Model-Based Prediction to Evaluate Residence Time of Hyaluronic Acid Based Dermal Fillers
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals and Study Design
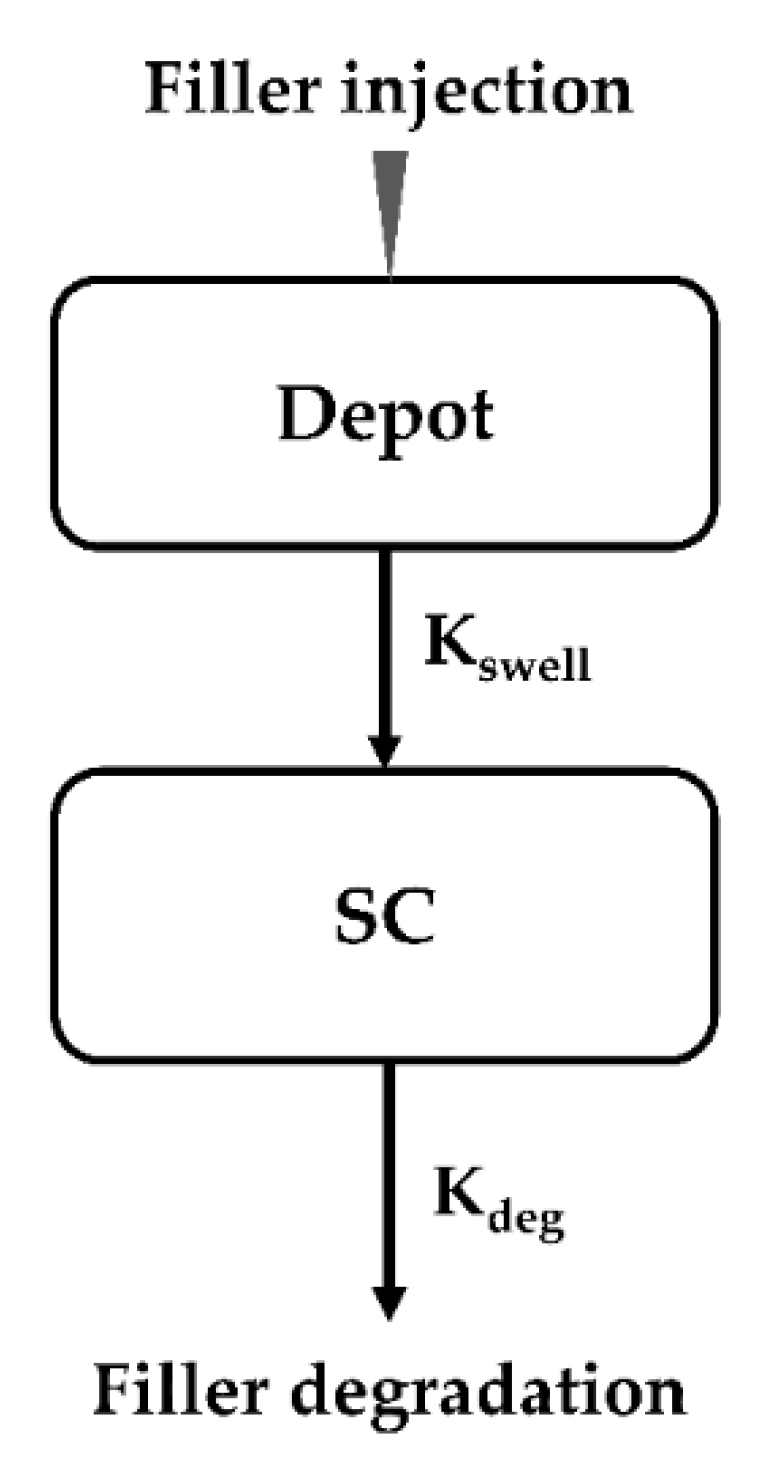
2.2. Establishment of Kinetic Model for Dermal Fillers
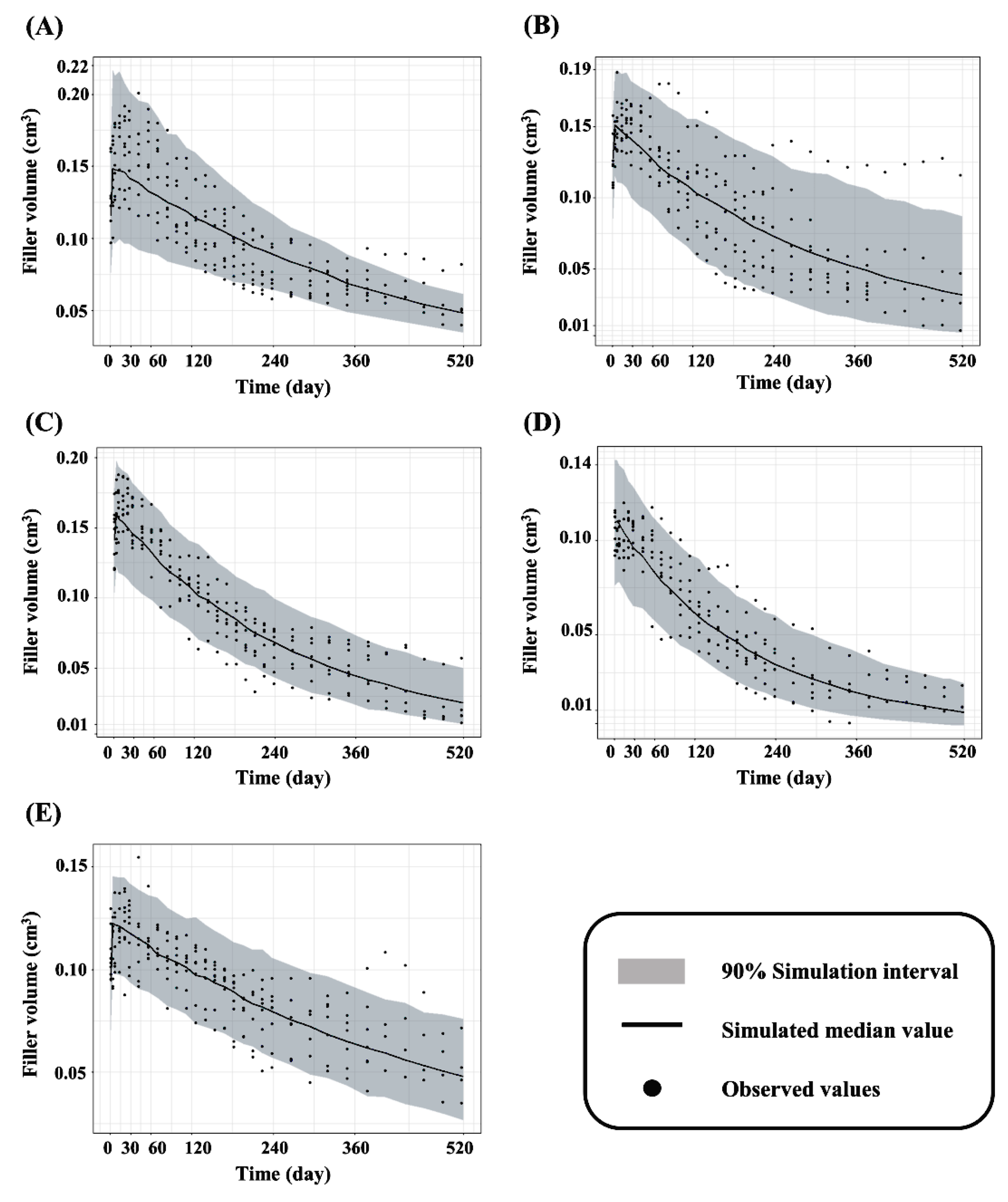
2.3. Model Diagnostics and Evaluation
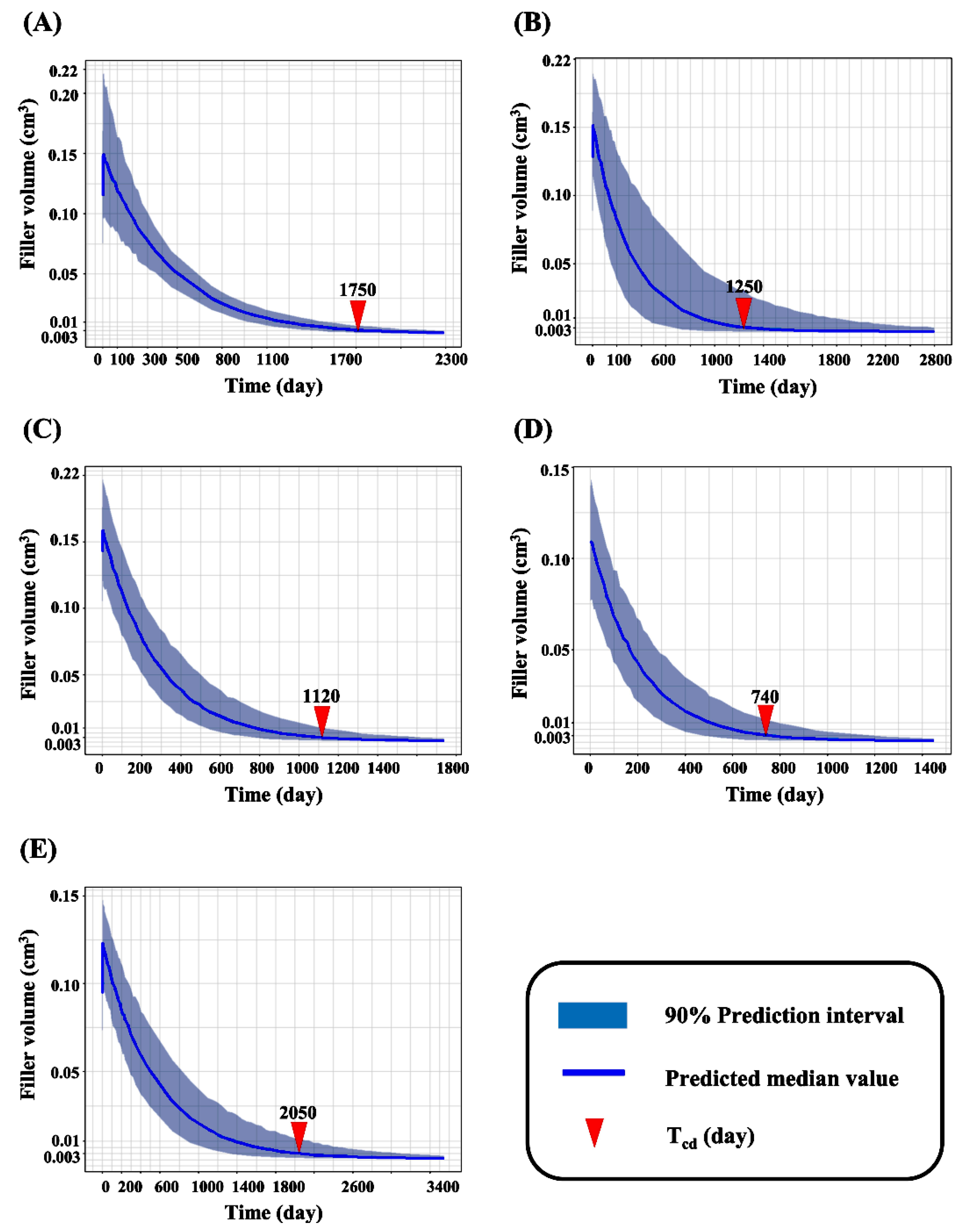
2.4. Simulation for Expected Time for Complete Decomposition
3. Results
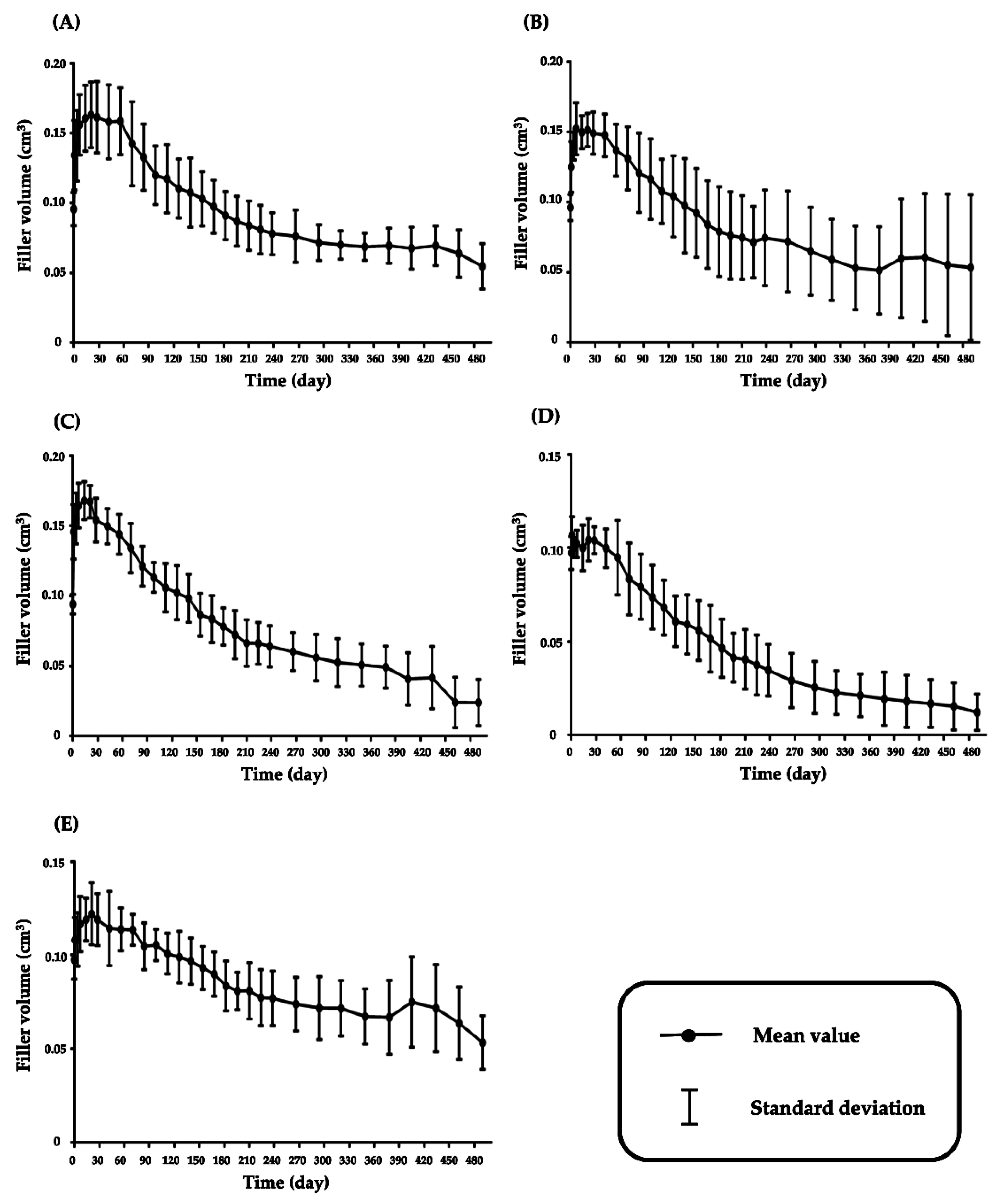
3.1. Volume Change of Fillers after Injection in Hairless Mice
