Therapeutic Cancer Vaccination with Ex Vivo RNA-Transfected Dendritic Cells—An Update
Abstract
1. Tumors and the Immune System
2. Therapeutic and Preventive Vaccination
3. Dendritic Cells as Cancer Vaccine
3.1. Dendritic Cell Maturation
3.2. Loading Dendritic Cells with Antigen
3.3. RNA-Transfection of Dendritic Cells
3.4. The Total Tumor RNA Approach
4. From the Bench to the Bedside
4.1. Clinical Efficacy
4.2. Safety of DC Vaccine Therapy
4.3. Challenges and Future Perspectives of DC Vaccine Therapy
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ichim, C.V. Revisiting immunosurveillance and immunostimulation: Implications for cancer Immunother. J. Transl. Med. 2005, 3, 8. [Google Scholar] [CrossRef]
- Wiemann, B.; Starnes, C.O. Coley’s toxins, tumor necrosis factor and cancer research: A historical perspective. Pharmacol. Ther. 1994, 64, 529–564. [Google Scholar] [CrossRef]
- Burnet, F.M. The concept of immunological surveillance. Prog. Exp. Tumor Res. 1970, 13, 1–27. [Google Scholar] [PubMed]
- Thomas, L. Discussion. In Cellular and Humoral Aspects of the Hypersensitive States; Lawrence, H.S., Ed.; Hoeber-Harper: New York, NY, USA, 1959. [Google Scholar]
- Dunn, G.P.; Bruce, A.T.; Ikeda, H.; Old, L.J.; Schreiber, R.D. Cancer immunoediting: From immunosurveillance to tumor escape. Nat. Immunol. 2002, 3, 991–998. [Google Scholar] [CrossRef] [PubMed]
- Swann, J.B.; Smyth, M.J. Immune surveillance of tumors. J. Clin. Investig. 2007, 117, 1137–1146. [Google Scholar] [CrossRef] [PubMed]
- Steinman, R.M.; Cohn, Z.A. Identification of a novel cell type in peripheral lymphoid organs of mice. I. Morphology, quantitation, tissue distribution. J. Exp. Med. 1973, 137, 1142–1162. [Google Scholar] [CrossRef] [PubMed]
- Aarntzen, E.H.; Figdor, C.G.; Adema, G.J.; Punt, C.J.; de Vries, I.J. Dendritic cell vaccination and immune monitoring. Cancer Immunol. Immunother. 2008, 57, 1559–1568. [Google Scholar] [CrossRef] [PubMed]
- Banchereau, J.; Steinman, R.M. Dendritic cells and the control of immunity. Nature 1998, 392, 245–252. [Google Scholar] [CrossRef] [PubMed]
- Palucka, K.; Banchereau, J. Cancer immunotherapy via dendritic cells. Nat. Rev. Cancer 2012, 12, 265–277. [Google Scholar] [CrossRef]
- Mayordomo, J.I.; Zorina, T.; Storkus, W.J.; Zitvogel, L.; Celluzzi, C.; Falo, L.D.; Melief, C.J.; Ildstad, S.T.; Kast, W.M.; DeLeo, A.B. Bone marrow-derived dendritic cells pulsed with synthetic tumour peptides elicit protective and therapeutic antitumour immunity. Nat. Med. 1995, 1, 1297–1302. [Google Scholar] [CrossRef]
- Zitvogel, L.; Mayordomo, J.I.; Tjandrawan, T.; DeLeo, A.B.; Clarke, M.R.; Lotze, M.T.; Storkus, W.J. Therapy of murine tumors with tumor peptide-pulsed dendritic cells: Dependence on T cells, B7 costimulation, and T helper cell 1-associated cytokines. J. Exp. Med. 1996, 183, 87–97. [Google Scholar] [CrossRef] [PubMed]
- Hsu, F.J.; Benike, C.; Fagnoni, F.; Liles, T.M.; Czerwinski, D.; Taidi, B.; Engleman, E.G.; Levy, R. Vaccination of patients with B-cell lymphoma using autologous antigen-pulsed dendritic cells. Nat. Med. 1996, 2, 52–58. [Google Scholar] [CrossRef] [PubMed]
- Nestle, F.O.; Banchereau, J.; Hart, D. Dendritic cells: On the move from bench to bedside. Nat. Med. 2001, 7, 761–765. [Google Scholar] [CrossRef] [PubMed]
- Bender, A.; Sapp, M.; Schuler, G.; Steinman, R.M.; Bhardwaj, N. Improved methods for the generation of dendritic cells from nonproliferating progenitors in human blood. J. Immunol. Methods 1996, 196, 121–135. [Google Scholar] [CrossRef]
- Nestle, F.O.; Alijagic, S.; Gilliet, M.; Sun, Y.; Grabbe, S.; Dummer, R.; Burg, G.; Schadendorf, D. Vaccination of melanoma patients with peptide- or tumor lysate-pulsed dendritic cells. Nat. Med. 1998, 4, 328–332. [Google Scholar] [CrossRef]
- Thurner, B.; Haendle, I.; Roder, C.; Dieckmann, D.; Keikavoussi, P.; Jonuleit, H.; Bender, A.; Maczek, C.; Schreiner, D.; von den, D.P.; et al. Vaccination with mage-3A1 peptide-pulsed mature, monocyte-derived dendritic cells expands specific cytotoxic T cells and induces regression of some metastases in advanced stage IV melanoma. J. Exp. Med. 1999, 190, 1669–1678. [Google Scholar] [CrossRef]
- De Vries, I.J.; Bernsen, M.R.; Lesterhuis, W.J.; Scharenborg, N.M.; Strijk, S.P.; Gerritsen, M.J.; Ruiter, D.J.; Figdor, C.G.; Punt, C.J.; Adema, G.J. Immunomonitoring tumor-specific T cells in delayed-type hypersensitivity skin biopsies after dendritic cell vaccination correlates with clinical outcome. J. Clin. Oncol. 2005, 23, 5779–5787. [Google Scholar] [CrossRef]
- Banchereau, J.; Palucka, A.K.; Dhodapkar, M.; Burkeholder, S.; Taquet, N.; Rolland, A.; Taquet, S.; Coquery, S.; Wittkowski, K.M.; Bhardwaj, N.; et al. Immune and clinical responses in patients with metastatic melanoma to CD34(+) progenitor-derived dendritic cell vaccine. Cancer Res. 2001, 61, 6451–6458. [Google Scholar]
- Beck, B.; Dorfel, D.; Lichtenegger, F.S.; Geiger, C.; Lindner, L.; Merk, M.; Schendel, D.J.; Subklewe, M. Effects of TLR agonists on maturation and function of 3-day dendritic cells from AML patients in complete remission. J. Transl. Med. 2011, 9, 151. [Google Scholar] [CrossRef]
- Kalinski, P.; Okada, H. Polarized dendritic cells as cancer vaccines: Directing effector-type T cells to tumors. Semin. Immunol. 2010, 22, 173–182. [Google Scholar] [CrossRef]
- Steinman, R.M.; Hemmi, H. Dendritic cells: Translating innate to adaptive immunity. Curr. Top. Microbiol. Immunol. 2006, 311, 17–58. [Google Scholar] [PubMed]
- Gallucci, S.; Matzinger, P. Danger signals: SOS to the immune system. Curr. Opin. Immunol. 2001, 13, 114–119. [Google Scholar] [CrossRef]
- De Vries, I.J.; Krooshoop, D.J.; Scharenborg, N.M.; Lesterhuis, W.J.; Diepstra, J.H.; Van Muijen, G.N.; Strijk, S.P.; Ruers, T.J.; Boerman, O.C.; Oyen, W.J.; et al. Effective migration of antigen-pulsed dendritic cells to lymph nodes in melanoma patients is determined by their maturation state. Cancer Res. 2003, 63, 12–17. [Google Scholar] [PubMed]
- Dhodapkar, M.V.; Steinman, R.M.; Krasovsky, J.; Munz, C.; Bhardwaj, N. Antigen-specific inhibition of effector T cell function in humans after injection of immature dendritic cells. J. Exp. Med. 2001, 193, 233–238. [Google Scholar] [CrossRef] [PubMed]
- Romani, N.; Reider, D.; Heuer, M.; Ebner, S.; Kampgen, E.; Eibl, B.; Niederwieser, D.; Schuler, G. Generation of mature dendritic cells from human blood. An improved method with special regard to clinical applicability. J. Immunol. Methods 1996, 196, 137–151. [Google Scholar] [CrossRef]
- Jonuleit, H.; Kuhn, U.; Muller, G.; Steinbrink, K.; Paragnik, L.; Schmitt, E.; Knop, J.; Enk, A.H. Pro-inflammatory cytokines and prostaglandins induce maturation of potent immunostimulatory dendritic cells under fetal calf serum-free conditions. Eur. J. Immunol. 1997, 27, 3135–3142. [Google Scholar] [CrossRef]
- Melief, C.J.; van der Burg, S.H. Immunotherapy of established (pre)malignant disease by synthetic long peptide vaccines. Nat. Rev. Cancer 2008, 8, 351–360. [Google Scholar] [CrossRef]
- Rains, N.; Cannan, R.J.; Chen, W.; Stubbs, R.S. Development of a dendritic cell (DC)-based vaccine for patients with advanced colorectal cancer. Hepatogastroenterology 2001, 48, 347–351. [Google Scholar]
- Nair, S.K.; Morse, M.; Boczkowski, D.; Cumming, R.I.; Vasovic, L.; Gilboa, E.; Lyerly, H.K. Induction of tumor-specific cytotoxic T lymphocytes in cancer patients by autologous tumor RNA-transfected dendritic cells. Ann. Surg. 2002, 235, 540–549. [Google Scholar] [CrossRef]
- Su, Z.; Dannull, J.; Heiser, A.; Yancey, D.; Pruitt, S.; Madden, J.; Coleman, D.; Niedzwiecki, D.; Gilboa, E.; Vieweg, J. Immunological and clinical responses in metastatic renal cancer patients vaccinated with tumor RNA-transfected dendritic cells. Cancer Res. 2003, 63, 2127–2133. [Google Scholar]
- Caruso, D.A.; Orme, L.M.; Neale, A.M.; Radcliff, F.J.; Amor, G.M.; Maixner, W.; Downie, P.; Hassall, T.E.; Tang, M.L.; Ashley, D.M. Results of a phase 1 study utilizing monocyte-derived dendritic cells pulsed with tumor RNA in children and young adults with brain cancer. Neuro Oncol. 2004, 6, 236–246. [Google Scholar] [CrossRef] [PubMed]
- Caruso, D.A.; Orme, L.M.; Amor, G.M.; Neale, A.M.; Radcliff, F.J.; Downie, P.; Tang, M.L.; Ashley, D.M. Results of a Phase I study utilizing monocyte-derived dendritic cells pulsed with tumor RNA in children with Stage 4 neuroblastoma. Cancer 2005, 103, 1280–1291. [Google Scholar] [CrossRef] [PubMed]
- Dannull, J.; Su, Z.; Rizzieri, D.; Yang, B.K.; Coleman, D.; Yancey, D.; Zhang, A.; Dahm, P.; Chao, N.; Gilboa, E.; et al. Enhancement of vaccine-mediated antitumor immunity in cancer patients after depletion of regulatory T cells. J. Clin. Investig. 2005, 115, 3623–3633. [Google Scholar] [CrossRef]
