Deep Eutectic Systems as Novel Vehicles for Assisting Drug Transdermal Delivery
Abstract
1. Introduction
2. Deep Eutectic Solvent (DES)
2.1. The General Rule of DES
2.2. Natural Deep Eutectic Solvent (NADES)
2.3. Therapeutic Deep Eutectic Solvent (THEDES)
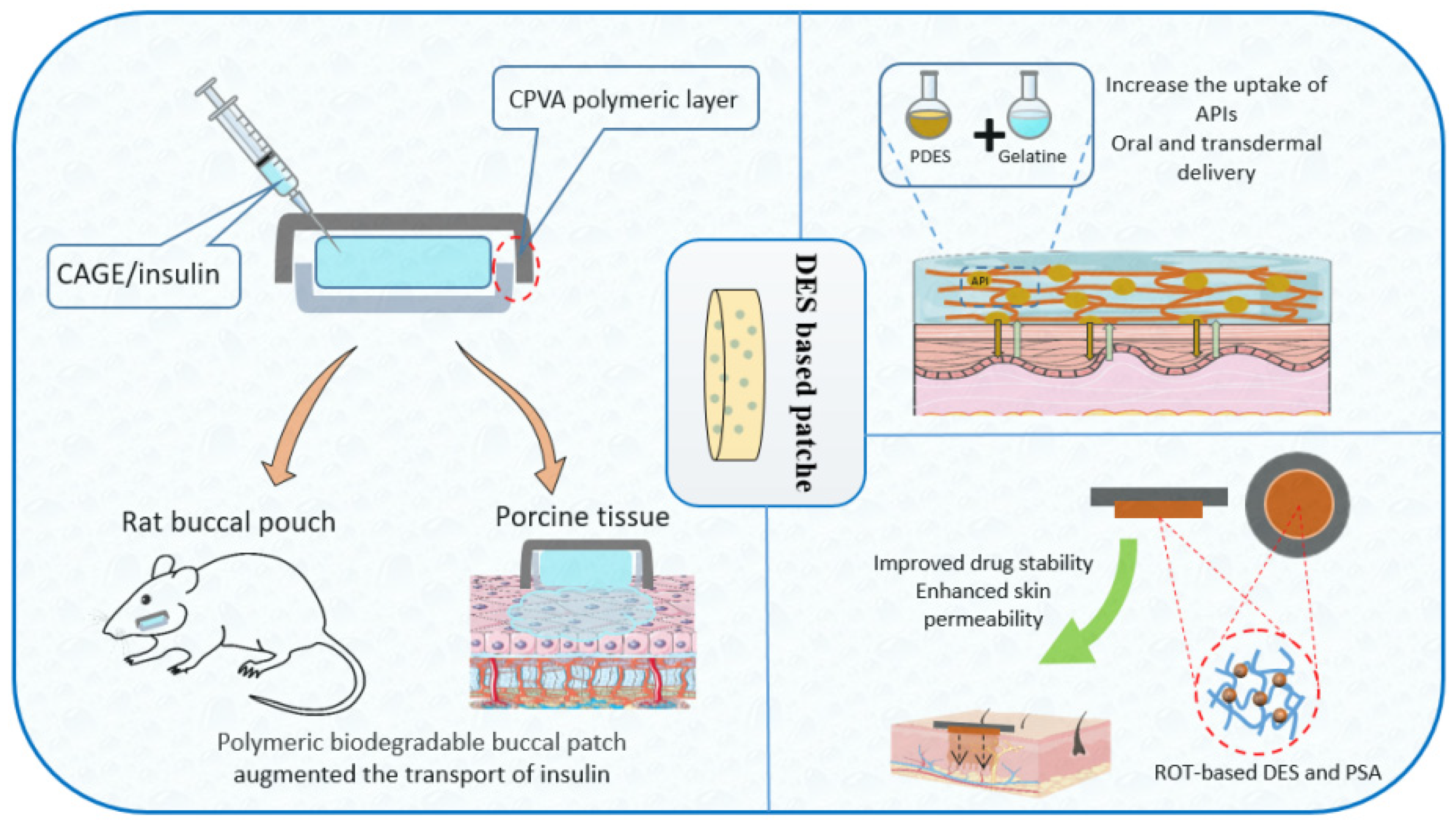
3. Role of DES in TDDS
3.1. DES as Stabilization Agents
3.2. DESs as Surfactants
3.3. DESs as Solvents
4. Influences of DES on SC
4.1. Liquid Modification
4.2. Keratins Modification
4.3. Lipid Exchange
5. Structure–Activity Relationship
5.1. Compositions
5.2. HBA/HBD Ratio
5.3. Substituents
6. Permeation Effect
6.1. Small Compounds
6.2. Biomacromolecule
6.2.1. Insulin
6.2.2. Protein
6.2.3. Polysaccharide
6.2.4. siRNA
6.2.5. Antigen Peptide
| API | HBA | HBD | Ratio | Application | References |
|---|---|---|---|---|---|
| Insulin | Choline Choline | Geranic acid Geranic acid | 1:4/1:2/1:1/2:1 1:1/1:2 | Provided a slow, consistent lowering of the blood glucose level over 12 h Promoted transdermal penetration of insulin | [30] [92] |
| Choline | Oleic acid | ||||
| BSA, OVA, Insulin | Choline | Geranic acid | 1:2 | Increased the ability of protein to penetrate the skin and lowed blood glucose | [108] |
| Dextran | Choline | Malic acid | 1:2 | Enhanced transdermal capacity and penetration | [109] |
| Choline | Geranic acid | 1:2 | Promoted transdermal penetration of macromolecules up to 150 kda | [83] | |
| siRNA | Choline | Geranic acid | 1:2 | Effective delivered of siRNA into the skin and suppress aberrant gene expression | [110,114] |
| Isovaleric acid | |||||
| Phenylpropionic acid | |||||
| 4-Phenolsulfonic acid | |||||
| Phenyl Phosphate | |||||
| Biphenyl-3-carboxylic acid | |||||
| IFN-α2 | Choline chloride | Fructose | 1:1 | Provided environmental protection and enhanced stability | [115] |
| Citric acid | |||||
| Malic acid | |||||
| RA-XII | Betaine | DL-Mandelic acid | 1:1 | Increased stability and permeability | [116] |
6.3. Rigid Nanoparticles
6.4. Complex Herb Extracts
7. DES-Based TDDS
7.1. DES-Based Microemulsions
7.2. DES-Based Hydrogel
7.3. DES Based Micelles
7.4. DES-Based Patches
7.5. Polymerized Drug-Based DESs
8. Toxicity of the DES in TDDS
9. Challenges and Prospect
Funding
Conflicts of Interest
References
- Md Moshikur, R.; Chowdhury, M.R.; Moniruzzaman, M.; Goto, M. Biocompatible ionic liquids and their applications in pharmaceutics. Green Chem. 2020, 22, 8116–8139. [Google Scholar] [CrossRef]
- Wu, H.; Deng, Z.; Zhou, B.; Qi, M.; Hong, M.; Ren, G. Improved transdermal permeability of ibuprofen by ionic liquid technology: Correlation between counterion structure and the physicochemical and biological properties. J. Mol. Liq. 2019, 283, 399–409. [Google Scholar] [CrossRef]
- Sidat, Z.; Marimuthu, T.; Kumar, P.; du Toit, L.C.; Kondiah, P.P.D.; Choonara, Y.E.; Pillay, V. Ionic Liquids as Potential and Synergistic Permeation Enhancers for Transdermal Drug Delivery. Pharmaceutics 2019, 11, 96. [Google Scholar] [CrossRef]
- Ramos, A.P.; Bouwstra, J.A.; Lafleur, M. Very Long Chain Lipids Favor the Formation of a Homogeneous Phase in Stratum Corneum Model Membranes. Langmuir 2020, 36, 13899–13907. [Google Scholar] [CrossRef] [PubMed]
- Santos de Almeida, T.; Júlio, A.; Saraiva, N.; Fernandes, A.S.; Araújo, M.E.M.; Baby, A.R.; Rosado, C.; Mota, J.P. Choline-versus imidazole-based ionic liquids as functional ingredients in topical delivery systems: Cytotoxicity, solubility, and skin permeation studies. Drug Dev. Ind. Pharm. 2017, 43, 1858–1865. [Google Scholar] [CrossRef] [PubMed]
- Qi, Q.M.; Duffy, M.; Curreri, A.M.; Balkaran, J.P.R.; Tanner, E.E.L.; Mitragotri, S. Comparison of Ionic Liquids and Chemical Permeation Enhancers for Transdermal Drug Delivery. Adv. Funct. Mater. 2020, 30, 2004257. [Google Scholar] [CrossRef]
- Yang, D.D.; Paterna, N.J.; Senetra, A.S.; Casey, K.R.; Trieu, P.D.; Caputo, G.A.; Vaden, T.D.; Carone, B.R. Synergistic interactions of ionic liquids and antimicrobials improve drug efficacy. iScience 2021, 24, 101853. [Google Scholar] [CrossRef]
- Jesus, A.R.; Soromenho, M.R.C.; Raposo, L.R.; Esperança, J.M.S.S.; Baptista, P.V.; Fernandes, A.R.; Reis, P.M. Enhancement of water solubility of poorly water-soluble drugs by new biocompatible N-acetyl amino acid N-alkyl cholinium-based ionic liquids. Eur. J. Pharm. Biopharm. 2019, 137, 227–232. [Google Scholar] [CrossRef]
- Babu, R.J.; Chen, L.; Kanikkannan, N. Fatty Alcohols, Fatty Acids, and Fatty Acid Esters as Penetration Enhancers. In Percutaneous Penetration Enhancers Chemical Methods in Penetration Enhancement: Modification of the Stratum Corneum; Dragicevic, N., Maibach, H.I., Eds.; Springer: Berlin/Heidelberg, Germany, 2015; pp. 133–150. [Google Scholar]
- Rambharose, S.; Kalhapure, R.S.; Jadhav, M.; Govender, T. Novel mono, di and tri-fatty acid esters bearing secondary amino acid ester head groups as transdermal permeation enhancers. New J. Chem. 2018, 42, 2232–2242. [Google Scholar] [CrossRef]
- Ben-Shabat, S.; Baruch, N.; Sintov, A.C. Conjugates of Unsaturated Fatty Acids with Propylene Glycol as Potentially Less-Irritant Skin Penetration Enhancers. Drug Dev. Ind. Pharm. 2007, 33, 1169–1175. [Google Scholar] [CrossRef]
- Eichner, A.; Stahlberg, S.; Sonnenberger, S.; Lange, S.; Dobner, B.; Ostermann, A.; Schrader, T.E.; Hauß, T.; Schroeter, A.; Huster, D.; et al. Influence of the penetration enhancer isopropyl myristate on stratum corneum lipid model membranes revealed by neutron diffraction and 2H NMR experiments. Biochim. Biophys. Acta (BBA) Biomembr. 2017, 1859, 745–755. [Google Scholar] [CrossRef] [PubMed]
- Perna, F.M.; Vitale, P.; Capriati, V. Deep eutectic solvents and their applications as green solvents. Curr. Opin. Green Sustain. Chem. 2020, 21, 27–33. [Google Scholar] [CrossRef]
- Yu, D.; Xue, Z.; Mu, T. Deep eutectic solvents as a green toolbox for synthesis. Cell Rep. Phys. Sci. 2022, 3, 100809. [Google Scholar] [CrossRef]
