Evaluation of a Medical Grade Thermoplastic Polyurethane for the Manufacture of an Implantable Medical Device: The Impact of FDM 3D-Printing and Gamma Sterilization
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Samples Preparation
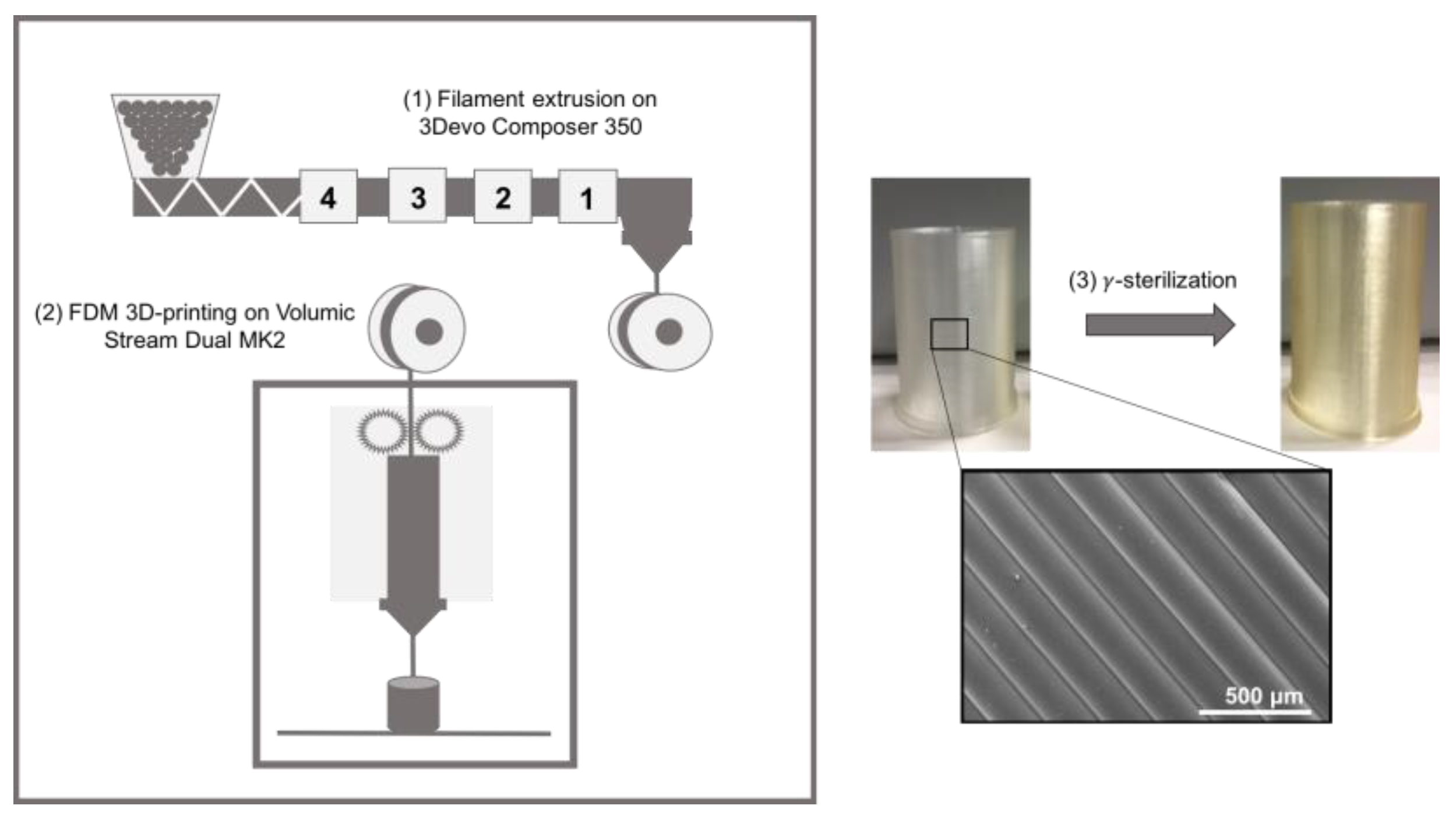
2.2.1. 3D-Printing (FDM)
2.2.2. Sterilization
2.3. Samples Characterization
2.3.1. Water Contact Angle Measurement (WCA)
2.3.2. Infrared Spectroscopy (ATR-FTIR)
2.3.3. Differential Scanning Calorimetry (DSC)
2.3.4. Size Exclusion Chromatography (SEC)
2.3.5. Tensile Tests
2.4. Biocompatibility Assessment
2.4.1. Cytotoxicity
2.4.2. Hemocompatibility Analysis
2.4.3. Hemolysis
2.4.4. Blood Cells Adhesion
2.5. Statistical Analysis
3. Results
3.1. Impact of FDM Manufacturing Process and Sterilization on TPU
3.2. Surface Properties of Sterilized 3DP Samples
3.3. Biological Properties of Sterilized 3DP Samples
3.3.1. Cytotoxicity
3.3.2. Hemocompatibility
Hemolysis
Blood Cells Adhesion
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lee, S.J.; Jo, H.H.; Lim, K.S.; Lim, D.; Lee, S.; Lee, J.H.; Kim, W.D.; Jeong, M.H.; Lim, J.Y.; Kwon, I.K.; et al. Heparin coating on 3D printed poly (l-lactic acid) biodegradable cardiovascular stent via mild surface modification approach for coronary artery implantation. Chem. Eng. J. 2019, 378, 122116. [Google Scholar] [CrossRef]
- Park, S.A.; Lee, S.J.; Lim, K.S.; Bae, I.H.; Lee, J.H.; Kim, W.D.; Jetong, M.H.; Palrk, J.-K. In vivo evaluation and characterization of a bio-absorbable drug-coated stent fabricated using a 3D-printing system. Mater. Lett. 2015, 141, 355–358. [Google Scholar] [CrossRef]
- Jiang, X.; Wu, S.; Kuss, M.; Kong, Y.; Shi, W.; Streubel, P.N.; Li, T.; Duan, B. 3D printing of multilayered scaffolds for rotator cuff tendon regeneration. Bioact. Mater. 2020, 5, 636–643. [Google Scholar] [CrossRef]
- Nagrath, M.; Sikora, A.; Graca, J.; Chinnici, J.L.; Rahman, S.U.; Reddy, S.G.; Ponnusamy, S.; Maddi, A.; Arany, P.R. Functionalized prosthetic interfaces using 3D printing: Generating infection-neutralizing prosthesis in dentistry. Mater. Today Commun. 2018, 15, 114–119. [Google Scholar] [CrossRef]
