Safe Practice of Y-Site Drug Administration: The Case of Colistin and Parenteral Nutrition
Abstract
1. Introduction
2. Experimental Section
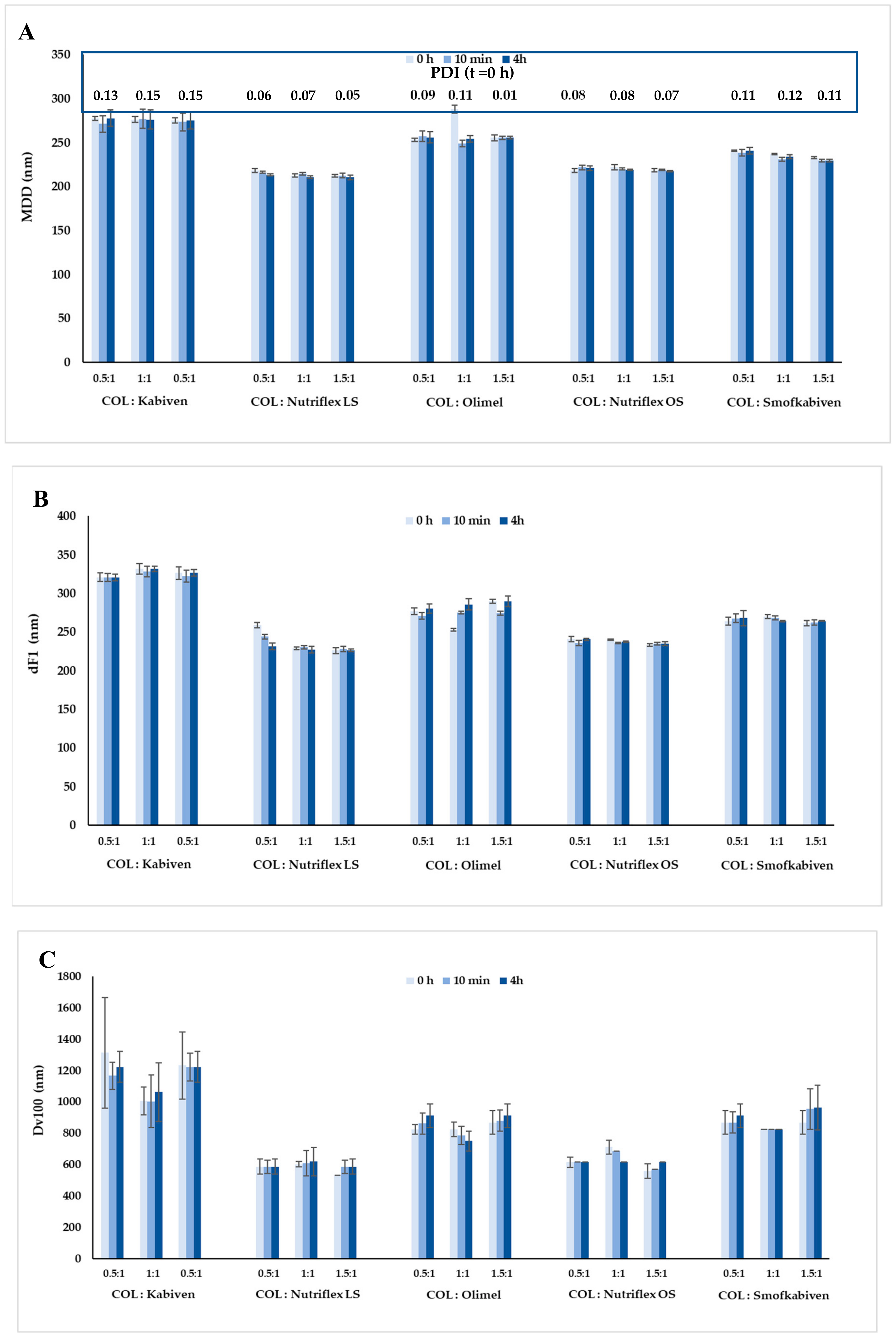
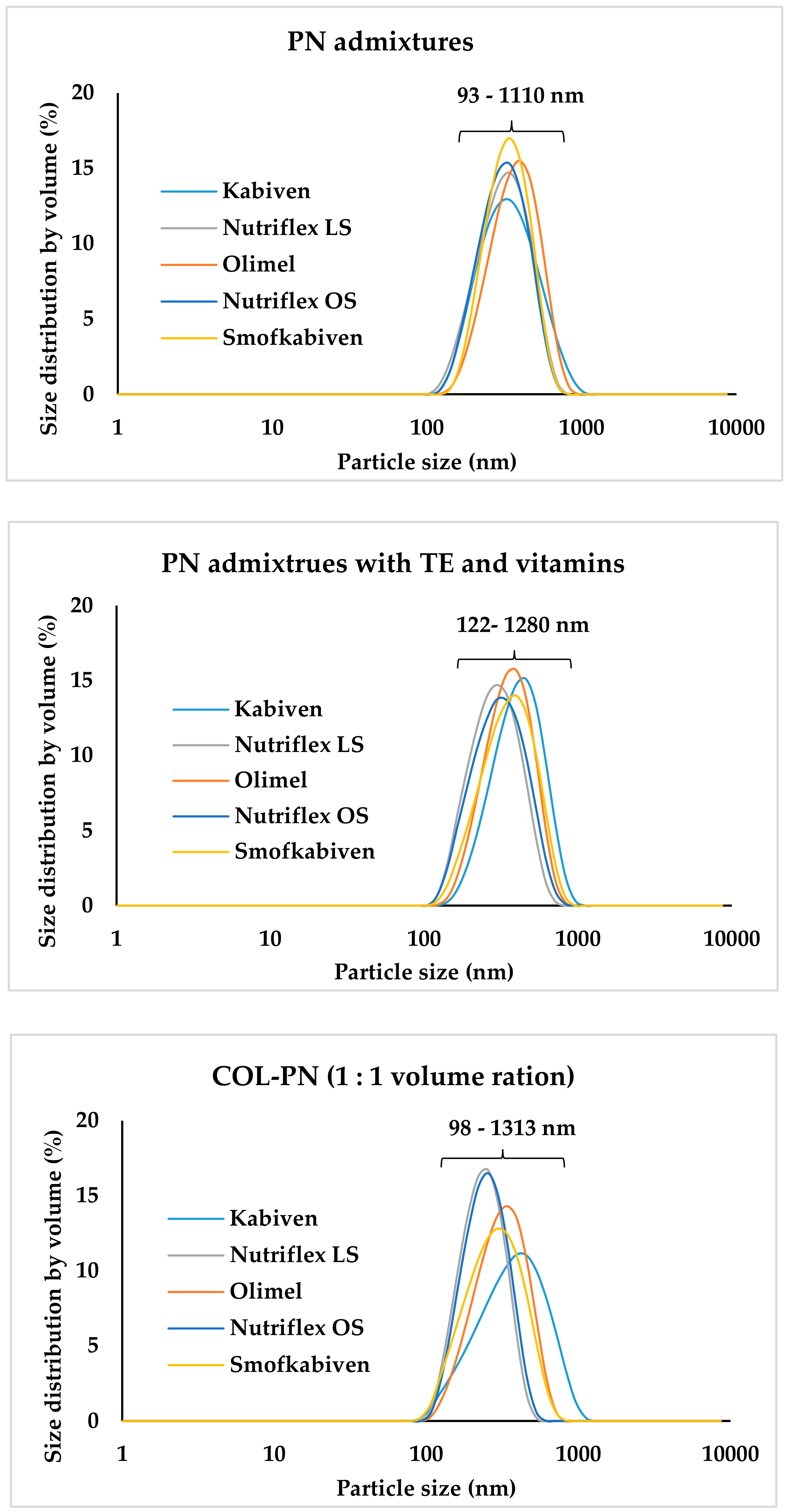
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Kanji, S.; Lam, J.; Johanson, C.; Singh, A.; Goddard, R.; Fairbairn, J.; Lloyd, T.; Monsour, D.; Kakal, J. Systematic review of physical and chemical compatibility of commonly used medications administered by continuous infusion in intensive care units. Crit. Care Med. 2010, 38, 1890–1898. [Google Scholar] [CrossRef]
- Trissel, L.A. Handbook on Injectable Drugs, 17th ed.; American Society of Health-System Pharmacists: Bethesda, MD, USA, 2013. [Google Scholar]
- Kaye, K.S.; Pogue, J.M.; Tran, T.B.; Nation, R.L.; Li, J. Agents of last resort: Polymyxin resistance. Infect. Dis. Clin. North. Am. 2016, 30, 391–414. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, M.; Grégoire, N.; Mégarbane, B.; Gobin, P.; Balayn, D.; Marchand, S.; Mimoz, O.; Coueta, W. Population pharmacokinetics of colistin methanesulfonate and colistin in critically ill patients with acute renal failure requiring intermittent hemodialysis. Antimicrob. Agents Chemother. 2016, 60, 1788–1793. [Google Scholar] [CrossRef] [PubMed]
- Bouchoud, L.; Fonzo-Christe, C.; Klingmüller, M.; Bonnabry, P. Compatibility of intravenous medications with parenteral nutrition: in vitro evaluation. J. Parenter. Enteral. Nutr. 2013, 37, 416–424. [Google Scholar] [CrossRef] [PubMed]
- Staven, V.; Iqbal, H.; Wang, S.; Grønlie, I.; Tho, I. Physical compatibility of total parenteral nutrition and drugs in Y-site administration to children from neonates to adolescents. J. Pharm. Pharmacol. 2016, 69, 448–462. [Google Scholar] [CrossRef]
- Gostyńska, A.; Stawny, M.; Dettlaff, K.; Jelińska, A. The interactions between ciprofloxacin and parenteral nutrition admixtures. Pharmaceutics 2019, 12, 27. [Google Scholar] [CrossRef]
- Stawny, M.; Nadolna, M.; Jelińska, A. In vitro compatibility studies of vancomycin with ready-to-use parenteral nutrition admixtures for safer clinical practice. Clin. Nutr. 2019, (in press). [CrossRef]
