Ex Vivo Conjunctival Retention and Transconjunctival Transport of Poorly Soluble Drugs Using Polymeric Micelles
Abstract
1. Introduction
2. Materials and Methods
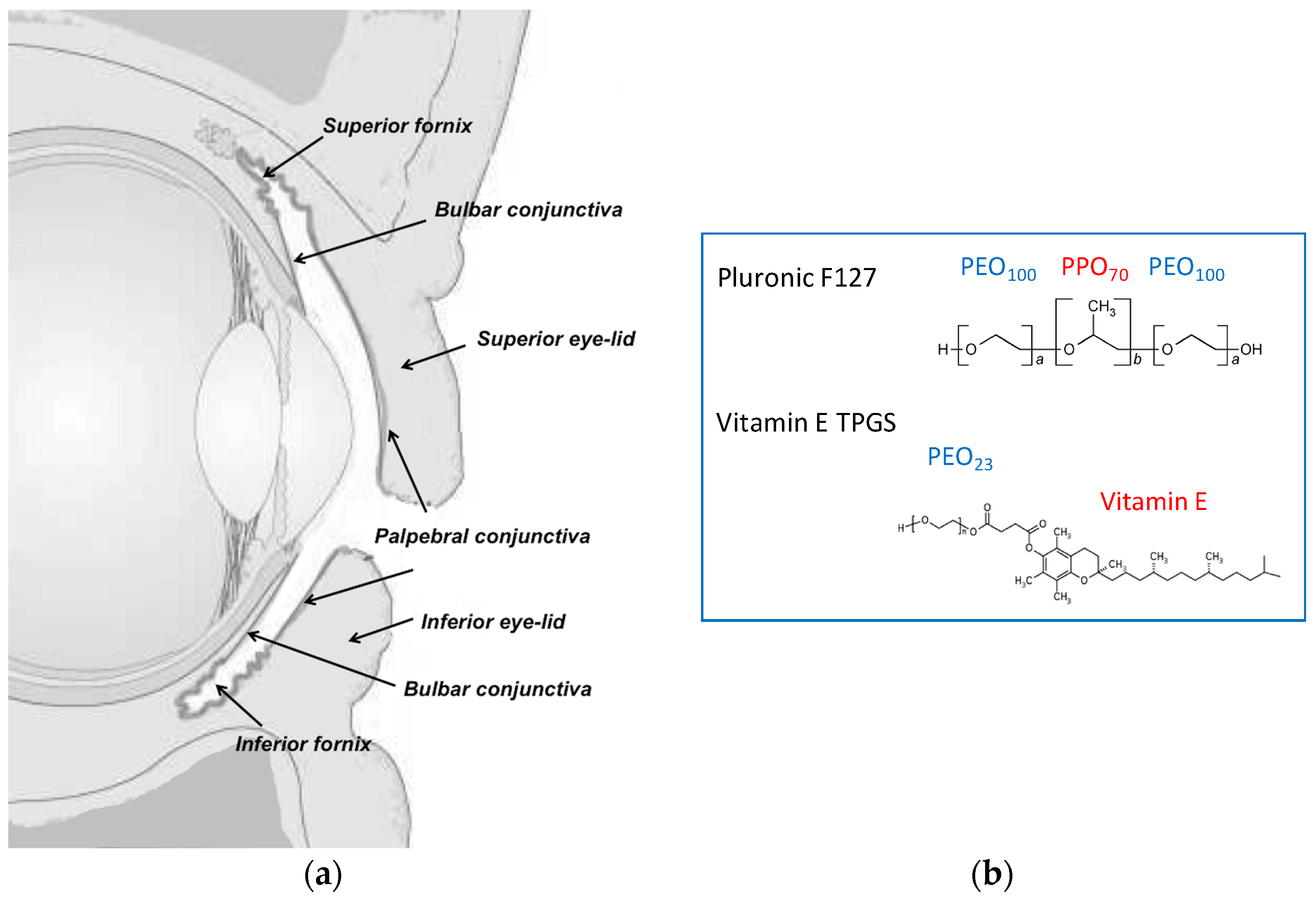
2.1. Materials
2.2. Micelle Preparation
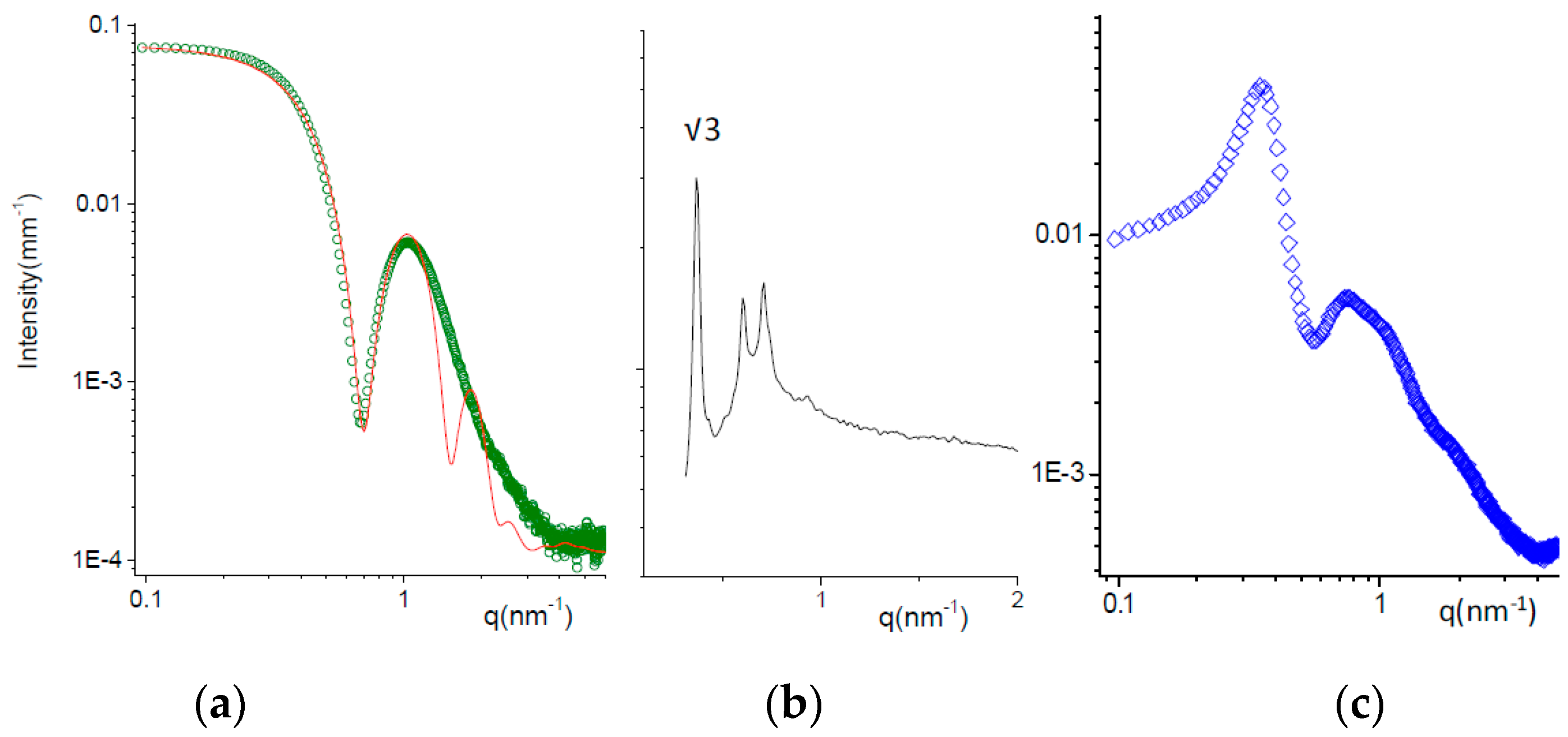
2.3. Micelle Characterization: Dynamic Light Scattering and SAXS Analysis
2.4. Drug Solubility Studies
2.5. Tissues
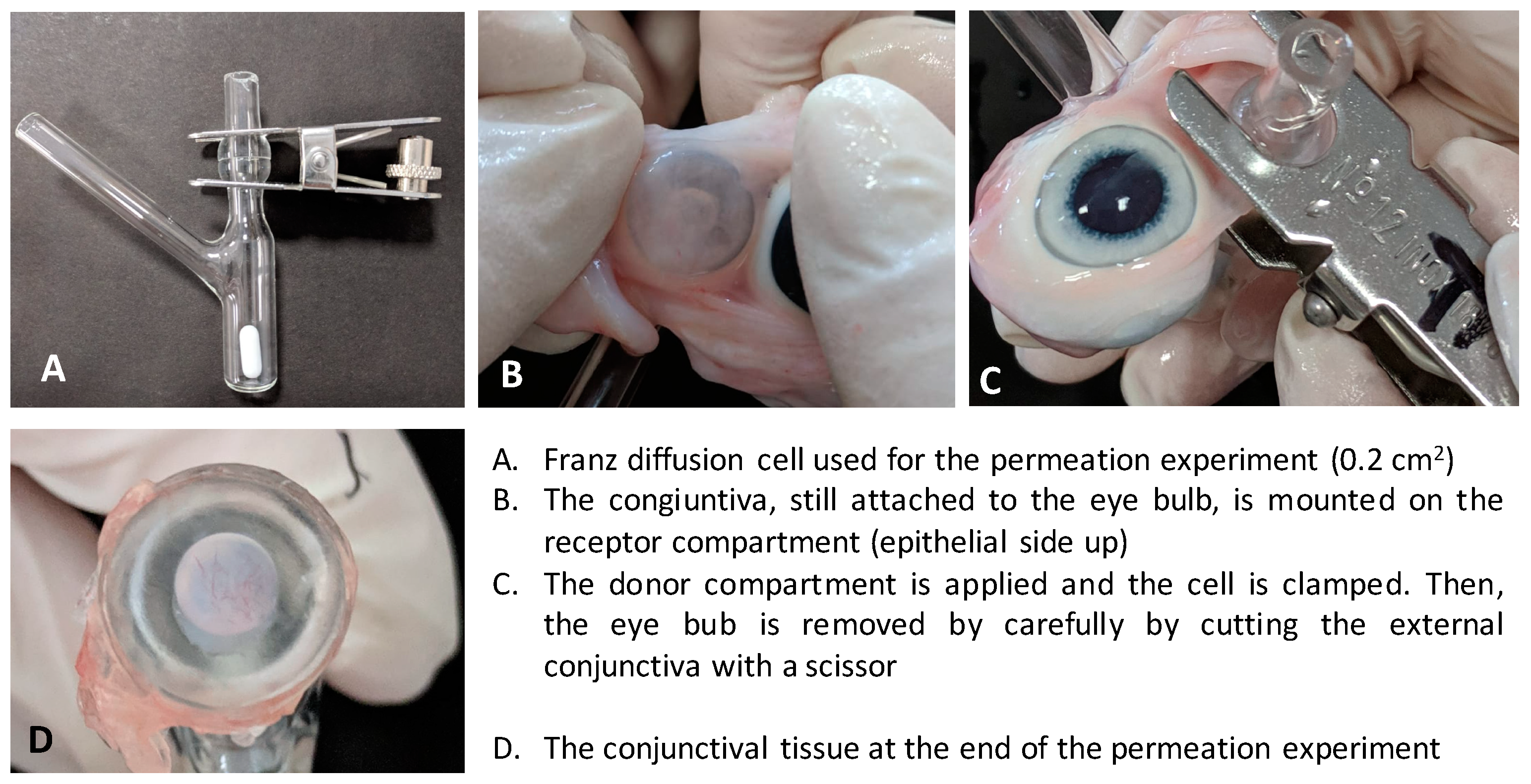
2.6. Transconjunctival Permeation Studies
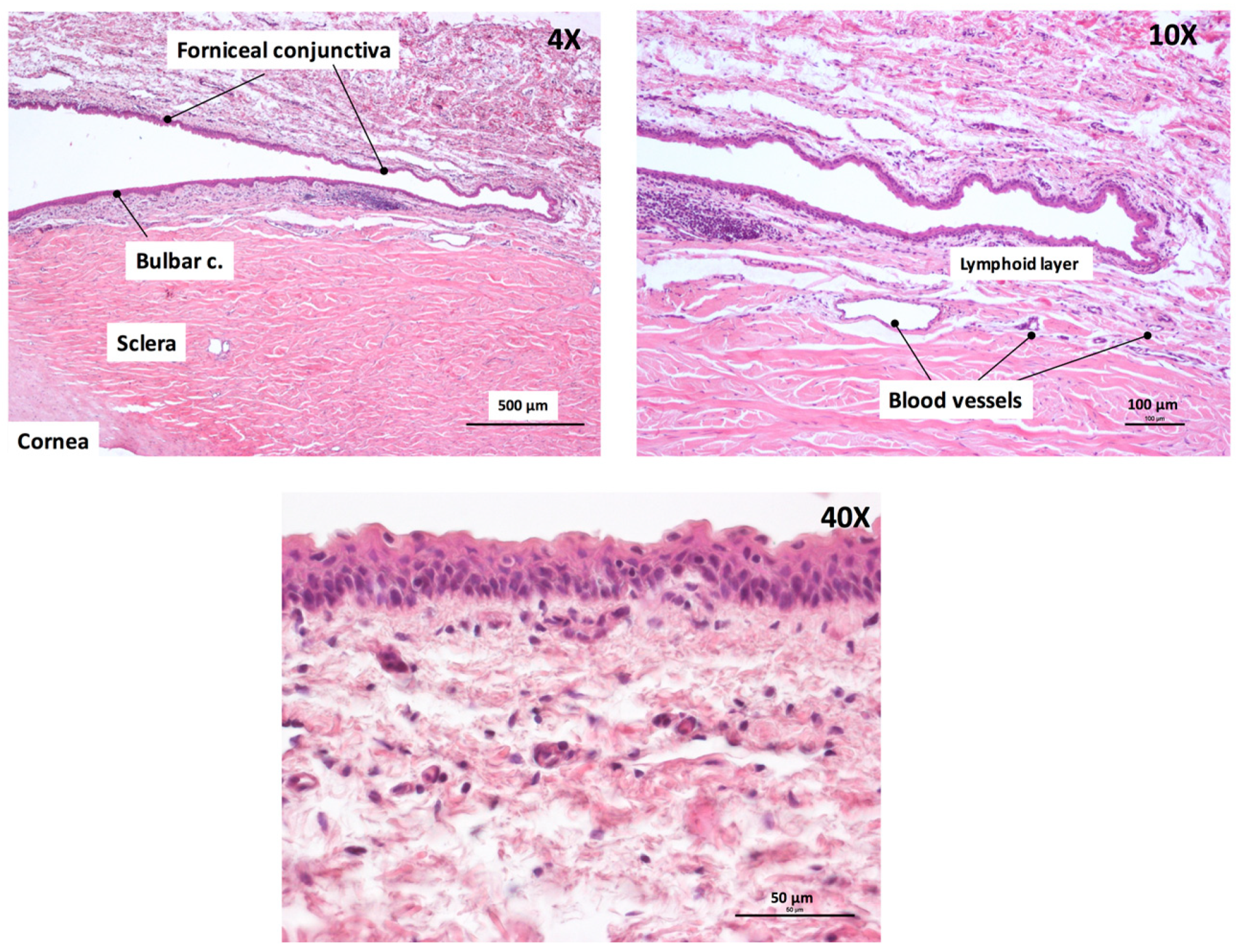
2.7. Conjunctiva Histology
2.8. Dexamethasone Release from Micelles
2.9. HPLC-UV Methods
2.10. Statistical Analysis
3. Results and Discussion
3.1. Tissue Characterization
3.2. Characterization of Blank Micelles
3.2.1. Light Scattering: Size and Zeta Potential
3.2.2. X-ray Scattering
3.3. Cyclosporine
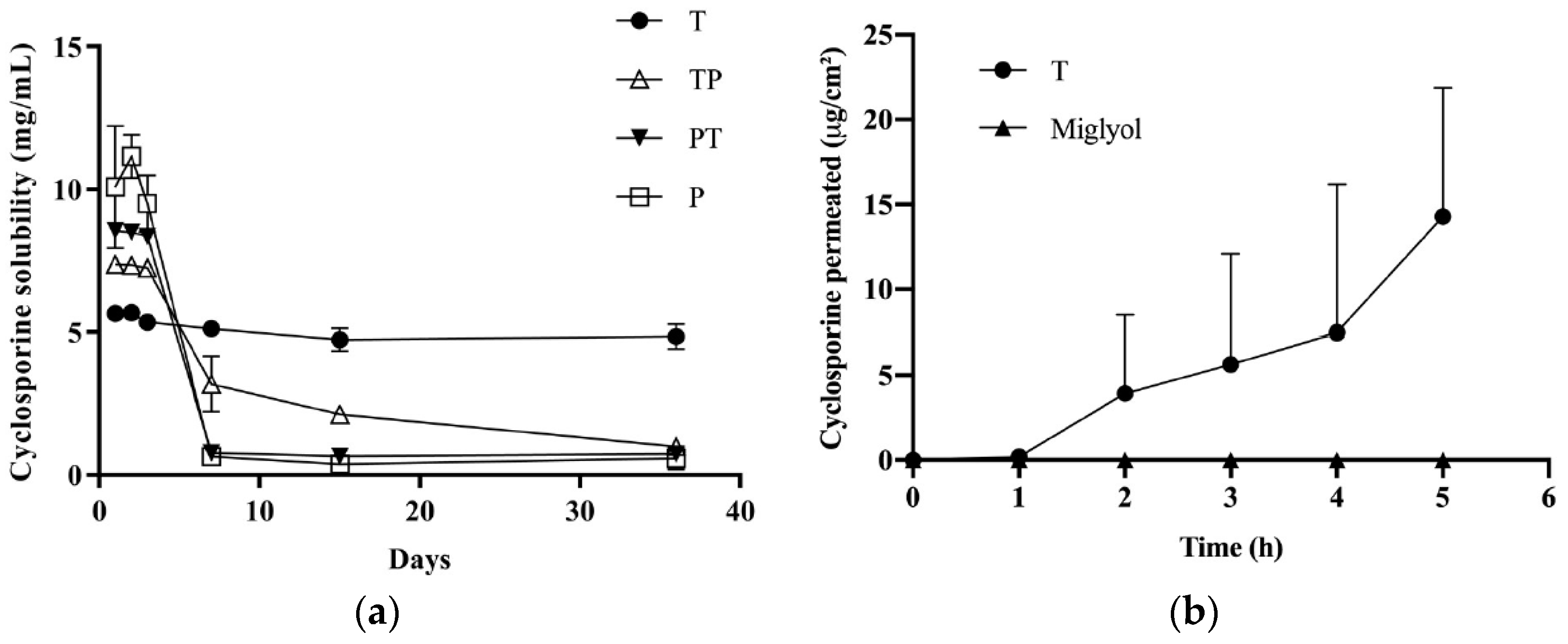
3.3.1. Cyclosporine Solubility
3.3.2. Cyclosporine Permeation across the Conjunctiva
3.4. Econazole Nitrate
3.4.1. Econazole Nitrate Solubility
3.4.2. Econazole Nitrate Retention in the Conjunctiva
3.5. Dexamethasone
3.5.1. Dexamethasone Solubility
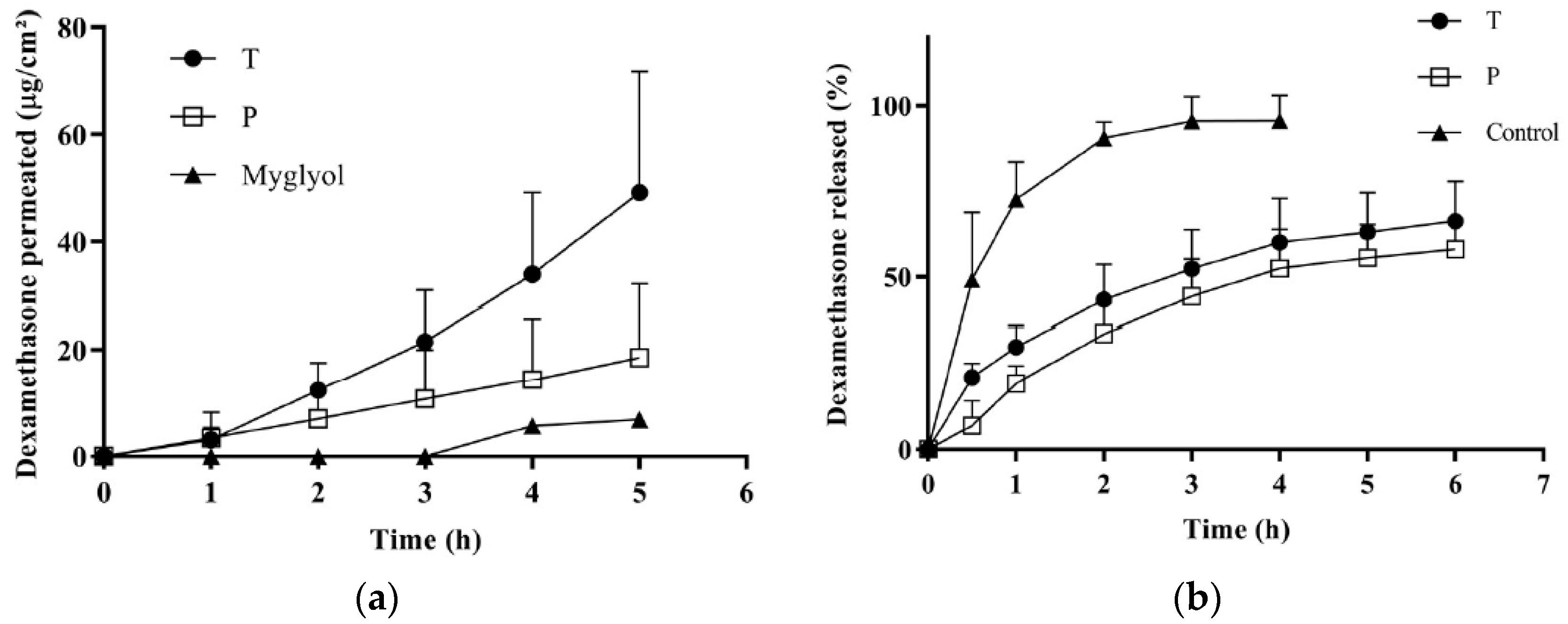
3.5.2. Dexamethasone Permeation across the Conjunctiva
3.5.3. Dexamethasone Release from Micelles
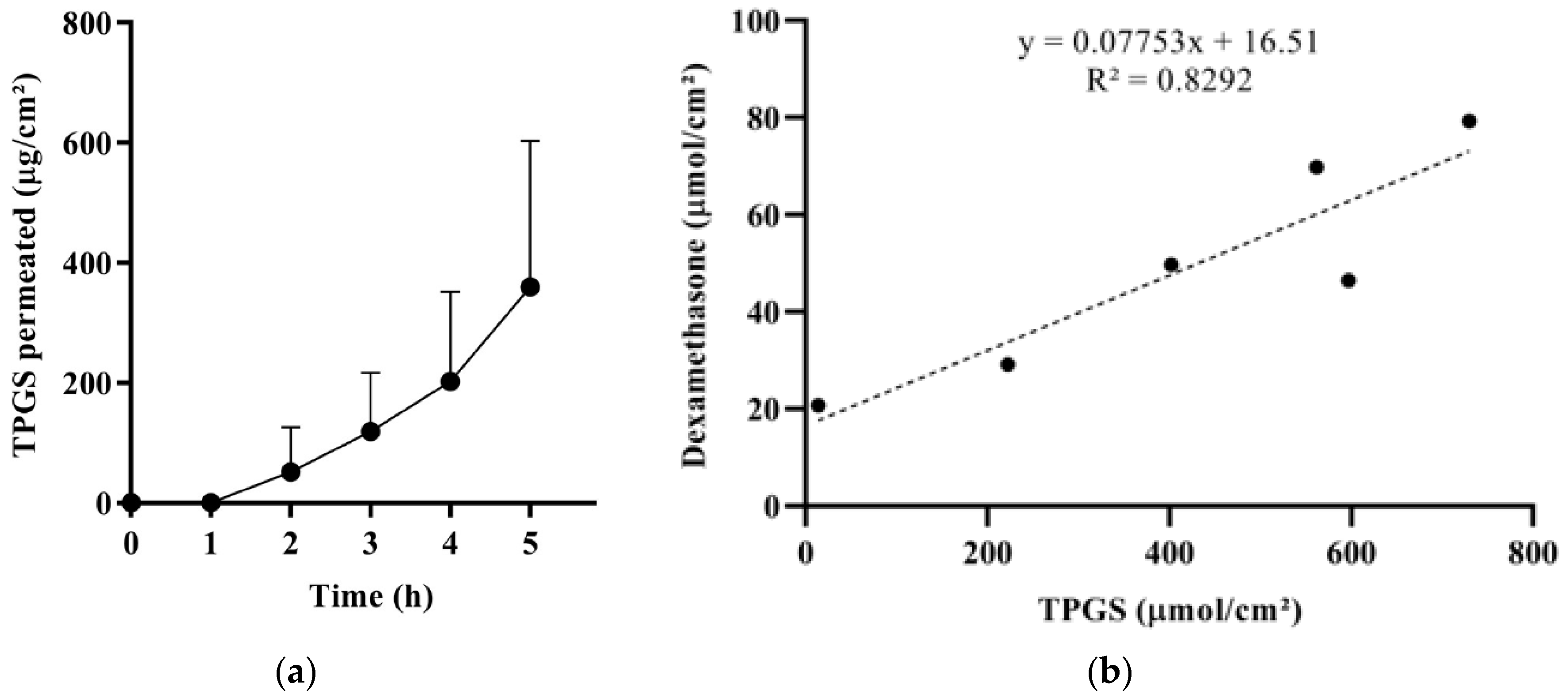
3.5.4. TPGS Transconjunctival Permeation and Mechanism of Enhancement
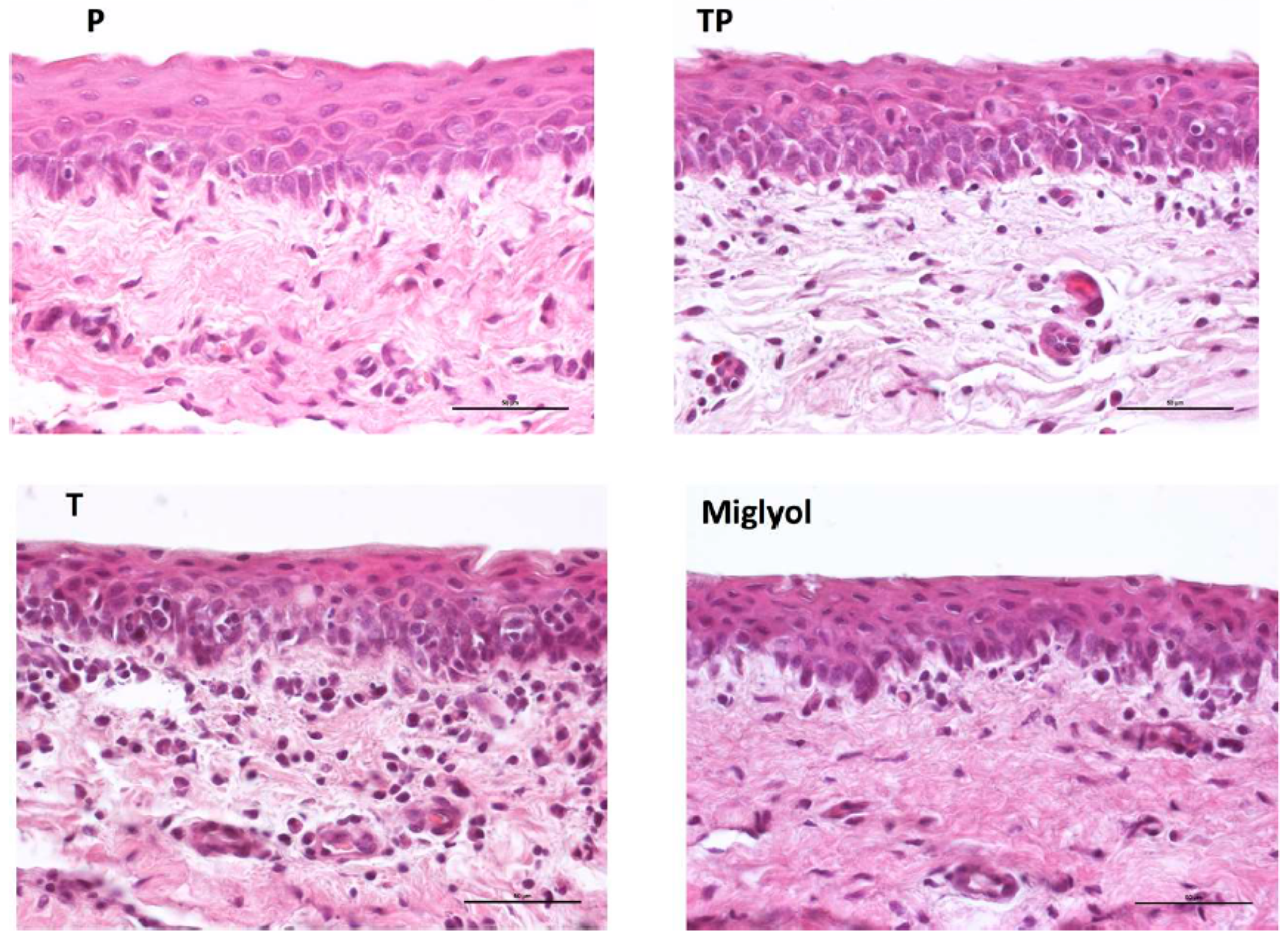
3.6. Effect of Micelle Application on the Conjunctival Tissue
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
References
- Hamalainen, K.M.; Kananen, K.; Auriola, S.; Kontturi, K.; Urtti, A. Characterization of paracellular and aqueous penetration routes in cornea, conjunctiva, and sclera. Investig. Ophthalmol. Vis. Sci. 1997, 38, 627–634. [Google Scholar]
- Grimaudo, M.A.; Pescina, S.; Padula, C.; Santi, P.; Concheiro, A.; Alvarez-Lorenzo, C.; Nicoli, S. Topical application of polymeric nanomicelles in ophthalmology: A review on research efforts for the noninvasive delivery of ocular therapeutics. Expert Opin. Drug Deliv. 2019, 16, 397–413. [Google Scholar] [CrossRef] [PubMed]
- Deshmukh, A.S.; Chauhan, P.N.; Noolvi, M.N.; Chaturvedi, K.; Ganguly, K.; Shukla, S.S.; Nadagouda, M.N.; Aminabhavi, T.M. Polymeric micelles: Basic research to clinical practice. Int. J. Pharm. 2017, 532, 249–268. [Google Scholar] [CrossRef] [PubMed]
- Varela-Garcia, A.; Concheiro, A.; Alvarez-Lorenzo, C. Soluplus micelles for acyclovir ocular delivery: Formulation and cornea and sclera permeability. Int. J. Pharm. 2018, 552, 39–47. [Google Scholar] [CrossRef] [PubMed]
- Noh, G.; Keum, T.; Seo, J.-E.; Choi, J.; Rakesh, B.; Shrawani, L.; Park, B.; Choi, Y.W.; Lee, S. Development and Evaluation of a Water Soluble Fluorometholone Eye Drop Formulation Employing Polymeric Micelle. Pharmaceutics 2018, 10, 208. [Google Scholar] [CrossRef] [PubMed]
