Polymeric Nanocarriers of Drug Delivery Systems in Cancer Therapy
Abstract
1. Introduction
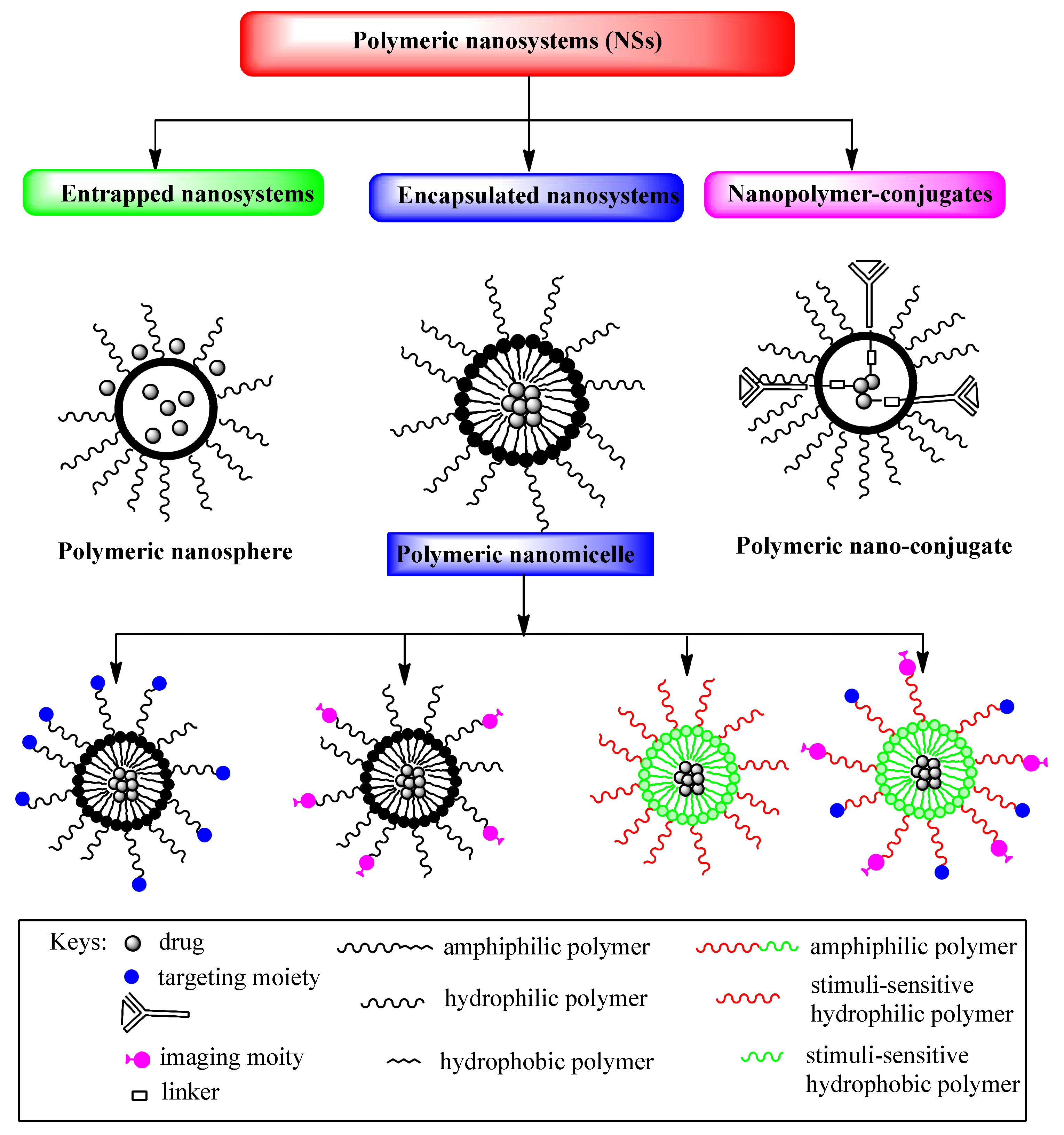
2. Polymeric Nanoparticles (NPs)
3. Amphiphilic Block Copolymers as Carriers in Drug Delivery Systems
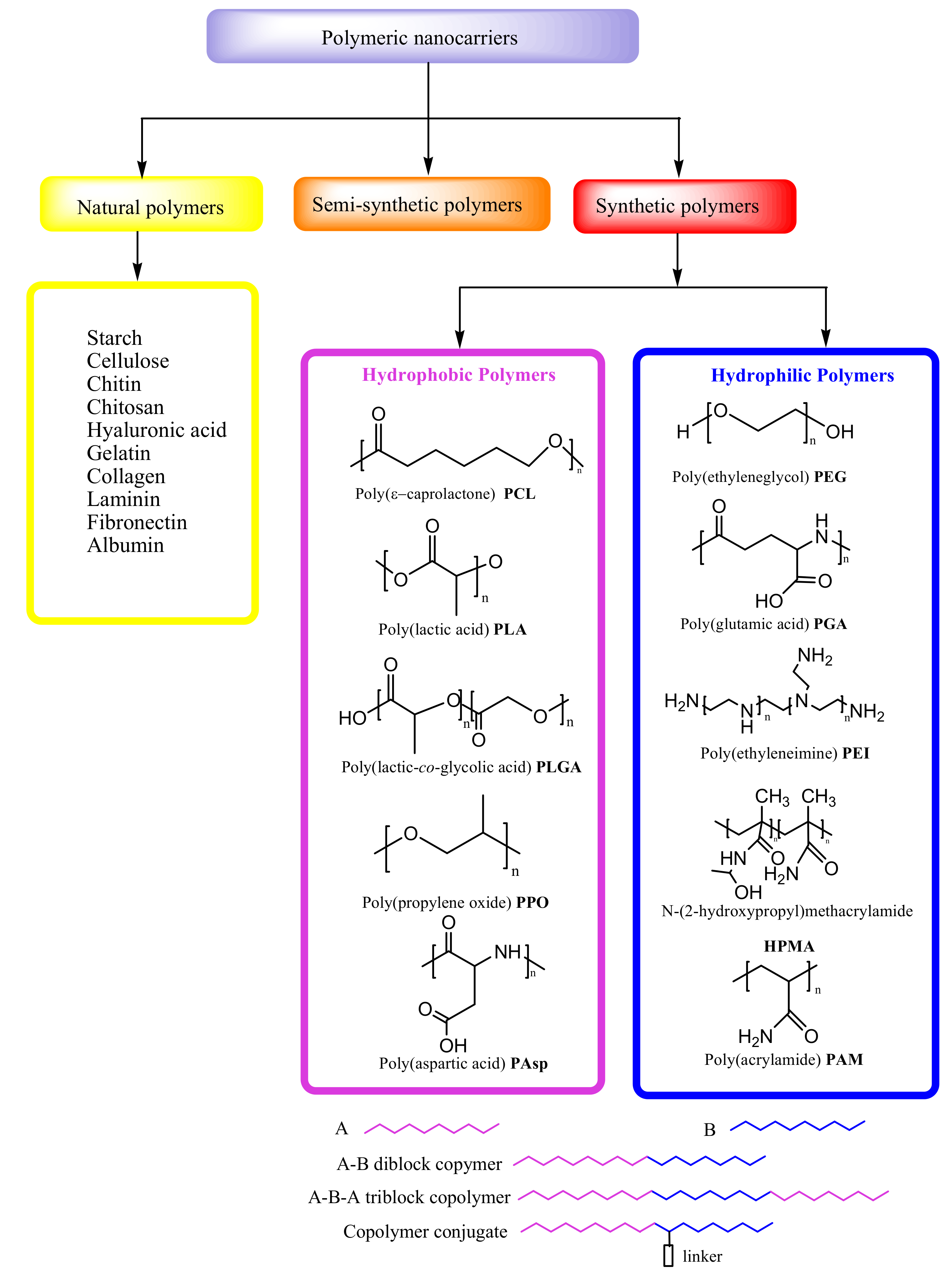
3.1. Hydrophobic and Hydrophilic Polymeric Nanocarriers
3.2. Block Copolymers of DDSs in Cancer Therapy
3.3. Polymer-Drug Conjugates in Cancer Therapy
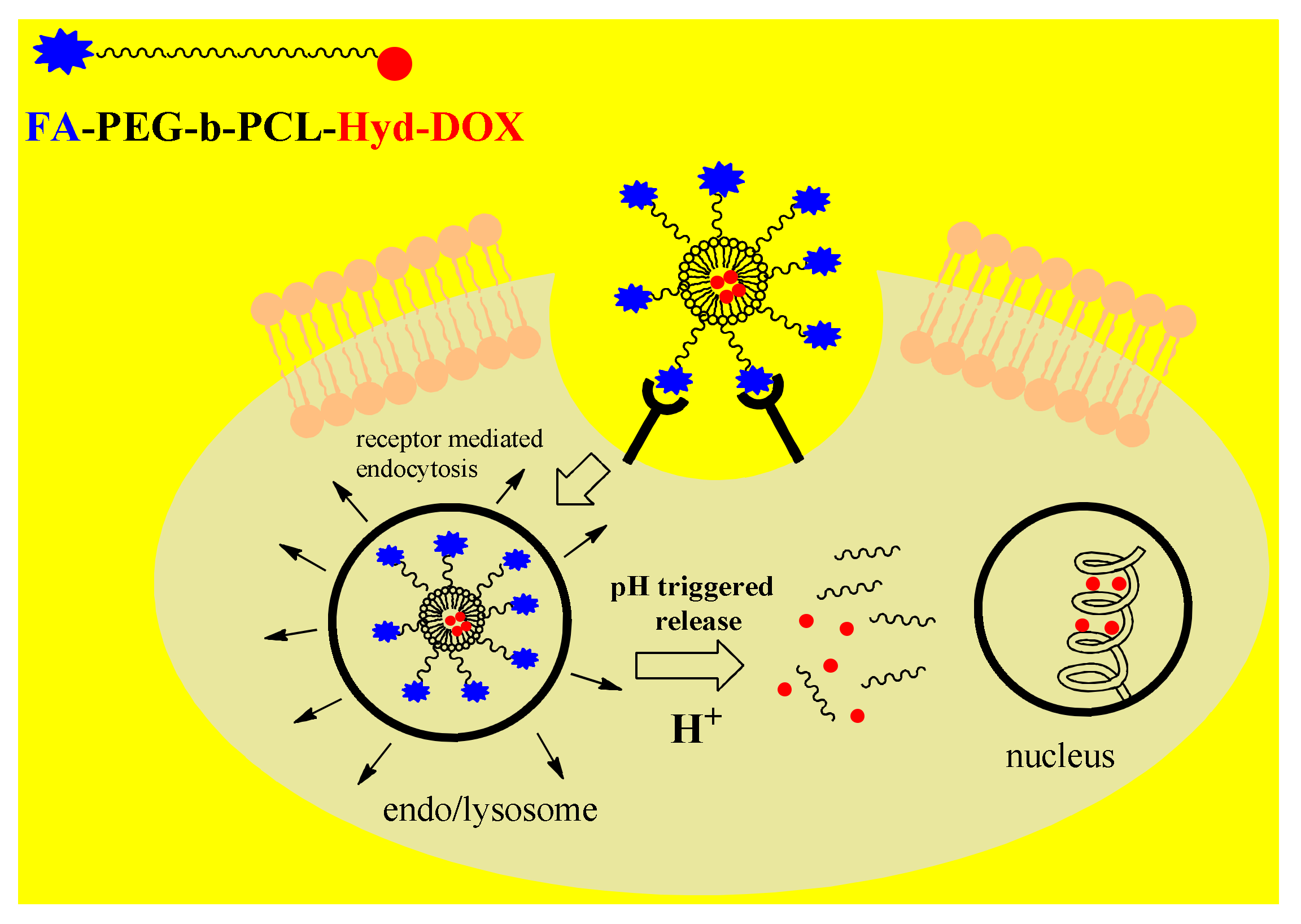
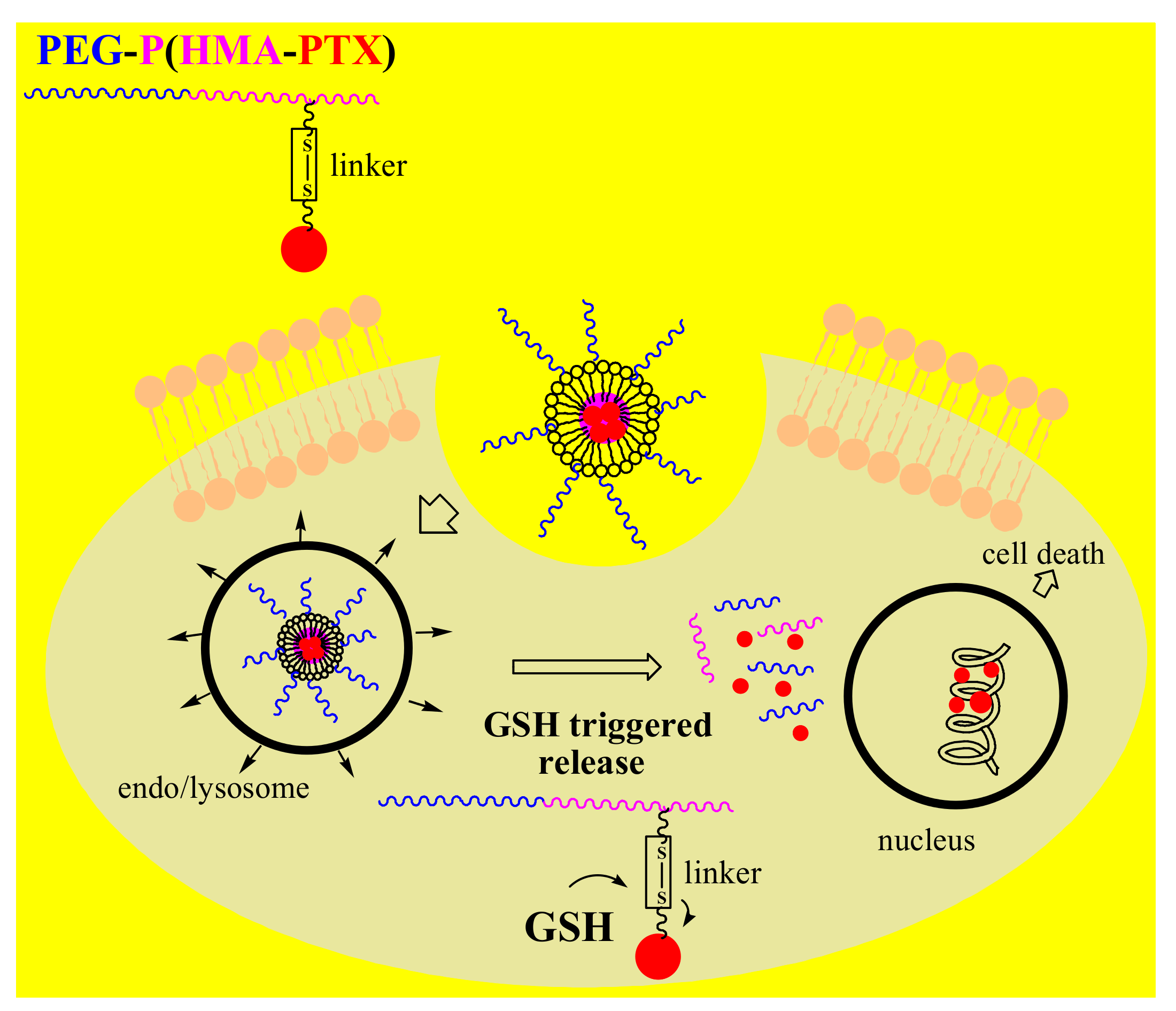
4. Stimuli-Responsive Polymer-Drug Conjugates
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Tran, S.; DeGiovanni, P.J.; Piel, B.; Rai, P. Cancer nanomedicine: A review of recent success in drug delivery. Clin. Transl. Med. 2017, 6, 44. [Google Scholar] [CrossRef] [PubMed]
- Bahrami, B.; Hojjat-Farsangi, M.; Mohammadi, H.; Anvari, E.; Ghalamfarsa, G.; Yousefi, M.; Jadidi-Niaragh, F. Nanoparticles and targeted drug delivery in cancer therapy. Immunol. Lett. 2017, 190, 64–83. [Google Scholar] [CrossRef]
- Pérez-Herrero, E.; Fernández-Medarde, A. Advanced targeted therapies in cancer: Drug nanocarriers, the future of chemotherapy. Eur. J. Pharm. Biopharm. 2015, 93, 52–79. [Google Scholar] [CrossRef] [PubMed]
- Avramović, N.; Ignjatović, N.; Savić, A. Platinum and ruthenium complexes as promising molecules in cancer therapy. Srp. Arh. Celok. Lek. 2019, 147, 105–109. [Google Scholar] [CrossRef]
- Escobar, Q.M.; Maschietto, M.; Krepischi, A.C.V.; Avramovic, N.; Tasic, L. Insights into the Chemical Biology of Childhood Embryonal Solid Tumors by NMR-Based Metabolomics. Biomolecules 2019, 9, 843. [Google Scholar] [CrossRef] [PubMed]
- Radic, T.; Coric, V.; Bukumiric, Z.; Pljesa-Ercegovac, M.; Djukic, T.; Avramovic, N.; Matic, M.; Mihailovic, S.; Dragicevic, D.; Dzamic, Z.; et al. GSTO1*CC Genotype (rs4925) Predicts Shorter Survival in Clear Cell Renal Cell Carcinoma Male Patients. Cancers 2019, 11, 2038. [Google Scholar] [CrossRef] [PubMed]
- Hossen, S.; Hossain, K.M.; Basher, M.K.; Mia, M.N.H.; Rahman, M.T.; Uddin, J.M. Smart nanocarrier-based drug delivery systems for cancer therapy andtoxicity studies: A review. J. Adv. Res. 2019, 15, 1–18. [Google Scholar] [CrossRef]
- Guo, X.; Wang, L.; Wei, X.; Zhou, S. Polymer-Based Drug Delivery Systems for Cancer Treatment. J. Polym. Sci. A Polym. Chem. 2016, 54, 3525–3550. [Google Scholar] [CrossRef]
- Parveen, S.; Arjmand, F.; Tabassum, S. Clinical developments of antitumor polymertherapeutics. RSCAdv. 2019, 9, 24699–24721. [Google Scholar] [CrossRef]
- Fathi, M.; Barar, J. Perspective highlights on biodegradable polymeric nanosystems for targeted therapy of solid tumors. Bioimpacts 2017, 7, 49–57. [Google Scholar] [CrossRef]
- Calzoni, E.; Cesaretti, A.; Polchi, A.; Di Michele, A.; Tancini, B.; Emiliani, C. Biocompatible Polymer Nanoparticles for Drug Delivery Applications in Cancer and Neurodegenerative Disorder Therapies. J. Funct. Biomater. 2019, 10, 4. [Google Scholar] [CrossRef] [PubMed]
- Cabral, H.; Kataoka, K. Progress of drug-loaded polymeric micelles into clinical studies. J. Control. Release 2014, 190, 465–476. [Google Scholar] [CrossRef] [PubMed]
- Allen, T.M. Ligand-targeted therapeutics in anticancer therapy. Nat. Rev. Cancer 2002, 2, 750–763. [Google Scholar] [CrossRef] [PubMed]
- Torchilin, V. Antibody-modified liposomes for cancer chemotherapy. Expert Opin. Drug Deliv. 2008, 5, 1003–1025. [Google Scholar] [CrossRef] [PubMed]