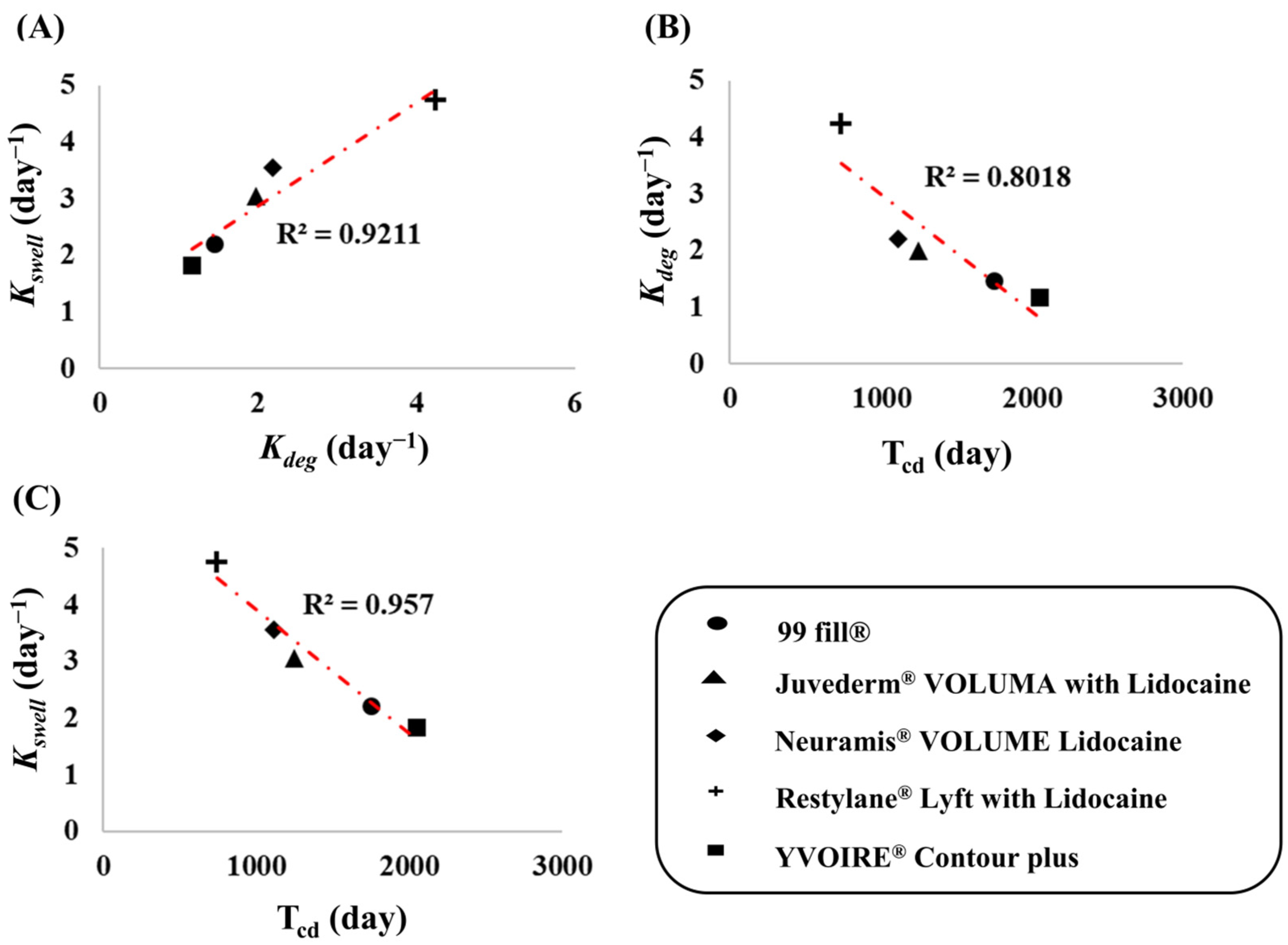
3.2. Kinetic Modeling for Swelling and Degradation
3.3. Simulation for Expected Time for Complete Decomposition of Dermal Fillers
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ballin, A.C.; Brandt, F.S.; Cazzaniga, A. Dermal fillers: An update. Am. J. Clin. Dermatol. 2015, 16, 271–283. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Carpintero, I.; Candelas, D.; Ruiz-Rodríguez, R. Dermal fillers: Types, indications, and complications. Actas Dermosifiliogr. 2010, 101, 381–393. