Effect of Experimental Electrical and Biological Parameters on Gene Transfer by Electroporation: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Eligibility Criteria
2.2. Search Strategy
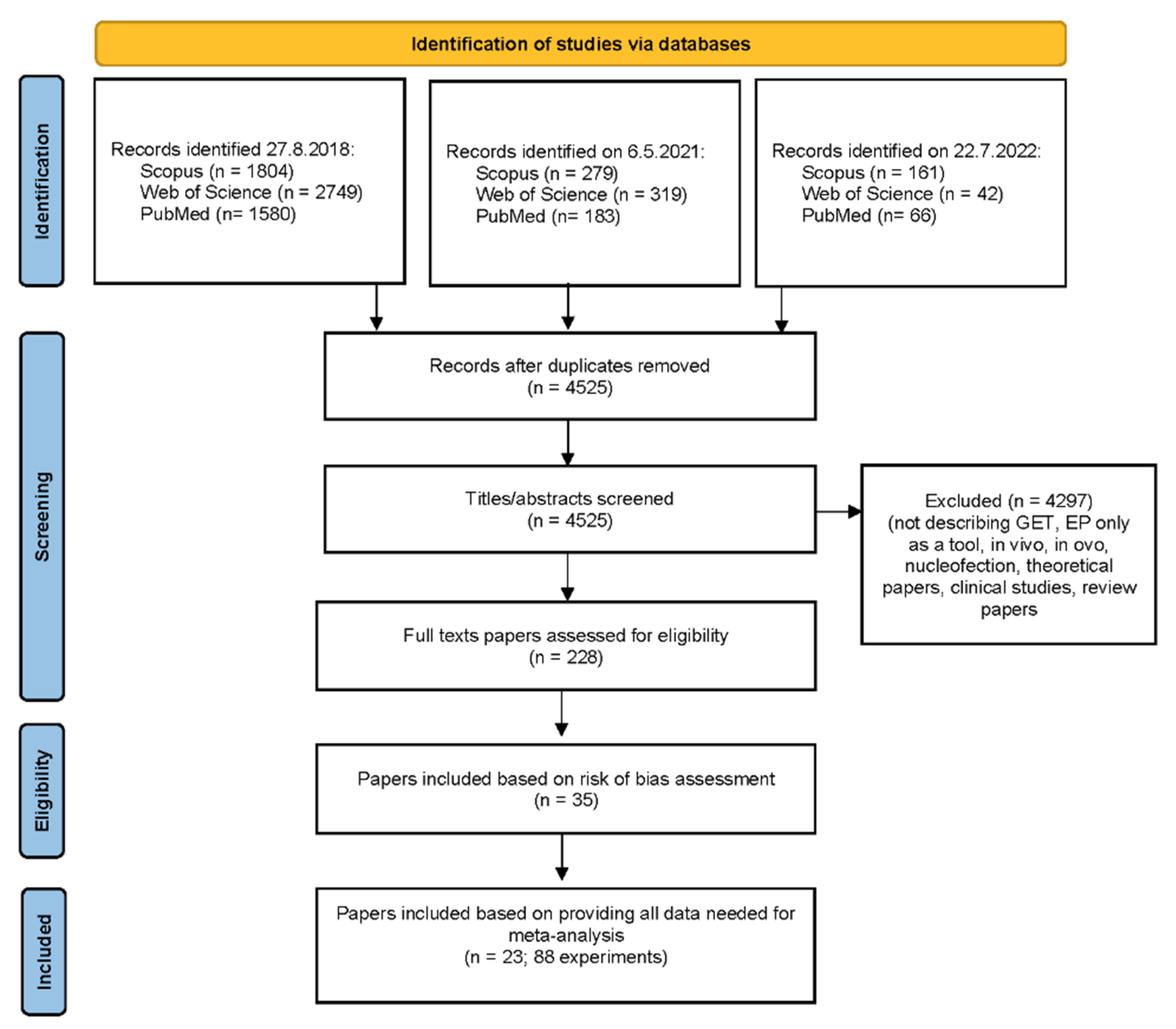
2.3. Paper Selection
2.4. Data Extraction
2.5. Assessment of Risk of Bias
2.6. Statistical Analysis
3. Results
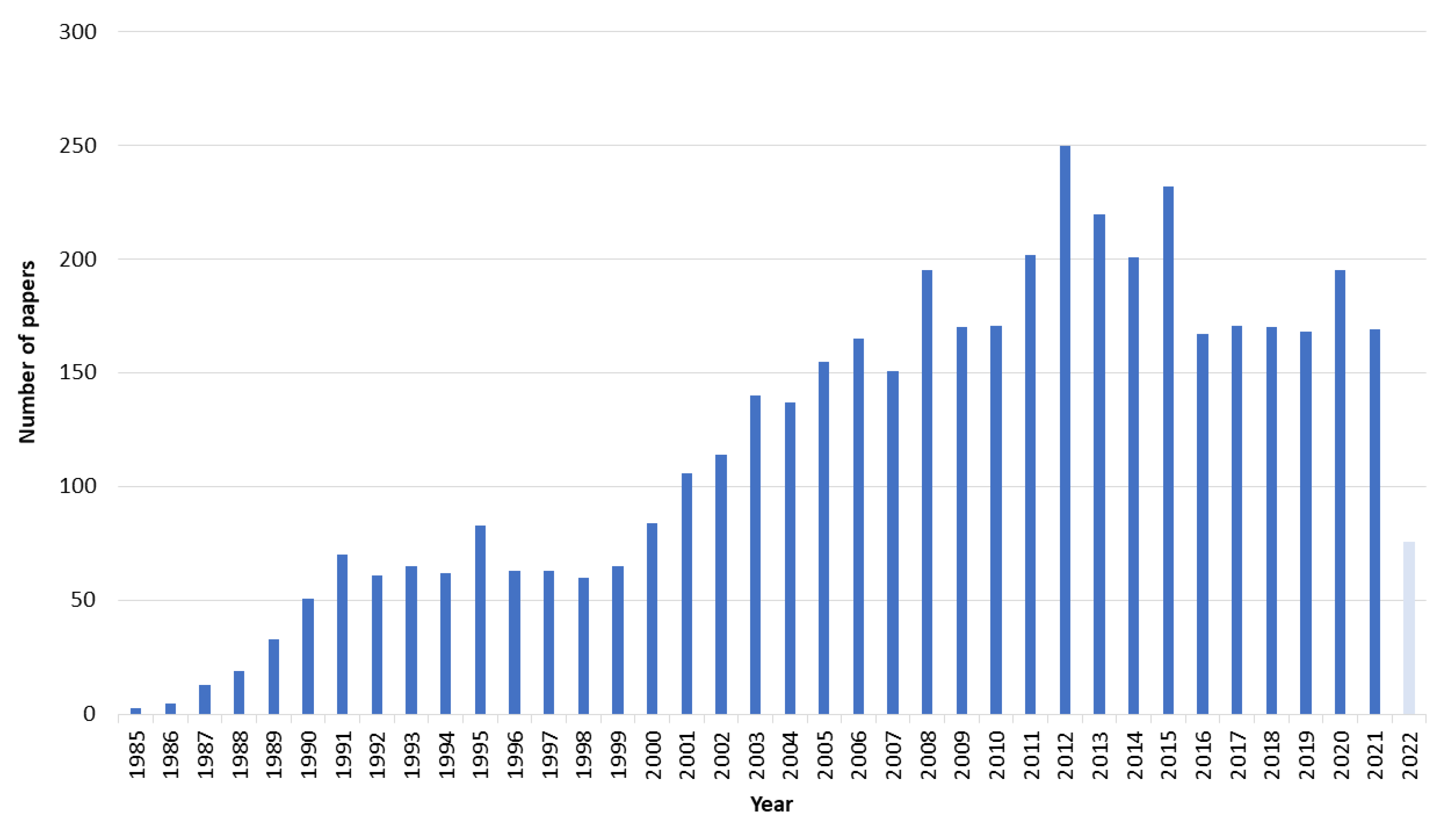
3.1. Paper Selection and Characteristics
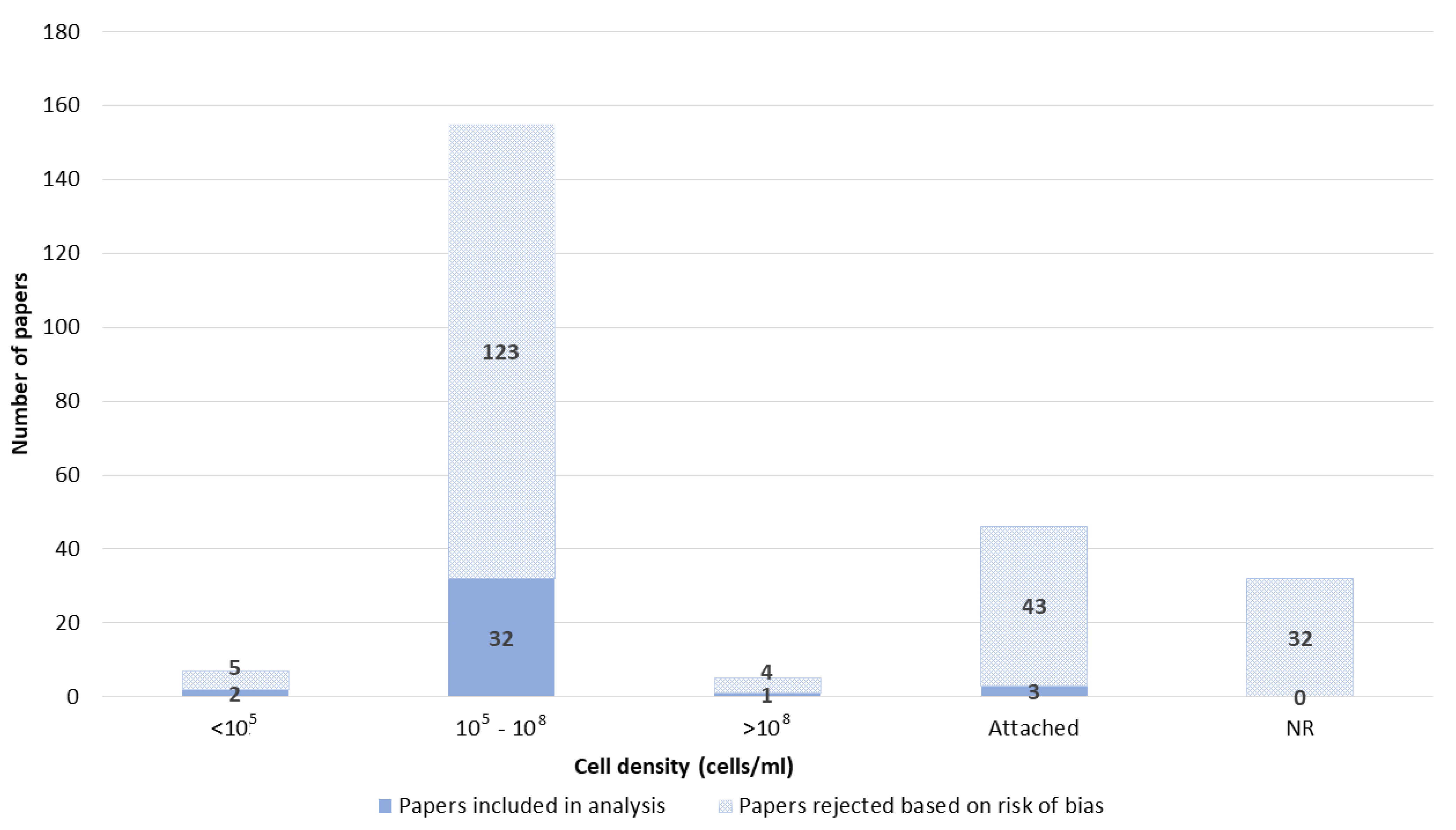
3.2. Risk of Bias Assesment
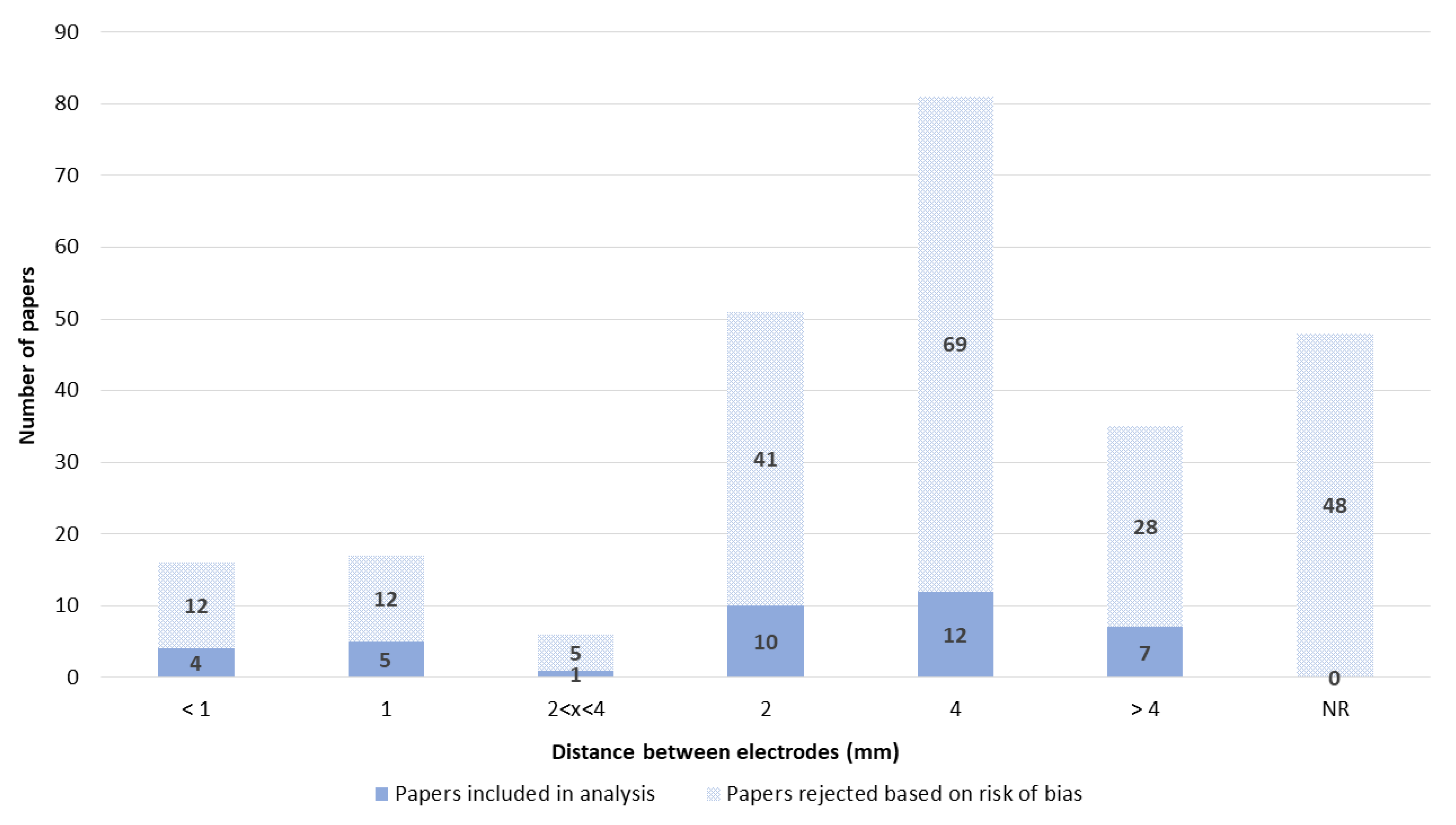
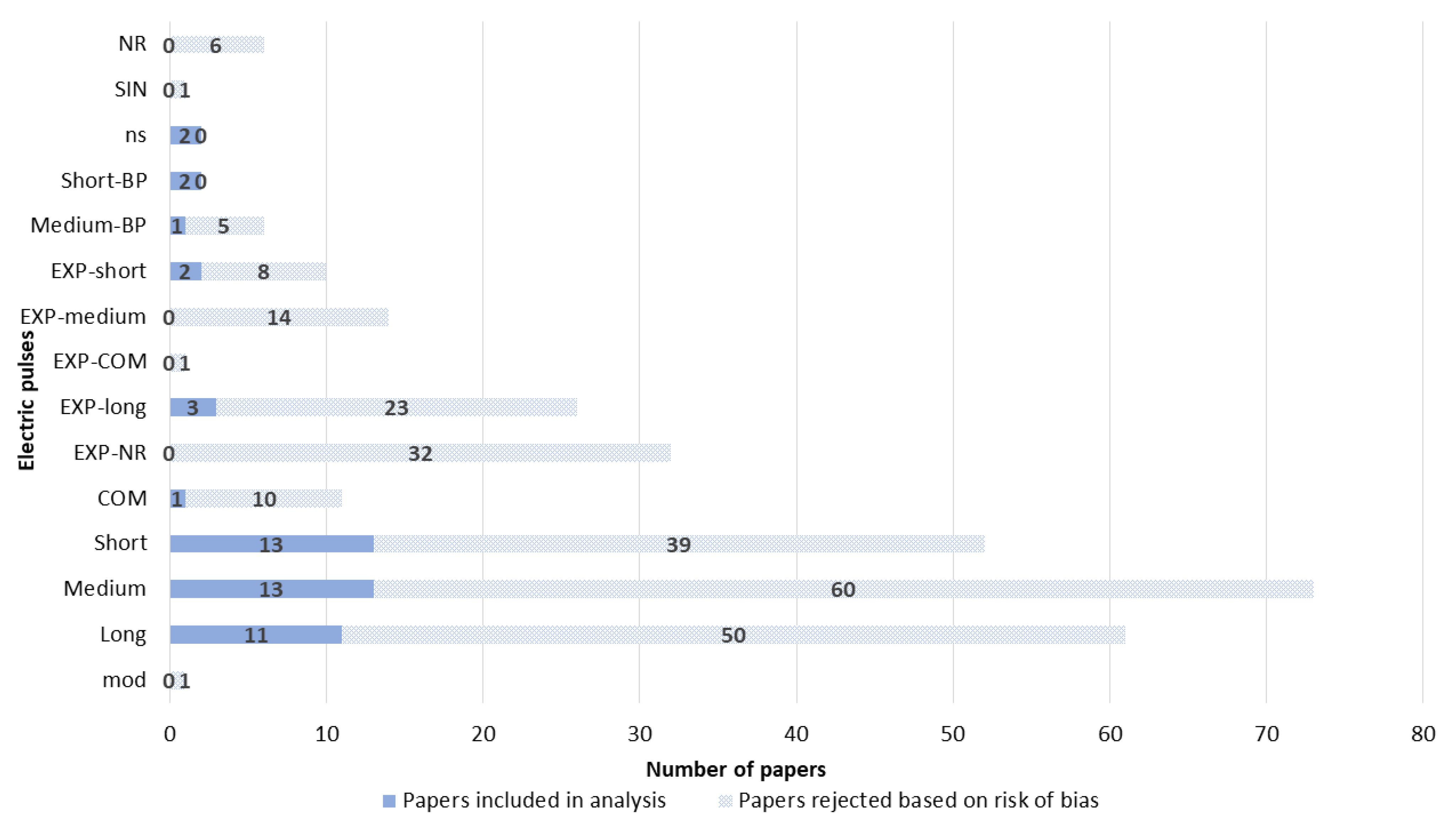
3.3. Electrodes
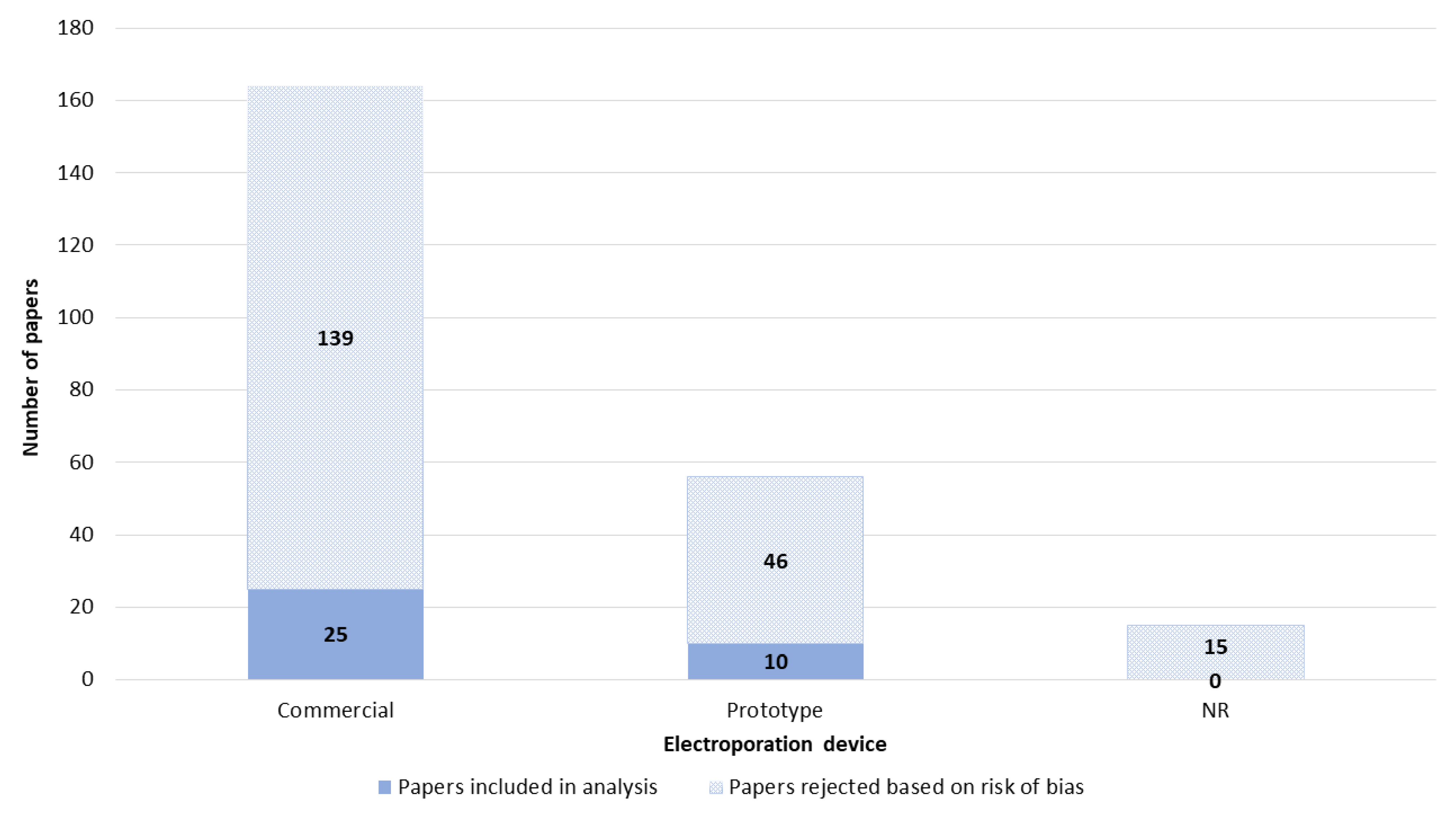
3.4. Electroporation Device
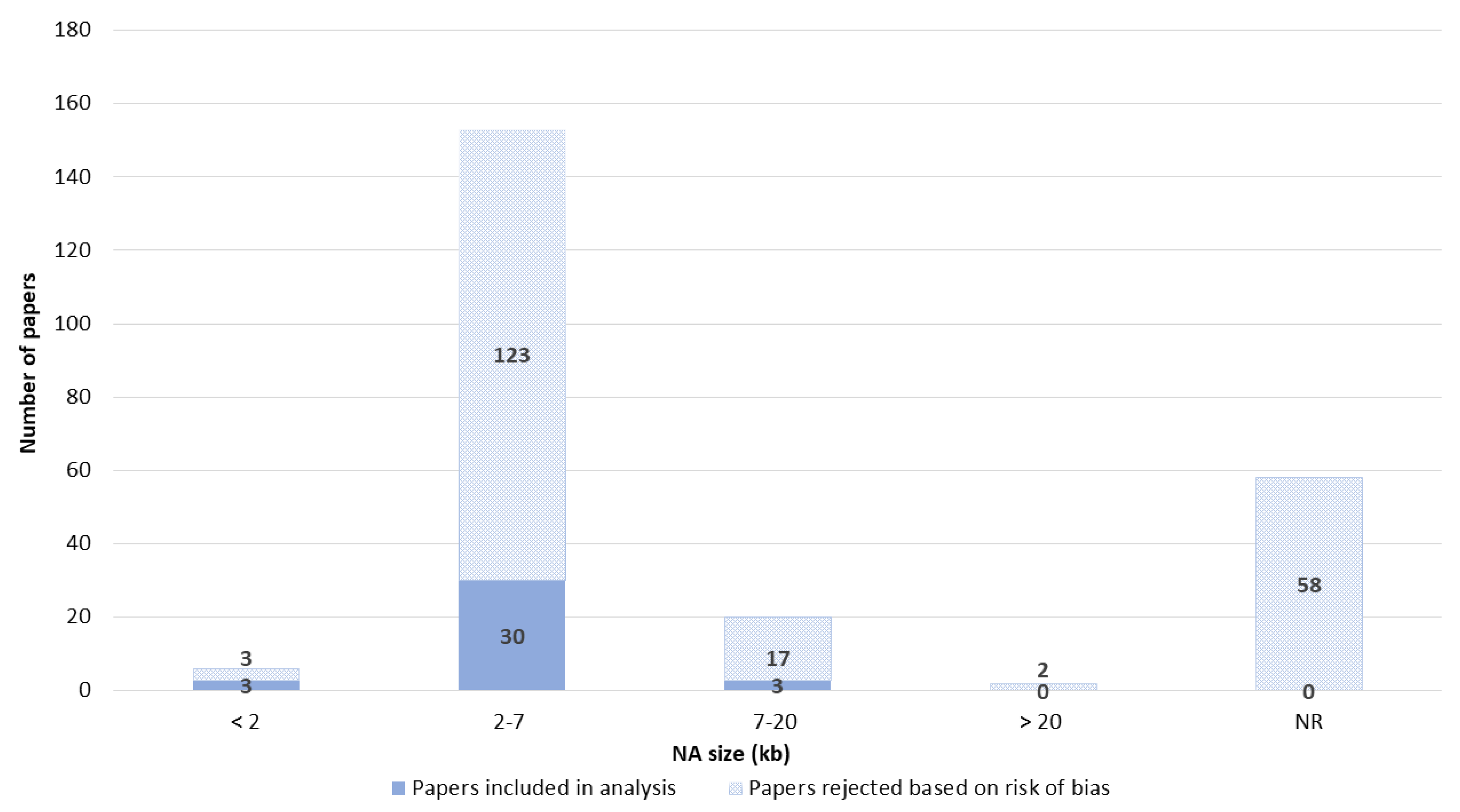
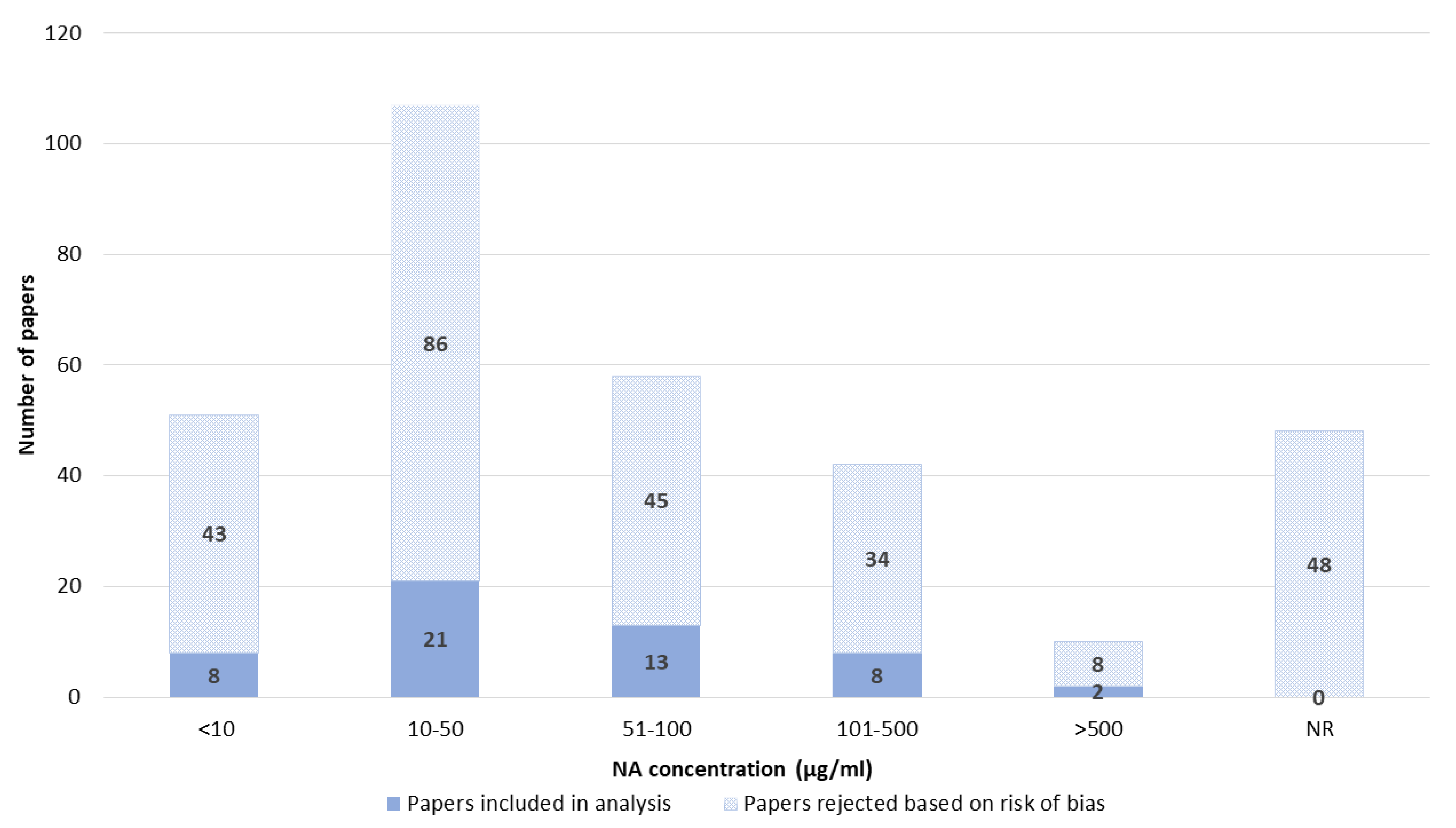
3.5. Nucleic Acids
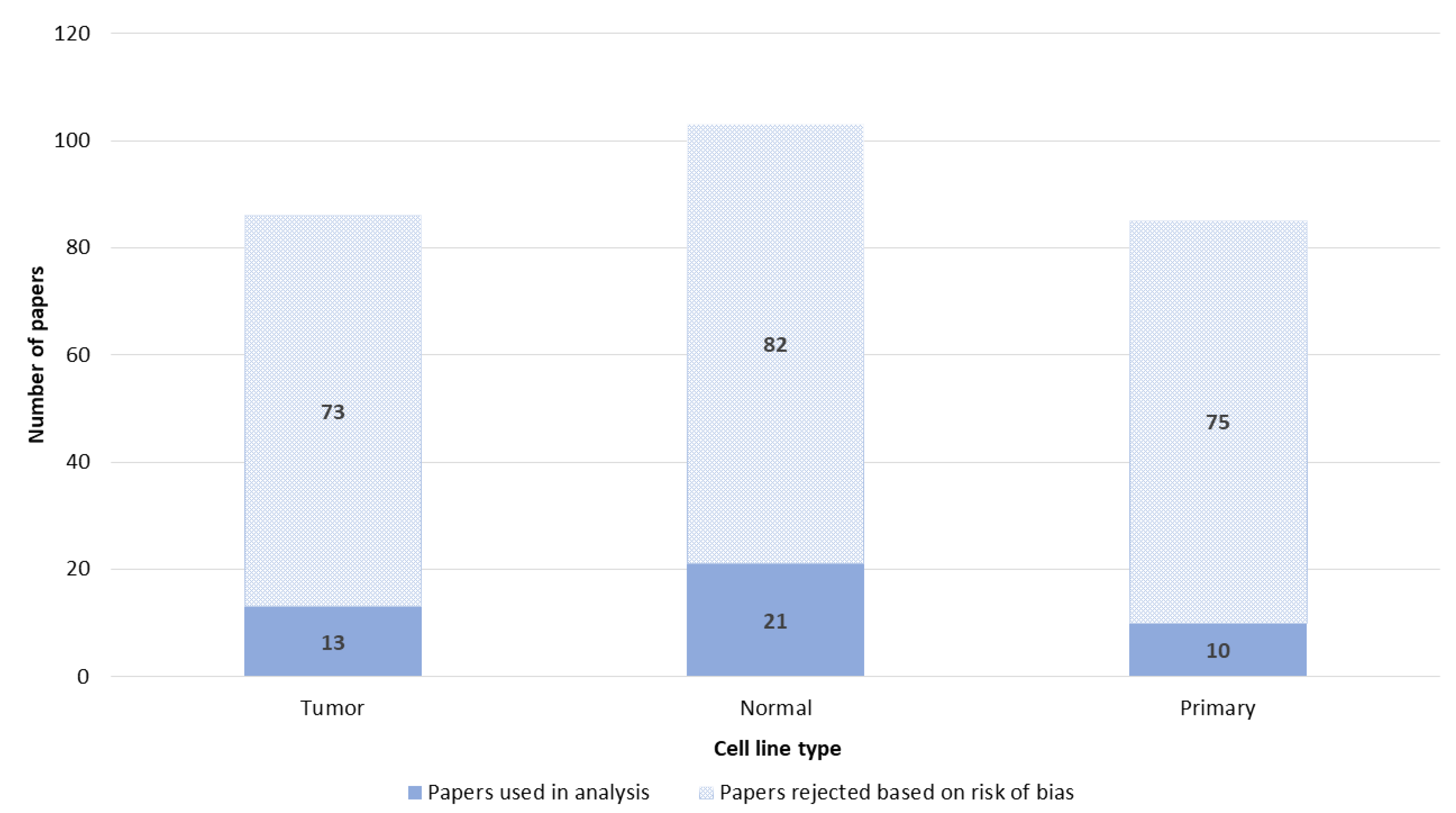
3.6. Cell Lines
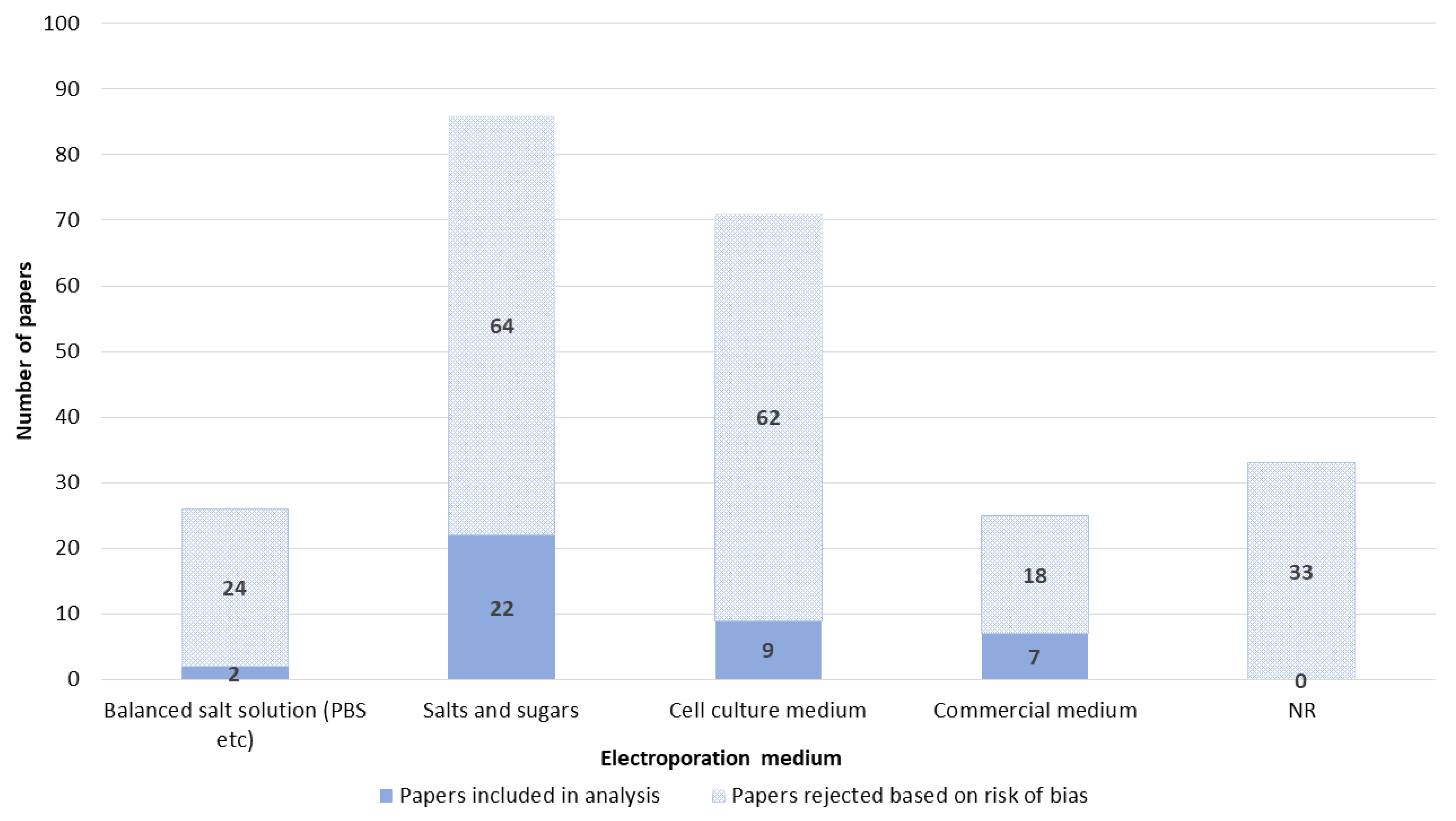
3.7. Electroporation Medium
3.8. Time at Which GET Was Measured
3.9. Sets of Parameters That Result in the Most Efficient GET
3.10. Pooled Overall GET Efficiency
3.11. Subgroup Analyses
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Neumann, E.; Schaefer-Ridder, M.; Wang, Y.; Hofschneider, P.H. Gene Transfer into Mouse Lyoma Cells by Electroporation in High Electric Fields. EMBO J. 1982, 1, 841–845. [Google Scholar] [CrossRef] [PubMed]
- Sachdev, S.; Potočnik, T.; Rems, L.; Miklavčič, D. Revisiting the Role of Pulsed Electric Fields in Overcoming the Barriers to in Vivo Gene Electrotransfer. Bioelectrochemistry 2022, 144, 107994. [Google Scholar] [CrossRef] [PubMed]
- Rosazza, C.; Haberl Meglic, S.; Zumbusch, A.; Rols, M.-P.; Miklavcic, D. Gene Electrotransfer: A Mechanistic Perspective. Curr. Gene Ther. 2016, 16, 98–129. [Google Scholar] [CrossRef]
- Cervia, L.D.; Chang, C.C.; Wang, L.; Yuan, F. Distinct Effects of Endosomal Escape and Inhibition of Endosomal Trafficking on Gene Delivery via Electrotransfection. PLoS ONE 2017, 12, e0171699. [Google Scholar] [CrossRef] [PubMed]
- Šatkauskas, S.; André, F.; Bureau, M.F.; Scherman, D.; Miklavčič, D.; Mir, L.M. Electrophoretic Component of Electric Pulses Determines the Efficacy of In Vivo DNA Electrotransfer. Hum. Gene Ther. 2005, 16, 1194–1201. [Google Scholar] [CrossRef] [PubMed]
- Haberl, S.; Kandušer, M.; Flisar, K.; Hodžić, D.; Bregar, V.B.V.B.; Miklavčič, D.; Escoffre, J.-M.J.-M.; Rols, M.-P.M..-P.; Pavlin, M. Effect of Different Parameters Used for in Vitro Gene Electrotransfer on Gene Expression Efficiency, Cell Viability and Visualization of Plasmid DNA at the Membrane Level. J. Gene Med. 2013, 15, 169–181. [Google Scholar] [CrossRef]
- Rosazza, C.; Phez, E.; Escoffre, J.M.; Cézanne, L.; Zumbusch, A.; Rols, M.P. Cholesterol Implications in Plasmid DNA Electrotransfer: Evidence for the Involvement of Endocytotic Pathways. Int. J. Pharm. 2012, 423, 134–143. [Google Scholar] [CrossRef]
- Maglietti, F.; Michinski, S.; Olaiz, N.; Castro, M.; Suárez, C.; Marshall, G. The Role of Ph Fronts in Tissue Electroporation Based Treatments. PLoS ONE 2013, 8, e80167. [Google Scholar] [CrossRef]
- Olaiz, N.; Signori, E.; Maglietti, F.; Soba, A.; Suárez, C.; Turjanski, P.; Michinski, S.; Marshall, G. Tissue Damage Modeling in Gene Electrotransfer: The Role of PH. Bioelectrochemistry 2014, 100, 105–111. [Google Scholar] [CrossRef]
- Kandušer, M.; Miklavčič, D.; Pavlin, M. Mechanisms Involved in Gene Electrotransfer Using High- and Low-Voltage Pulses--an in Vitro Study. Bioelectrochemistry 2009, 74, 265–271. [Google Scholar] [CrossRef]
- Potočnik, T.; Sachdev, S.; Polajžer, T.; Lebar, A.M.; Miklavčič, D. Efficient Gene Transfection by Electroporation—In Vitro and In Silico Study of Pulse Parameters. Appl. Sci. 2022, 12, 8237. [Google Scholar] [CrossRef]
- Golzio, M.; Teissié, J.; Rols, M.P. Direct Visualization at the Single-Cell Level of Electrically Mediated Gene Delivery. Proc. Natl. Acad. Sci. USA 2002, 99, 1292–1297. [Google Scholar] [CrossRef] [PubMed]
- Golzio, M.; Teissié, J.; Rols, M.P. Cell Synchronization Effect on Mammalian Cell Permeabilization and Gene Delivery by Electric Field. Biochim. Biophys. Acta—Biomembr. 2002, 1563, 23–28. [Google Scholar] [CrossRef] [PubMed]
- Rosazza, C.; Deschout, H.; Buntz, A.; Braeckmans, K.; Rols, M.-P.; Zumbusch, A. Endocytosis and Endosomal Trafficking of DNA After Gene Electrotransfer In Vitro. Mol. Ther.—Nucleic Acids 2016, 5, e286. [Google Scholar] [CrossRef] [PubMed]
- Paganin-Gioanni, A.; Bellard, E.; Escoffre, J.M.; Rols, M.P.; Teissié, J.; Golzio, M. Direct Visualization at the Single-Cell Level of SiRNA Electrotransfer into Cancer Cells. Proc. Natl. Acad. Sci. USA 2011, 108, 10443–10447. [Google Scholar] [CrossRef]
- Reberšek, M.; Miklavčič, D.; Bertacchini, C.; Sack, M. Cell Membrane Electroporation-Part 3: The Equipment. IEEE Electr. Insul. Mag. 2014, 30, 8–18. [Google Scholar] [CrossRef]
- Pirc, E.; Reberšek, M.; Miklavčič, D. Dosimetry in Electroporation-Based Technologies and Treatments. In Dosimetry in Bioelectromagnetics; Markov, M., Ed.; CRC Press: Boca Raton, FL, USA, 2017; pp. 233–268. [Google Scholar] [CrossRef]
- Mahnič-Kalamiza, S.; Miklavčič, D. Scratching the Electrode Surface: Insights into a High-Voltage Pulsed-Field Application from in Vitro & in Silico Studies in Indifferent Fluid. Electrochim. Acta 2020, 363, 137187. [Google Scholar] [CrossRef]
- Vižintin, A.; Marković, S.; Ščančar, J.; Miklavčič, D. Electroporation with Nanosecond Pulses and Bleomycin or Cisplatin Results in Efficient Cell Kill and Low Metal Release from Electrodes. Bioelectrochemistry 2021, 140, 107798. [Google Scholar] [CrossRef]
- Turjanski, P.; Olaiz, N.; Maglietti, F.; Michinski, S.; Suárez, C.; Molina, F.V.; Marshall, G. The Role of PH Fronts in Reversible Electroporation. PLoS ONE 2011, 6, e17303. [Google Scholar] [CrossRef]
- Faroja, M.; Ahmed, M.; Appelbaum, L.; Ben-David, E.; Moussa, M.; Sosna, J.; Nissenbaum, I.; Nahum Goldberg, S. Irreversible Electroporation Ablation: Is All the Damage Nonthermal? Radiology 2013, 266, 462–470. [Google Scholar] [CrossRef]
- Klein, N.; Guenther, E.; Botea, F.; Pautov, M.; Dima, S.; Tomescu, D.; Popescu, M.; Ivorra, A.; Stehling, M.; Popescu, I. The Combination of Electroporation and Electrolysis (E2) Employing Different Electrode Arrays for Ablation of Large Tissue Volumes. PLoS ONE 2019, 14, e0221393. [Google Scholar] [CrossRef] [PubMed]
- Yao, C.; Dong, S.; Zhao, Y.; Lv, Y.; Liu, H.; Gong, L.; Ma, J.; Wang, H.; Sun, Y. Bipolar Microsecond Pulses and Insulated Needle Electrodes for Reducing Muscle Contractions During Irreversible Electroporation. IEEE Trans. Biomed. Eng. 2017, 64, 2924–2937. [Google Scholar] [CrossRef] [PubMed]
- Mercadal, B.; Arena, C.B.; Davalos, R.V.; Ivorra, A. Avoiding Nerve Stimulation in Irreversible Electroporation: A Numerical Modeling Study. Phys. Med. Biol. 2017, 62, 8060–8079. [Google Scholar] [CrossRef]
- Aycock, K.N.; Zhao, Y.; Lorenzo, M.F.; Davalos, R.V. A Theoretical Argument for Extended Interpulse Delays in Therapeutic High-Frequency Irreversible Electroporation Treatments. IEEE Trans. Biomed. Eng. 2021, 68, 1999–2010. [Google Scholar] [CrossRef]
- Cvetkoska, A.; Maček-Lebar, A.; Trdina, P.; Miklavčič, D.; Reberšek, M. Muscle Contractions and Pain Sensation Accompanying High-Frequency Electroporation Pulses. Sci. Rep. 2022, 12, 8019. [Google Scholar] [CrossRef] [PubMed]
- Chopinet, L.; Batista-Napotnik, T.; Montigny, A.; Rebersek, M.; Teissié, J.; Rols, M.P.; Miklavčič, D. Nanosecond Electric Pulse Effects on Gene Expression. J. Membr. Biol. 2013, 246, 851. [Google Scholar] [CrossRef] [PubMed]
- Guo, S.; Jackson, D.L.; Burcus, N.I.; Chen, Y.J.; Xiao, S.; Heller, R. Gene Electrotransfer Enhanced by Nanosecond Pulsed Electric Fields. Mol. Ther.—Methods Clin. Dev. 2014, 1, 14043. [Google Scholar] [CrossRef] [PubMed]
- Chang, A.Y.; Liu, X.; Tian, H.; Hua, L.; Yang, Z.; Wang, S. Microfluidic Electroporation Coupling Pulses of Nanoseconds and Milliseconds to Facilitate Rapid Uptake and Enhanced Expression of DNA in Cell Therapy. Sci. Rep. 2020, 10, 6061. [Google Scholar] [CrossRef]
- Pucihar, G.; Kotnik, T.; Kandušer, M.; Miklavčič, D. The Influence of Medium Conductivity on Electropermeabilization and Survival of Cells in Vitro. Bioelectrochemistry 2001, 54, 107–115. [Google Scholar] [CrossRef]
- Ruzgys, P.; Novickij, V.; Novickij, J.; Šatkauskas, S. Nanosecond Range Electric Pulse Application as a Non-Viral Gene Delivery Method: Proof of Concept. Sci. Rep. 2018, 8, 15502. [Google Scholar] [CrossRef]
- Novickij, V.; Balevičiūtė, A.; Ruzgys, P.; Šatkauskas, S.; Novickij, J.; Zinkevičienė, A.; Girkontaitė, I. Sub-Microsecond Electrotransfection Using New Modality of High Frequency Electroporation. Bioelectrochemistry 2020, 136, 107594. [Google Scholar] [CrossRef] [PubMed]
- Potočnik, T.; Miklavčič, D.; Maček Lebar, A. Effect of Electroporation and Recovery Medium PH on Cell Membrane Permeabilization, Cell Survival and Gene Transfer Efficiency in Vitro. Bioelectrochemistry 2019, 130, 107342. [Google Scholar] [CrossRef] [PubMed]
- Kotnik, T.; Kramar, P.; Pucihar, G.; Miklavčič, D.; Tarek, M. Cell Membrane Electroporation—Part 1: The Phenomenon. IEEE Electr. Insul. Mag. 2012, 28, 14–23. [Google Scholar] [CrossRef]
- Pavlin, M.; Pavšelj, N.; Miklavčič, D. Dependence of Induced Transmembrane Potential on Cell Density, Arrangement, and Cell Position inside a Cell System. IEEE Trans. Biomed. Eng. 2002, 49, 605–612. [Google Scholar] [CrossRef] [PubMed]