- Cheng, W.W.; Allen, T.M. The use of single chain Fv as targeting agents for immunoliposomes: An update on immunoliposomal drugs for cancer treatment. Expert Opin. Drug Deliv. 2010, 7, 461–478. [Google Scholar] [CrossRef]
- Farokhzad, O.C.; Cheng, J.; Teply, B.A.; Sherifi, I.; Jon, S.; Kantoff, P.W.; Richie, J.P.; Langer, R. Targeted nanoparticle-aptamer bioconjugates for cancer chemotherapyin vivo. Proc. Natl. Acad. Sci. USA 2006, 103, 6315–6320. [Google Scholar] [CrossRef]
- Oba, M.; Vachutinsky, Y.; Miyata, K.; Kano, M.R.; Ikeda, S.; Nishiyama, N.; Itaka, K.; Miyazono, K.; Koyama, H.; Kataoka, K. Antiangiogenic gene therapy of solid tumor bysystemic injection of polyplex micelles loading plasmid DNA encoding soluble flt-1. Mol. Pharm. 2010, 7, 501–509. [Google Scholar] [CrossRef]
- Miura, Y.; Takenaka, T.; Toh, K.; Wu, S.; Nishihara, H.; Kano, M.R.; Ino, Y.; Nomoto, T.; Matsumoto, Y.; Koyama, H.; et al. Cyclic RGD-linked polymeric micelles for targeted delivery of platinum anticancer drugsto glioblastoma through the blood-brain tumor barrier. ACS Nano 2013, 7, 8583–8592. [Google Scholar] [CrossRef]
- Bae, Y.; Jang, W.D.; Nishiyama, N.; Fukushima, S.; Kataoka, K. Multifunctional poly-meric micelles with folate-mediated cancer cell targeting and pH-triggered drug releasing properties for active intracellular drug delivery. Mol. Biosyst. 2005, 1, 242–250. [Google Scholar] [CrossRef]
- Torchilin, V.P. Cell penetrating peptide-modified pharmaceuticalnanocarriers for intracellular drug and gene delivery. Biopolymers 2008, 90, 604–610. [Google Scholar] [CrossRef]
- Skatrud, P.L. The impact of multiple drug resistance (MDR) proteins on chemotherapy and drug discovery. Prog. Drug Res. 2002, 58, 99–131. [Google Scholar] [CrossRef] [PubMed]
- Dai, X.; Tan, C. Combination of microRNA therapeutics with small-molecule anticancer drugs: Mechanismof action and co-delivery nanocarriers. Adv. Drug Deliv. Rev. 2015, 81, 184–197. [Google Scholar] [CrossRef] [PubMed]
- Teo, P.Y.; Cheng, W.; Hedrick, J.L.; Yang, Y.Y. Co-delivery of drugs and plasmid DNA for cancer therapy. Adv. Drug Deliv. Rev. 2016, 98, 41–63. [Google Scholar] [CrossRef] [PubMed]
- Navarro, G.; Pan, J.; Torchilin, V.P. Micelle-like nanoparticles as carriers for DNA and siRNA. Mol. Pharm. 2015, 12, 301–313. [Google Scholar] [CrossRef] [PubMed]
- Alinejad, V.; Hossein Somi, M.; Baradaran, B.; Akbarzadeh, P.; Atyabi, F.; Kazerooni, H.; SamadiKafil, H.; AghebatiMaleki, L.; Siah Mansouri, H.; Yousefi, M. Co-delivery of IL17RB siRNA and doxorubicin bychitosan-based nanoparticles for enhanced anticancer efficacy in breast cancer cells. Biomed. Pharmacother. 2016, 83, 229–240. [Google Scholar] [CrossRef] [PubMed]
- Wei, W.; Lv, P.P.; Chen, X.M.; Yue, Z.G.; Fu, Q.; Liu, S.Y.; Yue, H.; Ma, G.H. Codelivery of mTERT siRNA and paclitaxel by chitosan-based nanoparticles promoted synergistic tumor suppression. Biomaterials 2013, 34, 3912–3923. [Google Scholar] [CrossRef]
- Li, N.; Huang, C.; Luan, Y.; Song, A.; Song, Y.; Garg, S. Active targeting co-delivery system based onpH-sensitive methoxy-poly(ethylene glycol)2K-poly(epsilon-caprolactone)4K-poly(glutamic acid)1K forenhanced cancer therapy. J. Colloid Interface Sci. 2016, 472, 90–98. [Google Scholar] [CrossRef]
- Pan, J.; Palmerston Mendes, L.; Yao, M.; Filipczak, N.; Garai, S.; Thakur, G.A.; Sarisozen, C.; Torchilin, V.P. Polyamidoamine dendrimers-based nanomedicine for combination therapy with siRNAand chemotherapeutics to overcome multidrug resistance. Eur. J. Pharm. Biopharm. 2019, 136, 18–28. [Google Scholar] [CrossRef]
