A Tumor-Immune Interaction Model for Synergistic Combinations of Anti PD-L1 and Ionizing Irradiation Treatment
Abstract
1. Introduction
2. Materials and Methods
2.1. Assumptions
2.2. Data Derivation from the Study
2.3. Mathematical Modeling
2.3.1. Modeling Therapy with IR (R) and IT (A)
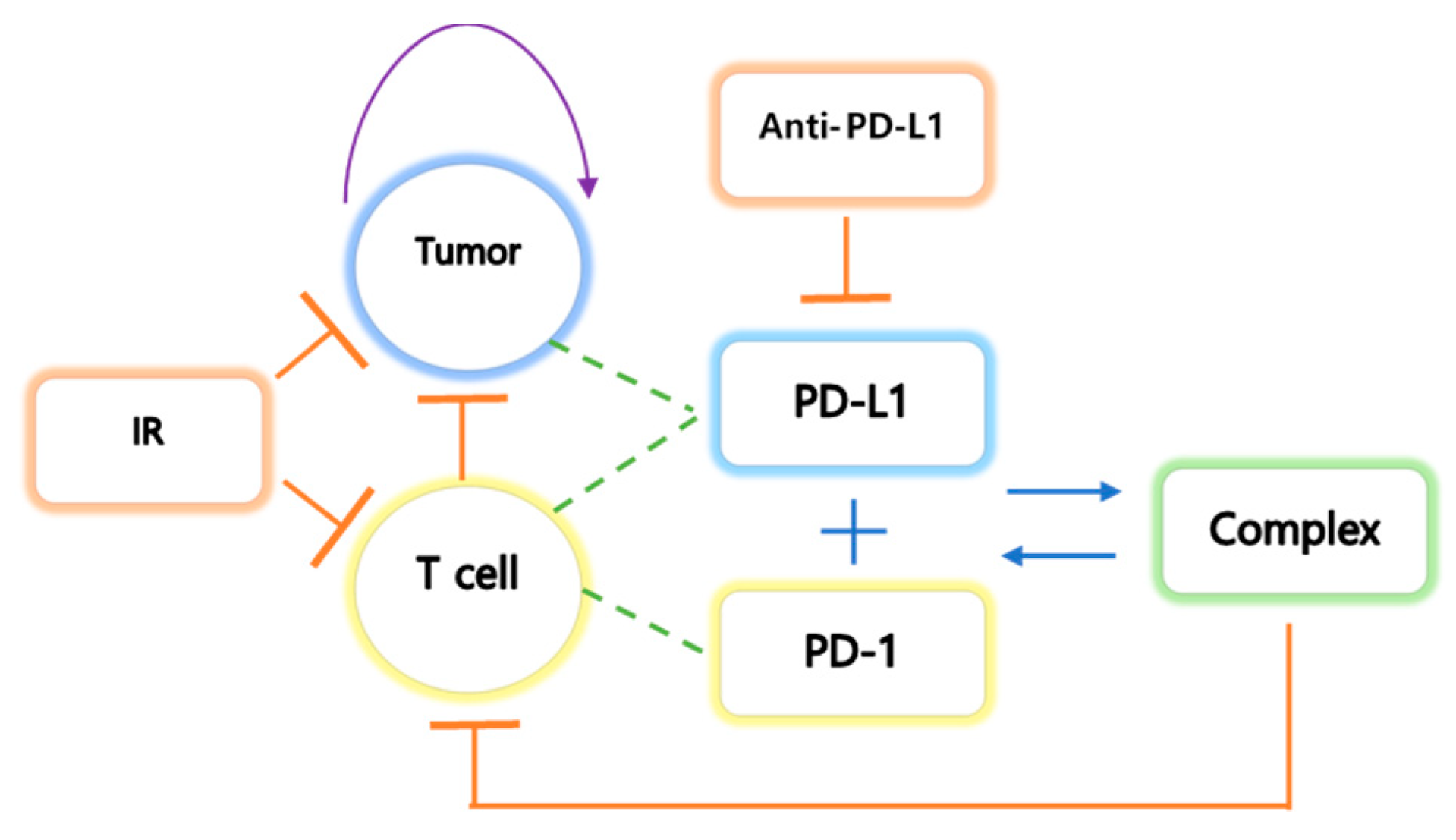
2.3.2. Compartmental Modeling of the Tumor(C), T Cell(T), PD-L1(PL), PD-1(PD), and Complex(S)
2.3.3. Model Reduction: Quasi Steady-State Approximation (QSSA)
3. Results
3.1. Simulation using the Cancer-Immune Model with IR and IT Therapy
3.2. The Expression Levels of PD-1 and PD-L1
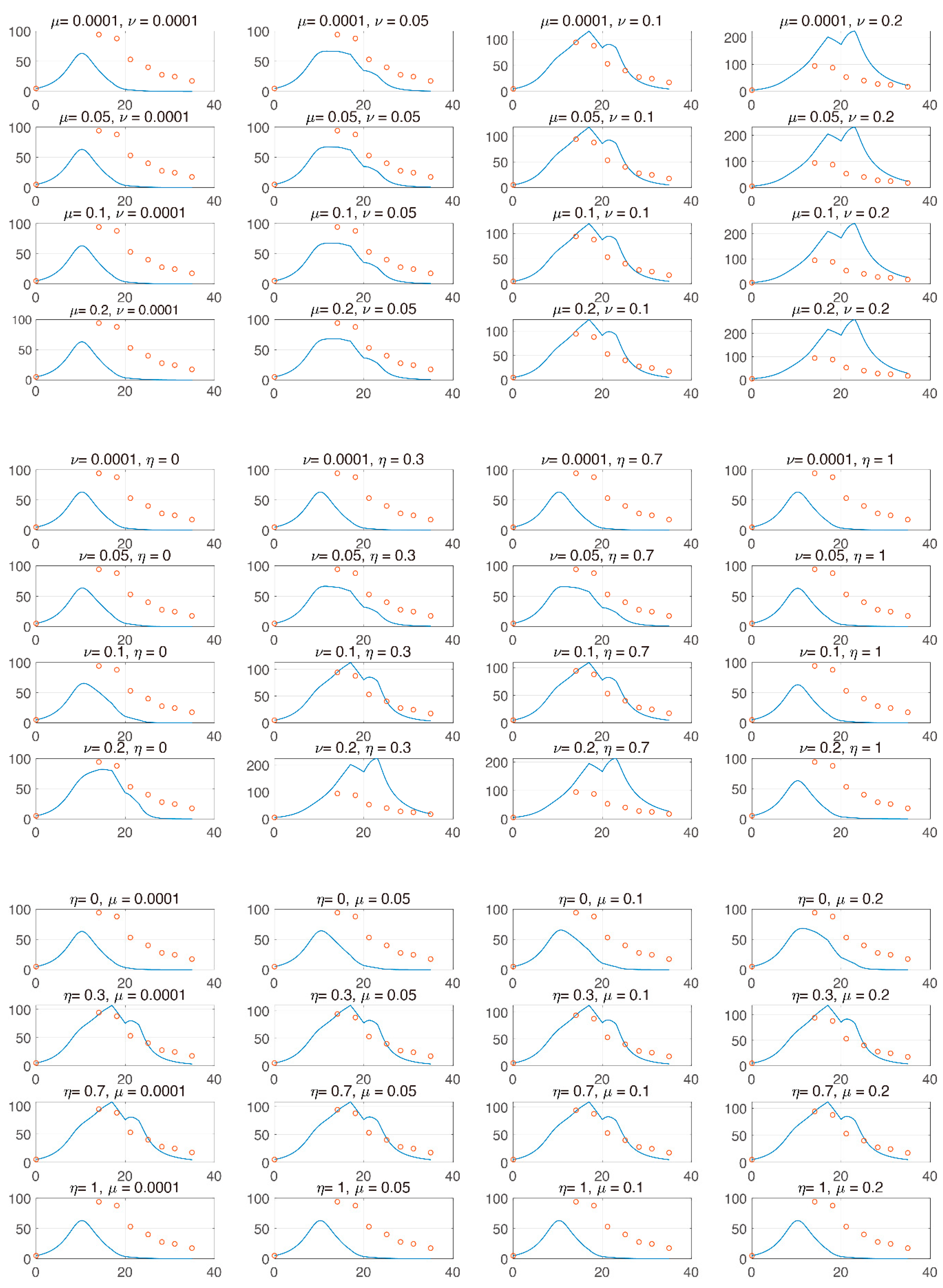
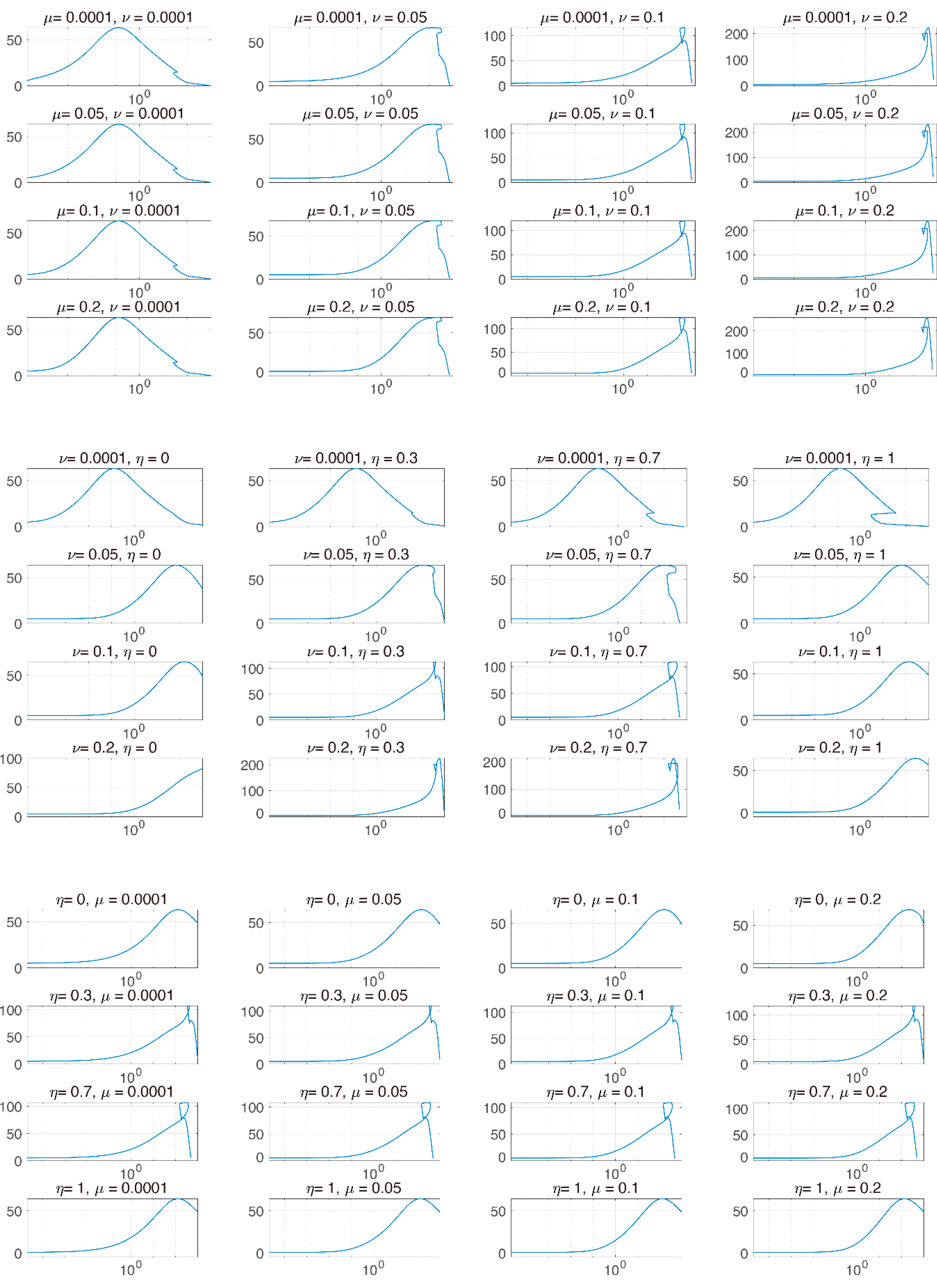
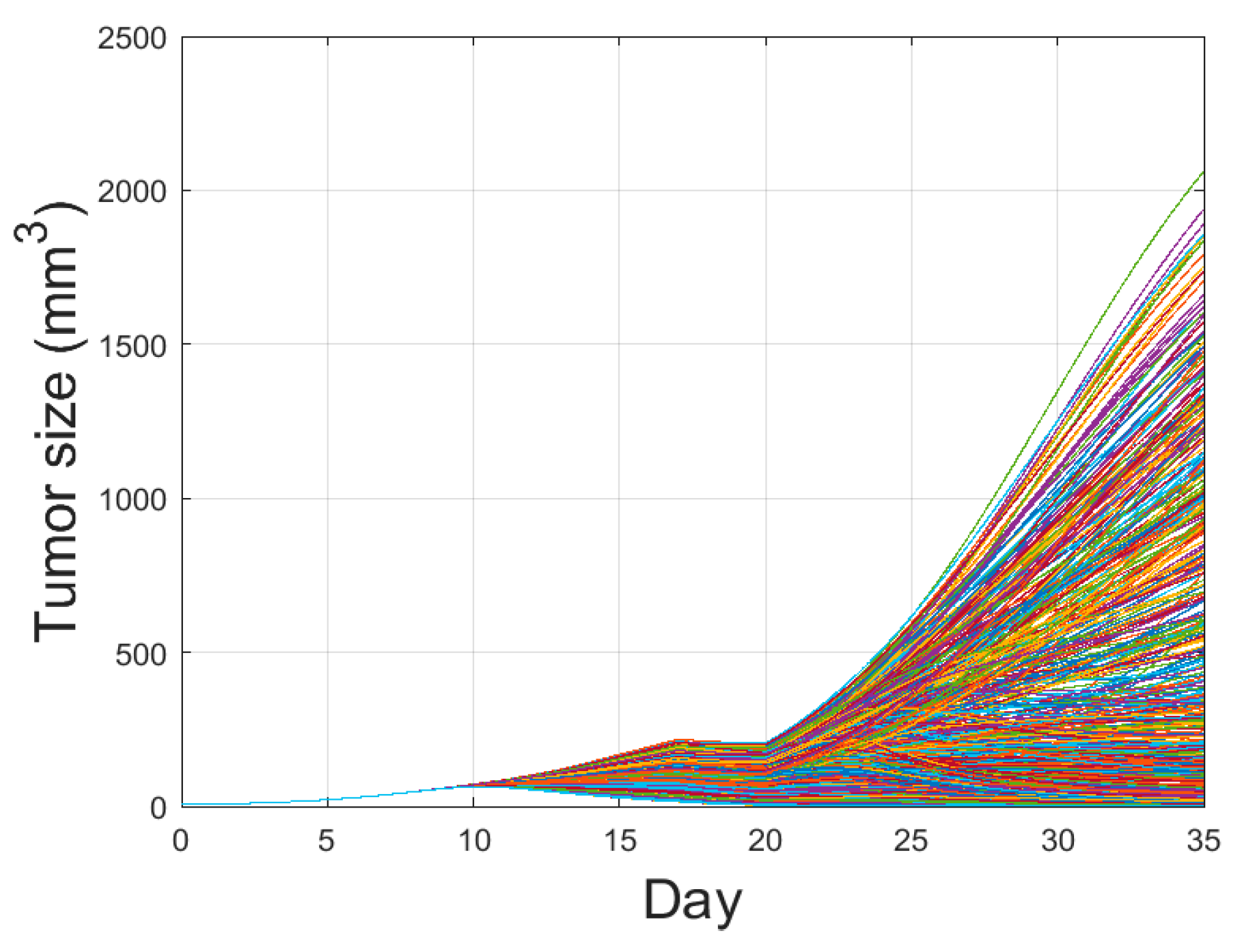
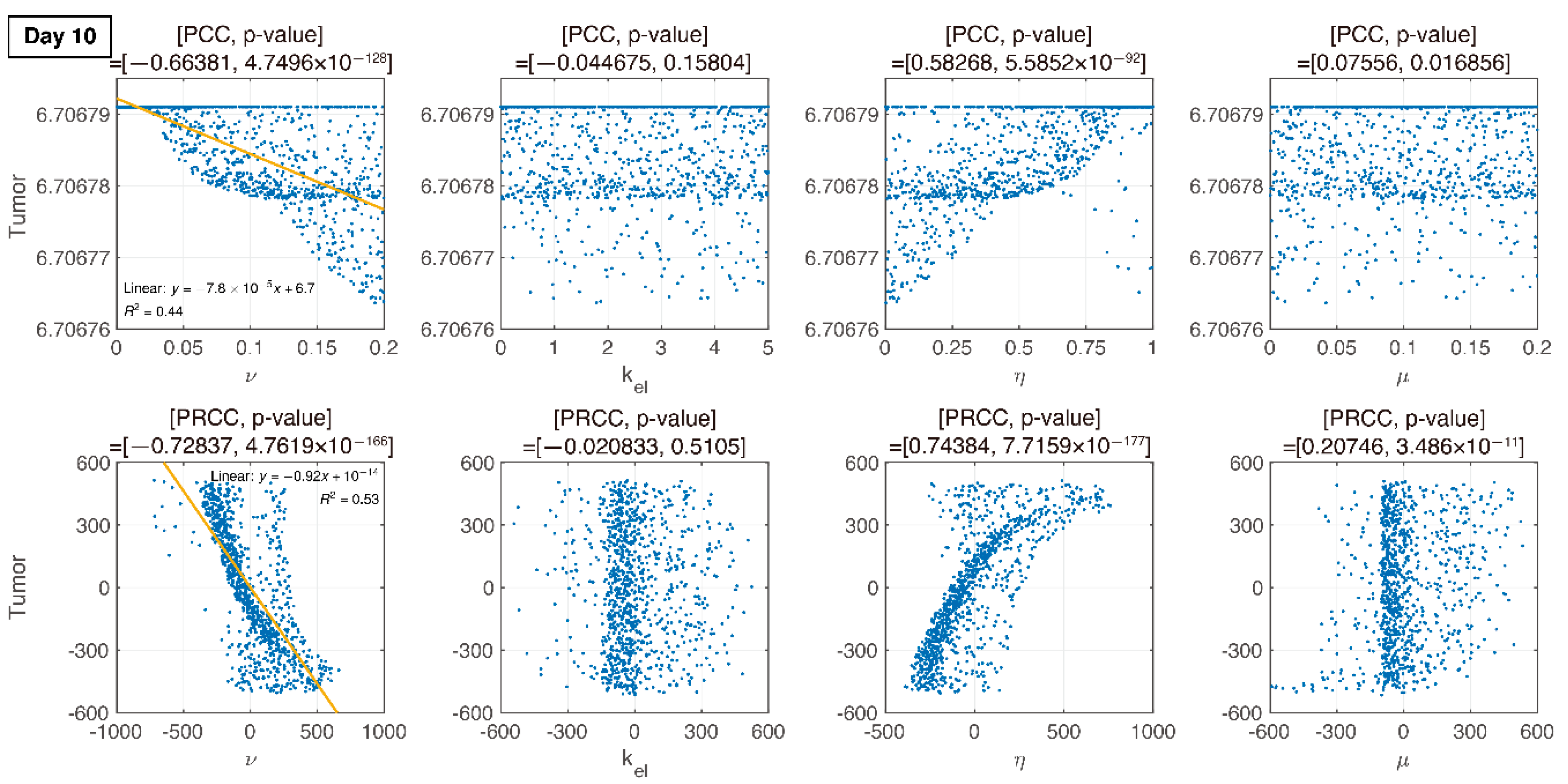
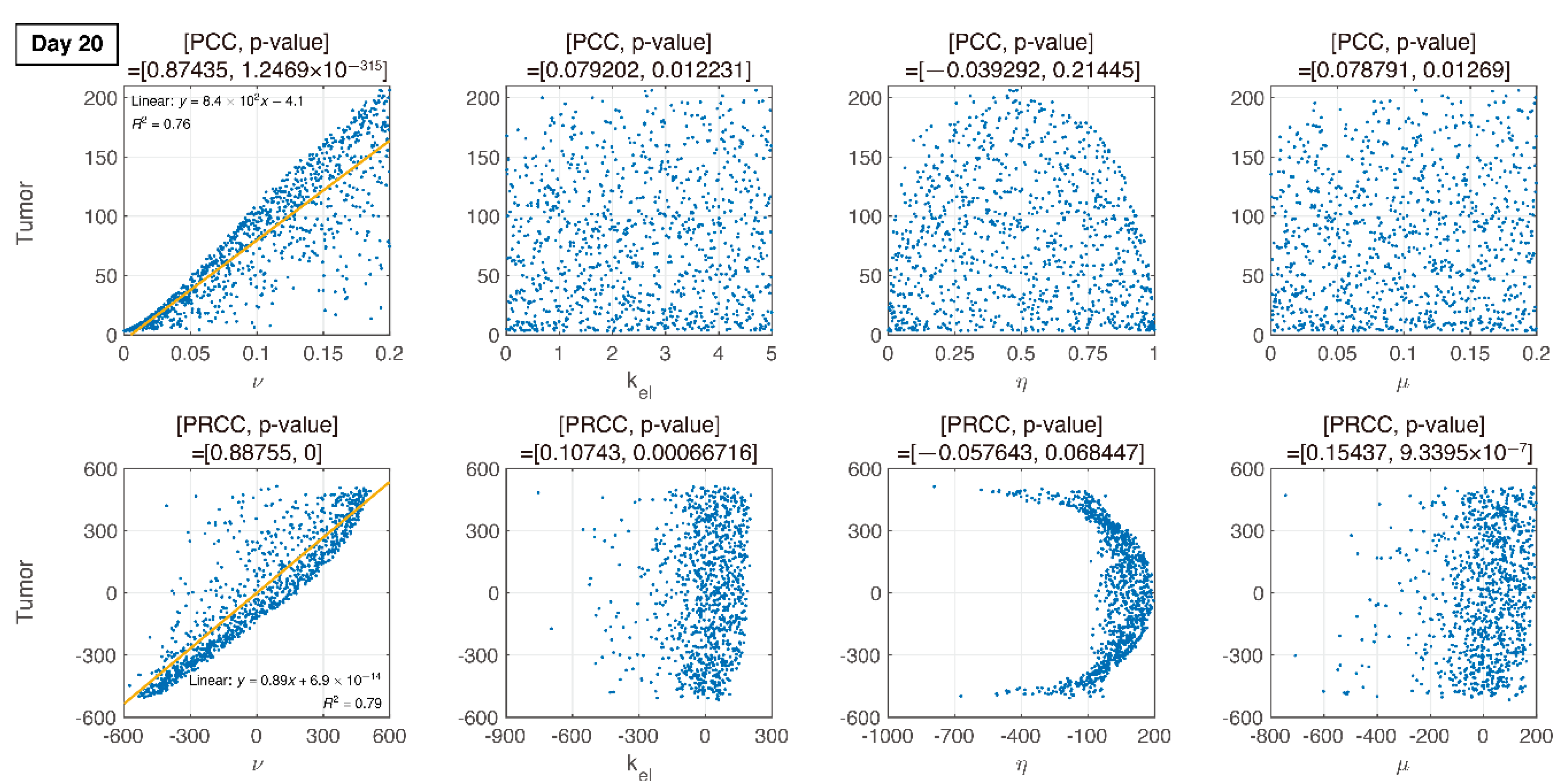
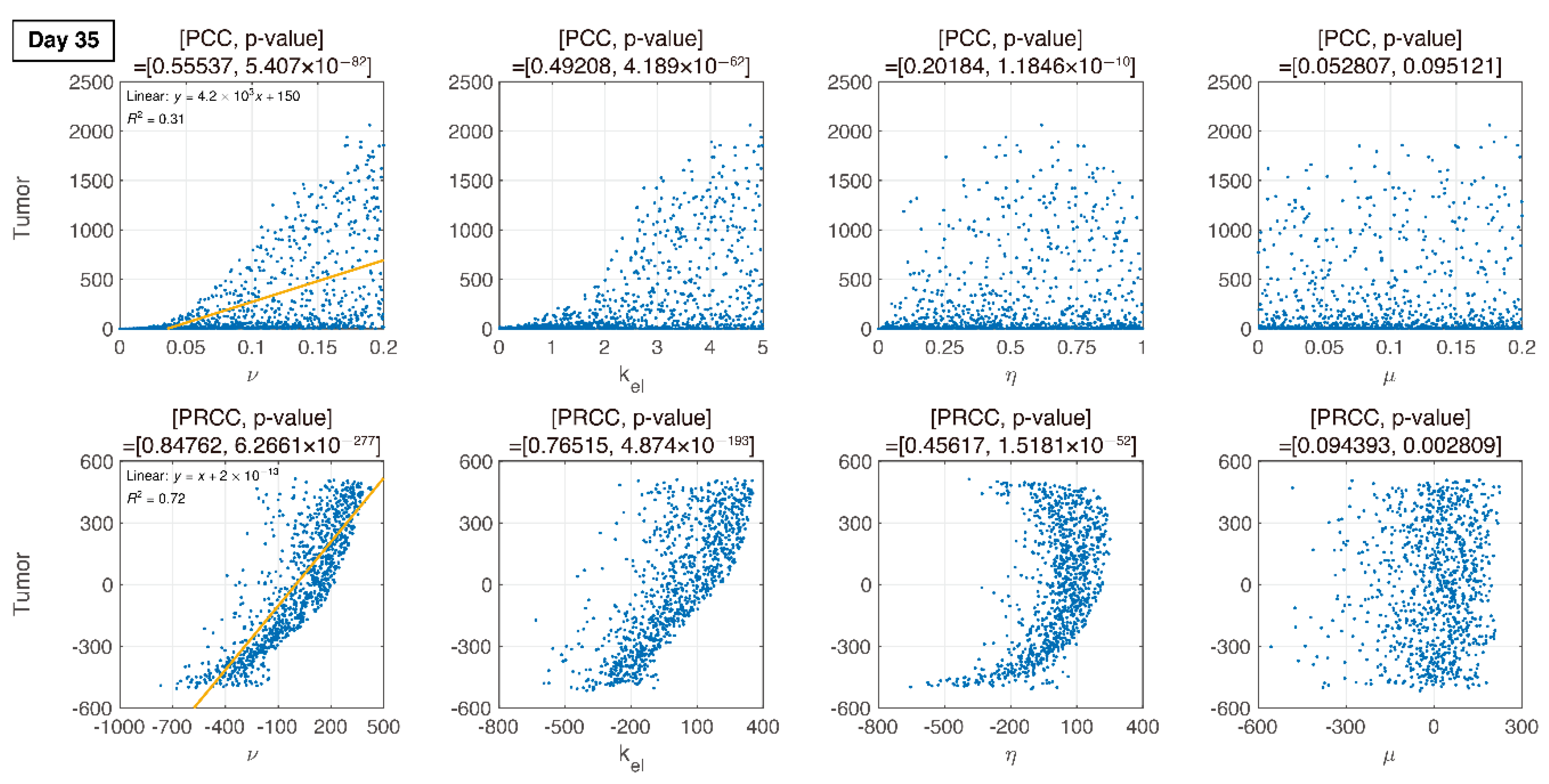
3.3. Global Sensitivity Analysis of the Parameter Space: Latin Hypercube Sampling Partial Rank Correlation Coefficient
3.4. Analysis of Changes in Parameters
4. Discussion and Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Shuptrine, C.W.; Surana, R.; Weiner, L.M. Monoclonal antibodies for the treatment of cancer. Semin. Cancer Biol. 2012, 22, 3–13. