Characterization of Phenolic Acid Antimicrobial and Antioxidant Structure–Property Relationships
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Modified Phenolic Acid (MPA) Synthesis
2.3. Antioxidant Characterization: Hydrogen Peroxide Scavenging
2.4. Antioxidant Characterization: Cell Protection from Hydrogen Peroxide
2.4.1. Cell Culture
2.4.2. PA Cytocompatibility
2.4.3. Hydrogen Peroxide Toxicity
2.4.4. PA Protective Effects
2.5. Antimicrobial Characterization
2.5.1. Preparation of Bacteria Strains
2.5.2. DMSO Bacterial Toxicity
2.5.3. PA IC50 and Log Reduction
2.5.4. MPA Colony Forming Unit Quantification
2.6. Statistical Analysis
3. Results and Discussion
3.1. PA Antioxidant Properties
3.2. PA Cytocompatibility and Protective Antioxidant Effect
3.3. PA Antimicrobial Properties
3.3.1. E. coli (Gram-Negative Bacteria)
3.3.2. Staph. epi. (Native and Drug-Resistant, Gram-Positive Bacteria)
3.3.3. Staph. aureus (Native and Drug-Resistant, Gram-Positive Bacteria)
3.4. Modified Phenolic Acid Characterization
3.4.1. Modified Phenolic Acid Antioxidant Properties
3.4.2. Modified Phenolic Acid Antimicrobial Properties
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Richards, M.J.; Edwards, J.R.; Culver, D.H.; Gaynes, R.P. Nosocomial infections in medical intensive care units in the United States. Crit. Care Med. 1999, 27, 887–892. [Google Scholar] [CrossRef] [PubMed]
- Raad, I.I.; Luna, M.; Khalil, S.M.; Costerton, J.W.; Lam, C.; Bodey, G.P. The relationship between the thrombotic and infectious complications of central venous catheters. JAMA 1994, 271, 1014–1016. [Google Scholar] [CrossRef] [PubMed]
