An Optimized Terpene Profile for a New Medical Cannabis Oil
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals and Solvents
2.2. Plant Material and Extraction of Cannabis Oils
2.3. Sample Preparation for Cannabinoid Analysis
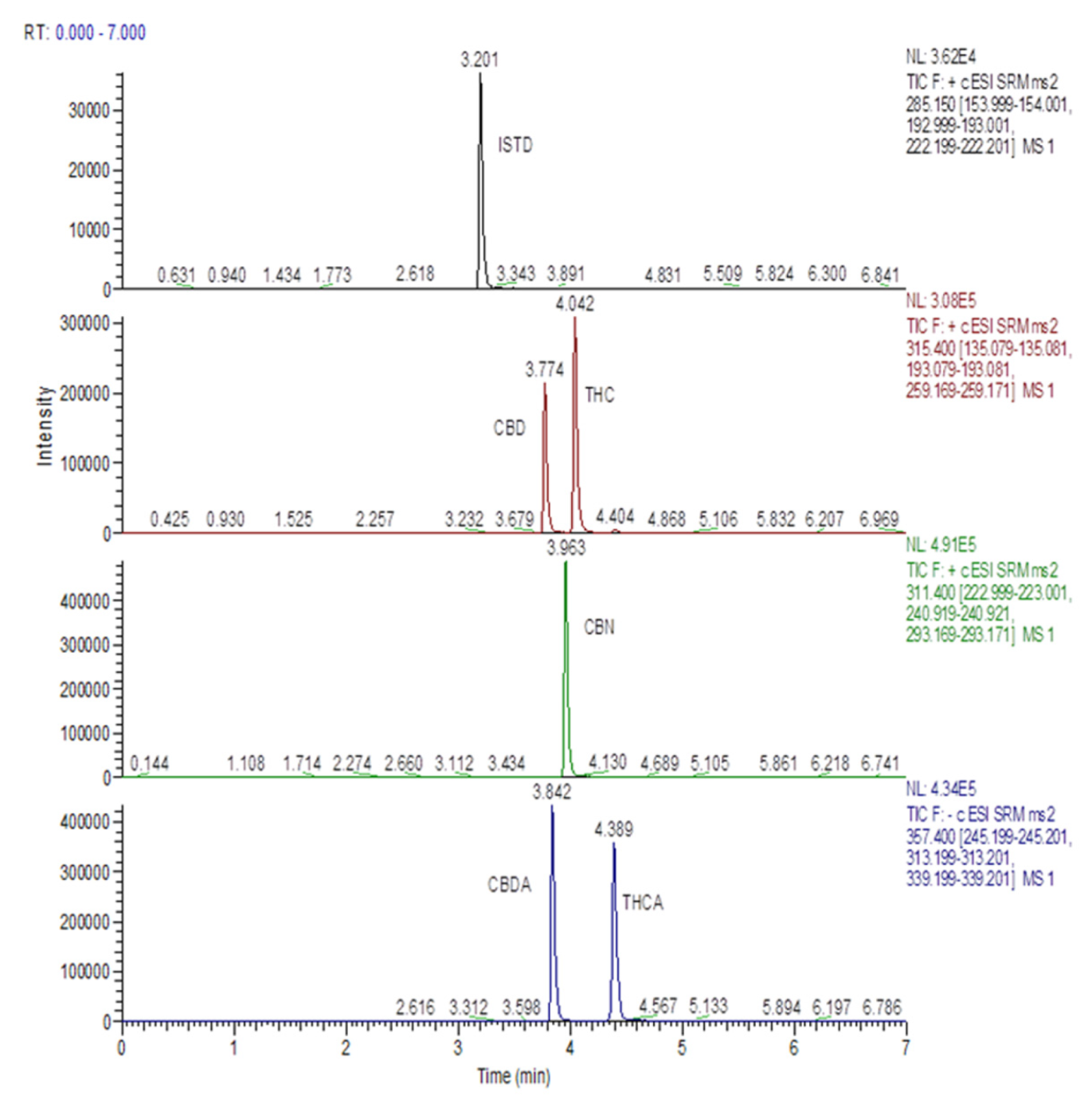
2.4. HPLC-MS/MS Analysis of Cannabinoids
2.5. Sample Preparation for Terpene Analysis
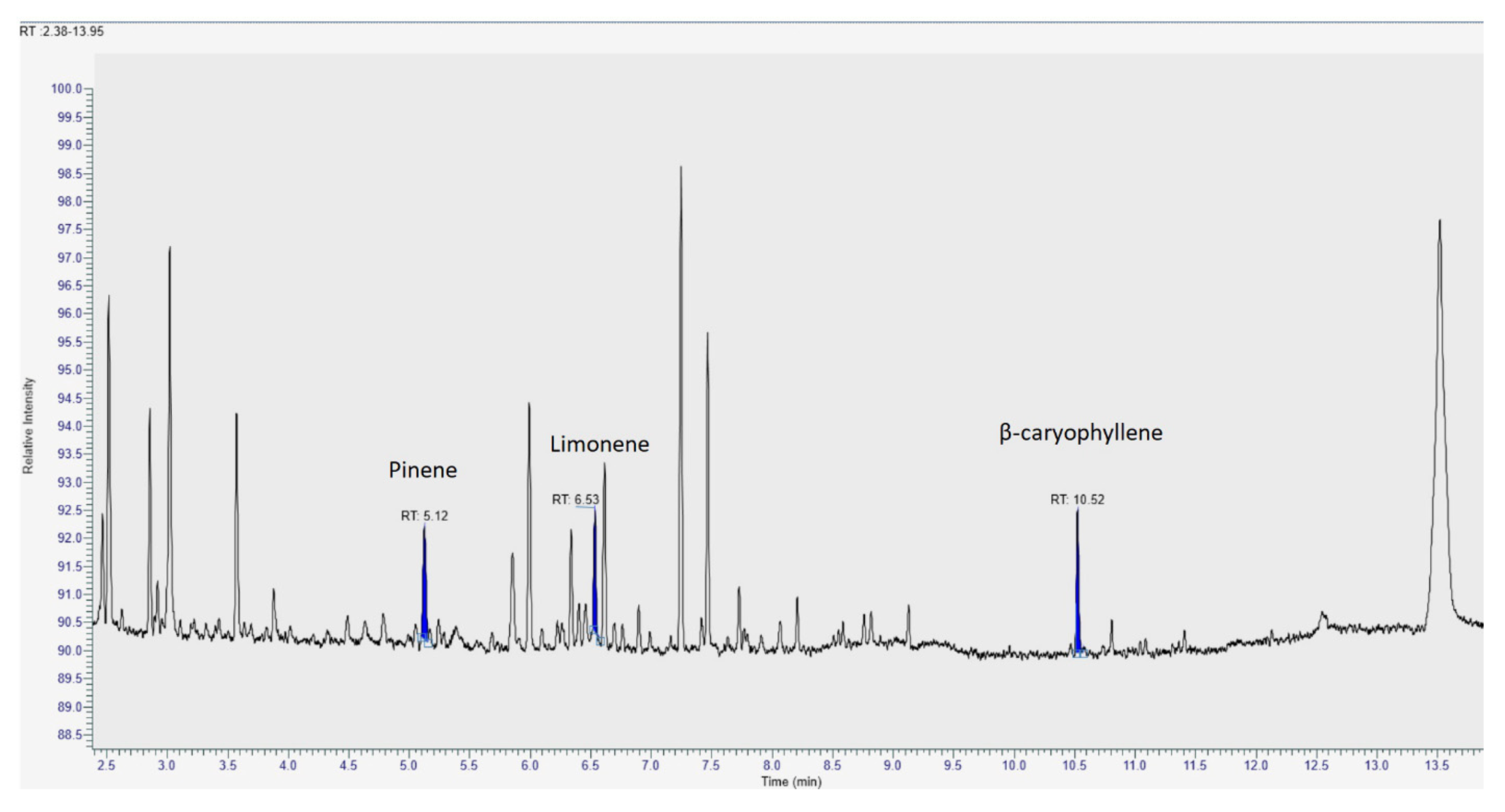
2.6. GC/MS Analysis of Terpenes
2.7. Statistical Analysis
3. Results
3.1. Extraction Methods for Medical Cannabis Oils
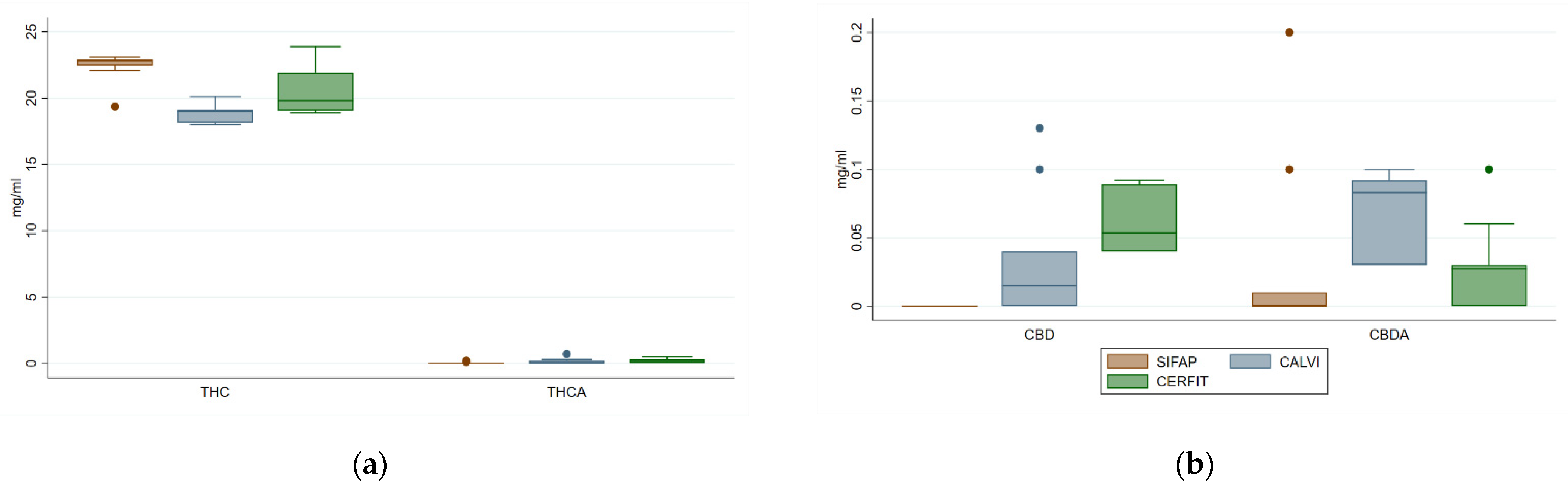
3.2. Cannabinoids in Medical Cannabis Oils
3.3. Terpenes in Medical Cannabis Oils
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Crocq, M.-A. History of Cannabis and the Endocannabinoid System. Dialogues Clin. Neurosci. 2020, 22, 223–228. [Google Scholar] [CrossRef] [PubMed]
- Heng, M.; McTague, M.F.; Lucas, R.C.; Harris, M.B.; Vrahas, M.S.; Weaver, M.J. Patient Perceptions of the Use of Medical Marijuana in the Treatment of Pain After Musculoskeletal Trauma: A Survey of Patients at 2 Trauma Centers in Massachusetts. J. Orthop. Trauma 2018, 32, e25–e30. [Google Scholar] [CrossRef] [PubMed]
- Kosiba, J.D.; Maisto, S.A.; Ditre, J.W. Patient-Reported Use of Medical Cannabis for Pain, Anxiety, and Depression Symptoms: Systematic Review and Meta-Analysis. Soc. Sci. Med. 2019, 233, 181–192. [Google Scholar] [CrossRef] [PubMed]
- Aran, A.; Harel, M.; Cassuto, H.; Polyansky, L.; Schnapp, A.; Wattad, N.; Shmueli, D.; Golan, D.; Castellanos, F.X. Cannabinoid Treatment for Autism: A Proof-of-Concept Randomized Trial. Mol. Autism 2021, 12, 6. [Google Scholar] [CrossRef]
- Uddin, M.S.; Mamun, A.A.; Sumsuzzman, D.M.; Ashraf, G.M.; Perveen, A.; Bungau, S.G.; Mousa, S.A.; El-Seedi, H.R.; Bin-Jumah, M.N.; Abdel-Daim, M.M. Emerging Promise of Cannabinoids for the Management of Pain and Associated Neuropathological Alterations in Alzheimer’s Disease. Front. Pharmacol. 2020, 11, 1097. [Google Scholar] [CrossRef]
- García-Gutiérrez, M.S.; Navarrete, F.; Gasparyan, A.; Austrich-Olivares, A.; Sala, F.; Manzanares, J. Cannabidiol: A Potential New Alternative for the Treatment of Anxiety, Depression, and Psychotic Disorders. Biomolecules 2020, 10, 1575. [Google Scholar] [CrossRef]
- Stella, B.; Baratta, F.; Della Pepa, C.; Arpicco, S.; Gastaldi, D.; Dosio, F. Cannabinoid Formulations and Delivery Systems: Current and Future Options to Treat Pain. Drugs 2021, 81, 1513–1557. [Google Scholar] [CrossRef]
- Filipiuc, L.E.; Ababei, D.C.; Alexa-Stratulat, T.; Pricope, C.V.; Bild, V.; Stefanescu, R.; Stanciu, G.D.; Tamba, B.-I. Major Phytocannabinoids and Their Related Compounds: Should We Only Search for Drugs That Act on Cannabinoid Receptors? Pharmaceutics 2021, 13, 1823. [Google Scholar] [CrossRef]
- Pattnaik, F.; Nanda, S.; Mohanty, S.; Dalai, A.K.; Kumar, V.; Ponnusamy, S.K.; Naik, S. Cannabis: Chemistry, Extraction and Therapeutic Applications. Chemosphere 2022, 289, 133012. [Google Scholar] [CrossRef]