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.S.; Mellman, I. Oncology meets immunology: The cancer-immunity cycle. Immunity 2013, 39, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Milberg, O.; Gong, C.; Jafarnejad, M.; Bartelink, I.H.; Wang, B.; Vicini, P.; Narwal, R.; Roskos, L.; Popel, A.S. A QSP Model for Predicting Clinical Responses to Monotherapy, Combination and Sequential Therapy Following CTLA-4, PD-1, and PD-L1 Checkpoint Blockade. Sci. Rep. 2019, 9, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Valentinuzzi, D.; Simončič, U.; Uršič, K.; Vrankar, M.; Turk, M.; Jeraj, R. Predicting tumour response to anti-PD-1 immunotherapy with computational modelling. Phys. Med. Biol. 2019, 64. [Google Scholar] [CrossRef]
- Wei, S.C.; Duffy, C.R.; Allison, J.P. Fundamental Mechanisms of Immune Checkpoint Blockade Therapy. Cancer Discov. 2018, 8, 1069–1086. [Google Scholar] [CrossRef]
- Iwai, Y.; Hamanishi, J.; Chamoto, K.; Honjo, T. Cancer immunotherapies targeting the PD-1 signaling pathway. J. Biomed. Sci. 2017, 24, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Weiner, L.M.; Dhodapkar, M.V.; Ferrone, S. Monoclonal antibodies for cancer immunotherapy. Lancet 2009, 373, 1033–1040. [Google Scholar] [CrossRef]
