Developments of Smart Drug-Delivery Systems Based on Magnetic Molecularly Imprinted Polymers for Targeted Cancer Therapy: A Short Review
Abstract
1. Introduction
1.1. Hurdles in Chemotherapy
1.2. Nano-Size Delivery Systems
1.3. Molecularly Imprinted Technology toward Drug-Delivery System (DDS)
1.4. Magnetic Molecularly Imprinted Polymers, Promising Hybrid Nano-DDS
2. State of the Art
3. Magnetic Molecularly Imprinted Polymer (MMIP) Preparation Methods
3.1. Choose of Suitable Monomers with the Aid of Computational Modeling
3.1.1. Cohesion Parameters
3.1.2. Interaction Mode and Energy Calculation
3.2. Imprinting Strategy, Advantages of MNPs Surface Imprinting in Drug Delivery
3.2.1. Optimizations in the Imprinting Process toward Enhancing the MMIPs Physicochemical Features
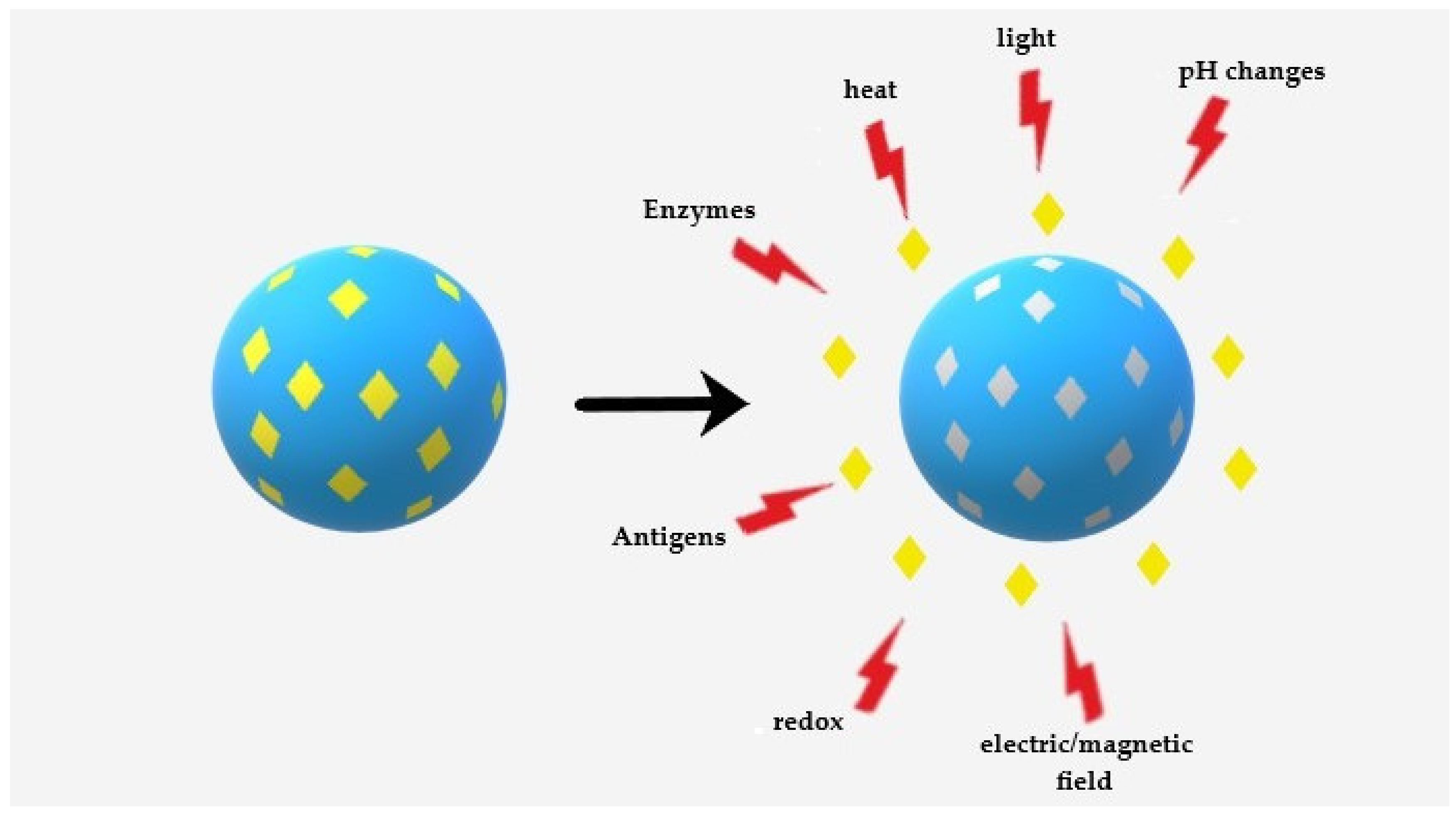
3.2.2. Stimuli-Sensitive MMIPs Triggered Release
3.2.3. Cyclodextrins as Comonomers
4. Results and Discussion
4.1. Particles Size and Loading Capacity
4.2. Toxicity and Degradability Studies
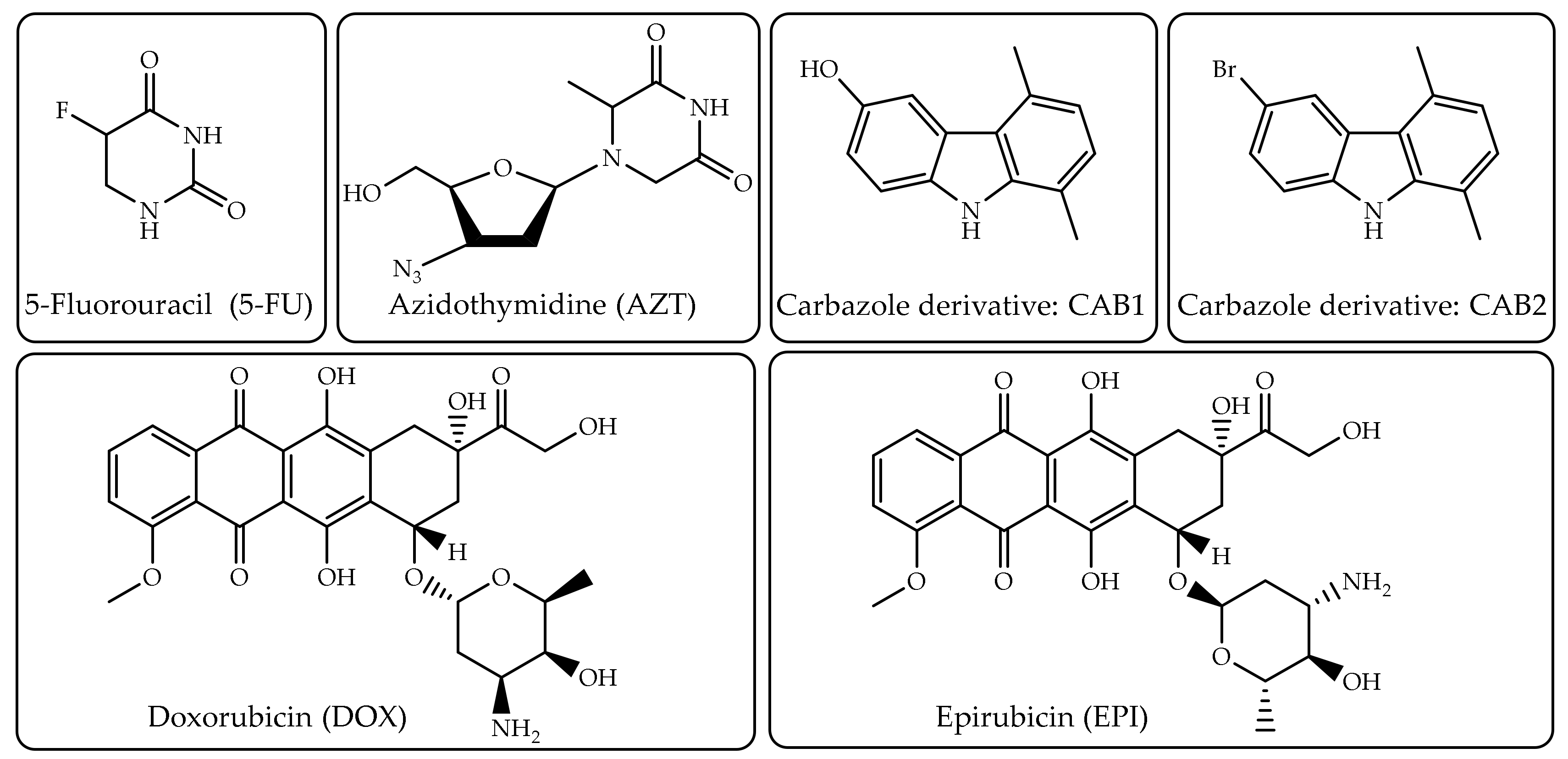
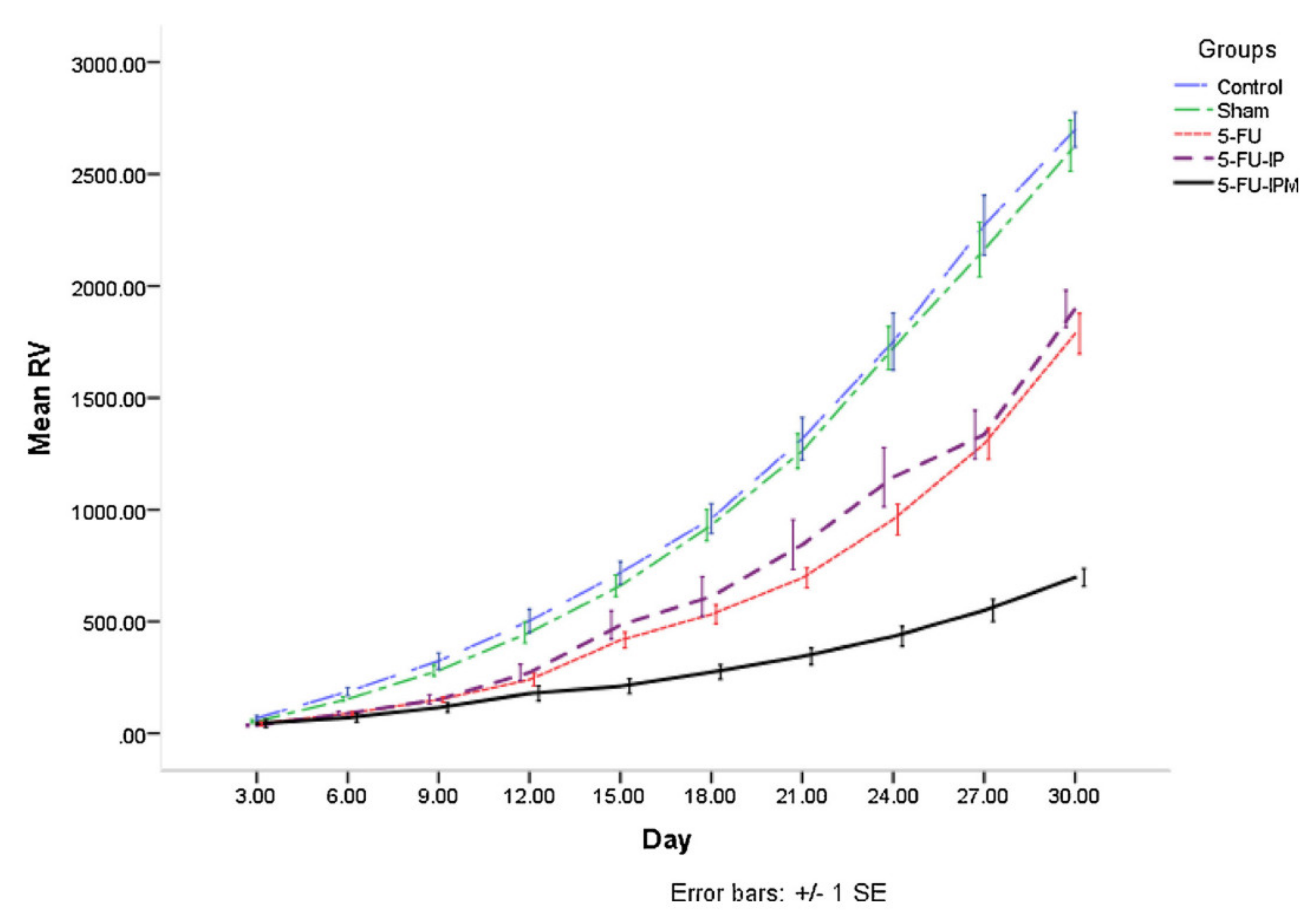
4.3. In-Vitro Drug Release Behavior and Cytotoxicity, In-Vivo Experimental Studies
5. Prospects and Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Moorthi, C.; Manavalan, R.; Kathiresan, K. Nanotherapeutics to overcome conventional cancer chemotherapy limitations. J. Pharm. Pharm. Sci. 2011, 14, 67–77. [Google Scholar] [CrossRef]
- Jahangirian, H.; Kalantari, K.; Izadiyan, Z.; Rafiee-Moghaddam, R.; Shameli, K.; Webster, T.J. A review of small molecules and drug delivery applications using gold and iron nanoparticles. Int. J. Nanomed. 2019, 14, 1633–1657. [Google Scholar] [CrossRef] [PubMed]
- Parisi, O.I.; Morelli, C.; Puoci, F.; Saturnino, C.; Caruso, A.; Sisci, D.; Trombino, G.E.; Picci, N.; Sinicropi, M.S. Magnetic molecularly imprinted polymers (MMIPs) for carbazole derivative release in targeted cancer therapy. J. Mater. Chem. B 2014, 2, 6619–6625. [Google Scholar] [CrossRef] [PubMed]
- Bodoki, A.E.; Iacob, B.-C.; Bodoki, E. Perspectives of Molecularly Imprinted Polymer-Based Drug Delivery Systems in Cancer Therapy. Polymers 2019, 11, 2085. [Google Scholar] [CrossRef] [PubMed]
- Senapati, S.; Mahanta, A.K.; Kumar, S.; Maiti, P. Controlled drug delivery vehicles for cancer treatment and their performance. Signal Transduct. Target. Ther. 2018, 3, 7. [Google Scholar] [CrossRef] [PubMed]
- Gardouh, A.R.; Barakat, B.M.; Qushawy, M.; Elkazaz, A.Y.; Sami, M.; Zaitone, S. Antitumor activity of a molecularly imprinted nanopreparation of 5-flurouracil against Ehrlich’s carcinoma solid tumors grown in mice: Comparison to free 5-flurouracil. Chem. Interact. 2018, 295, 52–63. [Google Scholar] [CrossRef] [PubMed]
- Ho, B.N.; Pfeffer, C.M.; Singh, A.T.K. Update on Nanotechnology-based Drug Delivery Systems in Cancer Treatment. Anticancer Res. 2017, 37, 5975–5981. [Google Scholar] [CrossRef]
- Yin, Q.; Shen, J.; Zhang, Z.; Yu, H.; Li, Y. Reversal of multidrug resistance by stimuli-responsive drug delivery systems for therapy of tumor. Adv. Drug Deliv. Rev. 2013, 65, 1699–1715. [Google Scholar] [CrossRef]
- Vangijzegem, T.; Stanicki, D.; Laurent, S. Magnetic iron oxide nanoparticles for drug delivery: Applications and characteristics. Expert Opin. Drug Deliv. 2019, 16, 69–78. [Google Scholar] [CrossRef]
- Xin, Y.; Yin, M.; Zhao, L.; Meng, F.; Luo, L. Recent progress on nanoparticle-based drug delivery systems for cancer therapy. Cancer Biol. Med. 2017, 14, 228. [Google Scholar] [CrossRef]
- Liu, Q.; Zhao, D.; Zhu, X.; Chen, H.; Yang, Y.; Xu, J.; Zhang, Q.; Fan, A.; Li, N.; Guo, C.; et al. Coloaded Nanoparticles of Paclitaxel and Piperlongumine for Enhancing Synergistic Antitumor Activities and Reducing Toxicity. J. Pharm. Sci. 2017, 106, 3066–3075. [Google Scholar] [CrossRef] [PubMed]
- Jacob, J.; Haponiuk, J.T.; Thomas, S.; Gopi, S. Biopolymer based nanomaterials in drug delivery systems: A review. Mater. Today Chem. 2018, 9, 43–55. [Google Scholar] [CrossRef]
- Elzoghby, A.O.; Freag, M.S.; Elkhodairy, K. Biopolymeric Nanoparticles for Targeted Drug Delivery to Brain Tumors; Elsevier Inc.: Amsterdam, The Netherlands, 2018. [Google Scholar]
- Bhunchu, S.; Rojsitthisak, P. Biopolymeric alginate-chitosan nanoparticles as drug delivery carriers for cancer therapy. Die Pharm. 2014, 69, 563–570. [Google Scholar]
- You, X.; Kang, Y.; Hollett, G.; Chen, X.; Zhao, W.; Gu, Z.; Wu, J. Polymeric nanoparticles for colon cancer therapy: Overview and perspectives. J. Mater. Chem. B 2016, 4, 7779–7792. [Google Scholar] [CrossRef]
- Kumari, A.; Yadav, S.K.; Yadav, S.C. Biodegradable polymeric nanoparticles based drug delivery systems. Colloids Surf. B Biointerfaces 2010, 75, 1–18. [Google Scholar] [CrossRef]
- Rivera, E. Current Status of Liposomal Anthracycline Therapy in Metastatic Breast Cancer. Clin. Breast Cancer 2003, 4 (Suppl. 2), S76–S83. [Google Scholar] [CrossRef]
- Anselmo, A.C.; Mitragotri, S. Nanoparticles in the clinic: An update. Bioeng. Transl. Med. 2019, 4, e10143. [Google Scholar] [CrossRef]
- Fan, Y.; Zhang, Q. Development of liposomal formulations: From concept to clinical investigations. Asian J. Pharm. Sci. 2013, 8, 81–87. [Google Scholar] [CrossRef]
- Ponzoni, M. Liposomal Chemotherapy BT-Encyclopedia of Cancer; Schwab, M., Ed.; Springer: Berlin/Heidelberg, Germany, 2011; pp. 2059–2063. [Google Scholar]
- Bobo, D.; Robinson, K.J.; Islam, J.; Thurecht, K.J.; Corrie, S.R. Nanoparticle-Based Medicines: A Review of FDA-Approved Materials and Clinical Trials to Date. Pharm. Res. 2016, 33, 2373–2387. [Google Scholar] [CrossRef]
