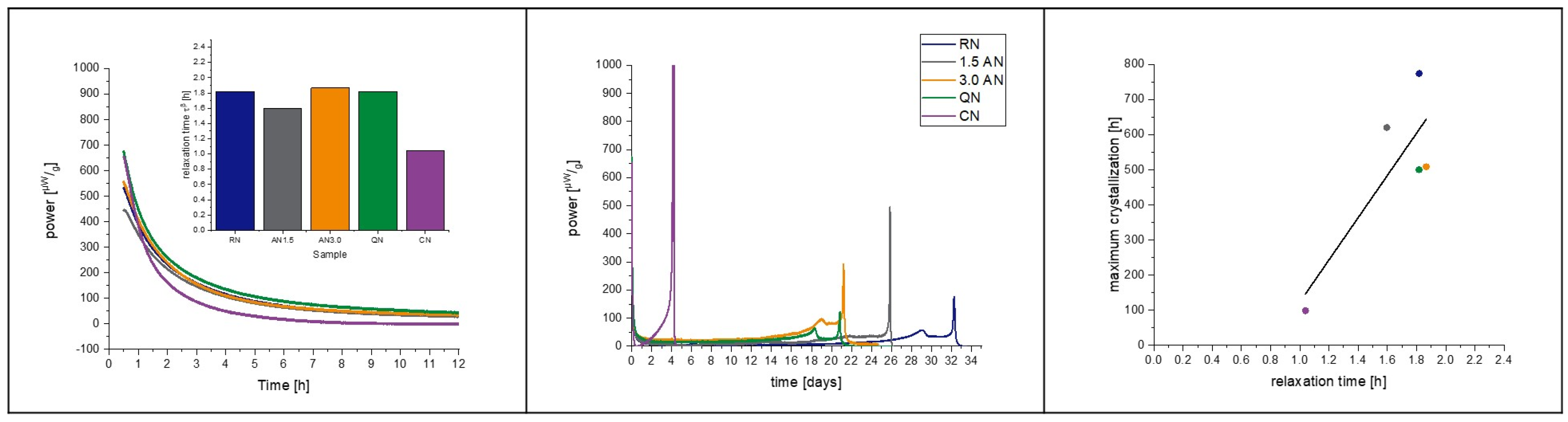
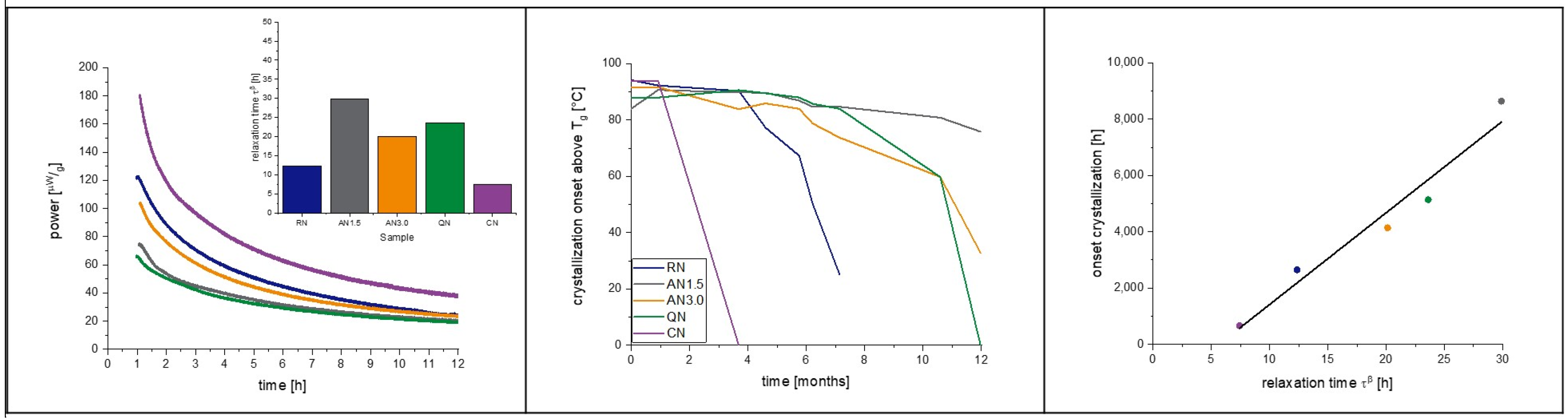
3.6. Isothermal Microcalorimetry
4. Discussion
4.1. Placebo Formulation Pre-Test
4.2. Protein-Containing Samples Equalized for Residual Moisture
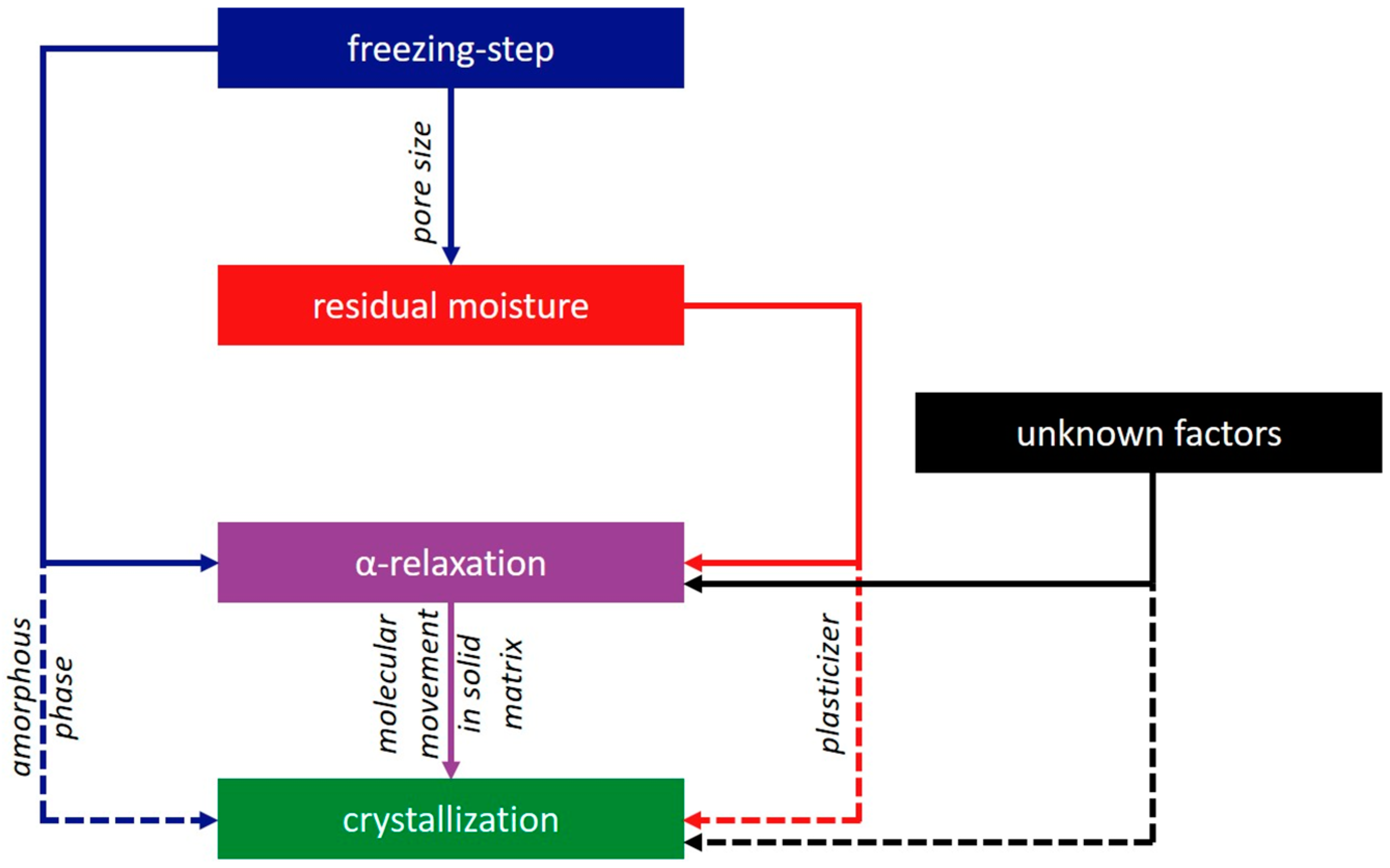
- The freezing step influences the residual moisture directly by determining the pore size of the formulation [36,37]. Furthermore, the freezing step influences α-relaxation directly by changes in the amorphous phase as well as indirectly by the residual moisture. Both effects are independent from each other.
- The residual moisture influences the α-relaxation and crystallization directly by plastization of the solid phase [31]. The effect on α-relaxation and crystallization is proportional.
