Pharmacotechnical Development of a Nasal Drug Delivery Composite Nanosystem Intended for Alzheimer’s Disease Treatment
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials (AD)
2.2. Characterization of the Active Pharmaceutical Ingredient
2.2.1. In Vitro Measurement of AChE and BuChE Activity
2.2.2. Parallel Artificial Membrane Permeability Assay (PAMPA)
2.3. Preparation of Liposomes
2.3.1. Formulation of Liposomes
2.3.2. Characterization of Liposomes by Dynamic Light Scattering
2.3.3. Entrapment Efficiency
2.3.4. UHPLC
2.4. Preparation of the In Situ-Forming Gel
2.4.1. Gel Formulation
2.4.2. Gel Characterization
Physicochemical Properties
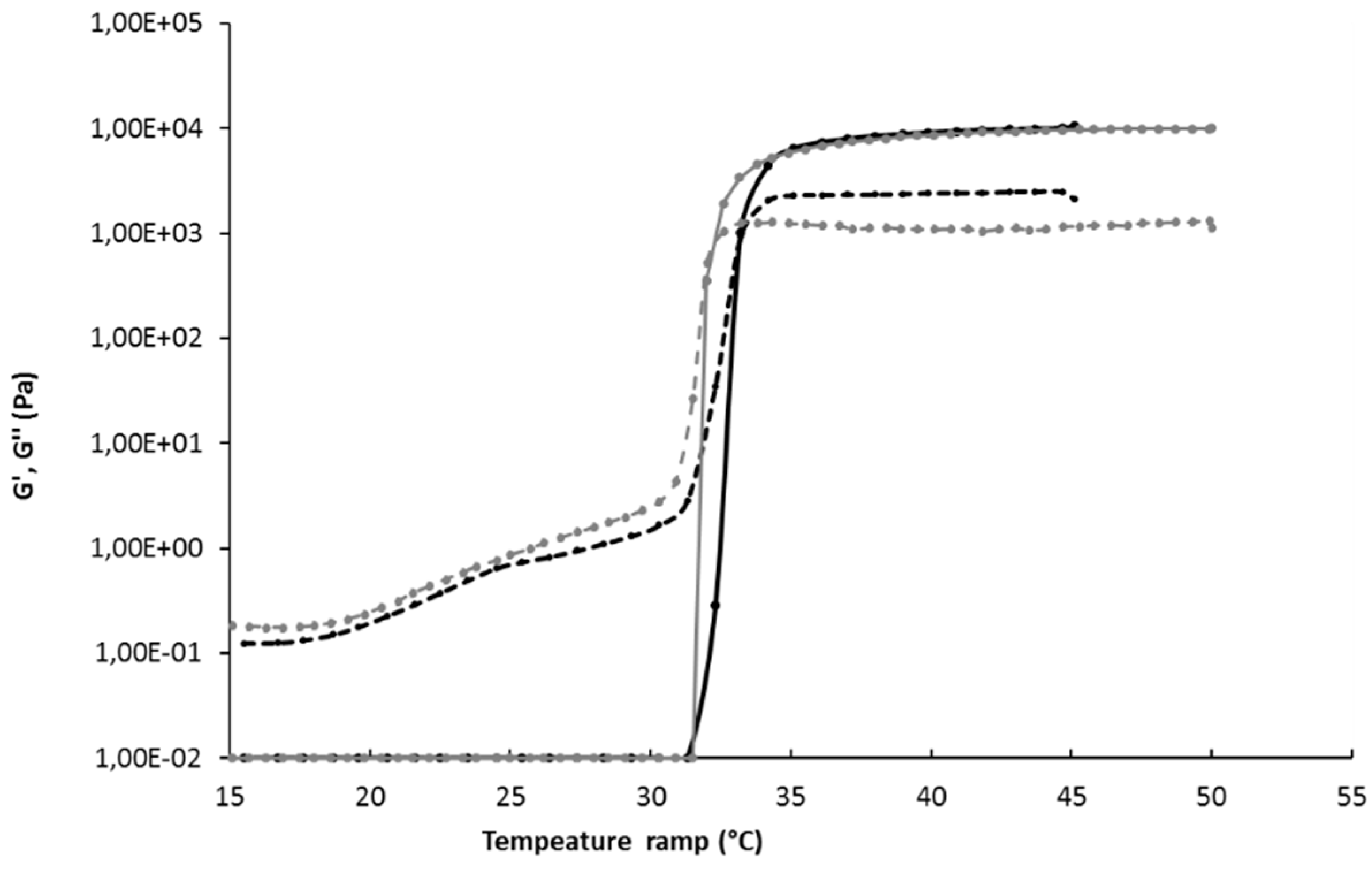
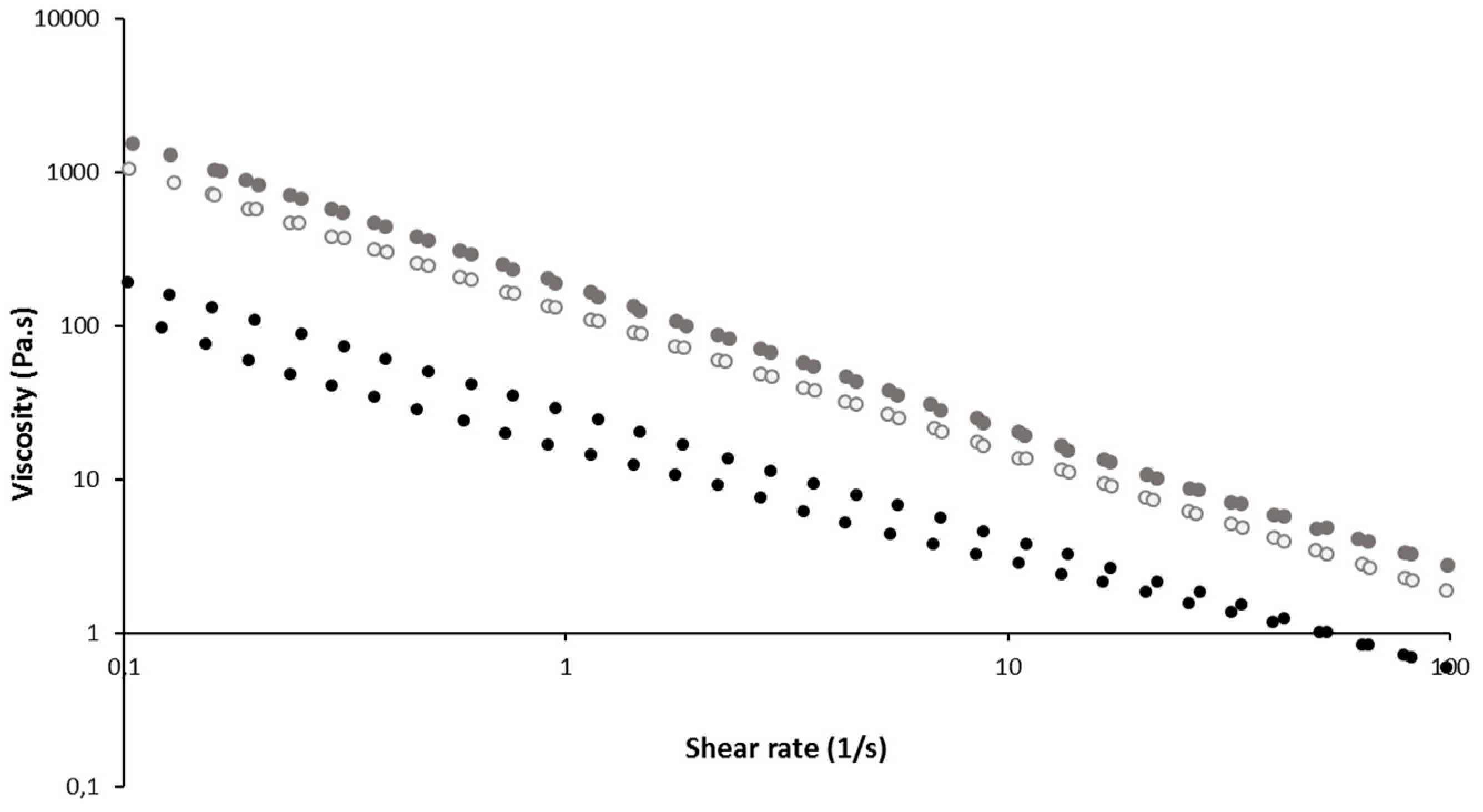
Rheological Evaluation
2.5. In Vitro Evaluation of Mucoadhesion
2.6. In Vitro Release Profile
2.7. Statistical Analysis
3. Results and Discussion
3.1. Characterization of the Active Pharmaceutical Ingredient (API)
3.2. Preparation of Liposomes
3.3. Preparation of in Situ-Forming Gel
3.4. Preparation of API Delivery Composite Formulation
3.5. Mucoadhesion Properties
3.6. API In Vitro Release
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Alzheimer’s Association 2016 Alzheimer’s disease facts and figures. Alzheimers Dement. 2016, 12, 459–509. [CrossRef]
- McGleenon, B.M.; Dynan, K.B.; Passmore, A.P. Acetylcholinesterase inhibitors in Alzheimer’s disease. Br. J. Clin. Pharm. 1999, 48, 471–480. [Google Scholar] [CrossRef]
- Darvesh, S.; Hopkins, D.A.; Geula, C. Neurobiology of butyrylcholinesterase. Nat. Rev. Neurosci. 2003, 4, 131–138. [Google Scholar] [CrossRef]
- Hoffmann, M.; Stiller, C.; Endres, E.; Scheiner, M.; Gunesch, S.; Sotriffer, C.; Maurice, T.; Decker, M. Highly Selective Butyrylcholinesterase Inhibitors with Tunable Duration of Action by Chemical Modification of Transferable Carbamate Units Exhibit Pronounced Neuroprotective Effect in an Alzheimer’s Disease Mouse Model. J. Med. Chem. 2019, 62, 9116–9140. [Google Scholar] [CrossRef]
- Jakki, S.L.; Senthil, V.; Yasam, V.R.; Chandrasekar, M.J.N.; Vijayaraghavan, C. The Blood Brain Barrier and its Role in Alzheimer’s Therapy: An Overview. Curr. Drug Targets 2018, 19, 155–169. [Google Scholar] [CrossRef]