- Chicaybam, L.; Barcelos, C.; Peixoto, B.; Carneiro, M.; Limia, C.G.; Redondo, P.; Lira, C.; Paraguassú-Braga, F.; De Vasconcelos, Z.F.M.; Barros, L.; et al. An Efficient Electroporation Protocol for the Genetic Modification of Mammalian Cells. Front. Bioeng. Biotechnol. 2017, 4, 99. [Google Scholar] [CrossRef]
- Potočnik, T.; Miklavčič, D.; Maček Lebar, A. Gene Transfer by Electroporation with High Frequency Bipolar Pulses in Vitro. Bioelectrochemistry 2021, 140, 107803. [Google Scholar] [CrossRef]
- Chopra, S.; Ruzgys, P.; Maciulevičius, M.; Jakutavičiute, M.; Šatka, S. Investigation of Plasmid DNA Delivery and Cell Viability Dynamics for Optimal Cell Electrotransfection in Vitro. Appl. Sci. 2020, 10, 6070. [Google Scholar] [CrossRef]
- Kos, S.; Tesic, N.; Kamensek, U.; Blagus, T.; Cemazar, M.; Kranjc, S.; Lavrencak, J.; Sersa, G. Improved Specificity of Gene Electrotransfer to Skin Using PDNA Under the Control of Collagen Tissue-Specific Promoter. J. Membr. Biol. 2015, 248, 919–928. [Google Scholar] [CrossRef]
- Mikolajewicz, N.; Komarova, S.V. Meta-Analytic Methodology for Basic Research: A Practical Guide. Front. Physiol. 2019, 10, 203. [Google Scholar] [CrossRef]
- Andreason, G.L.; Evans, G.A. Optimization of Electroporation for Transfection of Mammalian Cell Lines. Anal. Biochem. 1989, 180, 269–275. [Google Scholar] [CrossRef]
- Liu, L.; Johnson, C.; Fujimura, S.; Teque, F.; Levy, J.A. Transfection Optimization for Primary Human CD8+ Cells. J. Immunol. Methods 2011, 372, 22–29. [Google Scholar] [CrossRef]
- Liu, Y.; Bergan, R. Improved Intracellular Delivery of Oligonucleotides by Square Wave Electroporation. Antisense Nucleic Acid Drug Dev. 2001, 11, 7–14. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.C.; Lin, W.Y.; Jhang, Y.R.; Huang, S.H.; Wu, C.P.; Wu, H.T. Efficiency of DNA Transfection of Rat Heart Myoblast Cells H9c2(2-1) by Either Polyethyleneimine or Electroporation. Appl. Biochem. Biotechnol. 2011, 164, 1172–1182. [Google Scholar] [CrossRef] [PubMed]
- Lojk, J.; Mis, K.; Pirkmajer, S.; Pavlin, M. SiRNA Delivery into Cultured Primary Human Myoblasts--Optimization of Electroporation Parameters and Theoretical Analysis. Bioelectromagnetics 2015, 36, 551–563. [Google Scholar] [CrossRef] [PubMed]
- Lundqvist, A.; Noffz, G.; Pavlenko, M.; Saebøe-Larssen, S.; Fong, T.; Maitland, N.; Pisa, P.; Sæbøe-Larssen, S.; Fong, T.; Maitland, N.; et al. Nonviral and Viral Gene Transfer Into Different Subsets of Human Dendritic Cells Yield Comparable Efficiency of Transfection. J. Immunother. 2002, 25, 445–454. [Google Scholar] [CrossRef]
- Madeira, C.; Ribeiro, S.C.; Pinheiro, I.S.M.M.; Martins, S.A.M.M.; Andrade, P.Z.; da Silva, C.L.; Cabral, J.M.S.S. Gene Delivery to Human Bone Marrow Mesenchymal Stem Cells by Microporation. J. Biotechnol. 2011, 151, 130–136. [Google Scholar] [CrossRef]
- Madeira, C.; Ribeiro, S.C.C.; Turk, M.Z.Z.; Cabral, J.M.S.M.S. Optimization of Gene Delivery to HEK293T Cells by Microporation Using a Central Composite Design Methodology. Biotechnol. Lett. 2010, 32, 1393–1399. [Google Scholar] [CrossRef]
- Madeira, C.; Rodrigues, C.A.V.; Reis, M.S.C.; Ferreira, F.F.C.G.; Correia, R.E.S.M.; Diogo, M.M.; Cabral, J.M.S. Nonviral Gene Delivery to Neural Stem Cells with Minicircles by Microporation. Biomacromolecules 2013, 14, 1379–1387. [Google Scholar] [CrossRef]
- Marine, S.; Freeman, J.; Riccio, A.; Axenborg, M.-L.; Pihl, J.; Ketteler, R.; Aspengren, S. High-Throughput Transfection of Differentiated Primary Neurons from Rat Forebrain. J. Biomol. Screen. 2012, 17, 692–696. [Google Scholar] [CrossRef][Green Version]
- Marjanovič, I.; Haberl, S.S.; Miklavčič, D.; Kandušer, M.; Pavlin, M.; Marjanovic, I.; Haberl, S.S.; Miklavcic, D.; Kanduser, M.; Pavlin, M.; et al. Analysis and Comparison of Electrical Pulse Parameters for Gene Electrotransfer of Two Different Cell Lines. J. Membr. Biol. 2010, 236, 97–105. [Google Scholar] [CrossRef]
- Bodwell, J.; Swift, F.; Richardson, J. Long Duration Electroporation for Achieving High Level Expression of Glucocorticoid Receptors in Mammalian Cell Lines. J. Steroid Biochem. Mol. Biol. 1999, 68, 77–82. [Google Scholar] [CrossRef] [PubMed]
- Marjanovič, I.; Kandušer, M.; Miklavčič, D.; Keber, M.M.M.M.; Pavlin, M. Comparison of Flow Cytometry, Fluorescence Microscopy and Spectrofluorometry for Analysis of Gene Electrotransfer Efficiency. J. Membr. Biol. 2014, 247, 1259–1267. [Google Scholar] [CrossRef] [PubMed]
- Markelc, B.; Tevz, G.; Cemazar, M.; Kranjc, S.; Lavrencak, J.; Zegura, B.; Teissie, J.; Sersa, G. Muscle Gene Electrotransfer Is Increased by the Antioxidant Tempol in Mice. Gene Ther. 2012, 19, 312–320. [Google Scholar] [CrossRef]
- Markowicz, S.; Niedzielska, J.; Kruszewski, M.; Ołdak, T.; Gajkowska, A.; Machaj, E.K.; Skurzak, H.; Pojda, Z. Nonviral Transfection of Human Umbilical Cord Blood Dendritic Cells Is Feasible, but the Yield of Dendritic Cells with Transgene Expression Limits the Application of This Method in Cancer Immunotherapy. Acta Biochim. Pol. 2006, 53, 203–212. [Google Scholar] [CrossRef]
- Mars, T.; Strazisar, M.; Mis, K.; Kotnik, N.; Pegan, K.; Lojk, J.; Grubic, Z.; Pavlin, M. Electrotransfection and Lipofection Show Comparable Efficiency for In Vitro Gene Delivery of Primary Human Myoblasts. J. Membr. Biol. 2015, 248, 273–283. [Google Scholar] [CrossRef]
- May, R.D.R.D.R.D.; Tekari, A.; Frauchiger, D.A.D.A.; Krismer, A.; Benneker, L.M.L.M.; Gantenbein, B. Efficient Nonviral Transfection of Primary Intervertebral Disc Cells by Electroporation for Tissue Engineering Application. Tissue Eng. Part C Methods 2017, 23, 30–37. [Google Scholar] [CrossRef] [PubMed]
- Mignet, N.; Vandermeulen, G.; Pembouong, G.; Largeau, C.; Thompson, B.; Spanedda, M.-V.; Wasungu, L.; Rols, M.-P.; Bessodes, M.; Bureau, M.F.; et al. Cationic and Anionic Lipoplexes Inhibit Gene Transfection by Electroporation in Vivo. J. Gene Med. 2010, 12, 491–500. [Google Scholar] [CrossRef] [PubMed]
- Mohr, J.C.J.C.; de Pablo, J.J.J.J.; Palecek, S.P.S.P.; DePablo, J.J.; Palecek, S.P.S.P. Electroporation of Human Embryonic Stem Cells: Small and Macromolecule Loading and DNA Transfection. Biotechnol. Prog. 2006, 22, 825–834. [Google Scholar] [CrossRef]
- Müller, K.J.K.J.; Horbaschek, M.; Lucas, K.; Zimmermann, U.; Sukhorukov, V.L.V.L. Electrotransfection of Anchorage-Dependent Mammalian Cells. Exp. Cell Res. 2003, 288, 344–353. [Google Scholar] [CrossRef]
- Mun, J.-Y.; Shin, K.K.; Kwon, O.; Lim, Y.T.; Oh, D.-B. Minicircle Microporation-Based Non-Viral Gene Delivery Improved the Targeting of Mesenchymal Stem Cells to an Injury Site. Biomaterials 2016, 101, 310–320. [Google Scholar] [CrossRef]
- Nakayama, A.; Sato, M.; Shinohara, M.; Matsubara, S.; Yokomine, T.; Akasaka, E.; Yoshida, M.; Takao, S. Efficient Transfection of Primarily Cultured Porcine Embryonic Fibroblasts Using the Amaxa Nucleofection SystemTM. Cloning Stem. Cells. 2007, 9, 523–534. [Google Scholar] [CrossRef] [PubMed]
- Bosnjak, M.; Jesenko, T.; Kamensek, U.; Sersa, G.; Lavrencak, J.; Heller, L.; Cemazar, M. Electrotransfer of Different Control Plasmids Elicits Different Antitumor Effectiveness in B16.F10 Melanoma. Cancers 2018, 10, 37. [Google Scholar] [CrossRef] [PubMed]
- Niakan, S.; Heidari, B.; Akbari, G.; Nikousefat, Z. Comparison of Different Electroporation Parameters on Transfection Efciency of Sheep Testicular Cells. Cell J. 2016, 18, 425–437. [Google Scholar] [PubMed]
- Nickoloff, J.A.; Spirio, L.N.; Reynolds, R.J. A Comparison of Calcium Phosphate Coprecipitation and Electroporation. Mol. Biotechnol. 1998, 10, 93–101. [Google Scholar] [CrossRef]
- Ohse, M.; Tsuchida, K.; Tomita, H.; Taketo, A.; Kimoto, H.; Kusaoke, H. A New and Efficient Method for Gene Transfer into Mouse FM3A Cells Using Metaphase Chromosomes by Electroporation. Biosci. Biotechnol. Biochem. 1996, 60, 1879–1881. [Google Scholar] [CrossRef]
- Ołdak, T.; Kruszewski, M.; Machaj, E.K.; Gajkowska, A.; Pojda, Z. Optimisation of Transfection Conditions of CD34+ Hematopoietic Cells Derived from Human Umbilical Cord Blood. Acta Biochim. Pol. 2002, 49, 625–632. [Google Scholar] [CrossRef]
- Orio, J.; Bellard, E.; Baaziz, H.; Pichon, C.; Mouritzen, P.; Rols, M.-P.M.-P.; Teissié, J.; Golzio, M.; Chabot, S. Sub-Cellular Temporal and Spatial Distribution of Electrotransferred LNA/DNA Oligomer. J. RNAi Gene Silencing 2013, 9, 479–485. [Google Scholar]
- Pasquet, L.; Bellard, E.; Rols, M.P.; Golzio, M.; Teissie, J. Post-Pulse Addition of Trans-Cyclohexane-1,2-Diol Improves Electrotransfer Mediated Gene Expression in Mammalian Cells. Biochem. Biophys. Rep. 2016, 7, 287–294. [Google Scholar] [CrossRef]
- Pavlin, M.; Flisar, K.; Kandušer, M.; Kanduser, M.; Kandušer, M.; Kanduser, M. The Role of Electrophoresis in Gene Electrotransfer. J. Membr. Biol. 2010, 236, 75–79. [Google Scholar] [CrossRef]
- Pavlin, M.; Kandušer, M. New Insights into the Mechanisms of Gene Electrotransfer—Experimental and Theoretical Analysis. Sci. Rep. 2015, 5, 9132. [Google Scholar] [CrossRef]
- Pelofy, S.; Teissié, J.; Golzio, M.; Chabot, S. Chemically Modified Oligonucleotide–Increased Stability Negatively Correlates with Its Efficacy Despite Efficient Electrotransfer. J. Membr. Biol. 2012, 245, 565–571. [Google Scholar] [CrossRef] [PubMed]