- Wang, X.; Liow, S.S.; Wu, Q.; Li, C.; Owh, C.; Li, Z.; Loh, X.J.; Wu, Y.L. Codelivery for Paclitaxel and Bcl-2 Conversion Gene by PHB-PDMAEMA Amphiphilic Cationic Copolymer for Effective Drug Resistant Cancer Therapy. Macromol. Biosci. 2017, 17, 1700186. [Google Scholar] [CrossRef]
- Cheng, Q.; Du, L.; Meng, L.; Han, S.; Wei, T.; Wang, X.; Wu, Y.; Song, X.; Zhou, J.; Zheng, S.; et al. The Promising Nanocarrier for Doxorubicin and siRNA Co-delivery by PDMAEMA-based Amphiphilic Nanomicelles. ACS Appl. Mater. Interfaces 2016, 8, 4347–4356. [Google Scholar] [CrossRef]
- Cheng, H.; Yang, W.; Chen, H.; Liu, L.; Gao, F.; Yang, X.; Jiang, Q.; Zhang, Q.; Wang, Y. Surface modificationof mitoxantrone-loaded PLGA nanospheres with chitosan. Colloids Surf. B Biointerfaces 2009, 73, 212–218. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Hao, Y.; Li, H.; Zhao, Y.; Meng, D.; Li, D.; Shi, J.; Zhang, H.; Zhang, Z.; Zhang, Y. Co-delivery of doxorubicin and siRNA for glioma therapy by a brain targeting system: Angiopep-2-modifiedpoly(lactic-co-glycolic acid) nanoparticles. J. Drug Target 2015, 23, 832–846. [Google Scholar] [CrossRef] [PubMed]
- Cao, N.; Cheng, D.; Zou, S.; Ai, H.; Gao, J.; Shuai, X. The synergistic effect of hierarchical assemblies of siRNA and chemotherapeutic drugs co-delivered into hepatic cancer cells. Biomaterials 2011, 32, 2222–2232. [Google Scholar] [CrossRef] [PubMed]
- Navarro, G.; Sawant, R.R.; Biswas, S.; Essex, S.; Tros de Ilarduya, C.; Torchilin, V.P. P-glycoprotein silencingwith siRNA delivered by DOPE-modified PEI overcomes doxorubicin resistance in breast cancer cells. Nanomedicine 2012, 7, 65–78. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.Y.; Kuo, W.T.; Chou, M.J.; Huang, Y.Y. Co-delivery of anti-vascular endothelial growth factorsiRNA and doxorubicin by multifunctional polymeric micelle for tumor growth suppression. J. Biomed. Mater. Res. A 2011, 97, 330–338. [Google Scholar] [CrossRef]
- Knop, K.; Hoogenboom, R.; Fischer, D.; Schubert, U.S. Poly(ethylene glycol) in drug delivery: Pros and cons as well as potential alternatives. Angew. Chem. Int. Ed. 2010, 49, 6288–6308. [Google Scholar] [CrossRef]
- Zhou, S.; Deng, X.; Yang, H. Biodegradable poly(epsilon-caprolactone)-poly(ethylene glycol) block copolymers: Characterization and their use as drug carriers for a controlled delivery system. Biomaterials 2003, 24, 3563–3570. [Google Scholar] [CrossRef]
- Zhang, Z.; Qu, Q.; Li, J.; Zhou, S. The Effect of the Hydrophilic/Hydrophobic Ratio of Polymeric Micelles on their Endocytosis Pathways into Cells. Macromol. Biosci. 2013, 13, 789–798. [Google Scholar] [CrossRef]
- Çırpanlı, Y.; Allard, E.; Passirani, C.; Bilensoy, E.; Lemaire, L.; Çalış, S.; Benoit, J.P. Antitumoral activity ofcamptothecin-loaded nanoparticles in 9L rat glioma model. Int. J. Pharm. 2011, 403, 201–206. [Google Scholar] [CrossRef]
- Hu, J.; Fu, S.; Peng, Q.; Han, Y.; Xie, J.; Zan, N.; Chen, Y.; Fan, J. Paclitaxel-loaded polymeric nanoparticles combined with chronomodulated chemotherapy on lung cancer: In vitro and in vivo evaluation. Int. J.Pharm. 2017, 516, 313–322. [Google Scholar] [CrossRef]
- Hong, G.; Yuan, R.; Liang, B.; Shen, J.; Yang, X.; Shuai, X. Folate-functionalized polymeric micelle as hepatic carcinoma-targeted, MRI-ultrasensitive delivery system of antitumor drugs. Biomed. Microdevices 2008, 10, 693–700. [Google Scholar] [CrossRef] [PubMed]