- Darouiche, R.O.; Landon, G.C.; Patti, J.M.; Nguyen, L.L.; Fernau, R.C.; Mcdevitt, D.; Greene, C.; Foster, T.; Klima, M. Role of Staphylococcus aureus surface adhesins in orthopaedic device infections: Are results model-dependent? J. Med. Microbiol. 1997, 46, 75–79. [Google Scholar] [CrossRef] [PubMed]
- Esposito, M. Differential diagnosis and treatment strategies for biologic complications and failing oral implants: A review of the literature. Int. J. Oral Maxillofac. Implant. 1999, 14, 473–490. [Google Scholar]
- Ventola, C.L. The antibiotic resistance crisis: Part 1: Causes and threats. P T Peer Rev. J. Formul. Manag. 2015, 40, 277–283. [Google Scholar]
- O’Neill, J. Antimicrobial Resistance: Tackling a Crisis for the Health and Wealth of Nations. Rev. Antimicrob. Resist. 2016, 1, 1–16. [Google Scholar]
- Efem, S.E. Clinical observations on the wound healing properties of honey. Br. J. Surg. 1988, 75, 679–681. [Google Scholar] [CrossRef]
- Molan, P.C.; Betts, J.A. Clinical usage of honey as a wound dressing: An update. J. Wound Care 2004, 13, 353–356. [Google Scholar] [CrossRef]
- Wahdan, H.A. Causes of the antimicrobial activity of honey. Infection 1998, 26, 26–31. [Google Scholar] [CrossRef]
- Merkl, R.; Hrádková, I.; Filip, V.; Šmidrkal, J. Antimicrobial and antioxidant properties of phenolic acids alkyl esters. Czech J. Food Sci. 2010, 28, 275–279. [Google Scholar] [CrossRef]
- Nascimento, G.G.F.; Locatelli, J.; Freitas, P.C.; Silva, G.L. Antibacterial activity of plant extracts and phytochemicals on antibiotic-resistant bacteria. Braz. J. Microbiol. 2000, 31, 247–256. [Google Scholar] [CrossRef]