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.-E.; Sykes, J.M. Hyaluronic acid fillers: History and overview. Facial Plast. Surg. 2011, 27, 523–528. [Google Scholar] [CrossRef] [PubMed]
- De Boulle, K.; Glogau, R.; Kono, T.; Nathan, M.; Tezel, A.; Roca-Martinez, J.-X.; Paliwal, S.; Stroumpoulis, D. A review of the metabolism of 1,4-butanediol diglycidyl ether-crosslinked hyaluronic acid dermal fillers. Dermatol. Surg. 2013, 39, 1758–1766. [Google Scholar] [CrossRef]
- Haney, B. Aesthetic Procedures: Nurse Practitioner’s Guide to Cosmetic Dermatology, 1st ed.; Springer: Cham, Switzerland, 2020; pp. 139–189. [Google Scholar] [CrossRef]
- Zerbinati, N.; Moucchi, R.; Galadari, H.; Maccario, C.; Maggi, M.; Rauso, R.; Passi, A.; Esposito, C.; Sommatis, S. In Vitro Evaluation of the Biological Availability of Hyaluronic Acid Polyethylene Glycols-Cross-Linked Hydrogels to Bovine Testes Hyaluronidase. BioMed Res. Int. 2019, 12, 1–5. [Google Scholar] [CrossRef]
- Funt, D.; Tatjana, P. Dermal fillers in aesthetics: An overview of adverse events and treatment approaches. Clin. Cosmet. Investig. Dermatol. 2013, 6, 295–316. [Google Scholar] [CrossRef]
- Yeom, J.S.; Bhang, S.H.; Kim, B.S.; Seo, M.S.; Hwang, E.J.; Cho, I.H.; Park, J.K.; Hahn, S.K. Effect of cross-linking reagents for hyaluronic acid hydrogel dermal fillers on tissue augmentation and regeneration. Bioconjug. Chem. 2010, 21, 240–247. [Google Scholar] [CrossRef]
- Harvard Health: Health Information and Medical Information. Available online: https://www.health.harvard.edu/blog/dermal-fillers-the-good-the-bad-and-the-dangerous-2019071517234 (accessed on 10 November 2020).