- Wen, X.; Wu, Q.P.; Ke, S.; Ellis, L.; Charnsangavej, C.; Delpassand, A.S.; Wallace, S.; Li, C. Conjugation with (111)In-DTPA-poly(ethylene glycol)improves imaging of anti-EGF receptor antibody C225. J. Nucl. Med. 2001, 42, 1530–1537. [Google Scholar]
- Lee, H.; Hoang, B.; Fonge, H.; Reilly, R.M.; Allen, C. In vivo distribution of polymeric nanoparticles at the whole-body, tumor, and cellular levels. Pharm. Res. 2010, 27, 2343–2355. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.; Gao, X.; Su, L.; Xia, H.; Gu, G.; Pang, Z.; Jiang, X.; Yao, L.; Chen, J.; Chen, H. Aptamer-functionalized PEG–PLGA nanoparticles for enhance danti-glioma drug delivery. Biomaterials 2011, 32, 8010–8020. [Google Scholar] [CrossRef] [PubMed]
- Shafiei-Irannejad, V.; Samadi, N.; Salehi, R.; Yousefi, B.; Rahimi, M.; Akbarzadeh, A.; Zarghami, N. Reversion of Multidrug Resistance by Co-Encapsulation of Doxorubicin and Metformin in Poly(lactide-co-glycolide)-D-α-tocopheryl Polyethylene Glycol 1000 Succinate Nanoparticles. Pharm. Res. 2018, 35, 119. [Google Scholar] [CrossRef]
- Wang, H.; Zhao, Y.; Wu, Y.; Hu, Y.L.; Nan, K.; Nie, G.; Chen, H. Enhanced anti-tumor efficacy by co-deliveryof doxorubicin and paclitaxel with amphiphilic methoxy PEG-PLGA copolymer nanoparticles. Biomaterials 2011, 32, 8281–8290. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Chen, X.; Wang, Z.; Jing, X. Ultrafine PEG-PLA fibers loaded with both paclitaxel and doxorubicinhydrochloride and their in vitro cytotoxicity. Eur. J. Pharm. Biopharm. 2009, 72, 18–25. [Google Scholar] [CrossRef]
- Duong, H.H.; Yung, L.Y. Synergistic co-delivery of doxorubicin and paclitaxel using multi-functional micellesfor cancer treatment. Int. J. Pharm. 2013, 454, 486–495. [Google Scholar] [CrossRef]
- Lv, S.; Tang, Z.; Li, M.; Lin, J.; Song, W.; Liu, H.; Huang, Y.; Zhang, Y.; Chen, X. Co-delivery of doxorubicinand paclitaxel by PEG-polypeptide nanovehicle for the treatment of non-small cell lung cancer. Biomaterials 2014, 35, 6118–6129. [Google Scholar] [CrossRef]
- Matsumura, Y. Polymeric Micellar Delivery Systems in Oncology. Jpn. J. Clin. Oncol. 2008, 38, 793–802. [Google Scholar] [CrossRef]
- Wilson, R.H.; Plummer, R.; Adam, J.; Eatock, M.; Boddy, A.V.; Griffin, M.; Miller, R.; Matsumura, Y.; Shimizu, T.; Calvert, H. Phase I and pharmacokinetic study of NC-6004, a new platinum entity of cisplatin-conjugated polymer forming micelles. Clin. Oncol. 2008, 26, 2573. [Google Scholar] [CrossRef]
- Plummer, R.; Wilson, R.H.; Calvert, H.; Boddy, A.V.; Griffin, M.; Sludden, J.; Tilby, M.J.; Eatock, M.; Pearson, D.G.; Ottley, C.J. A Phase I clinical study of cisplatin-incorporated polymeric micelles (NC-6004) in patients with solid tumours T. Br. J. Cancer 2011, 104, 593–598. [Google Scholar] [CrossRef] [PubMed]
- Vega, J.; Ke, S.; Fan, Z.; Wallace, S.; Charsangavej, C.; Li, C. Targeting doxorubicin to epidermal growth factor receptors bysite-specific conjugation of C225 to poly(L-glutamic acid)through a polyethylene glycol spacer. Pharm. Res. 2003, 20, 826–832. [Google Scholar] [CrossRef] [PubMed]
- Yuan, J.D.; ZhuGe, D.L.; Tong, M.Q.; Lin, M.T.; Xu, X.F.; Tang, X.; Zhao, Y.Z.; Xu, H.L. pH-sensitive polymericnanoparticles of mPEG-PLGA-PGlu with hybrid core for simultaneous encapsulation of curcumin anddoxorubicin to kill the heterogeneous tumour cells in breast cancer. Artif. Cells Nanomed. Biotechnol. 2018, 46, 302–313. [Google Scholar] [CrossRef] [PubMed]
- Matsumura, Y. Poly (amino acid) micelle nanocarriers in preclinical and clinical studies. Adv. Drug Deliv. Rev. 2008, 60, 899–914. [Google Scholar] [CrossRef]