- Pelaz, B.; Alexiou, C.; Alvarez-Puebla, R.A.; Alves, F.; Andrews, A.M.; Ashraf, S.; Balogh, L.; Ballerini, L.; Bestetti, A.; Brendel, C.; et al. Diverse Applications of Nanomedicine. ACS Nano 2017, 11, 2313–2381. [Google Scholar] [CrossRef]
- Huynh, N.; Passirani, C.; Saulnier, P.; Benoit, J.P. Lipid nanocapsules: A new platform for nanomedicine. Int. J. Pharm. 2009, 379, 201–209. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Liu, R.; Huang, H.; Zhu, Q. Vinblastine-Loaded Nanoparticles with Enhanced Tumor-Targeting Efficiency and Decreasing Toxicity: Developed by One-Step Molecular Imprinting Process. Mol. Pharm. 2019, 16, 2675–2689. [Google Scholar] [CrossRef] [PubMed]
- Johnson, K.P.; Brooks, B.R.; Cohen, J.A.; Ford, C.C.; Goldstein, J.; Lisak, R.P.; Myers, L.W.; Panitch, H.; Rose, J.W.; Schiffer, R.B.; et al. Extended use of glatiramer acetate (Copaxone) is well tolerated and maintains its clinical effect on multiple sclerosis relapse rate and degree of disability. Copolymer 1 Multiple Sclerosis Study Group. Neurology 1998, 50, 701–708. [Google Scholar] [CrossRef] [PubMed]
- Duncan, R. Polymer therapeutics: Top 10 selling pharmaceuticals—What next? J. Control. Release 2014, 190, 371–380. [Google Scholar] [CrossRef] [PubMed]
- El-Say, K.M.; El-Sawy, H. Polymeric nanoparticles: Promising platform for drug delivery. Int. J. Pharm. 2017, 528, 675–691. [Google Scholar] [CrossRef]
- Kumari, P.; Ghosh, B.; Biswas, S. Nanocarriers for cancer-targeted drug delivery. J. Drug Target. 2016, 24, 179–191. [Google Scholar] [CrossRef]
- Kalaydina, R.-V.; Bajwa, K.; Qorri, B.; Decarlo, A.; Szewczuk, M.R. Recent advances in “smart” delivery systems for extended drug release in cancer therapy. Int. J. Nanomed. 2018, 13, 4727–4745. [Google Scholar] [CrossRef] [PubMed]
- Wilhelm, S.; Tavares, A.J.; Dai, Q.; Ohta, S.; Audet, J.; Dvorak, H.F.; Chan, W.C.W. Analysis of nanoparticle delivery to tumours. Nat. Rev. Mater. 2016, 1, 16014. [Google Scholar] [CrossRef]
- Cisterna, B.A.; Kamaly, N.; Choi, W.I.; Tavakkoli, A.; Farokhzad, O.C.; Vilos, C. Targeted nanoparticles for colorectal cancer. Nanomedicine 2016, 11, 2443–2456. [Google Scholar] [CrossRef]
- Liu, Z.; Jiao, Y.; Wang, Y.; Zhou, C.; Zhang, Z. Polysaccharides-based nanoparticles as drug delivery systems. Adv. Drug Deliv. Rev. 2008, 60, 1650–1662. [Google Scholar] [CrossRef]
- Bi, Y.; Hao, F.; Yan, G.; Teng, L.; Lee, R.J.; Xie, J. Actively Targeted Nanoparticles for Drug Delivery to Tumor. Curr. Drug Metab. 2016, 17, 763–782. [Google Scholar] [CrossRef] [PubMed]
- Tietze, R.; Zaloga, J.; Unterweger, H.; Lyer, S.; Friedrich, R.P.; Janko, C.; Pöttler, M.; Dürr, S.; Alexiou, C. Magnetic nanoparticle-based drug delivery for cancer therapy. Biochem. Biophys. Res. Commun. 2015, 468, 463–470. [Google Scholar] [CrossRef] [PubMed]
- Cegłowski, M.; Kurczewska, J.; Ruszkowski, P.; Schroeder, G. Application of paclitaxel-imprinted microparticles obtained using two different cross-linkers for prolonged drug delivery. Eur. Polym. J. 2019, 118, 328–336. [Google Scholar] [CrossRef]
- Choi, J.; Yong, K.; Choi, J.Y.; Cowie, A.C. Progress in Molecularly Imprinted Polymers for Biomedical Applications. Comb. Chem. High Throughput Screen. 2019, 22, 78–88. [Google Scholar] [CrossRef] [PubMed]
- Puoci, F.; Iemma, F.; Picci, N. Stimuli-responsive molecularly imprinted polymers for drug delivery: A review. Curr. Drug Deliv. 2008, 5, 85–96. [Google Scholar] [CrossRef] [PubMed]
- Vasapollo, G.; Del Sole, R.; Mergola, L.; Lazzoi, M.R.; Scardino, A.; Scorrano, S.; Mele, G. Molecularly Imprinted Polymers: Present and Future Prospective. Int. J. Mol. Sci. 2011, 12, 5908–5945. [Google Scholar] [CrossRef]
- Zaidi, S.A. Latest trends in molecular imprinted polymer based drug delivery systems. RSC Adv. 2016, 6, 88807–88819. [Google Scholar] [CrossRef]
- Uzun, L.; Turner, A.P. Molecularly-imprinted polymer sensors: Realising their potential. Biosens. Bioelectron. 2016, 76, 131–144. [Google Scholar] [CrossRef]
- Gui, R.; Jin, H.; Guo, H.; Wang, Z. Recent advances and future prospects in molecularly imprinted polymers-based electrochemical biosensors. Biosens. Bioelectron. 2018, 100, 56–70. [Google Scholar] [CrossRef]
- Xu, J.; Miao, H.; Wang, J.; Pan, G. Molecularly Imprinted Synthetic Antibodies: From Chemical Design to Biomedical Applications. Small 2020, 16, 1906644. [Google Scholar] [CrossRef]
- Muratsugu, S.; Shirai, S.; Tada, M. Recent progress in molecularly imprinted approach for catalysis. Tetrahedron Lett. 2020, 61, 151603. [Google Scholar] [CrossRef]
- Zhang, N.; Zhang, N.; Xu, Y.; Li, Z.; Yan, C.; Mei, K.; Ding, M.; Ding, S.; Guan, P.; Qian, L.; et al. Molecularly Imprinted Materials for Selective Biological Recognition. Macromol. Rapid Commun. 2019, 40, e1900096. [Google Scholar] [CrossRef] [PubMed]
- Luliński, P. Molecularly imprinted polymers based drug delivery devices: A way to application in modern pharmacotherapy. A review. Mater. Sci. Eng. C 2017, 76, 1344–1353. [Google Scholar] [CrossRef] [PubMed]
- Tuwahatu, C.A.; Yeung, C.C.; Lam, Y.W.; Roy, V.A.L. The molecularly imprinted polymer essentials: Curation of anticancer, ophthalmic, and projected gene therapy drug delivery systems. J. Control. Release 2018, 287, 24–34. [Google Scholar] [CrossRef]
- Luliński, P.; Maciejewska, D.; Klejn, D. Investigations of molecularly imprinted polymers for medical diagnostics and clinical analysis. Polym. Med. 2011, 41, 3–15. [Google Scholar]
- He, Y.; Zeng, S.; El-Aty, A.M.A.; Hacımüftüoğlu, A.; Yohannes, W.K.; Khan, M.; She, Y. Development of Water-Compatible Molecularly Imprinted Polymers Based on Functionalized β-Cyclodextrin for Controlled Release of Atropine. Polymers 2020, 12, 130. [Google Scholar] [CrossRef]