- Alexander, A.; Agrawal, M.; Uddin, A.; Siddique, S.; Shehata, A.M.; Shaker, M.A.; Ata Ur Rahman, S.; Abdul, M.I.M.; Shaker, M.A. Recent expansions of novel strategies towards the drug targeting into the brain. Int. J. Nanomed. 2019, 14, 5895–5909. [Google Scholar] [CrossRef]
- Crowe, T.P.; Greenlee, M.H.W.; Kanthasamy, A.G.; Hsu, W.H. Mechanism of intranasal drug delivery directly to the brain. Life Sci. 2018, 195, 44–52. [Google Scholar] [CrossRef]
- Hallschmid, M.; Benedict, C.; Schultes, B.; Perras, B.; Fehm, H.-L.; Kern, W.; Born, J. Towards the therapeutic use of intranasal neuropeptide administration in metabolic and cognitive disorders. Regul. Pept. 2008, 149, 79–83. [Google Scholar] [CrossRef]
- Martins, P.P.; Smyth, H.D.C.; Cui, Z. Strategies to facilitate or block nose-to-brain drug delivery. Int. J. Pharm. 2019, 570, 118635. [Google Scholar] [CrossRef]
- Misra, A.; Jogani, V.; Jinturkar, K.; Vyas, T. Recent patents review on intranasal administration for CNS drug delivery. Recent Pat. Drug Deliv. 2008, 2, 25–40. [Google Scholar] [CrossRef]
- Fonseca-Santos, B.; Gremião, M.P.D.; Chorilli, M. Nanotechnology-based drug delivery systems for the treatment of Alzheimer’s disease. Int. J. Nanomedicine 2015, 10, 4981–5003. [Google Scholar] [CrossRef]
- Al Asmari, A.K.; Ullah, Z.; Tariq, M.; Fatani, A. Preparation, characterization, and in vivo evaluation of intranasally administered liposomal formulation of donepezil. Drug Des. Devel. 2016, 10, 205–215. [Google Scholar]
- Yang, Z.-Z.; Zhang, Y.-Q.; Wang, Z.-Z.; Wu, K.; Lou, J.-N.; Qi, X.-R. Enhanced brain distribution and pharmacodynamics of rivastigmine by liposomes following intranasal administration. Int. J. Pharm. 2013, 452, 344–354. [Google Scholar] [CrossRef]
- Arumugam, K.; Subramanian, G.S.; Mallayasamy, S.R.; Averineni, R.K.; Reddy, M.S.; Udupa, N. A study of rivastigmine liposomes for delivery into the brain through intranasal route. Acta Pharm. 2008, 58, 287–297. [Google Scholar] [CrossRef]
- Ismail, M.F.; Elmeshad, A.N.; Salem, N.A.-H. Potential therapeutic effect of nanobased formulation of rivastigmine on rat model of Alzheimer’s disease. Int. J. Nanomed. 2013, 8, 393–406. [Google Scholar] [CrossRef]
- Amidon, G.L.; Lennernäs, H.; Shah, V.P.; Crison, J.R. A theoretical basis for a biopharmaceutic drug classification: The correlation of in vitro drug product dissolution and in vivo bioavailability. Pharm. Res. 1995, 12, 413–420. [Google Scholar] [CrossRef]