4.3. Protein-Containing Samples
4.4. Comparison of IMC and DSC as Methods for Crystallization Prediction
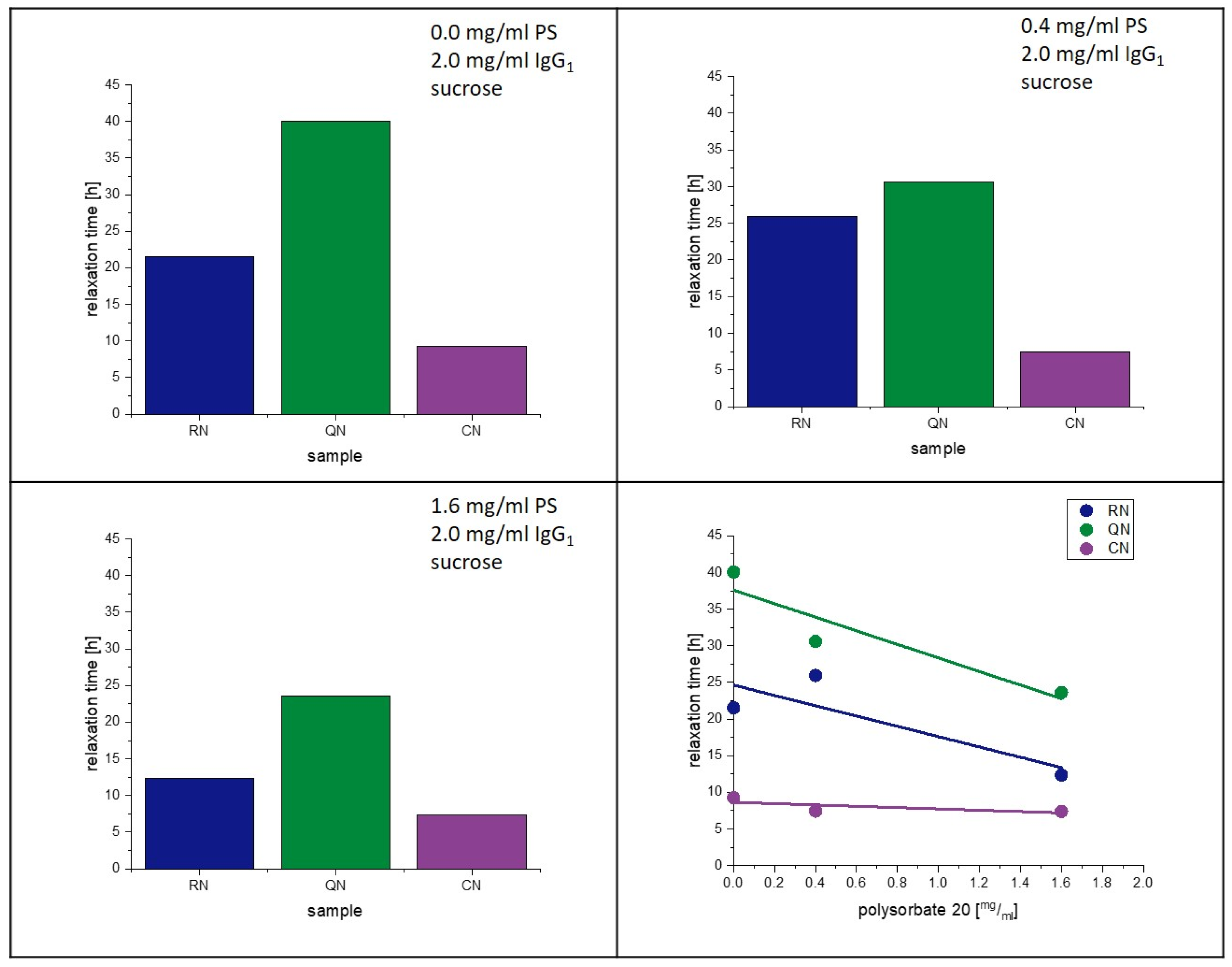
4.5. Influence of PS20 on Relaxation and Crystallization
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| RN | AN1.5 | AN3.0 | QN | CN | |
|---|---|---|---|---|---|
| Tg [°C] | 60.21 ± 0.04 | 60.09 ± 0.03 | 60.16 ± 0.04 | 72.03 ± 0.06 | 48.98 ± 0.08 |
| Δcp [Jg−1K−1] | 0.55 ± 0.02 | 0.42 ± 0.05 | 0.52 ± 0.08 | 0.46 ± 0.04 | 0.51 ± 0.06 |
| [Jg−1] | 19.21 ± 0.72 | 14.81 ± 1.76 | 18.18 ± 2.82 | 21.45 ± 1.90 | 12.33 ± 1.46 |
| [Jg−1] | 11.02 ± 0.41 | 8.48 ± 1.01 | 10.42 ± 1.62 | 14.61 ± 1.29 | 4.62 ± 0.55 |
| crystallization onset [°C] | 97.20 ± 2.57 | 100.10 ± 0.42 | 101.65 ± 0.25 | 119.90 ± 0.28 | 91.96 ± 1.10 |
| crystallization energy [Jg−1] | 70.48 ± 0.95 | 66.47 ± 0.30 | 71.20 ± 7.71 | 62.66 ± 2.31 | 64.15 ± 1.09 |
| RN | AN1.5 | AN3.0 | QN | CN | |
|---|---|---|---|---|---|
| Tg [°C] | 62.26 ± 2.73 | 64.10 ± 1.41 | 62.09 ± 2.74 | 66.20 ± 3.19 | 50.45 ± 2.67 |
| Δcp [Jg−1K−1] | 0.52 ± 0.12 | 0.62 ± 0.02 | 0.560 ± 0.03 | 0.48 ± 0.07 | 0.52 ± 0.10 |
| [Jg−1] | 19.23 ± 5.21 | 24.04 ± 0.95 | 20.73 ± 2.07 | 19.92 ± 3.79 | 13.14 ± 3.31 |
| [Jg−1] | 11.49 ± 3.12 | 14.82 ± 0.58 | 12.35 ± 1.23 | 12.67 ± 2.41 | 5.40 ± 1.36 |
| crystallization onset [°C] | 103.87 ± 5.53 | 106.15 ± 2.84 | 96.80 ± 1.31 | 99.23 ± 6.61 | 87.15 ± 1.10 |
| crystallization energy [Jg−1] | 66.75 ± 1.54 | 74.32 ± 0.29 | 69.41 ± 1.00 | 68.74 ± 1.10 | 62.69 ± 1.10 |
| RN | AN1.5 | AN3.0 | QN | CN | |
|---|---|---|---|---|---|
| Tg [°C] | 62.27 ± 2.93 | 60.19 ± 1.74 | 60.01 ± 2.47 | 64.05 ± 3.06 | 48.29 ± 3.11 |
| Δcp [Jg−1K−1] | 0.54 ± 0.02 | 0.47 ± 0.03 | 0.57 ± 0.01 | 0.29 ± 0.01 | 0.44 ± 0.01 |
| [Jg−1] | 20.20 ± 1.85 | 16.64 ± 1.54 | 20.04 ± 0.90 | 11.13 ± 0.92 | 10.14 ± 0.93 |
| [Jg−1] | 12.07 ± 1.10 | 9.55 ± 0.88 | 11.45 ± 0.51 | 6.85 ± 0.57 | 3.61 ± 0.33 |
| crystallization onset [°C] | 96.21 ± 2.83 | 84.05 ± 1.16 | 93.74 ± 0.26 | 88.01 ± 1.54 | 88.11 ± 2.86 |
| crystallization energy [Jg−1] | 64.79 ± 0.04 | 57.62 ± 1.23 | 62.42 ± 1.23 | 60.85 ± 0.98 | 63.68 ± 3.02 |
| RN | AN1.5 | AN3.0 | QN | CN | |
|---|---|---|---|---|---|
| Tg [°C] | 80.32 ± 0.04 | 84.14 ± 0.24 | 80.31 ± 0.42 | 84.14 ± 0.42 | 63.82 ± 0.70 |
| Δcp [Jg−1K−1] | 0.30 ± 0.01 | 0.34 ± 0.10 | 0.31 ± 0.02 | 0.33 ± 0.02 | 0.40 ± 0.02 |
| [Jg−1] | 16.71 ± 0.63 | 20.05 ± 5.97 | 16.76 ± 1.10 | 19.58 ± 1.28 | 15.37 ± 0.72 |
| [Jg−1] | 12.18 ± 0.46 | 14.96 ± 4.46 | 12.21 ± 0.80 | 14.61 ± 0.96 | 9.43 ± 0.44 |
| crystallization onset [°C] | † | † | † | † | † |
| crystallization energy [Jg−1] | † | † | † | † | † |
| RN | AN1.5 | AN3.0 | QN | CN | |
| Tg [°C] | 104.46 ± 0.09 | 104.09 ± 0.04 | 104.08 ± 0.16 | 106.51 ± 0.23 | 100.11 ± 0.11 |
