Comparison of Genetically Engineered Immunodeficient Animal Models for Nonclinical Testing of Stem Cell Therapies
Abstract
1. Introduction
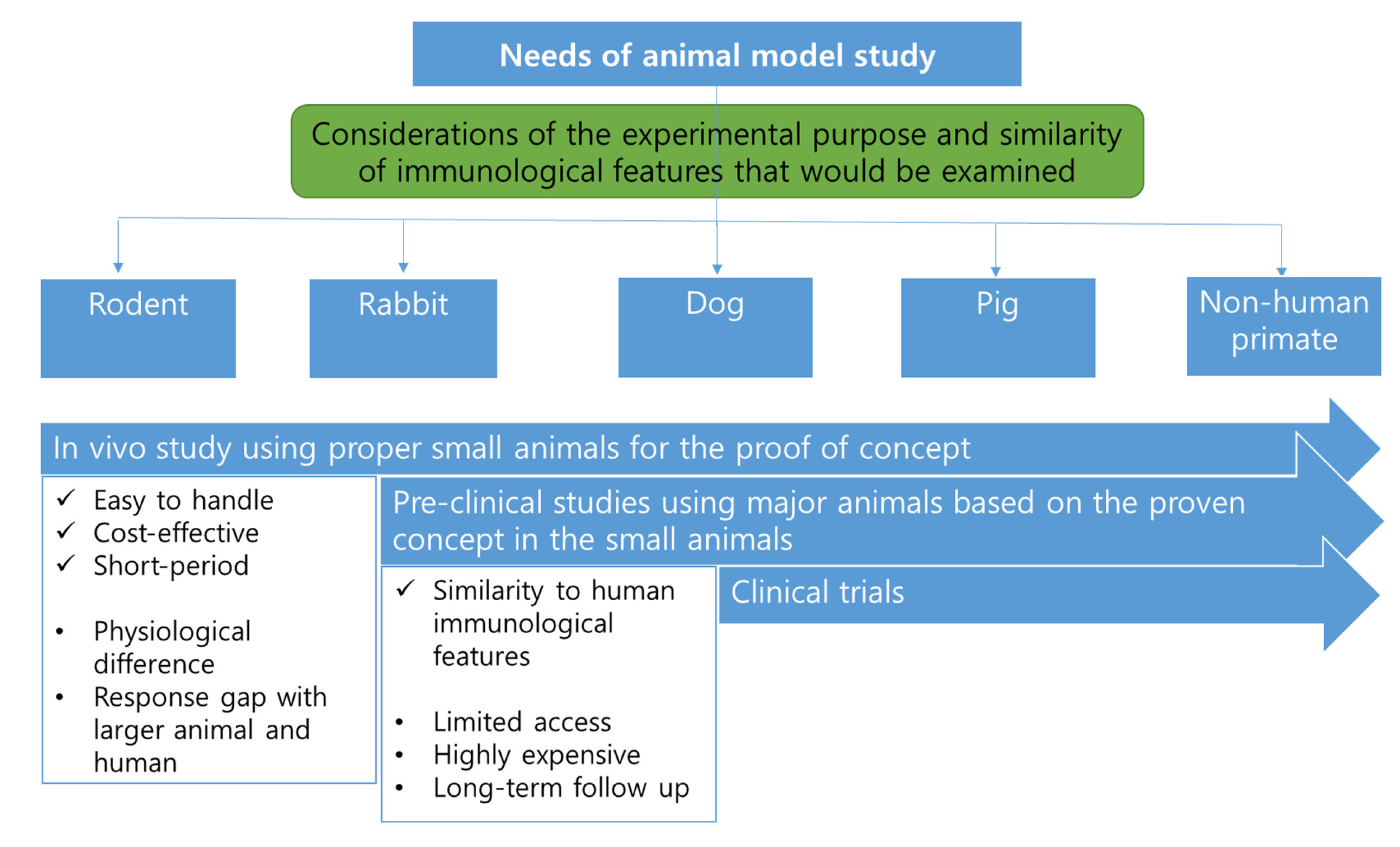
2. Immunodeficient Animals
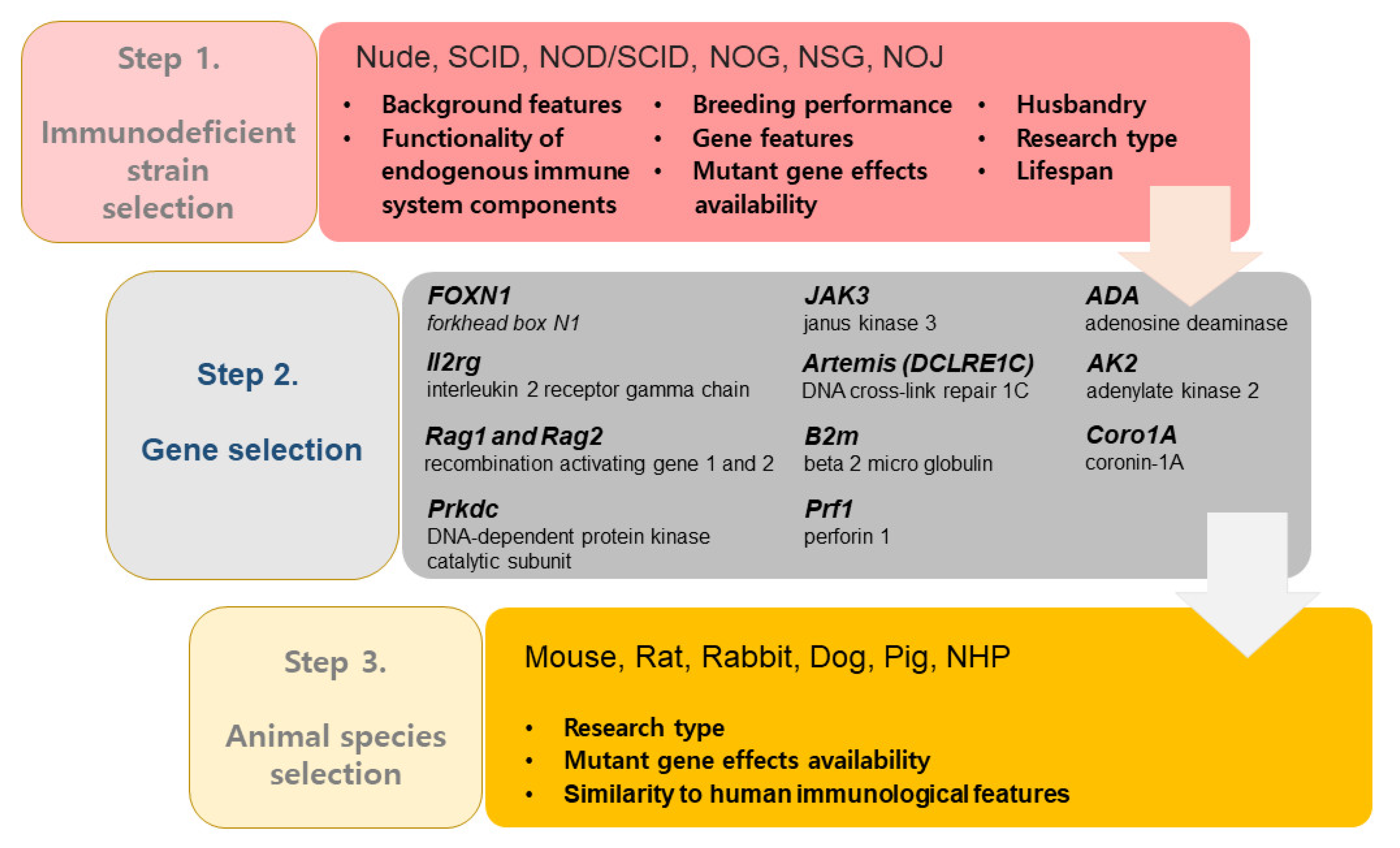
2.1. Genes Involved in Immunodeficiency
2.1.1. Forkhead Box N1 (FOXN1)
2.1.2. DNA-Dependent Protein Kinase Catalytic Subunit (PRKDC)
2.1.3. Interleukin 2 Receptor Subunit Gamma (IL2RG)
2.1.4. Recombination Activating Gene 1 and 2 (RAG1 and RAG2)
2.1.5. Janus Kinase 3 (JAK3)
2.1.6. Artemis (DCLRE1C)
2.1.7. Beta-2-Microglobulin (B2M) and Perforin 1
2.1.8. Adenosine Deaminase (ADA) and Adenylate Kinase 2 (AK2)
2.1.9. Coronin-1A (CORO1A)
2.2. Types and Characteristics of Immunodeficient Animals
2.2.1. Nude Animals
2.2.2. SCID Animals
2.2.3. SCID/Beige Animals
2.2.4. NOD/SCID Animals
2.2.5. NOD/SCID-Based Immunocompromised Animals
3. Comparison of Animal Species Used for Producing SCID Animals
3.1. Types of Animals
3.1.1. Mouse and Rat
3.1.2. Rabbit
3.1.3. Dog
3.1.4. Pig
3.1.5. NHP
4. Experimental Applications of Immunodeficient Animals
4.1. Rat and Mouse
4.2. Rabbit
4.3. Dog
4.4. Pig
4.5. NHP
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Aristizabal, A.M.; Caicedo, L.A.; Martinez, J.M.; Moreno, M.; Echeverri, G.J. Clinical xenotransplantation, a closer reality: Literature review. Cirugía Española 2017, 95, 62–72. [Google Scholar] [CrossRef] [PubMed]
- Deschamps, J.Y.; Roux, F.A.; Sai, P.; Gouin, E. History of xenotransplantation. Xenotransplantation 2005, 12, 91–109. [Google Scholar] [CrossRef] [PubMed]
- Cooper, D.K.C.; Ekser, B.; Tector, A.J. A brief history of clinical xenotransplantation. Int. J. Surg. 2015, 23, 205–210. [Google Scholar] [CrossRef] [PubMed]
- Cooper, D.K.C.; Gaston, R.; Eckhoff, D.; Ladowski, J.; Yamamoto, T.; Wang, L.; Iwase, H.; Hara, H.; Tector, M.; Tector, A.J. Xenotransplantation-the current status and prospects. Br. Med. Bull. 2018, 125, 5–14. [Google Scholar] [CrossRef]
- Cooper, D.K.; Gollackner, B.; Sachs, D.H. Will the pig solve the transplantation backlog? Annu. Rev. Med. 2002, 53, 133–147. [Google Scholar] [CrossRef]
- Kojima, Y.; Hayakawa, F.; Morishita, T.; Sugimoto, K.; Minamikawa, Y.; Iwase, M.; Yamamoto, H.; Hirano, D.; Imoto, N.; Shimada, K.; et al. YM155 induces apoptosis through proteasome-dependent degradation of MCL-1 in primary effusion lymphoma. Pharm. Res. 2017, 120, 242–251. [Google Scholar] [CrossRef]
- Rongvaux, A.; Takizawa, H.; Strowig, T.; Willinger, T.; Eynon, E.E.; Flavell, R.A.; Manz, M.G. Human Hemato-Lymphoid System Mice: Current Use and Future Potential for Medicine. Annu. Rev. Immunol. 2013, 31, 635–674. [Google Scholar] [CrossRef]
- Takenaka, K.; Prasolava, T.K.; Wang, J.C.Y.; Mortin-Toth, S.M.; Khalouei, S.; Gan, O.I.; Dick, J.E.; Danska, J.S. Polymorphism in Sirpa modulates engraftment of human hematopoietic stem cells. Nat. Immunol. 2007, 8, 1313–1323. [Google Scholar] [CrossRef]
- Strowig, T.; Rongvaux, A.; Rathinam, C.; Takizawa, H.; Borsotti, C.; Philbrick, W.; Eynon, E.E.; Manz, M.G.; Flavell, R.A. Transgenic expression of human signal regulatory protein alpha in Rag2(-/-)gamma(-/-)(c) mice improves engraftment of human hematopoietic cells in humanized mice. Proc. Natl. Acad. Sci. USA 2011, 108, 13218–13223. [Google Scholar] [CrossRef]
- Yamauchi, T.; Takenaka, K.; Urata, S.; Shima, T.; Kikushige, Y.; Tokuyama, T.; Iwamoto, C.; Nishihara, M.; Iwasaki, H.; Miyamoto, T.; et al. Polymorphic Sirpa is the genetic determinant for NOD-based mouse lines to achieve efficient human cell engraftment. Blood 2013, 121, 1316–1325. [Google Scholar] [CrossRef]
- He, J.J.; Rong, Z.L.; Fu, X.M.; Xu, Y. A Safety Checkpoint to Eliminate Cancer Risk of the Immune Evasive Cells Derived from Human Embryonic Stem Cells. Stem Cells 2017, 35, 1154–1161. [Google Scholar] [CrossRef] [PubMed]
- Rong, Z.L.; Fu, X.M.; Wang, M.Y.; Xu, Y. A Scalable Approach to Prevent Teratoma Formation of Human Embryonic Stem Cells. J. Biol. Chem. 2012, 287, 32338–32345. [Google Scholar] [CrossRef] [PubMed]
- Zhao, T.; Zhang, Z.N.; Westenskow, P.D.; Todorova, D.; Hu, Z.; Lin, T.; Rong, Z.; Kim, J.; He, J.; Wang, M.; et al. Humanized Mice Reveal Differential Immunogenicity of Cells Derived from Autologous Induced Pluripotent Stem Cells. Cell Stem Cell 2015, 17, 353–359. [Google Scholar] [CrossRef] [PubMed]
- Koboziev, I.; Jones-Hall, Y.; Valentine, J.F.; Reinoso Webb, C.; Furr, K.L.; Grisham, M.B. Use of Humanized Mice to Study the Pathogenesis of Autoimmune and Inflammatory Diseases. Inflamm. Bowel. Dis. 2015, 21, 1652–1673. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.J.; Duan, Z.Y.; Zhao, Y. Mouse models with human immunity and their application in biomedical research. J. Cell Mol. Med. 2009, 13, 1043–1058. [Google Scholar] [CrossRef]
- Kaestner, K.H.; Knochel, W.; Martinez, D.E. Unified nomenclature for the winged helix/forkhead transcription factors. Genes Dev. 2000, 14, 142–146. [Google Scholar]
- Pignata, C.; Fiore, M.; Guzzetta, V.; Castaldo, A.; Sebastio, G.; Porta, F.; Guarino, A. Congenital Alopecia and nail dystrophy associated with severe functional T-cell immunodeficiency in two sibs. Am. J. Med. Genet. 1996, 65, 167–170. [Google Scholar] [CrossRef]
- Zuklys, S.; Handel, A.; Zhanybekova, S.; Govani, F.; Keller, M.; Maio, S.; Mayer, C.E.; Teh, H.Y.; Hafen, K.; Gallone, G.; et al. Foxn1 regulates key target genes essential for T cell development in postnatal thymic epithelial cells. Nat. Immunol. 2016, 17, 1206. [Google Scholar] [CrossRef]
- Frank, J.; Pignata, C.; Panteleyev, A.A.; Prowse, D.M.; Baden, H.; Weiner, L.; Gaetaniello, L.; Ahmad, W.; Pozzi, N.; Cserhalmi-Friedman, P.B.; et al. Exposing the human nude phenotype. Nature 1999, 398, 473–474. [Google Scholar] [CrossRef]
- Palamaro, L.; Romano, R.; Fusco, A.; Giardino, G.; Gallo, V.; Pignata, C. FOXN1 in Organ Development and Human Diseases. Int. Rev. Immunol. 2014, 33, 83–93. [Google Scholar] [CrossRef]
