Photoinduced Antibacterial Activity and Cytotoxicity of CdS Stabilized on Mesoporous Aluminosilicates and Silicates
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. MCM-41 and MCM-41/HNTs Synthesis
2.3. SBA-15 Synthesis
2.4. Modification of HNTs and SBA-15
2.5. Synthesis of CdS
2.6. Samples Characterization
2.7. Antibacterial Assay
2.8. Cell Viability Tests
3. Results
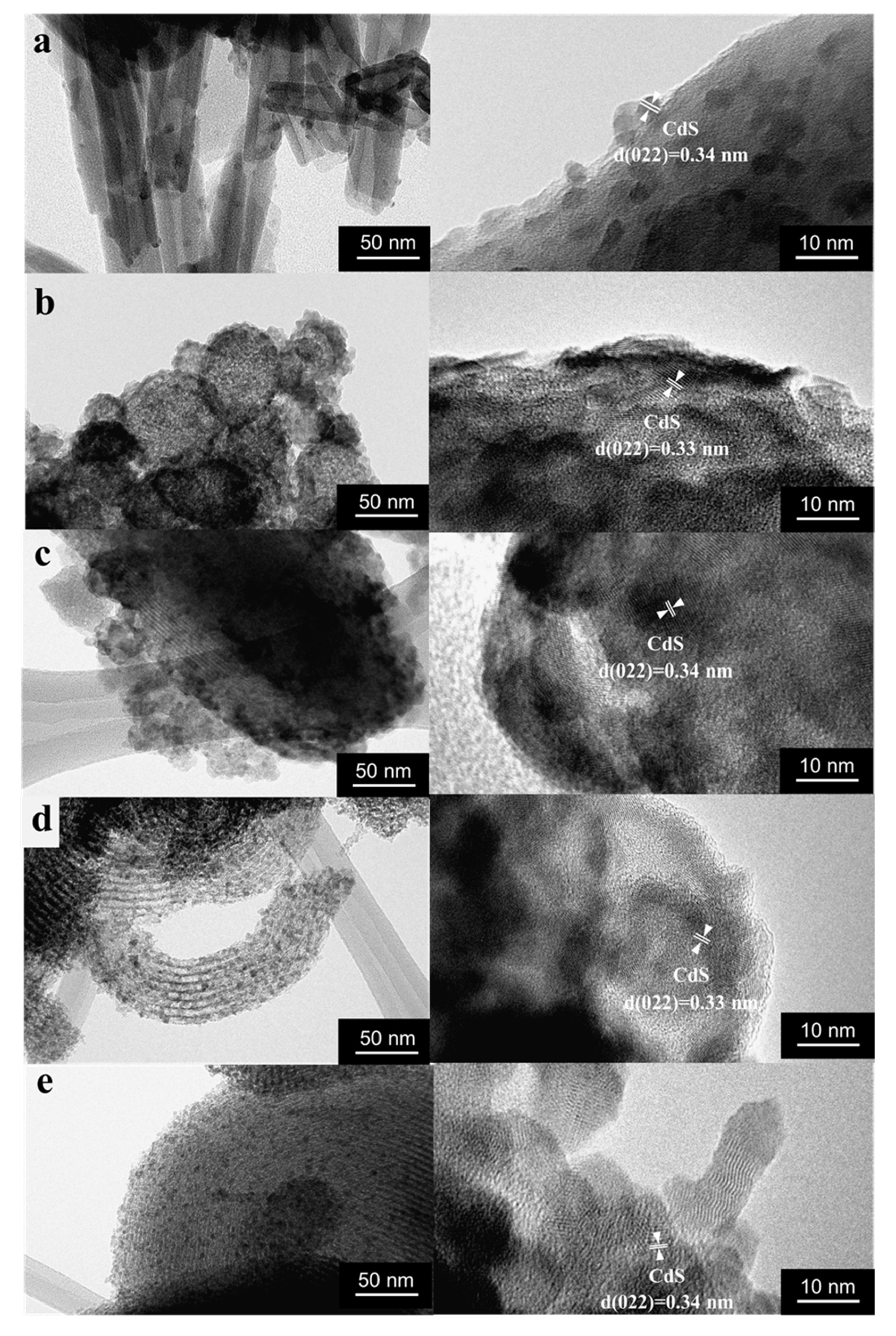
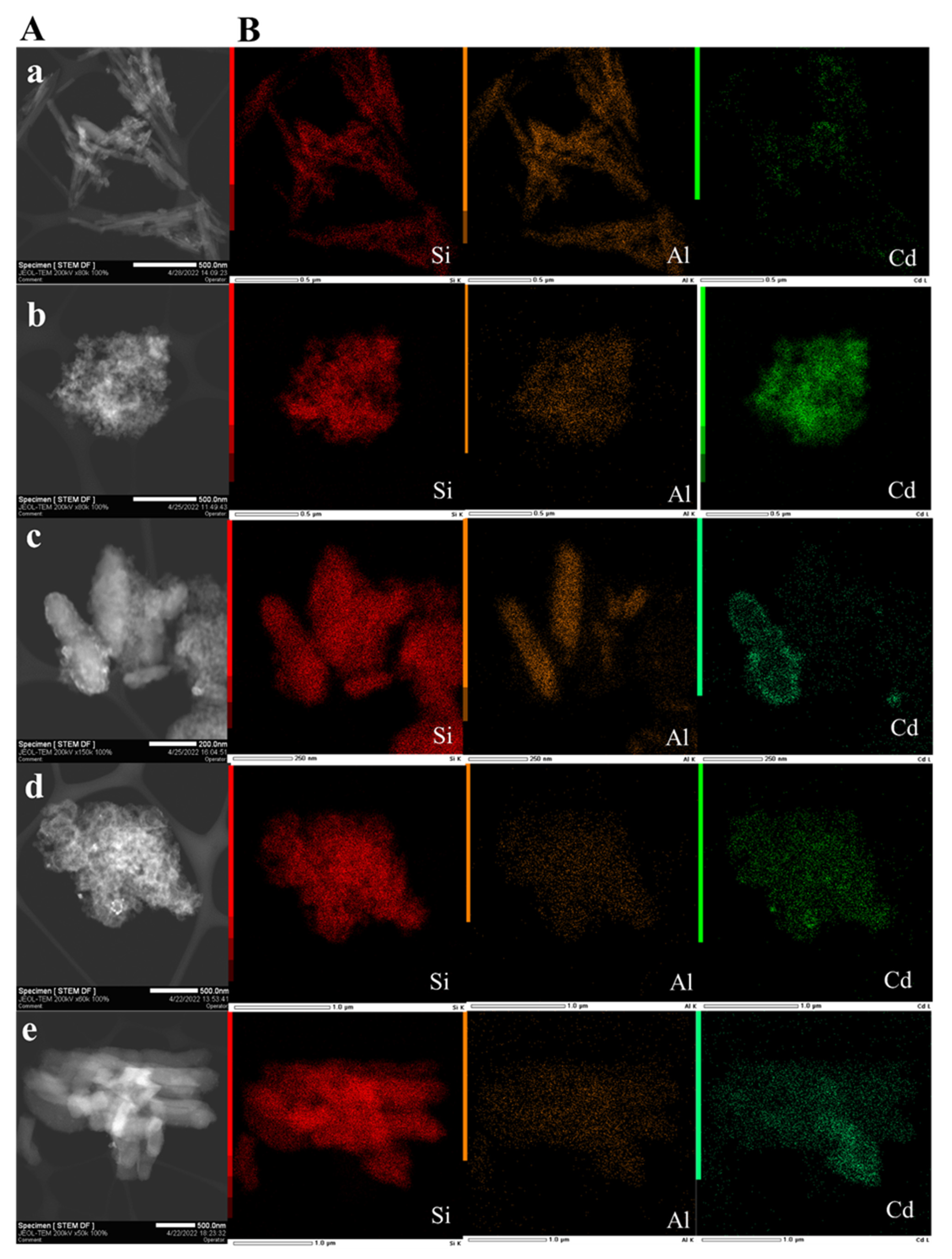
3.1. Structure and Morphology
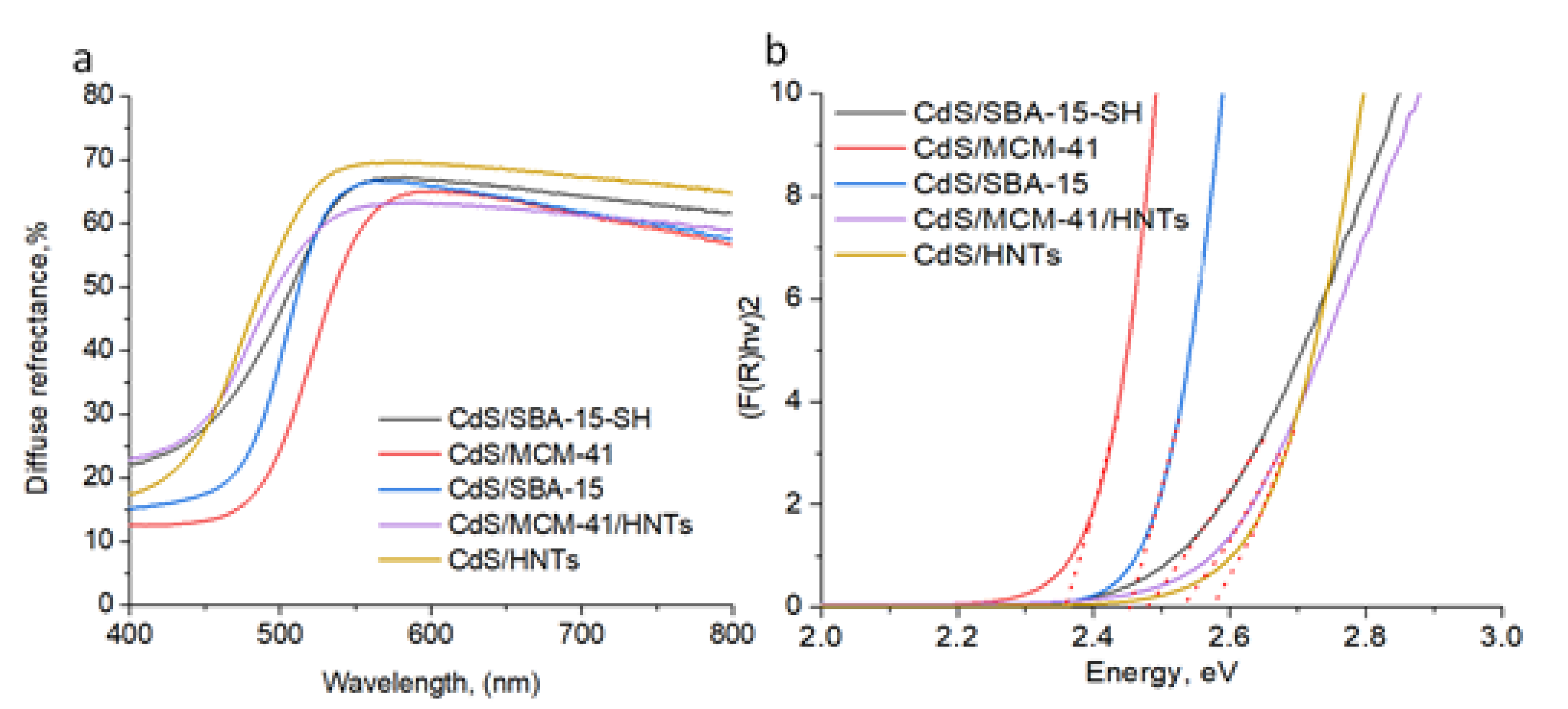
3.2. Spectral Characteristics
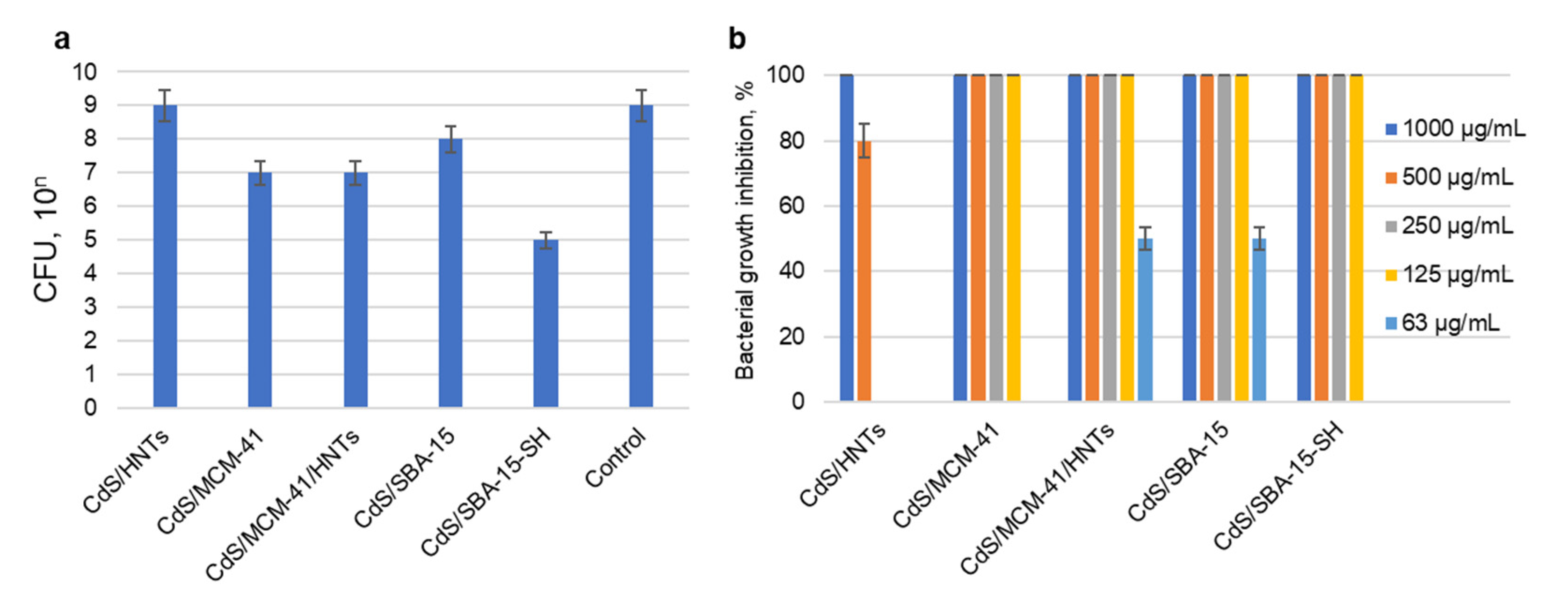
3.3. Antibacterial Activity Againts S. aureus
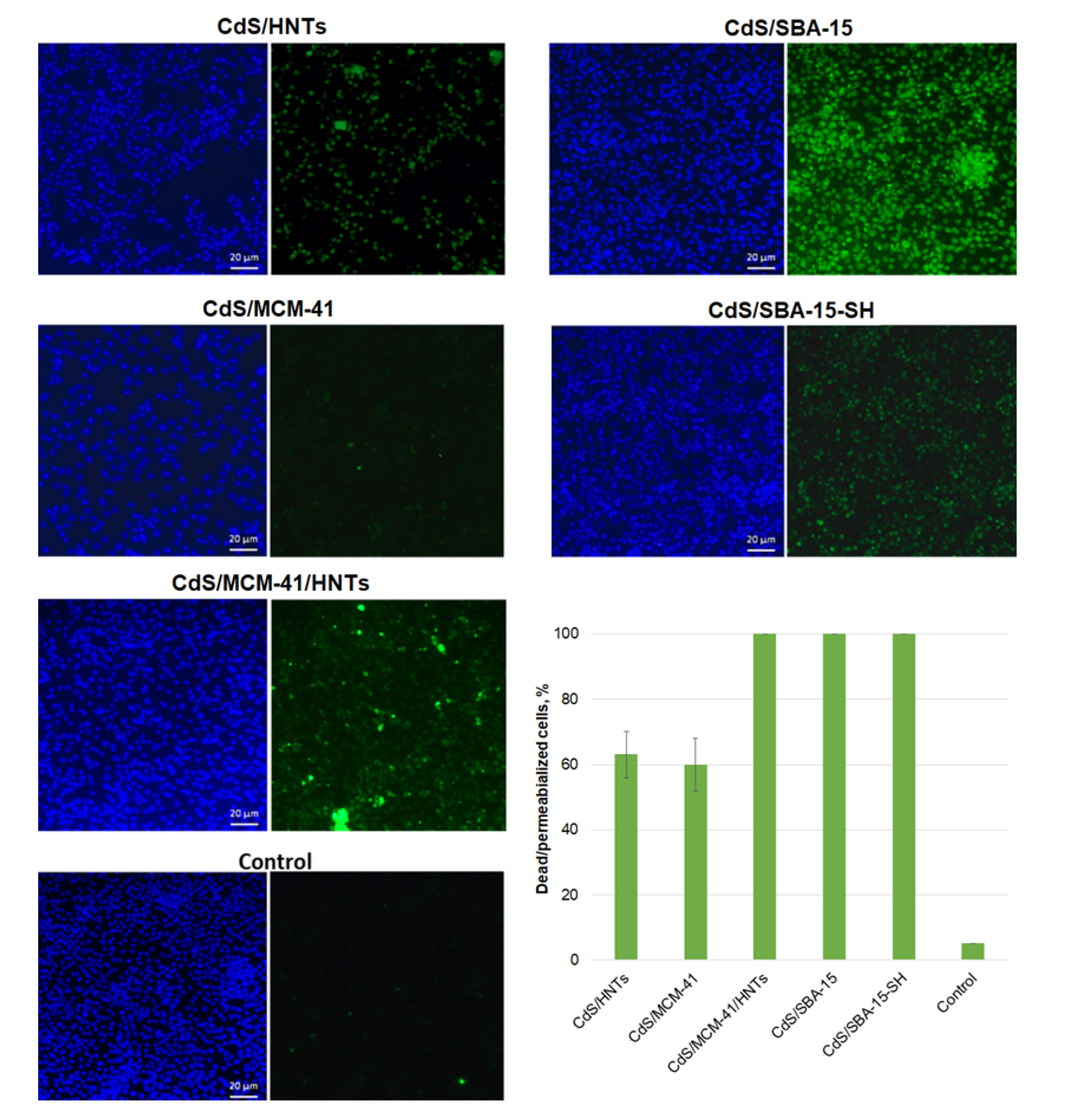
3.4. Cell Viability
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Fair, R.J.; Tor, Y. Antibiotics and bacterial resistance in the 21st century. Perspect. Med. Chem. 2014, 6, 25–64. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhao, J.; Huang, S.; Ravisankar, P.; Zhu, H. Two-dimensional nanomaterials for photoinduced antibacterial applications. ACS Appl. Bio Mater. 2020, 3, 8188–8210. [Google Scholar] [CrossRef] [PubMed]
- Dizaj, S.M.; Lotfipour, F.; Barzegar-Jalali, M.; Zarrintan, M.H.; Adibkia, K. Antimicrobial activity of the metals and metal oxide nanoparticles. Mater. Sci. Eng. C 2014, 44, 278–284. [Google Scholar] [CrossRef] [PubMed]
