Arming Filamentous Bacteriophage, a Nature-Made Nanoparticle, for New Vaccine and Immunotherapeutic Strategies
Abstract
1. Introduction
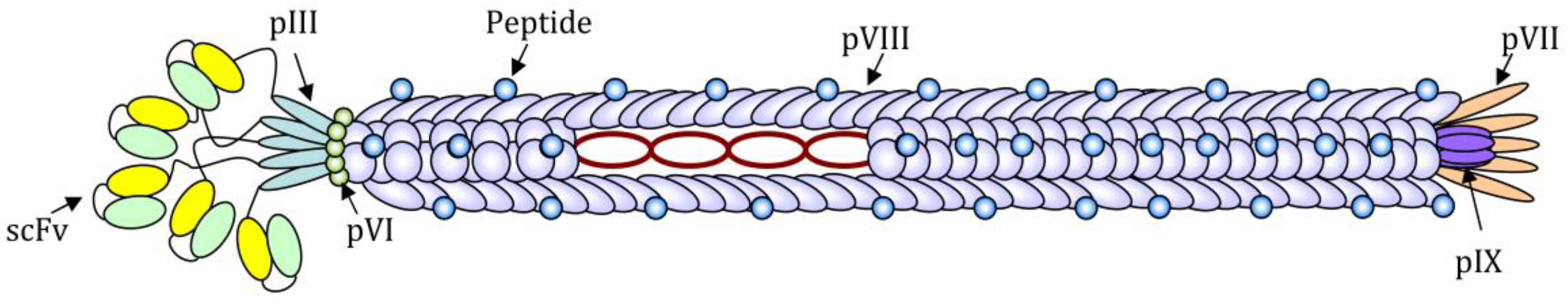
2. The Use of Filamentous Bacteriophages for the Induction and Analysis of Antibody Response
3. Filamentous Bacteriophage Nanocarriers for the Induction of Cellular Immune Responses
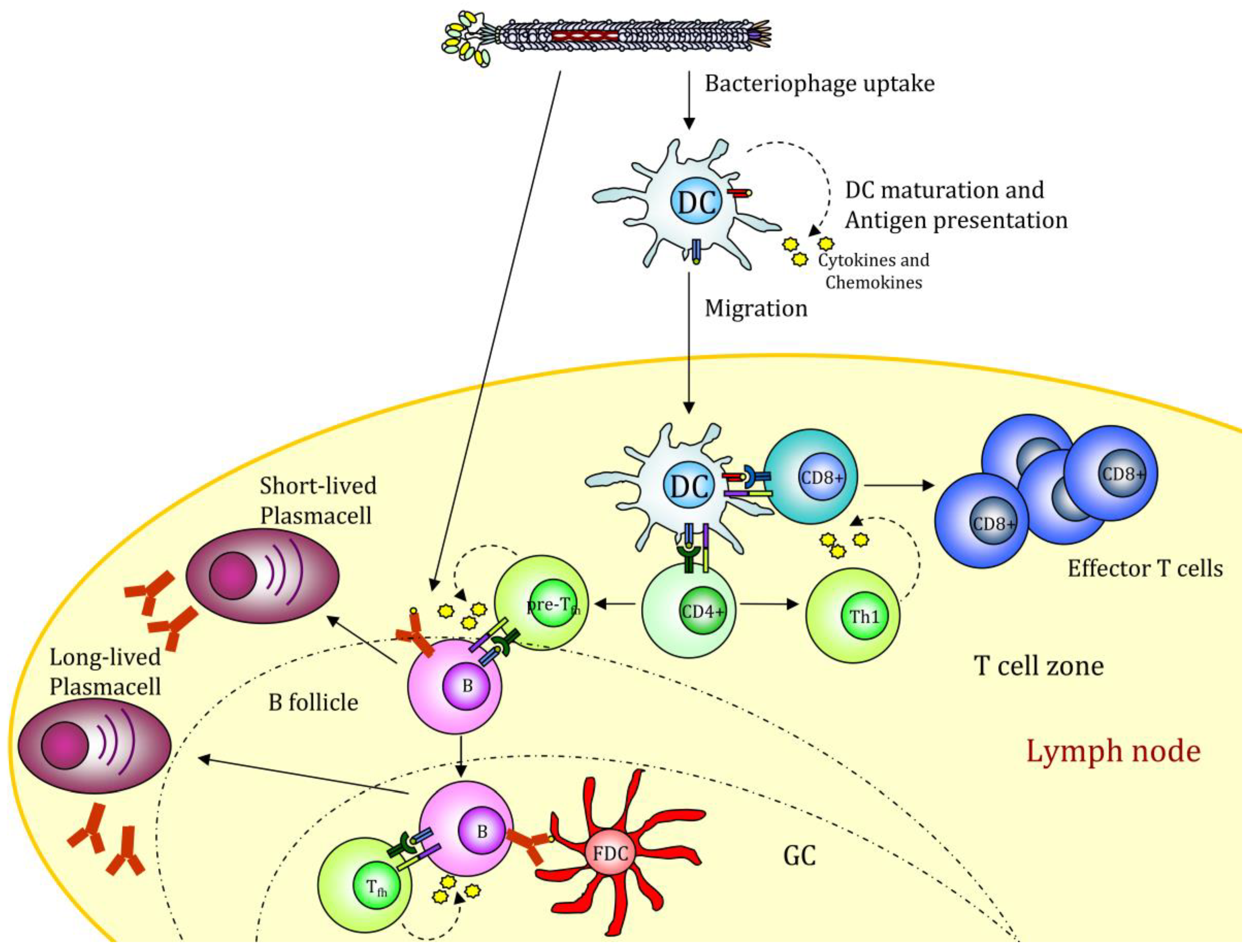
3.1. Use of Filamentous Bacteriophages for Cell Targeting
3.2. Further Improvement by Coupling Immunologically Active Molecules to Filamentous Bacteriophages
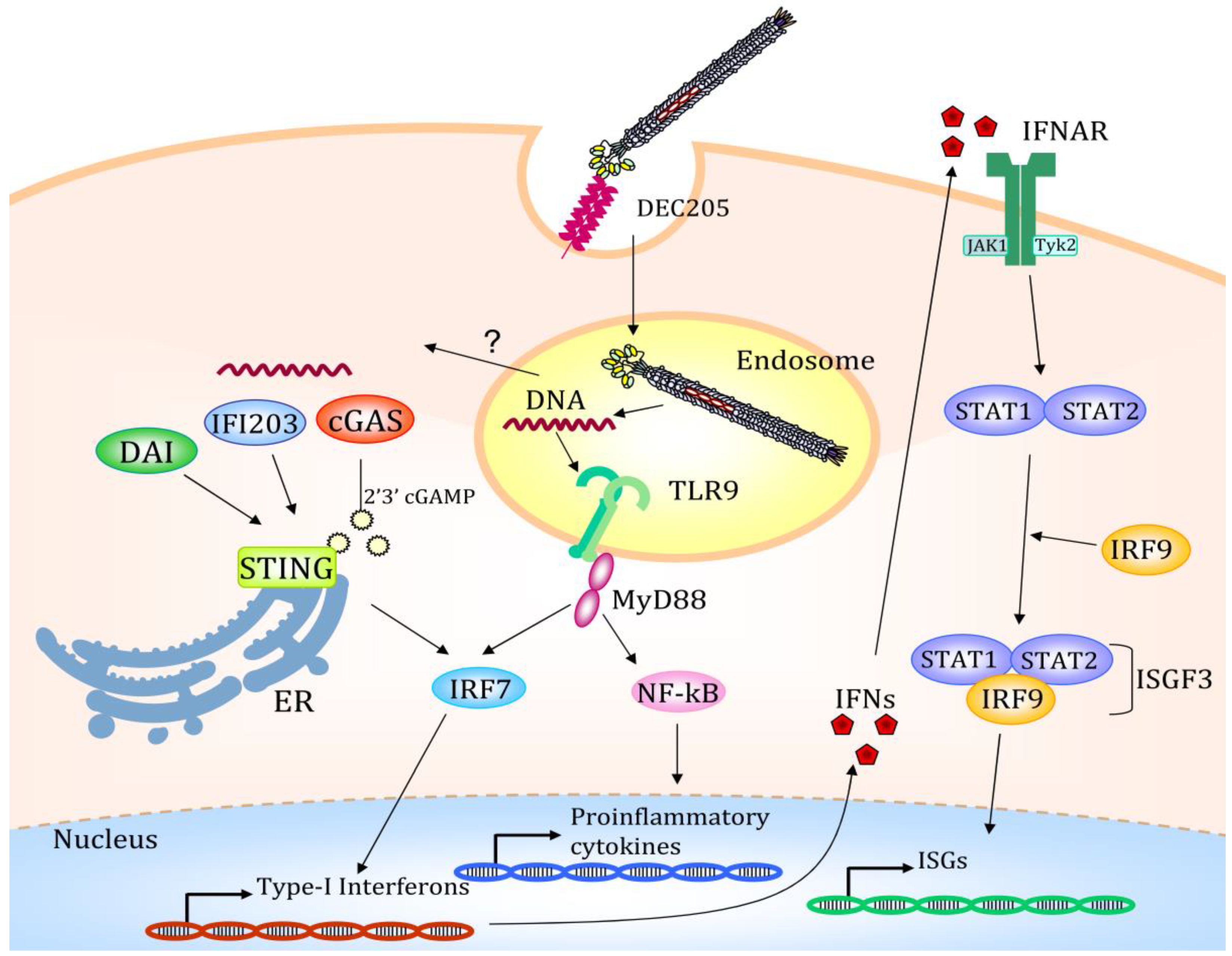
4. Altered Gene Expression after Filamentous Bacteriophage Endocytosis
5. General Considerations about Bacteriophages as Pharmaceutics
Author Contributions
Funding
Conflicts of Interest
References
- Reardon, S. Phage therapy gets revitalized. Nature 2014, 510, 15–16. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, C. Phage therapy’s latest makeover. Nat. Biotechnol. 2019, 37, 581–586. [Google Scholar] [CrossRef] [PubMed]
- Dedrick, R.M.; Guerrero-Bustamante, C.A.; Garlena, R.A.; Russell, D.A.; Ford, K.; Harris, K.; Gilmour, K.C.; Soothill, J.; Jacobs-Sera, D.; Schooley, R.T.; et al. Engineered bacteriophages for treatment of a patient with a disseminated drug-resistant Mycobacterium abscessus. Nat. Med. 2019, 25, 730–733. [Google Scholar] [CrossRef] [PubMed]
- Prisco, A.; De Berardinis, P. Filamentous Bacteriophage Fd as an Antigen Delivery System in Vaccination. Int. J. Mol. Sci. 2012, 13, 5179–5194. [Google Scholar] [CrossRef] [PubMed]
- Hassapis, K.A.; Stylianou, D.C.; Kostrikis, L.G. Architectural Insight into Inovirus-Associated Vectors (IAVs) and Development of IAV-Based Vaccines Inducing Humoral and Cellular Responses: Implications in HIV-1 Vaccines. Viruses 2014, 6, 5047–5076. [Google Scholar] [CrossRef] [PubMed]
- Sartorius, R.; Russo, D.; D’Apice, L.; De Berardinis, P.; Berardinis, P. Filamentous Bacteriophages: An Antigen and Gene Delivery System. In Innovation in Vaccinology; Springer Science and Business Media LLC: Berlin/Heidelberg, Germany, 2012; pp. 123–134. [Google Scholar]
- Henry, K.A.; Arbabi-Ghahroudi, M.; Scott, J.K. Beyond phage display: Non-traditional applications of the filamentous bacteriophage as a vaccine carrier, therapeutic biologic, and bioconjugation scaffold. Front. Microbiol. 2015, 6, 755. [Google Scholar] [CrossRef] [PubMed]
- Aghebati-Maleki, L.; Bakhshinejad, B.; Baradaran, B.; Motallebnezhad, M.; Aghebati-Maleki, A.; Nickho, H.; Yousefi, M.; Majidi, J. Phage display as a promising approach for vaccine development. J. Biomed. Sci. 2016, 23, 66. [Google Scholar] [CrossRef] [PubMed]
- Ju, Z.; Sun, W. Drug delivery vectors based on filamentous bacteriophages and phage-mimetic nanoparticles. Drug Deliv. 2017, 24, 1898–1908. [Google Scholar] [CrossRef]