- Agrawal, M.; Saraf, S.; Saraf, S.; Antimisiaris, S.G.; Chougule, M.B.; Shoyele, S.A.; Alexander, A. Nose-to-brain drug delivery: An update on clinical challenges and progress towards approval of anti-Alzheimer drugs. J. Control. Release 2018, 281, 139–177. [Google Scholar] [CrossRef]
- Bulbake, U.; Doppalapudi, S.; Kommineni, N.; Khan, W. Liposomal Formulations in Clinical Use: An Updated Review. Pharmaceutics 2017, 9, 12. [Google Scholar] [CrossRef]
- Sharma, D.; Ali, A.A.E.; Trivedi, L.R. An Updated Review on: Liposomes as drug delivery system. PharmaTutor 2018, 6, 50–62. [Google Scholar] [CrossRef]
- Peppas, N.A.; Bures, P.; Leobandung, W.; Ichikawa, H. Hydrogels in pharmaceutical formulations. Eur. J. Pharm. Biopharm. 2000, 50, 27–46. [Google Scholar] [CrossRef]
- Giuliano, E.; Paolino, D.; Fresta, M.; Cosco, D. Mucosal Applications of Poloxamer 407-Based Hydrogels: An Overview. Pharmaceutics 2018, 10, 159. [Google Scholar] [CrossRef]
- Soliman, G.M.; Fetih, G.; Abbas, A.M. Thermosensitive bioadhesive gels for the vaginal delivery of sildenafil citrate: In vitro characterization and clinical evaluation in women using clomiphene citrate for induction of ovulation. Drug Dev. Ind Pharm 2017, 43, 399–408. [Google Scholar] [CrossRef]
- Wang, Y.; Jiang, S.; Wang, H.; Bie, H. A mucoadhesive, thermoreversible in situ nasal gel of geniposide for neurodegenerative diseases. PLoS ONE 2017, 12, e0189478. [Google Scholar] [CrossRef]
- Aderibigbe, B.A. In Situ-Based Gels for Nose to Brain Delivery for the Treatment of Neurological Diseases. Pharmaceutics 2018, 10, 40. [Google Scholar] [CrossRef]
- Mura, P.; Mennini, N.; Nativi, C.; Richichi, B. In situ mucoadhesive-thermosensitive liposomal gel as a novel vehicle for nasal extended delivery of opiorphin. Eur. J. Pharm. Biopharm. 2018, 122, 54–61. [Google Scholar] [CrossRef]
- Jain, D.S.; Bajaj, A.N.; Athawale, R.B.; Shikhande, S.S.; Pandey, A.; Goel, P.N.; Gude, R.P.; Patil, S.; Raut, P. Thermosensitive PLA based nanodispersion for targeting brain tumor via intranasal route. Mater. Sci. Eng. C. Mater. Biol. Appl. 2016, 63, 411–421. [Google Scholar] [CrossRef]
- Bitter, C.; Suter-Zimmermann, K.; Surber, C. Nasal drug delivery in humans. Curr. Probl. Derm. 2011, 40, 20–35. [Google Scholar]
- Nasal preparations. In European Pharmacopoeia; ed. 9.5; EDQM Council of Europe: Strasbourg, France, 2008; pp. 940–942.