- Hamaguchi, T.; Matsumura, Y.; Suzuki, M.; Shimizu, K.; Goda, R.; Nakamura, I.; Nakatomi, I.; Yokoyama, M.; Kataoka, K.; Kakizoe, T. NK105, a paclitaxel-incorporating micellar nanoparticle formulation, can extend in vivo antitumour activity and reduce the neurotoxicity of paclitaxel. Br. J. Cancer 2005, 92, 1240–1246. [Google Scholar] [CrossRef]
- Hamaguchi, T.; Kato, K.; Yasui, H.; Morizane, C.; Ikeda, M.; Ueno, H.; Muro, K.; Yamada, Y.; Okusaka, T.; Shirao, K.; et al. A phase I and pharmacokinetic study of NK105, a paclitaxel-incorporating micellar nanoparticle formulation. Br. J. Cancer 2007, 97, 170–176. [Google Scholar] [CrossRef]
- Vilar, G.; Puche, J.T.; Albericio, F. Polymers and drug delivery systems. Curr. Drug Deliv. 2012, 9, 367–394. [Google Scholar] [CrossRef]
- Venne, A.; Li, S.; Mandeville, R.; Kabanov, A.; Alakhov, V. Hypersensitizing effect of pluronic L61 on cytotoxic activity, transport and subcellular distribution of doxorubicin in multiple drug-resistant cells. Cancer Res. 1996, 56, 3626–3629. [Google Scholar]
- Valle, J.W.; Armstrong, A.; Newman, C.; Alakhov, V.; Pietrzynski, G.; Brewer, J.; Campbell, S.; Corrie, P.; Rowinsky, E.K.; Ranson, M. A phase 2 study of SP1049C, doxorubicin in P-glycoprotein-targeting pluronics, in patients with advanced adenocarcinoma of the esophagus and gastroesophageal junction. Investig. New Drugs 2010, 29, 1029–1037. [Google Scholar] [CrossRef]
- Hu, D.; Chen, L.; Qu, Y.; Peng, J.; Chu, B.; Shi, K.; Hao, Y.; Zhong, L.; Wang, M.; Qian, Z. Oxygen-generating Hybrid Polymeric Nanoparticles with Encapsulated Doxorubicin and Chlorin e6 for Trimodal Imaging-Guided Combined Chemo-Photodynamic Therapy. Theranostics 2018, 8, 1558–1574. [Google Scholar] [CrossRef] [PubMed]
- Jin, M.; Jin, G.; Kang, L.; Chen, L.; Gao, Z.; Huang, W. Smart polymeric nanoparticles with pH-responsive and PEG-detachable properties for co-delivering paclitaxel and survivin siRNA to enhance antitumor outcomes. Int. J. Nanomed. 2018, 13, 2405–2426. [Google Scholar] [CrossRef] [PubMed]
- Greenwald, R.B.; Pendri, A.; Conover, C.D.; Lee, C.; Choe, Y.H.; Gilbert, C.; Martinez, A.; Xia, Y.; Wu, D.; Hsue, M. Camptothecin-20-PEG ester transport forms: The effect of spacer groups on antitumor activity. Bioorg. Med. Chem. 1998, 6, 551–562. [Google Scholar] [CrossRef]
- Fraier, D.; Frigerio, E.; Brianceschi, G.; Casati, M.; Benecchi, A.; James, C. Determination of MAG-Camptothecin, a new polymer-bound Camptothecin derivative, and free Camptothecin in dog plasma by HPLC with fluorimetricdetection. J. Pharm. Biomed. Anal. 2000, 19, 505–514. [Google Scholar] [CrossRef]
- Singer, J.W.; Bhatt, R.; Tulinsky, J.; Buhler, K.R.; Heasley, E.; Klein, P.; James, C. Water-soluble poly-(l-glutamic acid)–Gly-camptothecin conjugates enhance camptothecin stability and efficacy in vivo. J. Control. Release 2001, 74, 243–247. [Google Scholar] [CrossRef]
- Pastorino, F.; Loi, M.; Sapra, P.; Becherini, P.; Cilli, M.; Emionite, L.; Ribatti, D.; Greenberger, L.M.; Horak, I.D.; Ponzoni, M. Tumor Regression and Curability of Preclinical Neuroblastoma Models by PEGylated SN38 (EZN-2208), a Novel Topoisomerase I Inhibitor. Clin. Cancer Res. 2010, 16, 4809–4821. [Google Scholar] [CrossRef]
- Sapra, P.; Zhao, H.; Mehlig, M.; Malaby, J.; Kraft, P.; Longley, C.; Greenberger, L.M.; Horak, I.D. Novel Delivery of SN38 Markedly Inhibits Tumor Growth in Xenografts, Including a Camptothecin-11–Refractory Model. Clin. Cancer Res. 2008, 14, 1888–1896. [Google Scholar] [CrossRef]
