Boosting the Brain Delivery of Atazanavir through Nanostructured Lipid Carrier-Based Approach for Mitigating NeuroAIDS
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Animals Used
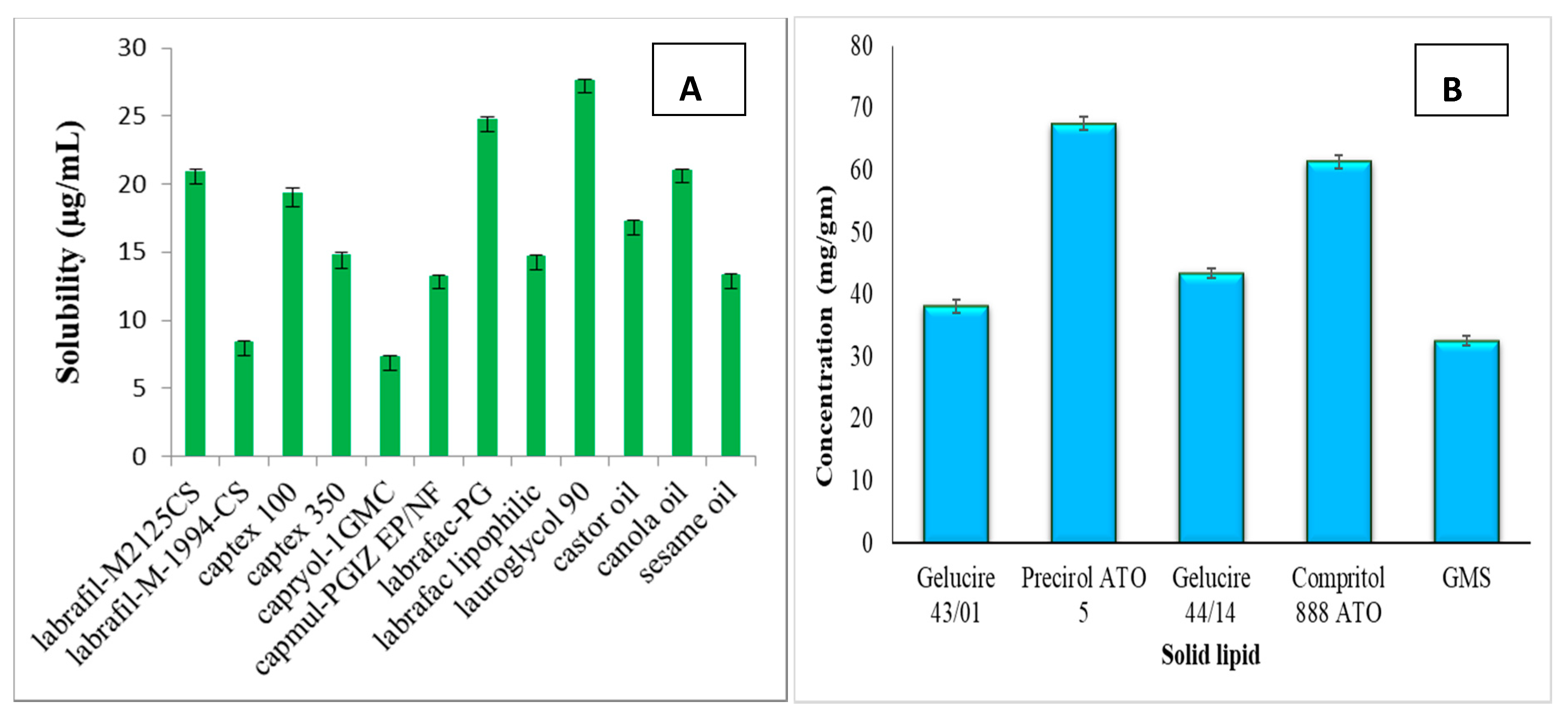
2.3. Screening of Lipids and Surfactants
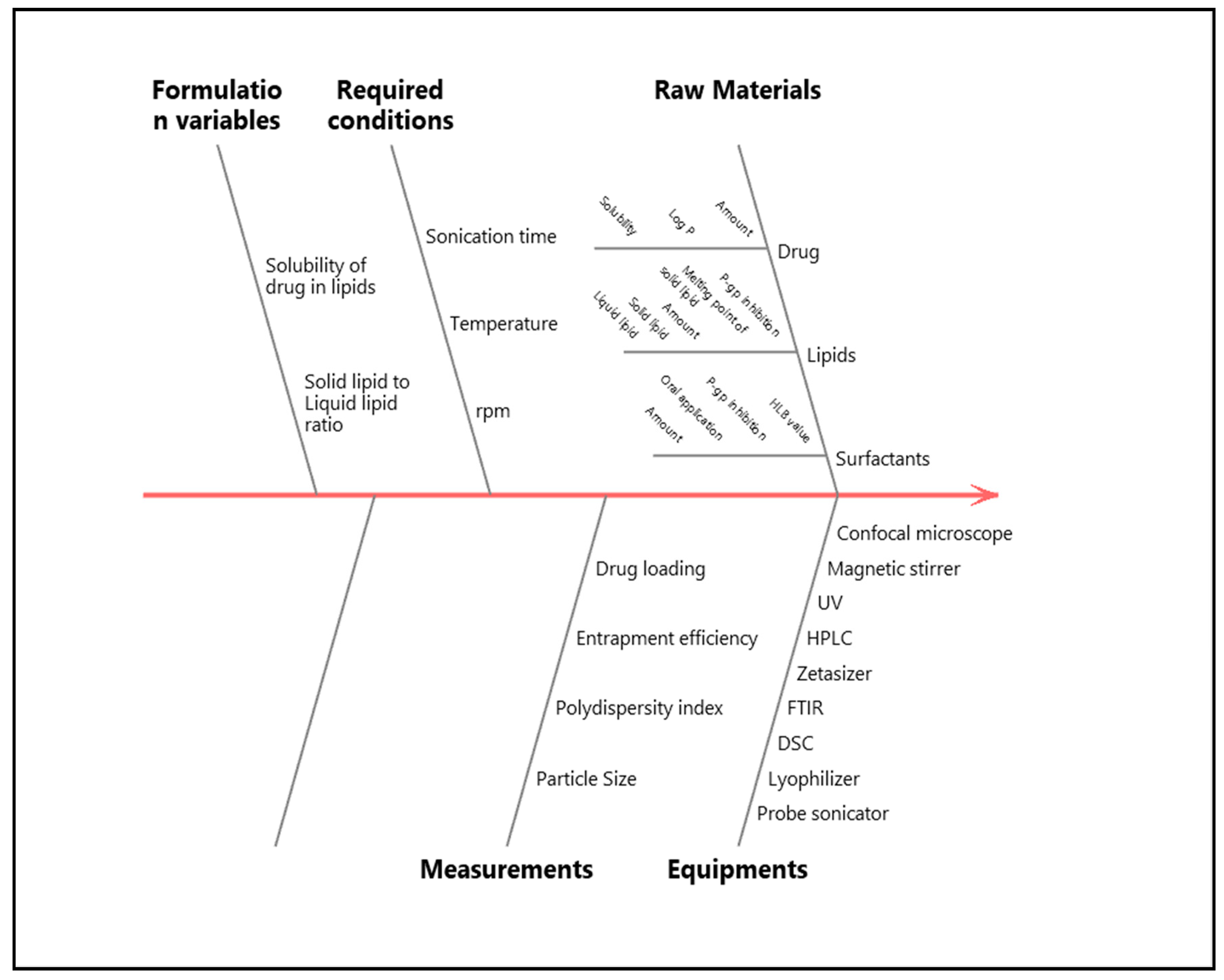
2.4. Quality by Design (QbD) Approach
2.5. Preparation of ATZ-NLC
2.6. Optimizing ATZ-NLC by the Box–Behnken Design (BBD)
2.7. Lyophilization of Optimized ATZ-NLC
2.8. Evaluation of Optimized ATZ-NLC
2.8.1. Particle Size, Polydispersity Index (PDI), and Zeta Potential (ZP)
2.8.2. Drug Entrapment Efficiency (%EE) and Drug Loading Capacity (%LC)
2.8.3. Transmission Electron Microscopy (TEM)
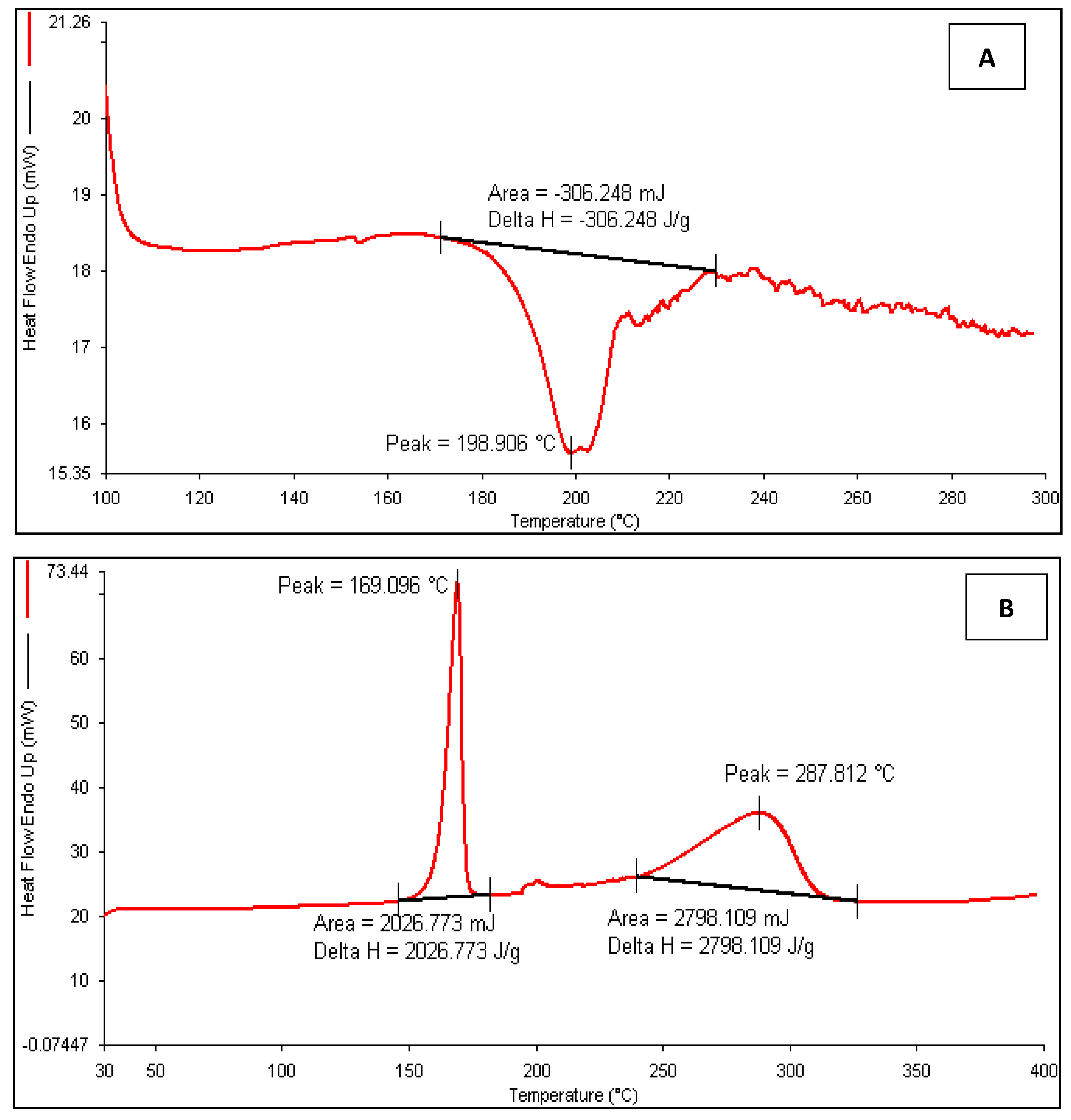
2.8.4. Differential Scanning Calorimetry (DSC)
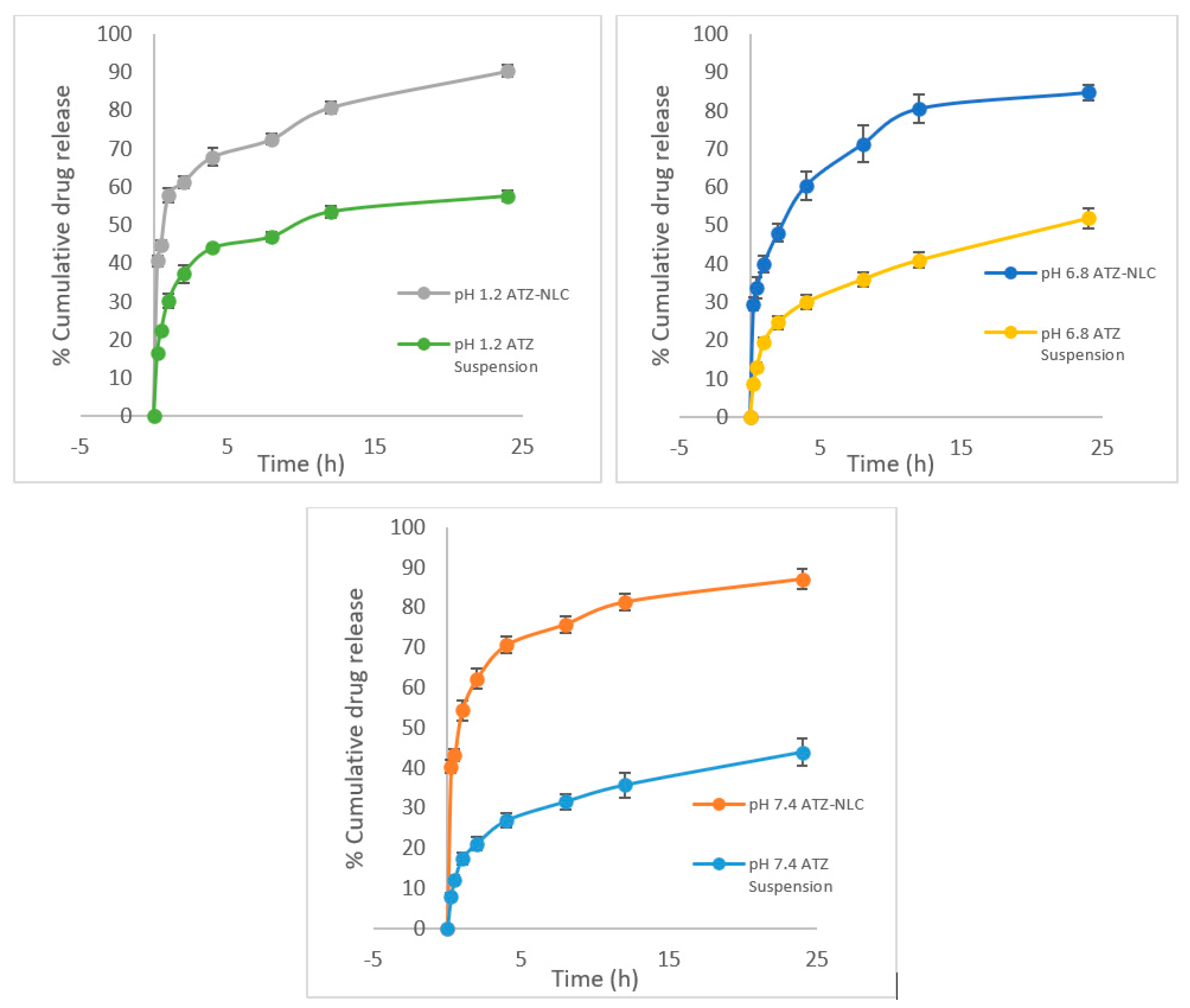
2.9. In Vitro Release Study
2.10. Quantification of ATZ by HPLC
2.11. Ex Vivo Permeation Study
2.12. Confocal Laser Scanning Microscopy (CLSM)
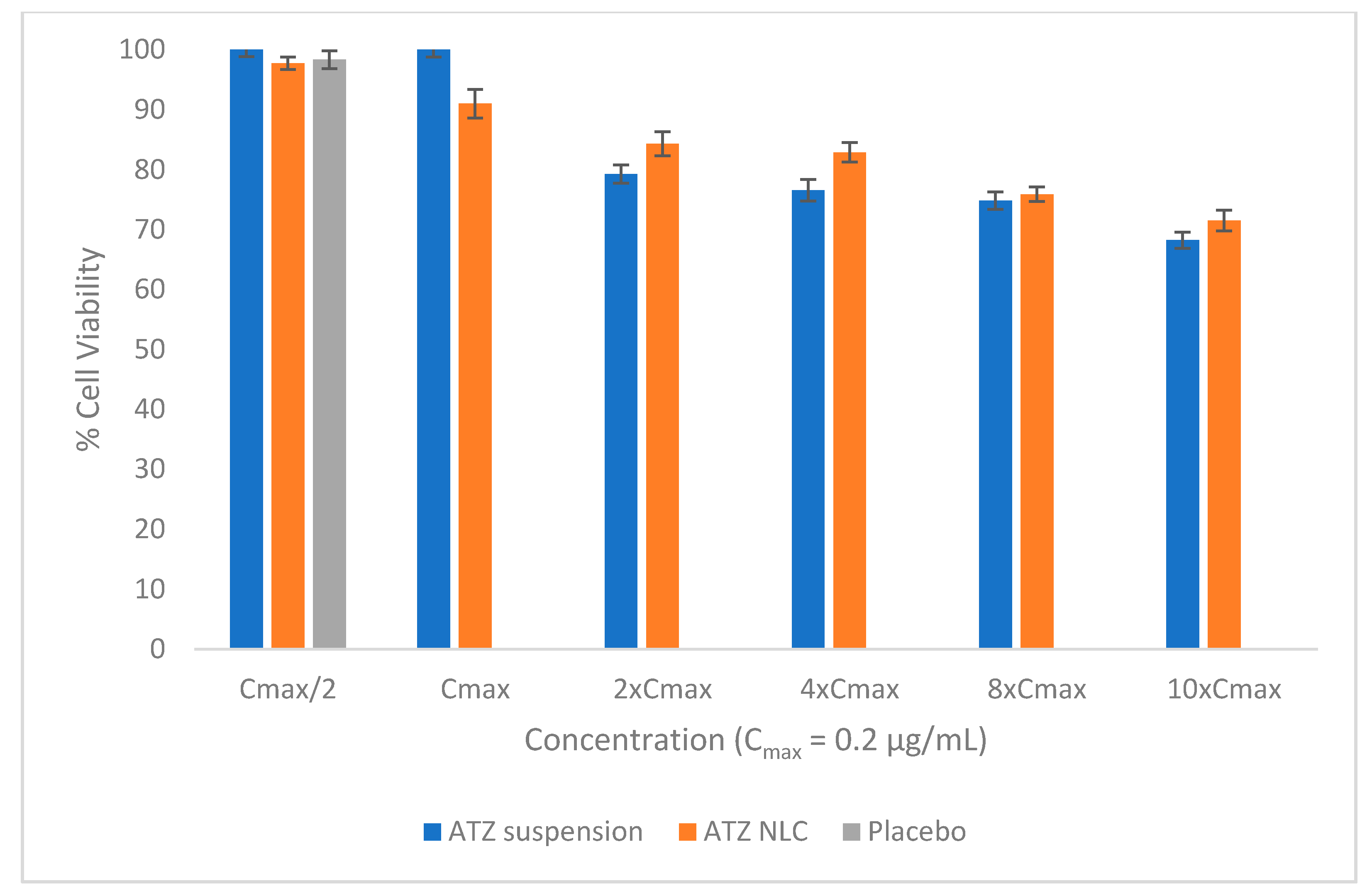
2.13. In Vitro Cell Viability Study
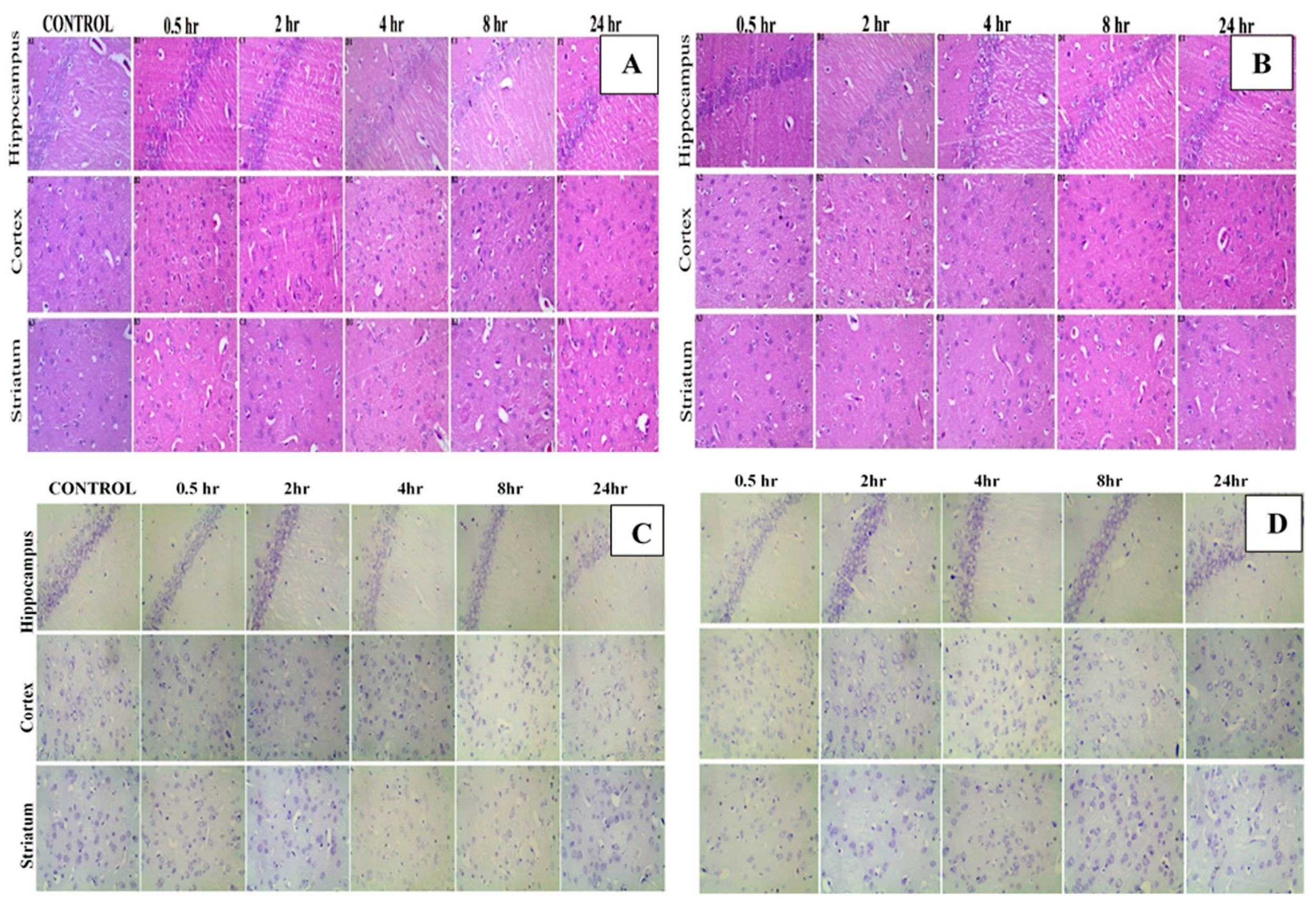
2.14. Histopathological Examination
2.15. In Vivo Study
2.15.1. Isolation and Extraction of Plasma and Brain Samples
2.15.2. Data Analysis
2.16. Stability Studies
2.17. Statistical Analysis
3. Result and Discussion
3.1. Screening of Lipids and Surfactants
3.2. Preparation and Optimization of ATZ-NLC
3.2.1. Fitting of Data to the Design
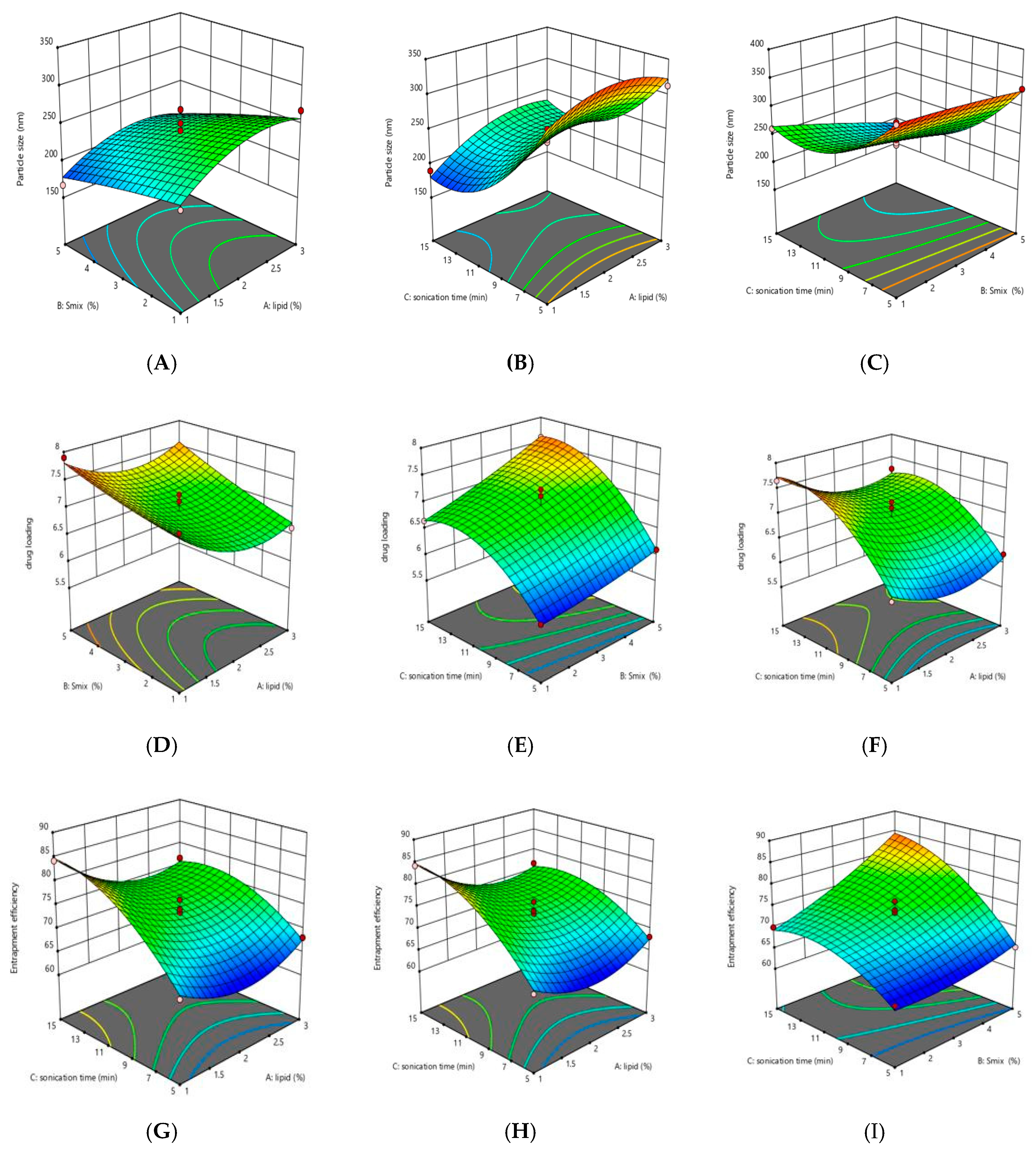
3.2.2. Response Analysis
3.2.3. Contour Plots
3.2.4. Validation of Experimental Design
3.3. Evaluation of Optimized ATZ-NLC
3.3.1. Particle Size, PDI, and ZP
3.3.2. Drug Entrapment Efficiency (% EE) and Drug Loading Capacity (% LC)
3.3.3. Transmission Electron Microscopy (TEM)
3.3.4. Differential Scanning Calorimetry (DSC)
3.4. In Vitro Release Study
3.5. Ex Vivo Permeation Study
3.6. Confocal Laser Scanning Microscopy
3.7. In Vitro Cell Viability Study
3.8. Histopathological Examination
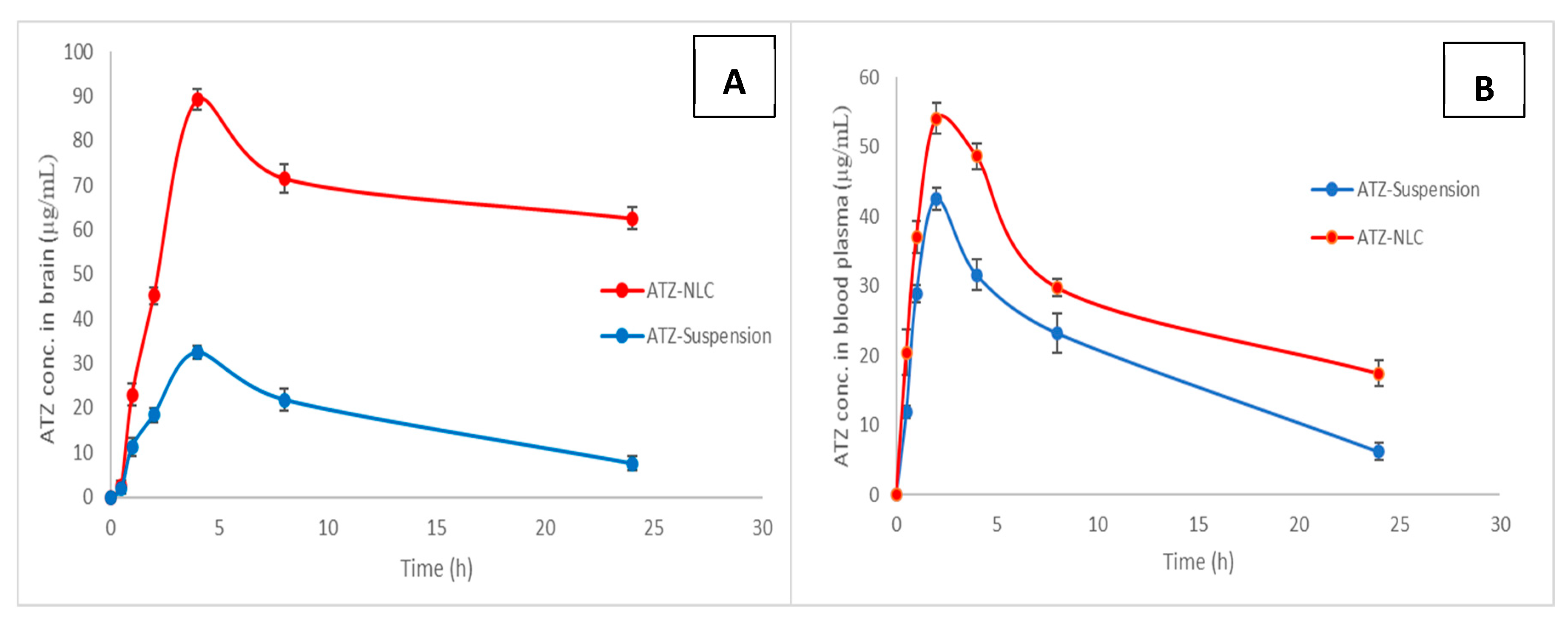
3.9. In Vivo Study
3.10. Stability Studies
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| AIDS | Acquired immunodeficiency syndrome |
| WHO | World Health Organization |
| UNAIDS | The Joint United Nations Program on HIV/AIDS |
| HIV | Human immunodeficiency virus |
| HAART | Highly active antiretroviral therapy |
| ARV | Antiretroviral |
| CNS | Central nervous system |
| BBB | Blood–brain barrier |
| P-gp | P-glycoprotein |
| NLC | Nanostructured lipid carriers |
| ATZ | Atazanavir |
| SLN | Solid lipid nanoparticle |
| SNEDDS | Self-nanoemulsifying drug delivery system |
| SLB | Solid–liquid binary lipid |
| QbD | Quality by Design |
| BBD | Box–Behnken design |
| RSM | Response surface methodology |
| PDI | Polydispersity index |
| ZP | Zeta potential |
| EE | Entrapment efficiency |
| LC | Loading capacity |
| TEM | Transmission electron microscopy |
| DSC | Differential scanning calorimetry |
| PBS | Phosphate buffer saline |