- Monroe, M.B.B.; Easley, A.D.; Grant, K.; Fletcher, G.K.; Boyer, C.; Maitland, D.J. Multifunctional shape memory polymer foams with bio-inspired antimicrobials. ChemPhysChem 2017, 19, 1999–2008. [Google Scholar] [CrossRef] [PubMed]
- Dietrich, H.; Pour Nikfardjam, M.S. Influence of Phenolic Compounds and Tannins on Wine-Related Microorganisms. In Biology of Microorganisms on Grapes, in Must and in Wine; König, H., Unden, G., Fröhlich, J., Eds.; Springer International Publishing: Cham, Switzerland, 2017; pp. 421–454. ISBN 978-3-319-60021-5. [Google Scholar]
- Borges, A.; Ferreira, C.; Saavedra, M.J.; Simões, M. Antibacterial Activity and Mode of Action of Ferulic and Gallic Acids Against Pathogenic Bacteria. Microb. Drug Resist. 2013, 19, 256–265. [Google Scholar] [CrossRef] [PubMed]
- Sroka, Z.; Cisowski, W. Hydrogen peroxide scavenging, antioxidant and anti-radical activity of some phenolic acids. Food Chem. Toxicol. 2003, 41, 753–758. [Google Scholar] [CrossRef]
- Natella, F.; Nardini, M.; Di Felice, M.; Scaccini, C. Benzoic and cinnamic acid derivatives as antioxidants: Structure-activity relation. J. Agric. Food Chem. 1999, 47, 1453–1459. [Google Scholar] [CrossRef]
- Oldham, K.M.; Wise, S.R.; Chen, L.; Stacewicz-Sapuntzakis, M.; Burns, J.; Bowen, P.E. A longitudinal evaluation of oxidative stress in trauma patients. J. Parenter. Enter. Nutr. 2002, 26, 189–197. [Google Scholar] [CrossRef]
- Majtan, J. Honey: An immunomodulator in wound healing. Wound Repair Regen. 2014, 22, 187–192. [Google Scholar] [CrossRef]