- Zhang, H. Molecularly Imprinted Nanoparticles for Biomedical Applications. Adv. Mater. 2019, 32, 1806328. [Google Scholar] [CrossRef]
- Dramou, P.; Zuo, P.; He, H.; Pham-Huy, L.A.; Zou, W.; Xiao, D.; Pham-Huy, C.; Ndorbor, T. Anticancer loading and controlled release of novel water-compatible magnetic nanomaterials as drug delivery agents, coupled to a computational modeling approach. J. Mater. Chem. B 2013, 1, 4099–4109. [Google Scholar] [CrossRef]
- Bai, J.; Zhang, Y.; Chen, L.; Yan, H.; Zhang, C.; Liu, L.; Xu, X. Synthesis and characterization of paclitaxel-imprinted microparticles for controlled release of an anticancer drug. Mater. Sci. Eng. C 2018, 92, 338–348. [Google Scholar] [CrossRef]
- Zhang, K.; Guan, X.; Qiu, Y.; Wang, N.; Zhang, X.; Zhang, H. A pH/glutathione double responsive drug delivery system using molecular imprint technique for drug loading. Appl. Surf. Sci. 2016, 389, 1208–1213. [Google Scholar] [CrossRef]
- Asadi, E.; Ezzati, N.; Abdouss, M.; Leblanc, R.M.; Wilson, J.N.; Azodi-Deilami, S. In vitro/in vivo study of novel anti-cancer, biodegradable cross-linked tannic acid for fabrication of 5-fluorouracil-targeting drug delivery nano-device based on a molecular imprinted polymer. RSC Adv. 2016, 6, 37308–37318. [Google Scholar] [CrossRef]
- Esfandyari-Manesh, M.; Darvishi, B.; Ishkuh, F.A.; Shahmoradi, E.; Mohammadi, A.; Javanbakht, M.; Dinarvand, R.; Atyabi, F. Paclitaxel molecularly imprinted polymer-PEG-folate nanoparticles for targeting anticancer delivery: Characterization and cellular cytotoxicity. Mater. Sci. Eng. C 2016, 62, 626–633. [Google Scholar] [CrossRef] [PubMed]
- Madadian-Bozorg, N.; Zahedi, P.; Shamsi, M.; Safarian, S. Poly (methacrylic acid)-based molecularly imprinted polymer nanoparticles containing 5-fluourouracil used in colon cancer therapy potentially. Polym. Adv. Technol. 2018, 29, 2401–2409. [Google Scholar] [CrossRef]
- Samarth, N.B.; Kamble, V.; Mahanwar, P.A.; Rane, A.V.; Abitha, V.K. A historical perspective and the development of molecular imprinting polymer-A review. Chem. Int. 2015, 4, 202–210. [Google Scholar]
- Sellergren, B.; Allender, C. Molecularly imprinted polymers: A bridge to advanced drug delivery. Adv. Drug Deliv. Rev. 2005, 57, 1733–1741. [Google Scholar] [CrossRef]
- Widder, K.J.; Senyel, A.E.; Scarpelli, D.G.; Senyei, A.E. Magnetic Microspheres: A Model System for Site Specific Drug Delivery in Vivo. Exp. Biol. Med. 1978, 158, 141–146. [Google Scholar] [CrossRef]
- Dinc, M.; Esen, C.; Mizaikoff, B. Recent advances on core–shell magnetic molecularly imprinted polymers for biomacromolecules. TrAC Trends Anal. Chem. 2019, 114, 202–217. [Google Scholar] [CrossRef]
- Ji, K.; Luo, X.; He, L.; Liao, S.; Hu, L.; Han, J.; Chen, C.; Liu, Y.; Tan, N. Preparation of hollow magnetic molecularly imprinted polymer and its application in silybin recognition and controlled release. J. Pharm. Biomed. Anal. 2020, 180, 113036. [Google Scholar] [CrossRef]
- Talavat, L.; Güner, A. Thermodynamic computational calculations for preparation 5-fluorouracil magnetic moleculary imprinted polymers and their application in controlled drug release. Inorg. Chem. Commun. 2019, 103, 119–127. [Google Scholar] [CrossRef]
- Wackerlig, J.; Lieberzeit, P.A. Molecularly imprinted polymer nanoparticles in chemical sensing—Synthesis, characterisation and application. Sens. Actuators B Chem. 2015, 207, 144–157. [Google Scholar] [CrossRef]
- Canfarotta, F.; Lezina, L.; Guerreiro, A.; Czulak, J.; Petukhov, A.; Daks, A.; Smolinska-Kempisty, K.; Poma, A.; Piletsky, S.; Barlev, N.A.; et al. Specific Drug Delivery to Cancer Cells with Double-Imprinted Nanoparticles against Epidermal Growth Factor Receptor. Nano Lett. 2018, 18, 4641–4646. [Google Scholar] [CrossRef] [PubMed]
- Asadi, E.; Abdouss, M.; Leblanc, R.M.; Ezzati, N.; Wilson, J.N.; Kordestani, D. Synthesis, characterization and in vivo drug delivery study of a biodegradable nano-structured molecularly imprinted polymer based on cross-linker of fructose. Polymers 2016, 97, 226–237. [Google Scholar] [CrossRef]
- Hashemi-Moghaddam, H.; Kazemi-Bagsangani, S.; Jamili, M.; Zavareh, S. Evaluation of magnetic nanoparticles coated by 5-fluorouracil imprinted polymer for controlled drug delivery in mouse breast cancer model. Int. J. Pharm. 2016, 497, 228–238. [Google Scholar] [CrossRef] [PubMed]
- Hashemi-Moghaddam, H.; Zavareh, S.; Karimpour, S.; Madanchi, H. Evaluation of molecularly imprinted polymer based on HER2 epitope for targeted drug delivery in ovarian cancer mouse model. React. Funct. Polym. 2017, 121, 82–90. [Google Scholar] [CrossRef]
- Lin, M.M.; Kim, D.K.; El Haj, A.J.; Dobson, J. Development of Superparamagnetic Iron Oxide Nanoparticles (SPIONS) for Translation to Clinical Applications. IEEE Trans. NanoBiosci. 2008, 7, 298–305. [Google Scholar] [CrossRef]
- Cazares-Cortes, E.; Nerantzaki, M.; Fresnais, J.; Wilhelm, C.; Griffete, N.; Menager, C. Magnetic Nanoparticles Create Hot Spots in Polymer Matrix for Controlled Drug Release. Nanomaterials 2018, 8, 850. [Google Scholar] [CrossRef] [PubMed]
- Tietze, R.; Lyer, S.; Dürr, S.; Struffert, T.; Engelhorn, T.; Schwarz, M.; Eckert, E.; Göen, T.; Vasylyev, S.; Peukert, W.; et al. Efficient drug-delivery using magnetic nanoparticles—Biodistribution and therapeutic effects in tumour bearing rabbits. Nanomed. Nanotechnol. Biol. Med. 2013, 9, 961–971. [Google Scholar] [CrossRef]
- Pham, H.N.; Pham, T.H.G.; Nguyen, D.T.; Phan, Q.T.; Le, T.T.H.; Ha, P.T.; Do, H.M.; Hoang, T.M.N.; Nguyen, X.P. Magnetic inductive heating of organs of mouse models treated by copolymer coated Fe3O4 nanoparticles. Adv. Nat. Sci. Nanosci. Nanotechnol. 2017, 8, 025013. [Google Scholar] [CrossRef]