| HPLC | High performance liquid chromatographic method |
| CLSM | Confocal laser scanning microscopy |
| MTT | Methylthiazoletetrazolium |
| H&E | Hematoxylin and eosin |
| CV | Cresyl violet |
| ANOVA | Analysis of variance (ANOVA |
| GRAS | Generally recognized as safe |
| HLB | Hydrophilic–lipophilic balance |
References
- Chakraborty, T.; Das, M.K.; Dutta, L.; Mukherjee, B.; Das, S.; Sarma, A. Successful Delivery of Zidovudine-Loaded Docosanol Nanostructured Lipid Carriers (Docosanol NLCs) into Rat Brain. In Surface Modification of Nanoparticles for Targeted Drug Delivery; Springer Science and Business Media LLC: Berlin, Germany, 2019; pp. 245–276. [Google Scholar]
- Unaids.org. Switzerland: UNAIDS. 2018. Available online: https://www.unaids.org/en/regionscountries/countries/india (accessed on 17 June 2020).
- Sarma, A.; Das, M.K. Formulation by Design (FbD) approach to develop Tenofovir Disoproxil Fumarate loaded Nanostructured Lipid Carriers (NLCs) for the aptness of nose to brain delivery. J. Drug Deliv. Ther. 2019, 9, 148–159. [Google Scholar] [CrossRef]
- Pokharkar, V.; Patil-Gadhe, A.; Palla, P. Efavirenz loaded nanostructured lipid carrier engineered for brain targeting through intranasal route: In-vivo pharmacokinetic and toxicity study. Biomed. Pharmacother. 2017, 94, 150–164. [Google Scholar] [CrossRef] [PubMed]
- Chiappetta, D.A.; Höcht, C.; Opezzo, J.A.W.; Sosnik, A. Intranasal administration of antiretroviral-loaded micelles for anatomical targeting to the brain in HIV. Nanomedicine 2013, 8, 223–237. [Google Scholar] [CrossRef] [PubMed]
- Dalpiaz, A.; Ferraro, L.; Perrone, D.; Leo, E.; Iannuccelli, V.; Pavan, B.; Paganetto, G.; Beggiato, S.; Scalia, S. Brain Uptake of a Zidovudine Prodrug after Nasal Administration of Solid Lipid Microparticles. Mol. Pharm. 2014, 11, 1550–1561. [Google Scholar] [CrossRef] [PubMed]
- Nair, M.; Jayant, R.D.; Kaushik, A.; Sagar, V. Getting into the brain: Potential of nanotechnology in the management of NeuroAIDS. Adv. Drug Deliv. Rev. 2016, 103, 202–217. [Google Scholar] [CrossRef] [PubMed]
- Raymond, A.; Diaz, P.; Chevelon, S.; Agudelo, M.; Yndartarias, A.; Ding, H.; Kaushik, A.; Jayant, R.D.; Nikkhah-Moshaie, R.; Roy, U.; et al. Microglia-derived HIV Nef+ exosome impairment of the blood–brain barrier is treatable by nanomedicine-based delivery of Nef peptides. J. NeuroVirology 2016, 22, 129–139. [Google Scholar] [CrossRef]
- Alex, A.; Paul, W.; Chacko, A.J.; Sharma, C.P. Enhanced delivery of lopinavir to the CNS using Compritol®-based solid lipid nanoparticles. Ther. Deliv. 2011, 2, 25–35. [Google Scholar] [CrossRef]
- Makwana, V.; Jain, R.; Patel, K.; Nivsarkar, M.; Joshi, A. Solid lipid nanoparticles (SLN) of Efavirenz as lymph targeting drug delivery system: Elucidation of mechanism of uptake using chylomicron flow blocking approach. Int. J. Pharm. 2015, 495, 439–446. [Google Scholar] [CrossRef] [PubMed]
