PLGA-Based Nanoparticles for Neuroprotective Drug Delivery in Neurodegenerative Diseases
Abstract
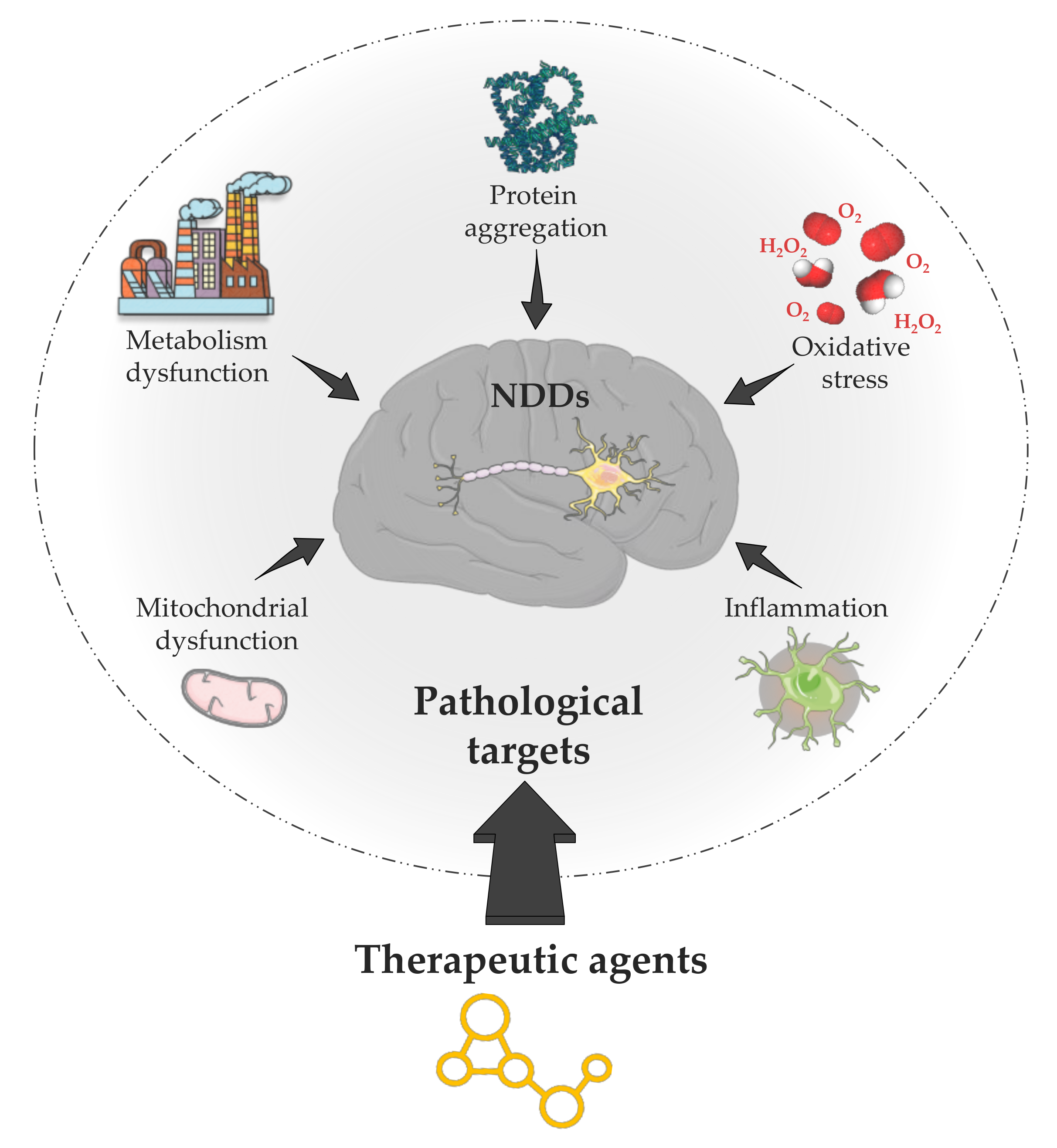
1. Introduction
| Disease | Prevalence (Per 100,000 Person) | Symptoms | Affected Areas |
|---|---|---|---|
| AD | 511–690 [5] | Memory impairment, changes in thinking, judgment, language, behavioral changes, etc. | Cortex, hippocampus, brainstem |
| PD | 100–200 [6] | Rest tremors, slowness of movement, decrease of spontaneous mobility, muscular stiffness, etc. | Basal ganglia, cortex |
| HD | 5.96–13.7 [7] | Motor disorders, breathing difficulties, speech and swallowing disorders, etc. | Striatum and other basal ganglia regions |
| ALS | 3.92–4.96 [8] | Progressive muscle paralysis, muscle atrophy, spasticity, breathing and swallowing disorders, etc. | Motor cortex, spinal cord |
| MS | 35.87–35.95 [9] | Numbness in a limb, vision problems, electric shock sensations in a limb or the back, movement problems, etc. | Brain, spinal cord, optic nerve |
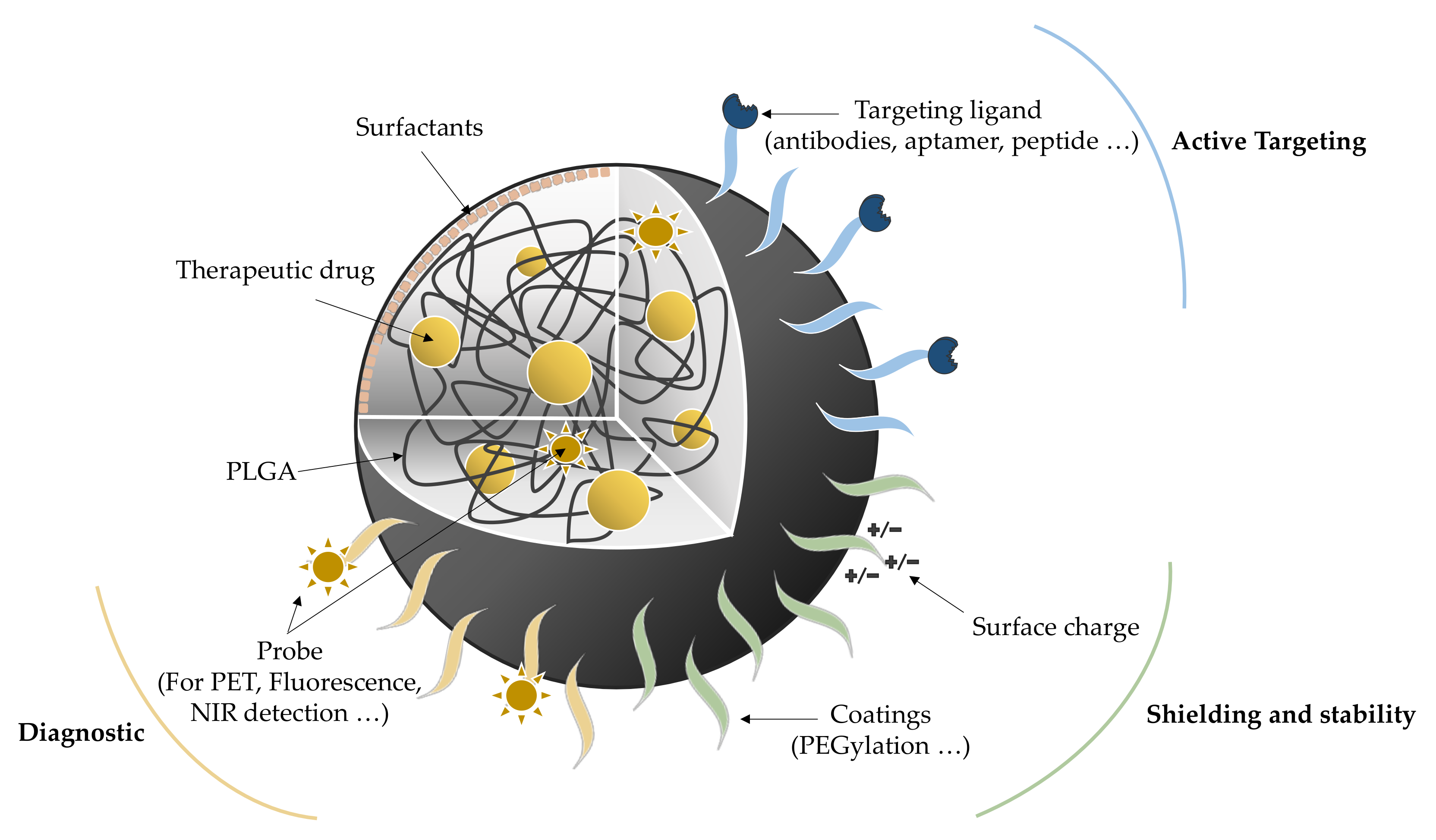
2. PLGA NPs
2.1. PLGA NP Formulation and Optimization
2.2. Administration Routes
3. PLGA NPs for Neuroprotective Drug Delivery in Neurological Disorder Therapy
3.1. Alzheimer’s Disease
3.1.1. Thymoquinone
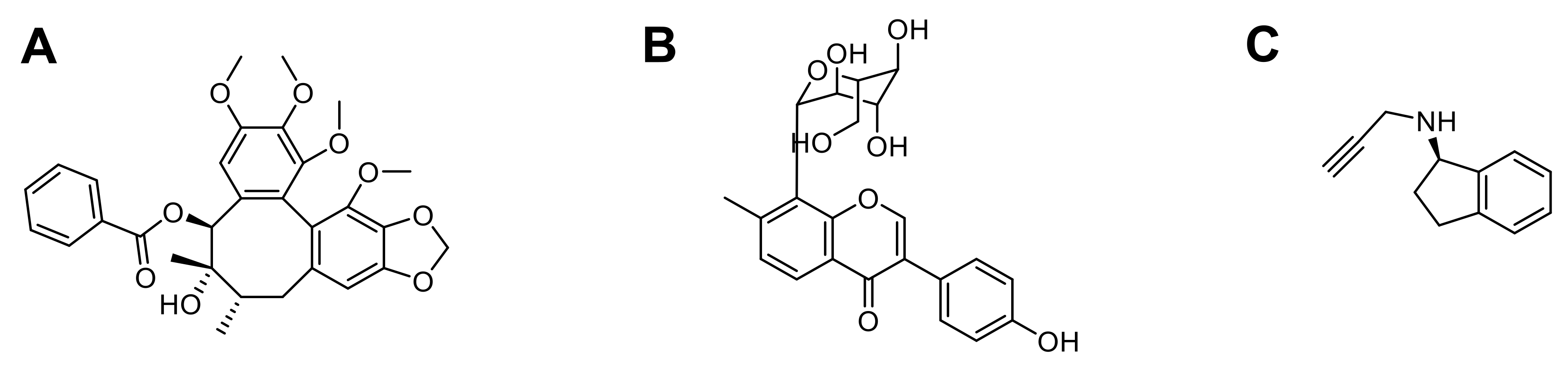
3.1.2. Huperzine A
3.1.3. Rhynchophylline
3.2. Parkinson’s Disease
3.2.1. Schisantherin A
3.2.2. Puerarin
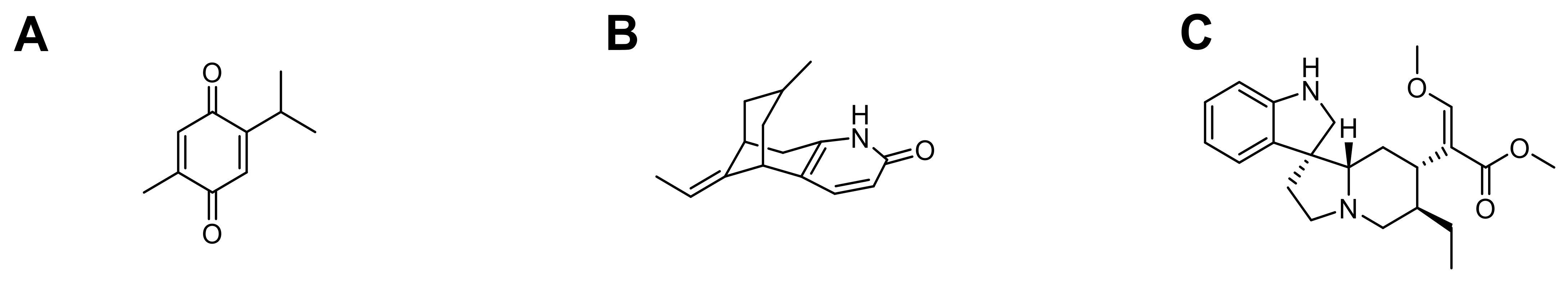
3.2.3. Rasagiline
3.3. Huntington’s Disease
3.3.1. Oligonucleotides QBP1, NT17, and PGQ9P2
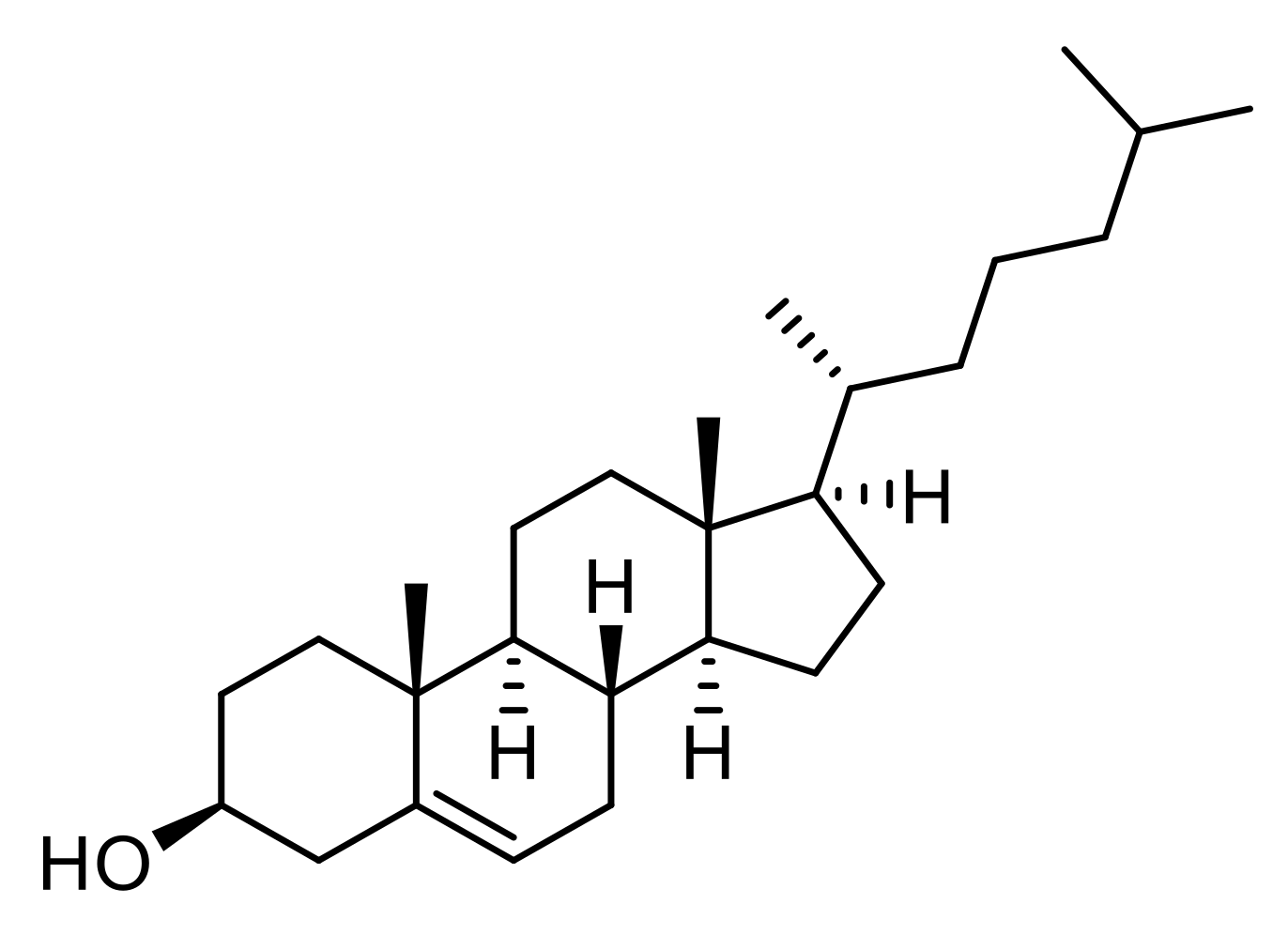
3.3.2. Cholesterol
3.4. Amyotrophic Lateral Sclerosis
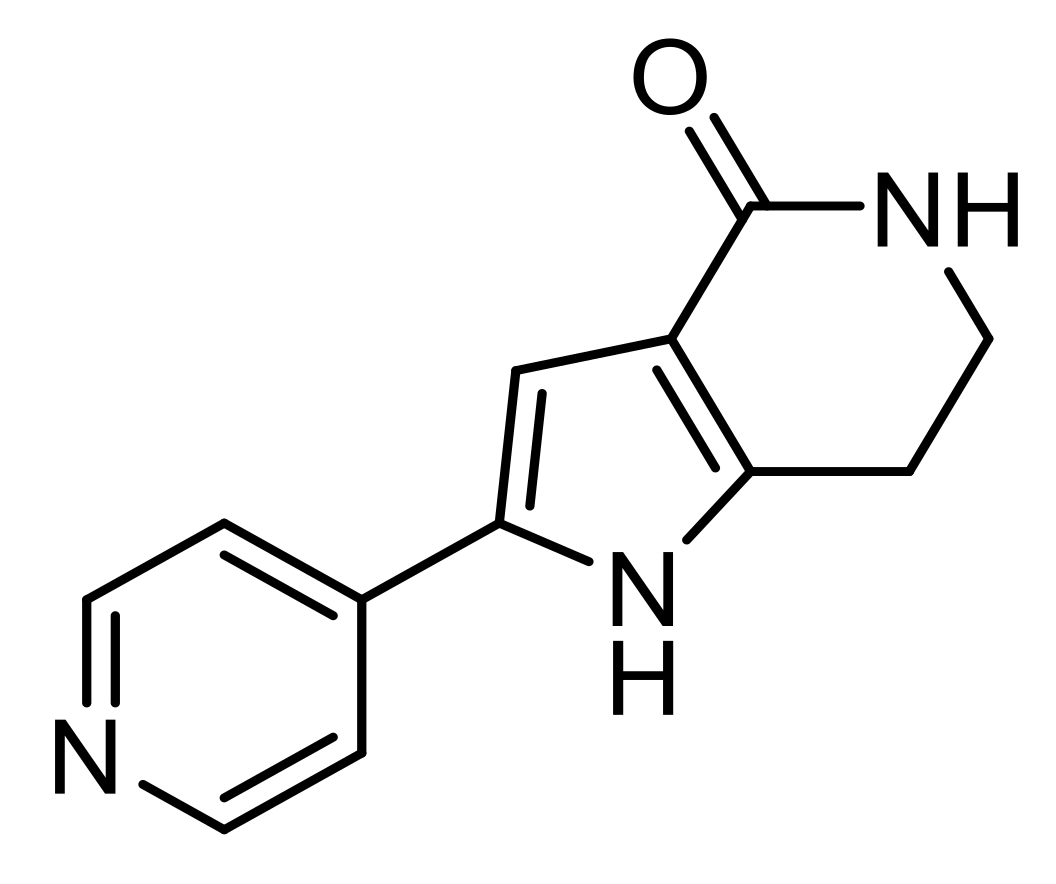
PHA-767491
3.5. Multiple Sclerosis
3.5.1. Interferon-Beta-1a
3.5.2. Leukemia Inhibitory Factor
3.5.3. Proteolipid Protein (PLP139–151)
| Active Substance | Encapsulation Method | NP Properties | Model of Use | Neuroprotective Activities | Ref. |
|---|---|---|---|---|---|
| Alzheimer’s Disease | |||||
| Rosmarinic acid | Emulsion/solvent diffusion | ø: 60 to 80 nm PdI: 0.047 ζ potential: 2 to 8 mV E.E.: 25 to 40% | In vitro: HBMECs, Has and Aβ-insulted SK-N-MC cells | Eliminate peroxynitrite anions Reduce inflammatory responses Disrupt the integrity of HBMEC cytoskeletal microfibrils Decrease the structural density of TJ Increase the permeability and reduce the TEER by targeting HBMECs with the use of 83-14 Mab | [89] |
| Curcumin Curcuma longa | Single emulsion/solvent evaporation | ø: 150 to 200 nm ζ Potential: −30 to −20 mV | In vitro: GI-1 glioma cells | Inhibit Aβ fibril formation Reduce brain amyloid level and plaque burden Free-radical scavenging activity | [90] |
| Rhynchophylline Uncaria rhynchophylla | Nanoprecipitation | ø: 145.2 nm PdI: 0.133 D.L.: 10.3% E.E.: 60% | In vitro: bEnd.3 and PC12 cells In vivo: healthy C57BL/6 mice | Inhibit soluble Aβ-induced hyperexcitability of hippocampal neurons Reduce cell apoptosis significantly | [58] |
| Quecertin | Double emulsion/solvent evaporation | ø < 145 nm | In vitro: human neuronal cells (SH-SY5Y) In vivo: BALB/c mice | Inhibit AChE and secretase enzymes Disrupt amyloid aggregates Prevent and inhibit Aβ1-42 fibrils formation Dissolve Aβ1-42 aggregates | [91] |
| iA β5 | Nanoprecipitation | ø: 153 to 166 nm PdI: 0.090 to 0.100 ζ potential: −10.1 to −13 mV E.E.: 63 ± 9% | Porcine brain capillary endothelial cells | N/A | [92] |
| Phytol | Emulsion/solvent evaporation | ø: 177.4 ± 5.9 nm ζ potential: −32.8 ± 2.2 mV D.L.: 56% E.E.: 92% | In vitro: Neuro2a cells In vivo: healthy male Wistar rats, rats injected with scopolamine | Anti-cholinesterase and anti-oxidative Disrupt amyloid aggregates Attenuate the impairment of learning and memory induced by scopolamine in rat brain Prevent oxidation of proteins and lipids against scopolamine-induced oxidative damage | [93,94] |
| Thymoquinone | Single emulsion/solvent evaporation | ø: 226.3 ± 4.6 nm PdI: 0.143 ζ potential: −45.7 ± 2.6 mV E.E.: 69.5 ± 3.0% | In vivo: STZ-induced Alzheimer model mice | Act on neurosis, ROS formation, free radical scavenging capacity, etc. Prevent Aβ aggregates and hyperphosphorylated τ-protein tangles | [53] |
| Nigella sativa oil and Plasmid DNA | Modified solvent diffusion | ø: 600 to 700 nm PdI: 0.229 ζ potential: + 35.6 mV D.L.: 13.0% E.E.: 73.8% | In vitro: murine neuroblastoma (N2a) cells | Inhibit radicals O2 Promote neurite outgrowth and neuroregeneration | [95] |