- Crozier, J.A.; Advani, P.P.; Plant, B.L.; Anthony, T.H.; Jaslowski, J.; Moreno-Aspitia, A.; Perez, E.A. N0436 (Alliance): A phase II trial of irinotecan plus cetuximab in patients with metastatic breast cancer previously exposed to anthracycline and/or taxane-containing therapy. Clin. Breast Cancer 2016, 16, 23–30. [Google Scholar] [CrossRef]
- Duncan, R.; Vicent, M.J. Do HPMA copolymer conjugates have a future as clinically useful nanomedicines? A critical overview of current status and future opportunities. Adv. Drug Deliv. Rev. 2010, 62, 272–282. [Google Scholar] [CrossRef]
- Seymour, L.W.; Ferry, D.R.; Kerr, D.J.; Rea, D.; Whitlock, M.; Poyner, R.; Boivin, C.; Hesslewood, S.; Twelves, C.; Blackie, R.; et al. Phase II studies of polymer-doxorubicin (PK1, FCE28068) in the treatment of breast, lung and colorectal cancer. Int. J. Oncol. 2009, 34, 1629–1636. [Google Scholar] [CrossRef]
- Terwogt, J.M.M.; ten BokkelHuinink, W.W.; Schellens, J.H.M.; Schot, M.; Mandjes, I.; Zurlo, M.; Rocchetti, M.; Rosing, H.; Koopman, F.M.; Beijnen, J.H. Phase I clinical and pharmacokinetic study of PNU166945, a novel water-soluble polymer-conjugated prodrug of paclitaxel. Anticancer Drugs 2001, 12, 315–323. [Google Scholar] [CrossRef] [PubMed]
- Campone, M.; Rademaker-Lakhai, J.M.; Bennouna, J.; Howell, S.B.; Nowotnik, D.P.; Beijnen, J.H.; Schellens, J.H. Phase I and pharmacokinetic trial of AP5346, a DACH-platinum-polymer conjugate, administered weekly for three out of every 4 weeks to advanced solid tumor patients. Cancer Chemother. Pharmacol. 2007, 60, 523–533. [Google Scholar] [CrossRef] [PubMed]
- Rice, J.R.; Howell, S.B. AP-5346. Drugs Future 2004, 29, 561. [Google Scholar] [CrossRef]
- Kelland, L. Broadening the clinical use of platinum drug–based chemotherapy with new analogues. ExpertOpin. Investig. Drugs 2007, 16, 1009–1021. [Google Scholar] [CrossRef] [PubMed]
- Nowotnik, D.P.; Cvitkovic, E. ProLindac™(AP5346): A review of the development of an HPMA DACH platinum Polymer Therapeutic. Adv. Drug Deliv. Rev. 2009, 61, 1214–1219. [Google Scholar] [CrossRef] [PubMed]
- Pan, J.; Rostamizadeh, K.; Filipczak, N.; Torchilin, V. Polymeric Co-Delivery Systems in Cancer Treatment: An Overview on Component Drugs Dosage Ratio Effect. Molecules 2019, 24, 1035. [Google Scholar] [CrossRef]
- Das, L.; Vinayak, M. Long-term effect of curcumin down-regulates expression of tumor necrosis factor-alphaand interleukin-6 via modulation of E26 transformation-specific protein and nuclear factor-κB transcription factors in livers of lymphoma bearing mice. Leuk. Lymphoma 2014, 55, 2627–2636. [Google Scholar] [CrossRef]
- Tuorkey, M.J. Curcumin a potent cancer preventive agent: Mechanisms of cancer cell killing. Interv. Med.Appl. Sci. 2014, 6, 139–146. [Google Scholar] [CrossRef]
- Wang, B.L.; Shen, Y.M.; Zhang, Q.W.; Li, Y.L.; Luo, M.; Liu, Z.; Li, Y.; Qian, Z.Y.; Gao, X.; Shi, H.S. Codelivery of curcumin and doxorubicin by MPEG-PCL results in improved efficacy of systemically administered chemotherapy in mice with lung cancer. Int. J. Nanomed. 2013, 8, 3521–3531. [Google Scholar] [CrossRef]
- Duan, J.; Mansour, H.M.; Zhang, Y.; Deng, X.; Chen, Y.; Wang, J.; Pan, Y.; Zhao, J. Reversion of multidrug resistance by co-encapsulation of doxorubicin and curcumin in chitosan/poly(butyl cyanoacrylate)nanoparticles. Int. J. Pharm. 2012, 426, 193–201. [Google Scholar] [CrossRef]