- Crespo, E.A.; Silva, L.P.; Martins, M.A.R.; Fernandez, L.; Ortega, J.; Ferreira, O.; Sadowski, G.; Held, C.; Pinho, S.P.; Coutinho, J.A.P. Characterization and Modeling of the Liquid Phase of Deep Eutectic Solvents Based on Fatty Acids/Alcohols and Choline Chloride. Ind. Eng. Chem. Res. 2017, 56, 12192–12202. [Google Scholar] [CrossRef]
- Zhao, Z.; Li, M.; Zheng, L.; Yang, Y.; Cui, X.; Xu, T.; Zhang, W.; Wang, C. Noninvasive transdermal delivery of mesoporous silica nanoparticles using deep eutectic solvent. J. Control. Release 2022, 343, 43–56. [Google Scholar] [CrossRef]
- Smith, E.L.; Abbott, A.P.; Ryder, K.S. Deep Eutectic Solvents (DESs) and Their Applications. Chem. Rev. 2014, 114, 11060–11082. [Google Scholar] [CrossRef]
- Álvarez, M.S.; Zhang, Y. Sketching neoteric solvents for boosting drugs bioavailability. J. Control. Release 2019, 311–312, 225–232. [Google Scholar] [CrossRef]
- Zhang, Q.; De Oliveira Vigier, K.; Royer, S.; Jérôme, F. Deep eutectic solvents: Syntheses, properties and applications. Chem. Soc. Rev. 2012, 41, 7108–7146. [Google Scholar] [CrossRef]
- Ge, X.; Gu, C.; Wang, X.; Tu, J. Deep eutectic solvents (DESs)-derived advanced functional materials for energy and environmental applications: Challenges, opportunities, and future vision. J. Mater. Chem. A 2017, 5, 8209–8229. [Google Scholar] [CrossRef]
- Ko, J.; Mandal, A.; Dhawan, S.; Shevachman, M.; Mitragotri, S.; Joshi, N. Clinical translation of choline and geranic acid deep eutectic solvent. Bioeng. Transl. Med. 2021, 6, e10191. [Google Scholar] [CrossRef]
- Angsantikul, P.; Peng, K.; Curreri, A.M.; Chua, Y.; Chen, K.Z.; Ehondor, J.; Mitragotri, S. Ionic Liquids and Deep Eutectic Solvents for Enhanced Delivery of Antibodies in the Gastrointestinal Tract. Adv. Funct. Mater. 2021, 31, 2002912. [Google Scholar] [CrossRef]
- Peng, K.; Shi, Y.; LaBarbiera, A.; Mitragotri, S. Mucoadhesive Ionic Liquid Gel Patches for Oral Delivery. ACS Biomater. Sci. Eng. 2020. [Google Scholar] [CrossRef]
- Vaidya, A.; Mitragotri, S. Ionic liquid-mediated delivery of insulin to buccal mucosa. J. Control. Release 2020, 327, 26–34. [Google Scholar] [CrossRef] [PubMed]
- Florindo, C.; Oliveira, F.S.; Rebelo, L.P.N.; Fernandes, A.M.; Marrucho, I.M. Insights into the Synthesis and Properties of Deep Eutectic Solvents Based on Cholinium Chloride and Carboxylic Acids. ACS Sustain. Chem. Eng. 2014, 2, 2416–2425. [Google Scholar] [CrossRef]
- Gutiérrez, M.C.; Ferrer, M.L.; Mateo, C.R.; del Monte, F. Freeze-Drying of Aqueous Solutions of Deep Eutectic Solvents: A Suitable Approach to Deep Eutectic Suspensions of Self-Assembled Structures. Langmuir 2009, 25, 5509–5515. [Google Scholar] [CrossRef] [PubMed]
- Qader, I.B.; Prasad, K. Recent Developments on Ionic Liquids and Deep Eutectic Solvents for Drug Delivery Applications. Pharm. Res. 2022. [Google Scholar] [CrossRef] [PubMed]
- Płotka-Wasylka, J.; de la Guardia, M.; Andruch, V.; Vilková, M. Deep eutectic solvents vs ionic liquids: Similarities and differences. Microchem. J. 2020, 159, 105539. [Google Scholar] [CrossRef]
- Tanner, E.E.L.; Curreri, A.M.; Balkaran, J.P.R.; Selig-Wober, N.C.; Yang, A.B.; Kendig, C.; Fluhr, M.P.; Kim, N.; Mitragotri, S. Design Principles of Ionic Liquids for Transdermal Drug Delivery. Adv. Mater. 2019, 31, 1901103. [Google Scholar] [CrossRef]
- Tanner, E.E.L.; Ibsen, K.N.; Mitragotri, S. Transdermal insulin delivery using choline-based ionic liquids (CAGE). J. Control. Release 2018, 286, 137–144. [Google Scholar] [CrossRef]
- Paiva, A.; Craveiro, R.; Aroso, I.; Martins, M.; Reis, R.L.; Duarte, A.R.C. Natural Deep Eutectic Solvents—Solvents for the 21st Century. ACS Sustain. Chem. Eng. 2014, 2, 1063–1071. [Google Scholar] [CrossRef]
- Dai, Y.; Witkamp, G.-J.; Verpoorte, R.; Choi, Y.H. Natural Deep Eutectic Solvents as a New Extraction Media for Phenolic Metabolites in Carthamus tinctorius L. Anal. Chem. 2013, 85, 6272–6278. [Google Scholar] [CrossRef] [PubMed]
- Osowska, N.; Ruzik, L. New Potentials in the Extraction of Trace Metal Using Natural Deep Eutectic Solvents (NADES). Food Anal. Methods 2019, 12, 926–935. [Google Scholar] [CrossRef]
- Radošević, K.; Ćurko, N.; Gaurina Srček, V.; Cvjetko Bubalo, M.; Tomašević, M.; Kovačević Ganić, K.; Radojčić Redovniković, I. Natural deep eutectic solvents as beneficial extractants for enhancement of plant extracts bioactivity. LWT 2016, 73, 45–51. [Google Scholar] [CrossRef]
- Dai, Y.; Verpoorte, R.; Choi, Y.H. Natural deep eutectic solvents providing enhanced stability of natural colorants from safflower (Carthamus tinctorius). Food Chem. 2014, 159, 116–121. [Google Scholar] [CrossRef]
- Jeliński, T.; Przybyłek, M.; Cysewski, P. Natural Deep Eutectic Solvents as Agents for Improving Solubility, Stability and Delivery of Curcumin. Pharm. Res. 2019, 36, 116. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, Y.; Su, J.; Zhang, X.; Wang, J.; Tu, Q. Preparation of a Multifunctional Wound Dressing Based on a Natural Deep Eutectic Solvent. ACS Sustain. Chem. Eng. 2020, 8, 14243–14252. [Google Scholar] [CrossRef]
- Wikene, K.O.; Rukke, H.V.; Bruzell, E.; Tønnesen, H.H. Investigation of the antimicrobial effect of natural deep eutectic solvents (NADES) as solvents in antimicrobial photodynamic therapy. J. Photochem. Photobiol. B Biol. 2017, 171, 27–33. [Google Scholar] [CrossRef]
- Radošević, K.; Čanak, I.; Panić, M.; Markov, K.; Bubalo, M.C.; Frece, J.; Srček, V.G.; Redovniković, I.R. Antimicrobial, cytotoxic and antioxidative evaluation of natural deep eutectic solvents. Environ. Sci. Pollut. Res. 2018, 25, 14188–14196. [Google Scholar] [CrossRef]
- Mbous, Y.P.; Hayyan, M.; Wong, W.F.; Looi, C.Y.; Hashim, M.A. Unraveling the cytotoxicity and metabolic pathways of binary natural deep eutectic solvent systems. Sci. Rep. 2017, 7, 41257. [Google Scholar] [CrossRef]
- Wang, J.-D.; Fu, L.-N.; Wang, L.-T.; Cai, Z.-H.; Wang, Y.-Q.; Yang, Q.; Fu, Y.-J. Simultaneous transformation and extraction of resveratrol from Polygonum cuspidatum using acidic natural deep eutectic solvent. Ind. Crops Prod. 2021, 173, 114140. [Google Scholar] [CrossRef]