- Mlost, J.; Bryk, M.; Starowicz, K. Cannabidiol for Pain Treatment: Focus on Pharmacology and Mechanism of Action. Int. J. Mol. Sci. 2020, 21, 8870. [Google Scholar] [CrossRef]
- Pavlovic, R.; Nenna, G.; Calvi, L.; Panseri, S.; Borgonovo, G.; Giupponi, L.; Cannazza, G.; Giorgi, A. Quality Traits of “Cannabidiol Oils”: Cannabinoids Content, Terpene Fingerprint and Oxidation Stability of European Commercially Available Preparations. Molecules 2018, 23, 1230. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bettiol, A.; Lombardi, N.; Crescioli, G.; Maggini, V.; Gallo, E.; Mugelli, A.; Firenzuoli, F.; Baronti, R.; Vannacci, A. Galenic Preparations of Therapeutic Differ in Cannabinoids Concentration: A Quantitative Analysis of Variability and Possible Clinical Implications. Front. Pharmacol. 2018, 9, 1543. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baron, E.P. Medicinal Properties of Cannabinoids, Terpenes, and Flavonoids in Cannabis, and Benefits in Migraine, Headache, and Pain: An Update on Current Evidence and Cannabis Science. Headache 2018, 58, 1139–1186. [Google Scholar] [CrossRef] [PubMed]
- Erridge, S.; Mangal, N.; Salazar, O.; Pacchetti, B.; Sodergren, M.H. Cannflavins—From Plant to Patient: A Scoping Review. Fitoterapia 2020, 146, 104712. [Google Scholar] [CrossRef]
- Sommano, S.R.; Chittasupho, C.; Ruksiriwanich, W.; Jantrawut, P. The Cannabis Terpenes. Molecules 2020, 25, 5792. [Google Scholar] [CrossRef] [PubMed]
- Bautista, J.L.; Yu, S.; Tian, L. Flavonoids in: Biosynthesis, Bioactivities, and Biotechnology. ACS Omega 2021, 6, 5119–5123. [Google Scholar] [CrossRef]
- Antonisamy, P.; Duraipandiyan, V.; Ignacimuthu, S. Anti-Inflammatory, Analgesic and Antipyretic Effects of Friedelin Isolated from Azima Tetracantha Lam. in Mouse and Rat Models. J. Pharm. Pharmacol. 2011, 63, 1070–1077. [Google Scholar] [CrossRef]
- Miyazawa, M.; Yamafuji, C. Inhibition of Acetylcholinesterase Activity by Bicyclic Monoterpenoids. J. Agric. Food Chem. 2005, 53, 1765–1768. [Google Scholar] [CrossRef]
- Liktor-Busa, E.; Keresztes, A.; LaVigne, J.; Streicher, J.M.; Largent-Milnes, T.M. Analgesic Potential of Terpenes Derived from. Pharmacol. Rev. 2021, 73, 98–126. [Google Scholar] [CrossRef]
- Gertsch, J.; Leonti, M.; Raduner, S.; Racz, I.; Chen, J.-Z.; Xie, X.-Q.; Altmann, K.-H.; Karsak, M.; Zimmer, A. Beta-Caryophyllene Is a Dietary Cannabinoid. Proc. Natl. Acad. Sci. USA 2008, 105, 9099–9104. [Google Scholar] [CrossRef] [Green Version]
- Hanuš, L.O.; Hod, Y. Terpenes/Terpenoids in: Are They Important? Med Cannabis Cannabinoids 2020, 3, 25–60. [Google Scholar] [CrossRef] [PubMed]
- Scurria, A.; Sciortino, M.; Presentato, A.; Lino, C.; Piacenza, E.; Albanese, L.; Zabini, F.; Meneguzzo, F.; Nuzzo, D.; Pagliaro, M.; et al. Volatile Compounds of Lemon and Grapefruit IntegroPectin. Molecules 2020, 26, 51. [Google Scholar] [CrossRef]