- Vaishya, R.D.; Gokulgandhi, M.; Patel, S.; Minocha, M.; Mitra, A.K. Novel dexamethasone-loaded nanomicelles for the intermediate and posterior segment uveitis. AAPS PharmSciTech 2014, 15, 1238–1251. [Google Scholar] [CrossRef] [PubMed]
- Grimaudo, M.A.; Pescina, S.; Padula, C.; Santi, P.; Concheiro, A.; Alvarez-Lorenzo, C.; Nicoli, S. Poloxamer 407/TPGS Mixed Micelles as Promising Carriers for Cyclosporine Ocular Delivery. Mol. Pharm. 2018, 15, 571–584. [Google Scholar] [CrossRef]
- Civiale, C.; Licciardi, M.; Cavallaro, G.; Giammona, G.; Mazzone, M.G. Polyhydroxyethylaspartamide-based micelles for ocular drug delivery. Int. J. Pharm. 2009, 378, 177–186. [Google Scholar] [CrossRef]
- Liaw, J.; Chang, S.F.; Hsiao, F.C. In vivo gene delivery into ocular tissues by eye drops of poly(ethylene oxide)-poly(propylene oxide)-poly(ethylene oxide) (PEO-PPO-PEO) polymeric micelles. Gene Ther. 2001, 8, 999–1004. [Google Scholar] [CrossRef][Green Version]
- Elmowafy, E.; Gad, H.; Biondo, F.; Casettari, L.; Soliman, M.E. Exploring optimized methoxy poly(ethylene glycol)-block-poly(ε-caprolactone) crystalline cored micelles in anti-glaucoma pharmacotherapy. Int. J. Pharm. 2019, 566, 573–584. [Google Scholar] [CrossRef]
- Hou, Y.; Zhang, F.; Lan, J.; Sun, F.; Li, J.; Li, M.; Song, K.; Wu, X. Ultra-small micelles based on polyoxyl 15 hydroxystearate for ocular delivery of myricetin: Optimization, in vitro, and in vivo evaluation. Drug Deliv. 2019, 26, 158–167. [Google Scholar] [CrossRef] [PubMed]
- Sun, F.; Zheng, Z.; Lan, J.; Li, X.; Li, M.; Song, K.; Wu, X. New micelle myricetin formulation for ocular delivery: Improved stability, solubility, and ocular anti-inflammatory treatment. Drug Deliv. 2019, 26, 575–585. [Google Scholar] [CrossRef] [PubMed]
- Lorenzo-Veiga, B.; Sigurdsson, H.H.; Loftsson, T.; Alvarez-Lorenzo, C. Cyclodextrin–Amphiphilic Copolymer Supramolecular Assemblies for the Ocular Delivery of Natamycin. Nanomaterials 2019, 9, 745. [Google Scholar] [CrossRef] [PubMed]
- Bongiovì, F.; Di Prima, G.; Palumbo, F.S.; Licciardi, M.; Pitarresi, G.; Giammona, G. Hyaluronic Acid-Based Micelles as Ocular Platform to Modulate the Loading, Release, and Corneal Permeation of Corticosteroids. Macromol. Biosci. 2017, 17. [Google Scholar] [CrossRef] [PubMed]
- Trinh, H.M.; Cholkar, K.; Joseph, M.; Yang, X.; Mitra, A.K. Clear, Aqueous Topical Drop of Triamcinolone Acetonide. AAPS PharmSciTech 2017, 18, 2466–2478. [Google Scholar] [CrossRef] [PubMed]
- Kang, H.; Cha, K.-H.; Cho, W.; Park, J.; Park, H.J.; Sun, B.K.; Hyun, S.-M.; Hwang, S.-J. Cyclosporine Amicellar delivery system for dry eyes. Int. J. Nanomed. 2016, 11, 2921–2933. [Google Scholar]
- Harvey, T.M.; Fernandez, A.; Patel, R.; Goldman, D.; Ciralsky, J. Conjunctival anatomy and physiology. In Ocular Surface Disease: Cornea, Conjunctiva and Tear Film; Holland, E.J., Mannis, M.J., Lee, W.B., Eds.; Elsevier Saunders: Philadelphia, PA, USA, 2013; pp. 23–27. [Google Scholar]