- Jiang, H.; Hegde, S.; Knolhoff, B.L.; Zhu, Y.; Herndon, J.M.; Meyer, M.A.; Nywening, T.M.; Hawkins, W.G.; Shapiro, I.M.; Weaver, D.T.; et al. Targeting focal adhesion kinase renders pancreatic cancers responsive to checkpoint immunotherapy. Nat. Med. 2016, 22, 851–860. [Google Scholar] [CrossRef]
- Esteva, F.J.; Hubbard-Lucey, V.M.; Tang, J.; Pusztai, L. Immunotherapy and targeted therapy combinations in metastatic breast cancer. Lancet Oncol. 2019, 20, e175–e186. [Google Scholar] [CrossRef]
- dePillis, L.G.; Eladdadi, A.; Radunskaya, A.E. Modeling cancer-immune responses to therapy. J. Pharmacokinet. Pharmacodyn. 2014, 41, 461–478. [Google Scholar] [CrossRef]
- Serre, R.; Benzekry, S.; Padovani, L.; Meille, C.; Andre, N.; Ciccolini, J.; Barlesi, F.; Muracciole, X.; Barbolosi, D. Mathematical modeling of cancer immunotherapy and its synergy with radiotherapy. Cancer Res. 2016, 76, 4931–4940. [Google Scholar] [CrossRef] [PubMed]
- Wei, S.C.; Anang, N.-A.A.S.; Sharma, R.; Andrews, M.C.; Reuben, A.; Levine, J.H.; Cogdill, A.P.; Mancuso, J.J.; Wargo, J.A.; Pe’er, D.; et al. Combination anti--CTLA-4 plus anti--PD-1 checkpoint blockade utilizes cellular mechanisms partially distinct from monotherapies. Proc. Natl. Acad. Sci. USA 2019, 116, 22699–22709. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.L.; Jang, S.R.J. A mathematical model of tumor-immune interactions with an immune checkpoint inhibitor. Appl. Math. Comput. 2019, 362, 124523. [Google Scholar] [CrossRef]