- Goode, H.F.; Cowley, H.C.; Walker, B.E.; Howdle, P.D.; Webster, N.R. Decreased antioxidant status and increased lipid peroxidation in patients with septic shock and secondary organ dysfunction. Crit. Care Med. 1995, 23, 645–651. [Google Scholar] [CrossRef]
- Takeda, K.; Shimada, Y.; Amano, M.; Sakai, T.; Okada, T.; Yoshiya, I. Plasma lipid peroxides and alpha-tocopherol in critically ill patients. Crit. Care Med. 1984, 12, 957–959. [Google Scholar] [CrossRef]
- Boosalis, M.G.; Snowdon, D.A.; Tully, C.L.; Gross, M.D. Acute phase response and plasma carotenoid concentrations in older women: Findings from the nun study. Nutrition 1996, 12, 475–478. [Google Scholar] [CrossRef]
- Chatterjee, N.S.; Panda, S.K.; Navitha, M.; Asha, K.K.; Anandan, R.; Mathew, S. Vanillic acid and coumaric acid grafted chitosan derivatives: Improved grafting ratio and potential application in functional food. J. Food Sci. Technol. 2015, 52, 7153–7162. [Google Scholar] [CrossRef]
- Campos, F.M.; Couto, J.A.; Figueiredo, A.R.; Tóth, I.V.; Rangel, A.O.S.S.; Hogg, T.A. Cell membrane damage induced by phenolic acids on wine lactic acid bacteria. Int. J. Food Microbiol. 2009, 135, 144–151. [Google Scholar] [CrossRef] [PubMed]
- Salaheen, S.; Peng, M.; Joo, J.; Teramoto, H.; Biswas, D. Eradication and sensitization of methicillin resistant Staphylococcus aureus to methicillin with bioactive extracts of berry pomace. Front. Microbiol. 2017, 8, 253. [Google Scholar] [CrossRef] [PubMed]
- Alves, M.J.; Ferreira, I.C.F.R.; Froufe, H.J.C.; Abreu, R.M.V.; Martins, A.; Pintado, M. Antimicrobial activity of phenolic compounds identified in wild mushrooms, SAR analysis and docking studies. J. Appl. Microbiol. 2013, 115, 346–357. [Google Scholar] [CrossRef]