- Ramsay, E.; Del Amo, E.M.; Toropainen, E.; Tengvall-Unadike, U.; Ranta, V.-P.; Urtti, A.; Ruponen, M. Corneal and conjunctival drug permeability: Systematic comparison and pharmacokinetic impact in the eye. Eur. J. Pharm. Sci. 2018, 119, 83–89. [Google Scholar] [CrossRef] [PubMed]
- Ranta, V.P.; Urtti, A. Transscleral drug delivery to the posterior eye: Prospects of pharmacokinetic modeling. Adv. Drug Deliv. Rev. 2006, 58, 1164–1181. [Google Scholar] [CrossRef] [PubMed]
- Hosoya, K.; Lee, V.H.; Kim, K.J. Roles of the conjunctiva in ocular drug delivery: A review of conjunctival transport mechanisms and their regulation. Eur. J. Pharm. Biopharm. 2005, 60, 227–240. [Google Scholar] [CrossRef]
- Vaajanen, A.; Vapaatalo, H. A Single Drop in the Eye - Effects on the Whole Body? Open Ophthalmol. J. 2017, 11, 305–314. [Google Scholar] [CrossRef]
- Urtti, A.; Salminen, L. Minimizing systemic absorption of topically administered ophthalmic drugs. Surv. Ophthalmol. 1993, 37, 435–456. [Google Scholar] [CrossRef]
- Lallemand, F.; Felt-Baeyens, O.; Besseghir, K.; Behar-Cohen, F.; Gurny, R. Cyclosporine A delivery to the eye: A pharmaceutical challenge. Eur. J. Pharm. Biopharm. 2003, 56, 307–318. [Google Scholar] [CrossRef]
- Cholkar, K.; Gilger, B.C.; Mitra, A.K. Topical, Aqueous, Clear Cyclosporine Formulation Design for Anterior and Posterior Ocular Delivery. Transl. Vis. Sci. Technol. 2015, 4, 1. [Google Scholar] [CrossRef] [PubMed]
- Prajna, N.V.; John, R.K.; Nirmalan, P.K.; Lalitha, P.; Srinivasan, M. A randomised clinical trial comparing 2% econazole and 5% natamycin for the treatment of fungal keratitis. Br. J. Ophthalmol. 2003, 87, 1235–1237. [Google Scholar] [CrossRef] [PubMed]
- Lakhani, P.; Patil, A.; Majumdar, S. Challenges in the Polyene- and Azole-Based Pharmacotherapy of Ocular Fungal Infections. J. Ocul. Pharmacol. Ther. 2019, 35, 6–22. [Google Scholar] [CrossRef] [PubMed]
- Díaz-Tomé, V.; Luaces-Rodríguez, A.; Silva-Rodríguez, J.; Blanco-Dorado, S.; García-Quintanilla, L.; Llovo-Taboada, J.; Blanco-Méndez, J.; García-Otero, X.; Varela-Fernández, R.; Herranz, M.; et al. Ophthalmic Econazole Hydrogels for the Treatment of Fungal Keratitis. J. Pharm. Sci. 2018, 107, 1342–1351. [Google Scholar] [CrossRef] [PubMed]
- Zeng, S.; Wen, K.-K.; Workalemahu, G.; Sohn, E.H.; Wu, M.; Chirco, K.R.; Flamme-Wiese, M.J.; Liu, X.; Stone, E.M.; Tucker, B.A. Imidazole Compounds for Protecting Choroidal Endothelial Cells from Complement Injury. Sci. Rep. 2018, 8, 13387. [Google Scholar] [CrossRef] [PubMed]
- Pescina, S.; Macaluso, C.; Gioia, G.A.; Padula, C.; Santi, P.; Nicoli, S. Mydriatics release from solid and semi-solid ophthalmic formulations using different in vitro methods. Drug Dev. Ind. Pharm. 2017, 43, 1472–1479. [Google Scholar] [CrossRef]