- Gradwohl, M.; Chai, F.; Payen, J.; Guerreschi, P.; Marchetti, P.; Blanchemain, N. Effects of Two Melt Extrusion Based Additive Manufacturing Technologies and Common Sterilization Methods on the Properties of a Medical Grade PLGA Copolymer. Polymers 2021, 13, 572. [Google Scholar] [CrossRef] [PubMed]
- Haleem, A.; Javaid, M.; Khan, R.H.; Suman, R. 3D printing applications in bone tissue engineering. J. Clin. Orthop. Trauma 2020, 11, S118–S124. [Google Scholar] [CrossRef] [PubMed]
- Jiao, Z.; Luo, B.; Xiang, S.; Ma, H.; Yu, Y.; Yang, W. 3D printing of HA/PCL composite tissue engineering scaffolds. Advanced Ind. Eng. Polym. Res. 2019, 2, 196–202. [Google Scholar] [CrossRef]
- Lal, H.; Patralekh, M.K. 3D printing and its applications in orthopaedic trauma: A technological marvel. J. Clin. Orthop. Trauma 2018, 9, 260–268. [Google Scholar] [CrossRef]
- Kang, J.; Zhang, J.; Zheng, J.; Wang, L.; Li, D.; Liu, S. 3D-printed PEEK implant for mandibular defects repair—A new method. J. Mech. Behav. Biomed. Mater. 2021, 116, 104335. [Google Scholar] [CrossRef]
- Oladapo, B.I.; Ismail, S.O.; Bowoto, O.K.; Omigbodun, F.T.; Olawumi, M.A.; Muhammad, M.A. Lattice design and 3D-printing of PEEK with Ca10(OH)(PO4)3 and in-vitro bio-composite for bone implant. Int. J. Biol. Macromol. 2020, 165, 50–62. [Google Scholar] [CrossRef]
- Oladapo, B.I.; Zahedi, S.A.; Ismail, S.O. Assessing 3D printing of Poly(ether-ether-ketone) and cellular cHAp to increase biointerfaces as a biomedical material. Colloids Surf. B Biointerfaces 2021, 203, 111726. [Google Scholar] [CrossRef] [PubMed]
- Verstraete, G.; Samaro, A.; Grymonpré, W.; Vanhoorne, V.; Van Snick, B.; Boone, M.N.; Hellemans, T.; Van Hoorebeke, L.; Remon, J.P.; Vervaet, C. 3D printing of high drug loaded dosage forms using thermoplastic polyurethanes. Int. J. Pharm. 2018, 536, 318–325. [Google Scholar] [CrossRef] [PubMed]
- Rahmatabadi, D.; Ghasemi, I.; Baniassadi, M.; Abrinia, K.; Baghani, M. 3D printing of PLA-TPU with different component ratios: Fracture toughness, mechanical properties, and morphology. J. Mater. Res. Technol. 2022, 21, 3970–3981. [Google Scholar] [CrossRef]