- Booth, J.K.; Page, J.E.; Bohlmann, J. Terpene Synthases from Cannabis Sativa. PLoS ONE 2017, 12, e0173911. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Marchini, M.; Charvoz, C.; Dujourdy, L.; Baldovini, N.; Filippi, J.-J. Multidimensional Analysis of Cannabis Volatile Constituents: Identification of 5,5-Dimethyl-1-vinylbicyclo[2.1.1]hexane as a Volatile Marker of Hashish, the Resin of Cannabis sativa L. J. Chromatogr. A 2014, 1370, 200–215. [Google Scholar] [CrossRef] [PubMed]
- Du, Z.-C.; Xia, Z.-S.; Zhang, M.-Z.; Wei, Y.-T.; Malhotra, N.; Saputra, F.; Audira, G.; Roldan, M.J.M.; Hsiao, C.-D.; Hao, E.-W.; et al. Sub-Lethal Camphor Exposure Triggers Oxidative Stress, Cardiotoxicity, and Cardiac Physiology Alterations in Zebrafish Embryos. Cardiovasc. Toxicol. 2021, 21, 901–913. [Google Scholar] [CrossRef] [PubMed]
- Chueca, B.; Pagán, R.; García-Gonzalo, D. Oxygenated Monoterpenes Citral and Carvacrol Cause Oxidative Damage in Escherichia Coli without the Involvement of Tricarboxylic Acid Cycle and Fenton Reaction. Int. J. Food Microbiol. 2014, 189, 126–131. [Google Scholar] [CrossRef]
- Booth, J.K.; Bohlmann, J. Terpenes in Cannabis Sativa—From Plant Genome to Humans. Plant Sci. 2019, 284, 67–72. [Google Scholar] [CrossRef]
- Crescioli, G.; Lombardi, N.; Bettiol, A.; Menniti-Ippolito, F.; Da Cas, R.; Parrilli, M.; Del Lungo, M.; Gallo, E.; Mugelli, A.; Maggini, V.; et al. Adverse Events Following Cannabis for Medical Use in Tuscany: An Analysis of the Italian Phytovigilance Database. Br. J. Clin. Pharmacol. 2020, 86, 106–120. [Google Scholar] [CrossRef]
- Ramella, A.; Roda, G.; Pavlovic, R.; Cas, M.D.; Casagni, E.; Mosconi, G.; Cecati, F.; Minghetti, P.; Grizzetti, C. Impact of Lipid Sources on Quality Traits of Medical Cannabis-Based Oil Preparations. Molecules 2020, 25, 2986. [Google Scholar] [CrossRef]
- Calvi, L.; Pentimalli, D.; Panseri, S.; Giupponi, L.; Gelmini, F.; Beretta, G.; Vitali, D.; Bruno, M.; Zilio, E.; Pavlovic, R.; et al. Comprehensive Quality Evaluation of Medical Cannabis sativa L. Inflorescence and Macerated Oils Based on HS-SPME Coupled to GC-MS and LC-HRMS (q-Exactive Orbitrap®) Approach. J. Pharm. Biomed. Anal. 2018, 150, 208–219. [Google Scholar] [CrossRef]
- Ternelli, M.; Brighenti, V.; Anceschi, L.; Poto, M.; Bertelli, D.; Licata, M.; Pellati, F. Innovative Methods for the Preparation of Medical Cannabis Oils with a High Content of Both Cannabinoids and Terpenes. J. Pharm. Biomed. Anal. 2020, 186, 113296. [Google Scholar] [CrossRef] [PubMed]
- Pacifici, R.; Marchei, E.; Salvatore, F.; Guandalini, L.; Busardò, F.P.; Pichini, S. Evaluation of Cannabinoids Concentration and Stability in Standardized Preparations of Cannabis Tea and Cannabis Oil by Ultra-High Performance Liquid Chromatography Tandem Mass Spectrometry. Clin. Chem. Lab. Med. 2017, 55, 1555–1563. [Google Scholar] [CrossRef] [PubMed]