- Behrouzkia, Z.; Joveini, Z.; Keshavarzi, B.; Eyvazzadeh, N.; Aghdam, R.Z. Hyperthermia: How Can It Be Used? Oman Med. J. 2016, 31, 89–97. [Google Scholar] [CrossRef]
- Sanchez, C.; Shea, K.J.; Kitagawa, S.; Schubert, U. Hybrid materials themed issue Cluster-based inorganic–organic hybrid materialsw. Chem. Soc. Rev. 2011, 40, 453–1152. [Google Scholar]
- Martinelli, C.; Pucci, C.; Ciofani, G. Nanostructured carriers as innovative tools for cancer diagnosis and therapy. APL Bioeng. 2019, 3, 011502. [Google Scholar] [CrossRef] [PubMed]
- Shi, J.; Kantoff, P.W.; Wooster, R.; Farokhzad, O.C. Cancer nanomedicine: Progress, challenges and opportunities. Nat. Rev. Cancer 2017, 17, 20–37. [Google Scholar] [CrossRef]
- Thiesen, B.; Jordan, A. Clinical applications of magnetic nanoparticles for hyperthermia. Int. J. Hyperth. 2008, 24, 467–474. [Google Scholar] [CrossRef] [PubMed]
- Maier-Hauff, K.; Ulrich, F.; Nestler, D.; Niehoff, H.; Wust, P.; Thiesen, B.; Orawa, H.; Budach, V.; Jordan, A. Efficacy and safety of intratumoral thermotherapy using magnetic iron-oxide nanoparticles combined with external beam radiotherapy on patients with recurrent glioblastoma multiforme. J. Neuro-Oncol. 2011, 103, 317–324. [Google Scholar] [CrossRef] [PubMed]
- Griffete, N.; Fresnais, J.; Espinosa, A.; Wilhelm, C.; Bée, A.; Menager, C. Design of magnetic molecularly imprinted polymer nanoparticles for controlled release of doxorubicin under an alternative magnetic field in athermal conditions. Nanoscale 2015, 7, 18891–18896. [Google Scholar] [CrossRef]
- Kaamyabi, S.; Habibi, D.; Amini, M.M. Preparation and characterization of the pH and thermosensitive magnetic molecular imprinted nanoparticle polymer for the cancer drug delivery. Bioorg. Med. Chem. Lett. 2016, 26, 2349–2354. [Google Scholar] [CrossRef]
- Zavareh, S.; Mahdi, M.; Erfanian, S.; Hashemi-Moghaddam, H. Synthesis of polydopamine as a new and biocompatible coating of magnetic nanoparticles for delivery of doxorubicin in mouse breast adenocarcinoma. Cancer Chemother. Pharmacol. 2016, 78, 1073–1084. [Google Scholar] [CrossRef]
- Li, L.; Chen, L.; Zhang, H.; Yang, Y.-Z.; Liu, X.-G.; Chen, Y. Temperature and magnetism bi-responsive molecularly imprinted polymers: Preparation, adsorption mechanism and properties as drug delivery system for sustained release of 5-fluorouracil. Mater. Sci. Eng. C 2016, 61, 158–168. [Google Scholar] [CrossRef]
- Hassanpour, A.; Irandoust, M.; Soleimani, E.; Zhaleh, H.; Irandust, M. Increasing the anticancer activity of azidothymidine toward the breast cancer via rational design of magnetic drug carrier based on molecular imprinting technology. Mater. Sci. Eng. C 2019, 103, 109771. [Google Scholar] [CrossRef]
- Luo, L.; Jin, X.; Zhang, P.; Cheng, H.; Li, Y.; Du, T.; Zou, B.; Gou, M. Efficient intravesical therapy of bladder cancer with cationic doxorubicin nanoassemblies. Int. J. Nanomed. 2016, 11, 4535–4544. [Google Scholar] [CrossRef]
- O’Brien, M.E.R.; Borthwick, A.; Rigg, A.; Leary, A.; Assersohn, L.; Last, K.; Tan, S.; Milan, S.; Tait, D.; Smith, I.E. Mortality within 30 days of chemotherapy: A clinical governance benchmarking issue for oncology patients. Br. J. Cancer 2006, 95, 1632–1636. [Google Scholar] [CrossRef] [PubMed]
- Yoffe, S.; Leshuk, T.; Everett, P.; Gu, F.X. Superparamagnetic iron oxide nanoparticles (SPIONs): Synthesis and surface modification techniques for use with MRI and other biomedical applications. Curr. Pharm. Des. 2013, 19, 493–509. [Google Scholar] [CrossRef] [PubMed]
- Yáñez-Sedeño, P.; Campuzano, S.; Pingarrón, J.M.; Campuzano, S. Electrochemical sensors based on magnetic molecularly imprinted polymers: A review. Anal. Chim. Acta 2017, 960, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Ali, A.; Zafar, H.; Zia, M.; Haq, I.U.; Phull, A.R.; Ali, J.S.; Hussain, A. Synthesis, characterization, applications, and challenges of iron oxide nanoparticles. Nanotechnol. Sci. Appl. 2016, 9, 49–67. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Han, M.; Guan, G.; Wang, S.; Liu, R.; Zhang, Z. Highly-Controllable Molecular Imprinting at Superparamagnetic Iron Oxide Nanoparticles for Ultrafast Enrichment and Separation. J. Phys. Chem. C 2011, 115, 17320–17327. [Google Scholar] [CrossRef]
- Hemmati, K.; Sahraei, R.; Ghaemy, M. Synthesis and characterization of a novel magnetic molecularly imprinted polymer with incorporated graphene oxide for drug delivery. Polymers 2016, 101, 257–268. [Google Scholar] [CrossRef]
- Rostamizadeh, K.; Vahedpour, M.; Bozorgi, S. Synthesis, characterization and evaluation of computationally designed nanoparticles of molecular imprinted polymers as drug delivery systems. Int. J. Pharm. 2012, 424, 67–75. [Google Scholar] [CrossRef]
- Mojica, E.-R.E. Screening of different computational models for the preparation of sol–gel imprinted materials. J. Mol. Model. 2013, 19, 3911–3923. [Google Scholar] [CrossRef]
- Gholivand, K.; Khodadadian, M.; Ahmadi, F. Computer aided-molecular design and synthesis of a high selective molecularly imprinted polymer for solid-phase extraction of furosemide from human plasma. Anal. Chim. Acta 2010, 658, 225–232. [Google Scholar] [CrossRef]
- Prasad, B.B.; Rai, G. Study on monomer suitability toward the template in molecularly imprinted polymer: An ab initio approach. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2012, 88, 82–89. [Google Scholar] [CrossRef]
- Wu, L.; Sun, B.; Li, Y.; Chang, W. Study properties of molecular imprinting polymer using a computational approach. Analyst 2003, 128, 944–949. [Google Scholar] [CrossRef]