- Potocnakova, L.; Bhide, M.; Pulzova, L.B. An Introduction to B-Cell Epitope Mapping and In Silico Epitope Prediction. J. Immunol. Res. 2016, 2016, 6760830. [Google Scholar] [CrossRef]
- Wang, L.F.; Yu, M. Epitope Identification and Discovery Using Phage Display Libraries: Applications in Vaccine Development and Diagnostics. Curr. Drug Targets 2004, 5, 1–15. [Google Scholar] [CrossRef]
- Villa-Mancera, A.; Reynoso-Palomar, A.; Utrera-Quintana, F.; Carreon-Luna, L. Cathepsin L1 mimotopes with adjuvant quil a induces a th1/th2 immune response and confers significant protection against fasciola hepatica infection in goats. Parasitol. Res. 2014, 113, 243–250. [Google Scholar] [CrossRef] [PubMed]
- Schiavone, M.; Fiume, G.; Caivano, A.; De Laurentiis, A.; Falcone, C.; Masci, F.F.; Iaccino, E.; Mimmi, S.; Palmieri, C.; Pisano, A.; et al. Design and Characterization of a Peptide Mimotope of the HIV-1 gp120 Bridging Sheet. Int. J. Mol. Sci. 2012, 13, 5674–5699. [Google Scholar] [CrossRef] [PubMed]
- Xin, H.; Glee, P.; Adams, A.; Mohiuddin, F.; Eberle, K. Design of a mimotope-peptide based double epitope vaccine against disseminated candidiasis. Vaccine 2019, 37, 2430–2438. [Google Scholar] [CrossRef] [PubMed]
- Mertens, P.; Walgraffe, D.; Laurent, T.; Deschrevel, N.; Letesson, J.J.; De Bolle, X. Selection of Phage-displayed Peptides Recognised by Monoclonal Antibodies Directed against the Lipopolysaccharide of Brucella. Int. Rev. Immunol. 2001, 20, 181–199. [Google Scholar] [CrossRef] [PubMed]
- Khurana, S.; Suguitan, A.L.; Rivera, Y.; Simmons, C.P.; Lanzavecchia, A.; Sallusto, F.; Manischewitz, J.; King, L.R.; Subbarao, K.; Golding, H. Antigenic Fingerprinting of H5N1 Avian Influenza Using Convalescent Sera and Monoclonal Antibodies Reveals Potential Vaccine and Diagnostic Targets. PLoS Med. 2009, 6, e1000049. [Google Scholar] [CrossRef] [PubMed]
- Khurana, S.; Chearwae, W.; Castellino, F.; Manischewitz, J.; King, L.R.; Honorkiewicz, A.; Rock, M.T.; Edwards, K.M.; Del Giudice, G.; Rappuoli, R.; et al. Vaccines with MF59 Adjuvant Expand the Antibody Repertoire to Target Protective Sites of Pandemic Avian H5N1 Influenza Virus. Sci. Transl. Med. 2010, 2. [Google Scholar] [CrossRef] [PubMed]
- Ravichandran, S.; Hahn, M.; Belaunzarán-Zamudio, P.F.; Ramos-Castañeda, J.; Nájera-Cancino, G.; Caballero-Sosa, S.; Navarro-Fuentes, K.R.; Ruiz-Palacios, G.; Golding, H.; Beigel, J.H.; et al. Differential human antibody repertoires following Zika infection and the implications for serodiagnostics and disease outcome. Nat. Commun. 2019, 10, 1943. [Google Scholar] [CrossRef]