- Yao, J.; Li, C.; Xiao, L.; Wu, Y.; Wu, Q.; Cui, Z.; Wang, B. Influence of natural deep eutectic solvents on stability and structure of cellulase. J. Mol. Liq. 2022, 346, 118238. [Google Scholar] [CrossRef]
- Stott, P.W.; Williams, A.C.; Barry, B.W. Transdermal delivery from eutectic systems: Enhanced permeation of a model drug, ibuprofen. J. Control. Release 1998, 50, 297–308. [Google Scholar] [CrossRef]
- Aroso, I.M.; Craveiro, R.; Rocha, Â.; Dionísio, M.; Barreiros, S.; Reis, R.L.; Paiva, A.; Duarte, A.R.C. Design of controlled release systems for THEDES—Therapeutic deep eutectic solvents, using supercritical fluid technology. Int. J. Pharm. 2015, 492, 73–79. [Google Scholar] [CrossRef] [PubMed]
- Abbott, A.P.; Ahmed, E.I.; Prasad, K.; Qader, I.B.; Ryder, K.S. Liquid pharmaceuticals formulation by eutectic formation. Fluid Phase Equilibria 2017, 448, 2–8. [Google Scholar] [CrossRef]
- Farooq, M.Q.; Abbasi, N.M.; Smith, E.A.; Petrich, J.W.; Anderson, J.L. Characterizing the Solvation Characteristics of Deep Eutectic Solvents Composed of Active Pharmaceutical Ingredients as a Hydrogen Bond Donor and/or Acceptor. ACS Sustain. Chem. Eng. 2022, 10, 3066–3078. [Google Scholar] [CrossRef]
- Silva, E.; Oliveira, F.; Silva, J.M.; Matias, A.; Reis, R.L.; Duarte, A.R.C. Optimal Design of THEDES Based on Perillyl Alcohol and Ibuprofen. Pharmaceutics 2020, 12, 1121. [Google Scholar] [CrossRef] [PubMed]
- Duarte, A.R.C.; Ferreira, A.S.D.; Barreiros, S.; Cabrita, E.; Reis, R.L.; Paiva, A. A comparison between pure active pharmaceutical ingredients and therapeutic deep eutectic solvents: Solubility and permeability studies. Eur. J. Pharm. Biopharm. 2017, 114, 296–304. [Google Scholar] [CrossRef]
- Mano, F.; Martins, M.; Sá-Nogueira, I.; Barreiros, S.; Borges, J.P.; Reis, R.L.; Duarte, A.R.C.; Paiva, A. Production of Electrospun Fast-Dissolving Drug Delivery Systems with Therapeutic Eutectic Systems Encapsulated in Gelatin. AAPS PharmSciTech 2017, 18, 2579–2585. [Google Scholar] [CrossRef]
- Yin, T.; Wu, J.; Yuan, J.; Wang, X. Therapeutic deep eutectic solvent based on osthole and paeonol: Preparation, characterization, and permeation behavior. J. Mol. Liq. 2022, 346, 117133. [Google Scholar] [CrossRef]
- Bolla, G.; Nangia, A. Pharmaceutical cocrystals: Walking the talk. Chem. Commun. 2016, 52, 8342–8360. [Google Scholar] [CrossRef]
- Souza, H.K.S.; Mateus, N.; de Freitas, V.; Gonçalves, M.P.; Cruz, L. Chemical/Color Stability and Rheological Properties of Cyanidin-3-Glucoside in Deep Eutectic Solvents as a Gateway to Design Task-Specific Bioactive Compounds. ACS Sustain. Chem. Eng. 2020, 8, 16184–16196. [Google Scholar] [CrossRef]
- Sakuragi, M.; Yano, R.; Hasnol, S.; Kusakabe, K. Evaluation of microemulsions containing a large amount of a deep eutectic solvent as a potential transdermal carrier of resveratrol. Jpn. J. Appl. Phys. 2020, 59. [Google Scholar] [CrossRef]
- Liu, Y.; Wu, Y.; Liu, J.; Wang, W.; Yang, Q.; Yang, G. Deep eutectic solvents: Recent advances in fabrication approaches and pharmaceutical applications. Int. J. Pharm. 2022, 622, 121811. [Google Scholar] [CrossRef] [PubMed]
- Yadav, N.; Bhakuni, K.; Bisht, M.; Bahadur, I.; Venkatesu, P. Expanding the Potential Role of Deep Eutectic Solvents toward Facilitating the Structural and Thermal Stability of α-Chymotrypsin. ACS Sustain. Chem. Eng. 2020, 8, 10151–10160. [Google Scholar] [CrossRef]
- Hossain, S.S.; Paul, S.; Samanta, A. Structural Stability and Conformational Dynamics of Cytochrome c in Hydrated Deep Eutectic Solvents. J. Phys. Chem. B 2021, 125, 5757–5765. [Google Scholar] [CrossRef]
- Olivares, B.; Martínez, F.; Rivas, L.; Calderón, C.; M. Munita, J.; R. Campodonico, P. A Natural Deep Eutectic Solvent Formulated to Stabilize β-Lactam Antibiotics. Sci. Rep. 2018, 8, 14900. [Google Scholar] [CrossRef]
- Toledo, M.L.; Pereira, M.M.; Freire, M.G.; Silva, J.P.A.; Coutinho, J.A.P.; Tavares, A.P.M. Laccase Activation in Deep Eutectic Solvents. ACS Sustain. Chem. Eng. 2019, 7, 11806–11814. [Google Scholar] [CrossRef]
- Delorme, A.E.; Andanson, J.-M.; Verney, V. Improving laccase thermostability with aqueous natural deep eutectic solvents. Int. J. Biol. Macromol. 2020, 163, 919–926. [Google Scholar] [CrossRef]
- Liu, C.; Qu, X.; Song, L.; Shang, R.; Wan, X.; Fang, L. Investigation on the effect of deep eutectic formation on drug-polymer miscibility and skin permeability of rotigotine drug-in-adhesive patch. Int. J. Pharm. 2020, 574, 118852. [Google Scholar] [CrossRef]
- Rebello, S.; Asok, A.K.; Mundayoor, S.; Jisha, M.S. Surfactants: Toxicity, remediation and green surfactants. Environ. Chem. Lett. 2014, 12, 275–287. [Google Scholar] [CrossRef]
- McDonald, S.; Murphy, T.; Imberti, S.; Warr, G.G.; Atkin, R. Amphiphilically Nanostructured Deep Eutectic Solvents. J. Phys. Chem. Lett. 2018, 9, 3922–3927. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Fernandez, A.; Leung, A.E.; Kelley, E.G.; Jackson, A.J. Complex by design: Hydrotrope-induced micellar growth in deep eutectic solvents. J. Colloid Interface Sci. 2021, 581, 292–298. [Google Scholar] [CrossRef] [PubMed]
- Atri, R.S.; Sanchez-Fernandez, A.; Hammond, O.S.; Manasi, I.; Doutch, J.; Tellam, J.P.; Edler, K.J. Morphology Modulation of Ionic Surfactant Micelles in Ternary Deep Eutectic Solvents. J. Phys. Chem. B 2020, 124, 6004–6014. [Google Scholar] [CrossRef] [PubMed]
- Buzolic, J.J.; Li, H.; Aman, Z.M.; Warr, G.G.; Atkin, R. Self-assembled nanostructure induced in deep eutectic solvents via an amphiphilic hydrogen bond donor. J. Colloid Interface Sci. 2022, 616, 121–128. [Google Scholar] [CrossRef] [PubMed]
- Agatemor, C.; Ibsen, K.N.; Tanner, E.E.L.; Mitragotri, S. Ionic liquids for addressing unmet needs in healthcare. Bioeng. Transl. Med. 2018, 3, 7–25. [Google Scholar] [CrossRef]
- Wu, J.; Yin, T. Amphiphilic Deep Eutectic Solvent Based on Lidocaine and Lauric Acid: Formation of Microemulsion and Gel. Langmuir 2022, 38, 1170–1177. [Google Scholar] [CrossRef]