- Guo, O.; Li, X.; Yang, Y.; Wei, J.; Zhao, Q.; Luo, F.; Qian, Z. Enhanced 4T1 breast carcinoma anticancer activity by co-delivery of doxorubicin and curcumin with core-shell drug-carrier based on heparin modified poly(L-lactide) grafted polyethylenimine cationic nanoparticles. J. Biomed. Nanotechnol. 2014, 10, 227–237. [Google Scholar] [CrossRef] [PubMed]
- Danson, S.; Ferry, D.; Alakhov, V.; Margison, J.; Kerr, D.; Jowle, D.; Brampton, M.; Halbert, G.; Ranson, M. Phase I dose escalation and pharmacokinetic study of pluronicpolymer-bound doxorubicin (SP1049C) in patients with ad-vanced cancer. Br. J. Cancer 2004, 90, 2085–2091. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Yu, L.; Han, L.; Sha, X.; Fang, X. Difunctional Pluroniccopolymer micelles for paclitaxel delivery: Synergistic effect of folate-mediated targeting and Pluronic-mediated overcoming multidrugresistance in tumor cell lines. Int. J. Pharm. 2007, 337, 63–73. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Zhang, W.; Huang, Y.; Gao, F.; Sha, X.; Fang, X. Pluronic-based functional polymeric mixed micellesfor co-delivery of doxorubicin and paclitaxel to multidrug resistant tumor. Int. J. Pharm. 2015, 488, 44–58. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Fan, X.; Li, L. pH-sensitive polymeric micelles formed by doxorubicin conjugated prodrugs forco-delivery of doxorubicin and paclitaxel. Carbohydr. Polym. 2016, 137, 19–29. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Agarwal, P.; Zhao, S.; Xu, R.X.; Yu, J.; Lu, X.; He, X. Hyaluronic acid-decorated dual responsivenanoparticles of Pluronic F127, PLGA, and chitosan for targeted co-delivery of doxorubicin and irinotecan toeliminate cancer stem-like cells. Biomaterials 2015, 72, 74–89. [Google Scholar] [CrossRef]
- Tang, S.; Yin, Q.; Su, J.; Sun, H.; Meng, Q.; Chen, Y.; Chen, L.; Huang, Y.; Gu, W.; Xu, M.; et al. Inhibition ofmetastasis and growth of breast cancer by pH-sensitive poly (beta-amino ester) nanoparticles co-deliveringtwo siRNA and paclitaxel. Biomaterials 2015, 48, 1–15. [Google Scholar] [CrossRef]
- Pasut, G.; Veronese, F.M. PEG conjugates in clinical development or use as anticancer agents: An overview. Adv. Drug Deliv. Rev. 2009, 61, 1177–1188. [Google Scholar] [CrossRef]
- Kopecek, J.; Kopeckova, P. HPMA copolymers: Origins, early developments, present, and future. Adv. Drug Deliv. Rev. 2010, 62, 122–149. [Google Scholar] [CrossRef]
- Hopewel, W.; Duncan, R.; Wilding, D.; Chakrabarti, K. Preclinical evaluation of the cardiotoxicity of PK2: A novel HPMA copolymer–doxorubicin–galactosamine conjugate antitumouragent. Hum. Exp. Toxicol. 2001, 20, 461–470. [Google Scholar] [CrossRef]
- Julyan, P.J.; Seymour, L.W.; Ferry, D.R.; Daryani, S.; Boivin, C.M.; Doran, J.; David, M.; Anderson, D.; Christodoulou, C.; Young, A.M.; et al. Preliminary clinical study of the distribution of HPMA copolymers bearing doxorubicin and galactosamine. J. Control. Release 1999, 57, 281–290. [Google Scholar] [CrossRef]
- Seymour, L.W.; Ferry, D.R.; Anderson, D.; Hesslewood, S.; Julyan, P.J.; Poyner, R.; Doran, J.; Young, A.M.; Burtles, S.; Kerr, D.J. Hepatic Drug Targeting: Phase I Evaluation of Polymer-Bound Doxorubicin. J. Clin. Oncol. 2002, 20, 1668–1676. [Google Scholar] [CrossRef] [PubMed]
- Bae, Y.; Nishiyama, N.; Fukushima, S.; Koyama, H.; Yasuhiro, M.; Kataoka, K. Preparation and biological characterization of polymeric micelle drug carriers with intracellular pH-triggered drug release property: Tumor permeability, controlled subcellular drug distribution, and enhanced invivo antitumor efficacy. Bioconjug.Chem. 2005, 16, 122–130. [Google Scholar] [CrossRef]