- Luzar, J.; Štrukelj, B.; Lunder, M. Phage display peptide libraries in molecular allergology: From epitope mapping to mimotope based immunotherapy. Allergy 2016, 71, 1526–1532. [Google Scholar] [CrossRef]
- Zahirović, A.; Lunder, M. Microbial Delivery Vehicles for Allergens and Allergen-Derived Peptides in Immunotherapy of Allergic Diseases. Front. Microbiol. 2018, 9, 1449. [Google Scholar] [CrossRef]
- Jensen-Jarolim, E.; Leitner, A.; Kalchhauser, H.; Zürcher, A.; Ganglberger, E.; Bohle, B.; Scheiner, O.; Boltz-Nitulescu, G.; Breiteneder, H. Peptide mimotopes displayed by phage inhibit antibody binding to Bet v 1, the major birch pollen allergen, and induce specific IgG response in mice. FASEB J. 1998, 12, 1635–1642. [Google Scholar] [CrossRef]
- Yang, Y.; Cao, M.J.; Alcocer, M.; Liu, Q.M.; Fei, D.X.; Mao, H.Y.; Liu, G.M. Mapping and characterization of antigenic epitopes of arginine kinase of Scylla paramamosain. Mol. Immunol. 2015, 65, 310–320. [Google Scholar] [CrossRef] [PubMed]
- Tonelli, R.R.; Colli, W.; Alves, M.J. Selection of binding targets in parasites using phage-display and aptamer libraries in vivo and in vitro. Front. Immunol. 2012, 3, 419. [Google Scholar] [CrossRef] [PubMed]
- Shi, H.; Dong, S.; Zhang, X.; Chen, X.; Gao, X.; Wang, L. Phage vaccines displaying YGKDVKDLFDYAQE epitope induce protection against systemic candidiasis in mouse model. Vaccine 2018, 36, 5717–5724. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Sun, M.; Fang, J.; Yang, Q.; Tong, H.; Wang, L. Protective immune responses against systemic candidiasis mediated by phage-displayed specific epitope of Candida albicans heat shock protein 90 in C57BL/6J mice. Vaccine 2006, 24, 6065–6073. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Shi, H.; Dong, S.; Li, Y.; Wang, M.; Huai, Y.; Zhang, X.; Chen, X.; Mao, C.; Gao, X.; et al. Nontoxic engineered virus nanofibers as an efficient agent for the prevention and detection of fungal infection. Nano Res. 2018, 11, 2248–2255. [Google Scholar] [CrossRef]
- Chen, F.; Jiang, R.; Wang, Y.; Zhu, M.; Zhang, X.; Dong, S.; Shi, H.; Wang, L. Recombinant phage elicits protective immune response against systemic s. Globosa infection in mouse model. Sci. Rep. 2017, 7, 42024. [Google Scholar] [CrossRef]
- Mantile, F.; Basile, C.; Cicatiello, V.; De Falco, D.; Caivano, A.; De Berardinis, P.; Prisco, A. A multimeric immunogen for the induction of immune memory to beta-amyloid. Immunol. Cell Biol. 2011, 89, 604–609. [Google Scholar] [CrossRef] [PubMed]
- Trovato, M.; De Berardinis, P. Novel antigen delivery systems. World J. Virol. 2015, 4, 156–168. [Google Scholar] [CrossRef]
- Frenkel, D.; Katz, O.; Solomon, B. Immunization against Alzheimer’s β-amyloid plaques via EFRH phage administration. Proc. Natl. Acad. Sci. USA 2000, 97, 11455–11459. [Google Scholar] [CrossRef]