| Anthocyanins Natural pigments | Emulsion/solvent evaporation | ø: 165 nm PdI: 0.400 ζ potential: −12 mV E.E.: 60% | In vitro: human neuronal cells (SH-SY5Y) | Inhibit Aβ1–42-induced ROS generation Anti-oxidant | [96] |
| Huperzine A | Emulsion/solvent evaporation | ø: 78.1 to 153.2 nm PdI: 0.182 to 0.229 ζ potential: −21.2 to +35.6 mV D.L.: 5 to 25% E.E.: 83.2 to 73.8% | In vitro: 16HBE and SH-SY5Y cells In vivo: KM mice | Inhibit acetylcholinesterase Disrupt amyloid aggregates | [55] |
| Selegiline Methamphetamine derivative | Emulsion/solvent evaporation | ø: 217 to 246 nm PdI: 0.295 to 0.331 ζ potential: 34.7 to 38.0 mV E.E.: 3.3 to 11.7% | In vitro: fibrils (fAβ1-40 and fAβ1-42) | Inhibit MAO-B and Aβ fibrils formation Increase the destabilizing effects of the drug by increasing selegiline concentration and incubation time | [97] |
| Parkinson’s Disease | |||||
| Schisantherin A | Flash nanoprecipitation | ø: 70.6 ± 2.2 nm PdI: 0.104 ± 0.012 ζ potential: −24.7 ± 3.5 mV D.L.: 28.0 ± 0.8% E.E.: 91.1 ± 2.6% | In vitro: SH-SY5Y and MDCK cells In vivo: larval zebrafish, MPTP-induced zebrafish and male Sprague-Dawley rats | Reduce tyrosine hydroxylase TH+ DA neuronal death Prevent MPTP-induced decrease in TH+ region Reverse locomotor deficiency Stronger neuroprotective effects in MPP+ -induced SH-SY5Y cell injury model | [98] |
| Puearin | Antisolvent precipitation | ø: 88.4 ± 1.7 nm PdI: 0.047 ± 0.007 ζ potential: −18.9 ± 2.8 mV D.L.: 43.0 ± 1.6% E.E.: 89.5 ± 1.7% | In vitro: MDCK and SH-SY5Y cells In vivo: zebrafish and their embryos, male Sprague−Dawley (SD) rats, male C57BL/6 mice (with MPTP group) | Reduce tyrosine hydroxylase TH+ DA neuronal death Reduce behavioral deficits and associated mobility impairments in MPTP-mediated neurotoxicity in mice Reduce TH+ neuron loss and associated neurotoxicity | [63] |
| Levodopa | Double emulsion/solvent evaporation | ø: 329.0 to 383.7 nm PdI: 0.384 to 0.426 ζ potential: −4.5 to −20.8 mV E.E.: 50.5 to 73.0% | In vitro: PC12 cells In vivo: healthy CD57/BL6 mice (with MPTP group) | Improve locomotor activity over time Improve brain delivery by intranasal route Enhance dopamine concentration in the brain compared to the blood | [99] |
| Rasagiline | Double emulsion/solvent evaporation | ø: 221.7 ± 5.7 nm PdI: 0.388 ± 0.860 ζ potential: −36.1 ± 4.4 mV E.E.: 29.2 ± 1.8% | In vivo: healthy and Parkinson Wistar rats | Inhibit MAO-B enzyme Prevent neuronal damage caused by oxidative stress | [67] |
| Huntington’s Disease | |||||
| Peptides QBP1, NT17, and PGQ9P2 | Nanoprecipitation | ø: 158 to 180 nm PdI: 0.031 to 0.066 ζ potential: −23.3 to −27.5 mV | In vitro: MDCK, Neuro 2A and PC12 cell model of HD In vivo: healthy mice, Drosophila, larvae, and adult fly model of HD | Inhibit mHtt aggregation Restore motor activity | [74] |
| Cholesterol | Nanoprecipitation | ø: 200 to 300 nm PdI: 0.090 to 0.300 ζ potential: −8 to −12 mV D.L.: 0.7 to 2.5% E.E.: 48 to 68% | In vitro: neural stem (NS) cell model of HD In vivo: genotyping of R6/2 mouse | Restore synaptic alterations and delaying cognitive | [77] |
| Amyotrophic Lateral Sclerosis | |||||
| PHA-767491 | Nanoprecipitation | ø: 141 to 155 nm PdI ≤ 0.150 D.L.: 24% E.E.: 12 to 18% | In vitro: SH-SY5Y cell model of ALS | Reduce TDP-43 phosphorylation | [81] |
| Multiple Sclerosis | |||||
| Interferon-β-1a | Double emulsion/solvent evaporation | ø: 150 to 165 nm PdI: 0.081 to 0.093 ζ potential: 17.7 to 18.8 mV D.L.: 0.7 to 2.5% E.E.: 95.9% | In vitro: human blood plasma, primary hepatocytes from male Wistar rats In vivo: Wistar male rats | N/A | [85] |
| LIF | Double emulsion/solvent evaporation | ø: 126 ± 50 nm | In vitro: oligodendrocyte precursor cells from neonatal Sprague–Dawley rats | Induce maturation of OPCs to myelin-competent oligodendrocytes within 3 days Promote high-quality myelin repair | [88] |
| Proteolipid Protein | Double emulsion/solvent evaporation | ø: 195.1 ± 10.1 nm PdI: 0.220 ζ potential: −20 mV D.L.: 20% E.E.: 40% | N/A | N/A | [100] |
4. New Candidates for PLGA NPs Vectorization
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Scott, A.M. Global Burden of Neuropsychiatric Disorders. Alzheimer’s Dis. Int. World’s Alzheimer’s Rep. 2017, 2015, 10. [Google Scholar]
- Feigin, V.L.; Krishnamurthi, R.V.; Theadom, A.M.; Abajobir, A.A.; Mishra, S.R.; Ahmed, M.B.; Abate, K.H.; Mengistie, M.A.; Wakayo, T.; Abd-Allah, F.; et al. Global, regional, and national burden of neurological disorders during 1990–2015: A systematic analysis for the Global Burden of Disease Study 2015. Lancet Neurol. 2017, 16, 877–897. [Google Scholar] [CrossRef]