- Advanced Chemistry Development, Inc. ACD/i-lab, Advanced Chemistry Development 2019. Available online: https://ilab.acdlabs.com/iLab2/ (accessed on 12 April 2019).
- Doucet, M.; Cho, J.H.; Alina, G.; Bakker, J.; Bouwman, W.; Butler, P.; Campbell, K.; Gonzales, M.; Heenan, G.; Jackson, A.; et al. SasView Version 4.2.1; Zenodo, 10 February 2019. [Google Scholar] [CrossRef]
- Wang, W.; Sasaki, H.; Chien, D.S.; Lee, V.H. Lipophilicity influence on conjunctival drug penetration in the pigmented rabbit: A comparison with corneal penetration. Curr. Eye Res. 1991, 10, 571–579. [Google Scholar] [CrossRef]
- Ahmed, I.; Gokhale, R.D.; Shah, M.V.; Patton, T.F. Physicochemical determinants of drug diffusion across the conjunctiva, sclera, and cornea. J. Pharm. Sci 1987, 76, 583–586. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, H.; Ichikawa, M.; Yamamura, K.; Nishida, K.; Nakamura, J. Ocular membrane permeability of hydrophilic drugs for ocular peptide delivery. J. Pharm. Pharmacol. 1997, 49, 135–139. [Google Scholar] [CrossRef] [PubMed]
- Horibe, Y.; Hosoya, K.; Kim, K.J.; Ogiso, T.; Lee, V.H. Polar solute transport across the pigmented rabbit conjunctiva: Size dependence and the influence of 8-bromo cyclic adenosine monophosphate. Pharm. Res. 1997, 14, 1246–1251. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, H.; Yamamura, K.; Mukai, T.; Nishida, K.; Nakamura, J.; Nakashima, M.; Ichikawa, M. Modification of ocular permeability of peptide drugs by absorption promoters. Biol. Pharm. Bull. 2000, 23, 1524–1527. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Sasaki, H.; Yamamura, K.; Tei, C.; Nishida, K.; Nakamura, J. Ocular permeability of FITC-dextran with absorption promoter for ocular delivery of peptide drug. J. Drug Target 1995, 3, 129–135. [Google Scholar] [CrossRef] [PubMed]
- Kompella, U.B.; Sundaram, S.; Raghava, S.; Escobar, E.R. Luteinizing hormone-releasing hormone agonist and transferrin functionalizations enhance nanoparticle delivery in a novel bovine ex vivo eye model. Mol. Vis. 2006, 12, 1185–1198. [Google Scholar]
- Ramsay, E.; Ruponen, M.; Picardat, T.; Tengvall, U.; Tuomainen, M.; Auriola, S.; Toropainen, E.; Urtti, A.; Del Amo, E.M. Impact of Chemical Structure on Conjunctival Drug Permeability: Adopting Porcine Conjunctiva and Cassette Dosing for Construction of In Silico Model. J. Pharm. Sci. 2017, 106, 2463–2471. [Google Scholar] [CrossRef]
- Gukasyan, H.; Kim, K.L.; Lee, V. The Conjunctival Barrier in Ocular Drug Delivery. In Drug Absorption Studies In Situ, In Vitro and In Silico Models; Ehrhardt, C., Kim, K.J., Eds.; Springer: Boston, MA, USA, 2008. [Google Scholar]
- Pepić, I.; Lovrić, J.; Filipović-Grčić, J. How do polymeric micelles cross epithelial barriers? Eur. J. Pharm. Sci. 2013, 50, 42–55. [Google Scholar] [CrossRef]