- Das, S.; Ng, W.K.; Tan, R.B. Are nanostructured lipid carriers (NLCs) better than solid lipid nanoparticles (SLNs): Development, characterizations and comparative evaluations of clotrimazole-loaded SLNs and NLCs? Eur. J. Pharm. Sci. 2012, 47, 139–151. [Google Scholar] [CrossRef]
- Kasongo, K.W.; Shegokar, R.; Müller, R.H.; Walker, R.B. Formulation development and in vitro evaluation of didanosine-loaded nanostructured lipid carriers for the potential treatment of AIDS dementia complex. Drug Dev. Ind. Pharm. 2010, 37, 396–407. [Google Scholar] [CrossRef]
- Annu; Rehman, S.; Shadab; Baboota, S.; Ali, J. Analyzing Nanotheraputics-Based Approaches for the Management of Psychotic Disorders. J. Pharm. Sci. 2019, 108, 3757–3768. [Google Scholar] [CrossRef]
- Wagner, S.; Zensi, A.; Wien, S.L.; Tschickardt, S.E.; Maier, W.; Vogel, T.; Worek, F.; Pietrzik, C.U.; Kreuter, J.; Von Briesen, H. Uptake Mechanism of ApoE-Modified Nanoparticles on Brain Capillary Endothelial Cells as a Blood-Brain Barrier Model. PLoS ONE 2012, 7, e32568. [Google Scholar] [CrossRef]
- Clark, A.J.; Davis, M.E. Increased brain uptake of targeted nanoparticles by adding an acid-cleavable linkage between transferrin and the nanoparticle core. Proc. Natl. Acad. Sci. USA 2015, 112, 12486–12491. [Google Scholar] [CrossRef]
- Van Rooy, I.; Cakir-Tascioglu, S.; Hennink, W.E.; Storm, G.; Schiffelers, R.M.; Mastrobattista, E. In Vivo Methods to Study Uptake of Nanoparticles into the Brain. Pharm. Res. 2011, 28, 456–471. [Google Scholar] [CrossRef] [PubMed]
- Santa-Maria, A.R.; Walter, F.R.; Valkai, S.; Brás, A.R.; Mészáros, M.; Kincses, A.; Klepe, A.; Gaspar, D.; Castanho, M.A.; Zimányi, L.; et al. Lidocaine turns the surface charge of biological membranes more positive and changes the permeability of blood-brain barrier culture models. Biochim. Biophys. Acta Biomembr. 2019, 1861, 1579–1591. [Google Scholar] [CrossRef] [PubMed]
- Singh, G.; Pai, R.S. Atazanavir-loaded Eudragit RL 100 nanoparticles to improve oral bioavailability: Optimization and in vitro/in vivo appraisal. Drug Deliv. 2016, 23, 532–539. [Google Scholar] [CrossRef]
- Singh, G.; Pai, R.S. Optimized self-nanoemulsifying drug delivery system of atazanavir with enhanced oral bioavailability:in vitro/in vivocharacterization. Expert Opin. Drug Deliv. 2014, 11, 1023–1032. [Google Scholar] [CrossRef]
- Chattopadhyay, N.; Zastre, J.; Wong, H.-L.; Wu, X.Y.; Bendayan, R. Solid Lipid Nanoparticles Enhance the Delivery of the HIV Protease Inhibitor, Atazanavir, by a Human Brain Endothelial Cell Line. Pharm. Res. 2008, 25, 2262–2271. [Google Scholar] [CrossRef]
- Müller, R.; Radtke, M.; Wissing, S. Solid lipid nanoparticles (SLN) and nanostructured lipid carriers (NLC) in cosmetic and dermatological preparations. Adv. Drug Deliv. Rev. 2002, 54, S131–S155. [Google Scholar] [CrossRef]
- Chatterjee, B.; Almurisi, S.H.; Dukhan, A.A.M.; Mandal, U.K.; Sengupta, P. Controversies with self-emulsifying drug delivery system from pharmacokinetic point of view. Drug Deliv. 2016, 23, 3639–3652. [Google Scholar] [CrossRef]