- Engels, C.; Schieber, A.; Gänzle, M.G. Sinapic acid derivatives in defatted Oriental mustard (Brassica juncea L.) seed meal extracts using UHPLC-DAD-ESI-MS n and identification of compounds with antibacterial activity. Eur. Food Res. Technol. 2012, 234, 535–542. [Google Scholar] [CrossRef]
- Park, E.-S.; Moon, W.-S.; Song, M.-J.; Kim, M.-N.; Chung, K.-H.; Yoon, J.-S. Antimicrobial activity of phenol and benzoic acid derivatives. Int. Biodeterior. Biodegrad. 2001, 47, 209–214. [Google Scholar] [CrossRef]
- Kuete, V.; Nana, F.; Ngameni, B.; Mbaveng, A.T.; Keumedjio, F.; Ngadjui, B.T. Antimicrobial activity of the crude extract, fractions and compounds from stem bark of Ficus ovata (Moraceae). J. Ethnopharmacol. 2009, 124, 556–561. [Google Scholar] [CrossRef]
- Varì, R.; D’Archivio, M.; Filesi, C.; Carotenuto, S.; Scazzocchio, B.; Santangelo, C.; Giovannini, C.; Masella, R. Protocatechuic acid induces antioxidant/detoxifying enzyme expression through JNK-mediated Nrf2 activation in murine macrophages. J. Nutr. Biochem. 2011, 22, 409–417. [Google Scholar] [CrossRef]
- Bahorun, T.; Gressier, B.; Trotin, F.; Brunet, C.; Dine, T.; Luyckx, M.; Vasseur, J.; Cazin, M.; Cazin, J.C.; Pinkas, M. Oxygen species scavenging activity of phenolic extracts from hawthorn fresh plant organs and pharmaceutical preparations. Arzneimittelforschung 1996, 46, 1086–1089. [Google Scholar]
- Tsutomu, M.K. In ISO/TC194 Biological evaluation of medical devices. Environ. Mutagen Res. 2005, 27, 129–131. [Google Scholar]