- Peng, H.; Wu, Y.; Zhang, Y. Efficient Delivery of DNA and Morpholinos into Mouse Preimplantation Embryos by Electroporation. PLoS ONE 2012, 7, e43748. [Google Scholar] [CrossRef] [PubMed]
- Bosnjak, M.; Lorente, B.C.; Pogacar, Z.; Makovsek, V.; Cemazar, M. Different Incubation Times of Cells After Gene Electrotransfer in Fetal Bovine Serum Affect Cell Viability, but Not Transfection Efficiency. J. Membr. Biol. 2014, 247, 421–428. [Google Scholar] [CrossRef] [PubMed]
- Peng, L.; Xiong, W.; Cai, Y.; Chen, Y.; He, Y.; Yang, J.; Jin, J.; Li, H. A Simple, Rapid Method for Evaluation of Transfection Efficiency Based on Fluorescent Dye. Bioengineered 2017, 8, 225–231. [Google Scholar] [CrossRef] [PubMed]
- Prosen, L.; Markelc, B.; Dolinsek, T.; Music, B.; Cemazar, M.; Sersa, G. Mcam Silencing With RNA Interference Using Magnetofection Has Antitumor Effect in Murine Melanoma. Mol. Ther. Nucleic Acids 2014, 3, e205. [Google Scholar] [CrossRef] [PubMed]
- Rebersek, M.; Kanduser, M.; Miklavcic, D. Pipette Tip with Integrated Electrodes for Gene Electrotransfer of Cells in Suspension: A Feasibility Study in CHO Cells. Radiol. Oncol. 2011, 45, 204–208. [Google Scholar] [CrossRef]
- Regnier, V.; Le Doan, T.; Préat, V. Parameters Controlling Topical Delivery of Oligonucleotides by Electroporation. J. Drug Target. 1998, 5, 275–289. [Google Scholar] [CrossRef]
- Rizk, A.; Rabie, B.M. Electroporation for Transfection and Differentiation of Dental Pulp Stem Cells. Biores. Open Access 2013, 2, 155–162. [Google Scholar] [CrossRef]
- Rols, M.-P.; Coulet, D.; Teissie, J. Highly Efficient Transfection of Mammalian Cells by Electric Field Pulses. Application to Large Volumes of Cell Culture by Using a Flow System. Eur. J. Biochem. 1992, 206, 115–121. [Google Scholar] [CrossRef]
- Rols, M.-P.; Delteil, C.; Golzio, M.; Teissié, J. In Vitro and Ex Vivo Electrically Mediated Permeabilization and Gene Transfer in Murine Melanoma. Bioelectrochemistry Bioenerg. 1998, 47, 129–134. [Google Scholar] [CrossRef]
- Ross, J.W.; Whyte, J.J.; Zhao, J.; Samuel, M.; Wells, K.D.; Prather, R.S. Optimization of Square-Wave Electroporation for Transfection of Porcine Fetal Fibroblasts. Transgenic Res. 2010, 19, 611–620. [Google Scholar] [CrossRef] [PubMed]
- Ruybal, P.; Gravisaco, M.J.; Barcala, V.; Escalada, A.; Cremaschi, G.; Taboga, O.; Waldner, C.; Mongini, C. Transgene Expression Enhancement in T-Lymphoma Cell Lines. Int. Immunopharmacol. 2005, 5, 1685–1692. [Google Scholar] [CrossRef] [PubMed]
- Salimzadeh, L.; Jaberipour, M.; Hosseini, A.; Ghaderi, A. Non-Viral Transfection Methods Optimized for Gene Delivery to a Lung Cancer Cell Line. Avicenna J. Med. Biotechnol. 2013, 5, 68–77. [Google Scholar]
- Bradshaw, H.D.; Parson, W.W.; Sheffer, M.; Lioubin, P.J.; Mulvihill, E.R.; Gordon, M.P. Design, Construction, and Use of an Electroporator for Plant Protoplasts and Animal Cells. Anal. Biochem. 1987, 166, 342–348. [Google Scholar] [CrossRef] [PubMed]
- Satyabhama, S.; Epstein, A.L. Short-Term Efficient Expression of Transfected DNA in Human Hematopoietic Cells by Electroporation: Definition of Parameters and Use of Chemical Stimulators. DNA 1988, 7, 203–209. [Google Scholar] [CrossRef]
- Schakowski, F.; Buttgereit, P.; Mazur, M.; Märten, A.; Schöttker, B.; Gorschlüter, M.; Schmidt-Wolf, I.G.H. Novel Non-Viral Method for Transfection of Primary Leukemia Cells and Cell Lines. Genet. Vaccines Ther. 2004, 2, 1. [Google Scholar] [CrossRef] [PubMed]
- Schakowski, F.; Gorschlüter, M.; Junghans, C.; Schroff, M.; Buttgereit, P.; Ziske, C.; Schöttker, B.; König-Merediz, S.A.; Sauerbruch, T.; Wittig, B.; et al. A Novel Minimal-Size Vector (MIDGE) Improves Transgene Expression in Colon Carcinoma Cells and Avoids Transfection of Undesired DNA. Mol. Ther. 2001, 3, 793–800. [Google Scholar] [CrossRef]
- Seo, H.W.; Kim, T.M.; Choi, J.W.; Han, B.K.; Song, G.; Han, J.Y. Evaluation of Combinatorial Cis-Regulatory Elements for Stable Gene Expression in Chicken Cells. BMC Biotechnol. 2010, 10, 69. [Google Scholar] [CrossRef]
- Smirnikhina, S.A.; Lavrov, A.V.; Bochkov, N.P. Dynamics of Elimination of Plasmids and Expression of VEGF121 Gene Transfected into Human Mesenchymal Stem Cells by Different Methods. Bull. Exp. Biol. Med. 2011, 151, 121–125. [Google Scholar] [CrossRef]
- Spandidos, D.A.D.A. Electric Field-Mediated Gene Transfer (Electroporation) into Mouse Friend and Human K562 Erythroleukemic Cells. Gene Anal. Tech. 1987, 4, 50–56. [Google Scholar] [CrossRef]
- Stacey, K.J.; Ross, I.A.N.L.; Hume, D.A. Electroporation and DNA-Dependent Cell Death in Murine Macrophages. Immunol. Cell. Biol. 1993, 71, 75–85. [Google Scholar] [CrossRef] [PubMed]
- Stopper, H.; Jones, H.; Zimmermann, U. Large Scale Transfection of Mouse L-Cells by Electropermeabilization. BBA—Biomembr. 1987, 900, 38–44. [Google Scholar] [CrossRef] [PubMed]
- Stopper, H.; Zimmermann, U.; Neil, G.A. Increased Efficiency of Transfection of Murine Hybridoma Cells with DNA by Electropermeabilization. J. Immunol. Methods 1988, 109, 145–151. [Google Scholar] [CrossRef] [PubMed]
- Stopper, H.; Zimmermann, U.; Wecker, E. High Yields of DNA-Transfer Into Mouse L-Cells by Electropermeabilization. Z. Fur Naturforsch.—Sect. C J. Biosci. 1985, 40, 929–932. [Google Scholar] [CrossRef]
- Bravo, S.B.; Garcia-Rendueles, M.E.R.R.; Perez-Romero, S.; Cameselle-Teijeiro, J.; Rodrigues, J.S.; Barreiro, F.; Alvarez, C. V Expression of Exogenous Proteins and Short Hairpin RNAs in Human Primary Thyrocytes. Anal. Biochem. 2010, 400, 219–228. [Google Scholar] [CrossRef]
- Stroh, T.; Erben, U.; Kühl, A.A.A.A.; Zeitz, M.; Siegmund, B. Combined Pulse Electroporation–A Novel Strategy for Highly Efficient Transfection of Human and Mouse Cells. PLoS ONE 2010, 5, e9488. [Google Scholar] [CrossRef]
- Sukharev, S.I.; Klenchin, V.A.; Serov, S.M.; Chernomordik, L.V.; Chizmadzhev, Y.A. Electroporation and Electrophoretic DNA Transfer into Cells. The Effect of DNA Interaction with Electropores. Biophys. J. 1992, 63, 1320–1327. [Google Scholar] [CrossRef]
- Sungailaitė, S.; Ruzgys, P.; Šatkauskienė, I.; Čepurnienė, K.; Šatkauskas, S. The Dependence of Efficiency of Transmembrane Molecular Transfer Using Electroporation on Medium Viscosity. J. Gene Med. 2015, 17, 80–86. [Google Scholar] [CrossRef]
- Tabar, M.S.; Hesaraki, M.; Esfandiari, F.; Samani, F.S.; Vakilian, H.; Baharvand, H.; Sharifi Tabar, M.; Hesaraki, M.; Esfandiari, F.; Sahraneshin Samani, F.; et al. Evaluating Electroporation and Lipofectamine Approaches for Transient and Stable Transgene Expressions in Human Fibroblasts and Embryonic Stem Cells. Cell J. 2015, 17, 438–450. [Google Scholar]
- Takahashi, M.; Furukawa, T.; Saitoh, H.; Aoki, A.; Koike, T.; Moriyama, Y.; Shibata, A. Gene Transfer into Human Leukemia Cell Lines by Electroporation: Experience with Exponentially Decaying and Square Wave Pulse. Leuk. Res. 1991, 15, 507–513. [Google Scholar] [CrossRef]
- Tanaka, M.; Yanagawa, Y.; Hirashima, N. Transfer of Small Interfering RNA by Single-Cell Electroporation in Cerebellar Cell Cultures. J. Neurosci. Methods 2009, 178, 80–86. [Google Scholar] [CrossRef] [PubMed]
- Taylor, W.; Gokay, K.E.; Capaccio, C.; Davis, E.; Glucksberg, M.; Dean, D.A. The Effects of Cyclic Stretch on Gene Transfer in Alveolar Epithelial Cells. Mol. Ther. 2003, 7, 542–549. [Google Scholar] [CrossRef]
- Teifel, M.; Heine, L.-T.; Milbredt, S.; Friedl, P. Optimization of Transfection of Human Endothelial Cells. Endothelium 1997, 5, 21–35. [Google Scholar] [CrossRef] [PubMed]
- Tekle, E.; Astumian, R.D.; Chock, P.B. Electroporation by Using Bipolar Oscillating Electric Field: An Improved Method for DNA Transfection of NIH 3T3 Cells. Proc. Natl. Acad. Sci. USA 1991, 88, 4230–4234. [Google Scholar] [CrossRef] [PubMed]
- Tesic, N.; Cemazar, M. In Vitro Targeted Gene Electrotransfer to Endothelial Cells with Plasmid DNA Containing Human Endothelin-1 Promoter. J. Membr. Biol. 2013, 246, 783–791. [Google Scholar] [CrossRef]
- Browne, C.J.; Pinyon, J.L.; Housley, D.M.; Crawford, E.N.; Lovell, N.H.; Klugmann, M.; Housley, G.D. Mapping of Bionic Array Electric Field Focusing in Plasmid DNA-Based Gene Electrotransfer. Gene Ther. 2016, 23, 369–379. [Google Scholar] [CrossRef]
- Tur-Kaspa, R.; Teicher, L.; Levine, B.J.; Skoultchi, A.I.; Shafritz, D.A. Use of Electroporation to Introduce Biologically Active Foreign Genes into Primary Rat Hepatocytes. Mol. Cell. Biol. 1986, 6, 716–718. [Google Scholar]
- Underhill, M.F.; Coley, C.; Birch, J.R.; Findlay, A.; Kallmeier, R.; Proud, C.G.; James, D.C. Engineering MRNA Translation Initiation to Enhance Transient Gene Expression in Chinese Hamster Ovary Cells. Biotechnol. Prog. 2003, 19, 121–129. [Google Scholar] [CrossRef] [PubMed]
- Van Gaal, E.V.B.; Oosting, R.S.; Van Eijk, R.; Bakowska, M.; Feyen, D.; Kok, R.J.; Hennink, W.E.; Crommelin, D.J.A.; Mastrobattista, E. DNA Nuclear Targeting Sequences for Non-Viral Gene Delivery. Pharm. Res. 2011, 28, 1707–1722. [Google Scholar] [CrossRef]
- van Leeuwen, E.B.M.B.; van der Veen, A.Y.Y.; Hoekstra, D.; Engberts, J.B.F.N.B.; Halie, M.R.R.; Van Der Meer, J.; Ruiters, M.H.J.H. Transfection of Small Numbers of Human Endothelial Cells by Electroporation and Synthetic Amphiphiles. Eur. J. Vasc. Endovasc. Surg. 1999, 17, 9–14. [Google Scholar] [CrossRef]