- Zamboni, F.; Ryan, E.; Culebras, M.; Collins, M.N. Labile crosslinked hyaluronic acid via urethane formation using bis(β-isocyanatoethyl) disulphide with tuneable physicochemical and immunomodulatory properties. Carbohydr. Polym. 2020, 245, 116501. [Google Scholar] [CrossRef]
- Tiwari, S.; Bahadur, P. Modified hyaluronic acid based materials for biomedical applications. Int. J. Biol. Macromol. 2019, 121, 556–571. [Google Scholar] [CrossRef]
- Mochizuki, M.; Aoi, N.; Gonda, K.; Hirabayashi, S.; Komuro, Y. Evaluation of the In Vivo Kinetics and Biostimulatory Effects of Subcutaneously Injected Hyaluronic Acid Filler. Plast. Reconstr. Surg. 2018, 142, 112–121. [Google Scholar] [CrossRef]
- Requena, L.; Requena, C.; Christensen, L.; Zimmermann, U.S.; Kutzner, H.; Cerroni, L. Adverse reactions to injectable soft tissue fillers. J. Am. Acad. Dermatol. 2011, 64, 1–34. [Google Scholar] [CrossRef] [PubMed]
- Lafaille, P.; Benedetto, A. Fillers: Contraindications, side effects and precautions. J. Cutan. Aesthet. Surg. 2010, 3, 16–19. [Google Scholar] [CrossRef] [PubMed]
- Buhren, B.A.; Schrumpf, H.; Hoff, N.P.; Bölke, E.; Hilton, S.; Gerber, P.A. Hyaluronidase: From clinical applications to molecular and cellular mechanisms. Eur. J. Med. Res. 2016, 21, 5. [Google Scholar] [CrossRef] [PubMed]
- Park, S.; Park, K.Y.; Yeo, I.K.; Cho, S.Y.; Ah, Y.C.; Koh, H.J.; Park, W.S.; Kim, B.J. Investigation of the degradation-retarding effect caused by the low swelling capacity of a novel hyaluronic acid filler developed by solid-phase crosslinking technology. Anna. Dermatol. 2014, 26, 357–362. [Google Scholar] [CrossRef] [PubMed]
- Huerta-Ángeles, G.; Nešporová, K.; Ambrožová, G.; Kubala, L.; Velebný, V. An Effective Translation: The Development of Hyaluronan-Based Medical Products from the Physicochemical, and Preclinical Aspects. Front. Bioeng. Biotechnol. 2018, 6, 62. [Google Scholar] [CrossRef]
- ISO 10993-6 Biological Evaluation of Medical Devices—Part 6: Tests for Local Effects after Implantation, 3rd ed.; The International Organization for Standardization (ISO): Geneva, Switzerland, 2016; pp. 5–6.