- Scholz, C.; Madry, M.M.; Kraemer, T.; Baumgartner, M.R. LC-MS/MS Analysis of Δ9-THC, CBN and CBD in Hair: Investigation of Artefacts. J. Anal. Toxicol. 2021, bkab056. [Google Scholar] [CrossRef] [PubMed]
- Grotenhermen, F. Pharmacokinetics and Pharmacodynamics of Cannabinoids. Clin. Pharmacokinet. 2003, 42, 327–360. [Google Scholar] [CrossRef] [PubMed]
- Filly, A.; Fernandez, X.; Minuti, M.; Visinoni, F.; Cravotto, G.; Chemat, F. Solvent-Free Microwave Extraction of Essential Oil from Aromatic Herbs: From Laboratory to Pilot and Industrial Scale. Food Chem. 2014, 150, 193–198. [Google Scholar] [CrossRef] [Green Version]
- Mwaurah, P.W.; Kumar, S.; Kumar, N.; Attkan, A.K.; Panghal, A.; Singh, V.K.; Garg, M.K. Novel Oil Extraction Technologies: Process Conditions, Quality Parameters, and Optimization. Compr. Rev. Food Sci. Food Saf. 2020, 19, 3–20. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wen, C.; Zhang, J.; Zhang, H.; Dzah, C.S.; Zandile, M.; Duan, Y.; Ma, H.; Luo, X. Advances in Ultrasound Assisted Extraction of Bioactive Compounds from Cash Crops—A Review. Ultrason. Sonochem. 2018, 48, 538–549. [Google Scholar] [CrossRef]
- Bitterling, H.; Lorenz, P.; Vetter, W.; Conrad, J.; Kammerer, D.R.; Stintzing, F.C. Rapid Spectrophotometric Method for Assessing Hydroperoxide Formation from Terpenes in Essential Oils upon Oxidative Conditions. J. Agric. Food Chem. 2020, 68, 9576–9584. [Google Scholar] [CrossRef]
- Turek, C.; Stintzing, F.C. Stability of Essential Oils: A Review. Comp. Rev. Food Sci. Food Saf. 2013, 12, 40–53. [Google Scholar] [CrossRef]
- Karlberg, A.-T.; Börje, A.; Duus Johansen, J.; Lidén, C.; Rastogi, S.; Roberts, D.; Uter, W.; White, I.R. Activation of Non-Sensitizing or Low-Sensitizing Fragrance Substances into Potent Sensitizers—Prehaptens and Prohaptens. Contact Dermat. 2013, 69, 323–334. [Google Scholar] [CrossRef] [Green Version]
- Karlberg, A.-T.; Lepoittevin, J.-P. One Hundred Years of Allergic Contact Dermatitis due to Oxidized Terpenes: What We Can Learn from Old Research on Turpentine Allergy. Contact Dermat. 2021, 85, 627–636. [Google Scholar] [CrossRef] [PubMed]
- Diepgen, T.L.; Ofenloch, R.; Bruze, M.; Cazzaniga, S.; Coenraads, P.J.; Elsner, P.; Goncalo, M.; Svensson, Å.; Naldi, L. Prevalence of Fragrance Contact Allergy in the General Population of Five European Countries: A Cross-Sectional Study. Br. J. Dermatol. 2015, 173, 1411–1419. [Google Scholar] [CrossRef]
- Agus, H.H. Terpene Toxicity and Oxidative Stress. In Toxicology; Elsevier: Amsterdam, The Netherlands, 2021; pp. 33–42. ISBN 9780128190920. [Google Scholar]
- Chubukov, V.; Mingardon, F.; Schackwitz, W.; Baidoo, E.E.K.; Alonso-Gutierrez, J.; Hu, Q.; Lee, T.S.; Keasling, J.D.; Mukhopadhyay, A. Acute Limonene Toxicity in Escherichia Coli Is Caused by Limonene Hydroperoxide and Alleviated by a Point Mutation in Alkyl Hydroperoxidase AhpC. Appl. Environ. Microbiol. 2015, 81, 4690–4696. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Anand, U.; Pacchetti, B.; Anand, P.; Sodergren, M.H. Cannabis-Based Medicines and Pain: A Review of Potential Synergistic and Entourage Effects. Pain Manag. 2021, 11, 395–403. [Google Scholar] [CrossRef] [PubMed]