- Salas, C. 4-Solution electrospinning of nanofibers. In Woodhead Publishing Series in Textiles; Afshari, M., Ed.; Woodhead Publishing: Cambridge, UK, 2017; pp. 73–108. [Google Scholar]
- Piletska, E.V.; Abd, B.H.; Krakowiak, A.S.; Parmar, A.; Pink, D.L.; Wall, K.S.; Wharton, L.; Moczko, E.; Whitcombe, M.J.; Karim, K.; et al. Magnetic high throughput screening system for the development of nano-sized molecularly imprinted polymers for controlled delivery of curcumin. Analyst 2015, 140, 3113–3120. [Google Scholar] [CrossRef] [PubMed]
- Korde, B.A.; Mankar, J.S.; Phule, S.; Krupadam, R.J. Nanoporous imprinted polymers (nanoMIPs) for controlled release of cancer drug. Mater. Sci. Eng. C 2019, 99, 222–230. [Google Scholar] [CrossRef]
- Diñeiro, Y.; Menéndez, M.I.; Blanco-López, M.C.; Castanon, M.J.L.; Miranda-Ordieres, A.J.; Tuñón-Blanco, P. Computational Approach to the Rational Design of Molecularly Imprinted Polymers for Voltammetric Sensing of Homovanillic Acid. Anal. Chem. 2005, 77, 6741–6746. [Google Scholar] [CrossRef] [PubMed]
- Pardeshi, S.; Patrikar, R.; Dhodapkar, R.S.; Kumar, A. Validation of computational approach to study monomer selectivity toward the template Gallic acid for rational molecularly imprinted polymer design. J. Mol. Model. 2012, 18, 4797–4810. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Li, X.; Li, Y.; Dong, C.; Jin, P.; Qi, J. Selective recognition of veterinary drugs residues by artificial antibodies designed using a computational approach. Biomaterials 2009, 30, 3205–3211. [Google Scholar] [CrossRef]
- Ramezanpour, M.; Leung, S.S.; Delgado-Magnero, K.; Bashe, B.; Thewalt, J.; Tieleman, D. Computational and experimental approaches for investigating nanoparticle-based drug delivery systems. Biochim. Biophys. Acta (BBA)-Biomembr. 2016, 1858, 1688–1709. [Google Scholar] [CrossRef]
- Morris, B.A. (Ed.) 8-Barrier. In Plastics Design Library; William Andrew Publishing: Oxford, UK, 2017; pp. 259–308. [Google Scholar]
- Meier, F.; Schott, B.; Riedel, D.; Mizaikoff, B. Computational and experimental study on the influence of the porogen on the selectivity of 4-nitrophenol molecularly imprinted polymers. Anal. Chim. Acta 2012, 744, 68–74. [Google Scholar] [CrossRef]
- Turner, N.W.; Piletska, E.V.; Karim, K.; Whitcombe, M.J.; Malecha, M.; Magan, N.; Baggiani, C.; Piletsky, S. Effect of the solvent on recognition properties of molecularly imprinted polymer specific for ochratoxin A. Biosens. Bioelectron. 2004, 20, 1060–1067. [Google Scholar] [CrossRef]
- Li, Y.; Ding, M.-J.; Wang, S.; Wang, R.-Y.; Wu, X.-L.; Wen, T.-T.; Yuan, L.-H.; Dai, P.; Lin, Y.-H.; Zhou, X. Preparation of Imprinted Polymers at Surface of Magnetic Nanoparticles for the Selective Extraction of Tadalafil from Medicines. ACS Appl. Mater. Interfaces 2011, 3, 3308–3315. [Google Scholar] [CrossRef]
- Dąbrowski, M.; Lach, P.; Cieplak, M.; Kutner, W. Nanostructured molecularly imprinted polymers for protein chemosensing. Biosens. Bioelectron. 2018, 102, 17–26. [Google Scholar] [CrossRef]
- Ansari, S.; Masoum, S. Molecularly imprinted polymers for capturing and sensing proteins: Current progress and future implications. TrAC Trends Anal. Chem. 2019, 114, 29–47. [Google Scholar] [CrossRef]
- Chen, Y.; Xie, Z.; Zhang, L.; Hu, X. Effective preparation of magnetic molecularly imprinted polymer nanoparticle for the rapid and selective extraction of cyfluthrin from honeysuckle. J. Biomater. Sci. Polym. Ed. 2020, 31, 954–968. [Google Scholar] [CrossRef] [PubMed]
- Dramou, P.; Zuo, P.; He, H.; Pham-Huy, L.A.; Zou, W.; Xiao, D.; Pham-Huy, C. Development of novel amphiphilic magnetic molecularly imprinted polymers compatible with biological fluids for solid phase extraction and physicochemical behavior study. J. Chromatogr. A 2013, 1317, 110–120. [Google Scholar] [CrossRef] [PubMed]
- Dirion, B.; Cobb, Z.; Schillinger, E.; Andersson, L.I.; Sellergren, B. Water-Compatible Molecularly Imprinted Polymers Obtained via High-Throughput Synthesis and Experimental Design. J. Am. Chem. Soc. 2003, 125, 15101–15109. [Google Scholar] [CrossRef]
- Lee, H.; Scherer, N.F.; Messersmith, P.B. Single-molecule mechanics of mussel adhesion. Proc. Natl. Acad. Sci. USA 2006, 103, 12999–13003. [Google Scholar] [CrossRef]
- Zhang, C.; Jia, X.; Wang, Y.; Zhang, M.; Yang, S.; Guo, J. Thermosensitive molecularly imprinted hydrogel cross-linked with N -malely chitosan for the recognition and separation of BSA. J. Sep. Sci. 2014, 37, 419–426. [Google Scholar] [CrossRef]
- Chen, F.; Jiang, X.; Kuang, T.; Chang, L.; Fu, D.; Yang, Z.; Yang, J.; Fan, P.; Fei, Z.; Zhong, M. Effect of nanoporous structure and polymer brushes on the ionic conductivity of poly(methacrylic acid)/anode aluminum oxide hybrid membranes. RSC Adv. 2015, 5, 70204–70210. [Google Scholar] [CrossRef]
- Chen, F.; Jiang, X.; Mi, H.-Y.; Chang, L.; Fu, D.; Yang, J.; Fan, P.; Zhong, M. Polyelectrolyte/mesoporous silica hybrid materials for the high performance multiple-detection of pH value and temperature. Polym. Chem. 2015, 6, 3529–3536. [Google Scholar] [CrossRef]
- Liu, W.; Zhao, H.; Yang, Y.-Z.; Liu, X.-G.; Xu, B. Reactive carbon microspheres prepared by surface-grafting 4-(chloromethyl)phenyltrimethoxysilane for preparing molecularly imprinted polymer. Appl. Surf. Sci. 2013, 277, 146–154. [Google Scholar] [CrossRef]
- Xu, L.; Pan, J.; Dai, J.; Li, X.; Hang, H.; Cao, Z.; Yan, Y. Preparation of thermal-responsive magnetic molecularly imprinted polymers for selective removal of antibiotics from aqueous solution. J. Hazard. Mater. 2012, 233, 48–56. [Google Scholar] [CrossRef]