- Lam, S.J.; Wong, E.H.H.; Boyer, C.; Qiao, G.G. Antimicrobial polymeric nanoparticles. Prog. Polym. Sci. 2018, 76, 40–64. [Google Scholar] [CrossRef]
- Aroso, R.T.; Schaberle, F.A.; Arnaut, L.G.; Pereira, M.M. Photodynamic disinfection and its role in controlling infectious diseases. Photochem. Photobiol. Sci. 2021, 20, 1497–1545. [Google Scholar] [CrossRef] [PubMed]
- Bing, W.; Chen, Z.; Sun, H.; Shi, P.; Gao, N.; Ren, J.; Qu, X. Visible-light-driven enhanced antibacterial and biofilm elimination activity of graphitic carbon nitride by embedded Ag nanoparticles. Nano Res. 2015, 8, 1648–1658. [Google Scholar] [CrossRef]
- Dai, X.; Zhao, Y.; Yu, Y.; Chen, X.; Wei, X.; Zhang, X.; Li, C. Single continuous near-infrared laser-triggered photodynamic and photothermal ablation of antibiotic-resistant bacteria using effective targeted copper sulfide nanoclusters. ACS Appl. Mater. Interfaces 2017, 9, 30470–30479. [Google Scholar] [CrossRef]
- Han, H.; Yang, J.; Li, X.; Qi, Y.; Yang, Z.; Han, Z.; Jiang, Y.; Stenzel, M.; Li, H.; Yin, Y.; et al. Shining light on transition metal sulfides: New choices as highly efficient antibacterial agents. Nano Res. 2021, 14, 2512–2534. [Google Scholar] [CrossRef]