- Babich, H.; Sedletcaia, A.; Kenigsberg, B. In Vitro Cytotoxicity of Protocatechuic Acid to Cultured Human Cells from Oral Tissue: Involvement in Oxidative Stress. Pharmacol. Toxicol. 2002, 91, 245–253. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Xie, S.; Ahmed, S.; Wang, F.; Gu, Y.; Zhang, C.; Chai, X.; Wu, Y.; Cai, J.; Cheng, G. Antimicrobial Activity and Resistance: Influencing Factors. Front. Pharmacol. 2017, 8, 364. [Google Scholar] [CrossRef] [PubMed]
- Nićiforović, N.; Abramovič, H. Sinapic Acid and Its Derivatives: Natural Sources and Bioactivity. Compr. Rev. Food Sci. Food Saf. 2014, 13, 34–51. [Google Scholar] [CrossRef]
- Estevinho, L.; Pereira, A.P.; Moreira, L.; Dias, L.G.; Pereira, E. Antioxidant and antimicrobial effects of phenolic compounds extracts of Northeast Portugal honey. Food Chem. Toxicol. 2008, 46, 3774–3779. [Google Scholar] [CrossRef]
- Briandet, R.; Herry, J.-M.; Bellon-Fontaine, M.-N. Determination of the van der Waals, electron donor and electron acceptor surface tension components of static Gram-positive microbial biofilms. Colloids Surf. B Biointerfaces 2001, 21, 299–310. [Google Scholar] [CrossRef]
- Christenson, E.M.; Anderson, J.M.; Hiltner, A. Antioxidant inhibition of poly(carbonate urethane)in vivo biodegradation. J. Biomed. Mater. Res. Part A 2006, 76A, 480–490. [Google Scholar] [CrossRef]
- Domínguez-Robles, J.; Martin, N.K.; Fong, M.L.; Stewart, S.A.; Irwin, N.J.; Rial-Hermida, M.I.; Donnelly, R.F.; Larrañeta, E. Antioxidant PLA Composites Containing Lignin for 3D Printing Applications: A Potential Material for Healthcare Applications. Pharmaceutics 2019, 11, 165. [Google Scholar] [CrossRef]