- Markovic, S.N.; Dietz, A.B.; Greiner, C.W.; Maas, M.L.; Butler, G.W.; Padley, D.J.; Bulur, P.A.; Allred, J.B.; Creagan, E.T.; Ingle, J.N.; et al. Preparing clinical-grade myeloid dendritic cells by electroporation-mediated transfection of in vitro amplified tumor-derived mRNA and safety testing in stage IV malignant melanoma. J. Transl. Med. 2006, 4, 35. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Amin, A.; Dudek, A.Z.; Logan, T.F.; Lance, R.S.; Holzbeierlein, J.M.; Knox, J.J.; Master, V.A.; Pal, S.K.; Miller, W.H., Jr.; Karsh, L.I.; et al. Survival with AGS-003, an autologous dendritic cell-based immunotherapy, in combination with sunitinib in unfavorable risk patients with advanced renal cell carcinoma (RCC): Phase 2 study results. J. Immunother. Cancer 2015, 3, 14. [Google Scholar] [CrossRef] [PubMed]
- Kyte, J.A.; Aamdal, S.; Dueland, S.; Saeboe-Larsen, S.; Inderberg, E.M.; Madsbu, U.E.; Skovlund, E.; Gaudernack, G.; Kvalheim, G. Immune response and long-term clinical outcome in advanced melanoma patients vaccinated with tumor-mRNA-transfected dendritic cells. Oncoimmunology 2016, 5, e1232237. [Google Scholar] [CrossRef]
- Vik-Mo, E.O.; Nyakas, M.; Mikkelsen, B.V.; Moe, M.C.; Due-Tonnesen, P.; Suso, E.M.; Saeboe-Larssen, S.; Sandberg, C.; Brinchmann, J.E.; Helseth, E.; et al. Therapeutic vaccination against autologous cancer stem cells with mRNA-transfected dendritic cells in patients with glioblastoma. Cancer Immunol. Immunother. 2013, 62, 1499–1509. [Google Scholar] [CrossRef]
- Mu, L.J.; Kyte, J.A.; Kvalheim, G.; Aamdal, S.; Dueland, S.; Hauser, M.; Hammerstad, H.; Waehre, H.; Raabe, N.; Gaudernack, G. Immunotherapy with allotumour mRNA-transfected dendritic cells in androgen-resistant prostate cancer patients. Br. J. Cancer 2005, 93, 749–756. [Google Scholar] [CrossRef]
- Wilgenhof, S.; Corthals, J.; Van Nuffel, A.M.; Benteyn, D.; Heirman, C.; Bonehill, A.; Thielemans, K.; Neyns, B. Long-term clinical outcome of melanoma patients treated with messenger RNA-electroporated dendritic cell therapy following complete resection of metastases. Cancer Immunol. Immunother. 2015, 64, 381–388. [Google Scholar] [CrossRef]
- Wilgenhof, S.; Van Nuffel, A.M.; Corthals, J.; Heirman, C.; Tuyaerts, S.; Benteyn, D.; De Coninck, A.; Van Riet, I.; Verfaillie, G.; Vandeloo, J.; et al. Therapeutic vaccination with an autologous mRNA electroporated dendritic cell vaccine in patients with advanced melanoma. J. Immunother. 2011, 34, 448–456. [Google Scholar] [CrossRef]
- Wilgenhof, S.; Van Nuffel, A.M.; Benteyn, D.; Corthals, J.; Aerts, C.; Heirman, C.; Van Riet, I.; Bonehill, A.; Thielemans, K.; Neyns, B. A phase IB study on intravenous synthetic mRNA electroporated dendritic cell immunotherapy in pretreated advanced melanoma patients. Ann. Oncol 2013, 24, 2686–2693. [Google Scholar] [CrossRef]
- Wilgenhof, S.; Corthals, J.; Heirman, C.; van Baren, N.; Lucas, S.; Kvistborg, P.; Thielemans, K.; Neyns, B. Phase II Study of Autologous Monocyte-Derived mRNA Electroporated Dendritic Cells (TriMixDC-MEL) Plus Ipilimumab in Patients with Pretreated Advanced Melanoma. J. Clin. Oncol. 2016, 34, 1330–1338. [Google Scholar] [CrossRef] [PubMed]
- Schuurhuis, D.H.; Verdijk, P.; Schreibelt, G.; Aarntzen, E.H.; Scharenborg, N.; de Boer, A.; van de Rakt, M.W.; Kerkhoff, M.; Gerritsen, M.J.; Eijckeler, F.; et al. In situ expression of tumor antigens by messenger RNA-electroporated dendritic cells in lymph nodes of melanoma patients. Cancer Res. 2009, 69, 2927–2934. [Google Scholar] [CrossRef] [PubMed]
- Aarntzen, E.H.; Schreibelt, G.; Bol, K.; Lesterhuis, W.J.; Croockewit, A.J.; de Wilt, J.H.; van Rossum, M.M.; Blokx, W.A.; Jacobs, J.F.; Duiveman-de Boer, T.; et al. Vaccination with mRNA-electroporated dendritic cells induces robust tumor antigen-specific CD4+ and CD8+ T cells responses in stage III and IV melanoma patients. Clin. Cancer Res. 2012, 18, 5460–5470. [Google Scholar] [CrossRef] [PubMed]
- Bol, K.F.; Figdor, C.G.; Aarntzen, E.H.; Welzen, M.E.; van Rossum, M.M.; Blokx, W.A.; van de Rakt, M.W.; Scharenborg, N.M.; de Boer, A.J.; Pots, J.M.; et al. Intranodal vaccination with mRNA-optimized dendritic cells in metastatic melanoma patients. Oncoimmunology 2015, 4, e1019197. [Google Scholar] [CrossRef] [PubMed]
- Bol, K.F.; Aarntzen, E.H.; Pots, J.M.; Olde Nordkamp, M.A.; van de Rakt, M.W.; Scharenborg, N.M.; de Boer, A.J.; van Oorschot, T.G.; Croockewit, S.A.; Blokx, W.A.; et al. Prophylactic vaccines are potent activators of monocyte-derived dendritic cells and drive effective anti-tumor responses in melanoma patients at the cost of toxicity. Cancer Immunol. Immunother. 2016, 65, 327–339. [Google Scholar] [CrossRef] [PubMed]
- Bol, K.F.; van den Bosch, T.; Schreibelt, G.; Mensink, H.W.; Keunen, J.E.; Kilic, E.; Japing, W.J.; Geul, K.W.; Westdorp, H.; Boudewijns, S.; et al. Adjuvant Dendritic Cell Vaccination in High-Risk Uveal Melanoma. Ophthalmology 2016, 123, 2265–2267. [Google Scholar] [CrossRef]
- Dannull, J.; Haley, N.R.; Archer, G.; Nair, S.; Boczkowski, D.; Harper, M.; De Rosa, N.; Pickett, N.; Mosca, P.J.; Burchette, J.; et al. Melanoma immunotherapy using mature DCs expressing the constitutive proteasome. J. Clin. Investig. 2013, 123, 3135–3145. [Google Scholar] [CrossRef]
- Borch, T.H.; Engell-Noerregaard, L.; Zeeberg, I.T.; Ellebaek, E.; Met, O.; Hansen, M.; Andersen, M.H.; thor, S.P.; Svane, I.M. mRNA-transfected dendritic cell vaccine in combination with metronomic cyclophosphamide as treatment for patients with advanced malignant melanoma. Oncoimmunology 2016, 5, e1207842. [Google Scholar] [CrossRef]
- Hobo, W.; Strobbe, L.; Maas, F.; Fredrix, H.; Greupink-Draaisma, A.; Esendam, B.; de Witte, T.; Preijers, F.; Levenga, H.; van Rees, B.; et al. Immunogenicity of dendritic cells pulsed with MAGE3, Survivin and B-cell maturation antigen mRNA for vaccination of multiple myeloma patients. Cancer Immunol. Immunother. 2013, 62, 1381–1392. [Google Scholar] [CrossRef]
- Wang, D.; Zhang, B.; Gao, H.; Ding, G.; Wu, Q.; Zhang, J.; Liao, L.; Chen, H. Clinical research of genetically modified dendritic cells in combination with cytokine-induced killer cell treatment in advanced renal cancer. BMC Cancer 2014, 14, 251. [Google Scholar] [CrossRef] [PubMed]
- Morse, M.A.; Nair, S.K.; Boczkowski, D.; Tyler, D.; Hurwitz, H.I.; Proia, A.; Clay, T.M.; Schlom, J.; Gilboa, E.; Lyerly, H.K. The feasibility and safety of immunotherapy with dendritic cells loaded with CEA mRNA following neoadjuvant chemoradiotherapy and resection of pancreatic cancer. Int. J. Gastrointest. Cancer 2002, 32, 1–6. [Google Scholar] [CrossRef]
- Morse, M.A.; Nair, S.K.; Mosca, P.J.; Hobeika, A.C.; Clay, T.M.; Deng, Y.; Boczkowski, D.; Proia, A.; Neidzwiecki, D.; Clavien, P.A.; et al. Immunotherapy with autologous, human dendritic cells transfected with carcinoembryonic antigen mRNA. Cancer Investig. 2003, 21, 341–349. [Google Scholar] [CrossRef]
- Lesterhuis, W.J.; de Vries, I.J.; Schreibelt, G.; Schuurhuis, D.H.; Aarntzen, E.H.; de Boer, A.; Scharenborg, N.M.; van de Rakt, M.; Hesselink, E.J.; Figdor, C.G.; et al. Immunogenicity of dendritic cells pulsed with CEA peptide or transfected with CEA mRNA for vaccination of colorectal cancer patients. Anticancer Res. 2010, 30, 5091–5097. [Google Scholar] [PubMed]
- Heiser, A.; Coleman, D.; Dannull, J.; Yancey, D.; Maurice, M.A.; Lallas, C.D.; Dahm, P.; Niedzwiecki, D.; Gilboa, E.; Vieweg, J. Autologous dendritic cells transfected with prostate-specific antigen RNA stimulate CTL responses against metastatic prostate tumors. J. Clin. Investig. 2002, 109, 409–417. [Google Scholar] [CrossRef] [PubMed]
- Su, Z.; Dannull, J.; Yang, B.K.; Dahm, P.; Coleman, D.; Yancey, D.; Sichi, S.; Niedzwiecki, D.; Boczkowski, D.; Gilboa, E.; et al. Telomerase mRNA-transfected dendritic cells stimulate antigen-specific CD8+ and CD4+ T cell responses in patients with metastatic prostate cancer. J. Immunol. 2005, 174, 3798–3807. [Google Scholar] [CrossRef]
- Khoury, H.J.; Collins, R.H., Jr.; Blum, W.; Stiff, P.S.; Elias, L.; Lebkowski, J.S.; Reddy, A.; Nishimoto, K.P.; Sen, D.; Wirth, E.D., III; et al. Immune responses and long-term disease recurrence status after telomerase-based dendritic cell immunotherapy in patients with acute myeloid leukemia. Cancer 2017, 123, 3061–3072. [Google Scholar] [CrossRef]
- Kongsted, P.; Borch, T.H.; Ellebaek, E.; Iversen, T.Z.; Andersen, R.; Met, O.; Hansen, M.; Lindberg, H.; Sengelov, L.; Svane, I.M. Dendritic cell vaccination in combination with docetaxel for patients with metastatic castration-resistant prostate cancer: A randomized phase II study. Cytotherapy 2017, 19, 500–513. [Google Scholar] [CrossRef]
- Hernando, J.J.; Park, T.W.; Fischer, H.P.; Zivanovic, O.; Braun, M.; Polcher, M.; Grunn, U.; Leutner, C.; Potzsch, B.; Kuhn, W. Vaccination with dendritic cells transfected with mRNA-encoded folate-receptor-alpha for relapsed metastatic ovarian cancer. Lancet Oncol. 2007, 8, 451–454. [Google Scholar] [CrossRef]
- Van Tendeloo, V.F.; Van de Velde, A.; Van Driessche, A.; Cools, N.; Anguille, S.; Ladell, K.; Gostick, E.; Vermeulen, K.; Pieters, K.; Nijs, G.; et al. Induction of complete and molecular remissions in acute myeloid leukemia by Wilms’ tumor 1 antigen-targeted dendritic cell vaccination. Proc. Natl. Acad. Sci. USA 2010, 107, 13824–13829. [Google Scholar] [CrossRef]