- Dey, P.C.; Ingti, B.; Bhattacharjee, A.; Choudhury, M.D.; Das, R.; Nath, S.S. Enhancement of antibacterial activity of synthesized ligand-free CdS nanocrystals due to silver doping. J. Basic Microbiol. 2021, 61, 27–36. [Google Scholar] [CrossRef]
- Shalabayev, Z.; Baláž, M.; Khan, N.; Nurlan, Y.; Augustyniak, A.; Daneu, N.; Tatykayev, B.; Dutková, E.; Burashev, G.; Casas-Luna, M.; et al. Sustainable synthesis of cadmium sulfide, with applicability in photocatalysis, hydrogen production, and as an antibacterial agent, using two mechanochemical protocols. Nanomaterials 2022, 12, 1250. [Google Scholar] [CrossRef]
- Stavitskaya, A.V.; Kozlova, E.A.; Kurenkova, A.Y.; Glotov, A.P.; Selischev, D.S.; Ivanov, E.V.; Kozlov, D.V.; Vinokurov, V.A.; Fakhrullin, R.F.; Lvov, Y.M. Ru/CdS quantum dots templated on clay nanotubes as visible-light-active photocatalysts: Optimization of S/Cd ratio and Ru content. Chem. Eur. J. 2020, 26, 13085–13092. [Google Scholar] [CrossRef] [PubMed]
- Vinokurov, V.A.; Stavitskaya, A.V.; Ivanov, E.V.; Gushchin, P.A.; Kozlov, D.V.; Kurenkova, A.Y.; Kolinko, P.A.; Kozlova, E.A.; Lvov, Y.M. Halloysite nanoclay based CdS formulations with high catalytic activity in hydrogen evolution reaction under visible light irradiation. ACS Sustain. Chem. Eng. 2017, 5, 11316–11323. [Google Scholar] [CrossRef]