- Deng, L.; Weichselbaum, R.R.; Fu, Y.; Deng, L.; Liang, H.; Burnette, B.; Beckett, M. Irradiation and anti—PD-L1 treatment synergistically promote antitumor immunity in mice Find the latest version: Irradiation and anti—PD-L1 treatment synergistically promote antitumor immunity in mice. J. Clin. Investig. 2014, 124, 687–695. [Google Scholar] [CrossRef]
- Chappell, M.; Chelliah, V.; Cherkaoui, M.; Derks, G.; Dumortier, T.; Evans, N.; Ferrarini, M.; Fornari, C.; Ghazal, P.; Guerriero, M.L.; et al. Mathematical Modelling for Combinations of Immuno-Oncology and Anti-Cancer Therapies. Rep. QSP UK Meet. 2015, 1–15. [Google Scholar] [CrossRef]
- Nikolopoulou, E.; Johnson, L.R.; Harris, D.; Nagy, J.D.; Stites, E.C.; Kuang, Y. Tumour-immune dynamics with an immune checkpoint inhibitor. Lett. Biomath. 2018, 5, S137–S159. [Google Scholar] [CrossRef]
- Lai, X.; Friedman, A. Combination therapy of cancer with cancer vaccine and immune checkpoint inhibitors: A mathematical model. PLoS ONE 2017, 12, e0178479. [Google Scholar] [CrossRef]
- Saltelli, A.; Tarantola, S.; Campolongo, F.; Ratto, M. Sensitivity Analysis in Practice: A Guide to Assessing Scientific Models; Wiley: Chichester, UK, 2004; ISBN 0470870931. [Google Scholar]