- Furuse, M. Molecular Basis of the Core Structure of Tight Junctions. Cold Spring Harb. Perspect. Biol. 2010, 2, a002907. [Google Scholar] [CrossRef]
- Pardridge, W.M. Drug transport across the blood-brain barrier. J. Cereb. Blood Flow Metab. 2012, 32, 1959–1972. [Google Scholar] [CrossRef] [PubMed]
- Nichols, E.; Szoeke, C.E.I.; Vollset, S.E.; Abbasi, N.; Abd-Allah, F.; Abdela, J.; Aichour, M.T.E.; Akinyemi, R.O.; Alahdab, F.; Asgedom, S.W.; et al. Global, regional, and national burden of Alzheimer’s disease and other dementias, 1990–2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol. 2019, 18, 88–106. [Google Scholar] [CrossRef]
- Tysnes, O.-B.; Storstein, A. Epidemiology of Parkinson’s disease. J. Neural Transm. 2017, 124, 901–905. [Google Scholar] [CrossRef] [PubMed]
- Baig, S.S.; Strong, M.; Quarrell, O.W. The global prevalence of Huntington’s disease: A systematic review and discussion. Neurodegener. Dis. Manag. 2016, 6, 331–343. [Google Scholar] [CrossRef]
- Xu, L.; Liu, T.; Liu, L.; Yao, X.; Chen, L.; Fan, D.; Zhan, S.; Wang, S. Global variation in prevalence and incidence of amyotrophic lateral sclerosis: A systematic review and meta-analysis. J. Neurol. 2020, 267, 944–953. [Google Scholar] [CrossRef] [PubMed]
- Walton, C.; King, R.; Rechtman, L.; Kaye, W.; Leray, E.; Marrie, R.A.; Robertson, N.; La Rocca, N.; Uitdehaag, B.; van der Mei, I.; et al. Rising prevalence of multiple sclerosis worldwide: Insights from the Atlas of MS, third edition. Mult. Scler. J. 2020, 26, 1816–1821. [Google Scholar] [CrossRef] [PubMed]
- Naqvi, S.; Panghal, A.; Flora, S.J.S. Nanotechnology: A Promising Approach for Delivery of Neuroprotective Drugs. Front. Neurosci. 2020, 14, 1–26. [Google Scholar] [CrossRef]
- Brambilla, D.; Le Droumaguet, B.; Nicolas, J.; Hashemi, S.H.; Wu, L.P.; Moghimi, S.M.; Couvreur, P.; Andrieux, K. Nanotechnologies for Alzheimer’s disease: Diagnosis, therapy, and safety issues. Nanomed. Nanotechnol. Biol. Med. 2011, 7, 521–540. [Google Scholar] [CrossRef]
- Leyva-Gómez, G.; Cortés, H.; Magaña, J.J.; Leyva-García, N.; Quintanar-Guerrero, D.; Florán, B. Nanoparticle technology for treatment of Parkinson’s disease: The role of surface phenomena in reaching the brain. Drug Discov. Today 2015, 20, 824–837. [Google Scholar] [CrossRef] [PubMed]
- Bourdenx, M.; Daniel, J.; Genin, E.; Soria, F.N.; Blanchard-Desce, M.; Bezard, E.; Dehay, B. Nanoparticles restore lysosomal acidification defects: Implications for Parkinson and other lysosomal-related diseases. Autophagy 2016, 12, 472–483. [Google Scholar] [CrossRef] [PubMed]
- Essa, D.; Kondiah, P.P.D.; Choonara, Y.E.; Pillay, V. The Design of Poly(lactide-co-glycolide) Nanocarriers for Medical Applications. Front. Bioeng. Biotechnol. 2020, 8, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Operti, M.C.; Bernhardt, A.; Grimm, S.; Engel, A.; Figdor, C.G.; Tagit, O. PLGA-based nanomedicines manufacturing: Technologies overview and challenges in industrial scale-up. Int. J. Pharm. 2021, 605, 120807. [Google Scholar] [CrossRef]
- Cano, A.; Sánchez-López, E.; Ettcheto, M.; López-Machado, A.; Espina, M.; Souto, E.B.; Galindo, R.; Camins, A.; García, M.L.; Turowski, P. Current advances in the development of novel polymeric nanoparticles for the treatment of neurodegenerative diseases. Nanomedicine 2020, 15, 1239–1261. [Google Scholar] [CrossRef]
- Makadia, H.K.; Siegel, S.J. Poly Lactic-co-Glycolic Acid (PLGA) as biodegradable controlled drug delivery carrier. Polymers 2011, 3, 1377–1397. [Google Scholar] [CrossRef]
- Anderson, J.M.; Shive, M.S. Biodegradation and biocompatibility of PLA and PLGA microspheres. Adv. Drug Deliv. Rev. 1997, 28, 5–24. [Google Scholar] [CrossRef]
- Danhier, F.; Ansorena, E.; Silva, J.M.; Coco, R.; Le Breton, A.; Préat, V. PLGA-based nanoparticles: An overview of biomedical applications. J. Control. Release 2012, 161, 505–522. [Google Scholar] [CrossRef]
- Zeng, J.; Martin, A.; Han, X.; Shirihai, O.S.; Grinstaff, M.W. Biodegradable plga nanoparticles restore lysosomal acidity and protect neural pc-12 cells against mitochondrial toxicity. Ind. Eng. Chem. Res. 2019, 58, 13910–13917. [Google Scholar] [CrossRef]
- Zhi, K.; Raji, B.; Nookala, A.R.; Khan, M.M.; Nguyen, X.H.; Sakshi, S.; Pourmotabbed, T.; Yallapu, M.M.; Kochat, H.; Tadrous, E.; et al. Plga nanoparticle-based formulations to cross the blood-brain barrier for drug delivery: From r&d to cgmp. Pharmaceutics 2021, 13, 500. [Google Scholar]