- Juretić, M.; Cetina-Čižmek, B.; Filipović-Grčić, J.; Hafner, A.; Lovrić, J.; Pepić, I. Biopharmaceutical evaluation of surface active ophthalmic excipients using in vitro and ex vivo corneal models. Eur. J. Pharm. Sci. 2018, 120, 133–141. [Google Scholar] [CrossRef]
- Huang, A.J.; Tseng, S.C.; Kenyon, K.R. Paracellular permeability of corneal and conjunctival epithelia. Investig. Ophthalmol. Vis. Sci. 1989, 30, 684–689. [Google Scholar]
- Tirnaksiz, F.; Robinson, J.R. Rheological, mucoadhesive and release properties of pluronic F-127 gel and pluronic F-127/polycarbophil mixed gel systems. Pharmazie 2005, 60, 518–523. [Google Scholar] [PubMed]
- Puig-Rigall, J.; Grillo, I.; Dreiss, C.A.; González-Gaitano, G. Structural and Spectroscopic Characterization of TPGS Micelles: Disruptive Role of Cyclodextrins and Kinetic Pathways. Langmuir 2017, 33, 4737–4747. [Google Scholar] [CrossRef] [PubMed]
- Dumée, L.F.; Lemoine, J.-B.; Ancel, A.; Hameed, N.; He, L.; Kong, L. Control of Partial Coalescence of Self-Assembled Metal Nano-Particles across Lyotropic Liquid Crystals Templates towards Long Range Meso-Porous Metal Frameworks Design. Nanomaterials 2015, 5, 1766–1781. [Google Scholar] [CrossRef] [PubMed]
- Mortensen, K.; Batsberg, W.; Hvidt, S. Effects of PEO-PPO Diblock Impurities on the Cubic Structure of Aqueous PEO-PPO-PEO Pluronics Micelles: Fcc and bcc Ordered Structures in F127. Macromolecules 2018, 41, 1720–1727. [Google Scholar] [CrossRef]
- Wanka, G.; Hoffmann, H.; Ulbricht, W. Phase Diagrams and Aggregation Behavior of Poly(oxyethylene)-Poly(oxypropylene)-Poly(oxyethylene) Triblock Copolymers in Aqueous Solutions. Macromolecules 1994, 27, 4145–4159. [Google Scholar] [CrossRef]
- Lam, Y.M.; Grigorieff, N.; Goldbeck-Wood, G. Direct visualisation of micelles of Pluronic block copolymers in aqueous solution by cryo-TEM. Phys. Chem. Chem. Phys. PCCP 1999, 1, 3331–3334. [Google Scholar] [CrossRef]
- Ahmed, T.A.; El-Say, K.M.; Ahmed, O.A.; Aljaeid, B.M. Superiority of TPGS-loaded micelles in the brain delivery of vinpocetine via administration of thermosensitive intranasal gel. Int. J. Nanomed. 2019, 14, 5555–5567. [Google Scholar] [CrossRef]
- Li, H.; Yan, L.; Tang, E.K.Y.; Zhang, Z.; Chen, W.; Liu, G.; Mo, J. Synthesis of TPGS/Curcumin Nanoparticles by Thin-Film Hydration and Evaluation of Their Anti-Colon Cancer Efficacy In Vitro and In Vivo. Front. Pharmacol. 2019, 10, 769. [Google Scholar]
- Sun, D.D.; Lee, P.I. Evolution of supersaturation of amorphous pharmaceuticals: Nonlinear rate of supersaturation generation regulated by matrix diffusion. Mol. Pharm. 2015, 12, 1203–1215. [Google Scholar] [CrossRef]
- Warren, D.B.; Benameur, H.; Porter, C.J.H.; Pouton, C.W. Using polymeric precipitation inhibitors to improve the absorption of poorly water-soluble drugs: A mechanistic basis for utility. J. Drug Target 2010, 18, 704–731. [Google Scholar] [CrossRef]