- Ellman, G.L.; Courtney, K.D.; Andres, V.; Feather-Stone, R.M. A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem. Pharm. 1961, 7, 88–95. [Google Scholar] [CrossRef]
- Rochais, C.; Lecoutey, C.; Gaven, F.; Giannoni, P.; Hamidouche, K.; Hedou, D.; Dubost, E.; Genest, D.; Yahiaoui, S.; Freret, T.; et al. Novel multitarget-directed ligands (MTDLs) with acetylcholinesterase (AChE) inhibitory and serotonergic subtype 4 receptor (5-HT4R) agonist activities as potential agents against Alzheimer’s disease: The design of donecopride. J. Med. Chem. 2015, 58, 3172–3187. [Google Scholar] [CrossRef]
- Jourdan, J.-P.; Since, M.; El Kihel, L.; Lecoutey, C.; Corvaisier, S.; Legay, R.; Sopkova-de Oliveira Santos, J.; Cresteil, T.; Malzert-Fréon, A.; Rochais, C.; et al. Novel benzylidenephenylpyrrolizinones with pleiotropic activities potentially useful in Alzheimer’s disease treatment. Eur. J. Med. Chem. 2016, 114, 365–379. [Google Scholar] [CrossRef]
- Bangham, A.D.; Standish, M.M.; Watkins, J.C. Diffusion of univalent ions across the lamellae of swollen phospholipids. J. Mol. Biol. 1965, 13, 238–252. [Google Scholar] [CrossRef]
- Schmolka, I.R. Artificial skin. I. Preparation and properties of pluronic F-127 gels for treatment of burns. J. Biomed. Mater. Res. 1972, 6, 571–582. [Google Scholar] [CrossRef] [PubMed]
- Greig, N.H.; Lahiri, D.K.; Sambamurti, K. Butyrylcholinesterase: An important new target in Alzheimer’s disease therapy. Int. Psychogeriatr. 2002, 14, 77–91. [Google Scholar] [CrossRef] [PubMed]
- Giacobini, E. Cholinesterase inhibitors: New roles and therapeutic alternatives. Pharm. Res. 2004, 50, 433–440. [Google Scholar] [CrossRef]
- Furukawa-Hibi, Y.; Alkam, T.; Nitta, A.; Matsuyama, A.; Mizoguchi, H.; Suzuki, K.; Moussaoui, S.; Yu, Q.-S.; Greig, N.H.; Nagai, T.; et al. Butyrylcholinesterase inhibitors ameliorate cognitive dysfunction induced by amyloid-β peptide in mice. Behav. Brain Res. 2011, 225, 222–229. [Google Scholar] [CrossRef]
- Elumalai, K.; Ali, M.A.; Elumalai, M.; Eluri, K.; Srinivasan, S. Acetylcholinesterase enzyme inhibitor activity of some novel pyrazinamide condensed 1,2,3,4-tetrahydropyrimidines. Biotechnol. Rep. 2015, 5, 1–6. [Google Scholar] [CrossRef]
- Dafferner, A.J.; Lushchekina, S.; Masson, P.; Xiao, G.; Schopfer, L.M.; Lockridge, O. Characterization of butyrylcholinesterase in bovine serum. Chem. -Biol. Interact. 2017, 266, 17–27. [Google Scholar] [CrossRef]
- Wu, J.; Tian, Y.; Wang, S.; Pistolozzi, M.; Jin, Y.; Zhou, T.; Roy, G.; Xu, L.; Tan, W. Design, synthesis and biological evaluation of bambuterol analogues as novel inhibitors of butyrylcholinesterase. Eur. J. Med. Chem. 2017, 126, 61–71. [Google Scholar] [CrossRef]
- Dighe, S.N.; Deora, G.S.; De la Mora, E.; Nachon, F.; Chan, S.; Parat, M.-O.; Brazzolotto, X.; Ross, B.P. Discovery and Structure–Activity Relationships of a Highly Selective Butyrylcholinesterase Inhibitor by Structure-Based Virtual Screening. J. Med. Chem. 2016, 59, 7683–7689. [Google Scholar] [CrossRef]
- Sun, H.; Nguyen, K.; Kerns, E.; Yan, Z.; Yu, K.R.; Shah, P.; Jadhav, A.; Xu, X. Highly predictive and interpretable models for PAMPA permeability. Bioorg. Med. Chem. 2017, 25, 1266–1276. [Google Scholar] [CrossRef]
- Masuda, Y. Cardiac effect of cholinesterase inhibitors used in Alzheimer’s disease--from basic research to bedside. Curr. Alzheimer Res. 2004, 1, 315–321. [Google Scholar] [CrossRef] [PubMed]
- Quraishi, M.S.; Jones, N.S.; Mason, J. The rheology of nasal mucus: A review. Clin. Otolaryngol. Allied Sci. 1998, 23, 403–413. [Google Scholar] [CrossRef] [PubMed]