- Van Meirvenne, S.; Straetman, L.; Heirman, C.; Dullaers, M.; De Greef, C.; Van Tendeloo, V.; Thielemans, K. Efficient Genetic Modification of Murine Dendritic Cells by Electroporation with MRNA. Cancer Gene Ther. 2002, 9, 787–797. [Google Scholar] [CrossRef] [PubMed]
- Van Tendeloo, V.F.I.; Ponsaerts, P.; Lardon, F.; Nijs, G.; Lenjou, M.; Van Broeckhoven, C.; Van Bockstaele, D.R.; Berneman, Z.N. Highly Efficient Gene Delivery by MRNA Electroporation in Human Hematopoietic Cells: Superiority to Lipofection and Passive Pulsing of MRNA and to Electroporation of Plasmid CDNA for Tumor Antigen Loading of Dendritic Cells. Blood 2001, 98, 49–56. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Zhang, X.; Wang, W.; Lee, L.J. Semicontinuous Flow Electroporation Chip for High-Throughput Transfection on Mammalian Cells. Anal. Chem. 2009, 81, 4414–4421. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Wasungu, L.; Escoffre, J.-M.; Valette, A.; Teissie, J.; Rols, M.-P. A 3D in Vitro Spheroid Model as a Way to Study the Mechanisms of Electroporation. Int. J. Pharm. 2009, 379, 278–284. [Google Scholar] [CrossRef]
- Wasungu, L.; Pillet, F.; Bellard, E.; Rols, M.-P.; Teissié, J. Shock Waves Associated with Electric Pulses Affect Cell Electro-Permeabilization. Bioelectrochemistry 2014, 100, 36–43. [Google Scholar] [CrossRef]
- Wawrzynska, M.; Bednarczyk, M.; Łakota, P.; Lubiszewska, M. Influence of Electroporation on Chicken Blastoderm Cell Viability in Vitro. Folia Biol. 2008, 56, 197–201. [Google Scholar] [CrossRef][Green Version]
- Buchser, W.J.W.J.; Pardinas, J.R.J.R.; Shi, Y.; Bixby, J.L.J.L.J.L.; Lemmon, V.P.V.P. 96-Well Electroporation Method for Transfection of Mammalian Central Neurons. Biotechniques 2006, 41, 619–624. [Google Scholar] [CrossRef]
- Wei, Z.; Li, X.; Zhao, D.; Yan, H.; Hu, Z.; Liang, Z.; Li, Z. Flow-Through Cell Electroporation Microchip Integrating Dielectrophoretic Viable Cell Sorting. Anal. Chem. 2014, 86, 10215–10222. [Google Scholar] [CrossRef]
- Wei, Z.; Zhao, D.; Li, X.; Wu, M.; Wang, W.; Huang, H.; Wang, X.; Du, Q.; Liang, Z.; Li, Z. A Laminar Flow Electroporation System for Efficient DNA and SiRNA Delivery. Anal. Chem. 2011, 83, 5881–5887. [Google Scholar] [CrossRef]
- Wei, Z.-L.; Ogawa, R.; Takasaki, I.; Zhao, Q.-L.; Zheng, H.-C.; Ahmed, K.; Hassan, M.A.; Kondo, T. Mild Hyperthermia Prior to Electroporation Increases Transfection Efficiency in HCT 116, HeLa S3 and SGC 7901 Cells. Biotechnol. Lett. 2010, 32, 367–371. [Google Scholar] [CrossRef]
- Wells, J.M.M.; Li, L.H.H.; Sen, A.; Jahreis, G.P.P.; Hui, S.W.W. Electroporation-Enhanced Gene Delivery in Mammary Tumors. Gene Ther. 2000, 7, 541–547. [Google Scholar] [CrossRef] [PubMed]
- Will, A.; Röllinghoff, M.; Gessner, A. Stable Transfection of Cloned Murine T Helper Cells. J. Immunol. Methods 1995, 188, 139–146. [Google Scholar] [CrossRef] [PubMed]
- Wolf, H.; Rols, M.P.; Boldt, E.; Neumann, E.; Teissié, J. Control by Pulse Parameters of Electric Field-Mediated Gene Transfer in Mammalian Cells. Biophys. J. 1994, 66, 524–531. [Google Scholar] [CrossRef]
- Wu, M.; Yuan, F. Membrane Binding of Plasmid DNA and Endocytic Pathways Are Involved in Electrotransfection of Mammalian Cells. PLoS ONE 2011, 6, e20923. [Google Scholar] [CrossRef]
- Xu, Y.; Lu, Y.; Xing, W. An Individually Addressable Suspended-Drop Electroporation System for High-Throughput Cell Transfection. Lab Chip 2014, 14, 686–690. [Google Scholar] [CrossRef]
- Xu, Y.; Su, S.; Zhou, C.; Lu, Y.; Xing, W. Cell Electroporation with a Three-Dimensional Microelectrode Array on a Printed Circuit Board. Bioelectrochemistry 2015, 102, 35–41. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.-C.; Huang, K.-S.; Chen, H.-Y.; Lin, Y.-C. Determination of Optimum Gene Transfection Conditions Using the Taguchi Method for an Electroporation Microchip. Sens. Actuators B Chem. 2008, 132, 551–557. [Google Scholar] [CrossRef]
- Bulysheva, A.A.A.; Burcus, N.; Lundberg, C.; Edelblute, C.M.C.M.; Francis, M.P.M.P.; Heller, R. Recellularized Human Dermis for Testing Gene Electrotransfer Ex Vivo. Biomed. Mater. 2016, 11, 35002. [Google Scholar] [CrossRef] [PubMed]
- Yao, S.; Rana, S.; Liu, D.; Wise, G.E.G.E. Electroporation Optimization to Deliver Plasmid DNA into Dental Follicle Cells. Biotechnol. J. 2009, 4, 1488–1496. [Google Scholar] [CrossRef]
- Yu, L.; Reynaud, F.; Falk, J.; Spencer, A.; Ding, Y.-D.Y.-D.; Baumlé, V.; Lu, R.; Castellani, V.; Rudkin, B.B.B.B.; Yuan, C.; et al. Highly Efficient Method for Gene Delivery into Mouse Dorsal Root Ganglia Neurons. Front. Mol. Neurosci. 2015, 8, 2. [Google Scholar] [CrossRef]
- Zhang, Z.; Qiu, S.; Zhang, X.; Chen, W. Optimized DNA Electroporation for Primary Human T Cell Engineering. BMC Biotechnol. 2018, 18, 4. [Google Scholar] [CrossRef] [PubMed]
- Zhou, R.; Norton, J.E.; Zhang, N.; Dean, D.A. Electroporation-Mediated Transfer of Plasmids to the Lung Results in Reduced TLR9 Signaling and Inflammation. Gene Ther. 2007, 14, 775–780. [Google Scholar] [CrossRef] [PubMed]
- Zizzi, A.; Minardi, D.; Ciavattini, A.; Giantomassi, F.; Montironi, R.; Muzzonigro, G.; Di Primio, R.; Lucarini, G. Green Fluorescent Protein as Indicator of Nonviral Transient Transfection Efficiency in Endometrial and Testicular Biopsies. Microsc Res Tech. 2010, 73, 229–233. [Google Scholar] [CrossRef] [PubMed]
- Znidar, K.; Bosnjak, M.; Semenova, N.; Pakhomova, O.; Heller, L.; Cemazar, M. Tumor Cell Death after Electrotransfer of Plasmid DNA Is Associated with Cytosolic DNA Sensor Upregulation. Oncotarget 2018, 9, 18665–18681. [Google Scholar] [CrossRef] [PubMed]
- Sieni, E.; Dettin, M.; De Robertis, M.; Bazzolo, B.; Conconi, M.T.; Zamuner, A.; Marino, R.; Keller, F.; Campana, L.G.; Signori, E. The Efficiency of Gene Electrotransfer in Breast-Cancer Cell Lines Cultured on a Novel Collagen-Free 3D Scaffold. Cancers 2020, 12, 1043. [Google Scholar] [CrossRef]
- Pinyon, J.L.; Klugmann, M.; Lovell, N.H.; Housley, G.D. Dual-Plasmid Bionic Array-Directed Gene Electrotransfer in HEK293 Cells and Cochlear Mesenchymal Cells Probes Transgene Expression and Cell Fate. Hum. Gene Ther. 2019, 30, 211–224. [Google Scholar] [CrossRef]
- Salkin, H.; Gönen, Z.B.; Ergen, E.; Bahar, D.; Çetin, M. Effects of TGF-β1 Overexpression on Biological Characteristics of Human Dental Pulp-Derived Mesenchymal Stromal Cells. Int. J. Stem Cells 2019, 12, 170–182. [Google Scholar] [CrossRef]
- Hyder, I.; Eghbalsaied, S.; Kues, W.A. Systematic Optimization of Square-Wave Electroporation Conditions for Bovine Primary Fibroblasts. BMC Mol. Cell Biol. 2020, 21, 9. [Google Scholar] [CrossRef]
- Bureau, M.F.F.; Gehl, J.; Deleuze, V.; Mir, L.M.M.; Scherman, D. Importance of Association between Permeabilization and Electrophoretic Forces for Intramuscular DNA Electrotransfer. Biochim. Biophys. Acta (BBA)—Gen. Subj. 2000, 1474, 353–359. [Google Scholar] [CrossRef]
- Sherba, J.J.; Hogquist, S.; Lin, H.; Shan, J.W.; Shreiber, D.I.; Zahn, J.D. The Effects of Electroporation Buffer Composition on Cell Viability and Electro-Transfection Efficiency. Sci. Rep. 2020, 10, 3053. [Google Scholar] [CrossRef]
- Heller, L.; Bulysheva, A.; Arpag, S.; Sales Conniff, A.; Kohena, K.; Shi, G.; Semenova, N.; Heller, R.; Cemazar, M. Growth Environment Influences B16.F10 Mouse Melanoma Cell Response to Gene Electrotransfer. Bioelectrochemistry 2021, 140, 107827. [Google Scholar] [CrossRef] [PubMed]
- Keyer, V.V.; Shevtsov, A.B.; Zaripov, M.M.; Baltabekova, A.Z.; Ramanculov, E.M.; Shustov, A. V Towards Development of Plasmacytoma Cells-Based Expression Systems Utilizing Alphavirus Vectors: An NS0-VEE Model. J. Virol. Methods 2019, 274, 113734. [Google Scholar] [CrossRef] [PubMed]
- Lissandrello, C.A.; Santos, J.A.; Hsi, P.; Welch, M.; Mott, V.L.; Kim, E.S.; Chesin, J.; Haroutunian, N.J.; Stoddard, A.G.; Czarnecki, A.; et al. High-Throughput Continuous-Flow Microfluidic Electroporation of MRNA into Primary Human T Cells for Applications in Cellular Therapy Manufacturing. Sci. Rep. 2020, 10, 18045. [Google Scholar] [CrossRef] [PubMed]
- Tereshchenko, V.; Bulygin, A.; Zavodskii, R.; Maksyutov, A.; Kurilin, V.; Fisher, M.; Semenyuk, N.; Aladev, S.; Sennikov, S. The Murine DCs Transfected with DNA-Plasmid Encoding CCR9 Demonstrate the Increased Migration to CCL25 and Thymic Cells in Vitro and to the Thymus in Vivo. Cytokine 2021, 142, 155473. [Google Scholar] [CrossRef]
- Zu, Y.; Liu, X.; Chang, A.Y.; Wang, S. Flow Micropillar Array Electroporation to Enhance Size Specific Transfection to a Large Population of Cells. Bioelectrochemistry 2020, 132, 107417. [Google Scholar] [CrossRef]
- Campillo-Davo, D.; Fujiki, F.; Van Den Bergh, J.M.J.; De Reu, H.; Smits, E.L.J.M.; Goossens, H.; Sugiyama, H.; Lion, E.; Berneman, Z.N.; Van Tendeloo, V. Efficient and Non-Genotoxic RNA-Based Engineering of Human T Cells Using Tumor-Specific t Cell Receptors with Minimal TCR Mispairing. Front. Immunol. 2018, 9, 2503. [Google Scholar] [CrossRef]
- Yudovich, D.; Bäckström, A.; Schmiderer, L.; Žemaitis, K.; Subramaniam, A.; Larsson, J. Combined Lentiviral- and RNA-Mediated CRISPR/Cas9 Delivery for Efficient and Traceable Gene Editing in Human Hematopoietic Stem and Progenitor Cells. Sci. Rep. 2020, 10, 22393. [Google Scholar] [CrossRef]
- Von Der Haar, K.; Jonczyk, R.; Lavrentieva, A.; Weyand, B.; Vogt, P.; Jochums, A.; Stahl, F.; Scheper, T.; Blume, C.A. Electroporation: A Sustainable and Cell Biology Preserving Cell Labeling Method for Adipogenous Mesenchymal Stem Cells. Biores. Open Access 2019, 8, 32–44. [Google Scholar] [CrossRef]