- Singh, R.; Kristensen, S.; Tønnesen, H.H. Influence of cosolvents, ionic strength and the method of sample preparation on the solubilization of curcumin by Pluronics and HP-gamma-cyclodextrin. Studies of curcumin and curcuminoids, XLIV. Pharmazie 2012, 67, 131–142. [Google Scholar] [PubMed]
- Overhoff, K.A.; McConville, J.T.; Yang, W.; Johnston, K.P.; Peters, J.I.; Williams, R.O. Effect of stabilizer on the maximum degree and extent of supersaturation and oral absorption of tacrolimus made by ultra-rapid freezing. Pharm. Res. 2008, 25, 167–175. [Google Scholar] [CrossRef] [PubMed]
- Guzmán, H.R.; Tawa, M.; Zhang, Z.; Ratanabanangkoon, P.; Shaw, P.; Gardner, C.R.; Chen, H.; Moreau, J.-P.; Almarsson, O.; Remenar, J.F. Combined use of crystalline salt forms and precipitation inhibitors to improve oral absorption of celecoxib from solid oral formulations. J. Pharm. Sci. 2007, 96, 2686–2702. [Google Scholar] [CrossRef] [PubMed]
- Saha, P.; Yang, J.J.; Lee, V.H. Existence of a p-glycoprotein drug efflux pump in cultured rabbit conjunctival epithelial cells. Investig. Ophthalmol. Vis. Sci. 1998, 39, 1221–1226. [Google Scholar]
- Vellonen, K.-S.; Hellinen, L.; Mannermaa, E.; Ruponen, M.; Urtti, A.; Kidron, H. Expression, activity and pharmacokinetic impact of ocular transporters. Adv. Drug Deliv. Rev. 2018, 126, 3–22. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.J.; Kim, K.J.; Lee, V.H. Role of P-glycoprotein in restricting propranolol transport in cultured rabbit conjunctival epithelial cell layers. Pharm. Res. 2000, 17, 533–538. [Google Scholar] [CrossRef]
- Ostacolo, C.; Caruso, C.; Tronino, D.; Troisi, S.; Laneri, S.; Pacente, L.; Del Prete, A.; Sacchi, A. Enhancement of corneal permeation of riboflavin-5’-phosphate through vitamin E TPGS: A promising approach in corneal trans-epithelial cross linking treatment. Int. J. Pharm. 2013, 440, 148–153. [Google Scholar] [CrossRef]
- Moiseev, R.V.; Morrison, P.; Steele, F.; Khutoryanskiy, V.V. Penetration Enhancers in Ocular Drug Delivery. Pharmaceutics 2019, 11, 321. [Google Scholar] [CrossRef]
- Collnot, E.-M.; Baldes, C.; Wempe, M.F.; Kappl, R.; Hüttermann, J.; Hyatt, J.A.; Edgar, K.J.; Schaefer, U.F.; Lehr, C.-M. Mechanism of inhibition of P-glycoprotein mediated efflux by vitamin E TPGS: Influence on ATPase activity and membrane fluidity. Mol. Pharm. 2007, 4, 465–474. [Google Scholar] [CrossRef]
- Baranowski, P.; Karolewicz, B.; Gajda, M.; Pluta, J. Ophthalmic drug dosage forms: Characterisation and research methods. Sci. World J. 2014, 2014, 861904. [Google Scholar] [CrossRef]
- Yu, Y.; Chen, D.; Li, Y.; Yang, W.; Tu, J.; Shen, Y. Improving the topical ocular pharmacokinetics of lyophilized cyclosporine A-loaded micelles: Formulation, in vitro and in vivo studies. Drug Deliv. 2018, 25, 888–899. [Google Scholar] [CrossRef] [PubMed]
- Di Prima, G.; Saladino, S.; Bongiovì, F.; Adamo, G.; Ghersi, G.; Pitarresi, G.; Giammona, G. Novel inulin-based mucoadhesive micelles loaded with corticosteroids as potential transcorneal permeation enhancers. Eur. J. Pharm. Biopharm. 2017, 117, 385–399. [Google Scholar] [CrossRef] [PubMed]