- Nasir, J.A.; Rehman, Z.U.; Shah, S.N.A.; Khan, A.; Butler, I.S.; Catlow, C.R.A. Recent developments and perspectives in CdS-based photocatalysts for water splitting. J. Mater. Chem. A 2020, 8, 20752–20780. [Google Scholar] [CrossRef]
- Li, Y.; Zhang, W.; Niu, J.; Chen, Y. Mechanism of photogenerated reactive oxygen species and correlation with the antibacterial properties of engineered metal-oxide nanoparticles. ACS Nano 2012, 6, 5164–5173. [Google Scholar] [CrossRef]
- Singh, I.R.; Chettri, U.; Maity, P.; Ghosh, A.K.; Joshi, S.R.; Mitra, S. Modulated antimicrobial activity and drug-protein interaction ability of zinc oxide and cadmium sulfide nanoparticles: Effect of doping with few first-row transition metals. J. Clust. Sci. 2022, 1–13. [Google Scholar] [CrossRef]
- Fakhrullina, G.I.; Akhatova, F.S.; Lvov, Y.M.; Fakhrullin, R.F. Toxicity of halloysite clay nanotubes in vivo: A Caenorhabditis elegans study. Environ. Sci. Nano 2015, 2, 54–59. [Google Scholar] [CrossRef]
- Long, Z.; Wu, Y.-P.; Gao, H.-Y.; Zhang, J.; Ou, X.; He, R.-R.; Liu, M. In vitro and in vivo toxicity evaluation of halloysite nanotubes. J. Mater. Chem. B 2018, 6, 7204–7216. [Google Scholar] [CrossRef]
- Glotov, A.; Levshakov, N.; Stavitskaya, A.; Artemova, M.; Gushchin, P.; Ivanov, E.; Vinokurov, V.; Lvov, Y. Templated self-assembly of ordered mesoporous silica on clay nanotubes. Chem. Commun. 2019, 55, 5507–5510. [Google Scholar] [CrossRef]