- Kim, J.H.; Kwon, T.-R.; Lee, S.E.; Jang, Y.N.; Han, H.S.; Mun, S.K.; Kim, B.J. Comparative Evaluation of the Effectiveness of Novel Hyaluronic Acid-Polynucleotide Complex Dermal filler. Sci. Rep. 2020, 10, 5127–5135. [Google Scholar] [CrossRef]
- Kim, J.S.; In, C.H.; Park, N.-J.; Kim, B.J.; Yoon, H.-S. Comparative study of rheological properties and preclinical data of porous polycaprolactone microsphere dermal fillers. J. Cosmet. Dermatol. 2019, 19, 596–604. [Google Scholar] [CrossRef]
- Kwon, T.-R.; Han, S.W.; Yeo, I.K.; Kim, J.H.; Kim, J.M.; Hong, J.-Y.; Lee, B.-C.; Lee, S.-E.; Moon, H.S.; Kwon, H.J.; et al. Biostimulatory effects of polydioxanone, poly-D, L lactic acid, and polycaprolactone fillers in mouse model. J. Cosmet. Dermatol. 2019, 18, 1002–1008. [Google Scholar] [CrossRef]
- Addor, F.A.S.; Guerra Neri, S.R.N. Injectable Polyethylene Glycol Gel as Dermal Filler: 01 Year Clinical and Ultrasound Follow-Up. J. Clin. Exp. Dermatol. Res. 2016, 7, 1–5. [Google Scholar] [CrossRef]
- Valachová, K.; Bañasová, M.; Topol’ská, D.; Sasinková, V.; Juránek, I.; Collins, M.N.; Šoltés, L. Influence of tiopronin, captopril and levamisole therapeutics on the oxidative degradation of hyaluronan. Carbohydr. Polym. 2015, 134, 516–523. [Google Scholar] [CrossRef]
- Valachová, K.; Topol’ská, D.; Mendichi, R.; Collins, M.N.; Sasinková, V.; Šoltés, L. Hydrogen peroxide generation by the Weissberger biogenic oxidative system during hyaluronan degradation. Carbohydr. Polym. 2016, 148, 189–193. [Google Scholar] [CrossRef] [PubMed]
- Shumate, G.T.; Chopra, R.; Jones, D.; Messina, D.J.; Hee, C.K. In Vivo Degradation of Crosslinked Hyaluronic Acid Fillers by Exogenous Hyaluronidases. Dermatol. Surg. 2018, 44, 1075–1083. [Google Scholar] [CrossRef] [PubMed]
- Chang, L.; Zhang, J.; Jiang, X. Comparative Properties of Hyaluronic Acid Hydrogel Cross-linked with 1, 4-Butanediol Diglycidyl Ether Assayed Using a Marine Hyaluronidase. IOP Conf. Ser. Mater. Sci. Eng. 2019, 493, 012007. [Google Scholar] [CrossRef]
- Lin, H.; Liu, J.; Zhang, K.; Fan, Y.; Zhang, X. Dynamic mechanical and swelling properties of maleated hyaluronic acid hydrogels. Carbohydr. Polym. 2015, 123, 381–389. [Google Scholar] [CrossRef]
- Rimmer, S. Biomedical Hydrogels. Biochemistry: Manufacture and Medical Applications, 1st ed.; Woodhead Publishing: Cambridge, UK, 2011; pp. 3–24. [Google Scholar] [CrossRef]
- Schott, H. Kinetics of swelling of polymers and their gels. J. Pharm. Sci. 1992, 81, 467–470. [Google Scholar] [CrossRef]
- Lee, D.Y.; Cheon, C.; Son, S.; Kim, Y.Z.; Kim, J.T.; Jang, J.W.; Kim, S.S. Influence of Molecular Weight on Swelling and Elastic Modulus of Hyaluronic Acid Dermal Fillers. Polym. Korea 2015, 39, 967–980. [Google Scholar] [CrossRef]
- Flynn, T.C.; Sarazin, D.; Bezzola, A.; Terrani, C.; Micheels, P. Comparative Histology of Intradermal Implantation of Mono and Biphasic Hyaluronic Acid Fillers. Dermatol. Surg. 2011, 37, 637–643. [Google Scholar] [CrossRef]
- Attenello, N.; Maas, C. Injectable Fillers: Review of Material and Properties. Facial Plast. Surg. 2015, 31, 29–34. [Google Scholar] [CrossRef]