- Coosemans, A.; Vanderstraeten, A.; Tuyaerts, S.; Verschuere, T.; Moerman, P.; Berneman, Z.N.; Vergote, I.; Amant, F.; Van Gool, S.W. Wilms’ Tumor Gene 1 (WT1)—Loaded dendritic cell immunotherapy in patients with uterine tumors: A phase I/II clinical trial. Anticancer Res. 2013, 33, 5495–5500. [Google Scholar] [PubMed]
- Coosemans, A.; Vanderstraeten, A.; Tuyaerts, S.; Verschuere, T.; Moerman, P.; Berneman, Z.; Vergote, I.; Amant, F.; Van Gool, S.W. Immunological response after WT1 mRNA-loaded dendritic cell immunotherapy in ovarian carcinoma and carcinosarcoma. Anticancer Res. 2013, 33, 3855–3859. [Google Scholar] [PubMed]
- Anguille, S.; Van de Velde, A.L.; Smits, E.L.; Van Tendeloo, V.F.; Juliusson, G.; Cools, N.; Nijs, G.; Stein, B.; Lion, E.; Van Driessche, A.; et al. Dendritic cell vaccination as postremission treatment to prevent or delay relapse in acute myeloid leukemia. Blood 2017, 130, 1713–1721. [Google Scholar] [CrossRef] [PubMed]
- Shindo, Y.; Hazama, S.; Maeda, Y.; Matsui, H.; Iida, M.; Suzuki, N.; Yoshimura, K.; Ueno, T.; Yoshino, S.; Sakai, K.; et al. Adoptive immunotherapy with MUC1-mRNA transfected dendritic cells and cytotoxic lymphocytes plus gemcitabine for unresectable pancreatic cancer. J. Transl. Med. 2014, 12, 175. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, D.A.; Batich, K.A.; Gunn, M.D.; Huang, M.N.; Sanchez-Perez, L.; Nair, S.K.; Congdon, K.L.; Reap, E.A.; Archer, G.E.; Desjardins, A.; et al. Tetanus toxoid and CCL3 improve dendritic cell vaccines in mice and glioblastoma patients. Nature 2015, 519, 366–369. [Google Scholar] [CrossRef]
- Maeda, Y.; Yoshimura, K.; Matsui, H.; Shindo, Y.; Tamesa, T.; Tokumitsu, Y.; Hashimoto, N.; Tokuhisa, Y.; Sakamoto, K.; Sakai, K.; et al. Dendritic cells transfected with heat-shock protein 70 messenger RNA for patients with hepatitis C virus-related hepatocellular carcinoma: A phase 1 dose escalation clinical trial. Cancer Immunol. Immunother. 2015, 64, 1047–1056. [Google Scholar] [CrossRef]
- Van Craenenbroeck, A.H.; Smits, E.L.; Anguille, S.; Van de Velde, A.; Stein, B.; Braeckman, T.; Van Camp, K.; Nijs, G.; Ieven, M.; Goossens, H.; et al. Induction of cytomegalovirus-specific T cell responses in healthy volunteers and allogeneic stem cell recipients using vaccination with messenger RNA-transfected dendritic cells. Transplantation 2015, 99, 120–127. [Google Scholar] [CrossRef]
- Batich, K.A.; Reap, E.A.; Archer, G.E.; Sanchez-Perez, L.; Nair, S.K.; Schmittling, R.J.; Norberg, P.; Xie, W.; Herndon, J.E.; Healy, P.; et al. Long-term Survival in Glioblastoma with Cytomegalovirus pp65-Targeted Vaccination. Clin. Cancer Res. 2017, 23, 1898–1909. [Google Scholar] [CrossRef]
- Reap, E.A.; Suryadevara, C.M.; Batich, K.A.; Sanchez-Perez, L.; Archer, G.E.; Schmittling, R.J.; Norberg, P.K.; Herndon, J.E.; Healy, P.; Congdon, K.L.; et al. Dendritic Cells Enhance Polyfunctionality of Adoptively Transferred T Cells That Target Cytomegalovirus in Glioblastoma. Cancer Res. 2018, 78, 256–264. [Google Scholar] [CrossRef]
- Gandhi, R.T.; Kwon, D.S.; Macklin, E.A.; Shopis, J.R.; McLean, A.P.; McBrine, N.; Flynn, T.; Peter, L.; Sbrolla, A.; Kaufmann, D.E.; et al. Immunization of HIV-1-Infected Persons with Autologous Dendritic Cells Transfected WIth mRNA Encoding HIV-1 Gag and Nef: Results of a Randomized, Placebo-Controlled Clinical Trial. J. Acquir. Immune Defic. Syndr. 2016, 71, 246–253. [Google Scholar] [CrossRef]
- Routy, J.P.; Boulassel, M.R.; Yassine-Diab, B.; Nicolette, C.; Healey, D.; Jain, R.; Landry, C.; Yegorov, O.; Tcherepanova, I.; Monesmith, T.; et al. Immunologic activity and safety of autologous HIV RNA-electroporated dendritic cells in HIV-1 infected patients receiving antiretroviral therapy. Clin. Immunol. 2010, 134, 140–147. [Google Scholar] [CrossRef] [PubMed]
- Jacobson, J.M.; Routy, J.P.; Welles, S.; DeBenedette, M.; Tcherepanova, I.; Angel, J.B.; Asmuth, D.M.; Stein, D.K.; Baril, J.G.; McKellar, M.; et al. Dendritic Cell Immunotherapy for HIV-1 Infection Using Autologous HIV-1 RNA: A Randomized, Double-Blind, Placebo-Controlled Clinical Trial. J. Acquir. Immune Defic. Syndr. 2016, 72, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Allard, S.D.; De Keersmaecker, B.; de Goede, A.L.; Verschuren, E.J.; Koetsveld, J.; Reedijk, M.L.; Wylock, C.; De Bel, A.V.; Vandeloo, J.; Pistoor, F.; et al. A phase I/IIa immunotherapy trial of HIV-1-infected patients with Tat, Rev and Nef expressing dendritic cells followed by treatment interruption. Clin. Immunol. 2012, 142, 252–268. [Google Scholar] [CrossRef]
- Van Gulck, E.; Vlieghe, E.; Vekemans, M.; Van Tendeloo, V.F.; Van de Velde, A.; Smits, E.; Angille, S.; Coos, N.; Goossens, H.; Mertens, L.; et al. mRNA-based dendritic cell vaccination induces potent antiviral T-cell responses in HIV-1-infected patients. AIDS 2012, 26, F1–F12. [Google Scholar] [CrossRef] [PubMed]
- Schuler, G. Dendritic cells in cancer immunotherapy. Eur. J. Immunol. 2010, 40, 2123–2130. [Google Scholar] [CrossRef]
- Gilboa, E.; Vieweg, J. Cancer immunotherapy with mRNA-transfected dendritic cells. Immunol. Rev. 2004, 199, 251–263. [Google Scholar] [CrossRef]
- Finn, O.J.; Rammensee, H.G. Is It Possible to Develop Cancer Vaccines to Neoantigens, What Are the Major Challenges, and How Can These Be Overcome? Neoantigens: Nothing New in Spite of the Name. Cold Spring Harb. Perspect. Biol. 2018, 10, a028829. [Google Scholar] [CrossRef]
- Loffler, M.W.; Kowalewski, D.J.; Backert, L.; Bernhardt, J.; Adam, P.; Schuster, H.; Dengler, F.; Backes, D.; Kopp, H.G.; Beckert, S.; et al. Mapping the HLA Ligandome of Colorectal Cancer Reveals an Imprint of Malignant Cell Transformation. Cancer Res. 2018, 78, 4627–4641. [Google Scholar] [CrossRef]
- Newey, A.; Griffiths, B.; Michaux, J.; Pak, H.S.; Stevenson, B.J.; Woolston, A.; Semiannikova, M.; Spain, G.; Barber, L.J.; Matthews, N.; et al. Immunopeptidomics of colorectal cancer organoids reveals a sparse HLA class I neoantigen landscape and no increase in neoantigens with interferon or MEK-inhibitor treatment. J. Immunother. Cancer 2019, 7, 309. [Google Scholar] [CrossRef]
- Sahin, U.; Derhovanessian, E.; Miller, M.; Kloke, B.P.; Simon, P.; Lower, M.; Bukur, V.; Tadmor, A.D.; Luxemburger, U.; Schrors, B.; et al. Personalized RNA mutanome vaccines mobilize poly-specific therapeutic immunity against cancer. Nature 2017, 547, 222–226. [Google Scholar] [CrossRef]
- Bonehill, A.; Heirman, C.; Tuyaerts, S.; Michiels, A.; Breckpot, K.; Brasseur, F.; Zhang, Y.; Van Der Bruggen, P.; Thielemans, K. Messenger RNA-electroporated dendritic cells presenting MAGE-A3 simultaneously in HLA class I and class II molecules. J. Immunol. 2004, 172, 6649–6657. [Google Scholar] [CrossRef] [PubMed]
- Van Lint, S.; Wilgenhof, S.; Heirman, C.; Corthals, J.; Breckpot, K.; Bonehill, A.; Neyns, B.; Thielemans, K. Optimized dendritic cell-based immunotherapy for melanoma: The TriMix-formula. Cancer Immunol. Immunother. 2014, 63, 959–967. [Google Scholar] [CrossRef] [PubMed]
- Calderhead, D.M.; DeBenedette, M.A.; Ketteringham, H.; Gamble, A.H.; Horvatinovich, J.M.; Tcherepanova, I.Y.; Nicolette, C.A.; Healey, D.G. Cytokine maturation followed by CD40L mRNA electroporation results in a clinically relevant dendritic cell product capable of inducing a potent proinflammatory CTL response. J. Immunother. 2008, 31, 731–741. [Google Scholar] [CrossRef] [PubMed]
- Pfeiffer, I.A.; Hoyer, S.; Gerer, K.F.; Voll, R.E.; Knippertz, I.; Guckel, E.; Schuler, G.; Schaft, N.; Dorrie, J. Triggering of NF-kappaB in cytokine-matured human DCs generates superior DCs for T-cell priming in cancer immunotherapy. Eur. J. Immunol. 2014, 44, 3413–3428. [Google Scholar] [CrossRef]
- Bosch, N.C.; Voll, R.E.; Voskens, C.J.; Gross, S.; Seliger, B.; Schuler, G.; Schaft, N.; D+Ârrie, J. NF-kappaB activation triggers NK-cell stimulation by monocyte-derived dendritic cells. Ther. Adv. Med. Oncol. 2019, 11, 1758835919891622. [Google Scholar] [CrossRef] [PubMed]
- Lennerz, V.; Fatho, M.; Gentilini, C.; Frye, R.A.; Lifke, A.; Ferel, D.; Wolfel, C.; Huber, C.; Wolfel, T. The response of autologous T cells to a human melanoma is dominated by mutated neoantigens. Proc. Natl. Acad. Sci. USA 2005, 102, 16013–16018. [Google Scholar] [CrossRef] [PubMed]
- Wood, L.D.; Parsons, D.W.; Jones, S.; Lin, J.; Sjoblom, T.; Leary, R.J.; Shen, D.; Boca, S.M.; Barber, T.; Ptak, J.; et al. The genomic landscapes of human breast and colorectal cancers. Science 2007, 318, 1108–1113. [Google Scholar] [CrossRef]
- Pleasance, E.D.; Cheetham, R.K.; Stephens, P.J.; McBride, D.J.; Humphray, S.J.; Greenman, C.D.; Varela, I.; Lin, M.L.; Ordonez, G.R.; Bignell, G.R.; et al. A comprehensive catalogue of somatic mutations from a human cancer genome. Nature 2010, 463, 191–196. [Google Scholar] [CrossRef]
- Kowalewski, D.J.; Schuster, H.; Backert, L.; Berlin, C.; Kahn, S.; Kanz, L.; Salih, H.R.; Rammensee, H.G.; Stevanovic, S.; Stickel, J.S. HLA ligandome analysis identifies the underlying specificities of spontaneous antileukemia immune responses in chronic lymphocytic leukemia (CLL). Proc. Natl. Acad. Sci. USA 2015, 112, E166–E175. [Google Scholar] [CrossRef]