- Lavie, V.; Becker, M.; Cohen-Kupiec, R.; Yacoby, I.; Koppel, R.; Wedenig, M.; Hutter-Paier, B.; Solomon, B. EFRH–Phage Immunization of Alzheimer’s Disease Animal Model Improves Behavioral Performance in Morris Water Maze Trials. J. Mol. Neurosci. 2004, 24, 105–114. [Google Scholar] [CrossRef]
- Frenkel, D.; Dewachter, I.; Van Leuven, F.; Solomon, B. Reduction of beta-amyloid plaques in brain of transgenic mouse model of Alzheimer’s disease by EFRH-phage immunization. Vaccine 2003, 21, 1060–1065. [Google Scholar] [CrossRef]
- Esposito, M.; Luccarini, I.; Cicatiello, V.; De Falco, D.; Fiorentini, A.; Barba, P.; Casamenti, F.; Prisco, A. Immunogenicity and therapeutic efficacy of phage-displayed beta-amyloid epitopes. Mol. Immunol. 2008, 45, 1056–1062. [Google Scholar] [CrossRef] [PubMed]
- Castiglione, F.; Mantile, F.; De Berardinis, P.; Prisco, A. How the Interval between Prime and Boost Injection Affects the Immune Response in a Computational Model of the Immune System. Comput. Math. Methods Med. 2012, 2012, 842329. [Google Scholar] [CrossRef] [PubMed]
- Mantile, F.; Trovato, M.; Santoni, A.; Barba, P.; Ottonello, S.; De Berardinis, P.; Prisco, A. Alum and Squalene-Oil-in-Water Emulsion Enhance the Titer and Avidity of Anti-Aβ Antibodies Induced by Multimeric Protein Antigen (1–11)E2, Preserving the Igg1-Skewed Isotype Distribution. PLoS ONE 2014, 9, e101474. [Google Scholar] [CrossRef] [PubMed]
- Prisco, A.; De Berardinis, P. Immunogenicity of B and T epitopes displayed on bacteriophage fd. In Immunogenicity; Villanueva, C.J., Ed.; Nova Science Publishers, Inc.: Hauppauge, NY, USA, 2011; pp. 205–216. [Google Scholar]
- Mantile, F.; Capasso, A.; De Berardinis, P.; Prisco, A. Identification of a Consolidation Phase in Immunological Memory. Front. Immunol. 2019, 10, 508. [Google Scholar] [CrossRef] [PubMed]
- Prisco, A.; De Berardinis, P. Memory immune response: A major challenge in vaccination. Biomol. Concepts 2012, 3, 479–486. [Google Scholar] [CrossRef] [PubMed]
- DeBerardinis, P. Recognition of HIV-derived B and T cell epitopes displayed on filamentous phages. Vaccine 1999, 17, 1434–1441. [Google Scholar] [CrossRef]
- D’Apice, L.; Sartorius, R.; Caivano, A.; Mascolo, D.; Del Pozzo, G.; Di Mase, D.S.; Ricca, E.; Pira, G.L.; Manca, F.; Malanga, D.; et al. Comparative analysis of new innovative vaccine formulations based on the use of procaryotic display systems. Vaccine 2007, 25, 1993–2000. [Google Scholar] [CrossRef] [PubMed]
- Ulivieri, C.; Citro, A.; Ivaldi, F.; Mascolo, D.; Ghittoni, R.; Fanigliulo, D.; Manca, F.; Baldari, C.T.; Li Pira, G.; Del Pozzo, G. Antigenic properties of hcmv peptides displayed by filamentous bacteriophages vs. Synthetic peptides. Immunol. Lett. 2008, 119, 62–70. [Google Scholar] [CrossRef] [PubMed]