- Nowaczyk, M.; Malcher, A.; Zimna, A.; Łabędź, W.; Kubaszewski, Ł.; Fiedorowicz, K.; Wierzbiński, K.; Rozwadowska, N.; Kurpisz, M. Transient and Stable Overexpression of Extracellular Superoxide Dismutase Is Positively Associated with the Myogenic Function of Human Skeletal Muscle-Derived Stem/Progenitor Cells. Antioxidants 2020, 9, 817. [Google Scholar] [CrossRef]
- Burrin, J.M.; Jameson, J.L. Regulation of Transfected Glycoprotein Hormone Alpha-Gene Expression in Primary Pituitary Cell Cultures. Mol. Endocrinol. 1989, 3, 1643–1651. [Google Scholar] [CrossRef]
- Yu, X.; Fang, X.; Xiao, H.; Zhao, Z.; Maak, S.; Wang, M.; Yang, R. The Effect of Acyl-CoA Synthetase Long-Chain Family Member 5 on Triglyceride Synthesis in Bovine Preadipocytes. Arch. Anim. Breed. 2019, 62, 257–264. [Google Scholar] [CrossRef] [PubMed]
- Gaebler, A.M.; Hennig, A.; Buczolich, K.; Weitz, J.; Welsch, T.; Stange, D.E.; Pape, K. Universal and Efficient Electroporation Protocol for Genetic Engineering of Gastrointestinal Organoids. J. Vis. Exp. 2020, 2020, e60704. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Zu, Y.; Wang, S. Cell Size-Specific Transfection by Micropillar Array Electroporation. In Methods in Molecular Biology; Humana Press Inc.: Totowa, NJ, USA, 2020; Volume 2050, pp. 3–12. [Google Scholar]
- Ishino, T.; Hashimoto, M.; Amagasa, M.; Saito, N.; Dochi, O.; Kirisawa, R.; Kitamura, H. Establishment of Protocol for Preparation of Gene-Edited Bovine Ear-Derived Fibroblasts for Somatic Cell Nuclear Transplantation. Biomed. Res. 2018, 39, 95–104. [Google Scholar] [CrossRef]
- Kopru, C.Z.; Cagnan, I.; Akar, I.; Esendagli, G.; Korkusuz, P.; Gunel-Ozcan, A. Dual Effect of Glucocorticoid-Induced Tumor Necrosis Factor–Related Receptor Ligand Carrying Mesenchymal Stromal Cells on Small Cell Lung Cancer: A Preliminary in Vitro Study. Cytotherapy 2018, 20, 930–940. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Lu, Y.; Cheng, J.; Xu, Y. Highly Uniform In-Situ Cell Electrotransfection of Adherent Cultures Using Grouped Interdigitated Electrodes. Bioelectrochemistry 2020, 132, 107435. [Google Scholar] [CrossRef] [PubMed]
- Chopra, S.; Ruzgys, P.; Maciulevičius, M.; Šatkauskas, S. Effect of Cell Passage Time on the Electrotransfection Efficiency. Biol. Bull. 2020, 47, 441–447. [Google Scholar] [CrossRef]
- Stolwijk, J.A.; Wegener, J. Impedance Analysis of Adherent Cells after in Situ Electroporation-Mediated Delivery of Bioactive Proteins, DNA and Nanoparticles in ΜL-Volumes. Sci. Rep. 2020, 10, 21331. [Google Scholar] [CrossRef]
- Zhu, Q.; Hamilton, M.; Vasquez, B.; He, M. 3D-Printing Enabled Micro-Assembly of a Microfluidic Electroporation System for 3D Tissue Engineering. Lab Chip 2019, 19, 2362–2372. [Google Scholar] [CrossRef]
- Ishino, T.; Kurita, H.; Kirisawa, R.; Shimamoto, Y.; Numano, R.; Kitamura, H. Introduction of a Plasmid and a Protein into Bovine and Swine Cells by Water-in-Oil Droplet Electroporation. J. Vet. Med. Sci. 2020, 82, 14–22. [Google Scholar] [CrossRef]
- Cao, F.; Xie, X.; Gollan, T.; Zhao, L.; Narsinh, K.; Lee, R.J.; Wu, J.C. Comparison of Gene-Transfer Efficiency in Human Embryonic Stem Cells. Mol. Imaging Biol. 2010, 12, 15–24. [Google Scholar] [CrossRef]
- Campillo-Davo, D.; Versteven, M.; Roex, G.; De Reu, H.; van der Heijden, S.; Anguille, S.; Berneman, Z.N.; Van Tendeloo, V.F.I.; Lion, E. Rapid Assessment of Functional Avidity of Tumor-Specific T Cell Receptors Using an Antigen-Presenting Tumor Cell Line Electroporated with Full-Length Tumor Antigen MRNA. Cancers 2020, 12, 256. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Chang, C.C.; Sylvers, J.; Yuan, F. A Statistical Framework for Determination of Minimal Plasmid Copy Number Required for Transgene Expression in Mammalian Cells. Bioelectrochemistry 2021, 138, 107731. [Google Scholar] [CrossRef]
- Bruter, A.V.; Kandarakov, O.F.; Belyavsky, A. V Persistence of Plasmid-Mediated Expression of Transgenes in Human Mesenchymal Stem Cells Depends Primarily on CpG Levels of Both Vector and Transgene. J. Gene Med. 2018, 20, e3009. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Yin, C.; Hu, L.; Chen, Z.; Zhao, F.; Li, D.; Ma, J.; Ma, X.; Su, P.; Qiu, W.; et al. MACF1 Overexpression by Transfecting the 21 Kbp Large Plasmid PEGFP-C1A-ACF7 Promotes Osteoblast Differentiation and Bone Formation. Hum. Gene Ther. 2018, 29, 259–270. [Google Scholar] [CrossRef]
- Aghvami, T.A.; Latifi-Navid, S.; Zahri, S.; Sagha, M. Optimization of PCMV3-Nog-C-GFPSpark Electro-Transfection in MCF-7, a Breast Cancer Cell Line. Res. J. Biotechnol. 2021, 16, 13–18. [Google Scholar] [CrossRef]
- Komel, T.; Bosnjak, M.; Kranjc Brezar, S.; De Robertis, M.; Mastrodonato, M.; Scillitani, G.; Pesole, G.; Signori, E.; Sersa, G.; Cemazar, M. Gene Electrotransfer of IL-2 and IL-12 Plasmids Effectively Eradicated Murine B16.F10 Melanoma. Bioelectrochemistry 2021, 141, 107843. [Google Scholar] [CrossRef]
- Radzevičiūtė, E.; Malyško-Ptašinskė, V.; Novickij, J.; Novickij, V.; Girkontaitė, I. Transfection by Electroporation of Cancer and Primary Cells Using Nanosecond and Microsecond Electric Fields. Pharmaceutics 2022, 14, 1239. [Google Scholar] [CrossRef]
- Eghbalsaied, S.; Kues, W.A. An Electrochemical Protocol for CRISPR-Mediated Gene-Editing of Sheep Embryonic Fibroblast Cells. Cells Tissues Organs 2021. Online ahead of print. [Google Scholar] [CrossRef]
- Bosnjak, M.; Znidar, K.; Sales Conniff, A.; Jesenko, T.; Markelc, B.; Semenova, N.; Tur, J.; Kohena, K.; Kranjc Brezar, S.; Heller, L.; et al. In Vitro and in Vivo Correlation of Skin and Cellular Responses to Nucleic Acid Delivery. Biomed. Pharmacother. 2022, 150, 113088. [Google Scholar] [CrossRef]
- Hun, T.; Zhang, Y.; Xu, Q.; Huang, D.; Wang, Q.; Li, Z.; Wang, W. In Situ Electroporation on PERFECT Filter for High-Efficiency and High-Viability Tumor Cell Labeling. Micromachines 2022, 13, 672. [Google Scholar] [CrossRef]
- Cegovnik, U. Setting Optimal Parameters for in Vitro Electrotransfection of B16F1, SA1, LPB, SCK, L929 and CHO Cells Using Predefined Exponentially Decaying Electric Pulses. Bioelectrochemistry 2004, 62, 73–82. [Google Scholar] [CrossRef] [PubMed]
- Patino, C.A.; Mukherjee, P.; Berns, E.J.; Moully, E.H.; Stan, L.; Mrksich, M.; Espinosa, H.D. High-Throughput Microfluidics Platform for Intracellular Delivery and Sampling of Biomolecules from Live Cells. ACS Nano 2022, 16, 7937–7946. [Google Scholar] [CrossRef] [PubMed]
- Nakami, W.N.; Nguhiu-Mwangi, J.; Kipyegon, A.N.; Ogugo, M.; Muteti, C.; Kemp, S. Comparative Efficiency for in Vitro Transfection of Goat Undifferentiated Spermatogonia Using Lipofectamine Reagents and Electroporation. Stem Cells Cloning Adv. Appl. 2022, 15, 11–20. [Google Scholar] [CrossRef]
- Radzevičiūtė, E.; Murauskas, A.; Ruzgys, P.; Šatkauskas, S.; Girkontaitė, I.; Novickij, J.; Novickij, V. Dielectrophoretic Manipulation of Cell Transfection Efficiency during Electroporation Using a Center Needle Electrode. Appl. Sci. 2021, 11, 7015. [Google Scholar] [CrossRef]
- Kardani, K.; Milani, A.; Bolhassani, A. Gene Delivery in Adherent and Suspension Cells Using the Combined Physical Methods. Cytotechnology 2022, 74, 245–257. [Google Scholar] [CrossRef] [PubMed]
- Tabak, S.; Feinshtein, V.; Schreiber-Avissar, S.; Beit-Yannai, E. Non-Pigmented Ciliary Epithelium-Derived Extracellular Vesicles Loaded with Smad7 Sirna Attenuate Wnt Signaling in Trabecular Meshwork Cells in Vitro. Pharmaceuticals 2021, 14, 858. [Google Scholar] [CrossRef] [PubMed]
- Bulygin, A.S.; Tereshchenko, V.P.; Zavodskii, R.Y.; Obleukhova, I.A.; Sennikov, S.V.; Silkov, A.N. Effect of DNA Constructions Electroporation on Dendritic Cells. Med. Immunol. 2021, 23, 653–658. [Google Scholar] [CrossRef]
- Kanazawa, T.; Hoashi, Y.; Ibaraki, H.; Takashima, Y.; Okada, H. Electroporation-Based Ex Vivo Gene Delivery into Dendritic Cells by Anionic Polymer-Coated Versatile Nuclear Localization Signal/PDNA Complex. Biol. Pharm. Bull. 2021, 44, 1866–1871. [Google Scholar] [CrossRef]
- Pelofy, S.; Bousquet, H.; Gibot, L.; Rols, M.P.; Golzio, M. Transfer of Small Interfering RNA by Electropermeabilization in Tumor Spheroids. Bioelectrochemistry 2021, 141, 107848. [Google Scholar] [CrossRef]
- Cemazar, M.; Sersa, G.; Wilson, J.; Tozer, G.M.G.M.; Hart, S.L.S.L.; Grosel, A.; Dachs, G.U. Effective Gene Transfer to Solid Tumors Using Different Nonviral Gene Delivery Techniques: Electroporation, Liposomes, and Integrin-Targeted Vector. Cancer Gene Ther. 2002, 9, 399–406. [Google Scholar] [CrossRef][Green Version]
- Cepurniene, K.; Ruzgys, P.; Treinys, R.; Satkauskiene, I.; Satkauskas, S.; Čepurnienė, K.; Ruzgys, P.; Treinys, R.; Šatkauskienė, I.; Šatkauskas, S.; et al. Influence of Plasmid Concentration on DNA Electrotransfer in Vitro Using High-Voltage and Low-Voltage Pulses. J. Membr. Biol. 2010, 236, 81–85. [Google Scholar] [CrossRef] [PubMed]
- Cervia, L.D.; Chang, C.-C.; Wang, L.; Mao, M.; Yuan, F. Enhancing Electrotransfection Efficiency through Improvement in Nuclear Entry of Plasmid DNA. Mol. Ther.—Nucleic Acids 2018, 11, 263–271. [Google Scholar] [CrossRef] [PubMed]