- McCall, R.L.; Sirianni, R.W. PLGA nanoparticles formed by single- or double-emulsion with vitamin E-TPGS. J. Vis. Exp. 2013, 82, 51015. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.; Zhang, C. Tuning the Size of Poly(lactic-co-glycolic Acid) (PLGA) Nanoparticles Fabricated by Nanoprecipitation. Biotechnol. J. 2018, 13, 1700203. [Google Scholar] [CrossRef] [PubMed]
- Ryu, S.; Park, S.; Lee, H.Y.; Lee, H.; Cho, C.W.; Baek, J.S. Biodegradable Nanoparticles-Loaded PLGA Microcapsule for the Enhanced Encapsulation Efficiency and Controlled Release of Hydrophilic Drug. Int. J. Mol. Sci. 2021, 22, 2792. [Google Scholar] [CrossRef]
- Moghimi, S.M.; Moghimi, S.M.; Hunter, A.C.; Murray, J.C. Long-circulating and target-specific nanoparticles: Theory to practice. Pharmacol. Rev. 2001, 53, 283–318. [Google Scholar]
- Hoshyar, N.; Gray, S.; Han, H.; Bao, G. The effect of nanoparticle size on in vivo pharmacokinetics and cellular interaction. Nanomedicine 2016, 11, 673–692. [Google Scholar] [CrossRef] [PubMed]
- Danaei, M.; Dehghankhold, M.; Ataei, S.; Hasanzadeh Davarani, F.; Javanmard, R.; Dokhani, A.; Khorasani, S.; Mozafari, M. Impact of Particle Size and Polydispersity Index on the Clinical Applications of Lipidic Nanocarrier Systems. Pharmaceutics 2018, 10, 57. [Google Scholar] [CrossRef]
- Kumar, A.; Dixit, C.K. Methods for characterization of nanoparticles. In Advances in Nanomedicine for the Delivery of Therapeutic Nucleic Acids; Elsevier: Amsterdam, The Netherlands, 2017; pp. 43–58. ISBN 9780081005637. [Google Scholar]
- Fröhlich, E. The role of surface charge in cellular uptake and cytotoxicity of medical nanoparticles. Int. J. Nanomed. 2012, 7, 5577. [Google Scholar] [CrossRef] [PubMed]
- Mulvihill, J.J.; Cunnane, E.M.; Ross, A.M.; Duskey, J.T.; Tosi, G.; Grabrucker, A.M. Drug delivery across the blood-brain barrier: Recent advances in the use of nanocarriers. Nanomedicine 2020, 15, 205–214. [Google Scholar] [CrossRef]
- Wünsch, A.; Mulac, D.; Langer, K. Lecithin coating as universal stabilization and functionalization strategy for nanosized drug carriers to overcome the blood–brain barrier. Int. J. Pharm. 2021, 593, 120146. [Google Scholar] [CrossRef]
- Yoo, J.; Park, C.; Yi, G.; Lee, D.; Koo, H. Active targeting strategies using biological ligands for nanoparticle drug delivery systems. Cancers 2019, 11, 640. [Google Scholar] [CrossRef]
- Ray, S.; Sinha, P.; Laha, B.; Maiti, S.; Bhattacharyya, U.K.; Nayak, A.K. Polysorbate 80 coated crosslinked chitosan nanoparticles of ropinirole hydrochloride for brain targeting. J. Drug Deliv. Sci. Technol. 2018, 48, 21–29. [Google Scholar] [CrossRef]
- Joshi, A.S.; Gahane, A.; Thakur, A.K. Deciphering the mechanism and structural features of polysorbate 80 during adsorption on PLGA nanoparticles by attenuated total reflectance-Fourier transform infrared spectroscopy. RSC Adv. 2016, 6, 108545–108557. [Google Scholar] [CrossRef]
- Schwartzberg, L.S.; Navari, R.M. Safety of Polysorbate 80 in the Oncology Setting. Adv. Ther. 2018, 35, 754–767. [Google Scholar] [CrossRef] [PubMed]
- Baskin, J.; Jeon, J.E.; Lewis, S.J.G. Nanoparticles for drug delivery in Parkinson’s disease. J. Neurol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Gulati, N.M.; Stewart, P.L.; Steinmetz, N.F. Bio inspired shielding strategies for nanoparticle drug delivery applications Graphical Abstract. Mol. Pharm 2018, 15, 2900–2909. [Google Scholar] [CrossRef] [PubMed]
- Suk, J.S.; Xu, Q.; Kim, N.; Hanes, J.; Ensign, L.M. PEGylation as a strategy for improving nanoparticle-based drug and gene delivery. Adv. Drug Deliv. Rev. 2016, 99, 28–51. [Google Scholar] [CrossRef]
- Hu, K.; Shi, Y.; Jiang, W.; Han, J.; Huang, S.; Jiang, X. Lactoferrin conjugated PEG-PLGA nanoparticles for brain delivery: Preparation, characterization and efficacy in Parkinsons disease. Int. J. Pharm. 2011, 415, 273–283. [Google Scholar] [CrossRef]
- Torres-Ortega, P.V.; Saludas, L.; Hanafy, A.S.; Garbayo, E.; Blanco-Prieto, M.J. Micro- and nanotechnology approaches to improve Parkinson’s disease therapy. J. Control. Release 2019, 295, 201–213. [Google Scholar] [CrossRef] [PubMed]
- Appelboom, G.; Detappe, A.; LoPresti, M.; Kunjachan, S.; Mitrasinovic, S.; Goldman, S.; Chang, S.D.; Tillement, O. Stereotactic modulation of blood-brain barrier permeability to enhance drug delivery. Neuro Oncol. 2016, 18, 1601–1609. [Google Scholar] [CrossRef]
- Jaiswal, M.; Dudhe, R.; Sharma, P.K. Nanoemulsion: An advanced mode of drug delivery system. 3 Biotech 2015, 5, 123–127. [Google Scholar] [CrossRef]