| Δcp [Jg−1K−1] | 0.46 ± 0.07 | 0.47 ± 0.01 | 0.41 ± 0.01 | 0.41 ± 0.02 | 0.52 ± 0.01 |
| [Jg−1] | 36.59 ± 5.37 | 36.82 ± 0.97 | 32.06 ± 0.89 | 33.66 ± 1.70 | 39.36 ± 0.79 |
| [Jg−1] | 29.68 ± 4.36 | 29.83 ± 0.78 | 25.98 ± 0.72 | 27.47 ± 1.39 | 31.50 ± 0.64 |
| crystallization onset [°C] | † | † | † | † | † |
| crystallization energy [Jg−1] | † | † | † | † | † |
Appendix B
References
- Mullard, A. 2020 FDA drug approvals. Nat. Rev. Drug Discov. 2021, 20, 85–90. [Google Scholar] [CrossRef] [PubMed]
- Chi, E.Y.; Krishnan, S.; Randolph, T.W.; Carpenter, J.F. Physical stability of proteins in aqueous solution: Mechanism and driving forces in nonnative protein aggregation. Pharm. Res. 2003, 20, 1325–1336. [Google Scholar] [CrossRef] [PubMed]
- Carpenter, J.F.; Pikal, M.J.; Chang, B.S.; Randolph, T.W. Rational design of stable lyophilized protein formulations: Some practical advice. Pharm. Res. 1997, 14, 969–975. [Google Scholar] [CrossRef] [PubMed]
- Carpenter, J.F.; Chang, B.S.; Garzon-Rodriguez, W.; Randolph, T.W. Rational design of stable lyophilized protein formulations: Theory and practice. Pharm. Biotechnol. 2002, 13, 109–133. [Google Scholar] [CrossRef] [PubMed]
- Pyne, A.; Surana, R.; Suryanarayanan, R. Crystallization of mannitol below Tg’ during freeze-drying in binary and ternary aqueous systems. Pharm. Res. 2002, 19, 901–908. [Google Scholar] [CrossRef]
- Costantino, H.R. Excipients for Use in Lyophilized Pharmaceutical Peptide, Protein and other Bioproducts. In Lyophilization of Biopharmaceuticals; AAPS Press: Arlington, VA, USA, 2004; pp. 139–228. ISBN 0971176760. [Google Scholar]
- Allison, S.D.; Chang, B.; Randolph, T.W.; Carpenter, J.F. Hydrogen bonding between sugar and protein is responsible for inhibition of dehydration-induced protein unfolding. Arch. Biochem. Biophys. 1999, 365, 289–298. [Google Scholar] [CrossRef]
- Cicerone, M.T.; Douglas, J.F. β-Relaxation governs protein stability in sugar-glass matrices. Soft Matter 2012, 8, 2983. [Google Scholar] [CrossRef]
- Liu, J.; Rigsbee, D.R.; Stotz, C.; Pikal, M.J. Dynamics of pharmaceutical amorphous solids: The study of enthalpy relaxation by isothermal microcalorimetry. J. Pharm. Sci. 2002, 91, 1853–1862. [Google Scholar] [CrossRef]
- Pikal, M.J.; Rigsbee, D.R. The stability of insulin in crystalline and amorphous solids: Observation of greater stability for the amorphous form. Pharm. Res. 1997, 14, 1379–1387. [Google Scholar] [CrossRef]
- Shamblin, S.L.; Tang, X.; Chang, L.; Hancock, B.C.; Pikal, M.J. Characterization of the Time Scales of Molecular Motion in Pharmaceutically Important Glasses. J. Phys. Chem. B 1999, 103, 4113–4121. [Google Scholar] [CrossRef]
- Cicerone, M.T.; Tellington, A.; Trost, L.; Sokolov, A. Substantially Improved Stability of Biological Agents in Dried Form. BioProcess Int. 2003. [Google Scholar]