- Tang, H.; Zhao, W.; Yu, J.; Zhao, C. Recent Development of pH-Responsive Polymers for Cancer Nanomedicine. Molecules 2018, 24, 4. [Google Scholar] [CrossRef] [PubMed]
- Kocak, G.; Tuncer, C.; Bütün, V. PH-Responsive polymers. Polym. Chem. 2017, 8, 144–176. [Google Scholar] [CrossRef]
- Bae, Y.; Jang, W.-D.; Nishiyama, N.; Fukushima, S.; Kataoka, K. Multifunctional polymeric micelles with folate-mediated cancer cell targeting and pH-triggered drug releasing properties for active intracellular drug delivery. Mol. BioSyst. 2005, 1, 242. [Google Scholar] [CrossRef] [PubMed]
- Chawla, J.S.; Amiji, M. Biodegradable poly(ε-caprolactone) nanoparticles for tumor-targeted delivery of tamoxifen. Int. J. Pharm. 2002, 249, 127–138. [Google Scholar] [CrossRef]
- Meléndez-Ortiz, H.I.; Varca, G.H.; Zavala-Lagunes, E.; Bucio, E. State of the art of smart polymers: From fundamentals to final applications. In Polymer Science: Research Advances, Practical Applications and Educational Aspects; Formatex Research Center: Badajoz, Spain, 2016; pp. 476–487. [Google Scholar]
- Guragain, S.; Bastakoti, B.P.; Malgras, V.; Nakashima, K.; Yamauchi, Y. Multi-Stimuli-Responsive Polymeric Materials. Chem.-A Eur. J. 2015, 21, 13164–13174. [Google Scholar] [CrossRef]
- Liu, F.; Urban, M.W. Recent advances and challenges in designing stimuli-responsive polymers. Prog. Polym. Sci. 2010, 35, 3–23. [Google Scholar] [CrossRef]
- Tang, W.; Du, W.; Guo, P.; Wu, N.; Du, K.; Xu, C.; Luo, Z.; Chang, R.; Zeng, A.; Jing, W.; et al. Molecularly Imprinted Solid Phase Extraction using Bismethacryloyl-β-cyclodextrin and Methacrylic Acid as Double Functional Monomers for Selective Analysis of Glycyrrhizic Acid in Aqueous Media. J. Chromatogr. Sci. 2017, 55, 166–173. [Google Scholar] [CrossRef]
- Asman, S.; Mohamad, S.; Sarih, N.M. Influence of polymer morphology on the adsorption behaviors of molecularly imprinted polymer-methacrylic acid functionalized β-cyclodextrin. J. Appl. Polym. Sci. 2015, 132. [Google Scholar] [CrossRef]
- Surikumaran, H.; Mohamad, S.; Sarih, N.M. Molecular Imprinted Polymer of Methacrylic Acid Functionalised β-Cyclodextrin for Selective Removal of 2,4-Dichlorophenol. Int. J. Mol. Sci. 2014, 15, 6111–6136. [Google Scholar] [CrossRef]
- Gao, S.; Liu, Y.; Jiang, J.; Ji, Q.; Fu, Y.; Zhao, L.; Li, C.; Ye, F. Physicochemical properties and fungicidal activity of inclusion complexes of fungicide chlorothalonil with β-cyclodextrin and hydroxypropyl-β-cyclodextrin. J. Mol. Liq. 2019, 293, 111513. [Google Scholar] [CrossRef]
- Sun, Y.; Ding, C.; Lin, Y.; Sun, W.; Liu, H.; Zhu, X.; Dai, Y.; Luo, C. Highly selective and sensitive chemiluminescence biosensor for adenosine detection based on carbon quantum dots catalyzing luminescence released from aptamers functionalized graphene@magnetic β-cyclodextrin polymers. Talanta 2018, 186, 238–247. [Google Scholar] [CrossRef] [PubMed]
- Jacob, S.; Nair, A. Cyclodextrin complexes: Perspective from drug delivery and formulation. Drug Dev. Res. 2018, 79, 201–217. [Google Scholar] [CrossRef] [PubMed]
- Gao, S.; Jiang, J.-Y.; Liu, Y.; Fu, Y.; Zhao, L.-X.; Li, C.; Ye, F. Enhanced Solubility, Stability, and Herbicidal Activity of the Herbicide Diuron by Complex Formation with β-Cyclodextrin. Polymers 2019, 11, 1396. [Google Scholar] [CrossRef] [PubMed]
- Kang, Y.; Duan, W.; Li, Y.; Kang, J.; Xie, J. Molecularly imprinted polymers of allyl-β-cyclodextrin and methacrylic acid for the solid-phase extraction of phthalate. Carbohydr. Polym. 2012, 88, 459–464. [Google Scholar]
- Liu, Y.; Liu, Y.; Liu, Z.; Hu, X.; Xu, Z. β-Cyclodextrin molecularly imprinted solid-phase microextraction coatings for selective recognition of polychlorophenols in water samples. Anal. Bioanal. Chem. 2018, 410, 509–519. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.J.; Cheng, L.; Zhang, L.; Zuo, Y. Graphene oxide based molecularly imprinted polymers modified with β-cyclodextrin for selective extraction of di(2-ethylhexyl) phthalate in environmental waters. J. Sep. Sci. 2019, 42, 1248–1256. [Google Scholar] [CrossRef]
- Xu, Z.F.; Wen, G.; Kuang, D.Z.; Zhang, F.X.; Tang, S.P. Selective separation of deltamethrin by molecularly imprinted polymers using a β-cyclodextrin derivative as the functional monomer. J. Environ. Sci. Health Part B 2013, 48, 336–343. [Google Scholar] [CrossRef]
- Zhu, C.; Zhang, X.; Wang, Q.; Zhang, R.; Wang, T. Thermo/pH dual responsive β -cyclodextrin magnetic microspheres for anti-cancer drug controlled release. J. Control. Release 2015, 213, e21–e22. [Google Scholar] [CrossRef]
- Sedghi, R.; Yassari, M.; Heidari, B. Thermo-responsive molecularly imprinted polymer containing magnetic nanoparticles: Synthesis, characterization and adsorption properties for curcumin. Colloids Surf. B Biointerfaces 2018, 162, 154–162. [Google Scholar] [CrossRef]
- Stöber, W.; Fink, A.; Bohn, E. Controlled growth of monodisperse silica spheres in the micron size range. J. Colloid Interface Sci. 1968, 26, 62–69. [Google Scholar] [CrossRef]
- Gagliardi, M.; Bertero, A.; Bifone, A. Molecularly Imprinted Biodegradable Nanoparticles. Sci. Rep. 2017, 7, 40046. [Google Scholar] [CrossRef] [PubMed]
- Chianella, I.; Guerreiro, A.; Moczko, E.; Caygill, J.S.; Piletska, E.V.; Sansalvador, I.M.P.D.V.; Whitcombe, M.J.; Piletsky, S. Direct Replacement of Antibodies with Molecularly Imprinted Polymer Nanoparticles in ELISA—Development of a Novel Assay for Vancomycin. Anal. Chem. 2013, 85, 8462–8468. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Xue, X.; Luo, Y.; Lin, T.Y.; Zhang, H.; Lac, D.; Xiao, K.; He, Y.; Jia, B.; Lam, K.S.; et al. Sub-100 nm, Long Tumor Retention SN-38-Loaded Photonic Micelles for Tri-modal Cancer Therapy HHS Public Access. J. Control. Release 2017, 261, 297–306. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Wang, J.; Mao, W.; Huang, J.; Wu, X.; Shen, Y.; Sui, M. Novel SN38 conjugate-forming nanoparticles as anticancer prodrug: In vitro and in vivo studies. J. Control. Release 2013, 166, 147–158. [Google Scholar] [CrossRef] [PubMed]