- Bel Haj Mohamed, N.; Haouari, M.; Zaaboub, Z.; Nafoutti, M.; Hassen, F.; Maaref, H.; Ben Ouada, H. Time resolved and temperature dependence of the radiative properties of thiol-capped CdS nanoparticles films. J. Nanopart. Res. 2014, 16, 2242. [Google Scholar] [CrossRef] [Green Version]
- Jin, Q.; Qu, F.; Jiang, J.; Dong, Y.; Guo, W.; Lin, H. A pH-sensitive controlled dual-drug release from meso-macroporous silica/multilayer-polyelectrolytes coated SBA-15 composites. J. Sol-Gel Sci. Technol. 2013, 66, 466–471. [Google Scholar] [CrossRef]
- Stavitskaya, A.; Glotov, A.; Pouresmaeil, F.; Potapenko, K.; Sitmukhanova, E.; Mazurova, K.; Ivanov, E.; Kozlova, E.; Vinokurov, V.; Lvov, Y. CdS quantum dots in hierarchical mesoporous silica templated on clay nanotubes: Implications for photocatalytic hydrogen production. ACS Appl. Nano Mater. 2022, 5, 605–614. [Google Scholar] [CrossRef]
- Boesewetter, D.E.; Collier, J.L.; Kim, A.M.; Riley, M.R. Alterations of A549 lung cell gene expression in response to biochemical toxins. Cell Biol. Toxicol. 2006, 22, 101–118. [Google Scholar] [CrossRef]