- Omotani, S.; Aoe, M.; Esaki, S.; Nagai, K.; Hatsuda, Y.; Mukai, J.; Teramachi, H.; Myotoku, M. Compatibility of intravenous fat emulsion with antibiotics for secondary piggyback infusion. Ann. Nutr. Metab. 2018, 73, 227–233. [Google Scholar] [CrossRef]
- Stawny, M.; Gostyńska, A.; Dettlaff, K.; Jelińska, A.; Główka, E.; Ogrodowczyk, M. Effect of Lipid Emulsion on Stability of Ampicillin in Total Parenteral Nutrition. Nutrients 2019, 11, 559. [Google Scholar] [CrossRef]
- Benlabed, M.; Perez, M.; Gaudy, R.; Genay, S.; Lannoy, D.; Barthelemy, C.; Odou, P.; Lebuffe, G.; Décaudin, B. Clinical implications of intravenous drug incompatibilities in critically ill patients. Anaesth. Crit. Care Pain Med. 2019, 38, 173–180. [Google Scholar] [CrossRef]
- Leopoldino, R.W.D.; Da Costa, T.X.; Da Costa, T.X.; Martins, R.R.; Oliveira, A.G. Potential drug incompatibilities in the neonatal intensive care unit: a network analysis approach. BMC Pharmacol. Toxicol. 2018, 19, 83. [Google Scholar] [CrossRef] [PubMed]
- European Directorate for Quality in Medicines and Healthcare (EDQM). 2.9.20. Particulate contamination: Visible particles. In European Pharmacopoeia 9.0; EDQM: Strasburg, France, 2017. [Google Scholar]
- The United States Pharmacopeia and National Formulary, 38th ed.; United States Pharmacopeial Convention: Rockville, MD, USA, 2015.
- Katip, W. Visual compatibility of colistin injection with other antibiotics during simulated Y-site administration. Am. J. Heal. Pharm. 2017, 74, 1099–1102. [Google Scholar] [CrossRef] [PubMed]
- Trissel, L.A.; Saenz, C.A.; Ogundele, A.B.; Ingram, D.S. Physical compatibility of pemetrexed disodium with other drugs during simulated Y-site administration. Am. J. Heal. Pharm. 2004, 61, 2289–2293. [Google Scholar] [CrossRef]
- Chan, P.; Bishop, A.; Kupiec, T.C.; Trissel, L.A.; Gole, D.; Jimidar, I.M.; Vermeersch, H. Compatibility of ceftobiprole medocaril with selected drugs during simulated Y-site administration. Am. J. Heal. Pharm. 2008, 65, 1545–1551. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, T.; Forest, J.-M.; LeClair, G. Compatibility of cloxacillin sodium with selected intravenous drugs during simulated y-site administration. Hosp. Pharm. 2015, 50, 214–220. [Google Scholar] [CrossRef] [PubMed]