- Marino, S.; Hogue, I.B.; Ray, C.J.; Kirschner, D.E. A methodology for performing global uncertainty and sensitivity analysis in systems biology. J. Theor. Biol. 2008, 254, 178–196. [Google Scholar] [CrossRef]
- Conover, W.J. A Distribution-Free Approach to Inducing Rank Correlation Among Input Variables. Commun. Stat. Simul. Comput. 1982, 11, 311–334. [Google Scholar] [CrossRef]
- Davenport, J.M. Rank Correlation Plots for Use with Correlated Input Variables. Commun. Stat. Simul. Comput. 1982, 11, 335–360. [Google Scholar] [CrossRef]
- Gong, J.; Le, T.Q.; Massarelli, E.; Hendifar, A.E.; Tuli, R. Radiation therapy and PD-1/PD-L1 blockade: The clinical development of an evolving anti-cancer combination. J. Immunother. Cancer 2018, 6, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Tang, C.; Welsh, J.W.; De Groot, P.; Massarelli, E.; Chang, J.Y.; Hess, K.R.; Basu, S.; Curran, M.A.; Cabanillas, M.E.; Subbiah, V.; et al. Ipilimumab with stereotactic ablative radiation therapy: Phase i results and immunologic correlates from peripheral T cells. Clin. Cancer Res. 2017, 23, 1388–1396. [Google Scholar] [CrossRef] [PubMed]