- Rahmatabadi, D.; Aberoumand, M.; Soltanmohammadi, H.; Soleyman, E.; Ghasemi, I.; Baniassadi, M.; Abrinia, K.; Bodaghi, M.; Baghani, M. 4D Printing Encapsulated Polycaprolactone—Thermoplastic Polyurethane with High Shape Memory Performances. Adv. Eng. Mater. 2022, 2201309. [Google Scholar] [CrossRef]
- Petrović, Z.S.; Ferguson, J. Polyurethane elastomers. Prog. Polym. Sci. 1991, 16, 695–836. [Google Scholar] [CrossRef]
- Takahara, A.; Okkema, A.Z.; Cooper, S.L.; Coury, A.J. Effect of surface hydrophilicity on ex vivo blood compatibility of segmented polyurethanes. Biomaterials 1991, 12, 324–334. [Google Scholar] [CrossRef]
- Kanyanta, V.; Ivankovic, A. Mechanical characterisation of polyurethane elastomer for biomedical applications. J. Mech. Behav. Biomed. Mater. 2010, 3, 51–62. [Google Scholar] [CrossRef]
- Boffito, M.; Sartori, S.; Mattu, C.; Ciardelli, G. 13-Polyurethanes for cardiac applications. In Advances in Polyurethane Biomaterials; Cooper, S.L., Guan, J., Eds.; Woodhead Publishing: Sawston, UK, 2016; pp. 387–416. [Google Scholar]
- Ghanbari, H.; Viatge, H.; Kidane, A.G.; Burriesci, G.; Tavakoli, M.; Seifalian, A.M. Polymeric heart valves: New materials, emerging hopes. Trends Biotechnol. 2009, 27, 359–367. [Google Scholar] [CrossRef]
- Scholz, P.; Wachtendorf, V.; Panne, U.; Weidner, S.M. Degradation of MDI-based polyether and polyester-polyurethanes in various environments—Effects on molecular mass and crosslinking. Polym. Test. 2019, 77, 105881. [Google Scholar] [CrossRef]
- Miller, A.T.; Safranski, D.L.; Smith, K.E.; Sycks, D.G.; Guldberg, R.E.; Gall, K. Fatigue of injection molded and 3D printed polycarbonate urethane in solution. Polymer 2017, 108, 121–134. [Google Scholar] [CrossRef]
- Wady, P.; Wasilewski, A.; Brock, L.; Edge, R.; Baidak, A.; McBride, C.; Leay, L.; Griffiths, A.; Vallés, C. Effect of ionising radiation on the mechanical and structural properties of 3D printed plastics. Addit. Manuf. 2020, 31, 100907. [Google Scholar] [CrossRef]
- Gorna, K.; Gogolewski, S. The effect of gamma radiation on molecular stability and mechanical properties of biodegradable polyurethanes for medical applications. Polym. Degrad. Stab. 2003, 79, 465–474. [Google Scholar] [CrossRef]