- Gosselet, F.; Loiola, R.A.; Roig, A.; Rosell, A.; Culot, M. Central nervous system delivery of molecules across the blood-brain barrier. Neurochem. Int. 2021, 144, 104952. [Google Scholar] [CrossRef]
- Sharma, D.; Sharma, R.K.; Sharma, N.; Gabrani, R.; Sharma, S.K.; Ali, J.; Dang, S. Nose-To-Brain Delivery of PLGA-Diazepam Nanoparticles. AAPS Pharmscitech 2015, 16, 1108–1121. [Google Scholar] [CrossRef] [PubMed]
- Masserini, M. Nanoparticles for Brain Drug Delivery. ISRN Biochem. 2013, 2013, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Tarawneh, R.; Galvin, J.E. Potential Future Neuroprotective Therapies for Neurodegenerative Disorders and Stroke. Clin. Geriatr. Med. 2010, 26, 125–147. [Google Scholar] [CrossRef] [PubMed]
- Tapias, V. Editorial: Mitochondrial Dysfunction and Neurodegeneration. Front. Neurosci. 2019, 13, 1372. [Google Scholar] [CrossRef] [PubMed]
- Murphy, M.P.; Levine, H. Alzheimer’s disease and the amyloid-β peptide. J. Alzheimer’s Dis. 2010, 19, 311–323. [Google Scholar] [CrossRef] [PubMed]
- Iqbal, K.; Liu, F.; Gong, C.-X.; Grundke-Iqbal, I. Tau in Alzheimer Disease and Related Tauopathies. Curr. Alzheimer Res. 2010, 7, 656–664. [Google Scholar] [CrossRef] [PubMed]
- Gallagher, D.J.; Mhaolaín, A.N.; Sperling, R.A.; Lawlor, B.A. Alzheimer’s Disease. In Neurodegenerative Disorders; Springer: London, UK, 2011; pp. 43–64. [Google Scholar]
- Armstrong, R.A. Risk factors for Alzheimer’s disease. Folia Neuropathol. 2019, 57, 87–105. [Google Scholar] [CrossRef]
- Huang, W.-J.; Zhang, X.; Chen, W.-W. Role of oxidative stress in Alzheimer’s disease. Biomed. Rep. 2016, 4, 519–522. [Google Scholar] [CrossRef]
- Yusuf, M.; Khan, M.; Alrobaian, M.M.; Alghamdi, S.A.; Warsi, M.H.; Sultana, S.; Khan, R.A. Brain targeted Polysorbate-80 coated PLGA thymoquinone nanoparticles for the treatment of Alzheimer’s disease, with biomechanistic insights. J. Drug Deliv. Sci. Technol. 2021, 61, 102214. [Google Scholar] [CrossRef]
- Elibol, B.; Beker, M.; Terzioglu-Usak, S.; Dalli, T.; Kilic, U. Thymoquinone administration ameliorates Alzheimer’s disease-like phenotype by promoting cell survival in the hippocampus of amyloid beta1–42 infused rat model. Phytomedicine 2020, 79, 153324. [Google Scholar] [CrossRef]
- Meng, Q.; Wang, A.; Hua, H.; Jiang, Y.; Wang, Y.; Mu, H.; Wu, Z.; Sun, K. Intranasal delivery of Huperzine A to the brain using lactoferrin-conjugated N-trimethylated chitosan surface-modified PLGA nanoparticles for treatment of Alzheimer’s disease. Int. J. Nanomed. 2018, 13, 705–718. [Google Scholar] [CrossRef] [PubMed]
- Ma, T.; Gong, K.; Yan, Y.; Zhang, L.; Tang, P.; Zhang, X.; Gong, Y. Huperzine A promotes hippocampal neurogenesis in vitro and in vivo. Brain Res. 2013, 1506, 35–43. [Google Scholar] [CrossRef]
- Shao, H.; Mi, Z.; Ji, W.G.; Zhang, C.H.; Zhang, T.; Ren, S.C.; Zhu, Z.R. Rhynchophylline Protects Against the Amyloid β-Induced Increase of Spontaneous Discharges in the Hippocampal CA1 Region of Rats. Neurochem. Res. 2015, 40, 2365–2373. [Google Scholar] [CrossRef] [PubMed]
- Xu, R.; Wang, J.; Xu, J.; Song, X.; Huang, H.; Feng, Y.; Fu, C. Rhynchophylline loaded-mPEG-PLGA nanoparticles coated with tween-80 for preliminary study in Alzheimer’s disease. Int. J. Nanomed. 2020, 15, 1149–1160. [Google Scholar] [CrossRef] [PubMed]
- Walsh, R.A.; Lynch, T.; Fahn, S. Parkinson’s Disease. In Neurodegenerative Disorders; Springer: London, UK, 2011; pp. 77–114. [Google Scholar]
- Sa, F.; Zhang, L.Q.; Chong, C.M.; Guo, B.J.; Li, S.; Zhang, Z.J.; Zheng, Y.; Hoi, P.M.; Lee, S.M.Y. Discovery of novel anti-parkinsonian effect of schisantherin A in in vitro and in vivo. Neurosci. Lett. 2015, 593, 7–12. [Google Scholar] [CrossRef]
- Huang, J.L.; Jing, X.; Tian, X.; Qin, M.C.; Xu, Z.H.; Wu, D.P.; Zhong, Z.G. Neuroprotective properties of panax notoginseng saponins via preventing oxidative stress injury in SAMP8 mice. Evid. Based Complement. Altern. Med. 2017, 2017. [Google Scholar] [CrossRef]
- Zhang, X.; Xiong, J.; Liu, S.; Wang, L.; Huang, J.; Liu, L.; Yang, J.; Zhang, G.; Guo, K.; Zhang, Z.; et al. Puerarin protects dopaminergic neurons in Parkinson’s disease models. Neuroscience 2014, 280, 88–98. [Google Scholar] [CrossRef]
- Chen, T.; Liu, W.; Xiong, S.; Li, D.; Fang, S.; Wu, Z.; Wang, Q.; Chen, X. Nanoparticles mediating the sustained puerarin release facilitate improved brain delivery to treat parkinson’s disease. ACS Appl. Mater. Interfaces 2019, 11, 45276–45289. [Google Scholar] [CrossRef]