- Di Tommaso, C.; Bourges, J.-L.; Valamanesh, F.; Trubitsyn, G.; Torriglia, A.; Jeanny, J.-C.; Behar-Cohen, F.; Gurny, R.; Möller, M. Novel micelle carriers for cyclosporin A topical ocular delivery: In vivo cornea penetration, ocular distribution and efficacy studies. Eur. J. Pharm. Biopharm. 2012, 81, 257–264. [Google Scholar] [CrossRef] [PubMed]
- Caruso, C.; Ostacolo, C.; Epstein, R.L.; Barbaro, G.; Troisi, S.; Capobianco, D. Transepithelial Corneal Cross-Linking With Vitamin E-Enhanced Riboflavin Solution and Abbreviated, Low-Dose UV-A: 24-Month Clinical Outcomes. Cornea 2016, 35, 145–150. [Google Scholar] [CrossRef] [PubMed]
- Fathalla, Z.M.A.; Vangala, A.; Longman, M.; Khaled, K.A.; Hussein, A.K.; El-Garhy, O.H.; Alany, R.G. Poloxamer-based thermoresponsive ketorolac tromethamine in situ gel preparations: Design, characterisation, toxicity and transcorneal permeation studies. Eur. J. Pharm. Biopharm. 2017, 114, 119–134. [Google Scholar] [CrossRef] [PubMed]
| Formulation | TPGS | Poloxamer 407 | Water | ||
|---|---|---|---|---|---|
| mmol | mg | mmol | mg | mL | |
| T | 0.1 | 151.4 | - | - | 5 |
| TP | 0.05 | 75.71 | 0.05 | 632 | 5 |
| PT | 0.025 | 38.26 | 0.075 | 947 | 5 |
| P | - | - | 0.1 | 1264 | 5 |
| Drug | Donor | Receptor Phase | Extraction Mixture | |
|---|---|---|---|---|
| Vehicle | Drug Conc. (mg/mL) | |||
| Cyclosporine | T | ss a | PBS + TPGS 1 mM b | CH3CN: 1% CH3COOH (87:13) |
| Miglyol® (solution) | 5 | PBS + TPGS 1 mM b | ||
| Dexamethasone | T c | ss a | PBS d | CH3CN: water (35:65) |
| P | ss a | PBS d | ||
| Miglyol® (dispersion) | 1.5 | PBS d | ||
| Econazole Nitrate | TP | ss a | PBS + TPGS 0.2 mM e | CH3CN: pH 4 buffer (60:40) |
| P | ss a | PBS + TPGS 0.2 mM e | ||
| T | ss a | PBS + TPGS 0.2 mM e | ||
| T | ss a | PBS + 1% BSA f | ||
| Miglyol® (dispersion) | 3.8 | PBS + 1% BSA f | ||
| Parameter | Cyclosporine | Dexamethasone | Econazole Nitrate | TPGS |
|---|---|---|---|---|
| Column (Waters, Milford, MA, USA) | Nova-Pack C18 (150 × 3.9 mm, 4 µm) | Nova-Pack C18 (150 × 3.9 mm, 4 µm) | XTerra RP18 (4.6 × 100 mm, 3.5 µm) | C18 Simmetry 300 (250 × 4.6 mm, 5 µm) |
| Oven temperature (°C) | 65 | 45 | 37 | 40 |
| Mobile phase | CH3CN: water with TFA 0.1% (65:35 v/v) | CH3CN: water (35:65 v/v) | CH3CN: pH 4 acetate buffer (70:30 v/v) | MeOH: pH 5.8 acetate buffer (97:3 v/v) |
| Flow (mL/min) | 1.6 | 1.0 | 1.0 | 2.0 |
| Absorption wavelength (nm) | 230 | 240 | 223 | 215 |
| Retention time (min) | ~5 | ~3 | ~2.5 | ~4 |
| Blank | pH | Zeta Potential (mV) | Solubility (mg/mL) | Size (nm) | % Intensity | PdI |
| T | 6.00 ± 0.02 a | −8.26 ± 4.03 | - | 13.86 ± 4.30 | 100.0% | 0.132 |
| TP | 6.94 ± 0.01 | −8.09 ± 1.87 | - | 19.93 ± 8.03 | 96.8% | 0.205 |
| P | 7.22 ± 0.01 | −3.32 ± 8.93 | - | See Table S2 | ||