- Liu, J. Physical characterization of pharmaceutical formulations in frozen and freeze-dried solid states: Techniques and applications in freeze-drying development. Pharm. Dev. Technol. 2006, 11, 3–28. [Google Scholar] [CrossRef] [PubMed]
- Cleland, J.L.; Lam, X.; Kendrick, B.; Yang, J.; Yang, T.; Overcashier, D.; Brooks, D.; Hsu, C.; Carpenter, J.F. A specific molar ratio of stabilizer to protein is required for storage stability of a lyophilized monoclonal antibody. J. Pharm. Sci. 2001, 90, 310–321. [Google Scholar] [CrossRef]
- Kedward, C.J.; MacNaughtan, W.; Blanshard, J.M.V.; Mitchell, J.R. Crystallization Kinetics of Lactose and Sucrose Based On Isothermal Differential Scanning Calorimetry. J. Food Sci. 1998, 63, 192–197. [Google Scholar] [CrossRef]
- Chieng, N.; Teo, X.; Cheah, M.H.; Choo, M.L.; Chung, J.; Hew, T.K.; Keng, P.S. Molecular Dynamics and Physical Stability of Pharmaceutical Co-amorphous Systems: Correlation Between Structural Relaxation Times Measured by Kohlrausch-Williams-Watts With the Width of the Glass Transition Temperature (ΔTg) and the Onset of Crystallization. J. Pharm. Sci. 2019, 108, 3848–3858. [Google Scholar] [CrossRef]
- Pikal, M.J.; Dellerman, K.M. Stability testing of pharmaceuticals by high-sensitivity isothermal calorimetry at 25 °C: Cephalosporins in the solid and aqueous solution states. Int. J. Pharm. 1989, 50, 233–252. [Google Scholar] [CrossRef]
- Bhugra, C.; Shmeis, R.; Krill, S.L.; Pikal, M.J. Predictions of onset of crystallization from experimental relaxation times I-correlation of molecular mobility from temperatures above the glass transition to temperatures below the glass transition. Pharm. Res. 2006, 23, 2277–2290. [Google Scholar] [CrossRef]
- Surana, R.; Suryanarayanan, R. Quantitation of crystallinity in substantially amorphous pharmaceuticals and study of crystallization kinetics by X-ray powder diffractometry. Powder Diffr. 2000, 15, 2–6. [Google Scholar] [CrossRef]
- Kim, A.I.; Akers, M.J.; Nail, S.L. The physical state of mannitol after freeze-drying: Effects of mannitol concentration, freezing rate, and a noncrystallizing cosolute. J. Pharm. Sci. 1998, 87, 931–935. [Google Scholar] [CrossRef]
- Iurian, S.; Bogdan, C.; Tomuță, I.; Szabó-Révész, P.; Chvatal, A.; Leucuța, S.E.; Moldovan, M.; Ambrus, R. Development of oral lyophilisates containing meloxicam nanocrystals using QbD approach. Eur. J. Pharm. Sci. 2017, 104, 356–365. [Google Scholar] [CrossRef]
- Haque, M.K.; Roos, Y.H. Crystallization and X-ray Diffraction of Crystals Formed in Water-Plasticized Amorphous Spray-dried and Freeze-dried Lactose/Protein Mixtures. J. Food Sci. 2005, 70, E359–E366. [Google Scholar] [CrossRef]