- Segre, J.A.; Nemhauser, J.L.; Taylor, B.A.; Nadeau, J.H.; Lander, E.S. Positional cloning of the nude locus: Genetic, physical, and transcription maps of the region and mutations in the mouse and rat. Genomics 1995, 28, 549–559. [Google Scholar] [CrossRef]
- Nehls, M.; Kyewski, B.; Messerle, M.; Waldschutz, R.; Schuddekopf, K.; Smith, A.J.H.; Boehm, T. Two genetically separable steps in the differentiation of thymic epithelium. Science 1996, 272, 886–889. [Google Scholar] [CrossRef] [PubMed]
- Nehls, M.; Pfeifer, D.; Schorpp, M.; Hedrich, H.; Boehm, T. New member of the winged-helix protein family disrupted in mouse and rat nude mutations. Nature 1994, 372, 103–107. [Google Scholar] [CrossRef] [PubMed]
- Flanagan, S.P. ‘Nude’, a new hairless gene with pleiotropic effects in the mouse. Genet. Res. 1966, 8, 295–309. [Google Scholar] [CrossRef] [PubMed]
- Brissette, J.L.; Li, J.; Kamimura, J.; Lee, D.; Dotto, G.P. The product of the mouse nude locus Whn, regulates the balance between epithelial cell growth and differentiation. Gene Dev. 1996, 10, 2212–2221. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.; Prowse, D.M.; Brissette, J.L. Association between mouse nude gene expression and the initiation of epithelial terminal differentiation. Dev. Biol. 1999, 208, 362–374. [Google Scholar] [CrossRef] [PubMed]
- Kopfmaier, P.; Mboneko, V.F. Anomalies in the Hormonal Status of Athymic Nude-Mice. J. Cancer Res. Clin. 1990, 116, 229–231. [Google Scholar] [CrossRef]
- Bevan, M.J. In a radiation chimaera, host H-2 antigens determine immune responsiveness of donor cytotoxic cells (Reprint from Nature 269, pg 417-418, 1977). J. Immunol. 2006, 176, 5–6. [Google Scholar]
- Miller, J.F.A.P. The Immunological Function of the Thymus and of the Thymus-Derived Lymphocytes. Transpl. P 1991, 23, 9–10. [Google Scholar]
- Nelms, K.; Keegan, A.D.; Zamorano, J.; Ryan, J.J.; Paul, W.E. The IL-4 receptor: Signaling mechanisms and biologic functions. Annu. Rev. Immunol. 1999, 17, 701–738. [Google Scholar] [CrossRef]
- Cao, X.Q.; Shores, E.W.; Huli, J.; Anver, M.R.; Kelsall, B.L.; Russell, S.M.; Drago, J.; Noguchi, M.; Grinberg, A.; Bloom, E.T.; et al. Defective Lymphoid Development in Mice Lacking Expression of the Common Cytokine Receptor-Gamma Chain. Immunity 1995, 2, 223–238. [Google Scholar] [CrossRef]
- Mahaney, B.L.; Meek, K.; Lees-Miller, S.P. Repair of ionizing radiation-induced DNA double-strand breaks by non-homologous end-joining. Biochem. J. 2009, 417, 639–650. [Google Scholar] [CrossRef] [PubMed]
- Franco, S.; Alt, F.W.; Manis, J.P. Pathways that suppress programmed DNA breaks from progressing to chromosomal breaks and translocations. DNA Repair. 2006, 5, 1030–1041. [Google Scholar] [CrossRef] [PubMed]
- Yan, C.T.; Boboila, C.; Souza, E.K.; Franco, S.; Hickernell, T.R.; Murphy, M.; Gumaste, S.; Geyer, M.; Zarrin, A.A.; Manis, J.P.; et al. IgH class switching and translocations use a robust non-classical end-joining pathway. Nature 2007, 449, U478–U479. [Google Scholar] [CrossRef]
- Shrivastav, M.; De Haro, L.P.; Nickoloff, J.A. Regulation of DNA double-strand break repair pathway choice. Cell Res. 2008, 18, 134–147. [Google Scholar] [CrossRef]
- Bosma, G.C.; Custer, R.P.; Bosma, M.J. A Severe Combined Immunodeficiency Mutation in the Mouse. Nature 1983, 301, 527–530. [Google Scholar] [CrossRef]
- O’Driscoll, M.; Jeggo, P.A. The role of double-strand break repair—Insights from human genetics. Nat. Rev. Genet. 2006, 7, 45–54. [Google Scholar] [CrossRef]
- van der Burg, M.; IJspeert, H.; Verkaik, N.S.; Turul, T.; Wiegant, W.W.; Morotomi-Yano, K.; Mari, P.O.; Tezcan, I.; Chen, D.J.; Zdzienicka, M.Z.; et al. A DNA-PKcs mutation in a radiosensitive T-B- SCID patient inhibits Artemis activation and nonhomologous end-joining. J. Clin. Investig. 2009, 119, 91–98. [Google Scholar] [CrossRef]
- Perryman, L.E. Molecular pathology of severe combined immunodeficiency in mice, horses, and dogs. Vet. Pathol. 2004, 41, 95–100. [Google Scholar] [CrossRef]
- Allenspach, E.; Rawlings, D.J.; Scharenberg, A.M. X-Linked Severe Combined Immunodeficiency. In GeneReviews((R)); Adam, M.P., Ardinger, H.H., Pagon, R.A., Wallace, S.E., Bean, L.J.H., Stephens, K., Amemiya, A., Eds.; University of Washington: Seattle, WA, USA, 1993. [Google Scholar]
- Bai, Q.L.; Liu, N.; Kong, X.D.; Xu, X.J.; Zhao, Z.H. Mutation analyses and prenatal diagnosis in families of X-linked severe combined immunodeficiency caused by IL2Rgamma gene novel mutation. Genet. Mol. Res. 2015, 14, 6164–6172. [Google Scholar] [CrossRef]
- Okano, T.; Nishikawa, T.; Watanabe, E.; Watanabe, T.; Takashima, T.; Yeh, T.W.; Yamashita, M.; Tanaka-Kubota, M.; Miyamoto, S.; Mitsuiki, N.; et al. Maternal T and B cell engraftment in two cases of X-linked severe combined immunodeficiency with IgG1 gammopathy. Clin. Immunol. 2017, 183, 112–120. [Google Scholar] [CrossRef] [PubMed]
- Buckley, R.H. Molecular defects in human severe combined immunodeficiency and approaches to immune reconstitution. Annu. Rev. Immunol. 2004, 22, 625–655. [Google Scholar] [CrossRef] [PubMed]
- Noguchi, M.; Yi, H.F.; Rosenblatt, H.M.; Filipovich, A.H.; Adelstein, S.; Modi, W.S.; Mcbride, O.W.; Leonard, W.J. Interleukin-2 Receptor Gamma Chain Mutation Results in X-Linked Severe Combined Immunodeficiency in Humans. Cell 1993, 73, 147–157. [Google Scholar] [CrossRef]
- Suzuki, S.; Iwamoto, M.; Saito, Y.; Fuchimoto, D.; Sembon, S.; Suzuki, M.; Mikawa, S.; Hashimoto, M.; Aoki, Y.; Najima, Y.; et al. Il2rg Gene-Targeted Severe Combined Immunodeficiency Pigs. Cell Stem Cell 2012, 10, 753–758. [Google Scholar] [CrossRef] [PubMed]
- Fuchs, S.; Rensing-Ehl, A.; Erlacher, M.; Vraetz, T.; Hartjes, L.; Janda, A.; Rizzi, M.; Lorenz, M.R.; Gilmour, K.; de Saint-Basile, G.; et al. Patients with T+/low NK+ IL-2 receptor gamma chain deficiency have differentially-impaired cytokine signaling resulting in severe combined immunodeficiency. Eur. J. Immunol. 2014, 44, 3129–3140. [Google Scholar] [CrossRef]