- Fernandez, H. Malaty Role of rasagiline in treating Parkinson’s disease: Effect on disease progression. Clin. Risk Manag. 2009, 5, 413. [Google Scholar] [CrossRef][Green Version]
- Hattori, N.; Takeda, A.; Takeda, S.; Nishimura, A.; Kitagawa, T.; Mochizuki, H.; Nagai, M.; Takahashi, R. Rasagiline monotherapy in early Parkinson’s disease: A phase 3, randomized study in Japan. Park. Relat. Disord. 2019, 60, 146–152. [Google Scholar] [CrossRef]
- Hauser, R.A.; Abler, V.; Eyal, E.; Eliaz, R.E. Efficacy of rasagiline in early Parkinson’s disease: A meta-analysis of data from the TEMPO and ADAGIO studies. Int. J. Neurosci. 2016, 126, 942–946. [Google Scholar] [CrossRef]
- Bali, N.R.; Salve, P.S. Impact of rasagiline nanoparticles on brain targeting efficiency via gellan gum based transdermal patch: A nanotheranostic perspective for Parkinsonism. Int. J. Biol. Macromol. 2020, 164, 1006–1024. [Google Scholar] [CrossRef]
- Myers, R.H. Huntington’s Disease Genetics. NeuroRx 2004, 1, 255–262. [Google Scholar] [CrossRef] [PubMed]
- Pender, N.P.; Koroshetz, W.J. Huntington’s Disease. In Neurodegenerative Disorders; Springer: London, UK, 2011; pp. 167–179. [Google Scholar]
- Ferrante, R.J.; Kowall, N.W.; Beal, M.F.; Richardson, E.P.; Bird, E.D.; Martin, J.B. Selective sparing of a class of striatal neurons in Huntington’s disease. Science 1985, 230, 561–563. [Google Scholar] [CrossRef]
- Rubinsztein, D.C. Lessons from animal models of Huntington’s disease. Trends Genet. 2002, 18, 202–209. [Google Scholar] [CrossRef]
- Sung, V.W.; Iyer, R.; Schilling, T.; Buzinec, P.N. Tetrabenazine Use in Patients With Huntington’s Disease (HD) Chorea (P2.009). Neurology 2017, 88, 545–551. [Google Scholar]
- Burra, G.; Thakur, A.K. Inhibition of polyglutamine aggregation by SIMILAR huntingtin N-terminal sequences: Prospective molecules for preclinical evaluation in Huntington’s disease. Biopolymers 2017, 108, e23021. [Google Scholar] [CrossRef] [PubMed]
- Joshi, A.S.; Singh, V.; Gahane, A.; Thakur, A.K. Biodegradable Nanoparticles Containing Mechanism Based Peptide Inhibitors Reduce Polyglutamine Aggregation in Cell Models and Alleviate Motor Symptoms in a Drosophila Model of Huntington’s Disease. ACS Chem. Neurosci. 2019, 10, 1603–1614. [Google Scholar] [CrossRef] [PubMed]
- Leoni, V.; Caccia, C. The impairment of cholesterol metabolism in Huntington disease. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 2015, 1851, 1095–1105. [Google Scholar] [CrossRef]
- Block, R.C.; Dorsey, E.R.; Beck, C.A.; Brenna, J.T.; Shoulson, I. Altered cholesterol and fatty acid metabolism in Huntington disease. J. Clin. Lipidol. 2010, 4, 17–23. [Google Scholar] [CrossRef]
- Valenza, M.; Chen, J.Y.; Di Paolo, E.; Ruozi, B.; Belletti, D.; Ferrari Bardile, C.; Leoni, V.; Caccia, C.; Brilli, E.; Di Donato, S.; et al. Cholesterol-loaded nanoparticles ameliorate synaptic and cognitive function in H untington’s disease mice. EMBO Mol. Med. 2015, 7, 1547–1564. [Google Scholar] [CrossRef] [PubMed]
- Hardiman, O. Amyotrophic Lateral Sclerosis. In Neurodegenerative Disorders; Springer: London, UK, 2011; Volume 59, pp. 143–166. [Google Scholar]
- Andrews, J.A.; Jackson, C.E.; Heiman-Patterson, T.D.; Bettica, P.; Brooks, B.R.; Pioro, E.P. Real-world evidence of riluzole effectiveness in treating amyotrophic lateral sclerosis. Amyotroph. Lateral Scler. Front. Degener. 2020, 21, 509–518. [Google Scholar] [CrossRef] [PubMed]
- Chung, Y.H.; Lin, C.W.; Huang, H.Y.; Chen, S.L.; Huang, H.J.; Sun, Y.C.; Lee, G.C.; Lee-Chen, G.J.; Chang, Y.C.; Hsieh-Li, H.M. Targeting Inflammation, PHA-767491 Shows a Broad Spectrum in Protein Aggregation Diseases. J. Mol. Neurosci. 2020, 70, 1140–1152. [Google Scholar] [CrossRef]
- Rojas-Prats, E.; Tosat-Bitrián, C.; Martínez-González, L.; Nozal, V.; Pérez, D.I.; Martínez, A. Increasing Brain Permeability of PHA-767491, a Cell Division Cycle 7 Kinase Inhibitor, with Biodegradable Polymeric Nanoparticles. Pharmaceutics 2021, 13, 180. [Google Scholar] [CrossRef] [PubMed]
- Dobson, R.; Giovannoni, G. Multiple sclerosis—A review. Eur. J. Neurol. 2019, 26, 27–40. [Google Scholar] [CrossRef]
- Chountoulesi, M.; Demetzos, C. Promising nanotechnology approaches in treatment of autoimmune diseases of central nervous system. Brain Sci. 2020, 10. [Google Scholar] [CrossRef] [PubMed]
- Clerico, M.; Contessa, G.; Durelli, L. Interferon-β 1a for the treatment of multiple sclerosis. Expert Opin. Biol. 2007, 7, 535–542. [Google Scholar] [CrossRef] [PubMed]