- Shi, Y.; Wang, Y.; Huang, W.; Wang, Y.; Wang, R.; Yuan, Y. Integration of metabolomics and transcriptomics to reveal metabolic characteristics and key targets associated with cisplatin resistance in nonsmall cell lung cancer. J. Proteome Res. 2019, 18, 3259–3267. [Google Scholar] [CrossRef]
- Mahajan, S.D.; Law, W.-C.; Aalinkeel, R.; Reynolds, J.; Nair, B.B.; Yong, K.-T.; Roy, I.; Prasad, P.N.; Schwartz, S.A. Nanoparticle-mediated targeted delivery of antiretrovirals to the brain. Meth. Enzymol. 2012, 509, 41–60. [Google Scholar] [CrossRef]
- Stavitskaya, A.V.; Novikov, A.A.; Kotelev, M.S.; Kopitsyn, D.S.; Rozhina, E.V.; Ishmukhametov, I.R.; Fakhrullin, R.F.; Ivanov, E.V.; Lvov, Y.M.; Vinokurov, V.A. Fluorescence and cytotoxicity of cadmium sulfide quantum dots stabilized on clay nanotubes. Nanomaterials 2018, 8, 391. [Google Scholar] [CrossRef] [Green Version]
- Lunawat, P.S.; Kumar, R.; Gupta, N.M. Structure sensitivity of nano-structured CdS/SBA-15 containing Au and Pt co-catalysts for the photocatalytic splitting of water. Catal. Lett. 2008, 121, 226–233. [Google Scholar] [CrossRef]
- Kundu, M.; Khosravi, A.A.; Kulkarni, S.K.; Singh, P. Synthesis and study of organically capped ultra small clusters of cadmium sulphide. J. Mater. Sci. 1997, 32, 245–258. [Google Scholar] [CrossRef]