- Lee, T.M.; Villareal, C.L.; Meyer, L.M. Y-Site compatibility of intravenous levetiracetam with commonly used critical care medications. Hosp. Pharm. 2019, (in press). [CrossRef]
- Staven, V.; Wang, S.; Grønlie, I.; Tho, I. Development and evaluation of a test program for Y-site compatibility testing of total parenteral nutrition and intravenous drugs. Nutr. J. 2015, 15, 29. [Google Scholar] [CrossRef]
- Wang, W. Tolerability of hypertonic injectables. Int. J. Pharm. 2015, 490, 308–315. [Google Scholar] [CrossRef]
- Gonyon, T.; Carter, P.W.; Dahlem, O.; DeNet, A.-R.; Owen, H.; Trouilly, J.-L. Container effects on the physicochemical properties of parenteral lipid emulsions. Nutrition 2008, 24, 1182–1188. [Google Scholar] [CrossRef]
- Washington, C. Stability of lipid emulsions for drug delivery. Adv. Drug Deliv. Rev. 1996, 20, 131–145. [Google Scholar] [CrossRef]
- Washington, C.; Athersuch, A.; Kynoch, D. The electrokinetic properties of phospholipid stabilized fat emulsions. IV. The effect of glucose and of pH. Int. J. Pharm. 1990, 64, 217–222. [Google Scholar] [CrossRef]
- Washington, C. The electrokinetic properties of phospholipid-stabilized fat emulsions. II. Droplet mobility in mixed electrolytes. Int. J. Pharm. 1990, 58, 13–17. [Google Scholar] [CrossRef]
- Télessy, I.; Balogh, J.; Csempesz, F.; Szente, V.; Dredán, J.; Zelkó, R. Comparison of the physicochemical properties of MCT-containing fat emulsions in total nutrient admixtures. Colloids Surf. B Biointerfaces 2009, 72, 75–79. [Google Scholar] [CrossRef] [PubMed]
- Hippalgaonkar, K.; Majumdar, S.; Kansara, V. Injectable lipid emulsions—advancements, opportunities and challenges. AAPS PharmSciTech 2010, 11, 1526–1540. [Google Scholar] [CrossRef] [PubMed]
- Garcia, J.; Garg, A.; Song, Y.; Fotios, A.; Andersen, C.; Garg, S. Compatibility of intravenous ibuprofen with lipids and parenteral nutrition, for use as a continuous infusion. PLoS ONE 2018, 13, e0190577. [Google Scholar] [CrossRef]
- Mediavilla, M.M.; Molina, A.; Navarro, L.; Grau, L.; Pujol, M.D.; Cardenete, J.; Cardona, D.; Riera, P. Physicochemical Compatibility of Amiodarone with Parenteral Nutrition. J. Parenter. Enter. Nutr. 2018, 43, 298–304. [Google Scholar] [CrossRef]