- Dosoky, N.S.; Setzer, W.N. Maternal Reproductive Toxicity of Some Essential Oils and Their Constituents. Int. J. Mol. Sci. 2021, 22, 2380. [Google Scholar] [CrossRef] [PubMed]
- Perestrelo, R.; Silva, C.; Fernandes, M.X.; Câmara, J.S. Prediction of Terpenoid Toxicity Based on a Quantitative Structure-Activity Relationship Model. Foods 2019, 8, 628. [Google Scholar] [CrossRef] [Green Version]
- Worth, T. Cannabi’s Chemical Synergies. Nature 2019, 572, S12–S13. [Google Scholar] [CrossRef]
- Weston-Green, K.; Clunas, H.; Jimenez Naranjo, C. A Review of the Potential Use of Pinene and Linalool as Terpene-Based Medicines for Brain Health: Discovering Novel Therapeutics in the Flavours and Fragrances of Cannabis. Front. Psychiatry 2021, 12, 583211. [Google Scholar] [CrossRef]
- Pamplona, F.A.; da Silva, L.R.; Coan, A.C. Potential Clinical Benefits of CBD-Rich Extracts Over Purified CBD in Treatment-Resistant Epilepsy: Observational Data Meta-Analysis. Front. Neurol. 2018, 9, 759. [Google Scholar] [CrossRef] [Green Version]
- Russo, E.B. Taming THC: Potential Cannabis Synergy and Phytocannabinoid-Terpenoid Entourage Effects. Br. J. Pharmacol. 2011, 163, 1344–1364. [Google Scholar] [CrossRef]
- Bilbrey, J.A.; Ortiz, Y.T.; Felix, J.S.; McMahon, L.R.; Wilkerson, J.L. Evaluation of the Terpenes β-Caryophyllene, α-Terpineol, and γ-Terpinene in the Mouse Chronic Constriction Injury Model of Neuropathic Pain: Possible Cannabinoid Receptor Involvement. Psychopharmacology 2021. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, N.; Takada, T.; Yamamura, Y.; Adachi, I.; Suzuki, H.; Kawakami, J. Inhibitory Effects of Terpenoids on Multidrug Resistance-Associated Protein 2- and Breast Cancer Resistance Protein-Mediated Transport. Drug Metab. Dispos. 2008, 36, 1206–1211. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schmitt, S.; Schaefer, U.F.; Doebler, L.; Reichling, J. Cooperative Interaction of Monoterpenes and Phenylpropanoids on the in Vitro Human Skin Permeation of Complex Composed Essential Oils. Planta Med. 2009, 75, 1381–1385. [Google Scholar] [CrossRef] [PubMed]
- Yamane, M.A.; Williams, A.C.; Barry, B.W. Terpene Penetration Enhancers in Propylene Glycol/water Co-Solvent Systems: Effectiveness and Mechanism of Action. J. Pharm. Pharmacol. 1995, 47, 978–989. [Google Scholar] [CrossRef]
- Senyiğit, T.; Padula, C.; Ozer, O.; Santi, P. Different Approaches for Improving Skin Accumulation of Topical Corticosteroids. Int. J. Pharm. 2009, 380, 155–160. [Google Scholar] [CrossRef]
- Furuishi, T.; Kato, Y.; Fukami, T.; Suzuki, T.; Endo, T.; Nagase, H.; Ueda, H.; Tomono, K. Effect of Terpenes on the Skin Permeation of Lomerizine Dihydrochloride. J. Pharm. Pharm. Sci. 2013, 16, 551–563. [Google Scholar] [CrossRef] [Green Version]