- Gambhire, V.; Narkhede, R.; Gujar, K. Design and evaluation of self-nanoemulsifying drug delivery systems for nebivolol hydrochloride. Asian, J. Pharm. 2014, 8, 200. [Google Scholar] [CrossRef]
- Devkar, T.B.; Tekade, A.R.; Khandelwal, K.R. Surface engineered nanostructured lipid carriers for efficient nose to brain delivery of ondansetron HCl using Delonix regia gum as a natural mucoadhesive polymer. Coll. Surf. B Biointerf. 2014, 122, 143–150. [Google Scholar] [CrossRef]
- Shete, H.; Patravale, V. Long chain lipid based tamoxifen NLC. Part I: Preformulation studies, formulation development and physicochemical characterization. Int. J. Pharm. 2013, 454, 573–583. [Google Scholar] [CrossRef]
- Iqbal, B.; Ali, J.; Baboota, S. Silymarin loaded nanostructured lipid carrier: From design and dermatokinetic study to mechanistic analysis of epidermal drug deposition enhancement. J. Mol. Liq. 2018, 255, 513–529. [Google Scholar] [CrossRef]
- Cunha, S.; Costa, C.P.; Loureiro, J.A.; Alves, J.; Peixoto, A.F.; Forbes, B.; Lobo, J.M.S.; Silva, A. Double Optimization of Rivastigmine-Loaded Nanostructured Lipid Carriers (NLC) for Nose-to-Brain Delivery Using the Quality by Design (QbD) Approach: Formulation Variables and Instrumental Parameters. Pharmaceutics 2020, 12, 599. [Google Scholar] [CrossRef]
- Elmowafy, M.; Ibrahim, H.M.; Ahmed, M.A.; Shalaby, K.; Salama, A.; Hefesha, H. Atorvastatin-loaded nanostructured lipid carriers (NLCs): Strategy to overcome oral delivery drawbacks. Drug Deliv. 2017, 24, 932–941. [Google Scholar] [CrossRef]
- Jazuli, I.; Nabi, B.; Moolakkadath, T.; Alam, T.; Baboota, S.; Ali, J.; Annu; Annu, I.J. Optimization of Nanostructured Lipid Carriers of Lurasidone Hydrochloride Using Box-Behnken Design for Brain Targeting: In Vitro and In Vivo Studies. J. Pharm. Sci. 2019, 108, 3082–3090. [Google Scholar] [CrossRef]
- Kudarha, R.; Dhas, N.L.; Pandey, A.; Belgamwar, V.S.; Ige, P.P. Box–Behnken study design for optimization of bicalutamide-loaded nanostructured lipid carrier: Stability assessment. Pharm. Dev. Technol. 2014, 20, 608–618. [Google Scholar] [CrossRef] [PubMed]
- Tsai, M.-J.; Wu, P.-C.; Huang, Y.-B.; Chang, J.-S.; Lin, C.-L.; Tsai, Y.-H.; Fang, J.-Y. Baicalein loaded in tocol nanostructured lipid carriers (tocol NLCs) for enhanced stability and brain targeting. Int. J. Pharm. 2012, 423, 461–470. [Google Scholar] [CrossRef]
- Duong, V.-A.; Nguyen, T.-T.-L.; Maeng, H.-J.; Chi, S.-C. Nanostructured lipid carriers containing ondansetron hydrochloride by cold high-pressure homogenization method: Preparation, characterization, and pharmacokinetic evaluation. J. Drug Deliv. Sci. Technol. 2019, 53. [Google Scholar] [CrossRef]
- Shah, B.; Khunt, D.; Bhatt, H.; Misra, M.; Padh, H. Intranasal delivery of venlafaxine loaded nanostructured lipid carrier: Risk assessment and QbD based optimization. J. Drug Deliv. Sci. Technol. 2016, 33, 37–50. [Google Scholar] [CrossRef]