- Riera, P.; Garrido-Alejos, G.; Cardenete, J.; Moliner, E.; Zapico-Muniz, E.; Cardona, D.; Garin, N. Physicochemical stability and sterility of standard parenteral nutrition solutions and simulated y-site admixtures for neonates. Nutr. Clin. Pr. 2018, 33, 694–700. [Google Scholar] [CrossRef]
- Taxis, K.; Barber, N. Ethnographic study of incidence and severity of intravenous drug errors. BMJ 2003, 326, 684–687. [Google Scholar] [CrossRef]
- Tissot, E.; Cornette, C.; Limat, S.; Mourand, J.-L.; Becker, M.; Etievent, J.-P.; Dupond, J.-L.; Jacquet, M.; Woronoff-Lemsi, M.-C. Observational study of potential risk factors of medication administration errors. Pharm. World Sci. 2003, 25, 264–268. [Google Scholar] [CrossRef]
- Husson, E.; Crauste-Manciet, S.; Hadj-Salah, E.; Séguier, J.-C.; Brossard, D. Compatibility of parenteral drugs with commercialized total parenteral admixtures during simulated Y-site infusion. Nutr. Clin. Metabol. 2003, 17, 72–79. [Google Scholar] [CrossRef]
- Klang, M. PFAT5 and the evolution of lipid admixture stability. J. Parenter. Enter. Nutr. 2015, 39, 67S–71S. [Google Scholar] [CrossRef] [PubMed]
- Mathonet, S.; Mahler, H.-C.; Esswein, S.T.; Mazaheri, M.; Cash, P.; Wuchner, K.; Kallmeyer, G.; Das, T.K.; Finkler, C.; Lennard, A. A biopharmaceutical industry perspective on the control of visible particles in biotechnology derived injectable drug products. PDA J. Pharm. Sci. Technol. 2016, 70, 392–408. [Google Scholar] [CrossRef] [PubMed]
| Sample | COL:PN | pH ± SD | Osmolality ± SD | Zeta Potential ± SD |
|---|---|---|---|---|
| (mOsm/kg) | (mV) | |||
| 0 h * | 0 h → 10 min → 4 h | 0 h → 10 min → 4 h | ||
| Kabiven + COL | 0.5:1 | 5.49 ± 0.01 | (807 ± 4) → (855 ± 3) ** → (857 ± 4) ** | (−10.87 ± 0.15) → (−10.73 ± 0.25) → (−8.88 ± 0.03) ** |
| 1:1 | 5.5 ± 0.01 | (671 ± 3) → (672 ± 4) → (673 ± 3) | (−12.03 ± 0.55) → (−11.88 ± 0.0.4) → (−9.35 ± 0.46) ** | |
| 1.5:1 | 5.5 ± 0.01 | (567 ± 5) → (565 ± 2) → (567 ± 4) | (−12.83 ± 0.59) → (−12.12 ± 0.33) → (−7.81 ± 0.25) ** | |
| Nutriflex LS + COL | 0.5:1 | 5.45 ± 0.01 | (1304 ± 5) → (1298 ± 4) → (1290 ± 7) | (−17.90 ± 0.70) → (−18.02 ± 0.41) → (−18.50 ± 0.89) |
| 1:1 | 5.45 ± 0.01 | (967 ± 2) → (977 ± 4) → (982 ± 3) | (−19.70 ± 0.36) → (−19.82 ± 0.04) → (−20.03 ± 0.97) | |