- Okamura, M.; Ebina, K.; Akimoto, S.; Yamazaki, I.; Uosaki, K. Luminescent properties of CdS nanoclusters dispersed in solution—effects of size and surface termination. J. Photochem. Photobiol. A 2006, 178, 156–161. [Google Scholar] [CrossRef] [Green Version]
- Derfus, A.M.; Chan, W.C.W.; Bhatia, S.N. Probing the cytotoxicity of semiconductor quantum dots. Nano Lett. 2004, 4, 11–18. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, L.; Nagesha, D.K.; Selvarasah, S.; Dokmeci, M.R.; Carrier, R.L. Toxicity of CdSe nanoparticles in Caco-2 cell cultures. J. Nanobiotechnol. 2008, 6, 11. [Google Scholar] [CrossRef] [Green Version]
- Rikans, L.E.; Yamano, T. Mechanisms of cadmium-mediated acute hepatotoxicity. J. Biochem. Mol. Toxicol. 2000, 14, 110–117. [Google Scholar] [CrossRef]
- López, E.; Figueroa, S.; Oset-Gasque, M.J.; González, M.P. Apoptosis and necrosis: Two distinct events induced by cadmium in cortical neurons in culture. Br. J. Pharmacol. 2003, 138, 901–911. [Google Scholar] [CrossRef] [PubMed]



| Sample | Cd Content, wt.% (ICP-EOS) | Zeta–Potential, mV | SBET, m2/g |
|---|---|---|---|
| CdS/HNTs | 4.1 | −20 | 67 |
| CdS/MCM-41 | 4.6 | −34 | - |
| CdS/MCM-41-HNTs | 4.3 | −50 | 746 |
| CdS/SBA-15 | 4.5 | −45 | 845 |