- Xiong, X.B.; Ma, Z.; Lai, R.; Lavasanifar, A. The therapeutic response to multifunctional polymeric nano-conjugates in the targeted cellular and subcellular delivery of doxorubicin. Biomaterials 2010, 31, 757–768. [Google Scholar] [CrossRef] [PubMed]
- Wu, P.; Opadele, A.E.; Onodera, Y.; Nam, J. Targeting Integrins in Cancer Nanomedicine: Applications in Cancer Diagnosis and Therapy. Cancers 2019, 11, 1783. [Google Scholar] [CrossRef] [PubMed]
- Shi, C.; Guo, X.; Qu, Q.; Tang, Z.; Wang, Y.; Zhou, S. Actively targeted delivery of anticancer drug to tumor cells by redox-responsive star-shaped micelles. Biomaterials 2014, 35, 8711–8722. [Google Scholar] [CrossRef]
- Chen, W.; Shah, L.A.; Yuan, L.; Siddiq, M.; Hu, J.; Yang, D. Polymer–paclitaxel conjugates based on disulfide linkers for controlled drug release. RSC Adv. 2015, 5, 7559–7566. [Google Scholar] [CrossRef]
| Polymer | Drug | Loading Mode | Function | Reference |
|---|---|---|---|---|
| PEG-PCL | Camptothecin (CPT) | Entrapment | Colon, breast, ovarian, lung and brain cancers | [39] |
| PCL-PEG-PCL | Paclitaxel (PTX) | Encapsulation | Lung cancers in combination with chrono-modulated chemotherapy | [40] |
| PLGA-PEG | Paclitaxel (PTX) | Encapsulation | Breast, pancreatic and ovarian and brain cancers | [44] |
| PLGA-TPGS | Doxorubicin(DOX)- Metformin (Met) | Encapsulation | Multidrug resistance P388 cancer cell lines | [45] |
| PEG-PGlu | Cisplatin | Encapsulation | Solid cancers | [50,51,52] |
| mPEG-PLGA-PGlu | Doxorubicin(DOX) | Encapsulation | Breast cancer | [54] |
| PEG-PAsp | Paclitaxel (PTX) | Entrapment | Advanced stomach cancer | [55,56,57] |
| PEO-b-PAsp | Doxorubicin | Entrapment | Pancreatic cancer | [58] |
| PEO-PPO-PEO | Doxorubicin. | Encapsulation | Metastatic adenocarcinoma of the esophagus and gastroesophageal junction | [59,60] |
| PCLLA-PEG-PCLLA | Doxorubicin (DOX) | Encapsulation | Breast cancer | [61] |
| PEI-PLA | Paclitaxel (PTX) | Entrapment | Lung cancer | [62] |
| PEG | Camptothecin (CPT)SN38 Irinotecan (C-11) | Copolymer-drug conjugation | Colorectal, metastatic breast cancer, platinum-resistant ovarian cancer and metastatic cervical cancer | [63,64,65,66,67,68] |
| HPMA | Doxorubicin (DOX) | Copolymer-drug conjugation | Lung and breast cancer | [69,70] |
| HPMA | Paclitaxel (PTX) | Copolymer-drug conjugation | Solid cancers | [71] |
| HPMA | Diaminocyclohexane(DACH)-platinum | Copolymer-drug conjugation | Solid cancer, ovarian cancer | [72,73,74,75] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Avramović, N.; Mandić, B.; Savić-Radojević, A.; Simić, T. Polymeric Nanocarriers of Drug Delivery Systems in Cancer Therapy. Pharmaceutics 2020, 12, 298. https://doi.org/10.3390/pharmaceutics12040298
Avramović N, Mandić B, Savić-Radojević A, Simić T. Polymeric Nanocarriers of Drug Delivery Systems in Cancer Therapy. Pharmaceutics. 2020; 12(4):298. https://doi.org/10.3390/pharmaceutics12040298
Chicago/Turabian StyleAvramović, Nataša, Boris Mandić, Ana Savić-Radojević, and Tatjana Simić. 2020. "Polymeric Nanocarriers of Drug Delivery Systems in Cancer Therapy" Pharmaceutics 12, no. 4: 298. https://doi.org/10.3390/pharmaceutics12040298
APA StyleAvramović, N., Mandić, B., Savić-Radojević, A., & Simić, T. (2020). Polymeric Nanocarriers of Drug Delivery Systems in Cancer Therapy. Pharmaceutics, 12(4), 298. https://doi.org/10.3390/pharmaceutics12040298