- Parisi, O.I.; Ruffo, M.; Malivindi, R.; Vattimo, A.F.; Pezzi, V.; Puoci, F. Molecularly Imprinted Polymers (MIPs) as Theranostic Systems for Sunitinib Controlled Release and Self-Monitoring in Cancer Therapy. Pharmaceutics 2020, 12, 41. [Google Scholar] [CrossRef] [PubMed]
- Feng, Z.-Q.; Yan, K.; Li, J.; Xu, X.; Yuan, T.; Wang, T.; Zheng, J. Magnetic Janus particles as a multifunctional drug delivery system for paclitaxel in efficient cancer treatment. Mater. Sci. Eng. C 2019, 104, 110001. [Google Scholar] [CrossRef]
- Mo, C.-E.; Chai, M.-H.; Zhang, L.-P.; Ran, R.-X.; Huang, Y.-P.; Liu, Z.-S. Floating molecularly imprinted polymers based on liquid crystalline and polyhedral oligomeric silsesquioxanes for capecitabine sustained release. Int. J. Pharm. 2018, 557, 293–303. [Google Scholar] [CrossRef]
- Boitard, C.; Curcio, A.; Rollet, A.-L.; Wilhelm, C.; Ménager, C.; Griffete, N. Biological Fate of Magnetic Protein-Specific Molecularly Imprinted Polymers: Toxicity and Degradation. ACS Appl. Mater. Interfaces 2019, 11, 35556–35565. [Google Scholar] [CrossRef]
- Vivek, R. Scient Open Access Exploring the World of Science Eco-friendly and Biocompatible Acrylic Resins-A Review. J. Dent. Oral Health 2017, 3, 1–3. [Google Scholar]
- Ai, J.; Biazar, E.; Jafarpour, M.; Montazeri, M.; Majdi, A.; Aminifard, S.; Zafari, M.; Akbari, H.R.; Rad, H.G. Nanotoxicology and nanoparticle safety in biomedical designs. Int. J. Nanomed. 2011, 6, 1117–1127. [Google Scholar] [CrossRef]
- Gu, L.; Fang, R.H.; Sailor, M.J.; Park, J.-H. In Vivo Clearance and Toxicity of Monodisperse Iron Oxide Nanocrystals. ACS Nano 2012, 6, 4947–4954. [Google Scholar] [CrossRef] [PubMed]
- Levy, M.; Luciani, N.; Alloyeau, D.; Elgrabli, D.; Deveaux, V.; Péchoux, C.; Chat, S.; Wang, G.; Vats, N.; Gendron, F.; et al. Long term in vivo biotransformation of iron oxide nanoparticles. Biomaterials 2011, 32, 3988–3999. [Google Scholar] [CrossRef] [PubMed]
- Levy, M.; Lagarde, F.; Maraloiu, V.A.; Blanchin, M.-G.; Gendron, F.; Wilhelm, C.; Gazeau, F. Degradability of superparamagnetic nanoparticles in a model of intracellular environment: Follow-up of magnetic, structural and chemical properties. Nanotechnology 2010, 21, 395103. [Google Scholar] [CrossRef] [PubMed]
- Hoshino, Y.; Koide, H.; Urakami, T.; Kanazawa, H.; Kodama, T.; Oku, N.; Shea, K.J. Recognition, Neutralization, and Clearance of Target Peptides in the Bloodstream of Living Mice by Molecularly Imprinted Polymer Nanoparticles: A Plastic Antibody. J. Am. Chem. Soc. 2010, 132, 6644–6645. [Google Scholar] [CrossRef]
- Cecchini, A.; Raffa, V.; Canfarotta, F.; Signore, G.; Piletsky, S.; Macdonald, M.; Cuschieri, A. In Vivo Recognition of Human Vascular Endothelial Growth Factor by Molecularly Imprinted Polymers. Nano Lett. 2017, 17, 2307–2312. [Google Scholar] [CrossRef]
- Koide, H.; Yoshimatsu, K.; Hoshino, Y.; Lee, S.-H.; Okajima, A.; Ariizumi, S.; Narita, Y.; Yonamine, Y.; Weisman, A.C.; Nishimura, Y.; et al. A polymer nanoparticle with engineered affinity for a vascular endothelial growth factor (VEGF165). Nat. Chem. 2017, 9, 715–722. [Google Scholar] [CrossRef]
- Dmour, I.; Taha, M.O. Natural and semisynthetic polymers in pharmaceutical nanotechnology. In Organic Materials as Smart Nanocarriers for Drug Delivery; William Andrew Publishing: Oxford, UK, 2018; pp. 35–100. [Google Scholar]
- Patra, J.K.; Das, G.; Fraceto, L.F.; Campos, E.V.R.; Rodriguez-Torres, M.D.P.; Acosta-Torres, L.S.; Diaz-Torres, L.; Grillo, R.; Swamy, M.K.; Sharma, S.; et al. Nano based drug delivery systems: Recent developments and future prospects. J. Nanobiotechnol. 2018, 16, 71. [Google Scholar] [CrossRef]
- Kyzas, G.Z.; Lazaridis, N.K.; Bikiaris, D. Optimization of chitosan and β-cyclodextrin molecularly imprinted polymer synthesis for dye adsorption. Carbohydr. Polym. 2013, 91, 198–208. [Google Scholar] [CrossRef]
- Guo, T.; Xia, Y.; Hao, G.; Song, M.; Zhang, B. Adsorptive separation of hemoglobin by molecularly imprinted chitosan beads. Biomaterials 2004, 25, 5905–5912. [Google Scholar] [CrossRef]
- Xu, L.; Huang, Y.-A.; Zhu, Q.; Ye, C. Chitosan in Molecularly-Imprinted Polymers: Current and Future Prospects. Int. J. Mol. Sci. 2015, 16, 18328–18347. [Google Scholar] [CrossRef] [PubMed]
- Zheng, X.-F.; Lian, Q.; Yang, H.; Wang, X. Surface Molecularly Imprinted Polymer of Chitosan Grafted Poly(methyl methacrylate) for 5-Fluorouracil and Controlled Release. Sci. Rep. 2016, 6, 21409. [Google Scholar] [CrossRef] [PubMed]
- Liu, G.; Li, T.; Yang, X.; She, Y.; Wang, M.; Wang, J.; Zhang, M.; Wang, S.; Jin, F.; Jin, M.; et al. Competitive fluorescence assay for specific recognition of atrazine by magnetic molecularly imprinted polymer based on Fe3O4-chitosan. Carbohydr. Polym. 2016, 137, 75–81. [Google Scholar] [CrossRef] [PubMed]
- Luckachan, G.E.; Pillai, C. Biodegradable Polymers-A Review on Recent Trends and Emerging Perspectives. J. Polym. Environ. 2011, 19, 637–676. [Google Scholar] [CrossRef]
- Anderson, F.A. Amended final report on the safety assessment of polyacrylamide and acrylamide residues in cosmetics. Int. J. Toxicol. 2005, 24 (Suppl. 2), 21–50. [Google Scholar]