- Schmalstieg, F.C.; Leonard, W.J.; Noguchi, M.; Berg, M.; Rudloff, H.E.; Denney, R.M.; Dave, S.K.; Brooks, E.G.; Goldman, A.S. Missense Mutation in Exon-7 of the Common Gamma-Chain Gene Causes a Moderate Form of X-Linked Combined Immunodeficiency. J. Clin. Investig. 1995, 95, 1169–1173. [Google Scholar] [CrossRef] [PubMed]
- DiSanto, J.P.; Muller, W.; Guy-Grand, D.; Fischer, A.; Rajewsky, K. Lymphoid development in mice with a targeted deletion of the interleukin 2 receptor gamma chain. Proc. Natl. Acad. Sci. USA 1995, 92, 377–381. [Google Scholar] [CrossRef]
- Yates, F.; Malassis-Seris, M.; Stockholm, D.; Bouneaud, C.; Larousserie, F.; Noguiez-Hellin, P.; Danos, O.; Kohn, D.B.; Fischer, A.; de Villartay, J.P.; et al. Gene therapy of RAG-2(-/-) mice: Sustained correction of the immunodeficiency. Blood 2002, 100, 3942–3949. [Google Scholar] [CrossRef]
- van Til, N.P.; de Boer, H.; Mashamba, N.; Wabik, A.; Huston, M.; Visser, T.P.; Fontana, E.; Poliani, P.L.; Cassani, B.; Zhang, F.; et al. Correction of murine Rag2 severe combined immunodeficiency by lentiviral gene therapy using a codon-optimized RAG2 therapeutic transgene. Mol. Ther. 2012, 20, 1968–1980. [Google Scholar] [CrossRef]
- Schwarz, K.; Gauss, G.H.; Ludwig, L.; Pannicke, U.; Li, Z.; Lindner, D.; Friedrich, W.; Seger, R.A.; Hansen-Hagge, T.E.; Desiderio, S.; et al. RAG mutations in human B cell-negative SCID. Science 1996, 274, 97–99. [Google Scholar] [CrossRef]
- Leonard, W.J.; O’Shea, J.J. JAKS AND STATS: Biological implications. Annu. Rev. Immunol. 1998, 16, 293–322. [Google Scholar] [CrossRef] [PubMed]
- Notarangelo, L.D.; Candotti, F. JAK3-deficient severe combined immunodeficiency. Immunol. Allergy Clin. 2000, 20, 97–111. [Google Scholar] [CrossRef]
- Taylor, N.; Candotti, F.; Smith, S.; Oakes, S.A.; Jahn, T.; Isakov, J.; Puck, J.M.; OShea, J.J.; Weinberg, K.; Johnston, J.A. Interleukin-4 signaling in B lymphocytes from patients with X-linked severe combined immunodeficiency. J. Biol. Chem. 1997, 272, 7314–7319. [Google Scholar] [CrossRef] [PubMed]
- Orlic, D.; Girard, L.; Lee, D.; Anderson, S.; Puck, J.; Bodine, D. Differential Expression of Interleukin-2 Receptor-Gamma (Gamma(Common)) and Its Partners, Il-2r, Il-4r, Il-7r, Il-9r, in Developing Thymocytes and Bone-Marrow of Mice. Exp. Hematol. 1995, 23, 820. [Google Scholar]
- Russell, S.M.; Johnston, J.A.; Noguchi, M.; Kawamura, M.; Bacon, C.M.; Friedmann, M.; Berg, M.; Mcvicar, D.W.; Witthuhn, B.A.; Silvennoinen, O.; et al. Interaction of Il-2r-Beta and Gamma(C) Chains with Jak1 and Jak3—Implications for Xscid and Xcid. Science 1994, 266, 1042–1045. [Google Scholar] [CrossRef] [PubMed]
- Buckley, R.H.; Schiff, R.I.; Schiff, S.E.; Markert, M.L.; Williams, L.W.; Harville, T.O.; Roberts, J.L.; Puck, J.M. Human severe combined immunodeficiency: Genetic, phenotypic, and functional diversity in one hundred eight infants. J. Pediatr. 1997, 130, 378–387. [Google Scholar] [CrossRef]
- Roberts, J.L.; Lengi, A.; Brown, S.M.; Chen, M.; Zhou, Y.J.; O’Shea, J.J.; Buckley, R.H. Janus kinase 3 (JAK3) deficiency: Clinical, immunologic, and molecular analyses of 10 patients and outcomes of stem cell transplantation. Blood 2004, 103, 2009–2018. [Google Scholar] [CrossRef]
- Mella, P.; Schumacher, R.F.; Cranston, T.; de Saint Basile, G.; Savoldi, G.; Notarangelo, L.D. Eleven novel JAK3 mutations in patients with severe combined immunodeficiency-including the first patients with mutations in the kinase domain. Hum. Mutat. 2001, 18, 355–356. [Google Scholar] [CrossRef]
- Schuetz, C.; Neven, B.; Dvorak, C.C.; Leroy, S.; Ege, M.J.; Pannicke, U.; Schwarz, K.; Schulz, A.S.; Hoenig, M.; Sparber-Sauer, M.; et al. SCID patients with ARTEMIS vs RAG deficiencies following HCT: Increased risk of late toxicity in ARTEMIS-deficient SCID. Blood 2014, 123, 281–289. [Google Scholar] [CrossRef]
- Cossu, F. Genetics of SCID. Ital. J. Pediatr. 2010, 36, 76. [Google Scholar] [CrossRef]
- Waide, E.H.; Dekkers, J.C.M.; Ross, J.W.; Rowland, R.R.R.; Wyatt, C.R.; Ewen, C.L.; Evans, A.B.; Thekkoot, D.M.; Boddicker, N.J.; Serão, N.V.; et al. Not all SCID pigs are created equally: Two independent mutations in the artemis gene cause SCID in Pigs. J. Immunol. 2015, 195, 3171–3179. [Google Scholar] [CrossRef] [PubMed]
- Powell, E.J.; Cunnick, J.E.; Knetter, S.M.; Loving, C.L.; Waide, E.H.; Dekkers, J.C.M.; Tuggle, C.K. NK cells are intrinsically functional in pigs with Severe Combined Immunodeficiency (SCID) caused by spontaneous mutations in the Artemis gene. Vet. Immunol. Immunopathol. 2016, 175, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Shultz, L.D.; Banuelos, S.; Lyons, B.; Samuels, R.; Burzenski, L.; Gott, B.; Lang, P.; Leif, J.; Appel, M.; Rossini, A.; et al. NOD/LtSz-Rag1nullPfpnull mice: A new model system with increased levels of human peripheral leukocyte and hematopoietic stem-cell engraftment. Transplantation 2003, 76, 1036–1042. [Google Scholar] [CrossRef]
- Voskoboinik, I.; Sutton, V.R.; Ciccone, A.; House, C.M.; Chia, J.; Darcy, P.K.; Yagita, H.; Trapani, J.A. Perforin activity and immune homeostasis: The common A91V polymorphism in perforin results in both presynaptic and postsynaptic defects in function. Blood 2007, 110, 1184–1190. [Google Scholar] [CrossRef] [PubMed]
- Kagi, D.; Ledermann, B.; Burki, K.; Seiler, P.; Odermatt, B.; Olsen, K.J.; Podack, E.R.; Zinkernagel, R.M.; Hengartner, H. Cytotoxicity mediated by T cells and natural killer cells is greatly impaired in perforin-deficient mice. Nature 1994, 369, 31–37. [Google Scholar] [CrossRef]
- Iqbal, M.A.; Hong, K.; Kim, J.H.; Choi, Y. Severe combined immunodeficiency pig as an emerging animal model for human diseases and regenerative medicines. BMB Rep. 2019, 52, 625–634. [Google Scholar] [CrossRef] [PubMed]
- Burkart, A.; Shi, X.; Chouinard, M.; Corvera, S. Adenylate Kinase 2 Links Mitochondrial Energy Metabolism to the Induction of the Unfolded Protein Response. J. Biol. Chem. 2011, 286, 4081–4089. [Google Scholar] [CrossRef] [PubMed]