- Ali, M.K.; Moshikur, R.M.; Wakabayashi, R.; Moniruzzaman, M.; Goto, M. Biocompatible Ionic Liquid-Mediated Micelles for Enhanced Transdermal Delivery of Paclitaxel. ACS Appl. Mater. Interfaces 2021, 13, 19745–19755. [Google Scholar] [CrossRef]
- Islam, M.R.; Uddin, S.; Chowdhury, M.R.; Wakabayashi, R.; Moniruzzaman, M.; Goto, M. Insulin Transdermal Delivery System for Diabetes Treatment Using a Biocompatible Ionic Liquid-Based Microemulsion. ACS Appl. Mater. Interfaces 2021, 13, 42461–42472. [Google Scholar] [CrossRef]
- Ellison, C.A.; Tankersley, K.O.; Obringer, C.M.; Carr, G.J.; Manwaring, J.; Rothe, H.; Duplan, H.; Géniès, C.; Grégoire, S.; Hewitt, N.J.; et al. Partition coefficient and diffusion coefficient determinations of 50 compounds in human intact skin, isolated skin layers and isolated stratum corneum lipids. Toxicol. In Vitro 2020, 69, 104990. [Google Scholar] [CrossRef]
- Rothe, H.; Obringer, C.; Manwaring, J.; Avci, C.; Wargniez, W.; Eilstein, J.; Hewitt, N.; Cubberley, R.; Duplan, H.; Lange, D.; et al. Comparison of protocols measuring diffusion and partition coefficients in the stratum corneum. J. Appl. Toxicol. 2017, 37, 806–816. [Google Scholar] [CrossRef]
- Gutiérrez, A.; Aparicio, S.; Atilhan, M. Design of arginine-based therapeutic deep eutectic solvents as drug solubilization vehicles for active pharmaceutical ingredients. Phys. Chem. Chem. Phys. 2019, 21, 10621–10634. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez, A.; Atilhan, M.; Aparicio, S. Theoretical Study on Deep Eutectic Solvents as Vehicles for the Delivery of Anesthetics. J. Phys. Chem. B 2020, 124, 1794–1805. [Google Scholar] [CrossRef] [PubMed]
- Chowdhury, M.R.; Moshikur, R.M.; Wakabayashi, R.; Tahara, Y.; Kamiya, N.; Moniruzzaman, M.; Goto, M. Development of a novel ionic liquid–curcumin complex to enhance its solubility, stability, and activity. Chem. Commun. 2019, 55, 7737–7740. [Google Scholar] [CrossRef]
- Chowdhury, M.R.; Moshikur, R.M.; Wakabayashi, R.; Tahara, Y.; Kamiya, N.; Moniruzzaman, M.; Goto, M. Ionic-Liquid-Based Paclitaxel Preparation: A New Potential Formulation for Cancer Treatment. Mol. Pharm. 2018, 15, 2484–2488. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez, A.; Atilhan, M.; Aparicio, S. A theoretical study on lidocaine solubility in deep eutectic solvents. Phys. Chem. Chem. Phys. 2018, 20, 27464–27473. [Google Scholar] [CrossRef]
- Shi, Y.; Zhao, Z.; Gao, Y.; Pan, D.C.; Salinas, A.K.; Tanner, E.E.L.; Guo, J.; Mitragotri, S. Oral delivery of sorafenib through spontaneous formation of ionic liquid nanocomplexes. J. Control. Release 2020, 322, 602–609. [Google Scholar] [CrossRef]
- Millard, J.W.; Alvarez-Núñez, F.A.; Yalkowsky, S.H. Solubilization by cosolvents: Establishing useful constants for the log–linear model. Int. J. Pharm. 2002, 245, 153–166. [Google Scholar] [CrossRef]
- Bhatt, J.; Mondal, D.; Bhojani, G.; Chatterjee, S.; Prasad, K. Preparation of bio-deep eutectic solvent triggered cephalopod shaped silver chloride-DNA hybrid material having antibacterial and bactericidal activity. Mater. Sci. Eng. C 2015, 56, 125–131. [Google Scholar] [CrossRef]
- Zahrina, I.; Nasikin, M.; Mulia, K.; Prajanto, M.; Yanuar, A. Molecular interactions between betaine monohydrate-glycerol deep eutectic solvents and palmitic acid: Computational and experimental studies. J. Mol. Liq. 2018, 251, 28–34. [Google Scholar] [CrossRef]
- Gaikwad, N.; Kudal, S.; Avachat, A.M. Choline-Amino Acid-Derived Bio-ionic Liquids for Solubility Enhancement of Zafirlukast. AAPS PharmSciTech 2022, 23, 146. [Google Scholar] [CrossRef]
- Warmińska, D.; Nowosielski, B.; Szewczyk, A.; Ruszkowski, J.; Prokopowicz, M. Effect of choline chloride based natural deep eutectic solvents on aqueous solubility and thermodynamic properties of acetaminophen. J. Mol. Liq. 2021, 323, 114834. [Google Scholar] [CrossRef]
- Qi, Q.M.; Mitragotri, S. Mechanistic study of transdermal delivery of macromolecules assisted by ionic liquids. J. Control. Release 2019, 311–312, 162–169. [Google Scholar] [CrossRef]
- Zheng, L.; Zhao, Z.; Yang, Y.; Li, Y.; Wang, C. Novel skin permeation enhancers based on amino acid ester ionic liquid: Design and permeation mechanism. Int J Pharm 2020, 576, 119031. [Google Scholar] [CrossRef] [PubMed]
- Pedro, S.N.; R. Freire, C.S.; Silvestre, A.J.D.; Freire, M.G. The Role of Ionic Liquids in the Pharmaceutical Field: An Overview of Relevant Applications. Int. J. Mol. Sci. 2020, 21, 8298. [Google Scholar] [CrossRef]
- Zakrewsky, M.; Lovejoy, K.S.; Kern, T.L.; Miller, T.E.; Le, V.; Nagy, A.; Goumas, A.M.; Iyer, R.S.; Del Sesto, R.E.; Koppisch, A.T.; et al. Ionic liquids as a class of materials for transdermal delivery and pathogen neutralization. Proc. Natl. Acad. Sci. USA 2014, 111, 13313–13318. [Google Scholar] [CrossRef] [PubMed]
- Jing, B.; Lan, N.; Qiu, J.; Zhu, Y. Interaction of Ionic Liquids with a Lipid Bilayer: A Biophysical Study of Ionic Liquid Cytotoxicity. J. Phys. Chem. B 2016, 120, 2781–2789. [Google Scholar] [CrossRef]
- Zhang, D.; Wang, H.-J.; Cui, X.-M.; Wang, C.-X. Evaluations of imidazolium ionic liquids as novel skin permeation enhancers for drug transdermal delivery. Pharm. Dev. Technol. 2017, 22, 511–520. [Google Scholar] [CrossRef]
- Barry, B.W. Novel mechanisms and devices to enable successful transdermal drug delivery. Eur. J. Pharm. Sci. 2001, 14, 101–114. [Google Scholar] [CrossRef]
- Ghanta, K.P.; Pal, T.; Mondal, S.; Bandyopadhyay, S. Microscopic Understanding of the Effect of Ionic Liquid on Protein from Molecular Simulation Studies. J. Phys. Chem. B 2020, 124, 3909–3921. [Google Scholar] [CrossRef]
- Zhang, Y.; Cao, Y.; Meng, X.; Li, C.; Wang, H.; Zhang, S. Enhancement of transdermal delivery of artemisinin using microemulsion vehicle based on ionic liquid and lidocaine ibuprofen. Colloids Surf. B Biointerfaces 2020, 189, 110886. [Google Scholar] [CrossRef]
- Jorge, L.R.; Harada, L.K.; Silva, E.C.; Campos, W.F.; Moreli, F.C.; Shimamoto, G.; Pereira, J.F.B.; Oliveira, J.M.; Tubino, M.; Vila, M.M.D.C.; et al. Non-invasive Transdermal Delivery of Human Insulin Using Ionic Liquids: In vitro Studies. Front. Pharmacol. 2020, 11, 243. [Google Scholar] [CrossRef] [PubMed]