- Dunnill, C.; Patton, T.; Brennan, J.; Barrett, J.; Dryden, M.; Cooke, J.; Leaper, D.; Georgopoulos, N.T. Reactive oxygen species (ROS) and wound healing: The functional role of ROS and emerging ROS-modulating technologies for augmentation of the healing process. Int. Wound J. 2017, 14, 89–96. [Google Scholar] [CrossRef]
- Narasimhan, B.; Belsare, D.; Pharande, D.; Mourya, V.; Dhake, A. Esters, amides and substituted derivatives of cinnamic acid: Synthesis, antimicrobial activity and QSAR investigations. Eur. J. Med. Chem. 2004, 39, 827–834. [Google Scholar] [CrossRef]
- Khatkar, A.; Nanda, A.; Kumar, P.; Narasimhan, B. Synthesis, antimicrobial evaluation and QSAR studies of P-coumaric acid derivatives. Arab. J. Chem. 2017, 10, S3804–S3815. [Google Scholar] [CrossRef]

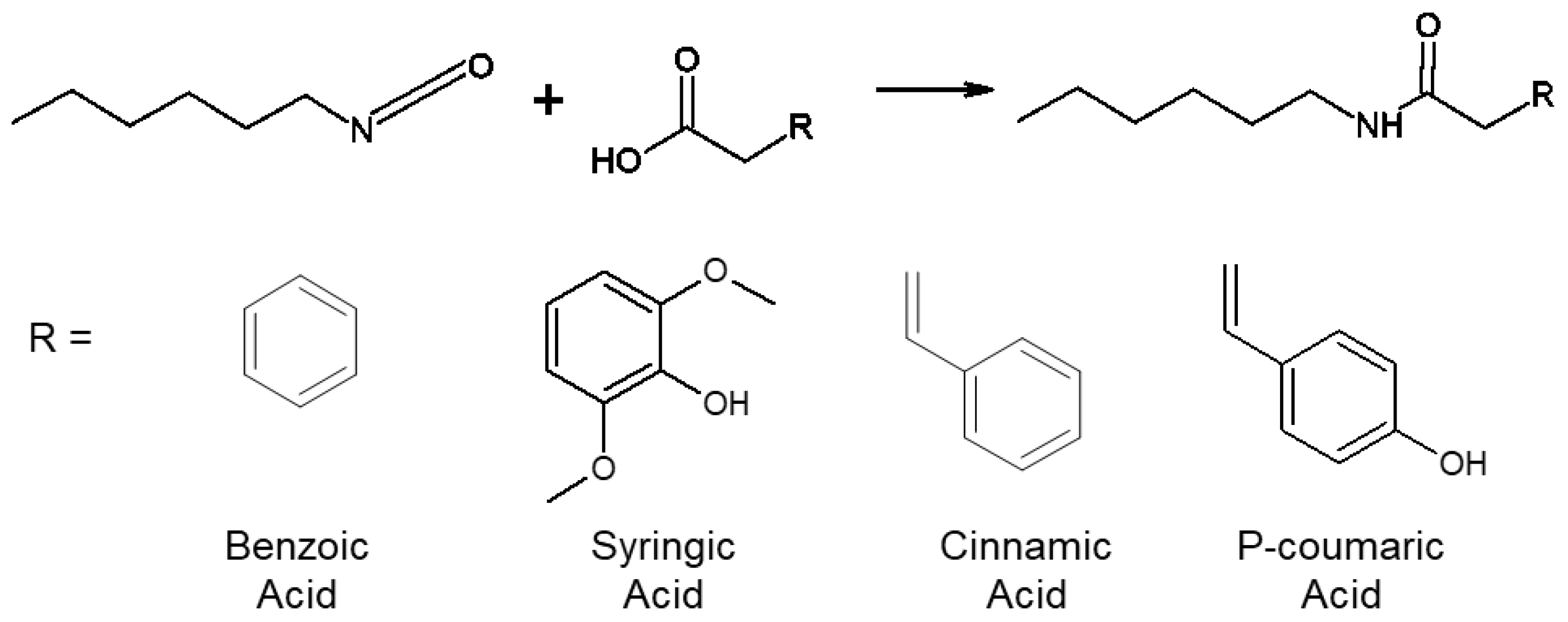


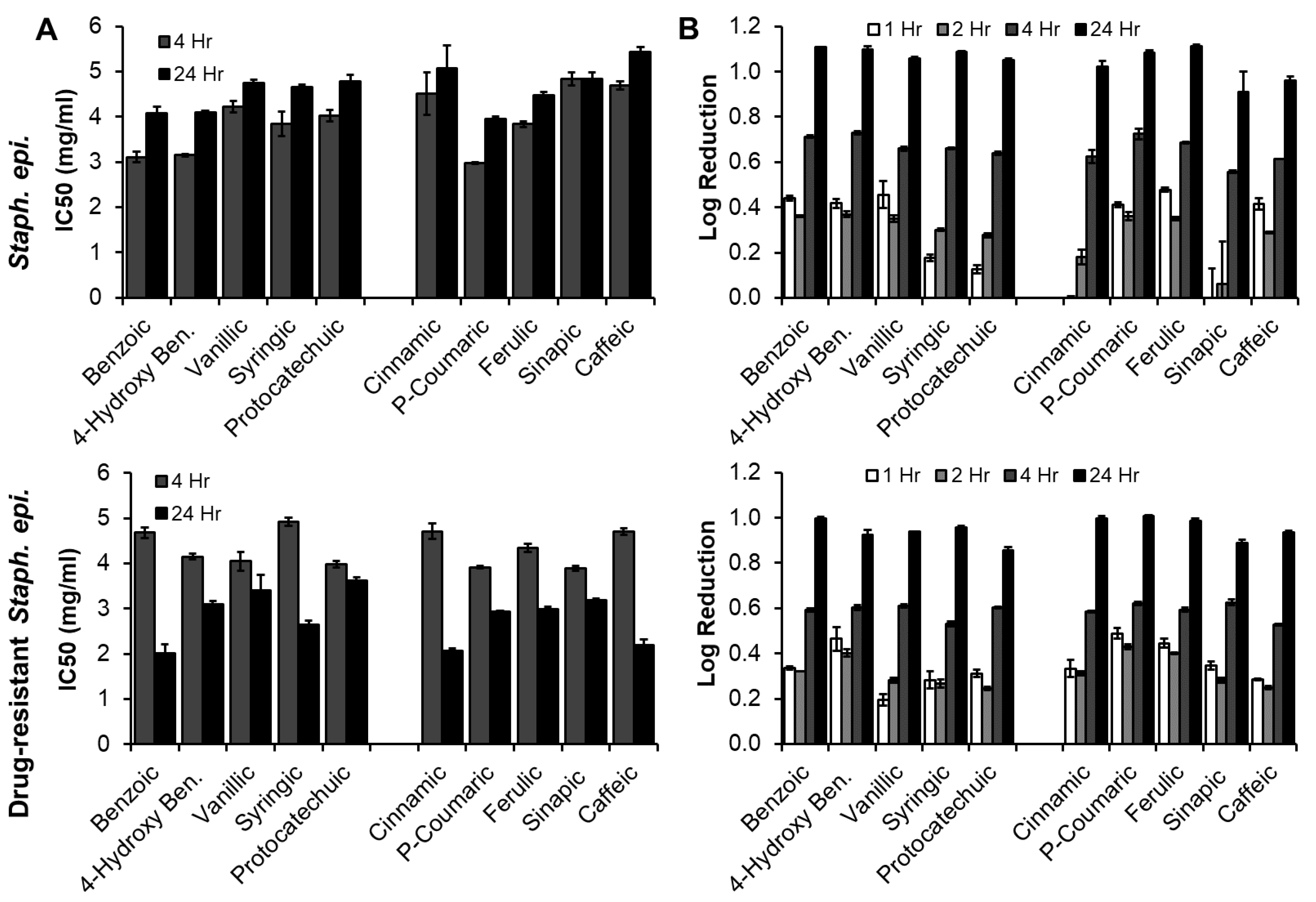
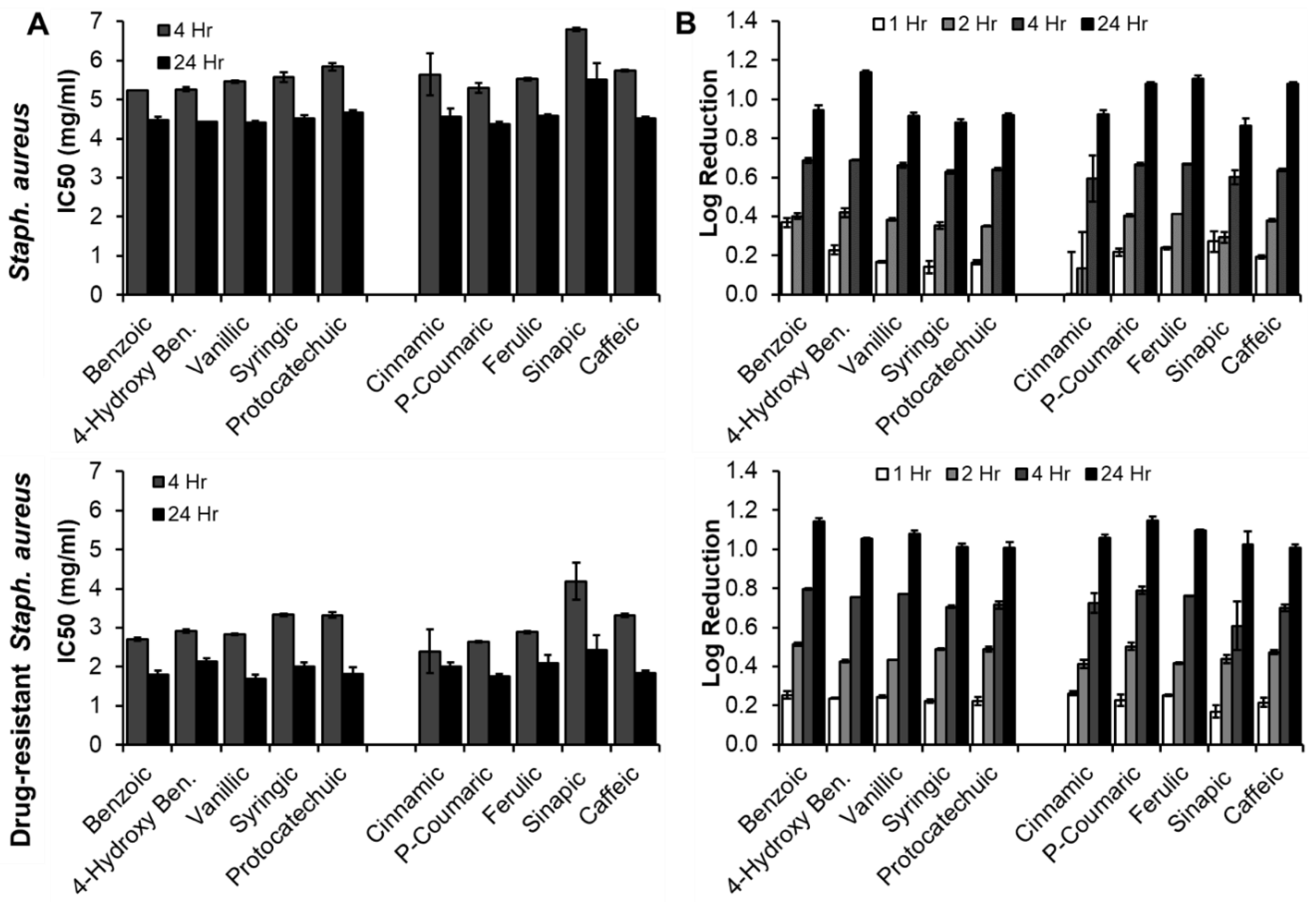

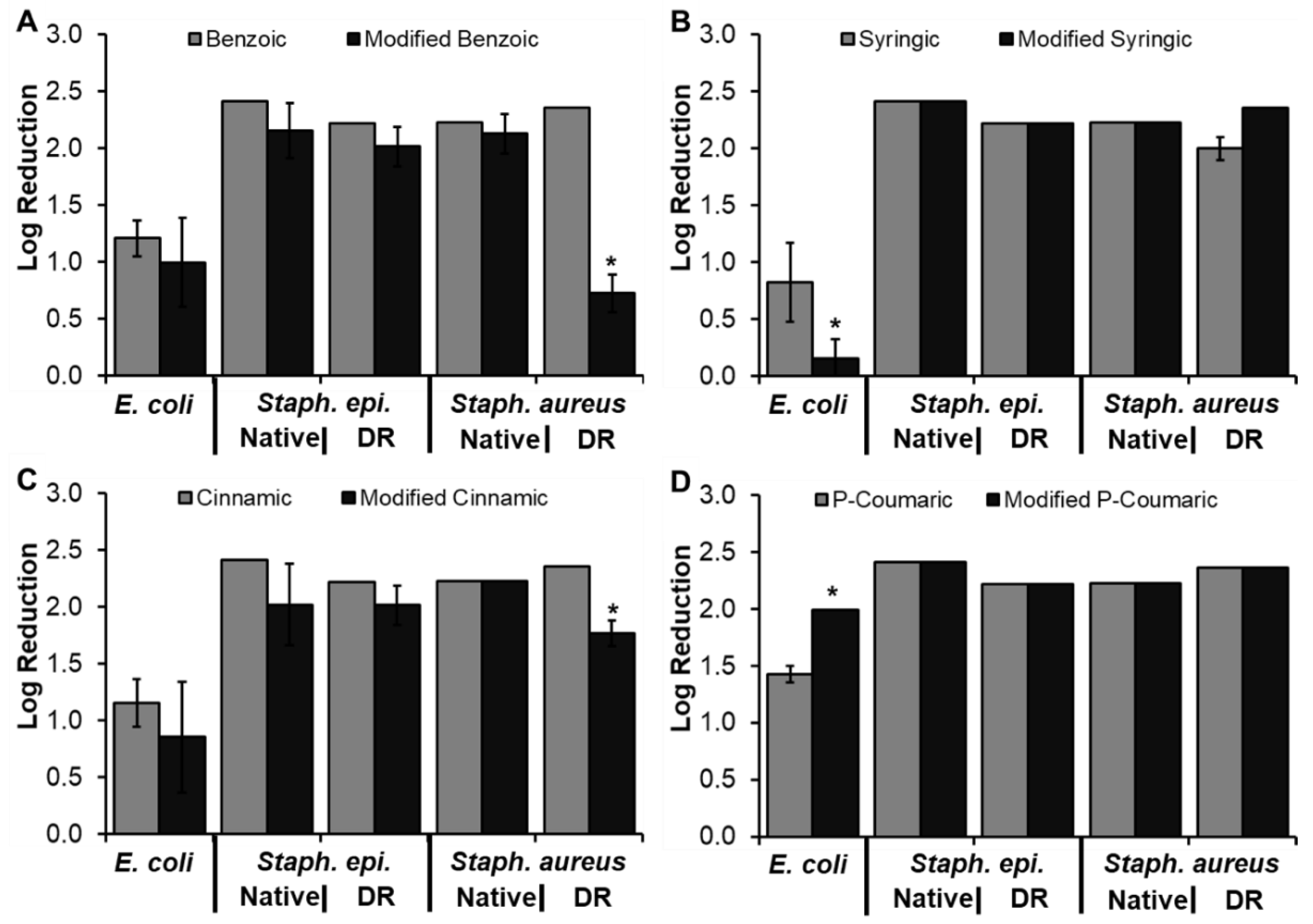
| Phenolic Acid | Antimicrobial Properties | Antioxidant Properties | |
|---|---|---|---|
| Gram Positive | Gram Negative | ||
| Cinnamic acid | Staphylococcus epidermidis (Staph. epi.) [12] Staphylococcus aureus (Staph. aureus); Multidrug-resistant Staph. aureus [11] | Escherichia coli (E. coli) [12] Multidrug-resistant E. coli [11] | N/A |
| P-coumaric acid | Staph. aureus [22] Methicillin-resistant Staph. aureus [24] | E. coli [25] Not effective against E. coli [22] | Relatively ineffective [16] |
| Ferulic acid | Staph. aureus [22] | E. coli [10] Not effective against E. coli [22] | 0.9X trolox efficacy [16] |
| Sinapic acid | Staph. aureus [26] | E. coli [26] | 6X trolox efficacy [16] |
| Caffeic acid | Methicillin-resistant Staph. aureus [24] | E. coli [10] | Scavenged 59% of H2O2 [15] 4X trolox efficacy [16] |