- Boczkowski, D.; Nair, S.K.; Snyder, D.; Gilboa, E. Dendritic cells pulsed with RNA are potent antigen-presenting cells in vitro and in vivo. J. Exp. Med. 1996, 184, 465–472. [Google Scholar] [CrossRef]
- Ashley, D.M.; Faiola, B.; Nair, S.; Hale, L.P.; Bigner, D.D.; Gilboa, E. Bone marrow-generated dendritic cells pulsed with tumor extracts or tumor RNA induce antitumor immunity against central nervous system tumors. J. Exp. Med. 1997, 186, 1177–1182. [Google Scholar] [CrossRef] [PubMed]
- Kyte, J.A.; Mu, L.; Aamdal, S.; Kvalheim, G.; Dueland, S.; Hauser, M.; Gullestad, H.P.; Ryder, T.; Lislerud, K.; Hammerstad, H.; et al. Phase I/II trial of melanoma therapy with dendritic cells transfected with autologous tumor-mRNA. Cancer Gene Ther. 2006, 13, 905–918. [Google Scholar] [CrossRef] [PubMed]
- Kyte, J.A.; Kvalheim, G.; Lislerud, K.; thor, S.P.; Dueland, S.; Aamdal, S.; Gaudernack, G. T cell responses in melanoma patients after vaccination with tumor-mRNA transfected dendritic cells. Cancer Immunol. Immunother. 2007, 56, 659–675. [Google Scholar] [CrossRef]
- Mocellin, S.; Mandruzzato, S.; Bronte, V.; Marincola, F.M. Cancer vaccines: Pessimism in check. Nat. Med. 2004, 10, 1278–1279. [Google Scholar] [CrossRef] [PubMed]
- Rosenberg, S.A.; Yang, J.C.; Restifo, N.P. Cancer immunotherapy: Moving beyond current vaccines. Nat. Med. 2004, 10, 909–915. [Google Scholar] [CrossRef] [PubMed]
- Timmerman, J.M.; Levy, R. Cancer vaccines: Pessimism in check. Nat. Med. 2004, 10, 1279–1280. [Google Scholar] [CrossRef] [PubMed]
- Timmerman, J.M.; Czerwinski, D.K.; Davis, T.A.; Hsu, F.J.; Benike, C.; Hao, Z.M.; Taidi, B.; Rajapaksa, R.; Caspar, C.B.; Okada, C.Y.; et al. Idiotype-pulsed dendritic cell vaccination for B-cell lymphoma: Clinical and immune responses in 35 patients. Blood 2002, 99, 1517–1526. [Google Scholar] [CrossRef]
- O’Rourke, M.G.; Johnson, M.; Lanagan, C.; See, J.; Yang, J.; Bell, J.R.; Slater, G.J.; Kerr, B.M.; Crowe, B.; Purdie, D.M.; et al. Durable complete clinical responses in a phase I/II trial using an autologous melanoma cell/dendritic cell vaccine. Cancer Immunol. Immunother. 2003, 52, 387–395. [Google Scholar] [CrossRef]
- O’Rourke, M.G.; Johnson, M.K.; Lanagan, C.M.; See, J.L.; O’Connor, L.E.; Slater, G.J.; Thomas, D.; Lopez, J.A.; Martinez, N.R.; Ellem, K.A.; et al. Dendritic cell immunotherapy for stage IV melanoma. Melanoma Res. 2007, 17, 316–322. [Google Scholar] [CrossRef]
- Ridgway, D. The first 1000 dendritic cell vaccinees. Cancer Investig. 2003, 21, 873–886. [Google Scholar] [CrossRef]
- Engell-Noerregaard, L.; Hansen, T.H.; Andersen, M.H.; thor, S.P.; Svane, I.M. Review of clinical studies on dendritic cell-based vaccination of patients with malignant melanoma: Assessment of correlation between clinical response and vaccine parameters. Cancer Immunol. Immunother. 2009, 58, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Kantoff, P.W.; Higano, C.S.; Shore, N.D.; Berger, E.R.; Small, E.J.; Penson, D.F.; Redfern, C.H.; Ferrari, A.C.; Dreicer, R.; Sims, R.B.; et al. Sipuleucel-T immunotherapy for castration-resistant prostate cancer. N. Engl. J. Med. 2010, 363, 411–422. [Google Scholar] [CrossRef] [PubMed]
- Wolchok, J.D.; Hoos, A.; O’Day, S.; Weber, J.S.; Hamid, O.; Lebbe, C.; Maio, M.; Binder, M.; Bohnsack, O.; Nichol, G.; et al. Guidelines for the evaluation of immune therapy activity in solid tumors: Immune-related response criteria. Clin. Cancer Res. 2009, 15, 7412–7420. [Google Scholar] [CrossRef] [PubMed]
- Hodi, F.S.; O’Day, S.J.; McDermott, D.F.; Weber, R.W.; Sosman, J.A.; Haanen, J.B.; Gonzalez, R.; Robert, C.; Schadendorf, D.; Hassel, J.C.; et al. Improved survival with ipilimumab in patients with metastatic melanoma. N. Engl. J. Med. 2010, 363, 711–723. [Google Scholar] [CrossRef] [PubMed]
- Hellmann, M.D.; Snyder, A. Making It Personal: Neoantigen Vaccines in Metastatic Melanoma. Immunity 2017, 47, 221–223. [Google Scholar] [CrossRef] [PubMed]
- Louahed, J.; Gruselle, O. Expression of defined genes identified by pretreatment tumor profiling: Association with clinical responses to the GSK MAGE-A3 immunotherapeutic in metastatic melanoma patients (EORTC 16032–18031). J. Clin. Oncol. 2008, 26, 15. [Google Scholar] [CrossRef]
- Gajewski, T.F.; Zha, Y. Association of gene expression profile in metastatic melanoma and survival to a dendritic cell-based vaccine. ASCO Meeting Abstract. J. Clin. Oncol. 2009, 27, 15. [Google Scholar]
- Harlin, H.; Meng, Y.; Peterson, A.C.; Zha, Y.; Tretiakova, M.; Slingluff, C.; McKee, M.; Gajewski, T.F. Chemokine expression in melanoma metastases associated with CD8+ T-cell recruitment. Cancer Res. 2009, 69, 3077–3085. [Google Scholar] [CrossRef]
- Fong, L.; Brockstedt, D.; Benike, C.; Breen, J.K.; Strang, G.; Ruegg, C.L.; Engleman, E.G. Dendritic cell-based xenoantigen vaccination for prostate cancer immunotherapy. J. Immunol. 2001, 167, 7150–7156. [Google Scholar] [CrossRef]
- Geiger, J.D.; Hutchinson, R.J.; Hohenkirk, L.F.; McKenna, E.A.; Yanik, G.A.; Levine, J.E.; Chang, A.E.; Braun, T.M.; Mule, J.J. Vaccination of pediatric solid tumor patients with tumor lysate-pulsed dendritic cells can expand specific T cells and mediate tumor regression. Cancer Res. 2001, 61, 8513–8519. [Google Scholar]
- Toungouz, M.; Libin, M.; Bulte, F.; Faid, L.; Lehmann, F.; Duriau, D.; Laporte, M.; Gangji, D.; Bruyns, C.; Lambermont, M.; et al. Transient expansion of peptide-specific lymphocytes producing IFN-gamma after vaccination with dendritic cells pulsed with MAGE peptides in patients with mage-A1/A3-positive tumors. J. Leukoc. Biol. 2001, 69, 937–943. [Google Scholar] [PubMed]
- Chang, A.E.; Redman, B.G.; Whitfield, J.R.; Nickoloff, B.J.; Braun, T.M.; Lee, P.P.; Geiger, J.D.; Mule, J.J. A phase I trial of tumor lysate-pulsed dendritic cells in the treatment of advanced cancer. Clin. Cancer Res. 2002, 8, 1021–1032. [Google Scholar]
- Vilella, R.; Benitez, D.; Mila, J.; Lozano, M.; Vilana, R.; Pomes, J.; Tomas, X.; Costa, J.; Vilalta, A.; Malvehy, J.; et al. Pilot study of treatment of biochemotherapy-refractory stage IV melanoma patients with autologous dendritic cells pulsed with a heterologous melanoma cell line lysate. Cancer Immunol. Immunother. 2004, 53, 651–658. [Google Scholar] [CrossRef] [PubMed]
- Chang, D.H.; Osman, K.; Connolly, J.; Kukreja, A.; Krasovsky, J.; Pack, M.; Hutchinson, A.; Geller, M.; Liu, N.; Annable, R.; et al. Sustained expansion of NKT cells and antigen-specific T cells after injection of alpha-galactosyl-ceramide loaded mature dendritic cells in cancer patients. J. Exp. Med. 2005, 201, 1503–1517. [Google Scholar] [CrossRef] [PubMed]
- Mazzolini, G.; Alfaro, C.; Sangro, B.; Feijoo, E.; Ruiz, J.; Benito, A.; Tirapu, I.; Arina, A.; Sola, J.; Herraiz, M.; et al. Intratumoral injection of dendritic cells engineered to secrete interleukin-12 by recombinant adenovirus in patients with metastatic gastrointestinal carcinomas. J. Clin. Oncol. 2005, 23, 999–1010. [Google Scholar] [CrossRef] [PubMed]
- Banchereau, J.; Ueno, H.; Dhodapkar, M.; Connolly, J.; Finholt, J.P.; Klechevsky, E.; Blanck, J.P.; Johnston, D.A.; Palucka, A.K.; Fay, J. Immune and clinical outcomes in patients with stage IV melanoma vaccinated with peptide-pulsed dendritic cells derived from CD34+ progenitors and activated with type I interferon. J. Immunother. 2005, 28, 505–516. [Google Scholar] [CrossRef] [PubMed]
- Loveland, B.E.; Zhao, A.; White, S.; Gan, H.; Hamilton, K.; Xing, P.X.; Pietersz, G.A.; Apostolopoulos, V.; Vaughan, H.; Karanikas, V.; et al. Mannan-MUC1-pulsed dendritic cell immunotherapy: A phase I trial in patients with adenocarcinoma. Clin. Cancer Res. 2006, 12, 869–877. [Google Scholar] [CrossRef]
- Stift, A.; Sachet, M.; Yagubian, R.; Bittermann, C.; Dubsky, P.; Brostjan, C.; Pfragner, R.; Niederle, B.; Jakesz, R.; Gnant, M.; et al. Dendritic cell vaccination in medullary thyroid carcinoma. Clin. Cancer Res. 2004, 10, 2944–2953. [Google Scholar] [CrossRef]
- Holtl, L.; Ramoner, R.; Zelle-Rieser, C.; Gander, H.; Putz, T.; Papesh, C.; Nussbaumer, W.; Falkensammer, C.; Bartsch, G.; Thurnher, M. Allogeneic dendritic cell vaccination against metastatic renal cell carcinoma with or without cyclophosphamide. Cancer Immunol. Immunother. 2005, 54, 663–670. [Google Scholar] [CrossRef]
- Hodge, J.W.; Ardiani, A.; Farsaci, B.; Kwilas, A.R.; Gameiro, S.R. The tipping point for combination therapy: Cancer vaccines with radiation, chemotherapy, or targeted small molecule inhibitors. Semin. Oncol. 2012, 39, 323–339. [Google Scholar] [CrossRef]
- Zhao, Y.; Qiao, G.; Wang, X.; Song, Y.; Zhou, X.; Jiang, N.; Zhou, L.; Huang, H.; Zhao, J.; Morse, M.A.; et al. Combination of DC/CIK adoptive T cell immunotherapy with chemotherapy in advanced non-small-cell lung cancer (NSCLC) patients: A prospective patients’ preference-based study (PPPS). Clin. Transl. Oncol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Yanagisawa, R.; Koizumi, T.; Koya, T.; Sano, K.; Koido, S.; Nagai, K.; Kobayashi, M.; Okamoto, M.; Sugiyama, H.; Shimodaira, S. WT1-pulsed Dendritic Cell Vaccine Combined with Chemotherapy for Resected Pancreatic Cancer in a Phase I Study. Anticancer Res. 2018, 38, 2217–2225. [Google Scholar] [CrossRef] [PubMed]