- Simmons, A.; Hyvarinen, J.; Poole-Warren, L. The effect of sterilisation on a poly(dimethylsiloxane)/poly(hexamethylene oxide) mixed macrodiol-based polyurethane elastomer. Biomaterials 2006, 27, 4484–4497. [Google Scholar] [CrossRef] [PubMed]
- Nouman, M.; Saunier, J.; Jubeli, E.; Marlière, C.; Yagoubi, N. Impact of sterilization and oxidation processes on the additive blooming observed on the surface of polyurethane. Eur. Polym. J. 2017, 90, 37–53. [Google Scholar] [CrossRef]
- Mrad, O.; Saunier, J.; Aymes Chodur, C.; Rosilio, V.; Agnely, F.; Aubert, P.; Vigneron, J.; Etcheberry, A.; Yagoubi, N. A comparison of plasma and electron beam-sterilization of PU catheters. Radiat. Phys. Chem. 2010, 79, 93–103. [Google Scholar] [CrossRef]
- Abraham, G.A.; Frontini, P.M.; Cuadrado, T.R. Physical and mechanical behavior of sterilized biomedical segmented polyurethanes. J. Appl. Polym. Sci. 1997, 65, 1193–1203. [Google Scholar] [CrossRef]
- Murray, K.A.; Kennedy, J.E.; McEvoy, B.; Vrain, O.; Ryan, D.; Cowman, R.; Higginbotham, C.L. The influence of electron beam irradiation conducted in air on the thermal, chemical, structural and surface properties of medical grade polyurethane. Eur. Polym. J. 2013, 49, 1782–1795. [Google Scholar] [CrossRef]
- Wetzel, C.; Schönfelder, J.; Schwarz, W.; Funk, R.H.W. Surface modification of polyurethane and silicone for therapeutic medical technics by means of electron beam. Surf. Coat. Technol. 2010, 205, 1618–1623. [Google Scholar] [CrossRef]
- Goodman, S.L. Sheep; pig; and human platelet–material interactions with model cardiovascular biomaterials. J. Biomed. Mater. Res. 1999, 45, 240–250. [Google Scholar] [CrossRef]
- Meijs, G.F.; McCarthy, S.J.; Rizzardo, E.; Chen, Y.C.; Chatelier, R.C.; Brandwood, A.; Schindhelm, K. Degradation of medical-grade polyurethane elastomers: The effect of hydrogen peroxide in vitro. J. Biomed. Mater. Res. 1993, 27, 345–356. [Google Scholar] [CrossRef]
- ISO 11137-2:2013. Available online: https://www.iso.org/cms/render/live/fr/sites/isoorg/contents/data/standard/06/24/62442.html (accessed on 13 May 2022).