- Cesnulevicius, K.; Timmer, M.; Wesemann, M.; Thomas, T.; Barkhausen, T.; Grothe, C. Nucleofection Is the Most Efficient Nonviral Transfection Method for Neuronal Stem Cells Derived from Ventral Mesencephali with No Changes in Cell Composition or Dopaminergic Fate. Stem Cells 2006, 24, 2776–2791. [Google Scholar] [CrossRef] [PubMed]
- Chabot, S.; Orio, J.; Castanier, R.; Bellard, E.; Nielsen, S.J.; Golzio, M.; Teissié, J. LNA-Based Oligonucleotide Electrotransfer for MiRNA Inhibition. Mol. Ther. 2012, 20, 1590–1598. [Google Scholar] [CrossRef]
- Chabot, S.; Orio, J.; Schmeer, M.; Schleef, M.; Golzio, M.; Teissié, J. Minicircle DNA Electrotransfer for Efficient Tissue-Targeted Gene Delivery. Gene Ther. 2012, 20, 62–68. [Google Scholar] [CrossRef]
- Chopinet, L.; Wasungu, L.; Rols, M.P. First Explanations for Differences in Electrotransfection Efficiency in Vitro and in Vivo Using Spheroid Model. Int. J. Pharm. 2012, 423, 7–15. [Google Scholar] [CrossRef]
- Coulberson, A.L.; Hud, N.V.; LeDoux, J.M.; Vilfan, I.D.; Prausnitz, M.R. Gene Packaging with Lipids, Peptides and Viruses Inhibits Transfection by Electroporation in Vitro. J. Control. Release 2003, 86, 361–370. [Google Scholar] [CrossRef]
- Delgado-Cañedo, A.; dos Santos, D.G.; Chies, J.A.B.; Kvitko, K.; Nardi, N.B. Optimization of an Electroporation Protocol Using the K562 Cell Line as a Model: Role of Cell Cycle Phase and Cytoplasmic DNAses. Cytotechnology 2006, 51, 141–148. [Google Scholar] [CrossRef]
- Delteil, C.; Teissié, J.; Rols, M.-P. Effect of Serum on in Vitro Electrically Mediated Gene Delivery and Expression in Mammalian Cells. Biochim. Biophys. Acta-Biomembr. 2000, 1467, 362–368. [Google Scholar] [CrossRef]
- Donate, A.; Heller, R. Assessment of Delivery Parameters with the Multi-Electrode Array for Development of a DNA Vaccine against Bacillus Anthracis. Bioelectrochemistry 2013, 94, 1–6. [Google Scholar] [CrossRef]
- Dullaers, M.; Breckpot, K.; Van Meirvenne, S.; Bonehill, A.; Tuyaerts, S.; Michiels, A.; Straetman, L.; Heirman, C.; De Greef, C.; Van Der Bruggen, P.; et al. Side-by-Side Comparison of Lentivirally Transduced and MRNA-Electroporated Dendritic Cells: Implications for Cancer Immunotherapy Protocols. Mol. Ther. 2004, 10, 768–779. [Google Scholar] [CrossRef] [PubMed]
- Ear, T.; Giguère, P.; Fleury, A.; Stankova, J.; Payet, M.D.; Dupuis, G. High Efficiency Transient Transfection of Genes in Human Umbilical Vein Endothelial Cells by Electroporation. J. Immunol. Methods 2001, 257, 41–49. [Google Scholar] [CrossRef] [PubMed]
- Escoffre, J.-M.; Nikolova, B.; Mallet, L.; Henri, J.; Favard, C.; Golzio, M.; Teissié, J.; Tsoneva, I.; Rols, M.-P. New Insights in the Gene Electrotransfer Process: Evidence for the Involvement of the Plasmid DNA Topology. Curr. Gene Ther. 2012, 12, 417–422. [Google Scholar] [CrossRef] [PubMed]
- Escoffre, J.M.M.; Kaddur, K.; Rols, M.P.P.; Bouakaz, A. In Vitro Gene Transfer by Electrosonoporation. Ultrasound Med. Biol. 2010, 36, 1746–1755. [Google Scholar] [CrossRef]
- Eslaminejad, T.; Nematollahi-Mahani, S.N.; Ansari, M. Cationic β-Cyclodextrin-Chitosan Conjugates as Potential Carrier for PmCherry-C1 Gene Delivery. Mol. Biotechnol. 2016, 58, 287–298. [Google Scholar] [CrossRef] [PubMed]
- Espinos, E.; Liu, J.H.; Bader, C.R.; Bernheim, L. Efficient Non-Viral DNA-Mediated Gene Transfer to Human Primary Myoblasts Using Electroporation. Neuromuscul. Disord. 2001, 11, 341–349. [Google Scholar] [CrossRef]
- Faurie, C.; Rebersek, M.; Golzio, M.; Kanduser, M.; Escoffre, J.-M.; Pavlin, M.; Teissie, J.; Miklavcic, D.; Rols, M.-P. Electro-Mediated Gene Transfer and Expression Are Controlled by the Life-Time of DNA/Membrane Complex Formation. J. Gene Med. 2010, 12, 117–125. [Google Scholar] [CrossRef]
- Faurie, C.; Phez, E.; Golzio, M.; Vossen, C.; Lesbordes, J.-C.J.-C.; Delteil, C.; Teissié, J.; Rols, M.-P.M.-P. Effect of Electric Field Vectoriality on Electrically Mediated Gene Delivery in Mammalian Cells. Biochim. Biophys. Acta (BBA)-Biomembr. 2004, 1665, 92–100. [Google Scholar] [CrossRef]
- Feril, L.B.; Ogawa, R.; Tachibana, K.; Kondo, T. Optimized Ultrasound-Mediated Gene Transfection in Cancer Cells. EMBO J. 1982, 1, 841–845. [Google Scholar] [CrossRef]
- Ferreira, E.; Potier, E.; Logeart-Avramoglou, D.; Salomskaite-Davalgiene, S.; Mir, L.M.M.; Petite, H. Optimization of a Gene Electrotransfer Method for Mesenchymal Stem Cell Transfection. Gene Ther. 2008, 15, 537–544. [Google Scholar] [CrossRef]
- Fountain, J.W.; Lockwood, W.K.; Collins, F.S. Transfection of Primary Human Skin Fibroblasts by Electroporation. Gene 1988, 68, 167–172. [Google Scholar] [CrossRef] [PubMed]
- García-Sánchez, T.; Guitart, M.; Rosell-Ferrer, J.; Gómez-Foix, A.M.; Bragós, R. A New Spiral Microelectrode Assembly for Electroporation and Impedance Measurements of Adherent Cell Monolayers. Biomed. Microdevices 2014, 16, 575–590. [Google Scholar] [CrossRef]
- Ghartey-Tagoe, E.B.; Babbin, B.A.; Nusrat, A.; Neish, A.S.; Prausnitz, M.R. Plasmid DNA and SiRNA Transfection of Intestinal Epithelial Monolayers by Electroporation. Int. J. Pharm. 2006, 315, 122–133. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, C.; Song, W.; Lahiri, D.K. Efficient DNA Transfection in Neuronal and Astrocytic Cell Lines. Mol. Biol. Rep. 2000, 27, 113–121. [Google Scholar] [CrossRef] [PubMed]
- Giroux, S.J.D.D.; Alves-Leiva, C.; Lécluse, Y.; Martin, P.; Albagli, O.; Godin, I. Gene Transfer to Pre-Hematopoietic and Committed Hematopoietic Precursors in the Early Mouse Yolk Sac: A Comparative Study between in Situ Electroporation and Retroviral Transduction. BMC Dev. Biol. 2007, 7, 79. [Google Scholar] [CrossRef]
- Goldstein, S.; Fordis, C.M.; Howard, B.H. Enhanced Transfection Efficiency and Improved Cell Survival after Electroporation of G2/M-Synchronized Cells and Treatment with Sodium Butyrate. Nucleic Acids Res. 1989, 17, 3959–3971. [Google Scholar] [CrossRef]
- Grabarek, J.B.; Plusa, B.; Glover, D.M.; Zernicka-Goetz, M. Efficient Delivery of DsRNA into Zona-Enclosed Mouse Oocytes and Preimplantation Embryos by Electroporation. Genesis 2002, 32, 269–276. [Google Scholar] [CrossRef]
- Grund, E.M.; Muise-Helmericks, R.C. Cost Efficient and Effective Gene Transfer into the Human Natural Killer Cell Line, NK92. J. Immunol. Methods 2005, 296, 31–36. [Google Scholar] [CrossRef]
- Guo, H.; Hao, R.; Wei, Y.; Sun, D.; Sun, S.; Zhang, Z. Optimization of Electrotransfection Conditions of Mammalian Cells with Different Biological Features. J. Membr. Biol. 2012, 245, 789–795. [Google Scholar] [CrossRef]
- Haberl, S.S.; Pavlin, M. Use of Collagen Gel as a Three-Dimensional In Vitro Model to Study Electropermeabilization and Gene Electrotransfer. J. Membr. Biol. 2010, 236, 87–95. [Google Scholar] [CrossRef]
- Harrison, R.L.; Byrne, B.J.; Tung, L. Electroporation-Mediated Gene Transfer in Cardiac Tissue. FEBS Lett. 1998, 435, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Hashemi, A.; Roohvand, F.; Ghahremani, M.H.; Aghasadeghi, M.R.; Vahabpour, R.; Motevali, F.; Memarnejadian, A. Optimization of Transfection Methods for Huh-7 and Vero Cells: A Comparative Study. Cytol. Genet. 2012, 46, 347–353. [Google Scholar] [CrossRef] [PubMed]
- Helledie, T.; Nurcombe, V.; Cool, S.M. A Simple and Reliable Electroporation Method for Human Bone Marrow Mesenchymal Stem Cells. Stem. Cells Dev. 2008, 17, 837–848. [Google Scholar] [CrossRef] [PubMed]
- Hernández, J.L.; Coll, T.; Ciudad, C.J. A Highly Efficient Electroporation Method for the Transfection of Endothelial Cells. Angiogenesis 2004, 7, 235–241. [Google Scholar] [CrossRef] [PubMed]
- Hilliard, C.M.; Fletcher, S.; Yeoh, G.C.T. Calcium Phosphate Transfection and Cell-Specific Expression of Heterologous Genes in Primary Fetal Rat Hepatocytes. Int. J. Biochem. Cell. Biol. 1996, 28, 639–650. [Google Scholar] [CrossRef]
- Hoerauf, W.W.W.; Cazares, V.A.V.A.V.A.; Subramani, A.; Stuenkel, E.L.E.L. Efficient Transfection of Dissociated Mouse Chromaffin Cells Using Small-Volume Electroporation. Cytotechnology 2015, 67, 573–583. [Google Scholar] [CrossRef]
- Hornstein, B.D.B.D.B.D.; Roman, D.; Arévalo-Soliz, L.M.L.M.; Engevik, M.A.M.A.; Zechiedrich, L. Effects of Circular DNA Length on Transfection Efficiency by Electroporation into HeLa Cells. PLoS ONE 2016, 11, e0167537. [Google Scholar] [CrossRef]
- Huang, H.; Wei, Z.; Huang, Y.; Zhao, D.; Zheng, L.; Cai, T.; Wu, M.; Wang, W.; Ding, X.; Zhou, Z.; et al. An Efficient and High-Throughput Electroporation Microchip Applicable for SiRNA Delivery. Lab Chip 2011, 11, 163–172. [Google Scholar] [CrossRef]
- Huang, K.-S.; Lin, Y.-C.; Su, C.-C.; Fang, C.-S. Enhancement of an Electroporation System for Gene Delivery Using Electrophoresis with a Planar Electrode. Lab Chip 2007, 7, 86–92. [Google Scholar] [CrossRef]
- Huang, S.; Deshmukh, H.; Rajagopalan, K.K.; Wang, S. Gold Nanoparticles Electroporation Enhanced Polyplex Delivery to Mammalian Cells. Electrophoresis 2014, 35, 1837–1845. [Google Scholar] [CrossRef]
- Hui, S.W.; Stoicheva, N.; Zhao, Y.L. High-Efficiency Loading, Transfection, and Fusion of Cells by Electroporation in Two-Phase Polymer Systems. Biophys. J. 1996, 71, 1123–1130. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ishibashi, T.; Takoh, K.; Kaji, H.; Abe, T.; Nishizawa, M. A Porous Membrane-Based Culture Substrate for Localized in Situ Electroporation of Adherent Mammalian Cells. Sens. Actuators B Chem. 2007, 128, 5–11. [Google Scholar] [CrossRef]