- Le, D.T.; Wang-Gillam, A.; Picozzi, V.; Greten, T.F.; Crocenzi, T.; Springett, G.; Morse, M.; Zeh, H.; Cohen, D.; Fine, R.L.; et al. Safety and survival with GVAX pancreas prime and Listeria Monocytogenes-expressing mesothelin (CRS-207) boost vaccines for metastatic pancreatic cancer. J. Clin. Oncol. 2015, 33, 1325–1333. [Google Scholar] [CrossRef] [PubMed]
- Massarelli, E.; William, W.; Johnson, F.; Kies, M.; Ferrarotto, R.; Guo, M.; Feng, L.; Lee, J.J.; Tran, H.; Kim, Y.U.; et al. Combining Immune Checkpoint Blockade and Tumor-Specific Vaccine for Patients With Incurable Human Papillomavirus 16-Related Cancer: A Phase 2 Clinical Trial. JAMA Oncol. 2019, 5, 67–73. [Google Scholar] [CrossRef] [PubMed]
- Ott, P.A.; Hu, Z.; Keskin, D.B.; Shukla, S.A.; Sun, J.; Bozym, D.J.; Zhang, W.; Luoma, A.; Giobbie-Hurder, A.; Peter, L.; et al. An immunogenic personal neoantigen vaccine for patients with melanoma. Nature 2017, 547, 217–221. [Google Scholar] [CrossRef]
- Weber, J.S.; Kudchadkar, R.R.; Yu, B.; Gallenstein, D.; Horak, C.E.; Inzunza, H.D.; Zhao, X.; Martinez, A.J.; Wang, W.; Gibney, G.; et al. Safety, efficacy, and biomarkers of nivolumab with vaccine in ipilimumab-refractory or -naive melanoma. J. Clin. Oncol. 2013, 31, 4311–4318. [Google Scholar] [CrossRef]
- Wijn, D.H.; Groeneveld, G.H.; Vollaard, A.M.; Muller, M.; Wallinga, J.; Gelderblom, H.; Smit, E.F. Influenza vaccination in patients with lung cancer receiving anti-programmed death receptor 1 immunotherapy does not induce immune-related adverse events. Eur. J. Cancer 2018, 104, 182–187. [Google Scholar] [CrossRef]
- Sprooten, J.; Ceusters, J.; Coosemans, A.; Agostinis, P.; De Vleeschouwer, S.; Zitvogel, L.; Kroemer, G.; Galluzzi, L.; Garg, A.D. Trial watch: Dendritic cell vaccination for cancer immunotherapy. Oncoimmunology 2019, 8, e1638212. [Google Scholar] [CrossRef]
- Garg, A.D.; More, S.; Rufo, N.; Mece, O.; Sassano, M.L.; Agostinis, P.; Zitvogel, L.; Kroemer, G.; Galluzzi, L. Trial watch: Immunogenic cell death induction by anticancer chemotherapeutics. Oncoimmunology 2017, 6, e1386829. [Google Scholar] [CrossRef]
- van Willigen, W.W.; Bloemendal, M.; Gerritsen, W.R.; Schreibelt, G.; De Vries, I.J.M.; Bol, K.F. Dendritic Cell Cancer Therapy: Vaccinating the Right Patient at the Right Time. Front. Immunol. 2018, 9, 2265. [Google Scholar] [CrossRef]
- Belderbos, R.A.; Aerts, J.G.J.V.; Vroman, H. Enhancing Dendritic Cell Therapy in Solid Tumors with Immunomodulating Conventional Treatment. Mol. Ther. Oncolyt. 2019, 13, 67–81. [Google Scholar] [CrossRef] [PubMed]
- Welters, M.J.; van der Sluis, T.C.; van Meir, H.; Loof, N.M.; van Ham, V.J.; van Duikeren, S.; Santegoets, S.J.; Arens, R.; de Kam, M.L.; Cohen, A.F.; et al. Vaccination during myeloid cell depletion by cancer chemotherapy fosters robust T cell responses. Sci. Transl. Med. 2016, 8, 334ra52. [Google Scholar] [CrossRef] [PubMed]
- Antonia, S.J.; Mirza, N.; Fricke, I.; Chiappori, A.; Thompson, P.; Williams, N.; Bepler, G.; Simon, G.; Janssen, W.; Lee, J.H.; et al. Combination of p53 cancer vaccine with chemotherapy in patients with extensive stage small cell lung cancer. Clin. Cancer Res. 2006, 12, 878–887. [Google Scholar] [CrossRef] [PubMed]
- Tanyi, J.L.; Bobisse, S.; Ophir, E.; Tuyaerts, S.; Roberti, A.; Genolet, R.; Baumgartner, P.; Stevenson, B.J.; Iseli, C.; Dangaj, D.; et al. Personalized cancer vaccine effectively mobilizes antitumor T cell immunity in ovarian cancer. Sci. Transl. Med. 2018, 10. [Google Scholar] [CrossRef]
- Ogasawara, M.; Miyashita, M.; Ota, S. Vaccination of Urological Cancer Patients With WT1 Peptide-Pulsed Dendritic Cells in Combination With Molecular Targeted Therapy or Conventional Chemotherapy Induces Immunological and Clinical Responses. Ther. Apher. Dial. 2018, 22, 266–277. [Google Scholar] [CrossRef]
- Hirooka, Y.; Itoh, A.; Kawashima, H.; Hara, K.; Nonogaki, K.; Kasugai, T.; Ohno, E.; Ishikawa, T.; Matsubara, H.; Ishigami, M.; et al. A combination therapy of gemcitabine with immunotherapy for patients with inoperable locally advanced pancreatic cancer. Pancreas 2009, 38, e69–e74. [Google Scholar] [CrossRef]
- Ito, Z.; Kan, S.; Bito, T.; Horiuchi, S.; Akasu, T.; Yoshida, S.; Kajihara, M.; Hokari, A.; Saruta, M.; Yoshida, N.; et al. Predicted Markers of Overall Survival in Pancreatic Cancer Patients Receiving Dendritic Cell Vaccinations Targeting WT1. Oncology 2019, 97, 135–148. [Google Scholar] [CrossRef]
- Bassani-Sternberg, M.; Digklia, A.; Huber, F.; Wagner, D.; Sempoux, C.; Stevenson, B.J.; Thierry, A.C.; Michaux, J.; Pak, H.; Racle, J.; et al. A Phase Ib Study of the Combination of Personalized Autologous Dendritic Cell Vaccine, Aspirin, and Standard of Care Adjuvant Chemotherapy Followed by Nivolumab for Resected Pancreatic Adenocarcinoma—A Proof of Antigen Discovery Feasibility in Three Patients. Front. Immunol. 2019, 10, 1832. [Google Scholar] [CrossRef]
- Koido, S.; Homma, S.; Okamoto, M.; Takakura, K.; Mori, M.; Yoshizaki, S.; Tsukinaga, S.; Odahara, S.; Koyama, S.; Imazu, H.; et al. Treatment with chemotherapy and dendritic cells pulsed with multiple Wilms’ tumor 1 (WT1)-specific MHC class I/II-restricted epitopes for pancreatic cancer. Clin. Cancer Res. 2014, 20, 4228–4239. [Google Scholar] [CrossRef]
- Hardwick, N.R.; Frankel, P.; Ruel, C.; Kilpatrick, J.; Tsai, W.; Kos, F.; Kaltcheva, T.; Leong, L.; Morgan, R.; Chung, V.; et al. p53-Reactive T Cells Are Associated with Clinical Benefit in Patients with Platinum-Resistant Epithelial Ovarian Cancer After Treatment with a p53 Vaccine and Gemcitabine Chemotherapy. Clin. Cancer Res. 2018, 24, 1315–1325. [Google Scholar] [CrossRef]
- Gujar, S.A.; Clements, D.; Lee, P.W. Two is better than one: Complementing oncolytic virotherapy with gemcitabine to potentiate antitumor immune responses. Oncoimmunology 2014, 3, e27622. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Tallon de, L.P.; Cecconi, V.; Hiltbrunner, S.; Yagita, H.; Friess, M.; Bode, B.; Opitz, I.; Vrugt, B.; Weder, W.; Stolzmann, P.; et al. Gemcitabine Synergizes with Immune Checkpoint Inhibitors and Overcomes Resistance in a Preclinical Model and Mesothelioma Patients. Clin. Cancer Res. 2018, 24, 6345–6354. [Google Scholar] [CrossRef] [PubMed]
- Galsky, M.D.; Wang, H.; Hahn, N.M.; Twardowski, P.; Pal, S.K.; Albany, C.; Fleming, M.T.; Starodub, A.; Hauke, R.J.; Yu, M.; et al. Phase 2 Trial of Gemcitabine, Cisplatin, plus Ipilimumab in Patients with Metastatic Urothelial Cancer and Impact of DNA Damage Response Gene Mutations on Outcomes. Eur. Urol. 2018, 73, 751–759. [Google Scholar] [CrossRef] [PubMed]
- Coventry, B.J.; Lilly, C.A.; Hersey, P.; Michele, A.; Bright, R.J. Prolonged repeated vaccine immuno-chemotherapy induces long-term clinical responses and survival for advanced metastatic melanoma. J. Immunother. Cancer 2014, 2, 9. [Google Scholar] [CrossRef][Green Version]
- Terheyden, P.; Becker, J.C.; Kampgen, E.; Brocker, E.B. Sequential interferon-alpha2b, interleukin-2 and fotemustine for patients with metastatic melanoma. Melanoma Res. 2000, 10, 475–482. [Google Scholar] [CrossRef]
- Maccalli, C.; Giannarelli, D.; Capocefalo, F.; Pilla, L.; Fonsatti, E.; Di Giacomo, A.M.; Parmiani, G.; Maio, M. Immunological markers and clinical outcome of advanced melanoma patients receiving ipilimumab plus fotemustine in the NIBIT-M1 study. Oncoimmunology 2016, 5, e1071007. [Google Scholar] [CrossRef]

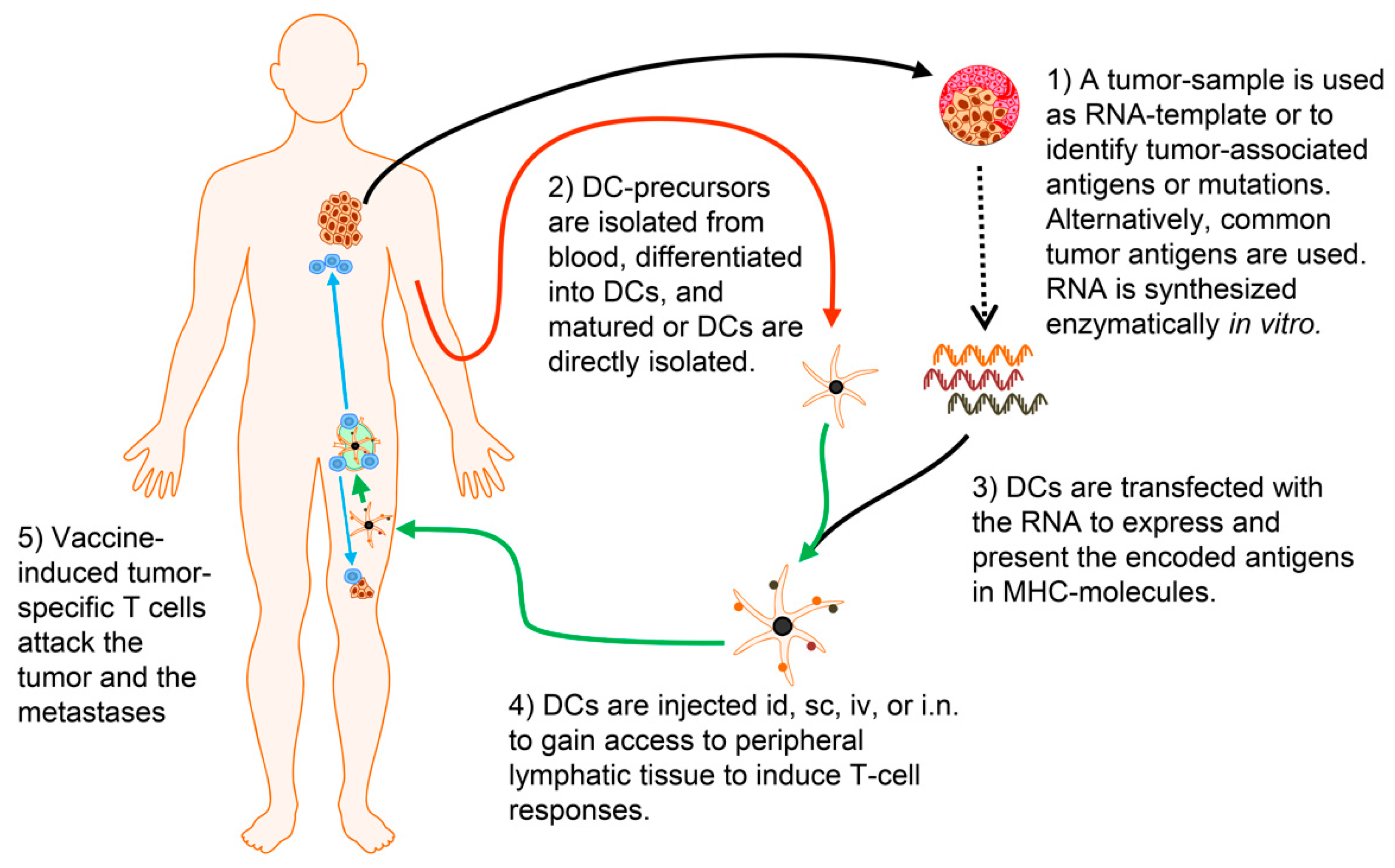
| RNA Used | Malignancy/Disease | Patients Vaccinated | Phase | DC Culturing | Maturation | Reference |
|---|---|---|---|---|---|---|
| Autologous tumor RNA (aT-RNA) | Colorectal ca m | 15 | I | Standard (including FCS) | No | [29] |
| Colon ca | 1 | 0 | Standard | TNFα | [30] | |
| Renal cell ca m | 10 | I | Standard | No | [31] | |
| Pediatric brain tumors | 7 | I | Standard | No | [32] | |
| Pediatric neuroblastoma stage IV | 8 | I | Standard | No | [33] | |
| Renal cell ca m, ovarian ca | 11 | ns | Standard | MCMM | [34] | |
| Melanoma stage IV | 6 | I | Standard (Clinimacs) | TNFα + PGE2 | [35] | |
| Renal cell ca m | 21 | II | Standard | TNFα + PGE2 + IFNγ + CD40L-mRNA | [36] | |
| Melanoma stage IV | 31 | I/II | Standard | MCMM | [37] | |
| aTSC-RNA (tumor stem cells) | Glioblastoma | 7 | I/II | Standard (5 days) | MCMM | [38] |
| Allogeneic tumor RNA (3 human cancer cell lines) | Prostate ca | 19 | I/II | Standard | MCMM | [39] |
| MAGE-A1- MAGE-A3-, MAGE-C2-, MelanA-, tyrosinase-, and/or gp100-DC-Lamp mRNA | Melanoma stage III and IV | 30 | I * | Standard (6 days) | TriMix or polyIC + CD40L-mRNA | [40] |
| MAGE-A3-, MAGE-C2-, tyrosinase-, gp100-DC-Lamp mRNA | Melanoma stage IIIC and IV | 35 | I * | Standard (6 days) | TriMix- mRNA | [41] |
| Melanoma stage IIIC and IV | 15 | IB | Standard (6 days) | TriMix-mRNA | [42] | |
| Melanoma stage IIIC and IV | 39 | II | Standard (6 days) | TriMix-mRNA | [43] | |
| gp100 or tyrosinase mRNA | Melanoma m stage III | 11 | I/II | Standard | MCM + TNFα + PGE2 | [44] |