- Park, K.J.; Lee, J.L.; Yoon, S.K.; Heo, C.; Park, B.W.; Kim, J.K. Radiomics-based prediction model for outcomes of PD-1/PD-L1 immunotherapy in metastatic urothelial carcinoma. Eur. Radiol. 2020. [Google Scholar] [CrossRef]
- Hamanishi, J.; Mandai, M.; Matsumura, N.; Abiko, K.; Baba, T.; Konishi, I. PD-1/PD-L1 blockade in cancer treatment: Perspectives and issues. Int. J. Clin. Oncol. 2016, 21, 462–473. [Google Scholar] [CrossRef] [PubMed]
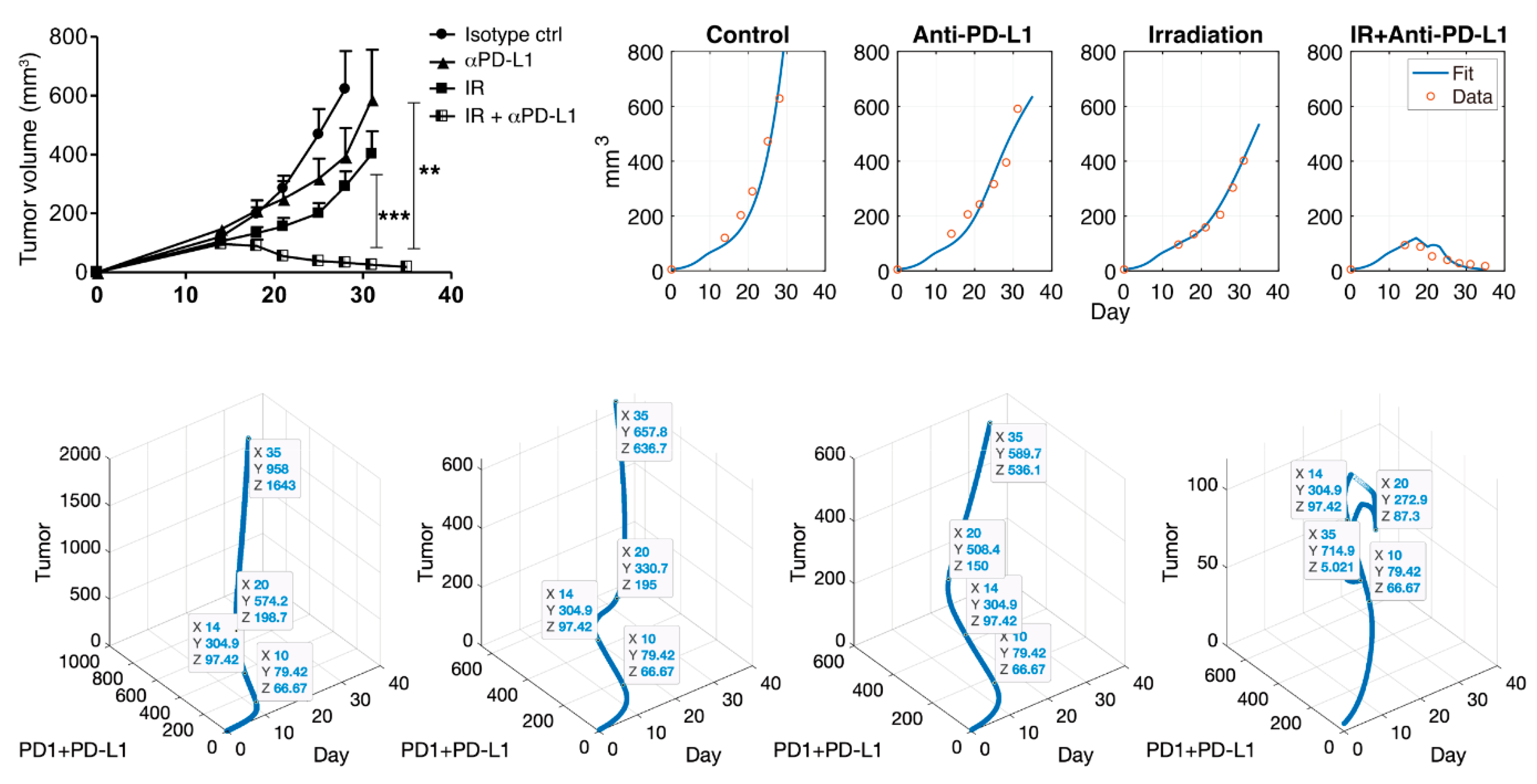

| Control | IR | IT (Anti-PD-L1) | Both | ||||
|---|---|---|---|---|---|---|---|
| 0 | 5 | 0 | 5 | 0 | 5 | 0 | 5 |
| 13.8587 | 120.487 | 14.0761 | 95.8553 | 13.9674 | 135.2759 | 14.0761 | 94.2126 |
| 18.0435 | 202.7944 | 18.0435 | 133.8006 | 18.2065 | 206.0865 | 18.0977 | 87.8069 |
| 21.0326 | 289.9808 | 21.087 | 158.5662 | 21.3043 | 242.3534 | 21.1413 | 53.0777 |
| 25.0543 | 472.4868 | 24.8913 | 204.7183 | 24.9457 | 316.4249 | 25.1087 | 40.0989 |
| 28.0977 | 628.6693 | 28.1522 | 303.4149 | 31.1413 | 591.012 | 28.2065 | 27.7271 |
| - | - | 30.9783 | 402.0936 | - | - | 31.087 | 24.56 |
| - | - | - | - | - | - | 34.9457 | 17.505 |
| Initial/Parameters | Description and Units | Estimated Values |
|---|---|---|
| Tumor initial volume () | 5 | |
| Initial lymphocytic density of CD8+ T cells () | 6 × 10−4 (Assumed) | |
| Initial concentration of PD-L1 ( | 1 × 10−5 (Assumed) | |