- Romano, R.; Palamaro, L.; Fusco, A.; Iannace, L.; Maio, S.; Vigliano, I.; Giardino, G.; Pignata, C. From Murine to Human Nude/SCID: The Thymus, T-Cell Development and the Missing Link. Clin. Dev. Immunol. 2012, 2012, 467101. [Google Scholar] [CrossRef]
- Shiow, L.R.; Roadcap, D.W.; Paris, K.; Watson, S.R.; Grigorova, I.L.; Lebet, T.; An, J.; Xu, Y.; Jenne, C.N.; Föger, N.; et al. The actin regulator coronin 1A is mutant in a thymic egress-deficient mouse strain and in a patient with severe combined immunodeficiency. Nat. Immunol. 2008, 9, 1307–1315. [Google Scholar] [CrossRef]
- Povlsen, C.O.; Jacobsen, G.K. Chemotherapy of a Human Malignant-Melanoma Transplanted in Nude Mouse. Cancer Res. 1975, 35, 2790–2796. [Google Scholar]
- Schmidt, M.; Good, R.A. Transplantation of Human Cancers to Nude Mice and Effects of Thymus Grafts. J. Natl. Cancer I 1975, 55, 81–87. [Google Scholar] [CrossRef] [PubMed]
- Schuddekopf, K.; Schorpp, M.; Boehm, T. The whn transcription factor encoded by the nude locus contains an evolutionarily conserved and functionally indispensable activation domain. Proc. Natl. Acad. Sci. USA 1996, 93, 9661–9664. [Google Scholar] [CrossRef] [PubMed]
- Mosier, D.E.; Gulizia, R.J.; Baird, S.M.; Wilson, D.B. Transfer of a Functional Human Immune-System to Mice with Severe Combined Immunodeficiency. Nature 1988, 335, 256–259. [Google Scholar] [CrossRef]
- Mccune, J.M.; Namikawa, R.; Kaneshima, H.; Shultz, L.D.; Lieberman, M.; Weissman, I.L. The Scid-Hu Mouse—Murine Model for the Analysis of Human Hematolymphoid Differentiation and Function. Science 1988, 241, 1632–1639. [Google Scholar] [CrossRef] [PubMed]
- Taghian, A.; Budach, W.; Zietman, A.; Freeman, J.; Gioioso, D.; Suit, H.D. Quantitative Comparison between the Transplantability of Human and Murine Tumors into the Brain of Ncr/Sed-Nu/Nu Nude and Severe Combined Immunodeficient Mice. Cancer Res. 1993, 53, 5018–5021. [Google Scholar]
- Roder, J.; Duwe, A. Beige Mutation in the Mouse Selectively Impairs Natural Killer Cell-Function. Nature 1979, 278, 451–453. [Google Scholar] [CrossRef]
- Mosier, D.E.; Stell, K.L.; Gulizia, R.J.; Torbett, B.E.; Gilmore, G.L. Homozygous Scid Scid Beige Beige Mice Have Low-Levels of Spontaneous or Neonatal T-Cell-Induced B-Cell Generation. J. Exp. Med. 1993, 177, 191–194. [Google Scholar] [CrossRef]
- Thomsen, M.; Galvani, S.; Canivet, C.; Kamar, N.; Bohler, T. Reconstitution of immunodeficient SCID/beige mice with human cells: Applications in preclinical studies. Toxicology 2008, 246, 18–23. [Google Scholar] [CrossRef]
- Makino, S.; Kunimoto, K.; Muraoka, Y.; Mizushima, Y.; Katagiri, K.; Tochino, Y. Breeding of a non-obese, diabetic strain of mice. Jikken Dobutsu 1980, 29, 1–13. [Google Scholar] [CrossRef]
- Kikutani, H.; Makino, S. The Murine Autoimmune Diabetes Model—Nod and Related Strains. Adv. Immunol. 1992, 51, 285–322. [Google Scholar] [CrossRef]
- Shultz, L.D.; Schweitzer, P.A.; Christianson, S.W.; Gott, B.; Schweitzer, I.B.; Tennent, B.; Mckenna, S.; Mobraaten, L.; Rajan, T.V.; Greiner, D.L.; et al. Multiple Defects in Innate and Adaptive Immunological Function in Nod/Ltsz-Scid Mice. J. Immunol. 1995, 154, 180–191. [Google Scholar] [PubMed]
- Christianson, S.W.; Greiner, D.L.; Hesselton, R.A.; Leif, J.H.; Wagar, E.J.; Schweitzer, I.B.; Rajan, T.V.; Gott, B.; Roopenian, D.C.; Shultz, L.D. Enhanced human CD4+ T cell engraftment in beta2-microglobulin-deficient NOD-scid mice. J. Immunol. 1997, 158, 3578–3586. [Google Scholar] [PubMed]
- Notarangelo, L.D.; Giliani, S.; Mazza, C.; Mella, P.; Savoldi, G.; Rodriguez-Perez, C.; Mazzolari, E.; Fiorini, M.; Duse, M.; Plebani, A.; et al. Of genes and phenotypes: The immunological and molecular spectrum of combined immune deficiency. Defects of the gamma(c)-JAK3 signaling pathway as a model. Immunol. Rev. 2000, 178, 39–48. [Google Scholar] [CrossRef] [PubMed]
- Okada, S.; Harada, H.; Ito, T.; Saito, T.; Suzu, S. Early development of human hematopoietic and acquired immune systems in new born NOD/Scid/Jak3null mice intrahepatic engrafted with cord blood-derived CD34 + cells. Int. J. Hematol. 2008, 88, 476–482. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Shultz, L.D.; Lyons, B.L.; Burzenski, L.M.; Gott, B.; Chen, X.; Chaleff, S.; Kotb, M.; Gillies, S.D.; King, M.; Mangada, J.; et al. Human lymphoid and myeloid cell development in NOD/LtSz-scid IL2R gamma null mice engrafted with mobilized human hemopoietic stem cells. J. Immunol. 2005, 174, 6477–6489. [Google Scholar] [CrossRef] [PubMed]
- Ito, M.; Hiramatsu, H.; Kobayashi, K.; Suzue, K.; Kawahata, M.; Hioki, K.; Ueyama, Y.; Koyanagi, Y.; Sugamura, K.; Tsuji, K.; et al. NOD/SCID/gamma(c)(null) mouse: An excellent recipient mouse model for engraftment of human cells. Blood 2002, 100, 3175–3182. [Google Scholar] [CrossRef]
- Sponzilli, I.; Notarangelo, L.D. Severe combined immunodeficiency (SCID): From molecular basis to clinical management. Acta Biomed. 2011, 8, 5–13. [Google Scholar]
- Meek, K.; Kienker, L.; Dallas, C.; Wang, W.; Dark, M.J.; Venta, P.J.; Huie, M.L.; Hirschhorn, R.; Bell, T. SCID in Jack Russell terriers: A new animal model of DNA-PKcs deficiency. J. Immunol. 2001, 167, 2142–2150. [Google Scholar] [CrossRef]
- Mashimo, T.; Takizawa, A.; Voigt, B.; Yoshimi, K.; Hiai, H.; Kuramoto, T.; Serikawa, T. Generation of Knockout Rats with X-Linked Severe Combined Immunodeficiency (X-SCID) Using Zinc-Finger Nucleases. PLoS ONE 2010, 5, e8870. [Google Scholar] [CrossRef]
- Sato, K.; Oiwa, R.; Kumita, W.; Henry, R.; Sakuma, T.; Ito, R.; Nozu, R.; Inoue, T.; Katano, I.; Sato, K.; et al. Generation of a Nonhuman Primate Model of Severe Combined Immunodeficiency Using Highly Efficient Genome Editing. Cell Stem Cell 2016, 19, 127–138. [Google Scholar] [CrossRef]
- Felsburg, P.J.; Somberg, R.L.; Hartnett, B.J.; Henthorn, P.S.; Carding, S.R. Canine X-linked severe combined immunodeficiency. Immunol. Res. 1998, 17, 63–73. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, M.; Nakano, K.; Matsunari, H.; Matsuda, T.; Maehara, M.; Kanai, T.; Kobayashi, M.; Matsumura, Y.; Sakai, R.; Kuramoto, M.; et al. Generation of Interleukin-2 Receptor Gamma Gene Knockout Pigs from Somatic Cells Genetically Modified by Zinc Finger Nuclease-Encoding mRNA. PLoS ONE 2013, 8, e76478. [Google Scholar] [CrossRef] [PubMed]