| Benzoic acid | Not effective against Staph. aureus [11] | Multidrug-resistant E. coli [11] | N/A |
| 4-hydroxy-benzoic acid | Staph. aureus [27] | E. coli [10] | Relatively ineffective [16] |
| Vanillic acid | Staph. aureus [22] Methicillin-resistant Staph. aureus [24] | E. coli [10] Not effective against E. coli [22] | Relatively ineffective [16] |
| Syringic acid | Some efficacy against E. coli [25] | 1.3X trolox efficacy [16] | |
| Protocatechuic acid | Methicillin-resistant Staph. aureus [24,28] | E. coli [10,28] | Scavenged 55% of H2O2 [15] Inhibits macrophage production of O2 and H2O2 [29] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, J.; Du, C.; Beaman, H.T.; Monroe, M.B.B. Characterization of Phenolic Acid Antimicrobial and Antioxidant Structure–Property Relationships. Pharmaceutics 2020, 12, 419. https://doi.org/10.3390/pharmaceutics12050419
Liu J, Du C, Beaman HT, Monroe MBB. Characterization of Phenolic Acid Antimicrobial and Antioxidant Structure–Property Relationships. Pharmaceutics. 2020; 12(5):419. https://doi.org/10.3390/pharmaceutics12050419
Chicago/Turabian StyleLiu, Jingyi, Changling Du, Henry T. Beaman, and Mary Beth B. Monroe. 2020. "Characterization of Phenolic Acid Antimicrobial and Antioxidant Structure–Property Relationships" Pharmaceutics 12, no. 5: 419. https://doi.org/10.3390/pharmaceutics12050419
APA StyleLiu, J., Du, C., Beaman, H. T., & Monroe, M. B. B. (2020). Characterization of Phenolic Acid Antimicrobial and Antioxidant Structure–Property Relationships. Pharmaceutics, 12(5), 419. https://doi.org/10.3390/pharmaceutics12050419