- Iversen, N.; Birkenes, B.; Torsdalen, K.; Djurovic, S. Electroporation by Nucleofector Is the Best Nonviral Transfection Technique in Human Endothelial and Smooth Muscle Cells. Genet. Vaccines Ther. 2005, 3, 2. [Google Scholar] [CrossRef] [PubMed]
- Jain, T.; Muthuswamy, J. Bio-Chip for Spatially Controlled Transfection of Nucleic Acid Payloads into Cells in a Culture. Lab Chip 2007, 7, 8. [Google Scholar] [CrossRef]
- Jiang, C.-K.; Connolly, D.; Blumenberg, M. Comparison of Methods for Transfection of Human Epidermal Keratinocytes. J. Investig. Dermatol. 1991, 97, 969–973. [Google Scholar] [CrossRef][Green Version]
- Joergensen, M.; Agerholm-Larsen, B.; Nielsen, P.E.; Gehl, J. Efficiency of Cellular Delivery of Antisense Peptide Nucleic Acid by Electroporation Depends on Charge and Electroporation Geometry. Oligonucleotides 2011, 21, 29–37. [Google Scholar] [CrossRef]
- Abdul Halim, N.S.S.; Fakiruddin, K.S.; Ali, S.A.; Yahaya, B.H.; Halim, N.; Fakiruddin, K.S.; Ali, S.A.; Yahaya, B.H. A Comparative Study of Non-Viral Gene Delivery Techniques to Human Adipose-Derived Mesenchymal Stem Cell. Int. J. Mol. Sci. 2014, 15, 15044–15060. [Google Scholar] [CrossRef]
- Jordan, E.T.E.T.; Collins, M.; Terefe, J.; Ugozzoli, L.; Rubio, T. Optimizing Electroporation Conditions in Primary and Other Difficult-to-Transfect Cells. J. Biomol. Tech. 2008, 19, 328–334. [Google Scholar]
- Kalina, J.; Kolmanová, A.; Mikuš, T.; Mičáková, A.; Trefil, P. Transfection of Cock Spermatogonial Cells via Electroporation and Lipofection. Czech J. Anim. Sci. 2003, 48, 279–284. [Google Scholar]
- Kang, J.-H.J.-H.; Toita, R.; Niidome, T.; Katayama, Y. Effective Delivery of DNA into Tumor Cells and Tissues by Electroporation of Polymer–DNA Complex. Cancer Lett. 2008, 265, 281–288. [Google Scholar] [CrossRef]
- Kawabata, I.; Umeda, T.; Yamamoto, K.; Okabe, S. Electroporation-Mediated Gene Transfer System Applied to Cultured CNS Neurons. Neuroreport 2004, 15, 971–975. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.A.; Lee, W.G.; Jung, N.C. Enhanced Electro-Mediated Gene Delivery Using Carrier Genes. Bioelectrochemistry 2010, 78, 186–190. [Google Scholar] [CrossRef]
- Kim, J.A.J.A.; Cho, K.; Shin, Y.S.Y.S.; Jung, N.; Chung, C.; Chang, J.K.J.K. A Multi-Channel Electroporation Microchip for Gene Transfection in Mammalian Cells. Biosens Bioelectron. 2007, 22, 3273–3277. [Google Scholar] [CrossRef]
- Kim, J.A.J.A.; Lee, W.G.W.G. Role of Weakly Polarized Nanoparticles in Electroporation. Nanoscale 2011, 3, 186–190. [Google Scholar] [CrossRef] [PubMed]
- Klenchin, V.A.; Sukharev, S.I.; Serov, S.M.; Chernomordik, L.V.; Chizmadzhev, Y.A. Electrically Induced DNA Uptake by Cells Is a Fast Process Involving DNA Electrophoresis. Biophys. J. 1991, 60, 804–811. [Google Scholar] [CrossRef] [PubMed]
- Knutson, J.C.; Yee, D. Electroporation: Parameters Affecting Transfer of DNA into Mammalian Cells. Anal. Biochem. 1987, 164, 44–52. [Google Scholar] [CrossRef] [PubMed]
- Kobelt, D.; Schleef, M.; Schmeer, M.; Aumann, J.; Schlag, P.M.; Walther, W. Performance of High Quality Minicircle DNA for In Vitro and In Vivo Gene Transfer. Mol. Biotechnol. 2013, 53, 80–89. [Google Scholar] [CrossRef]
- Amiri Yekta, A.; Dalman, A.; Sanati, M.H.; Fatemi, N.; Vazirinasab, H.; Zomorodipour, A.; Chehrazi, M.; Gourabi, H. Optimization of The Electroporation Conditions for Transfection of Human Factor IX into The Goat Fetal Fibroblasts. Cell J. 2013, 14, 270–275. [Google Scholar]
- Kotnis, R.A.A.; Thompson, M.M.M.; Eady, S.L.L.; Budd, J.S.S.; Bell, P.R.F.R.; James, R.F.L.F. Optimisation of Gene Transfer into Vascular Endothelial Cells Using Electroporation. Eur. J. Vasc. Endovasc. Surg. 1995, 9, 71–79. [Google Scholar] [CrossRef][Green Version]
- Kumar Pramod, R.; Kumar, R.; Mitra, A. Transgenic Expression of Green Fluorescent Protein in Caprine Embryos Produced through Electroporation-Aided Sperm-Mediated Gene Transfer. Gene 2016, 576, 505–511. [Google Scholar] [CrossRef]
- Kwee, S.; Nielsen, H.V.; Celis, J.E. Electropermeabilization of Human Cultured Cells Grown in Monolayers. Incorporation of Monoclonal Antibodies. Bioelectrochemistry Bioenerg. 1990, 23, 65–80. [Google Scholar] [CrossRef]
- Lakshmipathy, U.; Pelacho, B.; Sudo, K.; Linehan, J.L.; Coucouvanis, E.; Kaufman, D.S.; Verfaillie, C.M. Efficient Transfection of Embryonic and Adult Stem Cells. Stem Cells 2004, 22, 531–543. [Google Scholar] [CrossRef] [PubMed]
- Lam, A.P.; Dean, D.A. Cyclic Stretch-Induced Nuclear Localization of Transcription Factors Results in Increased Nuclear Targeting of Plasmids in Alveolar Epithelial Cells. J. Gene Med. 2008, 10, 668–678. [Google Scholar] [CrossRef] [PubMed]
- Lampreht Tratar, U.; Kos, S.; Kamensek, U.; Ota, M.; Tozon, N.; Sersa, G.; Cemazar, M. Antitumor Effect of Antibiotic Resistance Gene-Free Plasmids Encoding Interleukin-12 in Canine Melanoma Model. Cancer Gene Ther. 2018, 25, 260–273. [Google Scholar] [CrossRef] [PubMed]
- Landi, A.; Babiuk, L.A.; van Drunen Littel-van den Hurk, S. High Transfection Efficiency, Gene Expression, and Viability of Monocyte-Derived Human Dendritic Cells after Nonviral Gene Transfer. J. Leukoc. Biol. 2007, 82, 849–860. [Google Scholar] [CrossRef]
- Laurent, V.; Fraix, A.; Montier, T.; Cammas-Marion, S.; Ribault, C.; Benvegnu, T.; Jaffres, P.-A.; Loyer, P. Highly Efficient Gene Transfer into Hepatocyte-like HepaRG Cells: New Means for Drug Metabolism and Toxicity Studies. Biotechnol. J. 2010, 5, 314–320. [Google Scholar] [CrossRef]
- Lee, S.-L.S.-L.; Ock, S.-A.S.-A.; Yoo, J.-G.J.-G.; Kumar, B.M.M.; Choe, S.-Y.S.-Y.; Rho, G.-J.G.-J. Efficiency of Gene Transfection into Donor Cells for Nuclear Transfer of Bovine Embryos. Mol. Reprod. Dev. 2005, 72, 191–200. [Google Scholar] [CrossRef]
- Lehmann, M.H.; Berg, H. Electroporation Induced Gene Expression—A Case Study on Interleukin-10. Bioelectrochemistry Bioenerg. 1998, 47, 3–10. [Google Scholar] [CrossRef]
- Anderson, M.L.M.M.L.M.; Spandidos, D.A.D.A.; Coggins, J.R.J.R. Electroporation of Lymphoid Cells: Factors Affecting the Efficiency of Transfection. J. Biochem. Biophys. Methods 1991, 22, 207–222. [Google Scholar] [CrossRef]
- Lewis, E.W.; Rudo, T.J.; St John, M.A.R.; Chu, J.L.; Heinze, A.W.; Howard, B.H.; Engleka, K.A. Endothelial Cell DNA Transfer and Expression Using Petri Dish Electroporation and the Nonreplicating Vaccinia Virus/T7 RNA Polymerase Hybrid System. Gene Ther. 1999, 6, 1617–1625. [Google Scholar] [CrossRef][Green Version]
- Li, F.; Yamaguchi, K.; Okada, K.; Matsushita, K.; Enatsu, N.; Chiba, K.; Yue, H.; Fujisawa, M. Efficient Transfection of DNA into Primarily Cultured Rat Sertoli Cells by Electroporation1. Biol. Rep. 2013, 88, 61. [Google Scholar] [CrossRef]
- Li, G.-B.; Lu, G.-X. Gene Delivery Efficiency in Bone Marrow-Derived Dendritic Cells: Comparison of Four Methods and Optimization for Lentivirus Transduction. Mol. Biotechnol. 2009, 43, 250–256. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Yu, T.; Zhou, J.; Tu, W.; Gao, S.; Liu, X. Optimal Transfection Methods and Comparison of PK-15 and Dulac Cells for Rescue of Chimeric Porcine Circovirus Type 1-2. Virol. Methods. 2014, 208, 90–95. [Google Scholar] [CrossRef] [PubMed]
- Liang, X.; Potter, J.; Kumar, S.; Zou, Y.; Quintanilla, R.; Sridharan, M.; Carte, J.; Chen, W.; Roark, N.; Ranganathan, S.; et al. Rapid and Highly Efficient Mammalian Cell Engineering via Cas9 Protein Transfection. J. Biotechnol. 2015, 208, 44–53. [Google Scholar] [CrossRef]
- Liew, A.; André, F.M.; Lesueur, L.L.; De Ménorval, M.-A.; O’Brien, T.; Mir, L.M. Robust, Efficient, and Practical Electrogene Transfer Method for Human Mesenchymal Stem Cells Using Square Electric Pulses. Hum. Gene Ther. Methods 2013, 24, 289–297. [Google Scholar] [CrossRef]
- Lin, W.-Z.; Lee, S.S.T.; Cheung, W.-T. Efficient Expression of Foreign Genes in CHO DHFR(-) Cells by Electroporation. Biologicals 2009, 37, 277–281. [Google Scholar] [CrossRef]
- Lin, Y.-C.; Jen, C.-M.; Huang, M.-Y.; Wu, C.-Y.; Lin, X.-Z. Electroporation Microchips for Continuous Gene Transfection. Sens. Actuators B Chem. 2001, 79, 137–143. [Google Scholar] [CrossRef]
- Lin, Y.-C.; Li, M.; Fan, C.-S.; Wu, L.-W. A Microchip for Electroporation of Primary Endothelial Cells. Sens. Actuators A Phys. 2003, 108, 12–19. [Google Scholar] [CrossRef]
- Lin, Y.-C.; Li, M.; Wu, C.-C. Simulation and Experimental Demonstration of the Electric Field Assisted Electroporation Microchip for in Vitro Gene Delivery Enhancement. Lab Chip 2004, 4, 104–108. [Google Scholar] [CrossRef] [PubMed]
- Lara, A.R.; Ramírez, O.T. Plasmid DNA Production for Therapeutic Applications. Methods Mol. Biol. 2012, 824, 271–303. [Google Scholar] [CrossRef]
- Pucihar, G.; Tadej, A.E.; Ae, K.; Ae, J.T.; Miklavčič, D.; Pucihar, G.; Kotnik, Á.T.; Miklavčič, Á.D.; Teissié, J. Electropermeabilization of Dense Cell Suspensions. Eur. Biophys. J. 2007, 36, 173–185. [Google Scholar] [CrossRef] [PubMed]
- Molnar, M.J.M.J.M.J.; Gilbert, R.; Lu, Y.; Liu, A.-B.; Guo, A.; Larochelle, N.; Lochmuller, H.; Petrof, B.J.B.J.; Nalbantoglu, J.; Karpati, G.; et al. Factors Influencing the Efficacy, Longevity, and Safety of Electroporation-Assisted Plasmid-Based Gene Transfer into Mouse Muscles. Mol. Ther. 2004, 10, 447–455. [Google Scholar] [CrossRef]