- Ito, T.; Sendai, Y.; Yamazaki, S.; Seki-Soma, M.; Hirose, K.; Watanabe, M.; Fukawa, K.; Nakauchi, H. Generation of Recombination Activating Gene-1-Deficient Neonatal Piglets: A Model of T and B Cell Deficient Severe Combined Immune Deficiency. PLoS ONE 2014, 9, e113833. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.T.; Cho, B.; Ryu, J.; Ray, C.; Lee, E.J.; Yun, Y.J.; Ahn, S.; Lee, J.; Ji, D.Y.; Jue, N.; et al. Biallelic modification of IL2RG leads to severe combined immunodeficiency in pigs. Reprod. Biol. Endocrinol. 2016, 14, 74. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lee, K.; Kwon, D.N.; Ezashi, T.; Choi, Y.J.; Park, C.; Ericsson, A.C.; Brown, A.N.; Samuel, M.S.; Park, K.W.; Walters, E.M.; et al. Engraftment of human iPS cells and allogeneic porcine cells into pigs with inactivated RAG2 and accompanying severe combined immunodeficiency. Proc. Natl. Acad. Sci. USA 2014, 111, 7260–7265. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, S.; Iwamoto, M.; Hashimoto, M.; Suzuki, M.; Nakai, M.; Fuchimoto, D.; Sembon, S.; Eguchi-Ogawa, T.; Uenishi, H.; Onishi, A. Generation and characterization of RAG2 knockout pigs as animal model for severe combined immunodeficiency. Vet. Immunol. Immunop. 2016, 178, 37–49. [Google Scholar] [CrossRef]
- Hashikawa, Y.; Hayashi, R.; Tajima, M.; Okubo, T.; Azuma, S.; Kuwamura, M.; Takai, N.; Osada, Y.; Kunihiro, Y.; Mashimo, T.; et al. Generation of knockout rabbits with X-linked severe combined immunodeficiency (X-SCID) using CRISPR/Cas9. Sci. Rep. UK 2020, 10. [Google Scholar] [CrossRef]
- Powell, E.J.; Cunnick, J.E.; Tuggle, C.K. SCID pigs: An emerging large animal NK model. J. Rare Dis. Res. Treat. 2017, 2, 1–6. [Google Scholar]
- Floresco, S.B.; Geyer, M.A.; Gold, L.H.; Grace, A.A. Developing predictive animal models and establishing a preclinical trials network for assessing treatment effects on cognition in schizophrenia. Schizophr. Bull. 2005, 31, 888–894. [Google Scholar] [CrossRef]
- Goutianos, G.; Tzioura, A.; Kyparos, A.; Paschalis, V.; Margaritelis, N.V.; Veskoukis, A.S.; Zafeiridis, A.; Dipla, K.; Nikolaidis, M.G.; Vrabas, I.S. The rat adequately reflects human responses to exercise in blood biochemical profile: A comparative study. Physiol. Rep. 2015, 3, e12293. [Google Scholar] [CrossRef]
- Gibbs, R.A.; Weinstock, G.M.; Metzker, M.L.; Muzny, D.M.; Sodergren, E.J.; Scherer, S.; Scott, G.; Steffen, D.; Worley, K.C.; Burch, P.E.; et al. Genome sequence of the Brown Norway rat yields insights into mammalian evolution. Nature 2004, 428, 493–521. [Google Scholar] [CrossRef]
- Martignoni, M.; Groothuis, G.M.M.; de Kanter, R. Species differences between mouse, rat, dog, monkey and human CYP-mediated drug metabolism, inhibition and induction. Expert Opin. Drug Met. 2006, 2, 875–894. [Google Scholar] [CrossRef]
- Blais, E.M.; Rawls, K.D.; Dougherty, B.V.; Li, Z.I.; Kolling, G.L.; Ye, P.; Wallqvist, A.; Papin, J.A. Reconciled rat and human metabolic networks for comparative toxicogenomics and biomarker predictions. Nat. Commun. 2017, 8, 14250. [Google Scholar] [CrossRef]
- Ellenbroek, B.; Youn, J. Rodent models in neuroscience research: Is it a rat race? Dis. Model. Mech. 2016, 9, 1079–1087. [Google Scholar] [CrossRef]
- Robbins, T.W. Cross-species studies of cognition relevant to drug discovery: A translational approach. Br. J. Pharm. 2017, 174, 3191–3199. [Google Scholar] [CrossRef]
- Terrovitis, J.V.; Smith, R.R.; Marban, E. Assessment and Optimization of Cell Engraftment after Transplantation into the Heart. Circ. Res. 2010, 106, 479–494. [Google Scholar] [CrossRef]
- Fan, J.L.; Chen, Y.J.; Yan, H.Z.; Niimi, M.; Wang, Y.L.; Liang, J.Y. Principles and Applications of Rabbit Models for Atherosclerosis Research. J. Atheroscler. Thromb. 2018, 25, 213–220. [Google Scholar] [CrossRef]
- Mapara, M.; Thomas, B.S.; Bhat, K.M. Rabbit as an animal model for experimental research. Dent. Res. J. (Isfahan) 2012, 9, 111–118. [Google Scholar] [CrossRef]
- Schmitt, G.; Barrow, P.; Stephan-Gueldner, M. Alternatives to the Use of Nonhuman Primates in Regulatory Toxicology. In The Nonhuman Primate in Nonclinical Drug Development and Safety Assessment; Academic Press: Cambridge, MA, USA, 2015; pp. 337–355. [Google Scholar] [CrossRef]
- Fan, J.; Kitajima, S.; Watanabe, T.; Xu, J.; Zhang, J.; Liu, E.; Chen, Y.E. Rabbit models for the study of human atherosclerosis: From pathophysiological mechanisms to translational medicine. Pharmacol. Ther. 2015, 146, 104–119. [Google Scholar] [CrossRef]
- Ahn, S.J.; Hong, H.K.; Na, Y.M.; Park, S.J.; Ahn, J.; Oh, J.; Chung, J.Y.; Park, K.H.; Woo, S.J. Use of Rabbit Eyes in Pharmacokinetic Studies of Intraocular Drugs. J. Vis. Exp. 2016, 113, 53878. [Google Scholar] [CrossRef]
- Schafrum Macedo, A.; Cezaretti Feitosa, C.; Yoiti Kitamura Kawamoto, F.; Vinicius Tertuliano Marinho, P.; Dos Santos Dal-Bó, Í.; Fiuza Monteiro, B.; Prado, L.; Bregadioli, T.; Antonio Covino Diamante, G.; Ricardo Auada Ferrigno, C. Animal modeling in bone research-Should we follow the White Rabbit? Anim. Model. Exp. Med. 2019, 2, 162–168. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.S.; Lin, G.; Lue, T.F. Allogeneic and xenogeneic transplantation of adipose-derived stem cells in immunocompetent recipients without immunosuppressants. Stem Cells Dev. 2012, 21, 2770–2778. [Google Scholar] [CrossRef]
- Gomes, J.A.; Geraldes Monteiro, B.; Melo, G.B.; Smith, R.L.; Cavenaghi Pereira da Silva, M.; Lizier, N.F.; Kerkis, A.; Cerruti, H.; Kerkis, I. Corneal reconstruction with tissue-engineered cell sheets composed of human immature dental pulp stem cells. Invest. Ophthalmol. Vis. Sci. 2010, 51, 1408–1414. [Google Scholar] [CrossRef] [PubMed]