| gp100 and tyrosinase mRNA | Melanoma stage III and IV | 45 | I/II | Standard (5–7 days) | MCM + PGE2 + TNFα | [45] |
| Melanoma stage III and IV | 15 | I/II | Standard | TriMix-mRNA | [46] | |
| Melanoma stage III and IV | 28 | I/II | Standard | TLR-agonists from conventional vaccines | [47] | |
| Uveal melanoma | 23 | I/II | Standard | ns | [48] | |
| MelanA, MAGE-A3, gp100, and tyrosinase mRNA | Melanoma m | 12 | I | Standard (5 days) | MCMM | [49] |
| hTERT, survivin, and p53 mRNA | Advanced melanoma | 22 | I | Standard | ns | [50] |
| MAGE-A3, survivin, BCMA mRNA | Multiple myeloma stage II/III | 12 | I | Standard | MCMM | [51] |
| MUC1 and survivin mRNA | Renal cell carcinoma | 28 | I/II | Standard (4 days) | TNFα | [52] |
| CEA mRNA | Pancreatic cancer | 3 | ns | Standard | No | [53] |
| CEA expressing malignancies | 37 | I/II | Standard | No | [54] | |
| Colorectal cancer m | 5 | I/II | Standard | MCMM | [55] | |
| PSA mRNA | Prostate cancer m | 13 | I | Standard | No | [56] |
| hTERT mRNA, +/− LAMP | Prostate cancer m | 20 | I | Standard | MCMM | [57] |
| AML | 21 | II | ns | ns | [58] | |
| PSA, PAP, survivin, and hTERT mRNA | Castration-resistant prostate ca | 21 | II | Standard | ns | [59] |
| Folat receptor mRNA | Ovarian cancer m | 1 | 0 | Standard | MCMM | [60] |
| WT1 mRNA | AML | 10 | I/II | Standard (6 days) | TNFα+ PGE2 | [61] |
| Uterine cancer | 6 | I/II | Standard (6 days) | TNFα + IL1ß | [62] | |
| Ovarian | 2 | I/II | Standard (6 days) | TNFα + IL1ß | [63] | |
| WT1 mRNA +/− DC-Lamp | AML | 20 | II | Standard (6 days) | TNFα+ PGE2 | [64] |
| MUC1 mRNA | Pancreatic cancer | 42 | ns | Standard (6 days) | TNFα | [65] |
| CMV pp65.LAMP mRNA | Glioblastoma | 12 | I | Standard | MCMM | [66] |
| HSP70 | HCV-related hepatocarcinoma | 12 | I | Standard | TNFα | [67] |
| CMV pp65 mRNA | Hematopoietic stem cell transplantation | 7 | ns | Standard (6 days clinimacs) | TNFα, PGE2 | [68] |
| Glioblastoma | 11 | I | Standard from CD34+ | ns | [69] | |
| Glioblastoma | 9 | I | Standard | MCMM | [70] | |
| Gag and Nef mRNA | HIV infection | 10 | I/II | Standard (5 days) | MCMM | [71] |
| Gag, Vpr, Rev and Nef mRNA | HIV infection | 10 | I/II | Standard | TNFα + IFNγ+ PGE2 + CD40L mRNA | [72] |
| HIV infection | 35 | IIB | Standard | TNFa + IFNγ + PGE2 + CD40L-mRNA | [73] | |
| Gag-, Tat-, Rev-, and Nef-DC-Lamp mRNA | HIV infection | 17 | I/IIa | Standard (6 days) | MCMM | [74] |
| Gag- and Tat-Nef-Rev-DC-Lamp mRNA | HIV infection | 6 | I/II | Standard (clinimacs) | TNFα + PGE2 | [75] |
| Study Reference | # Pts | Disease and Combination Treatment | DC Culturing | Maturation | RNA-Transfection | Clinical Response |
|---|---|---|---|---|---|---|
| [35] | 6 | Melanoma, stage IV | Standard (Clinimacs) | TNFα + PGE2 | EP with aT-RNA | No objective clinical response |
| [44] | 11 | Melanoma stage III | Standard | MCM + TNFα + PGE2 | EP with gp100 or tyrosinase mRNA | No information regarding clinical responses, vaccine-related CTL-responses in 7 pts |
| [41] | 35 | Melanoma, m | Standard (6 days) | TriMix- mRNA | EP with MAGE-A3, -C2, tyrosinase, gp100 mRNA | Of 20 pts with measurable disease 11 pts SD, 9 pts PD. Of 15 pts without measurable disease 9 showed relapse. |
| [45] | 45 | Melanoma stage III and IV, m | Standard (5–7 days) | MCM + PGE2 + TNFα | EP with gp100 and tyrosinase mRNA | Stage III: median PFS 34.3 months; mOS not reached. Stage IV (19 pts): 6 SD, 1 PR, 12 PD; mOS 24.1 months (patients with positive immunomonitoring) |
| [49] | 12 | Melanoma, m | Standard (5 days) | MCMM | EP with gp100, MelanA, tyrosinase, and MAGE-A3 mRNA +/− IP siRNA | 1 pt PR 1 pt CR mOS 35 months |
| [42] | 15 | Melanoma | Standard (6 days) | TriMix-mRNA | EP with gp100-, tyrosinase-, MAGE-A3-, and -C2-DC-Lamp mRNA | 2 pts with CR 2 pts with PR 4 pts with SD |
| [46] | 15 | Melanoma | Standard | TriMix-mRNA | EP with gp100 and tyrosinase mRNA | Mpfs = 15.14 months mOS = 23.36 months 1 pt = not evaluable 7 pts with PD 2 pts with SD 1 pt with MR 3 pts with no evidence of disease |
| [40] | 30 | Melanoma (adjuvant) | Standard (6 days) | TriMix or polyIC + CD40L-mRNA | EP with MAGE-A1-, -A3-, -C2-, tyrosinase-, melanA-, and gp100-DC-Lamp RNA | mRFS = 22 months St IIIB/C = 18 months, OS = not reached St III = 36 months; OS = 6.2 years St IIB IIC II 24–27 months; OS = 5.3 years mOS = not reached |
| [47] | 28 | Melanoma stage III and IV | Standard | TLR-agonists from conventional vaccines | EP with gp100 and tyrosinase mRNA | 4 pts with SD |
| [37] | 31 | Advanced melanoma | Standard | MCMM | EP with aT-RNA | 1 pt with PR 3 pts with SD OS 10 months |
| [50] | 22 | Malignant melanoma Cyclophosphamide | Standard | ns | EP with hTERT, survivin, p53 mRNA | 9 pts with SD mPFS 3.1 months mOS 10.4 months |
| [43] | 39 | Pretreated advanced melanoma Ipilimumab | Standard (6 days) | TriMix-mRNA | EP with MAGE-A3-, -C2-, tyrosinase-, and gp100-DC-LAMP mRNA | 8 pts with CR 7 pts with PR 6 pts with SD mPFS 27 weeks mOS 59 weeks |
| [48] | 23 | Uveal melanoma | Standard | ns | EP with gp100 and tyrosinase mRNA | mDFS 34.5 months mOS 51.8 months |
| [60] | 1 | Advanced serous papillary ovarian cancer stage IIIc | Standard | MCMM | EP with folatR mRNA | 1 pt PR |
| [62] | 2 | Ovarian cancer | Standard (6 days) | TNFα + IL1ß | EP with WT1 mRNA | Patients with ovarian carcinosarcoma showed OS of 70 months (vs 15.5 months in historical controls). |
| [63] | 6 | Uterine cancer | Standard (6 days) | TNFα + IL1ß | EP with WT1 mRNA | OS of 10 to 11 months compared to 2–5 months historical controls |
| [31] | 10 | Renal cell carcinoma, stage III or IV | Standard | No | co-incubation with aT-RNA | 7 pts SD/slow progression |
| [34] | 11 | Renal cell cancer, m (10 pts), ovarial carcinoma (1pt) Ontak® | Standard | MCMM | EP with aT-RNA | Increase in tumor-specific CTL, no information on clinical responses |
| [52] | 28 | Renal cell cancer cytokine-induced killer cells | Standard (4 days) | TNFα | EP with MUC-1 and survivin mRNA | 4 pts with CR: 2 > 10 months; 2 > 15 months 7 pts with PR (6–21 months) 10 pts with SD (5–21 months) 6 pts with PD/1 death |
| [36] | 21 | Renal cell cancer sunitinib | Standard | TNFα + PGE2 + IFNγ + CD40L-mRNA | EP with aT-RNA | 5 pts with PR 8 pts with SD 13 pts with PR + SD 8 pts with PD Median OS:30.2 months |
| [56] | 13 | Prostate cancer, m | Standard | No | co-incubation with PSA mRNA | 1 pt decrease of PSA level, 5 pts reduction PSA log slope, 3 pts transient elimination of tumor cells in peripheral blood |
| [39] | 19 | Prostate cancer, androgen resistant | Standard | MCMM | EP with allogeneic tumor RNA (3 human cancer cell lines) | 11 pts SD (PSA) 13 pts decreased log slope PSA |
| [57] | 20 | Prostate cancer, m | Standard | MCMM | EP with hTERT mRNA +/− LAMP | No objective clinical response increase in hTERT-specific CTL and molecular clearence of circulating micrometastases |
| [59] | 21 | Castration-resistant prostate cancer docetaxel | Standard | ns | EP with PSA, PAP, survivin, hTERT mRNA | mPFS 5.5 months |
| [32] | 7 | Pediatric brain tumors | Standard | No | co-incubation with aT-RNA | 0 pt CR, 1 pt PR, 2 pts SD |
| [33] | 8 | Pediatric neuroblastoma stage IV | Standard | No | co-incubation with aT-RNA | No objective clinical response |
| [38] | 7 | Glioblastoma | Standard (5 days) | MCMM | EP with aT-RNA | Median PFS of 694 days vs. 236 days in historical controls Median OS of 759 days vs. 585 days in historical controls |
| [66] | 12 | Glioblastoma injection site preconditioned with tetanus toxoid | Standard | MCMM | EP with CMV pp65 mRNA | mPFS of 10.8 months; mOS 18.5 months |
| [69] | 11 | Glioblastoma temozolimide DCs mixed with GM-CSF | Standard from CD34+ | ns | EP with CMV pp65 mRNA | mPFS 25.3 months mOS 41.1 months |
| [70] | 9 | Glioblastoma adoptive T-cell transfer | Standard | MCMM | EP with CMV pp65 mRNA | increase in polyfunctinal pp65-specific T cells |
| [53] | 3 | Pancreatic adenocarcinoma, CEA expressing | Standard | No | co-incubation with CEA mRNA | 3 pts SD |
| [65] | 42 | Pancreatic cancer cytotoxic lymphocytes gemcitabine | Standard (6 days) | TNFα | EP with MUC-1 mRNA | 1 pt with CR, 3 pts with PR, 22 pts with SD16 pts with PD mOS 13.9 months 1-year survival rate 51.1% |
| [54] | 37 | CEA expressing cancer, m (24 tumor bearing, 13 tumor free) | Standard | No | co-incubation with CEA mRNA | 1 pt CR, 2 pts PR, 2 pts SD |
| [29] | 15 | Colorectal cancer, m | Standard (including FCS) | No | co-incubation with aT-RNA | No objective clinical response |
| [55] | 5 | Colorectal cancer, m | Standard | MCMM | EP with CEA mRNA | Median progression free survival of 26 months |
| [30] | 1 | Adenocarcinoma, m | Standard | TNFα | lipofection of aT-RNA | No objective clinical response |
| [51] | 12 | Multiple myeloma | Standard | MCMM | EP with BCMA, MAGE3, and survivin mRNA | After 25 months 10 of 12 pts still alive with 5 pts having SD, 5 pts having PD |
| [58] | 21 | AML | ns | ns | EP with hTERT mRNA, +/− LAMP | “vaccination with hTERT-DCs may be associated with favorable recurrence-free survival” |