- Islam, M.R.; Chowdhury, M.R.; Wakabayashi, R.; Tahara, Y.; Kamiya, N.; Moniruzzaman, M.; Goto, M. Choline and amino acid based biocompatible ionic liquid mediated transdermal delivery of the sparingly soluble drug acyclovir. Int. J. Pharm. 2020, 582, 119335. [Google Scholar] [CrossRef]
- Tao, G.-H.; He, L.; Sun, N.; Kou, Y. New generation ionic liquids: Cations derived from amino acids. Chem. Commun. 2005, 28, 3562–3564. [Google Scholar] [CrossRef] [PubMed]
- Yuan, J.; Zhou, N.; Wu, J.; Yin, T.; Jia, Y. Ionic liquids as effective additives to enhance the solubility and permeation for puerarin and ferulic acid. RSC Adv. 2022, 12, 3416–3422. [Google Scholar] [CrossRef] [PubMed]
- Yuan, J.; Wu, J.; Yin, T. Solubility and permeation enhancement of poor soluble drug by cholinium-amino acid based ionic liquids. J. Drug Deliv. Sci. Technol. 2020, 60, 102037. [Google Scholar] [CrossRef]
- Florindo, C.; Branco, L.C.; Marrucho, I.M. Quest for Green-Solvent Design: From Hydrophilic to Hydrophobic (Deep) Eutectic Solvents. ChemSusChem 2019, 12, 1549–1559. [Google Scholar] [CrossRef]
- Abranches, D.O.; Martins, M.A.R.; Silva, L.P.; Schaeffer, N.; Pinho, S.P.; Coutinho, J.A.P. Phenolic hydrogen bond donors in the formation of non-ionic deep eutectic solvents: The quest for type V DES. Chem. Commun. 2019, 55, 10253–10256. [Google Scholar] [CrossRef]
- Daadoue, S.; Al-Remawi, M.; Al-Mawla, L.; Idkaidek, N.; Khalid, R.M.; Al-Akayleh, F. Deep eutectic liquid as transdermal delivery vehicle of Risperidone. J. Mol. Liq. 2022, 345, 117347. [Google Scholar] [CrossRef]
- Wu, J.; Yin, T. Novel paeonol-matrine deep eutectic solvent: Physicochemical properties and cytotoxicity. J. Mol. Liq. 2022, 348, 118068. [Google Scholar] [CrossRef]
- Ijardar, S.P. Deep eutectic solvents composed of tetrabutylammonium bromide and PEG: Density, speed of sound and viscosity as a function of temperature. J. Chem. Thermodyn. 2020, 140, 105897. [Google Scholar] [CrossRef]
- Alhadid, A.; Mokrushina, L.; Minceva, M. Design of Deep Eutectic Systems: A Simple Approach for Preselecting Eutectic Mixture Constituents. Molecules 2020, 25, 1077. [Google Scholar] [CrossRef]
- Pedro, S.N.; Mendes, M.S.M.; Neves, B.M.; Almeida, I.F.; Costa, P.; Correia-Sá, I.; Vilela, C.; Freire, M.G.; Silvestre, A.J.D.; Freire, C.S.R. Deep Eutectic Solvent Formulations and Alginate-Based Hydrogels as a New Partnership for the Transdermal Administration of Anti-Inflammatory Drugs. Pharmaceutics 2022, 14, 827. [Google Scholar] [CrossRef] [PubMed]
- Chantereau, G.; Sharma, M.; Abednejad, A.; Neves, B.M.; Sèbe, G.; Coma, V.; Freire, M.G.; Freire, C.S.R.; Silvestre, A.J.D. Design of Nonsteroidal Anti-Inflammatory Drug-Based Ionic Liquids with Improved Water Solubility and Drug Delivery. ACS Sustain. Chem. Eng. 2019, 7, 14126–14134. [Google Scholar] [CrossRef]
- Münch, S.; Wohlrab, J.; Neubert, R.H.H. Dermal and transdermal delivery of pharmaceutically relevant macromolecules. Eur. J. Pharm. Biopharm. 2017, 119, 235–242. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Dong, Z.; Liu, W.; Wu, X.; He, H.; Lu, Y.; Wu, W.; Qi, J. Novel Pharmaceutical Strategies for Enhancing Skin Penetration of Biomacromolecules. Pharmaceuticals 2022, 15, 877. [Google Scholar] [CrossRef] [PubMed]
- Zhi, D.; Yang, T.; Zhang, T.; Yang, M.; Zhang, S.; Donnelly, R.F. Microneedles for gene and drug delivery in skin cancer therapy. J. Control. Release 2021, 335, 158–177. [Google Scholar] [CrossRef]
- Banerjee, A.; Ibsen, K.; Iwao, Y.; Zakrewsky, M.; Mitragotri, S. Transdermal Protein Delivery Using Choline and Geranate (CAGE) Deep Eutectic Solvent. Adv. Healthc. Mater. 2017, 6, 1601411. [Google Scholar] [CrossRef]
- Wu, X.; Chen, Z.; Li, Y.; Yu, Q.; Lu, Y.; Zhu, Q.; Li, Y.; An, D.; Qi, J.; Wu, W. Improving dermal delivery of hydrophilic macromolecules by biocompatible ionic liquid based on choline and malic acid. Int. J. Pharm. 2019, 558, 380–387. [Google Scholar] [CrossRef]
- Mandal, A.; Kumbhojkar, N.; Reilly, C.; Dharamdasani, V.; Ukidve, A.; Ingber, D.E.; Mitragotri, S. Treatment of psoriasis with NFKBIZ siRNA using topical ionic liquid formulations. Sci. Adv. 2020, 6, eabb6049. [Google Scholar] [CrossRef]
- Tahara, Y.; Morita, K.; Wakabayashi, R.; Kamiya, N.; Goto, M. Biocompatible Ionic Liquid Enhances Transdermal Antigen Peptide Delivery and Preventive Vaccination Effect. Mol. Pharm. 2020, 17, 3845–3856. [Google Scholar] [CrossRef]
- Uddin, S.; Islam, M.R.; Chowdhury, M.R.; Wakabayashi, R.; Kamiya, N.; Moniruzzaman, M.; Goto, M. Lipid-Based Ionic-Liquid-Mediated Nanodispersions as Biocompatible Carriers for the Enhanced Transdermal Delivery of a Peptide Drug. ACS Appl. Bio Mater. 2021, 4, 6256–6267. [Google Scholar] [CrossRef] [PubMed]
- Chowdhury, M.R.; Moshikur, R.M.; Wakabayashi, R.; Moniruzzaman, M.; Goto, M. Biocompatible ionic liquids assisted transdermal co-delivery of antigenic protein and adjuvant for cancer immunotherapy. Int. J. Pharm. 2021, 601, 120582. [Google Scholar] [CrossRef]
- Dharamdasani, V.; Mandal, A.; Qi, Q.M.; Suzuki, I.; Bentley, M.V.L.B.; Mitragotri, S. Topical delivery of siRNA into skin using ionic liquids. J. Control. Release 2020, 323, 475–482. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.S.; Lee, K.; Nam, M.W.; Jeong, K.M.; Lee, J.E.; Kim, N.W.; Yin, Y.; Lim, S.Y.; Yoo, D.E.; Lee, J.; et al. Natural deep eutectic solvents as a storage medium for human interferon-α2: A green and improved strategy for room-temperature biologics. J. Ind. Eng. Chem. 2018, 65, 343–348. [Google Scholar] [CrossRef]
- Liu, M.; Lai, Z.; Zhu, L.; Ding, X.; Tong, X.; Wang, Z.; Bi, Q.; Tan, N. Novel amorphous solid dispersion based on natural deep eutectic solvent for enhancing delivery of anti-tumor RA-XII by oral administration in rats. Eur. J. Pharm. Sci. 2021, 166, 105931. [Google Scholar] [CrossRef]