- Konidala, S.K.; Sujana, K.; Rani, A.P. New Validated RP-HPLC method for the Determination of Atazanavir Sulphate in Bulk and Dosage form. Der. Pharma. Chemica. 2012, 4, 1305–1310. [Google Scholar]
- Rehman, S.; Nabi, B.; Fazil, M.; Khan, S.; Bari, N.K.; Singh, R.; Ahmad, S.; Kumar, V.; Baboota, S.; Ali, J. Role of P-Glycoprotein Inhibitors in the Bioavailability Enhancement of Solid Dispersion of Darunavir. BioMed Res. Int. 2017, 2017, 1–17. [Google Scholar] [CrossRef]
- Neupane, Y.R.; Srivastava, M.; Ahmad, N.; Kumar, N.; Bhatnagar, A.; Kohli, K. Lipid based nanocarrier system for the potential oral delivery of decitabine: Formulation design, characterization, ex vivo, and in vivo assessment. Int. J. Pharm. 2014, 477, 601–612. [Google Scholar] [CrossRef]
- Sharma, S.; Rabbani, S.A.; Narang, J.K.; Pottoo, F.H.; Ali, J.; Kumar, S.; Baboota, S. Role of Rutin Nanoemulsion in Ameliorating Oxidative Stress: Pharmacokinetic and Pharmacodynamics Studies. Chem. Phys. Lipids 2020, 228, 104890. [Google Scholar] [CrossRef]
- Garg, B.; Beg, S.; Kumar, R.; Katare, O.; Singh, B. Nanostructured lipidic carriers of lopinavir for effective management of HIV-associated neurocognitive disorder. J. Drug Deliv. Sci. Technol. 2019, 53. [Google Scholar] [CrossRef]
- Kasongo, K.W.; Pardeike, J.; Müller, R.H.; Walker, R.B. Selection and Characterization of Suitable Lipid Excipients for use in the Manufacture of Didanosine-Loaded Solid Lipid Nanoparticles and Nanostructured Lipid Carriers. J. Pharm. Sci. 2011, 100, 5185–5196. [Google Scholar] [CrossRef] [PubMed]
- Alam, T.; Khan, S.; Gaba, B.; Haider, F.; Baboota, S.; Ali, J. Adaptation of Quality by Design-Based Development of Isradipine Nanostructured–Lipid Carrier and Its Evaluation for In Vitro Gut Permeation and In Vivo Solubilization Fate. J. Pharm. Sci. 2018, 107, 2914–2926. [Google Scholar] [CrossRef]
- Negi, L.M.; Jaggi, M.; Talegaonkar, S. Development of protocol for screening the formulation components and the assessment of common quality problems of nano-structured lipid carriers. Int. J. Pharm. 2014, 461, 403–410. [Google Scholar] [CrossRef]
- Meng, F.; Asghar, S.; Xu, Y.; Wang, J.; Jin, X.; Wang, Z.; Wang, J.; Ping, Q.; Zhou, J.; Xiao, Y. Design and evaluation of lipoprotein resembling curcumin-encapsulated protein-free nanostructured lipid carrier for brain targeting. Int. J. Pharm. 2016, 506, 46–56. [Google Scholar] [CrossRef]
- Kreuter, J. Influence of the Surface Properties on Nanoparticle-Mediated Transport of Drugs to the Brain. J. Nanosci. Nanotechnol. 2004, 4, 484–488. [Google Scholar] [CrossRef]
- Mandpe, L.; Pokharkar, V. Quality by design approach to understand the process of optimization of iloperidone nanostructured lipid carriers for oral bioavailability enhancement. Pharm. Dev. Technol. 2013, 20, 320–329. [Google Scholar] [CrossRef]