- Femenía-Font, A.; Balaguer-Fernández, C.; Merino, V.; Rodilla, V.; López-Castellano, A. Effect of Chemical Enhancers on the in Vitro Percutaneous Absorption of Sumatriptan Succinate. Eur. J. Pharm. Biopharm. 2005, 61, 50–55. [Google Scholar] [CrossRef]
- Guo, X.; Rong, Y.; Zhang, L.; Ye, J.-C. Enhancing Effect of Chiral Enhancer Linalool on Skin Permeation of Naproxen. Zhongguo Yi Xue Ke Xue Yuan Xue Bao 2016, 38, 55–61. [Google Scholar] [CrossRef]
- Sexton, M.; Shelton, K.; Haley, P.; West, M. Evaluation of Cannabinoid and Terpenoid Content: Cannabis Flower Compared to Supercritical CO2 Concentrate. Planta Med. 2018, 84, 234–241. [Google Scholar] [CrossRef] [Green Version]
- Pang, M.-H.; Kim, Y.; Jung, K.W.; Cho, S.; Lee, D.H. A Series of Case Studies: Practical Methodology for Identifying Antinociceptive Multi-Target Drugs. Drug Discov. Today 2012, 17, 425–434. [Google Scholar] [CrossRef]


| Compound Class | 2D Structure Image | PubChem Identifier: CID | Medicinal Properties | Reference |
|---|---|---|---|---|
| Cannabinoids | C₂₁H₃₀O₂ | |||
| Δ9-THC | 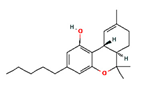 | 16078 | Psychoactive Antinociceptive, antiemetic, appetite stimulator, anti-inflammatory | [9] |
| CBD | 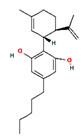 | 644019 | Nonpsychoactive Anticonvulsant, analgesic, anti-inflammatory, antiemetic, anxiolytic, neuroprotective, sleep-promoting | [10] |
| Flavonoids | C6C3C6 | |||
| apigenin | 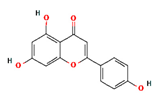 | 5280443 | Anti-inflammatory, anxiolytic | [13] |
| cannflavin B | 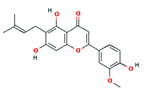 | 403815 | Anti-inflammatory | [14] |
| Terpenes | ||||
| Monoterpenes | C10H16 | |||
| α-pinene | 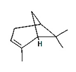 | 6654 | Neuroprotective | [18] |
| limonene | 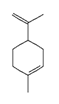 | 22311 | Anti-inflammatory, anxiolytic, sedative | [19] |
| myrcene |  | 31253 | Anti-inflammatory, analgesic, anxiolytic | [13] |
| Diterpene | C20H32 | |||
| phytol |  | 5280435 | Sleep-promoting | [13] |
| Triterpenes | C30H48 | |||
| friedelin | 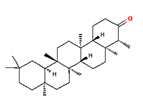 | 91472 | Anti-inflammatory, analgesic | [17] |
| Sesquiterpenes | C15H24 | |||
| α-humulene | 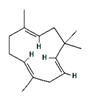 | 5281520 | Anti-inflammatory, analgesic | [13] |
| β-caryophyllene | 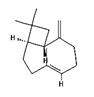 | 5281515 | Anti-inflammatory, antioxidant, anxiolytic, analgesic, neuro-protective | [20,21] |
| Phase | Temperature °C (Microwave Power) | Time (min) | Cycles |
|---|---|---|---|
| Pre-heating | from 25 to 100 (1500 Watt) | 5 | 5 |
| Decarboxylation | 100 (1500 Watt) | 10 | |
| Cooling | from 100 to 25 | 25 | |
| Rest | 25 | 55 |
| Time (min) | Solvent A (%) | Solvent B (%) |
|---|---|---|
| 0.00 | 90 | 10 |
| 0.10 | 90 | 10 |
| 2.00 | 10 | 90 |
| 5.00 | 10 | 90 |
| 5.10 | 90 | 10 |
| 7.00 | 90 | 10 |
| Electrospray Ionization Source | ESI |
|---|---|