- Yang, Q.; Wang, L.; Lu, D.N.; Gao, R.J.; Song, J.N.; Hua, P.Y.; Yuan, D.W. Prophylactic vaccination with phage-displayed epitope of C. albicans elicits protective immune responses against systemic candidiasis in C57BL/6 mice. Vaccine 2005, 23, 4088–4096. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Su, Q.; Dong, S.; Shi, H.; Gao, X.; Wang, L. Hybrid phage displaying SLAQVKYTSASSI induces protection againstCandida albicanschallenge in BALB/c mice. Hum. Vaccines Immunother. 2014, 10, 1057–1063. [Google Scholar] [CrossRef] [PubMed]
- De Berardinis, P.; Sartorius, R.; Fanutti, C.; Perham, R.N.; Del Pozzo, G.; Guardiola, J. Phage display of peptide epitopes from HIV-1 elicits strong cytolytic responses. Nat. Biotechnol. 2000, 18, 873–876. [Google Scholar] [CrossRef] [PubMed]
- De Berardinis, P.; Sartorius, R.; Caivano, A.; Mascolo, D.; Domingo, G.; Pozzo, G.; Gaubin, M.; Perham, R.; Piatier-Tonneau, D.; Guardiola, J. Use of Fusion Proteins and Procaryotic Display Systems for Delivery of HIV-1 Antigens: Development of Novel Vaccines for HIV-1 Infection. Curr. HIV Res. 2003, 1, 441–446. [Google Scholar] [CrossRef] [PubMed]
- Wan, Y.; Wu, Y.; Bian, J.; Wang, X.; Zhou, W.; Jia, Z.; Tan, Y.; Zhou, L. Induction of hepatitis B virus-specific cytotoxic T lymphocytes response in vivo by filamentous phage display vaccine. Vaccine 2001, 19, 2918–2923. [Google Scholar] [CrossRef]
- Mascolo, D.; Barba, P.; De Berardinis, P.; Di Rosa, F.; Del Pozzo, G. Phage display of a CTL epitope elicits a long-termin vivocytotoxic response. FEMS Immunol. Med. Microbiol. 2007, 50, 59–66. [Google Scholar] [CrossRef] [PubMed]
- Del Pozzo, G.; Mascolo, D.; Sartorius, R.; Citro, A.; Barba, P.; D’Apice, L.; De Berardinis, P. Triggering DTH and CTL activity by fd filamentous bacteriophages: Role of CD4+ T cells in memory responses. J. Biomed. Biotechnol. 2010, 2010, 894971. [Google Scholar] [CrossRef]
- Gaubin, M.; Fanutti, C.; Mishal, Z.; Durrbach, A.; De Berardinis, P.; Sartorius, R.; Del Pozzo, G.; Guardiola, J.; Perham, R.N.; Piatier-Tonneau, D. Processing of Filamentous Bacteriophage Virions in Antigen-Presenting Cells Targets Both HLA Class I and Class II Peptide Loading Compartments. DNA Cell Biol. 2003, 22, 11–18. [Google Scholar] [CrossRef]
- Wan, Y.; Wu, Y.; Zhou, J.; Zou, L.; Liang, Y.; Zhao, J.; Jia, Z.; Engberg, J.; Bian, J.; Zhou, W. Cross-presentation of phage particle antigen in MHC class II and endoplasmic reticulum marker-positive compartments. Eur. J. Immunol. 2005, 35, 2041–2050. [Google Scholar] [CrossRef]
- Fang, J.; Wang, G.; Yang, Q.; Song, J.; Wang, Y.; Wang, L. The potential of phage display virions expressing malignant tumor specific antigen MAGE-A1 epitope in murine model. Vaccine 2005, 23, 4860–4866. [Google Scholar] [CrossRef]