- Chu, C.R.; Szczodry, M.; Bruno, S. Animal models for cartilage regeneration and repair. Tissue Eng. Part. B Rev. 2010, 16, 105–115. [Google Scholar] [CrossRef] [PubMed]
- Smith, D.; Broadhead, C.; Descotes, G.; Fosse, R.; Hack, R.; Krauser, K.; Pfister, R.; Phillips, B.; Rabemampianina, Y.; Sanders, J.; et al. Preclinical safety evaluation using nonrodent species: An industry/welfare project to minimize dog use. ILAR J. 2002, 43 (Suppl. 1), S39–S42. [Google Scholar] [CrossRef]
- Hytonen, M.K.; Lohi, H. Canine models of human rare disorders. Rare Dis. 2016, 4, e1241362. [Google Scholar] [CrossRef]
- Box, R.J.; Spielmann, H. Use of the dog as non-rodent test species in the safety testing schedule associated with the registration of crop and plant protection products (pesticides): Present status. Arch. Toxicol. 2005, 79, 615–626. [Google Scholar] [CrossRef]
- Gilmore, K.M.; Greer, K.A. Why is the dog an ideal model for aging research? Exp. Gerontol. 2015, 71, 14–20. [Google Scholar] [CrossRef]
- Shifrine, M.; Taylor, N.; Rosenblatt, L.S.; Wilson, F. Seasonal variation in cell mediated immunity of clinically normal dogs. Exp. Hematol. 1980, 8, 318–326. [Google Scholar]
- Muir, P.; Hans, E.C.; Racette, M.; Volstad, N.; Sample, S.J.; Heaton, C.; Holzman, G.; Schaefer, S.L.; Bloom, D.D.; Bleedorn, J.A.; et al. Autologous bone marrow-derived mesenchymal stem cells modulate molecular markers of inflammation in dogs with cruciate ligament rupture. PLoS ONE 2016, 11, e0159095. [Google Scholar] [CrossRef]
- Black, L.L.; Gaynor, J.; Gahring, D.; Adams, C.; Aron, D.; Harman, S.; Gingerich, D.A.; Harman, R. Effect of adipose-derived mesenchymal stem and regenerative cells on lameness in dogs with chronic osteoarthritis of the coxofemoral joints: A randomized, double-blinded, multicenter, controlled trial. Vet. Ther. 2007, 8, 272–284. [Google Scholar] [PubMed]
- Gandolfi, F.; Vanelli, A.; Pennarossa, G.; Rahaman, M.; Acocella, F.; Brevini, T.A. Large animal models for cardiac stem cell therapies. Theriogenology 2011, 75, 1416–1425. [Google Scholar] [CrossRef] [PubMed]
- Rozkot, M.; Václavková, E.; Bělková, J. Minipigs as laboratory animals—review. Res. Pig Breed. 2015, 9, 10–14. [Google Scholar]
- Nunoya, T.; Shibuya, K.; Saitoh, T.; Yazawa, H.; Nakamura, K.; Baba, Y.; Hirai, T. Use of Miniature Pig for Biomedical Research, with Reference to Toxicologic Studies. J. Toxicol. Pathol. 2007, 20, 125–132. [Google Scholar] [CrossRef]
- Ganderup, N.C.; Harvey, W.; Mortensen, J.T.; Harrouk, W. The Minipig as Nonrodent Species in Toxicology-Where Are We Now? Int. J. Toxicol. 2012, 31, 507–528. [Google Scholar] [CrossRef]
- Dalgaard, L. Comparison of minipig, dog, monkey and human drug metabolism and disposition. J. Pharm. Toxicol. Methods 2015, 74, 80–92. [Google Scholar] [CrossRef]
- Baumann, A.; Flagella, K.; Forster, R.; de Haan, L.; Kronenberg, S.; Locher, M.; Richter, W.F.; Theil, F.P.; Todd, M. New challenges and opportunities in nonclinical safety testing of biologics. Regul. Toxicol. Pharm. 2014, 69, 226–233. [Google Scholar] [CrossRef]
- Boettcher, A.N.; Cunnick, J.E.; Powell, E.J.; Egner, T.K.; Charley, S.E.; Loving, C.L.; Tuggle, C.K. Porcine signal regulatory protein alpha binds to human CD47 to inhibit phagocytosis: Implications for human hematopoietic stem cell transplantation into severe combined immunodeficient pigs. Xenotransplantation 2019, 26, e12466. [Google Scholar] [CrossRef]
- Ha, C.W.; Park, Y.B.; Chung, J.Y.; Park, Y.G. Cartilage repair using composites of human umbilical cord blood-derived mesenchymal stem cells and hyaluronic acid hydrogel in a minipig model. Stem Cells Transl. Med. 2015, 4, 1044–1051. [Google Scholar] [CrossRef]
- Murata, D.; Tokunaga, S.; Tamura, T.; Kawaguchi, H.; Miyoshi, N.; Fujiki, M.; Nakayama, K.; Misumi, K. A preliminary study of osteochondral regeneration using a scaffold-free three-dimensional construct of porcine adipose tissue-derived mesenchymal stem cells. J. Orthop. Surg. Res. 2015, 10, 35. [Google Scholar] [CrossRef]
- Hotchkiss, G.G. Life Begins at Seventy; Sunstone Press: Santa Fe, NM, USA, 2015; p. 78. [Google Scholar]
- Young, S.S.; Schilling, A.M.; Skeans, S.; Ritacco, G. Short duration anaesthesia with medetomidine and ketamine in cynomolgus monkeys. Lab. Anim. 1999, 33, 162–168. [Google Scholar] [CrossRef]
- Orsi, A.; Rees, D.; Andreini, I.; Venturella, S.; Cinelli, S.; Oberto, G. Overview of the marmoset as a model in nonclinical development of pharmaceutical products. Regul. Toxicol. Pharm. 2011, 59, 19–27. [Google Scholar] [CrossRef]
- Kim, Y.Y.; Yun, J.W.; Kim, J.M.; Park, C.G.; Rosenwaks, Z.; Liu, H.C.; Kang, B.C.; Ku, S.Y. Gonadotropin ratio affects the in vitro growth of rhesus ovarian preantral follicles. J. Investig. Med. 2016, 64, 888–893. [Google Scholar] [CrossRef]
- Yun, J.W.; Kim, Y.Y.; Ahn, J.H.; Kang, B.C.; Ku, S.Y. Use of nonhuman primates for the development of bioengineered female reproductive organs. Tissue Eng. Regen. Med. 2016, 13, 323–334. [Google Scholar] [CrossRef]
- Mattison, J.A.; Roth, G.S.; Beasley, T.M.; Tilmont, E.M.; Handy, A.M.; Herbert, R.L.; Longo, D.L.; Allison, D.B.; Young, J.E.; Bryant, M.; et al. Impact of caloric restriction on health and survival in rhesus monkeys from the NIA study. Nature 2012, 489, 318. [Google Scholar] [CrossRef]
- Mubiru, J.N.; Garcia-Forey, M.; Cavazos, N.; Hemmat, P.; Dick, E.J., Jr.; Owston, M.A.; Bauer, C.A.; Shade, R.E.; Rogers, J. Serum prostate specific antigen changes in cynomolgus monkeys (Macaca fascicularis) on a high sugar high fat diet. Prostate 2012, 72, 469–475. [Google Scholar] [CrossRef]
- Bernacky, B.J.; Gibson, S.V.; Keeling, M.E.; Abee, C.R. Nonhuman primates. In Laboratory Animal Medicine; Fox, J.G., Anderson, L.C., Loew, F.M., Quimby, F.W., Eds.; Academic Press: San Diego, CA, USA, 2008; pp. 702–703. [Google Scholar]
- Zuhlke, U.; Weinbauer, G. The common marmoset (Callithrix jacchus) as a model in toxicology. Toxicol. Pathol. 2003, 31, 123–127. [Google Scholar] [CrossRef]