| CdS/SBA-15-SH | 4.7 | −48 | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stavitskaya, A.; Sitmukhanova, E.; Sayfutdinova, A.; Khusnetdenova, E.; Mazurova, K.; Cherednichenko, K.; Naumenko, E.; Fakhrullin, R. Photoinduced Antibacterial Activity and Cytotoxicity of CdS Stabilized on Mesoporous Aluminosilicates and Silicates. Pharmaceutics 2022, 14, 1309. https://doi.org/10.3390/pharmaceutics14071309
Stavitskaya A, Sitmukhanova E, Sayfutdinova A, Khusnetdenova E, Mazurova K, Cherednichenko K, Naumenko E, Fakhrullin R. Photoinduced Antibacterial Activity and Cytotoxicity of CdS Stabilized on Mesoporous Aluminosilicates and Silicates. Pharmaceutics. 2022; 14(7):1309. https://doi.org/10.3390/pharmaceutics14071309
Chicago/Turabian StyleStavitskaya, Anna, Eliza Sitmukhanova, Adeliya Sayfutdinova, Elnara Khusnetdenova, Kristina Mazurova, Kirill Cherednichenko, Ekaterina Naumenko, and Rawil Fakhrullin. 2022. "Photoinduced Antibacterial Activity and Cytotoxicity of CdS Stabilized on Mesoporous Aluminosilicates and Silicates" Pharmaceutics 14, no. 7: 1309. https://doi.org/10.3390/pharmaceutics14071309
APA StyleStavitskaya, A., Sitmukhanova, E., Sayfutdinova, A., Khusnetdenova, E., Mazurova, K., Cherednichenko, K., Naumenko, E., & Fakhrullin, R. (2022). Photoinduced Antibacterial Activity and Cytotoxicity of CdS Stabilized on Mesoporous Aluminosilicates and Silicates. Pharmaceutics, 14(7), 1309. https://doi.org/10.3390/pharmaceutics14071309