- Fatima, N.; Rehman, S.; Nabi, B.; Baboota, S.; Ali, J. Harnessing nanotechnology for enhanced topical delivery of clindamycin phosphate. J. Drug Deliv. Sci. Technol. 2019, 54, 101253. [Google Scholar] [CrossRef]
- De Souza, I.D.L.; Saez, V.; De Campos, V.E.B.; Mansur, C.R.E. Size and Vitamin E Release of Nanostructured Lipid Carriers with Different Liquid Lipids, Surfactants and Preparation Methods. Macromol. Symp. 2019, 383. [Google Scholar] [CrossRef]
- Jia, L.-J.; Zhang, D.-R.; Li, Z.-Y.; Feng, F.-F.; Wang, Y.; Dai, W.-T.; Duan, C.-X.; Zhang, Q. Preparation and characterization of silybin-loaded nanostructured lipid carriers. Drug Deliv. 2009, 17, 11–18. [Google Scholar] [CrossRef]
- Cavalcanti, S.M.T.; Nunes, C.; Lima, S.A.C.; Soares-Sobrinho, J.L.; Reis, S. Multiple Lipid Nanoparticles (MLN), a New Generation of Lipid Nanoparticles for Drug Delivery Systems: Lamivudine-MLN Experimental Design. Pharm. Res. 2017, 34, 1204–1216. [Google Scholar] [CrossRef]
- Alam, T.P.J.; Vohora, D.; Aqil, M.; Aqil, M.; Ali, A.; Sultana, Y. Optimization of nanostructured lipid carriers of lamotrigine for brain delivery: In vitro characterization and in vivo efficacy in epilepsy. Expert Opin. Drug Deliv. 2015, 12, 181–194. [Google Scholar] [CrossRef]
- Singh, S.K.; Dadhania, P.; Vuddanda, P.R.; Jain, A.; Velaga, S. Intranasal delivery of asenapine loaded nanostructured lipid carriers: Formulation, characterization, pharmacokinetic and behavioural assessment. RSC Adv. 2016, 6, 2032–2045. [Google Scholar] [CrossRef]
- Gabal, Y.M.; Kamel, A.O.; Sammour, O.A.; Elshafeey, A.H. Effect of surface charge on the brain delivery of nanostructured lipid carriers in situ gels via the nasal route. Int. J. Pharm. 2014, 473, 442–457. [Google Scholar] [CrossRef]
- Seyfoddin, A.; Al-Kassas, R. Development of solid lipid nanoparticles and nanostructured lipid carriers for improving ocular delivery of acyclovir. Drug Dev. Ind. Pharm. 2012, 39, 508–519. [Google Scholar] [CrossRef]
- Muthu, M.S.; Sahu, A.K.; Sonali; Abdulla, A.; Kaklotar, D.; Rajesh, C.V.; Singh, S.; Pandey, B.L. Solubilized delivery of paliperidone palmitate by d-alpha-tocopheryl polyethylene glycol 1000 succinate micelles for improved short-term psychotic management. Drug Deliv. 2014, 23, 230–237. [Google Scholar] [CrossRef] [PubMed]
- Jindal, A.B.; Bachhav, S.S.; Devarajan, P.V. In situ hybrid nano drug delivery system (IHN-DDS) of antiretroviral drug for simultaneous targeting to multiple viral reservoirs: An in vivo proof of concept. Int. J. Pharm. 2017, 521, 196–203. [Google Scholar] [CrossRef] [PubMed]
- Ahmadi, N.; Rostamizadeh, K.; Modarresi-Alam, A.R. Therapeutic Anti-Inflammatory Potential of Different Formulations Based on Coenzyme Q10-Loaded Nanostructured Lipid Carrier: In Vitro, Ex vivo, and In Vivo Evaluations. Eur. J. Lipid Sci. Technol. 2018, 120, 1800225. [Google Scholar] [CrossRef]