- Graf, C.; Gao, Q.; Schütz, I.; Noufele, C.N.; Ruan, W.; Posselt, U.; Korotianskiy, E.; Nordmeyer, D.; Rancan, F.; Hadam, S.; et al. Surface Functionalization of Silica Nanoparticles Supports Colloidal Stability in Physiological Media and Facilitates Internalization in Cells. Langmuir 2012, 28, 7598–7613. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Li, W.A.; Choi, Y.; Lewin, S.A.; Verbeke, C.S.; Dranoff, G.; Mooney, D.J. Injectable, spontaneously assembling, inorganic scaffolds modulate immune cells in vivo and increase vaccine efficacy. Nat. Biotechnol. 2015, 33, 64–72. [Google Scholar] [CrossRef] [PubMed]
- Ostrowski, A.; Nordmeyer, D.; Boreham, A.; Brodwolf, R.; Mundhenk, L.; Fluhr, J.W.; Lademann, J.; Graf, C.; Rühl, E.; Alexiev, U.; et al. Skin barrier disruptions in tape stripped and allergic dermatitis models have no effect on dermal penetration and systemic distribution of AHAPS-functionalized silica nanoparticles. Nanomed. Nanotechnol. Biol. Med. 2014, 10, 1571–1581. [Google Scholar] [CrossRef]
- Wang, C.; Zhu, J.; Zhang, D.; Yang, Y.; Zheng, L.; Qu, Y.; Yang, X.; Cui, X. Ionic liquid-microemulsions assisting in the transdermal delivery of Dencichine: Preparation, in-vitro and in-vivo evaluations, and investigation of the permeation mechanism. Int. J. Pharm. 2018, 535, 120–131. [Google Scholar] [CrossRef] [PubMed]
- Jamshidi, F.; Nouri, N.; Sereshti, H.; Shojaee Aliabadi, M.H. Synthesis of magnetic poly (acrylic acid-menthol deep eutectic solvent) hydrogel: Application for extraction of pesticides. J. Mol. Liq. 2020, 318, 114073. [Google Scholar] [CrossRef]
- Yu, Y.T.; Zhu, W.F.; Jin, C.; Chen, L.H.; Guan, Y. Research progress of the dosage form improvement and administration mode of triptolide. Chin. J. New Drugs 2016, 25, 1359–1362. [Google Scholar]
- Ning, Y.; Rao, Y.; Yu, Z.; Liang, W.; Li, F. Skin permeation profile and anti-inflammatory effect of anemonin extracted from weilingxian. Die Pharm. 2016, 71, 134–138. [Google Scholar] [CrossRef]
- Xiao, S.; Wang, L.; Han, W.; Gu, L.; Cui, X.; Wang, C. Novel Deep Eutectic Solvent–Hydrogel Systems for Synergistic Transdermal Delivery of Chinese Herb Medicine and Local Treatments for Rheumatoid Arthritis. Pharm. Res. 2022. [Google Scholar] [CrossRef]
- Xu, K.; Xu, P.; Wang, Y. Aqueous biphasic systems formed by hydrophilic and hydrophobic deep eutectic solvents for the partitioning of dyes. Talanta 2020, 213, 120839. [Google Scholar] [CrossRef]
- Moniruzzaman, M.; Tamura, M.; Tahara, Y.; Kamiya, N.; Goto, M. Ionic liquid-in-oil microemulsion as a potential carrier of sparingly soluble drug: Characterization and cytotoxicity evaluation. Int. J. Pharm. 2010, 400, 243–250. [Google Scholar] [CrossRef] [PubMed]
- Dhingra, D.; Bisht, M.; Bhawna, B.; Pandey, S. Enhanced solubility and improved stability of curcumin in novel water-in-deep eutectic solvent microemulsions. J. Mol. Liq. 2021, 339, 117037. [Google Scholar] [CrossRef]
- Sakuragi, M.; Yoshimura, H.; Kusakabe, K. Effects of structures of microemulsions containing a deep eutectic solvent on the entrapment amount and the skin permeation of resveratrol. Jpn. J. Appl. Phys. 2020, 59, 035002. [Google Scholar] [CrossRef]
- Ali, M.K.; Moshikur, R.M.; Wakabayashi, R.; Moniruzzaman, M.; Kamiya, N.; Goto, M. Biocompatible Ionic Liquid Surfactant-Based Microemulsion as a Potential Carrier for Sparingly Soluble Drugs. ACS Sustain. Chem. Eng. 2020, 8, 6263–6272. [Google Scholar] [CrossRef]
- Wang, W.; Cai, Y.; Liu, Y.; Zhao, Y.; Feng, J.; Liu, C. Microemulsions based on paeonol-menthol eutectic mixture for enhanced transdermal delivery: Formulation development and in vitro evaluation. Artif. Cells Nanomed. Biotechnol. 2017, 45, 1241–1246. [Google Scholar] [CrossRef]
- Negi, P.; Singh, B.; Sharma, G.; Beg, S.; Raza, K.; Katare, O.P. Phospholipid microemulsion-based hydrogel for enhanced topical delivery of lidocaine and prilocaine: QbD-based development and evaluation. Drug Deliv. 2016, 23, 941–957. [Google Scholar] [CrossRef]
- Jing, J.; Li, X.; Zhang, Y.; Liu, Y.; Lu, H.; Wang, J.; Wu, Y. pH-Responsive Regulation of a Surfactant-Free Microemulsion Based on Hydrophobic Deep Eutectic Solvents. Langmuir 2022, 38, 7898–7905. [Google Scholar] [CrossRef]
- Li, Y.; Hu, K.; Huang, C.; Hu, Y.; Ji, H.; Liu, S.; Gao, J. Improvement of solubility, stability and antioxidant activity of carotenoids using deep eutectic solvent-based microemulsions. Colloids Surf. B Biointerfaces 2022, 217, 112591. [Google Scholar] [CrossRef] [PubMed]
- Dhingra, D.; Behera, K.; Bhawna; Pandey, S. Formation of water-in-oil microemulsions within a hydrophobic deep eutectic solvent. Phys. Chem. Chem. Phys. 2021, 23, 10629–10635. [Google Scholar] [CrossRef] [PubMed]
- Onaciu, A.; Munteanu, R.A.; Moldovan, A.I.; Moldovan, C.S.; Berindan-Neagoe, I. Hydrogels Based Drug Delivery Synthesis, Characterization and Administration. Pharmaceutics 2019, 11, 432. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Li, S.; Yan, S. Starch as a reinforcement agent for poly(ionic liquid) hydrogels from deep eutectic solvent via frontal polymerization. Carbohydr. Polym. 2021, 263, 117996. [Google Scholar] [CrossRef]
- Li, W.; Zhao, X.; Huang, T.; Ren, Y.; Gong, W.; Guo, Y.; Wang, J.; Tu, Q. Preparation of sodium hyaluronate/dopamine/AgNPs hydrogel based on the natural eutetic solvent as an antibaterial wound dressing. Int. J. Biol. Macromol. 2021, 191, 60–70. [Google Scholar] [CrossRef] [PubMed]
- Silva, J.M.; Silva, E.; Reis, R.L. Therapeutic deep eutectic solvents assisted the encapsulation of curcumin in alginate-chitosan hydrogel beads. Sustain. Chem. Pharm. 2021, 24, 100553. [Google Scholar] [CrossRef]
- Rossetti, A.; Pizzetti, F.; Rossi, F.; Mauri, E.; Borghi, E.; Ottaviano, E.; Sacchetti, A. Synthesis and characterization of carbomer-based hydrogels for drug delivery applications. Int. J. Polym. Mater. Polym. Biomater. 2021, 70, 743–753. [Google Scholar] [CrossRef]
- Wang, H.; Li, J.; Yu, X.; Yan, G.; Tang, X.; Sun, Y.; Zeng, X.; Lin, L. Cellulose nanocrystalline hydrogel based on a choline chloride deep eutectic solvent as wearable strain sensor for human motion. Carbohydr. Polym. 2021, 255, 117443. [Google Scholar] [CrossRef]