- Wu, Y.; Wan, Y.; Bian, J.; Zhao, J.; Jia, Z.; Zhou, L.; Zhou, W.; Tan, Y. Phage display particles expressing tumor-specific antigens induce preventive and therapeutic anti-tumor immunity in murine p815 model. Int. J. Cancer 2002, 98, 748–753. [Google Scholar] [CrossRef]
- Sartorius, R.; Pisu, P.; D’Apice, L.; Pizzella, L.; Romano, C.; Cortese, G.; Giorgini, A.; Santoni, A.; Velotti, F.; De Berardinis, P. The use of filamentous bacteriophage fd to deliver MAGE-A10 or MAGE-A3 HLA-A2-restricted peptides and to induce strong antitumor CTL responses. J. Immunol. 2008, 180, 3719–3728. [Google Scholar] [CrossRef] [PubMed]
- Roehnisch, T.; Then, C.; Nagel, W.; Blumenthal, C.; Braciak, T.; Donzeau, M.; Böhm, T.; Flaig, M.; Bourquin, C.; Oduncu, F.S. Phage idiotype vaccination: First phase I/II clinical trial in patients with multiple myeloma. J. Transl. Med. 2014, 12, 119. [Google Scholar] [CrossRef] [PubMed]
- Bartolacci, C.; Andreani, C.; Curcio, C.; Occhipinti, S.; Massaccesi, L.; Giovarelli, M.; Galeazzi, R.; Iezzi, M.; Tilio, M.; Gambini, V.; et al. Phage-Based Anti-HER2 Vaccination Can Circumvent Immune Tolerance against Breast Cancer. Cancer Immunol. Res. 2018, 6, 1486–1498. [Google Scholar] [CrossRef] [PubMed]
- Eriksson, F.; Culp, W.D.; Massey, R.; Egevad, L.; Garland, D.; Persson, M.A.; Pisa, P. Tumor specific phage particles promote tumor regression in a mouse melanoma model. Cancer Immunol. Immunother. 2007, 56, 677–687. [Google Scholar] [CrossRef] [PubMed]
- Murgas, P.; Bustamante, N.; Araya, N.; Cruz-Gomez, S.; Duran, E.; Gaete, D.; Oyarce, C.; Lopez, E.; Herrada, A.A.; Ferreira, N.; et al. A filamentous bacteriophage targeted to carcinoembryonic antigen induces tumor regression in mouse models of colorectal cancer. Cancer Immunol. Immunother. 2018, 67, 183–193. [Google Scholar] [CrossRef] [PubMed]
- Eriksson, F.; Tsagozis, P.; Lundberg, K.; Parsa, R.; Mangsbo, S.M.; Persson, M.A.A.; Harris, R.A.; Pisa, P. Tumor-Specific Bacteriophages Induce Tumor Destruction through Activation of Tumor-Associated Macrophages. J. Immunol. 2009, 182, 3105–3111. [Google Scholar] [CrossRef] [PubMed]
- Hashiguchi, S.; Yamaguchi, Y.; Takeuchi, O.; Akira, S.; Sugimura, K. Immunological basis of M13 phage vaccine: Regulation under MyD88 and TLR9 signaling. Biochem. Biophys. Res. Commun. 2010, 402, 19–22. [Google Scholar] [CrossRef] [PubMed]
- Mori, K.; Kubo, T.; Kibayashi, Y.; Ohkuma, T.; Kaji, A. Anti-vaccinia virus effect of M13 bacteriophage DNA. Antivir. Res. 1996, 31, 79–86. [Google Scholar] [CrossRef]
- Gomes-Neto, J.F.; Sartorius, R.; Canto, F.B.; Almeida, T.S.; Dias, A.A.; Barbosa, C.H.D.; Melo, G.A.; Oliveira, A.C.; Aguiar, P.H.N.; Machado, C.R.; et al. Vaccination With Recombinant Filamentous fd Phages Against Parasite Infection Requires TLR9 Expression. Front. Immunol. 2018, 9, 1173. [Google Scholar] [CrossRef]