| Initial concentration of PD-1 ) | 1 × 10−5 (Assumed) | |
| Initial concentration of Anti-PD-L1 () in tissue | 4 | |
| Concentration of anti-PD-L1 | 0 | |
| Irradiation (G) | ||
| Tumor growth rate ( | 0.29428 | |
| Maximum tumor size () | 3 × 103 | |
| Maximum tumor death rate by T cells ( | 0.53643 | |
| Half maximum density of T cells | 1 × 103 | |
| Maximum tumor death rate by irradiation ( | 0.5 | |
| Half maximum irradiation | 8 | |
| Volume in mice ( | 50 | |
| T cell activation rate by cytokine | 10 | |
| T cell proliferation rate by cytokine | 1 × 102 | |
| Equilibrium constant ( | 1 | |
| Inhibition constant of PD-L1 and PD-1 | 10 | |
| Expression level ratio of PD-L1 to PD-1 in T cells (unitless) | 0.5 | |
| Half maximum inhibition of PD-L1 and PD-1 | 1 × 103 | |
| T cell death rate | 0.1 | |
| Maximum T cell death rate by irradiation ( | 1 | |
| Expression level of PD-L1 on activated T cells ( | 0.1 | |
| Degradation rate of PD-L1 ( | 1 × 10−2 | |
| Degradation rate of PD-1 ( | 1 × 10−2 | |
| Expression level of PD-L1 by tumor vs. T cells | 0.1 | |
| Maximum PD-L1 inhibition rate by anti-PD-L1 ( | 20 | |
| Half-maximum inhibition () | 1 | |
| Intercompartment distribution rate ) | 1.5 × 10−3 | |