- Nahar, Y.; Horne, J.; Truong, V.; Bissember, A.C.; Thickett, S.C. Preparation of thermoresponsive hydrogels via polymerizable deep eutectic monomer solvents. Polym. Chem. 2021, 12, 254–264. [Google Scholar] [CrossRef]
- Zhang, H.; Gao, T.; Jiang, L.; Meng, X.; Wang, J.; Ma, N.; Wei, H.; Zhang, X. Conductive and transparent poly (N-isopropylacrylamide) hydrogels with tunable LCST copolymerized by the green acrylamide-based deep eutectic solvent. Eur. Polym. J. 2022, 173, 111266. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, Y.; Lin, Z.; Huang, T.; Li, W.; Gong, W.; Guo, Y.; Su, J.; Wang, J.; Tu, Q. A green method of preparing a natural and degradable wound dressing containing aloe vera as an active ingredient. Compos. Part B Eng. 2021, 222, 109047. [Google Scholar] [CrossRef]
- Gao, Y.-R.; Zhang, W.-X.; Wei, Y.-N.; Li, Y.; Fei, T.; Shu, Y.; Wang, J.-H. Ionic liquids enable the preparation of a copper-loaded gel with transdermal delivery function for wound dressings. Biomater. Sci. 2022, 10, 1041–1052. [Google Scholar] [CrossRef] [PubMed]
- Moshikur, R.M.; Ali, M.K.; Moniruzzaman, M.; Goto, M. Recent advances in surface-active ionic liquid-assisted self-assembly systems for drug delivery. Curr. Opin. Colloid Interface Sci. 2021, 56, 101515. [Google Scholar] [CrossRef]
- Kumar, H.; Kaur, G. Scrutinizing Self-Assembly, Surface Activity and Aggregation Behavior of Mixtures of Imidazolium Based Ionic Liquids and Surfactants: A Comprehensive Review. Front. Chem. 2021, 9, 667941. [Google Scholar] [CrossRef] [PubMed]
- Chowdhury, M.R.; Moshikur, R.M.; Wakabayashi, R.; Tahara, Y.; Kamiya, N.; Moniruzzaman, M.; Goto, M. In vivo biocompatibility, pharmacokinetics, antitumor efficacy, and hypersensitivity evaluation of ionic liquid-mediated paclitaxel formulations. Int. J. Pharm. 2019, 565, 219–226. [Google Scholar] [CrossRef] [PubMed]
- Kurnik, I.S.; D’Angelo, N.A.; Mazzola, P.G.; Chorilli, M.; Kamei, D.T.; Pereira, J.F.B.; Vicente, A.A.; Lopes, A.M. Polymeric micelles using cholinium-based ionic liquids for the encapsulation and release of hydrophobic drug molecules. Biomater. Sci. 2021, 9, 2183–2196. [Google Scholar] [CrossRef]
- Qu, W.; Qader, I.B.; Abbott, A.P. Controlled release of pharmaceutical agents using eutectic modified gelatin. Drug Deliv. Transl. Res. 2022, 12, 1187–1194. [Google Scholar] [CrossRef]
- Yang, D.; Liu, C.; Piao, H.; Quan, P.; Fang, L. Enhanced Drug Loading in the Drug-in-Adhesive Transdermal Patch Utilizing a Drug–Ionic Liquid Strategy: Insight into the Role of Ionic Hydrogen Bonding. Mol. Pharm. 2021, 18, 1157–1166. [Google Scholar] [CrossRef]
- Sánchez-Leija, R.J.; Pojman, J.A.; Luna-Bárcenas, G.; Mota-Morales, J.D. Controlled release of lidocaine hydrochloride from polymerized drug-based deep-eutectic solvents. J. Mater. Chem. B 2014, 2, 7495–7501. [Google Scholar] [CrossRef] [PubMed]
- Mota-Morales, J.D.; Gutiérrez, M.C.; Ferrer, M.L.; Sanchez, I.C.; Elizalde-Peña, E.A.; Pojman, J.A.; Monte, F.D.; Luna-Bárcenas, G. Deep eutectic solvents as both active fillers and monomers for frontal polymerization. J. Polym. Sci. Part A Polym. Chem. 2013, 51, 1767–1773. [Google Scholar] [CrossRef]
- Serrano, M.C.; Gutiérrez, M.C.; Jiménez, R.; Ferrer, M.L.; Monte, F.d. Synthesis of novel lidocaine-releasing poly(diol-co-citrate) elastomers by using deep eutectic solvents. Chem. Commun. 2012, 48, 579–581. [Google Scholar] [CrossRef] [PubMed]
- Flieger, J.; Flieger, M. Ionic Liquids Toxicity—Benefits and Threats. Int. J. Mol. Sci. 2020, 21, 6267. [Google Scholar] [CrossRef] [PubMed]
- Holbrey, J.; Reichert, W.; Spear, S.; Swatloski, R.; Turner, M.B.; Visser, A.E. Getting started with ionic liquids: An experience-based tutorial on synthesis and handling. Abstr. Pap. Am. Chem. Soc. 2022, 224, 001-IEC. [Google Scholar]
- Perales, E.; García, C.B.; Lomba, L.; Aldea, L.; García, J.I.; Giner, B. Comparative ecotoxicology study of two neoteric solvents: Imidazolium ionic liquid vs. glycerol derivative. Ecotoxicol. Environ. Saf. 2016, 132, 429–434. [Google Scholar] [CrossRef] [PubMed]
- Radošević, K.; Železnjak, J.; Cvjetko Bubalo, M.; Radojčić Redovniković, I.; Slivac, I.; Gaurina Srček, V. Comparative in vitro study of cholinium-based ionic liquids and deep eutectic solvents toward fish cell line. Ecotoxicol. Environ. Saf. 2016, 131, 30–36. [Google Scholar] [CrossRef] [PubMed]
- Ahmadi, R.; Hemmateenejad, B.; Safavi, A.; Shojaeifard, Z.; Mohabbati, M.; Firuzi, O. Assessment of cytotoxicity of choline chloride-based natural deep eutectic solvents against human HEK-293 cells: A QSAR analysis. Chemosphere 2018, 209, 831–838. [Google Scholar] [CrossRef] [PubMed]
- Giner, B.; Lafuente, C.; Lapeña, D.; Errazquin, D.; Lomba, L. QSAR study for predicting the ecotoxicity of NADES towards Aliivibrio fischeri. Exploring the use of mixing rules. Ecotoxicol. Environ. Saf. 2020, 191, 110004. [Google Scholar] [CrossRef]
- Welss, T.; Basketter, D.A.; Schröder, K.R. In vitro skin irritation: Facts and future. State of the art review of mechanisms and models. Toxicol. In Vitro 2004, 18, 231–243. [Google Scholar] [CrossRef]
- Dobler, D.; Schmidts, T.; Klingenhöfer, I.; Runkel, F. Ionic liquids as ingredients in topical drug delivery systems. Int. J. Pharm. 2013, 441, 620–627. [Google Scholar] [CrossRef]
- Wu, X.; Yu, Q.; Wu, J.; Li, T.; Ding, N.; Wu, W.; Lu, Y.; Zhu, Q.; Chen, Z.; Qi, J. Ionic liquids containing ketoconazole improving topical treatment of T. Interdigitale infection by synergistic action. Int. J. Pharm. 2020, 589, 119842. [Google Scholar] [CrossRef] [PubMed]
- Marei, H.F.; Arafa, M.F.; Essa, E.A.; El Maghraby, G.M. Lidocaine as eutectic forming drug for enhanced transdermal delivery of nonsteroidal anti-inflammatory drugs. J. Drug Deliv. Sci. Technol. 2021, 61, 102338. [Google Scholar] [CrossRef]