- Sartorius, R.; Bettua, C.; D’Apice, L.; Caivano, A.; Trovato, M.; Russo, D.; Zanoni, I.; Granucci, F.; Mascolo, D.; Barba, P.; et al. Vaccination with filamentous bacteriophages targeting DEC-205 induces DC maturation and potent anti-tumor T-cell responses in the absence of adjuvants. Eur. J. Immunol. 2011, 41, 2573–2584. [Google Scholar] [CrossRef]
- Mahnke, K.; Guo, M.; Lee, S.; Sepulveda, H.; Swain, S.L.; Nussenzweig, M.; Steinman, R.M. The Dendritic Cell Receptor for Endocytosis, Dec-205, Can Recycle and Enhance Antigen Presentation via Major Histocompatibility Complex Class II–Positive Lysosomal Compartments. J. Cell Biol. 2000, 151, 673–684. [Google Scholar] [CrossRef] [PubMed]
- Sartorius, R.; D’Apice, L.; Trovato, M.; Cuccaro, F.; Costa, V.; De Leo, M.G.; Marzullo, V.M.; Biondo, C.; D’Auria, S.; De Matteis, M.A.; et al. Antigen delivery by filamentous bacteriophage fd displaying an anti-DEC-205 single-chain variable fragment confers adjuvanticity by triggering a TLR 9-mediated immune response. EMBO Mol. Med. 2015, 7, 973–988. [Google Scholar] [CrossRef] [PubMed]
- Krag, D.N.; Shukla, G.S.; Shen, G.P.; Pero, S.; Ashikaga, T.; Fuller, S.; Weaver, D.L.; Burdette-Radoux, S.; Thomas, C. Selection of Tumor-binding Ligands in Cancer Patients with Phage Display Libraries. Cancer Res. 2006, 66, 7724–7733. [Google Scholar] [CrossRef] [PubMed]
- Shukla, G.S.; Krag, D.N.; Peletskaya, E.N.; Pero, S.C.; Sun, Y.J.; Carman, C.L.; McCahill, L.E.; Roland, T.A. Intravenous Infusion of Phage-displayed Antibody Library in Human Cancer Patients: Enrichment and Cancer-Specificity of Tumor-Homing Phage-Antibodies. Cancer Immunol. Immunother. 2013, 62, 1397–1410. [Google Scholar] [CrossRef] [PubMed]
- Shadidi, M.; Sørensen, D.; Dybwad, A.; Furset, G.; Sioud, M. Mucosal vaccination with phage-displayed tumour antigens identified through proteomics-based strategy inhibits the growth and metastasis of 4T1 breast adenocarcinoma. Int. J. Oncol. 2008, 32, 241–247. [Google Scholar] [CrossRef]
- Ravn, U.; Gueneau, F.; Baerlocher, L.; Osteras, M.; Desmurs, M.; Malinge, P.; Magistrelli, G.; Farinelli, L.; Kosco-Vilbois, M.H.; Fischer, N. By-passing in vitro screening—Next generation sequencing technologies applied to antibody display and in silico candidate selection. Nucleic Acids Res. 2010, 38, e193. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, D.; Kohli, A.G.; Moser, F.; Endy, D.; Belcher, A.M. Refactored M13 Bacteriophage as a Platform for Tumor Cell Imaging and Drug Delivery. ACS Synth. Biol. 2012, 1, 576–582. [Google Scholar] [CrossRef]