| Ion Transfer Tube temperature | 350 °C |
| Vaporization temperature | 300 °C |
| Electrospray tension | 3.50 kV |
| Scanning acquisition | SIM |
| Isolation range | 2 m/z |
| Compound | Retention Time (tR, min) | MRM Transitions (m/z) | Collision Energy (eV) | Ion Spray Voltage (V) | Dwell Time (ms) | |
|---|---|---|---|---|---|---|
| CBDA | 3.84 | 357.4 | 245.2 | 28 | 3500 | 19.96 |
| 313.2 | 22 | |||||
| 339.2 | 19 | |||||
| Δ9-THCA | 4.39 | 357.4 | 245.2 | 28 | 3500 | 19.96 |
| 313.2 | 22 | |||||
| 339.2 | 19 | |||||
| CBD | 3.77 | 315.4 | 135.1 | 20 | 3500 | 19.96 |
| 193.1 | 22 | |||||
| 259.2 | 19 | |||||
| Δ9-THC | 4.04 | 315.4 | 135.1 | 20 | 3500 | 19.96 |
| 193.1 | 22 | |||||
| 259.2 | 19 | |||||
| CBN | 3.96 | 311.4 | 223.00 | 20 | 3500 | 19.96 |
| 240.92 | 18 | |||||
| 293.17 | 17 | |||||
| ISTD | 3.2 | 285.1 | 154.0 193.0 222.2 | |||
| METHOD | SIFAP [32] | CALVI [30] | CERFIT Present Work |
|---|---|---|---|
| Cannabis variety (5 g)/(T) | Bedrocan (RT) | Bedrocan (RT) | Bedrocan (−20 °C) |
| Matrix (50 mL) | MCT oil a | MCT oil a | MCT oil |
| Cannabis/Matrix ratio | 1:10 | 1:10 | 1:10 |
| Thawing | No | No | Yes/25 °C, 60 min fan oven |
| Decarboxylation step | Yes/115 °C, 40 min static oven | Yes/135 °C, 30 min fan oven | Yes/cyclic T b, 8 h vacuum microwave extractor |
| Freezing | No | No | Yes/−20 °C, 60 min |
| Extraction process | Turbo emulsion water bath (100 °C 40 min) | Turbo emulsion bath sonicator (35 KHz 30 min) | Turbo emulsion probe sonicator (200 W/30 KHz 8 min) |
| Oil heating step | Yes | No c | No d |
| Filtration | Yes/filter paper | Yes/filter paper | Yes/filter paper |
| Preparation time (hour) | 2 | 1.5 | 9.5 (8 nightlong) |
| Compound | Overall | MCO-1 SIFAP | MCO-2 CALVI | MCO-3 CERFIT | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| mg/mL | Mean (SD) | Median (Min–Max) | Mean (SD) | Median (Min–Max) | Mean (SD) | Median (Min–Max) | Mean (SD) | Median (Min–Max) | F Test p Value * | Chi2 Test p Value ° |
| CBDA | 0.04 (0.05) | 0.03 (0.00–0.20) | 0.03 (0.67) | 0.00 d (0.00–0.20) | 0.07 (0.03) | 0.08 e (0.03–0.10) | 0.03 (0.03) | 0.03 d (0.00–0.10) | 2.70 0.0854 | 8.13 0.0172 |
| CBD | 0.03 (0.04) | 0.02 (0.00–0.13) | NQ | NQ | 0.03 (0.05) | 0.02 (0.00–0.13) | 0.06 (0.02) | 0.05 (0.04–0.09) | 2.92 0.1044 | 3.86 0.0500 |
| CBN | 0.29 (0.35) | 0.17 (0.02–1.06) | 0.18 a (0.01) | 0.18 (0.16–0.19) | 0.17 a (0.14) | 0.14 (0.04–0.54) | 0.53 b (0.53) | 0.55 (0.02–1.06) | 4.33 0.0234 | 1.62 0.4447 |
| Δ9-THCA | 0.11 (0.18) | 0.00 (0.00–0.70) | 0.03 (0.67) | 0.00 d (0.00–0.20) | 0.14 (0.23) | 0.01 d (0.00–0.70) | 0.17 (0.18) | 0.15 e (0.00–0.50) | 1.84 0.1789 | 3.42 0.1809 |
| Δ9-THC | 20.58 (1.94) | 19.77 (17.99–23.87) | 22.42 a (1.11) | 22.82 d (19.37–23.11) | 18.80 b (0.72) | 19.00 e (17.99–20.13) | 20.52 c (1.75) | 19.82 f (18.90–23.87) | 20.32 0.0001 | 16.91 0.0002 |