- Ruel-Gariépy, E.; Leclair, G.; Hildgen, P.; Gupta, A.; Leroux, J.-C. Thermosensitive chitosan-based hydrogel containing liposomes for the delivery of hydrophilic molecules. J. Control. Release 2002, 82, 373–383. [Google Scholar] [CrossRef]
- Mayer, L.D.; Tai, L.C.; Ko, D.S.; Masin, D.; Ginsberg, R.S.; Cullis, P.R.; Bally, M.B. Influence of vesicle size, lipid composition, and drug-to-lipid ratio on the biological activity of liposomal doxorubicin in mice. Cancer Res. 1989, 49, 5922–5930. [Google Scholar]
- Ahmad, E.; Feng, Y.; Qi, J.; Fan, W.; Ma, Y.; He, H.; Xia, F.; Dong, X.; Zhao, W.; Lu, Y.; et al. Evidence of nose-to-brain delivery of nanoemulsions: Cargoes but not vehicles. Nanoscale 2017, 9, 1174–1183. [Google Scholar] [CrossRef]
- Oberdörster, G.; Oberdörster, E.; Oberdörster, J. Nanotoxicology: An Emerging Discipline Evolving from Studies of Ultrafine Particles. Environ. Health Perspect. 2005, 113, 823–839. [Google Scholar] [CrossRef]
- Morrison, E.E.; Costanzo, R.M. Morphology of the human olfactory epithelium. J. Comp. Neurol. 1990, 297, 1–13. [Google Scholar] [CrossRef]
- Yasir, M.; Sara, U.V.S.; Chauhan, I.; Gaur, P.K.; Singh, A.P.; Puri, D. Ameeduzzafar Solid lipid nanoparticles for nose to brain delivery of donepezil: Formulation, optimization by Box–Behnken design, in vitro and in vivo evaluation. Artif. CellsNanomed. Biotechnol. 2018, 46, 1838–1851. [Google Scholar]
- Bonaccorso, A.; Musumeci, T.; Serapide, M.F.; Pellitteri, R.; Uchegbu, I.F.; Puglisi, G. Nose to brain delivery in rats: Effect of surface charge of rhodamine B labeled nanocarriers on brain subregion localization. Colloids Surf. B Biointerfaces 2017, 154, 297–306. [Google Scholar] [CrossRef]
- Gänger, S.; Schindowski, K. Tailoring Formulations for Intranasal Nose-to-Brain Delivery: A Review on Architecture, Physico-Chemical Characteristics and Mucociliary Clearance of the Nasal Olfactory Mucosa. Pharmaceutics 2018, 10, 116. [Google Scholar] [CrossRef]
- Groo, A.-C.; Mircheva, K.; Bejaud, J.; Ailhas, C.; Panaiotov, I.; Saulnier, P.; Ivanova, T.; Lagarce, F. Development of 2D and 3D mucus models and their interactions with mucus-penetrating paclitaxel-loaded lipid nanocapsules. Pharm. Res. 2014, 31, 1753–1765. [Google Scholar] [CrossRef] [PubMed]
- Groo, A.-C.; Lagarce, F. Mucus models to evaluate nanomedicines for diffusion. Drug Discov. Today 2014, 19, 1097–1108. [Google Scholar] [CrossRef] [PubMed]
- Schlegel, P.N.; Histrelin Study Group. Efficacy and safety of histrelin subdermal implant in patients with advanced prostate cancer. J. Urol. 2006, 175, 1353–1358. [Google Scholar] [CrossRef]
- Thorn, R.M.S.; Greenman, J.; Austin, A. An in vitro study of antimicrobial activity and efficacy of iodine-generating hydrogel dressings. J. Wound Care 2006, 15, 305–310. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.-Q.; Kim, Y.-M.; Song, S.-C. Injectable and quadruple-functional hydrogel as an alternative to intravenous delivery for enhanced tumor targeting. Acs Appl. Mater. Interfaces 2019, 11, 34634–34644. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Yang, F.; Feng, L.; Yang, L.; Chen, L.; Wei, G.; Lu, W. In vivo retention of poloxamer-based in situ hydrogels for vaginal application in mouse and rat models. Acta Pharm. Sin. B 2017, 7, 502–509. [Google Scholar] [CrossRef]
- Russo, E.; Villa, C. Poloxamer Hydrogels for Biomedical Applications. Pharmaceutics 2019, 11, 671. [Google Scholar] [CrossRef]