- Ribeiro, S.; Mairhofer, J.; Madeira, C.; Diogo, M.M.; Lobato Da Silva, C.; Monteiro, G.; Grabherr, R.; Cabral, J.M. Plasmid DNA Size Does Affect Nonviral Gene Delivery Efficiency in Stem Cells. Cell. Reprogram. 2012, 14, 130–137. [Google Scholar] [CrossRef] [PubMed]
- Djuzenova, C.S.; Zimmermann, U.; Frank, H.; Sukhorukov, V.L.; Richter, E.; Fuhr, G. Effect of Medium Conductivity and Composition on the Uptake of Propidium Iodide into Electropermeabilized Myeloma Cells. Biochim. Biophys. Acta-Biomembr. 1996, 1284, 143–152. [Google Scholar] [CrossRef] [PubMed]
- Vižintin, A.; Vidmar, J.; Ščančar, J.; Miklavčič, D. Effect of Interphase and Interpulse Delay in High-Frequency Irreversible Electroporation Pulses on Cell Survival, Membrane Permeabilization and Electrode Material Release. Bioelectrochemistry 2020, 134, 107523. [Google Scholar] [CrossRef]
- Raso, J.; Frey, W.; Ferrari, G.; Pataro, G.; Knorr, D.; Teissie, J.; Miklavčič, D. Recommendations Guidelines on the Key Information to Be Reported in Studies of Application of PEF Technology in Food and Biotechnological Processes. Innov. Food Sci. Emerg. Technol. 2016, 37, 312–321. [Google Scholar] [CrossRef]
- Cemazar, M.; Sersa, G.; Frey, W.; Miklavcic, D.; Teissié, J. Recommendations and Requirements for Reporting on Applications of Electric Pulse Delivery for Electroporation of Biological Samples. Bioelectrochemistry 2018, 122, 69–76. [Google Scholar] [CrossRef] [PubMed]





| cDNA | Complementary DNA |
| CI | Confidence intervals |
| CMV | Cytomegalovirus |
| COM | Combination of pulses |
| DNA | Deoxyribonucleic acid |
| EXP-COM | Combination of exponential pulses |
| EXP-long | Long exponential pulses |
| EXP-medium | Medium exponential pulses |
| EXP-short | Short exponential pulses |
| HF-BP | High-frequency bipolar pulses |
| GET | Gene electrotransfer |
| gRNA | Guide RNA |
| medium-BP | Medium bipolar square wave pulse |
| MIDGE | Minimalistic, immunologically defined gene expression |
| mod | RF modulated square wave pulse |
| mRNA | Messenger RNA |
| NA | Nucleic acid |
| ns | Nanosecond pulses |
| pDNA | Plasmid DNA |
| RF | Radio frequency |
| RNA | Ribonucleic acid |
| short-BP | Short bipolar square wave pulse |
| shRNA | Short hairpin RNA |
| SIN | Sinusoidal pulses |
| siRNA | Silencing RNA |
| SV40 | Simian virus 40 |
| Appropriate | Poor | Inappropriate | |
|---|---|---|---|
| Controls | |||
| Were adequate control groups used? | At least control group without electroporation | Just control group without pDNA; not clear if it is control group without electroporation or without pDNA; other control groups | Not reported |
| Electrical parameters | |||
| Is the pulse generator adequately reported? | Output pulse parameters or design of the electroporator is known | The type of the electroporator is reported but the specifications of the output pulses or the design is not reported | Not reported |
| Are amplitude and duration of pulses adequately reported? | Amplitude and duration of pulses is reported | Amplitude and pulse waveform is known, but not the time constant | Not reported |
| Are the electrodes adequately reported? | Distance between the electrodes and the design (geometry to determine electric field distribution) is reported | Distance between the electrodes is reported but not the design (geometry to determine electric field distribution) | Not reported |
| Was electric pulses delivery controlled adequately? | The user measures output pulses with an oscilloscope | The user measures or in any other way assesses output pulses; pulse generators that measure output pulses | Not reported |
| Is the material of electrodes described? | Material of solid electrodes is reported or thickness and material of basal and plated material of the electrodes is reported | Basal and plated materials are reported but not the thickness | Not reported |
| Biological parameters | |||
| Is the amount of added NA reported? | Amount or concentration and volume | Just concentration or range reported | Not reported |
| Is the size of NA reported? | Size is described or manufacturer is reported | Not reported for all used pDNA-s | Not reported |
| Is the promoter used reported? | Promoter is reported or manufacturer is reported | Not reported for all used pDNA-s | Not reported |
| Is the number of cells used for GET reported? | Number is reported | Just concentration or range reported | Not reported |
| Are growth, electroporation medium, and other chemicals used reported? | Growth medium, electroporation medium and other chemicals used are reported | Electroporation medium is not reported | Not reported |
| Is it described if cells are in suspension or attached? | Described if cells are in suspension or attached | / | Not reported |
| Is the origin of cell line reported? | Origin of cell line is reported | Origin is not reported for all cell lines | Not reported |
| Is it described if it is normal, tumor cell line or primary cells? | Described if it is normal, tumor cell line or primary cells | / | Not reported |
| Is it reported when results were measured? | Reported when GET was measured | Just time frame reported | Not reported |
| Is the method used for evaluation of results reported? | Method used for evaluation of GET efficiency is reported | / | Not reported |
| Is reporting of results clear and adequate? (Defined if it is percentage of GET, overall GET) | Percentage of transfection and survival are reported | Only percentage of transfection is reported | Not reported |
| Parameter | No. of Experiments (No. of Papers) | Overall GET Efficiency (%) (95% CI) |
|---|---|---|
| Normal | 53 (15) | 23.6 (±1.8) |
| Tumor | 16 (7) | 39.9 (±17.3) |
| Primary | 19 (7) | 15.2 (±2.18) |
| Electroporation medium | ||
| Sugar and salt-based media | 43 (16) | 22.8 (±2.0) |
| Cell culture media | 32 (6) | 30.1 (±9.0) |
| Commercial electroporation media | 10 (4) | 20.1 (±6.9) |
| Balanced salt solutions | 3 (2) | 33.4 (±20.9) |
| Pulses | ||
| Short | 25 (9) | 17.6 (± 1.9) |
| Medium | 19 (10) | 28.2 (± 12.4) |
| Long | 20 (7) | 28.2 (±4.4) |
| Short-long and exp | 9 (3) | 50.6 (±12.9) |
| BP | 8 (2) | 16.2 (±9.9) |
| ns | 7 (2) | 15.7 (±11.6) |
| Promoter | ||
| CMV | 76 (20) | 23.8 (±1.6) |
| SV40 | 8 (2) | 51.2 (±29.3) |
| others | 4 (2) | 7.6 (±4.1) |
| Size of NA | ||
| <4 kb | 19 (9) | 26.2 (±2.6) |
| between 4 kb and 5 kb | 51 (11) | 25.0 (±4.2) |
| >5 kb | 18 (5) | 24.1 (±2.7) |
| Parameter | What Should Be Included/Reported |
|---|---|
| Controls | Cells which were not exposed to EP and NA Cells with NA but not exposed to EP Cells without NA but exposed to EP |
| Pulse Generator | Manufacturer and type In the case of prototype devices, the construction design should be reported. * |
| Electric pulses | Shape, duration, number, pulse repetition frequency and amplitude. Pulse delivery should always be monitored by an appropriate device and at least two figures of delivered pulses should be provided, one of a single pulse zoomed and another with reduced time scale, where all delivered pulses are displayed. * |
| Electrodes | Manufacturer and type of electrodes In the case of custom-made electrodes geometry, material should be reported, and in case of microelectrodes, the construction procedure should also be reported. * |
| NA concentration | Final NA concentration or amount of added NA and volume of suspension |
| NA size | Manufacturer and catalog number or specified NA size |
| Promoter | Manufacturer and catalog number or specified promoter |
| Number of cells | Number of cells exposed to GET or cell concentration and volume of suspension |
| Electroporation medium | Manufacturer and catalog number or specified composition of electroporation medium |
| Cells | Manufacturer and catalog number or specified type and origin of cell line. It should also be reported if cells were treated in suspension or attached. |
| Results | Transfection efficiency and cell survival should be reported. In addition, the method and time of transfection efficiency and cell survival measurement should be described. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Potočnik, T.; Maček Lebar, A.; Kos, Š.; Reberšek, M.; Pirc, E.; Serša, G.; Miklavčič, D. Effect of Experimental Electrical and Biological Parameters on Gene Transfer by Electroporation: A Systematic Review and Meta-Analysis. Pharmaceutics 2022, 14, 2700. https://doi.org/10.3390/pharmaceutics14122700
Potočnik T, Maček Lebar A, Kos Š, Reberšek M, Pirc E, Serša G, Miklavčič D. Effect of Experimental Electrical and Biological Parameters on Gene Transfer by Electroporation: A Systematic Review and Meta-Analysis. Pharmaceutics. 2022; 14(12):2700. https://doi.org/10.3390/pharmaceutics14122700
Chicago/Turabian StylePotočnik, Tjaša, Alenka Maček Lebar, Špela Kos, Matej Reberšek, Eva Pirc, Gregor Serša, and Damijan Miklavčič. 2022. "Effect of Experimental Electrical and Biological Parameters on Gene Transfer by Electroporation: A Systematic Review and Meta-Analysis" Pharmaceutics 14, no. 12: 2700. https://doi.org/10.3390/pharmaceutics14122700
APA StylePotočnik, T., Maček Lebar, A., Kos, Š., Reberšek, M., Pirc, E., Serša, G., & Miklavčič, D. (2022). Effect of Experimental Electrical and Biological Parameters on Gene Transfer by Electroporation: A Systematic Review and Meta-Analysis. Pharmaceutics, 14(12), 2700. https://doi.org/10.3390/pharmaceutics14122700