| Cyclosporine | pH | Zeta Potential (mV) | Solubility (mg/mL) ¶ | Size (nm) | % Intensity | PdI |
| T | 6.82 ± 0.07 | n.d. | 4.84 ± 0.45 | 12.96 ± 4.03 | 100.0% | 0.091 |
| TP | 6.88 ± 0.02 | −0.02 ± 0.20 e | 0.99 ± 0.23 | 20.31 ± 9.66 | 100.0% | 0.192 |
| P | 7.04 ± 0.01 | n.d. | 0.57 ± 0.38 | n.d. | ||
| Econazole Nitrate | pH | Zeta Potential (mV) | Solubility (mg/mL) # | Size | % Intensity | PdI |
| T | 3.36 b ± 0.02 | 9.13 ± 1.48 | 3.69 ± 0.20 | 7.21 ± 2.15 | 100.0% | 0.076 |
| T c | 5.1 d ± 0.14 | n.d. | n.d. | 12.02 ± 3.58 | 100.0% | 0.094 |
| TP | 4.23 ± 0.10 | 4.52 ± 1.68 | 4.15 ± 0.17 | 17.85 ± 8.50 | 99.5% | 0.19 |
| P | 4.23 ± 0.10 | −0.71 ± 1.48 | 3.79 ± 0.56 | n.d. | ||
| Dexamethasone | pH | Zeta Potential (mV) | Solubility (mg/mL) # | Size | % Intensity | PdI |
| T | 6.54 ± 0.16 | −11.39 ± 2.94 | 0.59 ± 0.03 | 13.44 ± 4.29 | 97.7% | 0.165 |
| TP | 6.94 ± 0.02 | −10.55 ± 4.69 | 0.53 ± 0.09 | 21.39 ± 11.54 | 99.6% | 0.222 |
| P | 7.10 ± 0.03 | −10.99 ± 3.30 | 1.11 ± 0.10 | n.d. |
| Formulation | Cyclosporine | Dexamethasone | Econazole | |||
|---|---|---|---|---|---|---|
| Conjunctiva Retention (µg/cm2) | P × 10−6 (cm/s) a | Conjunctiva Retention (µg/cm2) | P × 10−6 (cm/s) a | Conjunctiva Retention (µg/cm2) | P × 10−6 (cm/s) a | |
| T | 13.34 ± 15.92 | 0.46 ± 0.26 | 9.68 ± 4.92 | 3.26 ± 1.78 ¶ | 30.88 ± 7.93 #,b | - |
| 30.32 ± 5.23 #,c | - | |||||
| TP | n.d. | n.d. | n.d. | n.d. | 7.95 ± 2.32 | - |
| P | n.d. | n.d. | 3.53 ± 3.71 ¶ | 1.10 ± 0.60 | 5.41 ± 2.85 | - |
| Miglyol® | <LOQ | - | 4.96 ± 0.49 | - | 2.40 ± 0.44 | - |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pescina, S.; Grolli Lucca, L.; Govoni, P.; Padula, C.; Del Favero, E.; Cantù, L.; Santi, P.; Nicoli, S. Ex Vivo Conjunctival Retention and Transconjunctival Transport of Poorly Soluble Drugs Using Polymeric Micelles. Pharmaceutics 2019, 11, 476. https://doi.org/10.3390/pharmaceutics11090476
Pescina S, Grolli Lucca L, Govoni P, Padula C, Del Favero E, Cantù L, Santi P, Nicoli S. Ex Vivo Conjunctival Retention and Transconjunctival Transport of Poorly Soluble Drugs Using Polymeric Micelles. Pharmaceutics. 2019; 11(9):476. https://doi.org/10.3390/pharmaceutics11090476
Chicago/Turabian StylePescina, Silvia, Leticia Grolli Lucca, Paolo Govoni, Cristina Padula, Elena Del Favero, Laura Cantù, Patrizia Santi, and Sara Nicoli. 2019. "Ex Vivo Conjunctival Retention and Transconjunctival Transport of Poorly Soluble Drugs Using Polymeric Micelles" Pharmaceutics 11, no. 9: 476. https://doi.org/10.3390/pharmaceutics11090476
APA StylePescina, S., Grolli Lucca, L., Govoni, P., Padula, C., Del Favero, E., Cantù, L., Santi, P., & Nicoli, S. (2019). Ex Vivo Conjunctival Retention and Transconjunctival Transport of Poorly Soluble Drugs Using Polymeric Micelles. Pharmaceutics, 11(9), 476. https://doi.org/10.3390/pharmaceutics11090476