- Fodor-Kardos, A.; Kiss, Á.F.; Monostory, K.; Feczkó, T. Sustained: In vitro interferon-beta release and in vivo toxicity of PLGA and PEG-PLGA nanoparticles. RSC Adv. 2020, 10, 15893–15900. [Google Scholar] [CrossRef]
- Yue, X.; Wu, L.; Hu, W. The Regulation of Leukemia Inhibitory Factor. Cancer Cell Microenviron. 2015, 2, e877. [Google Scholar]
- Slaets, H.; Hendriks, J.J.A.; Stinissen, P.; Kilpatrick, T.J.; Hellings, N. Therapeutic potential of LIF in multiple sclerosis. Trends Mol. Med. 2010, 16, 493–500. [Google Scholar] [CrossRef]
- Rittchen, S.; Boyd, A.; Burns, A.; Park, J.; Fahmy, T.M.; Metcalfe, S.; Williams, A. Myelin repair invivo is increased by targeting oligodendrocyte precursor cells with nanoparticles encapsulating leukaemia inhibitory factor (LIF). Biomaterials 2015, 56, 78–85. [Google Scholar] [CrossRef]
- Kuo, Y.C.; Tsai, H.C. Rosmarinic acid- and curcumin-loaded polyacrylamide-cardiolipin-poly(lactide-co-glycolide) nanoparticles with conjugated 83–14 monoclonal antibody to protect β-amyloid-insulted neurons. Mater. Sci. Eng. C 2018, 91, 445–457. [Google Scholar] [CrossRef]
- Mathew, A.; Fukuda, T.; Nagaoka, Y.; Hasumura, T.; Morimoto, H.; Yoshida, Y.; Maekawa, T.; Venugopal, K.; Kumar, D.S. Curcumin loaded-PLGA nanoparticles conjugated with Tet-1 peptide for potential use in Alzheimer’s disease. PLoS ONE 2012, 7, e32616. [Google Scholar] [CrossRef] [PubMed]
- Sun, D.; Li, N.; Zhang, W.; Zhao, Z.; Mou, Z.; Huang, D.; Liu, J.; Wang, W. Design of PLGA-functionalized quercetin nanoparticles for potential use in Alzheimer’s disease. Colloids Surf. B Biointerfaces 2016, 148, 116–129. [Google Scholar] [CrossRef] [PubMed]
- Loureiro, J.A.; Gomes, B.; Fricker, G.; Coelho, M.A.N.; Rocha, S.; Pereira, M.C. Cellular uptake of PLGA nanoparticles targeted with anti-amyloid and anti-transferrin receptor antibodies for Alzheimer’s disease treatment. Colloids Surf. B Biointerfaces 2016, 145, 8–13. [Google Scholar] [CrossRef]
- Sathya, S.; Manogari, B.G.; Thamaraiselvi, K.; Vaidevi, S.; Ruckmani, K.; Devi, K.P. Phytol loaded PLGA nanoparticles ameliorate scopolamine-induced cognitive dysfunction by attenuating cholinesterase activity, oxidative stress and apoptosis in Wistar rat. Nutr. Neurosci. 2020, 8305, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Sathya, S.; Shanmuganathan, B.; Saranya, S.; Vaidevi, S.; Ruckmani, K.; Pandima Devi, K. Phytol-loaded PLGA nanoparticle as a modulator of Alzheimer’s toxic Aβ peptide aggregation and fibrillation associated with impaired neuronal cell function. Artif. CellsNanomed. Biotechnol. 2018, 46, 1719–1730. [Google Scholar] [CrossRef]
- Doolaanea, A.A.; Mansor, N.I.; Mohd Nor, N.H.; Mohamed, F. Co-encapsulation of Nigella sativa oil and plasmid DNA for enhanced gene therapy of Alzheimers disease. J. Microencapsul. 2016, 33, 114–126. [Google Scholar] [CrossRef]
- Amin, F.U.; Shah, S.A.; Badshah, H.; Khan, M.; Kim, M.O. Anthocyanins encapsulated by PLGA@PEG nanoparticles potentially improved its free radical scavenging capabilities via p38/JNK pathway against Aβ1-42-induced oxidative stress. J. Nanobiotechnol. 2017, 15, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Baysal, I.; Yabanoglu-Ciftci, S.; Tunc-Sarisozen, Y.; Ulubayram, K.; Ucar, G. Interaction of selegiline-loaded PLGA-b-PEG nanoparticles with beta-amyloid fibrils. J. Neural Transm. 2013, 120, 903–910. [Google Scholar] [CrossRef]
- Chen, T.; Li, C.; Li, Y.; Yi, X.; Wang, R.; Lee, S.M.Y.; Zheng, Y. Small-Sized mPEG-PLGA Nanoparticles of Schisantherin A with Sustained Release for Enhanced Brain Uptake and Anti-Parkinsonian Activity. Acs Appl. Mater. Interfaces 2017, 9, 9516–9527. [Google Scholar] [CrossRef] [PubMed]
- Arisoy, S.; Sayiner, O.; Comoglu, T.; Onal, D.; Atalay, O.; Pehlivanoglu, B. In vitro and in vivo evaluation of levodopa-loaded nanoparticles for nose to brain delivery. Pharm. Dev. Technol. 2020, 25, 735–747. [Google Scholar] [CrossRef] [PubMed]
- Lima, A.F.; Amado, I.R.; Pires, L.R. Poly(D,l-lactide-co-glycolide) (plga) nanoparticles loaded with proteolipid protein (plp)—exploring a new administration route. Polymer 2020, 12, 1–10. [Google Scholar]
- Mohd Sairazi, N.S.; Sirajudeen, K.N.S. Natural Products and Their Bioactive Compounds: Neuroprotective Potentials against Neurodegenerative Diseases. Evid. Based Complement. Altern. Med. 2020, 2020, 5–7. [Google Scholar] [CrossRef]
- Carrera, I.; Cacabelos, R. Current Drugs and Potential Future Neuroprotective Compounds for Parkinson’s Disease. Curr. Neuropharmacol. 2018, 17, 295–306. [Google Scholar] [CrossRef]