- Singh-Joy, S.D.; McLain, V.C. Safety assessment of poloxamers 101, 105, 108, 122, 123, 124, 181, 182, 183, 184, 185, 188, 212, 215, 217, 231, 234, 235, 237, 238, 282, 284, 288, 331, 333, 334, 335, 338, 401, 402, 403, and 407, poloxamer 105 benzoate, and poloxamer 182 dibenzoate as used in cosmetics. Int. J. Toxicol. 2008, 27, 93–128. [Google Scholar]
- Van Tomme, S.R.; Storm, G.; Hennink, W.E. In situ gelling hydrogels for pharmaceutical and biomedical applications. Int. J. Pharm. 2008, 355, 1–18. [Google Scholar] [CrossRef]
- Pereira, G.G.; Dimer, F.A.; Guterres, S.S.; Kechinski, C.P.; Granada, J.E.; Cardozo, N.S.M. Formulation and characterization of poloxamer 407®: Thermoreversible gel containing polymeric microparticles and hyaluronic acid. Química Nova 2013, 36, 1121–1125. [Google Scholar] [CrossRef]
- Dumortier, G.; Grossiord, J.L.; Agnely, F.; Chaumeil, J.C. A review of poloxamer 407 pharmaceutical and pharmacological characteristics. Pharm. Res. 2006, 23, 2709–2728. [Google Scholar] [CrossRef] [PubMed]
- Bochot, A.; Fattal, E.; Gulik, A.; Couarraze, G.; Couvreur, P. Liposomes Dispersed Within a Thermosensitive Gel: A New Dosage Form for Ocular Delivery of Oligonucleotides. Pharm. Res. 1998, 15, 1364–1369. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.; Lee, K.Y.C. Interaction of Poloxamers with Liposomes: An Isothermal Titration Calorimetry Study. J. Phys. Chem. B 2009, 113, 15522–15531. [Google Scholar] [CrossRef] [PubMed]
- Cho, E.C.; Lim, H.J.; Shim, J.; Park, J.Y.; Dan, N.; Kim, J.; Chang, I.-S. Effect of polymer characteristics on structure of polymer–liposome complexes. J. Colloid Interface Sci. 2007, 311, 243–252. [Google Scholar] [CrossRef] [PubMed]
- Pembouong, G.; Morellet, N.; Kral, T.; Hof, M.; Scherman, D.; Bureau, M.-F.; Mignet, N. A comprehensive study in triblock copolymer membrane interaction. J. Control. Release 2011, 151, 57–64. [Google Scholar] [CrossRef] [PubMed]
- Tian, J.L.; Ke, X.; Chen, Z.; Wang, C.J.; Zhang, Y.; Zhong, T.C. Melittin liposomes surface modified with poloxamer 188: In vitro characterization and in vivo evaluation. Pharmazie 2011, 66, 362–367. [Google Scholar]
- Karavasili, C.; Fatouros, D.G. Smart materials: In situ gel-forming systems for nasal delivery. Drug Discov. Today 2016, 21, 157–166. [Google Scholar] [CrossRef]
- Tuğcu-Demiröz, F. Development of in situ poloxamer-chitosan hydrogels for vaginal drug delivery of benzydamine hydrochloride: Textural, mucoadhesive and in vitro release properties. Marmara Pharm. J. 2017, 21, 762–770. [Google Scholar] [CrossRef]
- Mourtas, S.; Fotopoulou, S.; Duraj, S.; Sfika, V.; Tsakiroglou, C.; Antimisiaris, S.G. Liposomal drugs dispersed in hydrogels. Effect of liposome, drug and gel properties on drug release kinetics. Colloids Surf. B Biointerfaces 2007, 55, 212–221. [Google Scholar] [CrossRef]
- Lindner, L.H.; Hossann, M. Factors affecting drug release from liposomes. Curr. Opin. Drug Discov. Devel. 2010, 13, 111–123. [Google Scholar]
 low T°sol-gel transition,
low T°sol-gel transition,  acceptable T°sol-gel transition,
acceptable T°sol-gel transition,  high T°sol-gel transition; (b):
high T°sol-gel transition; (b):  acceptable osmolarity,
acceptable osmolarity,  High osmolarity.
High osmolarity.
 low T°sol-gel transition,
low T°sol-gel transition,  acceptable T°sol-gel transition,
acceptable T°sol-gel transition,  high T°sol-gel transition; (b):
high T°sol-gel transition; (b):  acceptable osmolarity,
acceptable osmolarity,  High osmolarity.