- Haque, M.K.; Roos, Y.H. Crystallization and X-ray diffraction of spray-dried and freeze-dried amorphous lactose. Carbohydr. Res. 2005, 340, 293–301. [Google Scholar] [CrossRef] [PubMed]
- Bhugra, C.; Rambhatla, S.; Bakri, A.; Duddu, S.P.; Miller, D.P.; Pikal, M.J.; Lechuga-Ballesteros, D. Prediction of the onset of crystallization of amorphous sucrose below the calorimetric glass transition temperature from correlations with mobility. J. Pharm. Sci. 2007, 96, 1258–1269. [Google Scholar] [CrossRef] [PubMed]
- Kawakami, K.; Pikal, M.J. Calorimetric investigation of the structural relaxation of amorphous materials: Evaluating validity of the methodologies. J. Pharm. Sci. 2005, 94, 948–965. [Google Scholar] [CrossRef]
- Bhugra, C.; Shmeis, R.; Krill, S.L.; Pikal, M.J. Different measures of molecular mobility: Comparison between calorimetric and thermally stimulated current relaxation times below Tg and correlation with dielectric relaxation times above Tg. J. Pharm. Sci. 2008, 97, 4498–4515. [Google Scholar] [CrossRef]
- Shamblin, S.L.; Hancock, B.C.; Pikal, M.J. Coupling between chemical reactivity and structural relaxation in pharmaceutical glasses. Pharm. Res. 2006, 23, 2254–2268. [Google Scholar] [CrossRef]
- Groёl, S.; Menzen, T.; Winter, G. Calorimetric Investigation of the Relaxation Phenomena in Amorphous Lyophilized Solids. Pharmaceutics 2021, 13, 1735. [Google Scholar] [CrossRef]
- Bhugra, C.; Shmeis, R.; Krill, S.L.; Pikal, M.J. Prediction of onset of crystallization from experimental relaxation times. II. Comparison between predicted and experimental onset times. J. Pharm. Sci. 2008, 97, 455–472. [Google Scholar] [CrossRef]
- Abdul-Fattah, A.M.; Truong-Le, V.; Yee, L.; Nguyen, L.; Kalonia, D.S.; Cicerone, M.T.; Pikal, M.J. Drying-induced variations in physico-chemical properties of amorphous pharmaceuticals and their impact on stability (I): Stability of a monoclonal antibody. J. Pharm. Sci. 2007, 96, 1983–2008. [Google Scholar] [CrossRef]
- Abdul-Fattah, A.M.; Dellerman, K.M.; Bogner, R.H.; Pikal, M.J. The effect of annealing on the stability of amorphous solids: Chemical stability of freeze-dried moxalactam. J. Pharm. Sci. 2007, 96, 1237–1250. [Google Scholar] [CrossRef]
- Luthra, S.A.; Hodge, I.M.; Utz, M.; Pikal, M.J. Correlation of annealing with chemical stability in lyophilized pharmaceutical glasses. J. Pharm. Sci. 2008, 97, 5240–5251. [Google Scholar] [CrossRef] [PubMed]
- Ilona Vollrath. Controlled Nucleation and Heat Flux Measurements as Innovative Technologies for Freeze-Drying. Available online: https://edoc.ub.uni-muenchen.de/23637/1/Vollrath_Ilona.pdf (accessed on 4 January 2023).
- NITROGEN|Air Liquide. Available online: https://mygas.airliquide.de/catalog-gas-products/stickstoff-technisch?language=en-DE (accessed on 2 February 2023).