- Lee, Y.W.; Kim, D.H.; Jeon, S.J.; Park, S.J.; Kim, J.M.; Jung, J.M.; Lee, H.E.; Bae, S.G.; Oh, H.K.; Ho Son, K.H.; et al. Neuroprotective effects of salvianolic acid B on an Aβ25-35 peptide-induced mouse model of Alzheimer’s disease. Eur. J. Pharmacol. 2013, 704, 70–77. [Google Scholar] [CrossRef]
- Wang, Z.-Y.; Liu, J.-G.; Li, H.; Yang, H.-M. Pharmacological Effects of Active Components of Chinese Herbal Medicine in the Treatment of Alzheimer’s Disease: A Review. Am. J. Chin. Med. 2016, 44, 1525–1541. [Google Scholar] [CrossRef] [PubMed]
- Emanuele, E. Can Trehalose Prevent Neurodegeneration? Insights from Experimental Studies. Curr. Drug Targets 2014, 15, 551–557. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.; Barkhordarian, H.; Emadi, S.; Chan, B.P.; Sierks, M.R. Trehalose differentially inhibits aggregation and neurotoxicity of beta-amyloid 40 and 42. Neurobiol. Dis. 2005, 20, 74–81. [Google Scholar] [CrossRef]
- Sarkar, S.; Davies, J.E.; Huang, Z.; Tunnacliffe, A.; Rubinsztein, D.C. Trehalose, a novel mTOR-independent autophagy enhancer, accelerates the clearance of mutant huntingtin and α-synuclein. J. Biol. Chem. 2007, 282, 5641–5652. [Google Scholar] [CrossRef]
- Khalifeh, M.; Barreto, G.E.; Sahebkar, A. Trehalose as a promising therapeutic candidate for the treatment of Parkinson’s disease. Br. J. Pharmacol. 2019, 176, 1173–1189. [Google Scholar] [CrossRef]
- Tanaka, M.; Machida, Y.; Niu, S.; Ikeda, T.; Jana, N.R.; Doi, H.; Kurosawa, M.; Nekooki, M.; Nukina, N. Trehalose alleviates polyglutamine-mediated pathology in a mouse model of Huntington disease. Nat. Med. 2004, 10, 148–154. [Google Scholar] [CrossRef] [PubMed]
- Halbe, L.; Rami, A. Trehalase localization in the cerebral cortex, hippocampus and cerebellum of mouse brains. J. Adv. Res. 2019, 18, 71–79. [Google Scholar] [CrossRef]
- Zhang, X.; Chen, G.; Wen, L.; Yang, F.; Shao, A.L.; Li, X.; Long, W.; Mu, L. Novel multiple agents loaded PLGA nanoparticles for brain delivery via inner ear administration: In vitro and in vivo evaluation. Eur. J. Pharm. Sci. 2013, 48, 595–603. [Google Scholar] [CrossRef]
- Zeng, G.; Tang, T.; Wu, H.J.; You, W.H.; Luo, J.K.; Lin, Y.; Liang, Q.H.; Li, X.Q.; Huang, X.; Yang, Q.D. Salvianolic acid b protects SH-SY5Y neuroblastoma cells from1-methyl-4- phenylpyridinium-induced apoptosis. Biol. Pharm. Bull. 2010, 33, 1337–1342. [Google Scholar] [CrossRef] [PubMed]
- Jiang, P.; Li, C.; Xiang, Z.; Jiao, B. Tanshinone IIA reduces the risk of Alzheimer’s disease by inhibiting iNOS, MMP-2 and NF-κBp65 transcription and translation in the temporal lobes of rat models of Alzheimer’s disease. Mol. Med. Rep. 2014, 10, 689–694. [Google Scholar] [CrossRef]
- Ren, B.; Zhang, Y.X.; Zhou, H.X.; Sun, F.W.; Zhang, Z.F.; Wei, Z.F.; Zhang, C.Y.; Si, D.W. Tanshinone IIA prevents the loss of nigrostriatal dopaminergic neurons by inhibiting NADPH oxidase and iNOS in the MPTP model of Parkinson’s disease. J. Neurol. Sci. 2015, 348, 142–152. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Feng, Y.; Fu, Q.; Li, L. Panax notoginsenoside Rb1 ameliorates Alzheimer’s disease by upregulating brain-derived neurotrophic factor and downregulating Tau protein expression. Exp. Ther. Med. 2013, 6, 826–830. [Google Scholar] [CrossRef]
- Anselmo, A.C.; Mitragotri, S. Nanoparticles in the clinic. Bioeng. Transl. Med. 2016, 1, 10–29. [Google Scholar] [CrossRef] [PubMed]
- US FDA CDER. Current Good Manufacturing Practice (CGMP) Regulations; FDA: Silver Spring, MD, USA, 2018; p. 5.
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cunha, A.; Gaubert, A.; Latxague, L.; Dehay, B. PLGA-Based Nanoparticles for Neuroprotective Drug Delivery in Neurodegenerative Diseases. Pharmaceutics 2021, 13, 1042. https://doi.org/10.3390/pharmaceutics13071042
Cunha A, Gaubert A, Latxague L, Dehay B. PLGA-Based Nanoparticles for Neuroprotective Drug Delivery in Neurodegenerative Diseases. Pharmaceutics. 2021; 13(7):1042. https://doi.org/10.3390/pharmaceutics13071042
Chicago/Turabian StyleCunha, Anthony, Alexandra Gaubert, Laurent Latxague, and Benjamin Dehay. 2021. "PLGA-Based Nanoparticles for Neuroprotective Drug Delivery in Neurodegenerative Diseases" Pharmaceutics 13, no. 7: 1042. https://doi.org/10.3390/pharmaceutics13071042
APA StyleCunha, A., Gaubert, A., Latxague, L., & Dehay, B. (2021). PLGA-Based Nanoparticles for Neuroprotective Drug Delivery in Neurodegenerative Diseases. Pharmaceutics, 13(7), 1042. https://doi.org/10.3390/pharmaceutics13071042