- Okano, H.; Hikishima, K.; Iriki, A.; Sasaki, E. The common marmoset as a novel animal model system for biomedical and neuroscience research applications. Semin. Fetal Neonatal Med. 2012, 17, 336–340. [Google Scholar] [CrossRef]
- Mansfield, K. Marmoset models commonly used in biomedical research. Comp. Med. 2003, 53, 383–392. [Google Scholar]
- Tardif, S.; Ross, C.; Bergman, P.; Fernandez, E.; Javors, M.; Salmon, A.; Spross, J.; Strong, R.; Richardson, A. Testing Efficacy of Administration of the Antiaging Drug Rapamycin in a Nonhuman Primate, the Common Marmoset. J. Gerontol. A-Biol. 2015, 70, 577–587. [Google Scholar] [CrossRef]
- Buse, E.; Habermann, G.; Osterburg, I.; Korte, R.; Weinbauer, G.F. Reproductive/developmental toxicity and immunotoxicity assessment in the nonhuman primate model. Toxicology 2003, 185, 221–227. [Google Scholar] [CrossRef]
- Korte, R.; Vogel, F.; Osterburg, I. The Primate as a Model for Hazard Assessment of Teratogens in Humans. Arch. Toxicol. 1987, 115–121. [Google Scholar]
- Daadi, M.M.; Barberi, T.; Shi, Q.; Lanford, R.E. Nonhuman primate models in translational regenerative medicine. Stem Cells Dev. 2014, 23 (Suppl. 1), 83–87. [Google Scholar] [CrossRef]
- Frost, P.A.; Chen, S.; Mezzles, M.J.; Voruganti, V.S.; Nava-Gonzalez, E.J.; Arriaga-Cazares, H.E.; Freed, K.A.; Comuzzie, A.G.; DeFronzo, R.A.; Kent, J.W., Jr.; et al. Successful pharmaceutical-grade streptozotocin (STZ)-induced hyperglycemia in a conscious tethered baboon (Papio hamadryas) model. J. Med. Primatol. 2015, 44, 202–217. [Google Scholar] [CrossRef]
- Samata, B.; Kikuchi, T.; Miyawaki, Y.; Morizane, A.; Mashimo, T.; Nakagawa, M.; Okita, K.; Takahashi, J. X-linked severe combined immunodeficiency (X-SCID) rats for xeno-transplantation and behavioral evaluation. J. Neurosci. Methods 2015, 243, 68–77. [Google Scholar] [CrossRef]
- Rigatti, L.H.; Toptan, T.; Newsome, J.T.; Moore, P.S.; Chang, Y. Identification and Characterization of Novel Rat Polyomavirus 2 in a Colony of X-SCID Rats by P-PIT assay. mSphere 2016, 1. [Google Scholar] [CrossRef]
- Besch-Williford, C.; Pesavento, P.; Hamilton, S.; Bauer, B.; Kapusinszky, B.; Phan, T.; Delwart, E.; Livingston, R.; Cushing, S.; Watanabe, R.; et al. A Naturally Transmitted Epitheliotropic Polyomavirus Pathogenic in Immunodeficient Rats: Characterization, Transmission, and Preliminary Epidemiologic Studies. Toxicol. Pathol. 2017, 45, 593–603. [Google Scholar] [CrossRef]
- Ohno, T.; Kai, T.; Miyasaka, Y.; Maruyama, H.; Ishih, A.; Kino, H. Intestinal immunity suppresses carrying capacity of rats for the model tapeworm, Hymenolepis diminuta. Parasitol. Int. 2018, 67, 357–361. [Google Scholar] [CrossRef]
- Tanaka, M.; Kuramochi, M.; Nakanishi, S.; Kuwamura, M.; Kuramoto, T. Rat polyomavirus 2 infection in a colony of X-linked severe combined immunodeficiency rats in Japan. J. Vet. Med. Sci. 2018, 80, 1400–1406. [Google Scholar] [CrossRef]
- Beldick, S.R.; Hong, J.; Altamentova, S.; Khazaei, M.; Hundal, A.; Zavvarian, M.M.; Rumajogee, P.; Chio, J.; Fehlings, M.G. Severe-combined immunodeficient rats can be used to generate a model of perinatal hypoxic-ischemic brain injury to facilitate studies of engrafted human neural stem cells. PLoS ONE 2018, 13, e0208105. [Google Scholar] [CrossRef]
- Watanabe, S.; Ohta, S.; Yajima, M.; Terashima, K.; Ito, M.; Mugishima, H.; Fujiwara, S.; Shimizu, K.; Honda, M.; Shimizu, N.; et al. Humanized NOD/SCID/IL2Rgamma(null) mice transplanted with hematopoietic stem cells under nonmyeloablative conditions show prolonged life spans and allow detailed analysis of human immunodeficiency virus type 1 pathogenesis. J. Virol. 2007, 81, 13259–13264. [Google Scholar] [CrossRef]
- Lagresle-Peyrou, C.; Benjelloun, F.; Hue, C.; Andre-Schmutz, I.; Bonhomme, D.; Forveille, M.; Beldjord, K.; Hacein-Bey-Abina, S.; De Villartay, J.P.; Charneau, P.; et al. Restoration of human B-cell differentiation into NOD-SCID mice engrafted with gene-corrected CD34+ cells isolated from Artemis or RAG1-deficient patients. Mol. Ther. 2008, 16, 396–403. [Google Scholar] [CrossRef]
- Beilhack, G.F.; Scheffold, Y.C.; Weissman, I.L.; Taylor, C.; Jerabek, L.; Burge, M.J.; Masek, M.A.; Shizuru, J.A. Purified allogeneic hematopoietic stem cell transplantation blocks diabetes pathogenesis in NOD mice. Diabetes 2003, 52, 59–68. [Google Scholar] [CrossRef]
- Song, J.; Wang, G.S.; Hoenerhoff, M.J.; Ruan, J.X.; Yang, D.S.; Zhang, J.F.; Yang, J.B.; Lester, P.A.; Sigler, R.; Bradley, M.; et al. Bacterial and Pneumocystis Infections in the Lungs of Gene-Knockout Rabbits with Severe Combined Immunodeficiency. Front. Immunol. 2018, 9, 429. [Google Scholar] [CrossRef]
- Hartnett, B.J.; Yao, D.P.; Suter, S.E.; Ellinwood, N.M.; Henthorn, P.S.; Moore, P.F.; McSweeney, P.A.; Nash, R.A.; Brown, J.D.; Weinberg, K.I.; et al. Transplantation of X-linked severe combined immunodeficient dogs with CD34(+) bone marrow cells. Biol. Blood Marrow. Transpl. 2002, 8, 188–197. [Google Scholar] [CrossRef]
- Goldschmidt, M.H.; Kennedy, J.S.; Kennedy, D.R.; Yuan, H.; Holt, D.E.; Casal, M.L.; Traas, A.M.; Mauldin, E.A.; Moore, P.F.; Henthorn, P.S.; et al. Severe papillomavirus infection progressing to metastatic squamous cell carcinoma in bone marrow-transplanted X-linked SCID dogs. J. Virol. 2006, 80, 6621–6628. [Google Scholar] [CrossRef]
- Verfuurden, B.; Wempe, F.; Reinink, P.; van Kooten, P.J.S.; Martens, E.; Gerritsen, R.; Vos, J.H.; Rutten, V.P.M.G.; Leegwater, P.A. Severe combined immunodeficiency in Frisian Water Dogs caused by a RAG1 mutation. Genes Immun. 2011, 12, 310–313. [Google Scholar] [CrossRef][Green Version]
- Burtner, C.R.; Beard, B.C.; Kennedy, D.R.; Wohlfahrt, M.E.; Adair, J.E.; Trobridge, G.D.; Scharenberg, A.M.; Torgerson, T.R.; Rawlings, D.J.; Felsburg, P.J.; et al. Intravenous injection of a foamy virus vector to correct canine SCID-X1. Blood 2014, 123, 3578–3584. [Google Scholar] [CrossRef]
- Huang, J.; Guo, X.G.; Fan, N.N.; Song, J.; Zhao, B.T.; Ouyang, Z.; Liu, Z.; Zhao, Y.; Yan, Q.M.; Yi, X.L.; et al. RAG1/2 Knockout Pigs with Severe Combined Immunodeficiency. J. Immunol. 2014, 193, 1496–1503. [Google Scholar] [CrossRef]