- Exon, J.H. A review of the toxicology of acrylamide. J. Toxicol. Environ. Health Part B 2006, 9, 397–412. [Google Scholar] [CrossRef]
- Darnell, M.; Sun, J.-Y.; Mehta, M.; Johnson, C.; Arany, P.R.; Suo, Z.; Mooney, D.J. Performance and biocompatibility of extremely tough alginate/polyacrylamide hydrogels. Biomaterials 2013, 34, 8042–8048. [Google Scholar] [CrossRef]
- Lutz, J.-F. Thermo-Switchable Materials Prepared Using the OEGMA-Platform. Adv. Mater. 2011, 23, 2237–2243. [Google Scholar] [CrossRef]
- Yang, H.; Wang, Q.; Chen, W.; Zhao, Y.; Yong, T.; Gan, L.; Xu, H.; Yang, X.-L. Hydrophilicity/Hydrophobicity Reversable and Redox-Sensitive Nanogels for Anticancer Drug Delivery. Mol. Pharm. 2015, 12, 1636–1647. [Google Scholar] [CrossRef]
- Sousa-Hervés, A.; Wedepohl, S.; Calderón, M. One-pot synthesis of doxorubicin-loaded multiresponsive nanogels based on hyperbranched polyglycerol. Chem. Commun. 2015, 51, 5264–5267. [Google Scholar] [CrossRef]
- Cazares-Cortes, E.; Espinosa, A.; Guigner, J.-M.; Michel, A.; Griffete, N.; Wilhelm, C.; Menager, C. Doxorubicin Intracellular Remote Release from Biocompatible Oligo(ethylene glycol) Methyl Ether Methacrylate-Based Magnetic Nanogels Triggered by Magnetic Hyperthermia. ACS Appl. Mater. Interfaces 2017, 9, 25775–25788. [Google Scholar] [CrossRef] [PubMed]
- Vougioukalakis, G.C.; Stamatopoulos, I.; Petzetakis, N.; Raptopoulou, C.P.; Psycharis, V.; Terzis, A.; Kyritsis, P.; Pitsikalis, M.; Hadjichristidis, N. Controlled Vinyl-Type Polymerization of Norbornene with a Nickel (II) Diphosphinoamine/Methylaluminoxane Catalytic System. Polymer 2009, 47, 5241–5250. [Google Scholar] [CrossRef]
- Hasanitabatabaei, M.; Mahdavi, H.; Zandi, S.; Kharrazi, M.J. HPLC analysis of eluted monomers from two composite resins cured with LED and halogen curing lights. J. Biomed. Mater. Res. Part B Appl. Biomater. 2009, 88, 191–196. [Google Scholar] [CrossRef]
- Scorrano, S.; Longo, L.; Vasapollo, G. Molecularly imprinted polymers for solid-phase extraction of 1-methyladenosine from human urine. Anal. Chim. Acta 2010, 659, 167–171. [Google Scholar] [CrossRef]
- Striegler†, S. Carbohydrate Recognition in Cross-Linked Sugar-Templated Poly(acrylates). Macromolecules 2003, 36, 1310–1317. [Google Scholar] [CrossRef]
- Piletsky, S.; Andersson, H.S.; Nicholls, I.A. Combined Hydrophobic and Electrostatic Interaction-Based Recognition in Molecularly Imprinted Polymers. Macromolecules 1999, 32, 633–636. [Google Scholar] [CrossRef]
- Mahmoudi, M.; Sant, S.; Wang, B.; Laurent, S.; Sen, T. Superparamagnetic iron oxide nanoparticles (SPIONs): Development, surface modification and applications in chemotherapy. Adv. Drug Deliv. Rev. 2011, 63, 24–46. [Google Scholar] [CrossRef]
- Jia, C.; Zhang, M.; Zhang, Y.; Ma, Z.-B.; Xiao, N.-N.; He, X.-W.; Li, W.-Y.; Zhang, Y.-K. Preparation of Dual-Template Epitope Imprinted Polymers for Targeted Fluorescence Imaging and Targeted Drug Delivery to Pancreatic Cancer BxPC-3 Cells. ACS Appl. Mater. Interfaces 2019, 11, 32431–32440. [Google Scholar] [CrossRef]
- Aykac, F.S.; Aydogan, C.; Yagci, Y. A robust strategy for the synthesis of miktoarm star copolymers by combination of ROP and photoinitiated free radical polymerization. Eur. Polym. J. 2018, 109, 499–505. [Google Scholar] [CrossRef]
- Bonardi, A.-H.; Zahouily, S.; Dietlin, C.; Graff, B.; Dumur, F.; Ibrahim-Ouali, M.; Gigmes, D.; Lalevée, J. Thermal Initiators as Additives for Photopolymerization of Methacrylates upon Blue Light. Coatings 2020, 10, 478. [Google Scholar] [CrossRef]
- Ensslin, A.S.; Koller, M.F. Convulsions and hypoglycemia due to tetramethyl succinonitrile intoxication in the polyvinyl chloride (PVC) industry: A 4-year follow-up. Int. J. Occup. Med. Environ. Health 2014, 27, 188–195. [Google Scholar] [CrossRef] [PubMed]
- Li, S.-D.; Huang, L. Stealth nanoparticles: High density but sheddable PEG is a key for tumor targeting. J. Control. Release 2010, 145, 178–181. [Google Scholar] [CrossRef] [PubMed]

| Polymerization Reaction | Monomer(s) | NP Size [nm] | Imprinted Polymer Thickness [nm] | Ref. |
|---|---|---|---|---|
| Thermal free radical | AA, MAA, MAM, 4-VP | 500 | n.m. | [50] |
| Thermal free radical | AA, AAM | 57 | 26–31 | [68,77] |
| Self-polymerization | DA | 80–100 | n.m. | [65,79] |
| Low temp. Photo-pol. | MAA | n.m. | n.m. | [3] |
| Thermal free radical | NIPAM | 80 | 40 | [78] |
| Thermal free radical | NIPAM | 152 | 50 | [80] |
| Thermal free radical | NIPAM, β-CD | 130–150 | 90–110 | [135] |
| Thermal free radical | AA, 4-VP | 200–400 | n.m. | [61] |
| Thermal free radical | MAA, ITA | ≥800 | n.m. | [81] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sanadgol, N.; Wackerlig, J. Developments of Smart Drug-Delivery Systems Based on Magnetic Molecularly Imprinted Polymers for Targeted Cancer Therapy: A Short Review. Pharmaceutics 2020, 12, 831. https://doi.org/10.3390/pharmaceutics12090831
Sanadgol N, Wackerlig J. Developments of Smart Drug-Delivery Systems Based on Magnetic Molecularly Imprinted Polymers for Targeted Cancer Therapy: A Short Review. Pharmaceutics. 2020; 12(9):831. https://doi.org/10.3390/pharmaceutics12090831
Chicago/Turabian StyleSanadgol, Nasim, and Judith Wackerlig. 2020. "Developments of Smart Drug-Delivery Systems Based on Magnetic Molecularly Imprinted Polymers for Targeted Cancer Therapy: A Short Review" Pharmaceutics 12, no. 9: 831. https://doi.org/10.3390/pharmaceutics12090831
APA StyleSanadgol, N., & Wackerlig, J. (2020). Developments of Smart Drug-Delivery Systems Based on Magnetic Molecularly Imprinted Polymers for Targeted Cancer Therapy: A Short Review. Pharmaceutics, 12(9), 831. https://doi.org/10.3390/pharmaceutics12090831