- Lo Presti, K.; Frieß, W. Adjustment of specific residual moisture levels in completely freeze-dried protein formulations by controlled spiking of small water volumes. Eur. J. Pharm. Biopharm. 2021, 169, 292–296. [Google Scholar] [CrossRef]
- Konstantinidis, A.K.; Kuu, W.; Otten, L.; Nail, S.L.; Sever, R.R. Controlled nucleation in freeze-drying: Effects on pore size in the dried product layer, mass transfer resistance, and primary drying rate. J. Pharm. Sci. 2011, 100, 3453–3470. [Google Scholar] [CrossRef] [PubMed]
- Geidobler, R.; Winter, G. Controlled ice nucleation in the field of freeze-drying: Fundamentals and technology review. Eur. J. Pharm. Biopharm. 2013, 85, 214–222. [Google Scholar] [CrossRef] [PubMed]
- Hottot, A.; Vessot, S.; Andrieu, J. Freeze drying of pharmaceuticals in vials: Influence of freezing protocol and sample configuration on ice morphology and freeze-dried cake texture. Chem. Eng. Process. Process Intensif. 2007, 46, 666–674. [Google Scholar] [CrossRef]
- Rambhatla, S.; Ramot, R.; Bhugra, C.; Pikal, M.J. Heat and mass transfer scale-up issues during freeze drying: II. Control and characterization of the degree of supercooling. AAPS PharmSciTech 2004, 5, e58. [Google Scholar] [CrossRef]
- Chang, B.S.; Kendrick, B.S.; Carpenter, J.F. Surface-induced denaturation of proteins during freezing and its inhibition by surfactants. J. Pharm. Sci. 1996, 85, 1325–1330. [Google Scholar] [CrossRef]
- Searles, J.A.; Carpenter, J.F.; Randolph, T.W. Annealing to optimize the primary drying rate, reduce freezing-induced drying rate heterogeneity, and determine T(g)’ in pharmaceutical lyophilization. J. Pharm. Sci. 2001, 90, 872–887. [Google Scholar] [CrossRef]
- Searles, J.A.; Carpenter, J.F.; Randolph, T.W. The ice nucleation temperature determines the primary drying rate of lyophilization for samples frozen on a temperature-controlled shelf. J. Pharm. Sci. 2001, 90, 860–871. [Google Scholar] [CrossRef]
- Kharatyan, T.; Gopireddy, S.R.; Ogawa, T.; Kodama, T.; Nishimoto, N.; Osada, S.; Scherließ, R.; Urbanetz, N.A. Quantitative Analysis of Glassy State Relaxation and Ostwald Ripening during Annealing Using Freeze-Drying Microscopy. Pharmaceutics 2022, 14, 1176. [Google Scholar] [CrossRef]
- Patel, S.M.; Doen, T.; Pikal, M.J. Determination of end point of primary drying in freeze-drying process control. AAPS PharmSciTech 2010, 11, 73–84. [Google Scholar] [CrossRef] [PubMed]
- Ehlers, S.; Schroeder, R.; Friess, W. Trouble with the Neighbor during Freeze-Drying: Rivalry About Energy. J. Pharm. Sci. 2021, 110, 1219–1226. [Google Scholar] [CrossRef] [PubMed]
- Makower, B.; Dye, W.B. Sugar Crystallization, Equilibrium Moisture Content and Crystallization of Amorphous Sucrose and Glucose. J. Agric. Food Chem. 1956, 4, 72–77. [Google Scholar] [CrossRef]
- Tomlinson, A.; Zarraga, I.E.; Demeule, B. Characterization of Polysorbate Ester Fractions and Implications in Protein Drug Product Stability. Mol. Pharm. 2020, 17, 2345–2353. [Google Scholar] [CrossRef]
- Kerwin, B.A. Polysorbates 20 and 80 used in the formulation of protein biotherapeutics: Structure and degradation pathways. J. Pharm. Sci. 2008, 97, 2924–2935. [Google Scholar] [CrossRef] [PubMed]
- Mittal, K.L. Determination of CMC of polysorbate 20 in aqueous solution by surface tension method. J. Pharm. Sci. 1972, 61, 1334–1335. [Google Scholar] [CrossRef]




| Excipient | 2 mg/mL 00PS_Suc | 2 mg/mL 04PS_Suc | 2 mg/mL 16PS_Suc | 50 mg/mL 16PS_Suc | 2 mg/mL 04PS_Tre |
|---|---|---|---|---|---|
| LMU1 | 2 | 2 | 2 | 50 | 2 |
| sucrose | 79.45 | 79.45 | 79.45 | 79.45 | - |
| trehalose | - | - | - | - | 79.45 |
| PS20 | - | 0.4 | 1.6 | 1.6 | 0.4 |