- Chen, N.; Dekkers, J.C.; Ewen, C.L.; Rowland, R.R. Porcine reproductive and respiratory syndrome virus replication and quasispecies evolution in pigs that lack adaptive immunity. Virus Res. 2015, 195, 246–249. [Google Scholar] [CrossRef]
- Ewen, C.L.; Cino-Ozuna, A.G.; He, H.; Kerrigan, M.A.; Dekkers, J.C.M.; Tuggle, C.K.; Rowland, R.R.R.; Wyatt, C.R. Analysis of blood leukocytes in a naturally occurring immunodeficiency of pigs shows the defect is localized to B and T cells. Vet. Immunol. Immunop. 2014, 162, 174–179. [Google Scholar] [CrossRef] [PubMed]
- Boettcher, A.N.; Li, Y.; Ahrens, A.P.; Kiupel, M.; Byrne, K.A.; Loving, C.L.; Cino-Ozuna, A.G.; Wiarda, J.E.; Adur, M.; Schultz, B.; et al. Novel Engraftment and T Cell Differentiation of Human Hematopoietic Cells in ART -/- IL2RG -/Y SCID Pigs. Front. Immunol. 2020, 11, 100. [Google Scholar] [CrossRef] [PubMed]
- Niu, Y.; Shen, B.; Cui, Y.; Chen, Y.; Wang, J.; Wang, L.; Kang, Y.; Zhao, X.; Si, W.; Li, W.; et al. Generation of gene-modified cynomolgus monkey via Cas9/RNA-mediated gene targeting in one-cell embryos. Cell 2014, 156, 836–843. [Google Scholar] [CrossRef] [PubMed]
- Kumita, W.; Sato, K.; Suzuki, Y.; Kurotaki, Y.; Harada, T.; Zhou, Y.; Kishi, N.; Sato, K.; Aiba, A.; Sakakibara, Y.; et al. Efficient generation of Knock-in/Knock-out marmoset embryo via CRISPR/Cas9 gene editing. Sci. Rep. UK 2019, 9, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Allen, T.M.; Brehm, M.A.; Bridges, S.; Ferguson, S.; Kumar, P.; Mirochnitchenko, O.; Palucka, K.; Pelanda, R.; Sanders-Beer, B.; Shultz, L.D.; et al. Humanized immune system mouse models: Progress, challenges and opportunities. Nat. Immunol. 2019, 20, 770–774. [Google Scholar] [CrossRef]
| Deleted Genes | Description | References |
|---|---|---|
| IL2rg | Generation of knockout rats with X-SCID using zinc-finger nucleases | [90] |
| IL2rg | Assessing in vivo function of human dopaminergic neurons using X-SCID rats | [149] |
| IL2rg | Evaluation of T-cell immune surveillance, critical for commensal polyomavirus control, in SCID rats | [150] |
| Prkdc, IL2rg | Identification, pathogenesis, and transmission of a novel polyomavirus in SCID F344 rats with null Prkdc and IL2rg genes | [151] |
| IL2rg | Identification of a major role for host immunity in determining the carrying capacity of H. diminuta in intestines of SCID rats | [152] |
| IL2rg | Detection of the rat polyomaviruses infection in a colony of X-SCID rats | [153] |
| Prkdc | Effects of hiPSC-NPCs in SCID rats as a model of neonatal hypoxic-ischemic brain injury | [154] |
| IL2rg | Analysis of human immunodeficiency virus type 1 pathogenesis in humanized NOD/SCID/Il2rg null mice transplanted with HSCs | [155] |
| Rag1 | Restoration of human B-cell differentiation into NOD-SCID mice | [156] |
| Deleted Genes | Description | References |
|---|---|---|
| IL2rg | Development and maintenance of stable strains of rabbits with X-SCID via the CRISPR/Cas9 system targeting Il2rg | [98] |
| IL2rg | Establishment of SCID rabbit models for the development of early diagnostics and therapeutics for immunodeficient patients | [158] |
| Deleted Gene | Description | References |
|---|---|---|
| IL2rg | Demonstration of CD34+ bone marrow cells to reconstitute normal B- and T-cell function in X-SCID dogs | [159] |
| IL2rg | Analysis of the response to papillomavirus infections progressing to metastatic squamous cell carcinoma. | [160] |
| Prkdc | Comparison of SCID animal models on the severity of the V(D)J recombination defects | [89] |
| Rag1 | Analysis of SCID dogs with Rag1 mutation | [161] |
| IL2rg | Intravenous injection of a foamy virus vector expressing the human IL2RG gene for the correction of SCID-X1 dogs | [162] |
| Deleted Genes | Description | References |
|---|---|---|
| IL2rg | Evaluation of preclinical regenerative stem cell strategies for clinical therapy | [45] |
| Rag2 | Demonstration of the growth of mature teratomas from human pluripotent stem cells in SCID pigs | [96] |
| Rag1/2 | Establishment of a SCID pig model by targeting porcine RAG1/2 genes via TALEN technology | [163] |
| IL2rg | Elucidation of the SCID phenotype by enumerating circulating white blood cell populations | [165] |
| Artemis | Generation of SCID pig model by spontaneous mutations in the Artemis gene | [63] |
| Artemis, IL2rg | Use of Artemis and IL2rg for SCID pigs lacking T, B, and NK cells | [166] |
| Species/Strain | Deleted Genes | Description | Reference |
|---|---|---|---|
| NHP/cynomolgus | Rag1 | Generation of gene-modified cynomolgus monkey via CRISPR/Cas9 system | [167] |
| NHP/marmoset | IL2rg | Development and evaluation of IL2rg knockout marmosets with immunodeficient phenotypes, possible to grow to adults | [91] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, Y.-Y.; Kim, J.-S.; Che, J.-H.; Ku, S.-Y.; Kang, B.-C.; Yun, J.-W. Comparison of Genetically Engineered Immunodeficient Animal Models for Nonclinical Testing of Stem Cell Therapies. Pharmaceutics 2021, 13, 130. https://doi.org/10.3390/pharmaceutics13020130
Kim Y-Y, Kim J-S, Che J-H, Ku S-Y, Kang B-C, Yun J-W. Comparison of Genetically Engineered Immunodeficient Animal Models for Nonclinical Testing of Stem Cell Therapies. Pharmaceutics. 2021; 13(2):130. https://doi.org/10.3390/pharmaceutics13020130
Chicago/Turabian StyleKim, Yoon-Young, Jin-Soo Kim, Jeong-Hwan Che, Seung-Yup Ku, Byeong-Cheol Kang, and Jun-Won Yun. 2021. "Comparison of Genetically Engineered Immunodeficient Animal Models for Nonclinical Testing of Stem Cell Therapies" Pharmaceutics 13, no. 2: 130. https://doi.org/10.3390/pharmaceutics13020130
APA StyleKim, Y.-Y., Kim, J.-S., Che, J.-H., Ku, S.-Y., Kang, B.-C., & Yun, J.-W. (2021). Comparison of Genetically Engineered Immunodeficient Animal Models for Nonclinical Testing of Stem Cell Therapies. Pharmaceutics, 13(2), 130. https://doi.org/10.3390/pharmaceutics13020130