High osmolarity.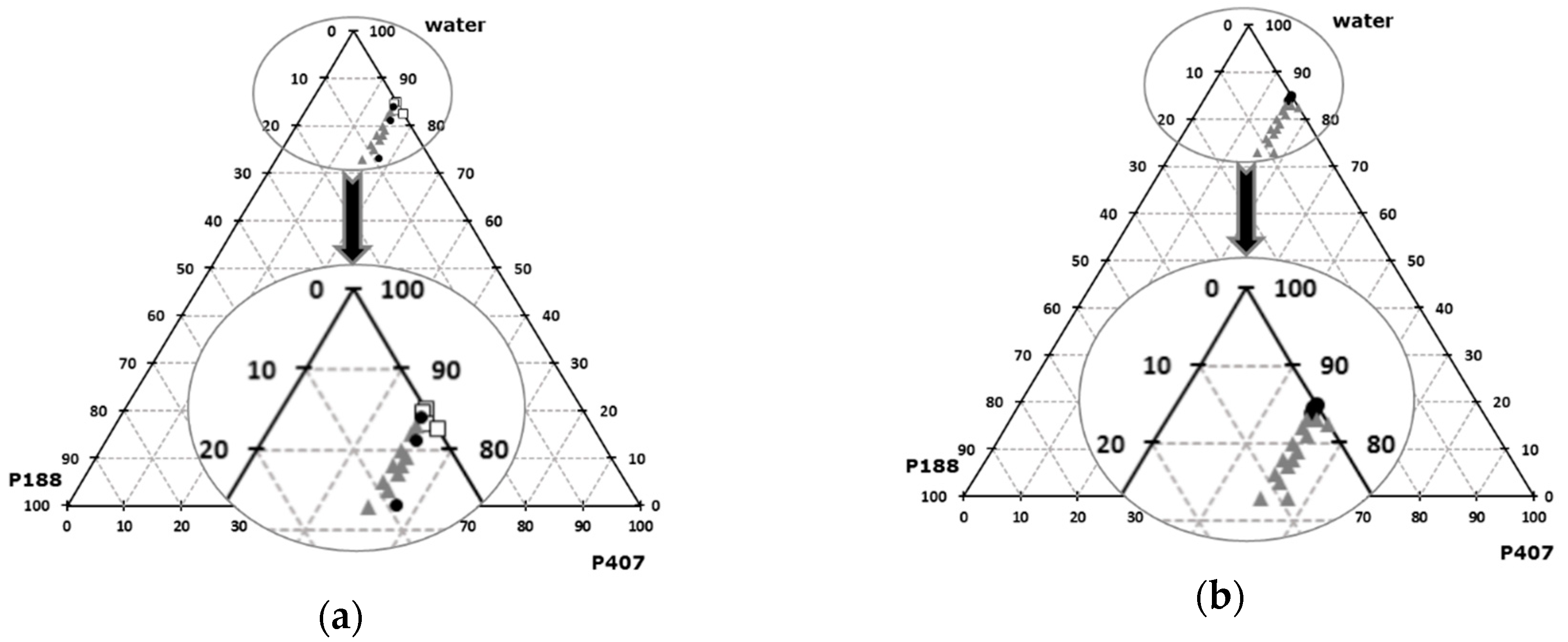

 detachment Force (mN),
detachment Force (mN),  adhesion work (mN.mm).
adhesion work (mN.mm).
 detachment Force (mN),
detachment Force (mN),  adhesion work (mN.mm).
adhesion work (mN.mm).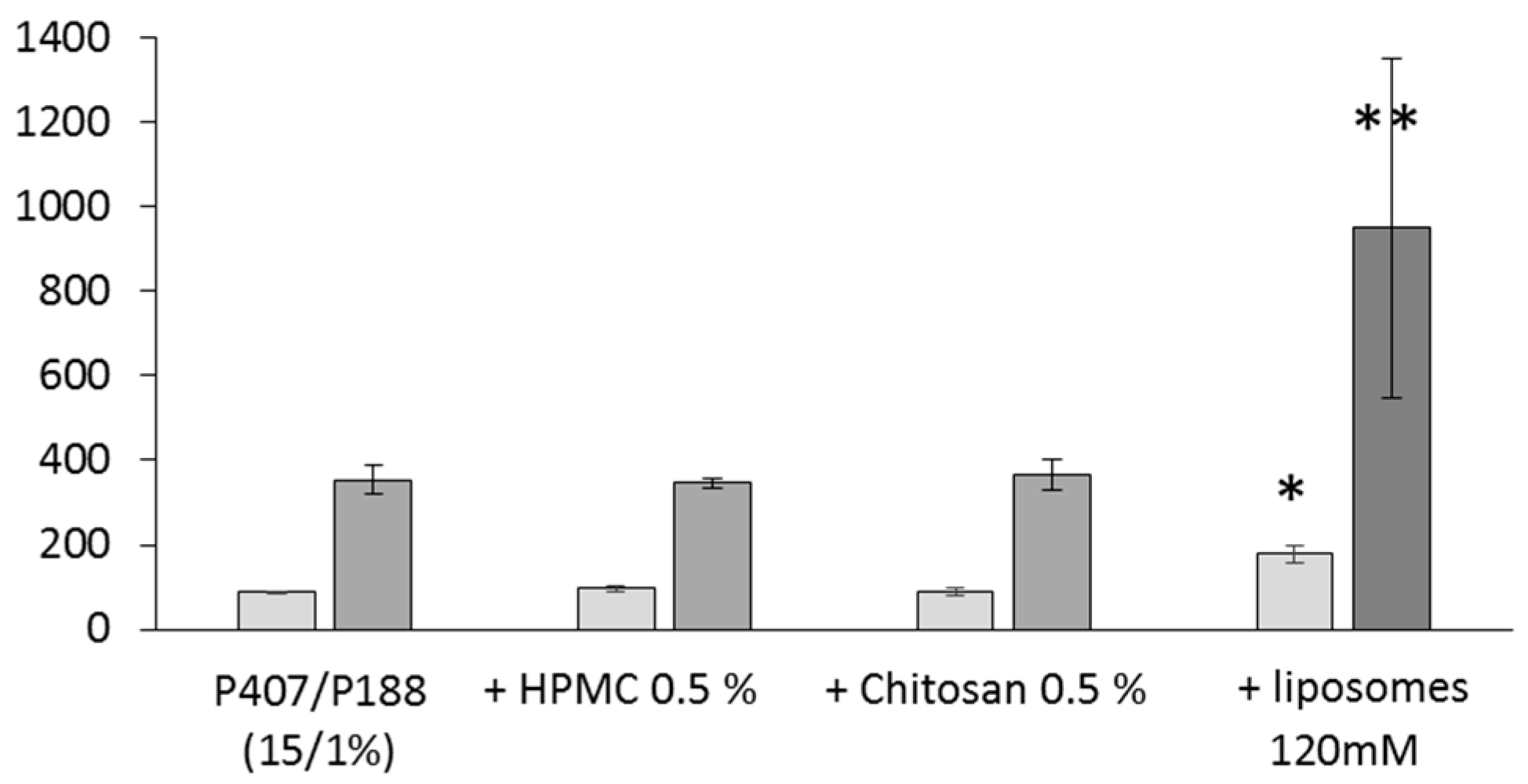
 Solution API 1 mg/mL,
Solution API 1 mg/mL,  API-loaded liposomes ,
API-loaded liposomes ,  Gel with API 1mg/mL,
Gel with API 1mg/mL,  API-loaded Liposomes in gel.