| 1.5:1 | 5.46 ± 0.01 | (821 ± 3) → (817 ± 3) → (818 ± 3) | (−21.40 ± 0.61) → (−21.15 ± 0.33) → (−18.60 ± 0.70) | |
| Olimel + COL | 0.5:1 | 6.30 ± 0.00 | (1096 ± 8) → (1122 ± 6) → (1119 ± 5) | (−11.17 ± 0.21) → (−11.26 ± 0.08) → (−13.83 ± 0.21) |
| 1:1 | 6.30 ± 0.00 | (831 ± 2) → (855 ± 4) → (853 ± 1) | (−12.23 ± 0.80) → (−12.14 ± 0.24) → (−11.30 ± 0.44) | |
| 1.5:1 | 6.31 ± 0.01 | (712 ± 6) → (728 ± 4) → (731 ± 3) | (−15.87 ± 2.26) → (−15.12 ± 0.72) → (−10.27 ± 0.15) ** | |
| Nutriflex OS + COL | 0.5:1 | 5.60 ± 0.01 | (1299 ± 4) → (1322 ± 3) → (1329 ± 9) | (−9.48 ± 0.27) → (−9.57 ± 0.41) → (−9.65 ± 0.41) |
| 1:1 | 5.60 ± 0.01 | (964 ± 3) → (976 ± 4) → (975 ± 4) | (−9.58 ± 0.80) → (−9.61 ± 0.28) → (−9.79 ± 0.28) | |
| 1.5:1 | 5.60 ± 0.01 | (818 ± 0) → (807 ± 4) → (804 ± 0) | (−10.24 ± 0.62) → (−9.98 ± 0.38) → (−10.20 ± 0.66) | |
| Smofkabiven + COL | 0.5:1 | 5.45 ± 0.01 | (1180 ± 4) → (1188 ± 7) → (1197 ± 5) | (−9.23 ± 0.39) → (−9.11 ± 0.07) → (−7.22 ± 0.14) ** |
| 1:1 | 5.45 ± 0.01 | (885 ± 3) → (891 ± 3) → (897 ± 6) | (−10.09 ± 0.53) → (−9.62 ± 0.41) → (−8.59 ± 0.33) ** | |
| 1.5:1 | 5.45 ± 0.00 | (758 ± 4) → (755 ± 3) → (756 ± 2) | (−10.63 ± 0.61) → (−10.11 ± 0.12) → (−7.29 ± 0.45) ** |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stawny, M.; Gostyńska, A.; Nadolna, M.; Jelińska, A. Safe Practice of Y-Site Drug Administration: The Case of Colistin and Parenteral Nutrition. Pharmaceutics 2020, 12, 292. https://doi.org/10.3390/pharmaceutics12030292
Stawny M, Gostyńska A, Nadolna M, Jelińska A. Safe Practice of Y-Site Drug Administration: The Case of Colistin and Parenteral Nutrition. Pharmaceutics. 2020; 12(3):292. https://doi.org/10.3390/pharmaceutics12030292
Chicago/Turabian StyleStawny, Maciej, Aleksandra Gostyńska, Malwina Nadolna, and Anna Jelińska. 2020. "Safe Practice of Y-Site Drug Administration: The Case of Colistin and Parenteral Nutrition" Pharmaceutics 12, no. 3: 292. https://doi.org/10.3390/pharmaceutics12030292
APA StyleStawny, M., Gostyńska, A., Nadolna, M., & Jelińska, A. (2020). Safe Practice of Y-Site Drug Administration: The Case of Colistin and Parenteral Nutrition. Pharmaceutics, 12(3), 292. https://doi.org/10.3390/pharmaceutics12030292