| Compound | MCOs Overall | MCO-1 SIFAP | MCO-2 CALVI | MCO-3 CERFIT | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| µg/mL | Mean (SD) | Median (Min–Max) | Mean (SD) | Median (Min–Max) | Mean (SD) | Median (Min–Max) | Mean (SD) | Median (Min–Max) | F Test p Value * | Chi2 Test p Value ° |
| α-Pinene | 24.82 (12.85) | 17.23 (12.60–47.10) | 14.86 a (0.88) | 15.05 d (12.60–15.90) | 17.10 b (1.38) | 17.23 e (14.08–18.23) | 42.50 c (2.41) | 42.23 f (39.00–47.10) | 834.73 0.0001 | 23.28 0.0001 |
| Limonene | 32.98 (13.96) | 31.12 (16.99–54.39) | 17.23 a (0.28) | 17.17 d (16.99–17.94) | 31.14 b (1.33) | 31.12 e (28.58–34.20) | 50.57 c (1.69) | 50.05 f (48.11–54.39) | 1786.70 0.0001 | 25.81 0.0001 |
| β-Caryophyllene | 56.77 (8.55) | 60.87 (41.34–68.25) | 64.36 a (1.73) | 64.22 d (61.55–68.25) | 60.53 b (1.31) | 60.87 e (57.09–61.86) | 45.41 c (2.74) | 45.32 f (41.34–50.20) | 245.76 0.0001 | 25.55 0.0001 |
| Total mono- di-tri Terpenes | 3998.60 (1528.39) | 4678.89 (1914.10–5405.44) | 1915.38 a (2.09) | 1914.67 d (1914.09–1921.09) | 4676.45 b (7.12) | 4678.89 e (4656.38–4679.95) | 5403.96 c (1.39) | 5404.39 f (5401.80–5405.44) | 1.8 × 106 0.0001 | 25.81 0.0001 |
| Total Sesquiterpenes | 248.18 (87.23) | 274.70 (119.45–341.34) | 132.6 a (4.82) | 134.11 d (119.45–137.02) | 274.27 b (1.63) | 274.70 e (269.98–276.04) | 337.65 c (1.97) | 338.14 f (334.23–341.34) | 1.1 × 104 0.0001 | 25.81 0.0001 |
| Total oxidized TP | NC | NC | 216.90 (2.60) | 214.00 (214.00–223.00) | NQ | NQ | NQ | NQ | NC | NC |
| Total TP | 4318.60 (1513.39) | 4952.00 (2257.00–5743.00) | 2264.50 a (4.40) | 2263.50 d (2257.00–2273.00) | 4950.10 b (7.20) | 4952.00 e (4930.00–4954.00) | 5741.20 c (1.32) | 5741.00 f (5739.00–5743.00) | 1.4 × 106 0.0001 | 25.81 0.0001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maggini, V.; Calvi, L.; Pelagatti, T.; Gallo, E.R.; Civati, C.; Privitera, C.; Squillante, F.; Maniglia, P.; Di Candia, D.; Spampatti, R.; et al. An Optimized Terpene Profile for a New Medical Cannabis Oil. Pharmaceutics 2022, 14, 298. https://doi.org/10.3390/pharmaceutics14020298
Maggini V, Calvi L, Pelagatti T, Gallo ER, Civati C, Privitera C, Squillante F, Maniglia P, Di Candia D, Spampatti R, et al. An Optimized Terpene Profile for a New Medical Cannabis Oil. Pharmaceutics. 2022; 14(2):298. https://doi.org/10.3390/pharmaceutics14020298
Chicago/Turabian StyleMaggini, Valentina, Lorenzo Calvi, Tommaso Pelagatti, Eugenia Rosaria Gallo, Celine Civati, Carlo Privitera, Flavio Squillante, Paolo Maniglia, Domenico Di Candia, Roberto Spampatti, and et al. 2022. "An Optimized Terpene Profile for a New Medical Cannabis Oil" Pharmaceutics 14, no. 2: 298. https://doi.org/10.3390/pharmaceutics14020298
APA StyleMaggini, V., Calvi, L., Pelagatti, T., Gallo, E. R., Civati, C., Privitera, C., Squillante, F., Maniglia, P., Di Candia, D., Spampatti, R., & Firenzuoli, F. (2022). An Optimized Terpene Profile for a New Medical Cannabis Oil. Pharmaceutics, 14(2), 298. https://doi.org/10.3390/pharmaceutics14020298