- Zakrewsky, M.; Banerjee, A.; Apte, S.; Kern, T.L.; Jones, M.R.; Sesto, R.E.D.; Koppisch, A.T.; Fox, D.T.; Mitragotri, S. Choline and Geranate Deep Eutectic Solvent as a Broad-Spectrum Antiseptic Agent for Preventive and Therapeutic Applications. Adv. Healthc. Mater. 2016, 5, 1282–1289. [Google Scholar] [CrossRef] [PubMed]
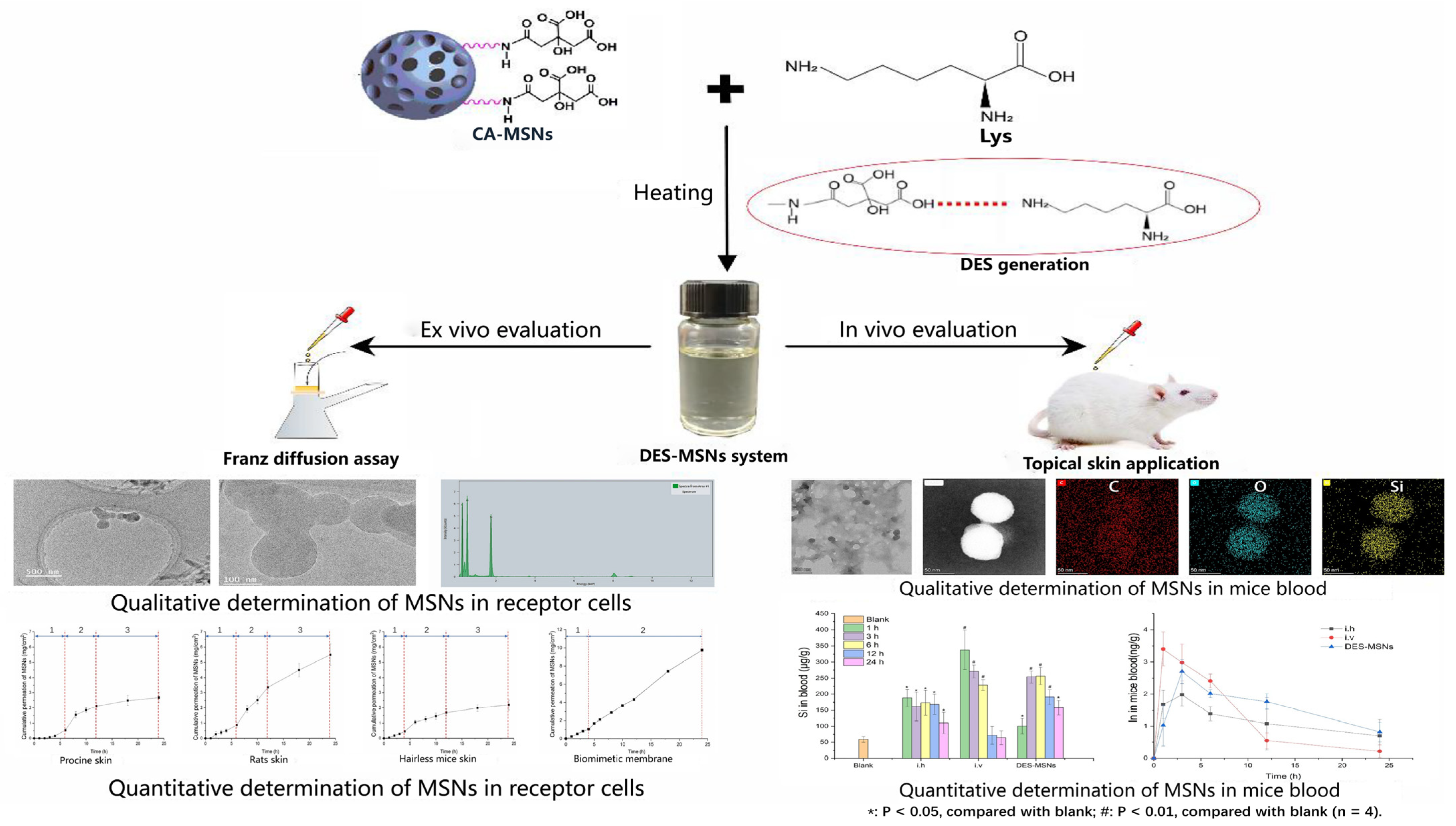
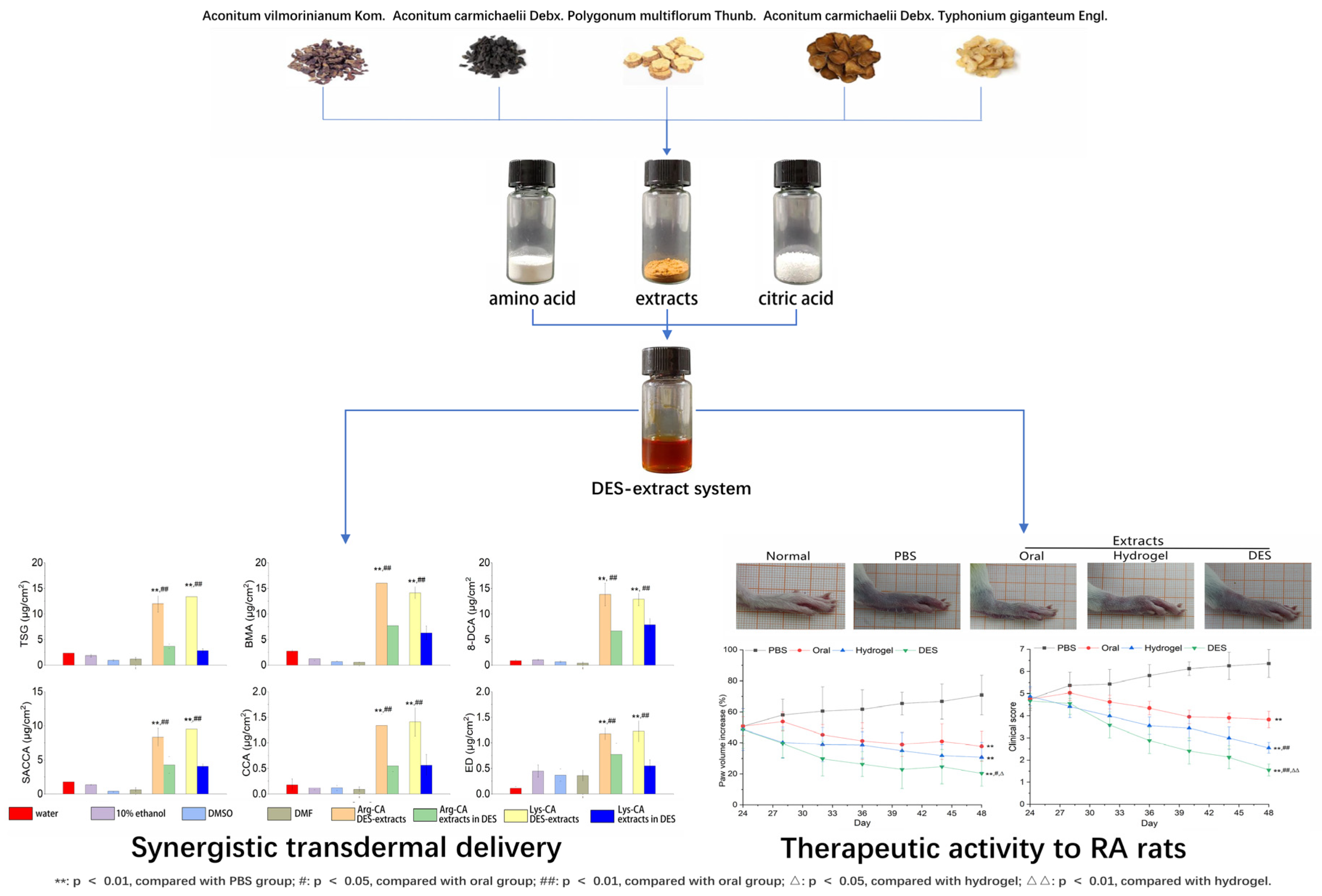
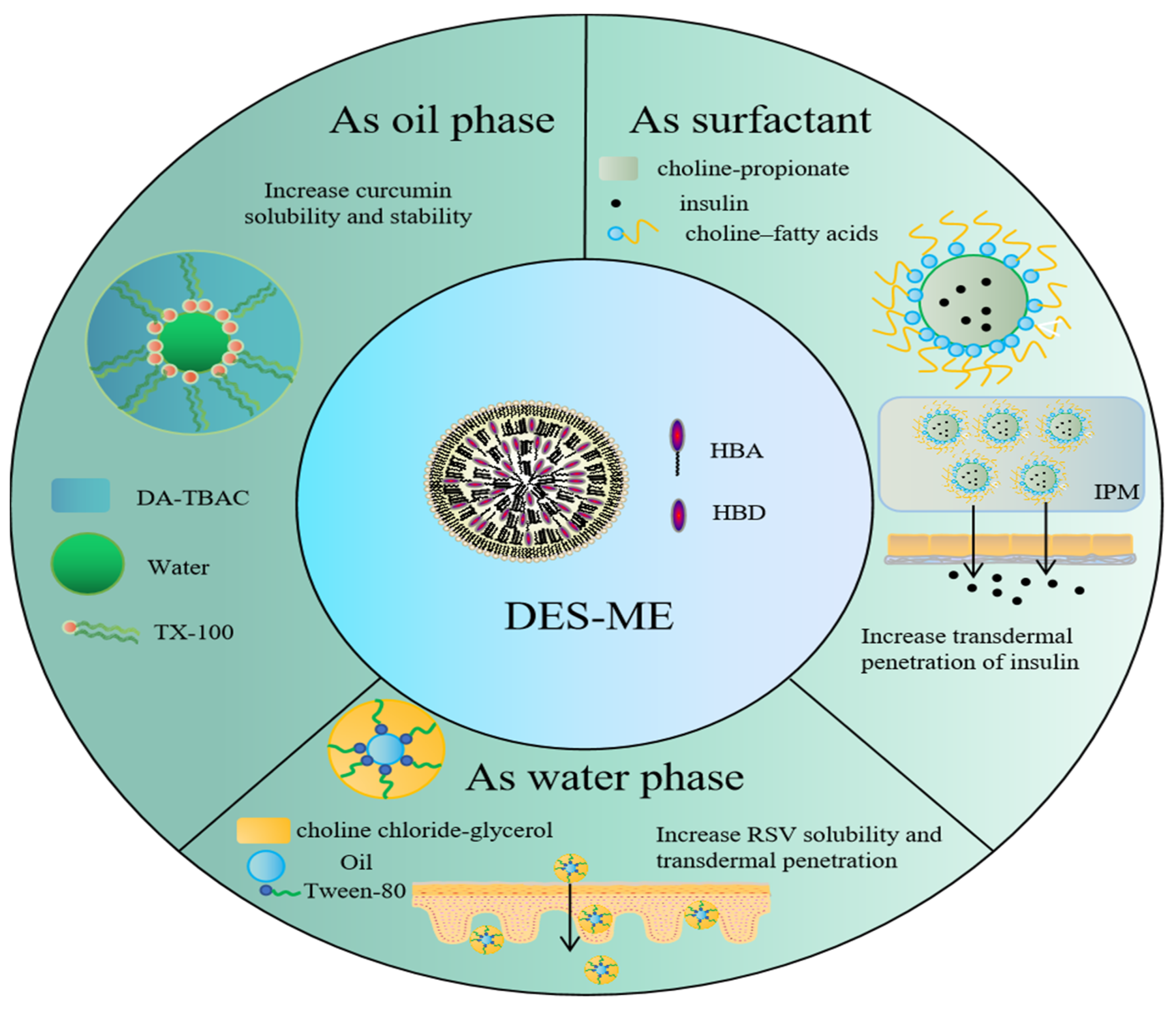
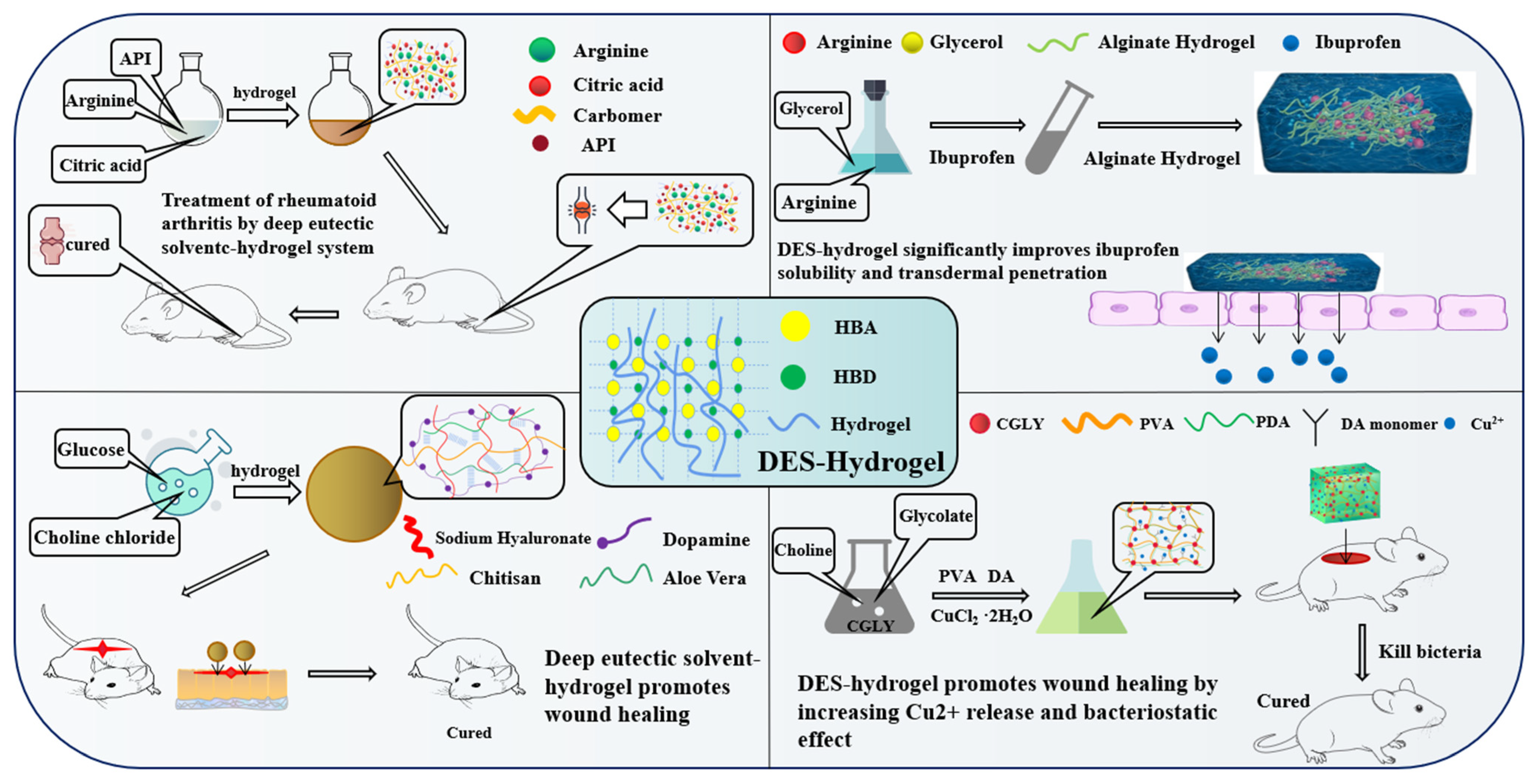

| HBA | HBD | Ratio | Application | References |
|---|---|---|---|---|
| Glucose | Lactic acid | 1:5 | As an efficient solvents for the extraction of phenolic | [32] |
| Proline | Malic acid | 1:1 | ||
| Choline chloride | Sucrose | 1:1/4:1 | ||
| Choline chloride | Glucose | 1:1/2:1/5:2 | ||
| Choline chloride | Sorbitol | 3:1/5:2 | ||
| Choline | 1,2-Propanediol | 1:1/1:1.5/1:2/1:3/2:1 | ||
| Choline chloride | Glucose | 2:1 | More efficient than methanol in extracting phenolic substances from grape skins | [34] |
| Xylose | 2:1 | |||
| Glycerin | 1:2 | |||
| Fructose | 1.9:1 | |||
| Malic acid | 1:1 | |||
| Glucose | Lactic acid | 1:5 | The stability of carthamin increased 2–8 times | [35] |
| Choline chloride | Sucrose | 1:1/4:1 | ||
| Proline | Malic acid | 1:1 | ||
| Choline chloride | Xylitol | 5:2 | ||
| Choline chloride | Glucose | 3:1/1:1/1:3 | Increased solubility of curcumin significantly | [36] |
| Fructose | ||||
| Sorbitol | ||||
| Xylitol | ||||
| Maltose | ||||
| Sucrose | ||||
| Glycerin | ||||
| Choline chloride | Mannose | 1:2 | Antibacterial, anti-inflammatory, nutritional supplement | [37] |
| Sucrose | Citric acid | 1:1 | Antibacterial | [38] |
| Fructose, glucose | Malic acid | 1:1:1 | ||
| Betaine, proline | Malic acid | 1:1:1 | Antioxidant activity | [39] |
| Choline chloride | Glucose | 2:1 | Anti-cancer activity | [40] |
| Choline chloride | Fructose | |||
| Choline chloride | Oxalic acid | 1:2 | Improved extraction efficiency and conversion rate | [41] |
| Betaine | 1,4-butanediol | 1:2 | Improve stability of cellulase | [42] |
| Choline chloride | 1,4-butanediol |
| HBA | HBD | Ratio (w/w) | Application | References |
|---|---|---|---|---|
| L-Menthol | Ibuprofen | (w/w) 20:80/25:75/30:70/35:65/ 40:60/50:50/60:40 | Improved solubility and transdermal penetration | [43,47] |
| 1,8-Cineole | ||||
| LD-Menthol | ||||
| Thymol | ||||
| Menthol | Ibuprofen | 1:3 | Development of THEDES with controlled release | [44] |
| Lidocaine | Ibuprofen | 1:1 | Improved the solubility of API | [46] |
| Menthol | Ibuprofen | 3:1 | Improved solubility and transdermal penetration | [48] |
| Choline chloride | Salicylic acid | 1:1 | Reduce waste of valuable compounds, increase the solubility of water-insoluble drugs, and overcome pharmacokinetic differences | [45] |
| Aspirin | 1:2 | |||
| Paracetamol | 1:1 | |||
| Thymol | 1:2 | |||
| Choline chloride | mandelic acid | 1:2 | Antibacterial | [49] |
| Atropine | Capric acid | 1:2 | Improved the solubility of API and avoid recrystallization | [46] |
| Dodecanoic acid | 1:2 | |||
| Lidocaine | Linoleic acid | 1:1 | ||
| Dodecanoic acid | 1:2 | |||
| Capric acid | 1:1/1:2/1:3 | |||
| 1,8-Octanediol | 1:1/1:2/1:4 | |||
| Tetracaine | 1:2 | |||
| Prilocaine | 1:3 | |||
| Vanillin | 3:2 | |||
| Osthole | Paeonol | 8:2 | 40 times increased in solubility | [50] |
| Surfactant | Oil Phase | Water Phase | API | Application | References |
|---|---|---|---|---|---|
| Lidocaine-lauric acid (1:1/4:6) | Propylene glycol | IPM | Lidocaine | Acts as a surfactant to facilitate morphological transformation and gel formation | [67] |
| Choline-linoleic acid(1:1), Span-20 | IPM | choline hydroxide- Propionic acid (1:1) | Insulin | Enhance the transdermal penetration of insulin through the intercellular pathway | [69] |
| Tween-80 | DL-menthol- Acetic acid (1:2) | Water | Carotenoids | Improvement of solubility, stability and antioxidant activity of carotenoids | [133] |
| DL-menthl- Octanoic acid (1:2) | |||||
| Triton X-100 | Tetra-n-butylammonium chloride- n-Decanoic acid (1:2) | Water | / | Participate in the formation of green, non-toxic, readily available emulsions | [134] |
| Triton X-100 | Tetra-n-butylammonium chloride- n-Decanoic acid (1:2) | Water | Curcumin | Improve the solubility and stability of curcumin | [127] |
| Tween-80, Span-20 | IPM | Choline chloride-Glycerin (1:2) | Resveratrol | Improved the solubility and skin penetration of resveratrol | [128] |
| Span-20 | IPM | Choline hydroxide-Oleic acid (1:1) | Celecoxib, Acyclovir | Increase the solubility of celecoxib and acyclovir | [129] |
| Cremophor EL, Glycerin | Menthol-Paeonol (6:4/5:5/4:6) | Water | Paeonol | Increased stability of paeonol and enhanced skin penetration in vitro | [130] |
| Tween-80 | Lidocaine-Prilocaine (1:1), | Water | Lidocaine | Better stability and permeation-promoting effect on Lidocaine | [131] |
| Tween-80, Span-20 | Lidocaine- Ibuprofen (1:2) | Water | Artemisinin | Enhanced transdermal delivery of artemisinin | [91] |
| / | Menthol-n-Octanoic acid (1:3) | Water | / | Preparation of pH-responsive microemulsions | [132] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, J.; Li, M.; Duan, L.; Lin, Y.; Cui, X.; Yang, Y.; Wang, C. Deep Eutectic Systems as Novel Vehicles for Assisting Drug Transdermal Delivery. Pharmaceutics 2022, 14, 2265. https://doi.org/10.3390/pharmaceutics14112265
Wang J, Li M, Duan L, Lin Y, Cui X, Yang Y, Wang C. Deep Eutectic Systems as Novel Vehicles for Assisting Drug Transdermal Delivery. Pharmaceutics. 2022; 14(11):2265. https://doi.org/10.3390/pharmaceutics14112265
Chicago/Turabian StyleWang, Jinbao, Mingjian Li, Langhuan Duan, Yameng Lin, Xiuming Cui, Ye Yang, and Chengxiao Wang. 2022. "Deep Eutectic Systems as Novel Vehicles for Assisting Drug Transdermal Delivery" Pharmaceutics 14, no. 11: 2265. https://doi.org/10.3390/pharmaceutics14112265
APA StyleWang, J., Li, M., Duan, L., Lin, Y., Cui, X., Yang, Y., & Wang, C. (2022). Deep Eutectic Systems as Novel Vehicles for Assisting Drug Transdermal Delivery. Pharmaceutics, 14(11), 2265. https://doi.org/10.3390/pharmaceutics14112265