| Process | Step Number [#] | Shelf Temperature [°C] | Hold Time [h] | Ramp Rate of the Shelf towards the Next Step [K/min] |
|---|---|---|---|---|
| RN | 1 | 20 | - | −1 |
| 2 | −5 | 1.00 | −1 | |
| 3 | −45 | 1.50 | * | |
| AN1.5 | 1 | 20 | - | −1 |
| 2 | −5 | 1.00 | −1 | |
| 3 | −45 | 1.50 | +1 | |
| 4 | −20 | 1.50 | −1 | |
| 5 | −45 | 1.50 | * | |
| AN3.0 | 1 | 20 | - | −1 |
| 2 | −5 | 1.00 | −1 | |
| 3 | −45 | 1.50 | +1 | |
| 4 | −20 | 3.00 | −1 | |
| 5 | −45 | 1.50 | * | |
| CN | 1 | 20 | - | −1 |
| 2 | −5 | 1.00 | † | |
| 3 | −5 | 2.00 | −1 | |
| 4 | −45 | 1.50 | * | |
| QN | 1 | 20 | - | quenching |
| 2 | −196 ‡ | 0.03 | - | |
| 3 | −45 | 1.500 | * |
| Excipient | 2 mg/mL 00PS_Suc | 2 mg/mL 04PS_Suc | 2 mg/mL 16PS_Suc | 50 mg/mL 16PS_Suc | 2 mg/mL 04PS_Tre |
|---|---|---|---|---|---|
| RN | 1.04 ± 0.03 | 0.84 ± 0.06 | 0.70 ± 0.06 | 0.31 ± 0.02 | 0.28 ± 0.05 |
| AN1.5 | 0.90 ± 0.02 | 0.77 ± 0.07 | 0.85 ± 0.13 | 0.39 ± 0.04 | 0.36 ± 0.02 |
| AN3.0 | 0.85 ± 0.00 | 0.93 ± 0.04 | 0.85 ± 0.01 | 0.35 ± 0.02 | 0.33 ± 0.03 |
| QN | 0.41 ± 0.04 | 0.49 ± 0.14 | 0.55 ± 0.02 | 0.17 ± 0.06 | 0.13 ± 0.01 |
| CN | 1.45 ± 0.10 | 1.42 ± 0.12 | 1.35 ± 0.02 | 1.34 ± 0.01 | 1.59 ± 0.22 |
| Process | Original | Moisture Equilibrated |
|---|---|---|
| RN | 0.70 ± 0.06 | 1.30 ± 0.14 |
| QN | 0.55 ± 0.02 | 1.15 ± 0.16 |
| CN | 1.35 ± 0.02 | 1.36 ± 0.12 |
| Excipient | 2 mg/mL 00PS_Suc | 2 mg/mL 04PS_Suc | 2 mg/mL 16PS_Suc | 50 mg/mL 16PS_Suc | 2 mg/mL 04PS_Tre |
|---|---|---|---|---|---|
| RN | 0.455 | 0.552 | 0.563 | 0.714 | 0.665 |
| AN1.5 | 0.404 | 0.594 | 0.549 | 0.747 | 0.739 |
| AN3.0 | 0.528 | 0.574 | 0.506 | 0.726 | 0.616 |
| QN | 1.080 | 0.982 | 0.914 | 2.270 | 0.982 |
| CN | 0.328 | 0.313 | 0.241 | 0.157 | 0.389 |
| Excipient | 2 mg/mL 00PS_Suc | 2 mg/mL 04PS_Suc | 2 mg/mL 16PS_Suc | 50 mg/mL 16PS_Suc | 2 mg/mL 04PS_Tre |
|---|---|---|---|---|---|
| RN | 60.21 ± 0.04 | 62.26 ± 2.73 | 62.27 ± 2.93 | 80.32 ± 0.04 | 104.46 ± 0.09 |
| AN1.5 | 60.09 ± 0.03 | 64.10 ± 1.41 | 60.19 ± 1.74 | 84.14 ± 0.24 | 104.09 ± 0.04 |
| AN3.0 | 60.16 ± 0.04 | 62.09 ± 2.74 | 60.01 ± 2.47 | 80.31 ± 0.42 | 104.08 ± 0.16 |
| QN | 72.03 ± 0.06 | 66.20 ± 3.19 | 64.05 ± 3.06 | 84.14 ± 0.42 | 106.51 ± 0.23 |
| CN | 48.98 ± 0.08 | 50.45 ± 2.67 | 48.29 ± 3.11 | 63.82 ± 0.70 | 100.11 ± 0.11 |
| Process | 2 mg/mL 00PS_Suc | 2 mg/mL 04PS_Suc | 2 mg/mL 16PS_Suc | 50 mg/mL 16PS_Suc | 2 mg/mL 04PS_Tre | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| 25 °C | 40 °C | 25 °C | 40 °C | 25 °C | 40 °C | 25 °C | 40 °C | 25 °C | 40 °C | |
| RN | - | - | - | - | X | X | - | - | - | - |
| AN1.5 | - | - | - | - | X | X | - | - | - | - |
| AN3.0 | - | - | - | - | X | X | - | - | - | - |
| QN | - | - | - | - | X | X | - | - | - | - |
| CN | - | - | - | X | X | X | - | - | - | - |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Groël, S.; Menzen, T.; Winter, G. Prediction of Unwanted Crystallization of Freeze-Dried Protein Formulations Using α-Relaxation Measurements. Pharmaceutics 2023, 15, 703. https://doi.org/10.3390/pharmaceutics15020703
Groël S, Menzen T, Winter G. Prediction of Unwanted Crystallization of Freeze-Dried Protein Formulations Using α-Relaxation Measurements. Pharmaceutics. 2023; 15(2):703. https://doi.org/10.3390/pharmaceutics15020703
Chicago/Turabian StyleGroël, Sebastian, Tim Menzen, and Gerhard Winter. 2023. "Prediction of Unwanted Crystallization of Freeze-Dried Protein Formulations Using α-Relaxation Measurements" Pharmaceutics 15, no. 2: 703. https://doi.org/10.3390/pharmaceutics15020703
APA StyleGroël, S., Menzen, T., & Winter, G. (2023). Prediction of Unwanted Crystallization of Freeze-Dried Protein Formulations Using α-Relaxation Measurements. Pharmaceutics, 15(2), 703. https://doi.org/10.3390/pharmaceutics15020703