- Nabi, B.; Rehman, S.; Aggarwal, S.; Baboota, S.; Ali, J. Quality by Design Adapted Chemically Engineered Lipid Architectonics for HIV Therapeutics and Intervention: Contriving of Formulation, Appraising the In vitro Parameters and In vivo Solubilization Potential. AAPS PharmSciTech 2020, 21, 1–17. [Google Scholar] [CrossRef]
- Forster, S.; Thumser, A.E.; Hood, S.R.; Plant, N. Characterization of Rhodamine-123 as a Tracer Dye for Use In In vitro Drug Transport Assays. PLoS ONE 2012, 7, e33253. [Google Scholar] [CrossRef]
- Rahman, H.S.; Rasedee, A.; Othman, H.H.; Chartrand, M.S.; Namvar, F.; Yeap, S.K.; Samad, N.A.; Andas, R.J.; Nadzri, N.M.; Anasamy, T.; et al. Acute Toxicity Study of Zerumbone-Loaded Nanostructured Lipid Carrier on BALB/c Mice Model. BioMed Res. Int. 2014, 2014, 1–15. [Google Scholar] [CrossRef]
- Singh, S.K.; Hidau, M.K.; Gautam, S.; Gupta, K.; Singh, K.P.; Singh, S.K. Glycol chitosan functionalized asenapine nanostructured lipid carriers for targeted brain delivery: Pharmacokinetic and teratogenic assessment. Int. J. Biol. Macromol. 2018, 108, 1092–1100. [Google Scholar] [CrossRef]
- Khan, N.; Shah, F.A.; Rana, I.; Ansari, M.M.; Din, F.U.; Rizvi, S.Z.H.; Aman, W.; Lee, G.-Y.; Lee, E.-S.; Kim, J.-K.; et al. Nanostructured lipid carriers-mediated brain delivery of carbamazepine for improved in vivo anticonvulsant and anxiolytic activity. Int. J. Pharm. 2020, 577, 119033. [Google Scholar] [CrossRef]
- Zhuang, C.-Y.; Li, N.; Wang, M.; Zhang, X.-N.; Pan, W.; Peng, J.-J.; Pan, Y.-S.; Tang, X. Preparation and characterization of vinpocetine loaded nanostructured lipid carriers (NLC) for improved oral bioavailability. Int. J. Pharm. 2010, 394, 179–185. [Google Scholar] [CrossRef]
- Misra, S.; Chopra, K.; Sinha, V.R.; Medhi, B. Galantamine-loaded solid–lipid nanoparticles for enhanced brain delivery: Preparation, characterization, in vitro and in vivo evaluations. Drug Deliv. 2015, 23, 1434–1443. [Google Scholar] [CrossRef]


| QTPP | ||
| QTPP | Target | Justification |
| Drug delivery system | Nanostructured lipid carriers | Offers augmentation in oral bioavailability |
| Dosage type | Controlled release | Enhanced drug absorption will be achieved |
| Route of administration | Oral | Patient compliant route offering lymphatic uptake. |
| Drug release | More than 80% | Essential for attained optimal therapeutic activity |
| CQA | ||
| CQA | Target | Justification |
| Particle size | Less than 250 nm | Absorption and bioavailability are improved |
| Entrapment efficiency | More than 70% | Improved therapeutic outcome and pharmacological activity. |
| Drug loading | More than 8% | Improved therapeutic outcome and pharmacological activity |
| Variables | Levels | ||
|---|---|---|---|
| Independent variables | Low (−1) | Medium (0) | High (+1) |
| Total Lipid (%) | 1 | 2 | 3 |
| Surfactant (%) | 1 | 3 | 5 |
| Sonication time (min) | 5 | 10 | 15 |
| Dependent variables | Goals | ||
| Particle size (nm) | Minimize | ||
| Entrapment efficiency (%) | Maximize | ||
| Drug Loading (%) | Maximize | ||
| Surfactants | % Transmittance |