- Wilhelm, C.; Rivaton, A.; Gardette, J.L. Infrared analysis of the photochemical behaviour of segmented polyurethanes: 3. Aromatic diisocyanate based polymers. Polymer 1998, 39, 1223–1232. [Google Scholar] [CrossRef]
- Jiang, S.; Yuan, C.; Guo, Z.; Bai, X. Effect of crosslink on tribological performance of polyurethane bearing material. Tribol. Int. 2019, 136, 276–284. [Google Scholar] [CrossRef]
- Bernard, M.; Jubeli, E.; Bakar, J.; Tortolano, L.; Saunier, J.; Yagoubi, N. Biocompatibility assessment of cyclic olefin copolymers: Impact of two additives on cytotoxicity, oxidative stress, inflammatory reactions, and hemocompatibility. J. Biomed. Mater. Res. Part A 2017, 105, 3333–3349. [Google Scholar] [CrossRef] [PubMed]





| Printing Parameters | Data |
|---|---|
| Flow (%) | 130 |
| Printing layer height (mm) | 0.15 |
| Nozzle Diameter (mm) | 0.4 |
| Nozzle temperature (°C) | 220 |
| Printing bed temperature (°C) | 50 |
| Printing speed (mm/min) | 1000 |
| Samples | Tg1 (°C) | Tg2 (°C) | Tm (°C) | ΔHm (J/g) | Tc (°C) | ΔHc (J/g) |
|---|---|---|---|---|---|---|
| Pellets | −50 ± 2 | n.d. | 164 ± 1 | 13.8 ± 0.5 | 84 ± 1 | 10.2 ± 0.4 |
| Filament | −45 ± 1 | 64 ± 3 | 163 ± 1 | 11.2 ± 0.5 | 85 ± 1 | 9.0 ± 0.5 |
| FDM printing | −48 ± 3 | 52 ± 2 | 163 ± 3 | 9.9 ± 3.4 | 88 ± 3 | 10.8 ± 0.7 |
| Ɣ-40 kGy | −47 ± 4 | 61 ± 3 | 165 ± 1 | 7.3 ± 3.2 | 86.0 ± 0.4 | 11.0 ± 0.5 |
| Samples | E (MPa) | σb (MPa) | εb (%) | |||
|---|---|---|---|---|---|---|
| Para | Perp | Para | Perp | Para | Perp | |
| FDM printing | 14 ± 1 | 14.0 ± 0.1 | 13 ± 2 | 8.0 ± 0.2 | 700 ± 20 | 380 ± 20 |
| Ɣ-40 kGy | 15.0 ± 0.5 | 14 ± 2 | 15 ± 1 | 8.0 ± 0.4 | 720 ± 70 | 440 ± 20 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
M’Bengue, M.-S.; Mesnard, T.; Chai, F.; Maton, M.; Gaucher, V.; Tabary, N.; García-Fernandez, M.-J.; Sobocinski, J.; Martel, B.; Blanchemain, N. Evaluation of a Medical Grade Thermoplastic Polyurethane for the Manufacture of an Implantable Medical Device: The Impact of FDM 3D-Printing and Gamma Sterilization. Pharmaceutics 2023, 15, 456. https://doi.org/10.3390/pharmaceutics15020456
M’Bengue M-S, Mesnard T, Chai F, Maton M, Gaucher V, Tabary N, García-Fernandez M-J, Sobocinski J, Martel B, Blanchemain N. Evaluation of a Medical Grade Thermoplastic Polyurethane for the Manufacture of an Implantable Medical Device: The Impact of FDM 3D-Printing and Gamma Sterilization. Pharmaceutics. 2023; 15(2):456. https://doi.org/10.3390/pharmaceutics15020456
Chicago/Turabian StyleM’Bengue, Marie-Stella, Thomas Mesnard, Feng Chai, Mickaël Maton, Valérie Gaucher, Nicolas Tabary, Maria-José García-Fernandez, Jonathan Sobocinski, Bernard Martel, and Nicolas Blanchemain. 2023. "Evaluation of a Medical Grade Thermoplastic Polyurethane for the Manufacture of an Implantable Medical Device: The Impact of FDM 3D-Printing and Gamma Sterilization" Pharmaceutics 15, no. 2: 456. https://doi.org/10.3390/pharmaceutics15020456
APA StyleM’Bengue, M.-S., Mesnard, T., Chai, F., Maton, M., Gaucher, V., Tabary, N., García-Fernandez, M.-J., Sobocinski, J., Martel, B., & Blanchemain, N. (2023). Evaluation of a Medical Grade Thermoplastic Polyurethane for the Manufacture of an Implantable Medical Device: The Impact of FDM 3D-Printing and Gamma Sterilization. Pharmaceutics, 15(2), 456. https://doi.org/10.3390/pharmaceutics15020456