API-loaded Liposomes in gel.
 Solution API 1 mg/mL,
Solution API 1 mg/mL,  API-loaded liposomes ,
API-loaded liposomes ,  Gel with API 1mg/mL,
Gel with API 1mg/mL,  API-loaded Liposomes in gel.
API-loaded Liposomes in gel.
| (h) AChE IC50 (nM | (eq) BuChE IC50 (nM) | |
|---|---|---|
| Donepezil | 11 ± 3 | - |
| Tacrine | - | 2 ± 1 |
| API | 14,520 ± 1,485 | 573 ± 40 |
| Hydrodynamic Diameter (nm) | PDI | ZP (mV) | %EE | %DL | Concentration (mg/mL) | |
|---|---|---|---|---|---|---|
| Blank liposomes | 119.0 ± 0.7 | 0.032 ± 0.004 | −5.5 ± 0.3 | - | - | - |
| API-Lip | 114.9 ± 2.4 | 0.048 ± 0.020 | −11.2 ± 1.4 | 11.1 ± 1.0 | 1.4 ± 0.1 | 1.2 ± 0.1 |
| Blank liposomes in gel | 140.8 ± 0.5 | 0.078 ± 0.004 | −6.6 ± 0.5 | - | - | - |
| Optimum | P407/P188 (15/1 wt %) | +API 1 mg/mL | +Liposomes 60 mM | +Liposomes 60 mM + API | |
|---|---|---|---|---|---|
| Tsol-gel (°C) | 32–35 | 34.5 ± 0.3 | 34.8 ± 1.2 | 30.9 ± 1.0 | 32.6 ± 1.0 |
| Osmolarity (mOsm) | 280 ± 20 | 277 ± 4 | 287 ± 6 | 280 ± 5 | 293 ± 5 |
| pH | 6 ± 3 | 6.5 | 6.5 | 5.5 | 5.5 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Adnet, T.; Groo, A.-C.; Picard, C.; Davis, A.; Corvaisier, S.; Since, M.; Bounoure, F.; Rochais, C.; Le Pluart, L.; Dallemagne, P.; et al. Pharmacotechnical Development of a Nasal Drug Delivery Composite Nanosystem Intended for Alzheimer’s Disease Treatment. Pharmaceutics 2020, 12, 251. https://doi.org/10.3390/pharmaceutics12030251
Adnet T, Groo A-C, Picard C, Davis A, Corvaisier S, Since M, Bounoure F, Rochais C, Le Pluart L, Dallemagne P, et al. Pharmacotechnical Development of a Nasal Drug Delivery Composite Nanosystem Intended for Alzheimer’s Disease Treatment. Pharmaceutics. 2020; 12(3):251. https://doi.org/10.3390/pharmaceutics12030251
Chicago/Turabian StyleAdnet, Thomas, Anne-Claire Groo, Céline Picard, Audrey Davis, Sophie Corvaisier, Marc Since, Frédéric Bounoure, Christophe Rochais, Loïc Le Pluart, Patrick Dallemagne, and et al. 2020. "Pharmacotechnical Development of a Nasal Drug Delivery Composite Nanosystem Intended for Alzheimer’s Disease Treatment" Pharmaceutics 12, no. 3: 251. https://doi.org/10.3390/pharmaceutics12030251
APA StyleAdnet, T., Groo, A.-C., Picard, C., Davis, A., Corvaisier, S., Since, M., Bounoure, F., Rochais, C., Le Pluart, L., Dallemagne, P., & Malzert-Fréon, A. (2020). Pharmacotechnical Development of a Nasal Drug Delivery Composite Nanosystem Intended for Alzheimer’s Disease Treatment. Pharmaceutics, 12(3), 251. https://doi.org/10.3390/pharmaceutics12030251