| Elimination rate of anti-PD-L1 in tissue ) | 0.1 | |
| Elimination rate of anti-PD-L1 ( | 0.05 | |
| Delay rate with the mean duration (1 | 0.15 | |
| Elimination rate of ionizing irradiation ( | 0.09 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Byun, J.H.; Yoon, I.-S.; Jeong, Y.D.; Kim, S.; Jung, I.H. A Tumor-Immune Interaction Model for Synergistic Combinations of Anti PD-L1 and Ionizing Irradiation Treatment. Pharmaceutics 2020, 12, 830. https://doi.org/10.3390/pharmaceutics12090830
Byun JH, Yoon I-S, Jeong YD, Kim S, Jung IH. A Tumor-Immune Interaction Model for Synergistic Combinations of Anti PD-L1 and Ionizing Irradiation Treatment. Pharmaceutics. 2020; 12(9):830. https://doi.org/10.3390/pharmaceutics12090830
Chicago/Turabian StyleByun, Jong Hyuk, In-Soo Yoon, Yong Dam Jeong, Sungchan Kim, and Il Hyo Jung. 2020. "A Tumor-Immune Interaction Model for Synergistic Combinations of Anti PD-L1 and Ionizing Irradiation Treatment" Pharmaceutics 12, no. 9: 830. https://doi.org/10.3390/pharmaceutics12090830
APA StyleByun, J. H., Yoon, I.-S., Jeong, Y. D., Kim, S., & Jung, I. H. (2020). A Tumor-Immune Interaction Model for Synergistic Combinations of Anti PD-L1 and Ionizing Irradiation Treatment. Pharmaceutics, 12(9), 830. https://doi.org/10.3390/pharmaceutics12090830