| [64] | 30 | AML | Standard (6 days) | TNFα+ PGE2 | EP with WT1 mRNA +/− DC-lamp | 9 pts with molecular remission 4 pts with SD relapse reduction rate of 25% |
| [68] | 7 | 4 healthy volunteers, 3 HSCT recipients | Standard (6 days clinimacs) | TNFα, PGE2 | EP with CMV pp65 mRNA | No survival data (vaccination to induce CMV cellular response) |
| [67] | 12 | HCV-related hepato-carcinoma | Standard | TNFα | EP with HSP70 mRNA | 2 pts with CR (min. 33 and 44 months) |
| [72] | 10 | HIV infection | Standard | TNFα + IFNγ+ PGE2 + CD40L mRNA | EP with Gag, Vpr, Rev, and Nef mRNA | 7 pts HIV-specific proliferative immune response |
| [74] | 17 | HIV infection | Standard (6 days) | MCMM | EP with Tat-, Rev-, or Nef-DC-Lamp mRNA | Vaccine-specific immune response |
| [75] | 6 | HIV infection | Standard (clinimacs) | TNFα + PGE2 | EP with Gag-DC-Lamp or Tat-Rev-Nef-DC-Lamp mRNA | Vaccine-specific immune response |
| [71] | 10 | HIV infection | Standard (5 days) | MCMM | EP with Gag and Nef mRNA | increased but short-lived CD4-responses against HIV gag and nef |
| [73] | 35 | HIV infection | Standard | TNFa + IFNγ + PGE2 + CD40L-mRNA | EP with Gag, Vpr, Rev, and Nef mRNA | none |
| Study (Reference) | # Pts | Disease + Combination Treatment | Transfection | Route and Target Dose | Safety Summary |
|---|---|---|---|---|---|
| [35] | 6 | Melanoma, stage IV | EP with aT-RNA | 5 × 106 sc in 3-weekly intervals for 4 cycles | 2 pts with fatigue (1 grade I, 1 grade II), 2 pts with nausea (1 grade I, 1 grade II), 1 pt with anorexia (grade II), 1 pt with arthralgia (grade I), 1 pt with confusion (grade I), 2 pts with diarrhea (grade I), 1 pt with hemorrhage (grade I), 1 pt with local reaction (grade I), 1 pt with myalgia (grade II), 1 pt with abdominal pain (grade II), 1 pt with bone pain (grade I), 1 pt with speech disorder (grade I), 1 pt with vomiting (grade I), 1 pt with wound infection (grade I) |
| [44] | 11 | Melanoma stage III | EP with gp100 or tyrosinase mRNA | 1.5 × 107 in biweekly intervals for 3 cycles | No side effects described |
| [41] | 35 | Melanoma, m | EP with MAGE-A3, -C2, tyrosinase, gp100 mRNA | 4.3 × 107 id 4 times in biweekly intervals; further vaccinations in case of residual vaccine after an 8 week interval | all pts: local reaction (grade II) 2 pts fever, myalgia, and asthenia grade II |
| [45] | 45 | Melanoma, m | EP with gp100 and tyrosinase mRNA | 12 × 106 cells 3 id times in biweekly intervals; 2 maintenance cycles for stable patients after 6 months respectively | local reaction: 23 pts grade I, 1 pt grade II flu like symptoms: 20 pts grade I, 10 pts grade II |
| [49] | 12 | Melanoma, m | EP with gp100, MelanA, tyrosinase, and MAGE-A3 mRNA +/− IP siRNA | 107 cells id 6 times in weekly intervals | No adverse events observed |
| [42] | 15 | Melanoma | EP with gp100-, tyrosinase-, MAGE-A3-, and -C2-DC-Lamp mRNA | Cohort 1: 2 × 107 id, 4 × 106 iv Cohort 2: 12 × 106 id, 12 × 106 iv Cohort 3: 4 × 106 id, 2 × 107 iv Cohort 4: 24 × 106 iv 4 vaccinations in biweekly intervals, 5th vaccination with 10 weeks interval | 11 pts local reaction grade II 3 pts chills grade II 8 pts flu like symptoms grade II 3 pts fever grade II |
| [46] | 15 | Melanoma | EP with tyrosinase and gp100 RNA | Up to 15 × 106 cells i.n. 3 times with maintenance cycles every 6 months | 4 pts local reaction grade I 4 pts flu like symptoms grade I |
| [40] | 30 | Melanoma (adjuvant) | EP with MAGE-A1-, -A3-, -C2-, tyrosinase-, MelanA-, and gp100-DC-Lamp RNA | ~24 × 106 id 4 to 6 times in biweekly intervals | 30 pts local reaction grade II 1 pt fever grade II 1 pt flu like symptoms 7 pts vitiligo |
| [47] | 28 | Melanoma stage III and IV | EP with gp100 and tyrosinase mRNA | 16 pts: 75 × 105 to 3 × 107 iv (2/3) and id (1/3) 12 pts: 15 × 105 to 16 × 107 intranodally 3 biweekly vaccinations per cycle, max 2 cycles in 6 months | flu-like: 11 pts grade I, 16 pts grade II, 1 pt grade III local reactions: 12 pts grade I, 13 pts grade II Hepatotoxicity: 9 pts grade I, 10 pts grade II, 5 pts grade III pneumonitis: 8 pts vitiligo 1 pt |
| [37] | 31 | Advanced melanoma | EP with aT-RNA | 4 weekly injection 2 × 107 intranodally (21) or id (10) then one id. 9 intranodally injected patients received IL-2 | Mild flu-like symptoms in some pts, pain in tumor, inflammatory reaction at injection site (grade I and II) 1 pt: vitiligo grade I no long term toxicity |
| [50] | 22 | Malignant melanomacyclophosphamide | EP with hTERT, survivin, p53 mRNA | 5 × 106 intermitting with cyclophosphamide for 6 cycles | Grade III: 1 pt: lung embolus from leukapheresis-catherization Grade I and II: 13 pts fatigue, 12 pts nausea, 7 pts diarrhea, 5 pts anemia, 1 pt neutropenia, 1 pt: hyperthyroidism, 1 pt vitiligo, 1 pt myalgia. (all not attributed to either vaccine or cyclophosphamide) |
| [43] | 39 | Pretreated advanced melanoma Ipilimumab | EP with MAGE-A3-, -C2-, tyrosinase-, and gp100-DC-LAMP mRNA | 4 × 106 id and 2 × 107 iv 1 h after Ipilimumab first 18 patients received one does DCs 2 weeks before Ipilimumab | DC-related: all pts: grade II injection site reactions 15 pts: grade I+II post-infusion chills 33 pts: grade I+II flu-like symptoms ICB-related: 14 pts: grade III+IV |
| [48] | 23 | Uveal melanoma | EP with gp100 and tyrosinase mRNA | up to 3 cycles of 3 biweekly iv and id injections in 6-month intervals | 21 pts: grade I and II flu-like symptoms, 20 pts: grade I and II local reactions, 1 pt vitiligo |
| [60] | 1 | Papillary ovarian cancer stage IIIc | EP with folat-R-mRNA | 2 to 50 × 106 id in monthly intervals for 10 cycles | No side effects |
| [62] | 2 | Ovarian cancer | EP with WT1 mRNA | 7–61 × 106 cells id 4 times in weekly intervals in Imiquimod pretreated skin | No signs of toxicity |
| [63] | 6 | Uterine cancer | EP with WT1 mRNA | 6–32 × 106 cells id 4 times in weekly intervals Imiquimod pretreated skin | 6 pts local reaction grade I |
| [31] | 10 | Stage III or IV renal cell carcinoma after nephrectomy | No EP, co-incubation with aT-RNA | 8 pts: 107 iv + 107 id every 2 weeks for 3 cycles 2 pts: 3 × 107 iv + 107 id every 2 weeks for 3 cycles | 5 pts with local reaction (grade I), 1 pt with anemia, 2 pts with dyspnea (both grade I, both considered unrelated to vaccine); |
| [34] | 11 | Renal cell cancer, m (10 pts), ovarial carcinoma (1pt) Ontak® (7 pts) | EP with aT-RNA | 107 id at biweekly intervals for 3 cycles | 4 pts with grade 1 rise of temperature and malaise (after Ontak®) 1 pt with elevation of RF (after Ontak®) 1 pt with transient ALT elevation (after Ontak) |
| [52] | 28 | Renal cell cancer cytokine-induced killer cells | EP with MUC-1 and Survivin mRNA | 2 × 107 to 5 × 107 cells sc 4 times in 2 days intervals | Flu like symptoms and fever grade I and II |
| [36] | 21 | Renal cell cancer sunitinib | EP with aT-RNA | 14 × 106 cells | Vaccine-related - all grade I or II: 7 pts: injection site erythema, 5 pts: Injection site induration, 4 pts rash, 3 pts diarrhea, 3 pts fatigue, 2 pts nausea, 2 pts headache, 1 pt decreased weight, 1 pt hypertension, 1 pt dysgeusia |
| [56] | 13 | Prostate cancer, m | No EP, co-incubation with PSA mRNA | 3 pts: 107 iv + 107 id for 3 cycles 3 pts: 3 × 107 iv + 107 id for 3 cycles 7 pts: 5 × 107 iv + 107 id for 3 cycles 2 week intervals | 4 pts with local reaction (grade I) 4 pts with grade I fever accompanied by flu-like symptoms following injection 1 pt with transiently elevated ANA and RF |
| [39] | 19 | Prostate cancer, androgen resistant | EP with allogeneic tumor RNA (3 human cancer cell lines) | 2 × 107 either intranodally (10 pts) or id (9 pts) weekly for 4 cycles | No grade II to IV side effects. Erythema at injection sites, increased size of draining lymph nodes, minor pain at injection site or small increase in hot flushes. |
| [57] | 20 | prostate cancer, m | EP with hTERT mRNA +/− LAMP | 107 id in weekly intervals (3 or 6 cycles) | 4 pts with constitutional symptoms (grade I) like fatigue or flu-like symptoms 18 pts with local reaction (grade I) 2 pts with transient elevation of ANA 1 pt with anemia and thrombocytopenia (grade III) considered unrelated to therapy |
| [59] | 21 | Castration- resistant prostate cancer Docetaxel | EP with PSA, PAP, survivin, hTERT mRNA | 5 × 106 twice during four Docetaxel-cycles, then one for 6 cycles, then only DCs every 3 months at patient decision | DC-related: local rash and pain only one pulmonary embolism related to leukapheresis procedure |
| [32] | 7 | Pediatric brain tumors | No EP, co-incubation with aT-RNA | 5 × 106/m2 iv + 5 × 106/m2 id | No measurable toxicity, no signs of autoimmunity |
| [33] | 8 | Pediatric neuroblastoma stage IV | No EP, co-incubation with aT-RNA | 5 × 106/m2 iv + 5 × 106/m2 id | No measurable toxicity, no signs of autoimmunity 1 pt with grade 1 skin reaction |
| [38] | 7 | Glioblastoma | EP with aT-RNA | 107 cells id; 2 vaccinations within first week, followed by 3 vaccinations in weekly intervals; rest of vaccinations in monthly intervals | fatigue: 6 pts grade I, 1 pt grade III 5 pts nausea/anorexia grade I pain: 3 pts grade 1, 1 pt grade II 1 pt constipation grade I |