|---|---|
| Span 20 | 0.744 |
| Poloxamer | 2.78 |
| Cremophor RH40 | 97.71 |
| Poloxamer 188 | 9.77 |
| Tween 20 | 45.57 |
| Tween 80 | 73.15 |
| Solutol HS15 | 48.5 |
| Std | Run | A: Total Lipid % | B: Surfactant % | C: Sonication Time (min) | Particle Size (nm) | Entrapment Efficiency % | Loading Capacity % |
|---|---|---|---|---|---|---|---|
| 4 | 1 | 3 | 5 | 10 | 208.4 | 82.93 | 7.5 |
| 16 | 2 | 2 | 3 | 10 | 237.4 | 70.98 | 6.85 |
| 13 | 3 | 2 | 3 | 10 | 231.8 | 73.43 | 7.24 |
| 17 | 4 | 2 | 3 | 10 | 235.4 | 76.18 | 7.12 |
| 10 | 5 | 2 | 5 | 5 | 331.4 | 65.22 | 6.1 |
| 2 | 6 | 3 | 1 | 10 | 268.2 | 70.08 | 6.63 |
| 8 | 7 | 3 | 3 | 15 | 227.8 | 76.62 | 7.25 |
| 9 | 8 | 2 | 1 | 5 | 349.2 | 63.66 | 5.71 |
| 15 | 9 | 2 | 3 | 10 | 241.4 | 74.12 | 6.78 |
| 3 | 10 | 1 | 5 | 10 | 167.1 | 86.75 | 7.9 |
| 14 | 11 | 2 | 3 | 10 | 251.3 | 72.19 | 6.72 |
| 5 | 12 | 1 | 3 | 5 | 321.4 | 66.98 | 6.2 |
| 12 | 13 | 2 | 5 | 15 | 192.4 | 83.43 | 7.6 |
| 7 | 14 | 1 | 3 | 15 | 189.9 | 84.23 | 7.65 |
| 6 | 15 | 3 | 3 | 5 | 312.8 | 68.12 | 6.18 |
| 11 | 16 | 2 | 1 | 15 | 261.4 | 69.98 | 6.65 |
| 1 | 17 | 1 | 1 | 10 | 212.6 | 77.92 | 7.36 |
| Period (Days) | Particle Size (nm) | PDI | Entrapment Efficiency (%) | Physical Appearance | Phase Separation | Precipitate Formation |
|---|---|---|---|---|---|---|
| 0 | 230.2 ± 4.14 | 0.342 ± 0.022 | 71.5 ± 4.13 | No change | No | No |
| 30 | 234.6 ± 5.26 | 0.357 ± 0.031 | 71.3 ± 2.48 | No change | No | No |
| 60 | 237.5 ± 4.76 | 0.401 ± 0.025 | 70.1 ± 1.46 | No change | No | No |
| 90 | 238.6 ± 5.28 | 0.418 ± 0.027 | 68.8 ± 3.29 | No change | No | No |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khan, S.A.; Rehman, S.; Nabi, B.; Iqubal, A.; Nehal, N.; Fahmy, U.A.; Kotta, S.; Baboota, S.; Md, S.; Ali, J. Boosting the Brain Delivery of Atazanavir through Nanostructured Lipid Carrier-Based Approach for Mitigating NeuroAIDS. Pharmaceutics 2020, 12, 1059. https://doi.org/10.3390/pharmaceutics12111059
Khan SA, Rehman S, Nabi B, Iqubal A, Nehal N, Fahmy UA, Kotta S, Baboota S, Md S, Ali J. Boosting the Brain Delivery of Atazanavir through Nanostructured Lipid Carrier-Based Approach for Mitigating NeuroAIDS. Pharmaceutics. 2020; 12(11):1059. https://doi.org/10.3390/pharmaceutics12111059
Chicago/Turabian StyleKhan, Saif Ahmad, Saleha Rehman, Bushra Nabi, Ashif Iqubal, Nida Nehal, Usama A. Fahmy, Sabna Kotta, Sanjula Baboota, Shadab Md, and Javed Ali. 2020. "Boosting the Brain Delivery of Atazanavir through Nanostructured Lipid Carrier-Based Approach for Mitigating NeuroAIDS" Pharmaceutics 12, no. 11: 1059. https://doi.org/10.3390/pharmaceutics12111059
APA StyleKhan, S. A., Rehman, S., Nabi, B., Iqubal, A., Nehal, N., Fahmy, U. A., Kotta, S., Baboota, S., Md, S., & Ali, J. (2020). Boosting the Brain Delivery of Atazanavir through Nanostructured Lipid Carrier-Based Approach for Mitigating NeuroAIDS. Pharmaceutics, 12(11), 1059. https://doi.org/10.3390/pharmaceutics12111059