- Kim, M.J.; Moon, W.-J.; Hur, M.S.; Han, S.H.; Lee, Y.W.; Choe, Y.B.; Ahn, K.J. Ex vivo magnetic resonance imaging using hyaluronic acid fillers: Differences between monophasic and biphasic fillers. Skin Res. Technol. 2017, 24, 16–19. [Google Scholar] [CrossRef]
- Park, K.Y.; Kim, H.K.; Kim, B.J. Comparative study of hyaluronic acid fillers by in vitro and in vivo testing. J. Eur. Acad. Dermatol. Venereol. 2013, 28, 565–568. [Google Scholar] [CrossRef]
| Trade Name | Parameters | Estimates (%RSE) | IIV (%RSE) | Bootstrap Median (2.5–97.5% Percentile) |
|---|---|---|---|---|
| 99 fill® | Kswell (day−1) | 2.2 (12. 6%) | 17.9% (20.3%) | 2.24 (1.84–2.85) |
| Kdeg (day−1) | 1.45 (8.8%) | - | 1.46 (1.26–1.73) | |
| Slope | 0 * | - | - | |
| Proportional residual variability CV% | 16.3% (13.1%) | - | - | |
| Juvederm® VOLUMA with Lidocaine | Kswell (day−1) | 3.04 (17.1%) | 46.5% (30.9%) | 3.06 (2.10–4.03) |
| Kdeg (day−1) | 1.98 (19.6%) | - | 1.99 (1.30–2.74) | |
| Slope | 1.15 (2.8%) | - | 1.15 (1.07–1.41) | |
| Proportional residual variability CV% | 13.8% (12.3%) | - | - | |
| Neuramis® VOLUME Lidocaine | Kswell (day−1) | 3.55 (8.5%) | 22.3% (13.5%) | 3.54 (3.03–4.19) |
| Kdeg (day−1) | 2.2 (8.8%) | - | 2.19 (1.87–2.63) | |
| Slope | 1.06 (11.1%) | - | 1.05 (0.754–1.33) | |
| Proportional residual variability CV% | 14.9% (14%) | - | - | |
| Restylane® Lyft with Lidocaine | Kswell (day−1) | 4.74 (13.1%) | 29% (22.3%) | 4.79 (3.87–5.90) |
| Kdeg (day−1) | 4.24 (11.6%) | - | 4.25 (3.52–5.23) | |
| Slope | 1.01 (18.3%) | - | 1.02 (0.436–1.53) | |
| Proportional residual variability CV% | 17.9% (11.9%) | - | - | |
| YVOIRE® Contour plus | Kswell (day−1) | 1.82 (10.2%) | 25% (38.5%) | 1.84 (1.50–2.25) |
| Kdeg (day−1) | 1.47 (10.7%) | - | 1.49 (1.20–1.83) | |
| Slope | 1.16 (14.5%) | - | 1.19 (0.891–2.88) | |
| Proportional residual variability CV% | 11.2% (10.8%) | - | - |
| Trade Name | Tcd Median (5–95% Quantile) |
|---|---|
| 99 fill | 1750 day (1450~2220 day) |
| Juvederm® VOLUMA with Lidocaine | 1250 day (630~2800 day) |
| Neuramis® VOLUME Lidocaine | 1120 day (760~1630 day) |
| Restylane® Lyft with Lidocaine | 740 day (490~1190 day) |
| YVOIRE® Contour plus | 2050 day (1360~3130 day) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ryu, H.-j.; Kwak, S.-s.; Rhee, C.-h.; Yang, G.-h.; Yun, H.-y.; Kang, W.-h. Model-Based Prediction to Evaluate Residence Time of Hyaluronic Acid Based Dermal Fillers. Pharmaceutics 2021, 13, 133. https://doi.org/10.3390/pharmaceutics13020133
Ryu H-j, Kwak S-s, Rhee C-h, Yang G-h, Yun H-y, Kang W-h. Model-Based Prediction to Evaluate Residence Time of Hyaluronic Acid Based Dermal Fillers. Pharmaceutics. 2021; 13(2):133. https://doi.org/10.3390/pharmaceutics13020133
Chicago/Turabian StyleRyu, Hyo-jeong, Seong-sung Kwak, Chang-hoon Rhee, Gi-hyeok Yang, Hwi-yeol Yun, and Won-ho Kang. 2021. "Model-Based Prediction to Evaluate Residence Time of Hyaluronic Acid Based Dermal Fillers" Pharmaceutics 13, no. 2: 133. https://doi.org/10.3390/pharmaceutics13020133
APA StyleRyu, H.-j., Kwak, S.-s., Rhee, C.-h., Yang, G.-h., Yun, H.-y., & Kang, W.-h. (2021). Model-Based Prediction to Evaluate Residence Time of Hyaluronic Acid Based Dermal Fillers. Pharmaceutics, 13(2), 133. https://doi.org/10.3390/pharmaceutics13020133