| [66] | 12 | Glioblastoma injection site preconditioned with tetanus toxoid | EP with CMV pp65 mRNA | 2 × 107 cells id 3 times in biweekly intervals followed by monthly intervals | None |
| [69] | 11 | Glioblastoma temozolimide DCs mixed with GM-CSF | EP with CMV pp65 mRNA | three times 2 × 107 in biweekly intervals then monthly 6 to 12 times into the groin | No AEs in response to DCs, but one grade III SAE in response to the co-injected GM-CSF |
| [70] | 9 | Glioblastoma adoptive T-cell transfer | EP with CMV pp65 mRNA | three times 2 × 107 in biweekly intervals iv | 2 pts reduced CD4 count (grade II), 1 pt reduced platelet count (grade I) 1 pt reduced Neutrophil and WBC count (grade II) 1 pt reduced hematocrit (grade I) |
| [53] | 3 | Pancreatic adenocarcinoma CEA expressing | No EP, co-incubation with CEA mRNA | 107 loaded and 107 unloaded DCs id monthly for 6 cycles | 1 pt with liver abscess, 1 pt with upper respiratory infection (both considered unrelated to vaccine) |
| [65] | 42 | Pancreatic cancer cytotoxic lymphocytes gemcitabine | EP with MUC-1 mRNA | 4 × 105 to 39 × 106 cells id in monthly intervals | several grade 3 and adverse events, but attributed to T-cell transfer |
| [54] | 37 | CEA expressing cancer m (24 tumor bearing, 13 tumor free) | No EP, co-incubation with mRNA encoding CEA | 11 pts: 107 iv weekly for 4 weeks 4 pts: 3 × 107 iv + 106 id every 2 weeks for 4 cycles 14 pts: 108 iv + 106 id every 2 weeks for 4 cycles 8 patients additionally received 1.2 × 106 units IL-2 s.c. group 2: 13 pts: 3 × 107 iv + 106 id every 2 weeks for 4 cycles | No acute toxicities (no evidence of anaphylactic reactions or other cardiopulmonary compromise) Rise of body temperature of 0.28 °C (0.5 °F) Rise of mean arterial pressure of 6mm Hg Unrelated or tumor-related: 1 pt with rise in hepatic transaminases (from grade I to grade III) 1 pt with myelodysplastic syndrome 6 months after completing therapy 1 pt with an upper extremity deep vein thrombosis |
| [29] | 15 | Colorectal cancer, m | co-incubation aT-RNA | 4 × 106 iv every 4 weeks for 4 cycles | 2 pts with transient rigor and malaise |
| [55] | 5 | Colorectal cancer, m | EP with CEA mRNA | 5 × 106 id, 1.1 × 107 iv on day 0, 7 and 15. 3 cycles. | Flu like symptoms grade I, fever grade I, local reaction grade I |
| [30] | 1 | Adenocarcinoma, m | No EP, lipofection of aT-RNA | 3 × 107 iv + 106 id every 4 weeks for 4 cycles | No toxicities observed |
| [51] | 12 | Multiple myeloma | EP with BCMA, MAGE3, and survivin mRNA | 15 × 106 cells iv and 8 × 106 cells id 3 times in biweekly intervals | 8 pts local reaction grade I 10 pts fever, chills, malaise, muscle pain grade I/II |
| [58] | 21 | AML | EP with hTERT mRNA, +/− LAMP | 3 to 32 vaccinations with 107 DCs, first 6x in weekly intervals later biweekly | 1 pt idiopathic thrombocytopenia purpura (grade III) no other severe toxicities reported |
| [64] | 30 | AML | EP with WT1 mRNA +/− DC-Lamp | 5 × 106, 107 or 2 × 107 cells id in biweekly intervals followed by bimonthly vaccinations | all pts: local reaction at injection site (grade I) 1 pt pain in draining lymph nodes 1 pt drop of platelet count after 1st vaccination 1 pt flare up of pre-existing inflammation of the Achilles tendon |
| [67] | 12 | HCV-related hepato-carcinoma | EP with HSP70 mRNA | 3 times 107 to 3 × 107 with 3 week interval | 1 pt: grade I: ALT/AST increase 3 pts grade II: hyperglycemia, ALT increase, ALT/AST increase 1 pt grade III liver abscess (not treatment related) |
| [72] | 10 | HIV infection | EP with Gag, Vpr, Rev and Nef mRNA | 107 id in monthly intervals for 4 cycles | 6 patients with either fatigue (grade I), or local reaction at injection site (grade I), flu-like-symptoms (grade I), one pt with each: headache (grade I), diarrhea (grade I), axillary pain (grade I), RF increase (grade I). nausea (grade 1), increase in creatinine (grade I), hematochezia (grade I), eye inflammation (grade I), insomnia (grade I), SCC (grade II), reflux (grade II), GI pain (grade III), appendicitis (grade III), anemia (grade I) |
| [74] | 17 | HIV infection | EP with Tat-DC-Lamp, Rev-DC-Lamp or Nef-DC-Lamp mRNA | 3 × 107 cells sc and id 4 times in 4 week intervals | 16 pts: local reactions (grade I) |
| [75] | 6 | HIV infection | Gag-DC-Lamp or Tat-Rev-Nef-DC-Lamp mRNA | 107 cells sc (50%) and id (50%) 4 times in monthly intervals | 1 pt fever 6 pts local reaction |
| [68] | 7 | 4 healthy volunteers, 3 HSCT recipients | EP with CMV pp65 mRNA | 4 times 107 (HTSC-patients) or 105 (HV) id at weekly intervals | 7 pts local reaction grade II (all) 2 HVs headache grade I 1 HV myalgia grade I 1 pt moderate gastrointestinal GVHD grade II (HSCT) |
| [71] | 10 | HIV infection | EP with Gag and Nef mRNA | 4 × 5 × 106–15 × 106 DCs at week 0,2,6,10 | no AEs larger grade II |
| [73] | 35 | HIV infection | EP with Gag, Vpr, Rev, and Nef mRNA | 4 id-injections of at least 107 DCs with 4 week intervals | 25 pts local reactions (grade I) possibly related: headache, nausea, depression dizziness vivid dreams lymphadenopathy, rashes |
| NCT-Number | Country | Title | Antigen | Transfection * | Phase | Status |
|---|---|---|---|---|---|---|
| NCT01983748 | Germany | Dendritic Cells Plus Autologous Tumor RNA in Uveal Melanoma | aT-RNA | EP | III | recruiting |
| NCT03615404 | USA | Cytomegalovirus (CMV) RNA-Pulsed Dendritic Cells for Pediatric Patients and Young Adults with WHO Grade IV Glioma, Recurrent Malignant Glioma, or Recurrent Medulloblastoma | CMV-pp65-LAMP | pulsed | I | active, not recruiting |
| NCT02405338 | Norway | DC Vaccination for Post-remission Therapy in AML | WT1 Prame | transfected | I/II | active, not recruiting |
| NCT02465268 | USA | Vaccine Therapy for the Treatment of Newly Diagnosed Glioblastoma Multiforme | CMV pp65-LAMP | pulsed | II | recruiting |
| NCT02649582 | Belgium | Adjuvant Dendritic Cell-Immunotherapy Plus Temozolomide in Glioblastoma Patients | WT1 | loaded | I/II | recruiting |
| NCT01456104 | USA | Immune Responses to Autologous Langerhans-Type Dendritic Cells Electroporated with mRNA Encoding a Tumor-Associated Antigen in Patients With Malignancy: A Single-Arm Phase I Trial in Melanoma | mTRP2 | EP | I | active, not recruiting |
| NCT03083054 | Brazil | Cellular Immunotherapy for Patients with High Risk Myelodysplastic Syndromes and Acute Myeloid Leukemia | WT1 | EP | I/II | active, not recruiting |
| NCT04157127 | USA | Th-1 Dendritic Cell Immunotherapy Plus Standard Chemotherapy for Pancreatic Adenocarcinoma | ns | loaded | I | not yet recruiting |
| NCT01995708 | USA | CT7, MAGE-A3, and WT1 mRNA-electroporated Autologous Langerhans-type Dendritic Cells as Consolidation for Multiple Myeloma Patients Undergoing Autologous Stem Cell Transplantation | Mage-A3, Mage-C1, WT1 | EP | I | active, not recruiting |
| NCT01197625 | Norway | Vaccine Therapy in Curative Resected Prostate Cancer Patients | aT-RNA, hTERT, survivin | loaded | I/II | active, not recruiting |
| NCT01686334 | Belgium | Efficacy Study of Dendritic Cell Vaccination in Patients with Acute Myeloid Leukemia in Remission | WT1 | EP | II | recruiting |
| NCT03548571 | Norway | Dendritic Cell Immunotherapy Against Cancer Stem Cells in Glioblastoma Patients Receiving Standard Therapy | aTSC-RNA, survivin, hTERT | transfected | II/III | recruiting |
| NCT02366728 | USA | DC Migration Study for Newly Diagnosed GBM | CMV pp65-LAMP | pulsed | II | active, not recruiting |
| NCT02808416 | China | Personalized Cellular Vaccine for Brain Metastases (PERCELLVAC3) | ns | pulsed | I | active, not recruiting |
| NCT02649829 | Belgium | Autologous Dendritic Cell Vaccination in Mesothelioma | WT1 | loaded | I/II | recruiting |
| NCT00639639 | USA | Vaccine Therapy in Treating Patients with Newly Diagnosed Glioblastoma Multiforme | CMV pp65-LAMP | loaded | I | active, not recruiting |
| NCT02709616 | China | Personalized Cellular Vaccine for Glioblastoma (PERCELLVAC) | individually selected TAAs | pulsed | I | active, not recruiting |
| NCT03927222 | USA | Immunotherapy Targeted Against Cytomegalovirus in Patients with Newly Diagnosed WHO Grade IV Unmethylated Glioma | CMV pp65-LAMP | pulsed | II | recruiting |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dörrie, J.; Schaft, N.; Schuler, G.; Schuler-Thurner, B. Therapeutic Cancer Vaccination with Ex Vivo RNA-Transfected Dendritic Cells—An Update. Pharmaceutics 2020, 12, 92. https://doi.org/10.3390/pharmaceutics12020092
Dörrie J, Schaft N, Schuler G, Schuler-Thurner B. Therapeutic Cancer Vaccination with Ex Vivo RNA-Transfected Dendritic Cells—An Update. Pharmaceutics. 2020; 12(2):92. https://doi.org/10.3390/pharmaceutics12020092
Chicago/Turabian StyleDörrie, Jan, Niels Schaft, Gerold Schuler, and Beatrice Schuler-Thurner. 2020. "Therapeutic Cancer Vaccination with Ex Vivo RNA-Transfected Dendritic Cells—An Update" Pharmaceutics 12, no. 2: 92. https://doi.org/10.3390/pharmaceutics12020092
APA StyleDörrie, J., Schaft, N., Schuler, G., & Schuler-Thurner, B. (2020). Therapeutic Cancer Vaccination with Ex Vivo RNA-Transfected Dendritic Cells—An Update. Pharmaceutics, 12(2), 92. https://doi.org/10.3390/pharmaceutics12020092