- Lee, K.J.; Lee, J.H.; Chung, H.K.; Ju, E.J.; Song, S.Y.; Jeong, S.Y.; Choi, E.K. Application of peptide displaying phage as a novel diagnostic probe for human lung adenocarcinoma. Amino Acids 2016, 48, 1079–1086. [Google Scholar] [CrossRef]
- Namdee, K.; Khongkow, M.; Boonrungsiman, S.; Nittayasut, N.; Asavarut, P.; Temisak, S.; Saengkrit, N.; Puttipipatkhachorn, S.; Hajitou, A.; Ruxrungtham, K.; et al. Thermoresponsive Bacteriophage Nanocarrier as a Gene Delivery Vector Targeted to the Gastrointestinal Tract. Mol. Ther. Nucleic Acids 2018, 12, 33–44. [Google Scholar] [CrossRef]
- Bedi, D.; Gillespie, J.W.; Petrenko, V.A.; Ebner, A.; Leitner, M.; Hinterdorfer, P.; Petrenko, V.A. Targeted Delivery of siRNA into Breast Cancer Cells via Phage Fusion Proteins. Mol. Pharm. 2013, 10, 551–559. [Google Scholar] [CrossRef]
- Marvin, D.A.; Symmons, M.F.; Straus, S.K. Structure and assembly of filamentous bacteriophages. Prog. Biophys. Mol. Biol. 2014, 114, 80–122. [Google Scholar] [CrossRef] [PubMed]
- Stopar, D.; Spruijt, R.B.; Wolfs, C.J.A.M.; Hemminga, M.A. Protein–lipid interactions of bacteriophage m13 major coat protein. Biochim. Biophys. Acta (BBA) Biomembr. 2003, 1611, 5–15. [Google Scholar] [CrossRef]
- Sartorius, R.; D’Apice, L.; Barba, P.; Cipria, D.; Grauso, L.; Cutignano, A.; De Berardinis, P. Vectorized Delivery of Alpha-GalactosylCeramide and Tumor Antigen on Filamentous Bacteriophage fd Induces Protective Immunity by Enhancing Tumor-Specific T Cell Response. Front. Immunol. 2018, 9, 1496. [Google Scholar] [CrossRef] [PubMed]
- Parekh, V.V.; Wilson, M.T.; Olivares-Villagómez, D.; Singh, A.K.; Wu, L.; Wang, C.R.; Joyce, S.; Van Kaer, L. Glycolipid antigen induces long-term natural killer T cell anergy in mice. J. Clin. Investig. 2005, 115, 2572–2583. [Google Scholar] [CrossRef] [PubMed]
- Foca, A.; Liberto, M.C.; Quirino, A.; Marascio, N.; Zicca, E.; Pavia, G. Gut inflammation and immunity: What is the role of the human gut virome? Mediat. Inflamm. 2015, 2015, 326032. [Google Scholar] [CrossRef] [PubMed]
- D’Apice, L.; Costa, V.; Sartorius, R.; Trovato, M.; Aprile, M.; De Berardinis, P. Stimulation of Innate and Adaptive Immunity by Using Filamentous Bacteriophage fd Targeted to DEC-205. J. Immunol. Res. 2015, 2015, 585078. [Google Scholar] [CrossRef] [PubMed]
- Chatterjee, B.; Smed-Sörensen, A.; Cohn, L.; Chalouni, C.; Vandlen, R.; Lee, B.C.; Widger, J.; Keler, T.; Delamarre, L.; Mellman, I. Internalization and endosomal degradation of receptor-bound antigens regulate the efficiency of cross presentation by human dendritic cells. Blood 2012, 120, 2011–2020. [Google Scholar] [CrossRef]
- Foti, M.; Granucci, F.; Pelizzola, M.; Beretta, O.; Ricciardi-Castagnoli, P. Dendritic cells in pathogen recognition and induction of immune responses: A functional genomics approach. J. Leukoc. Biol. 2006, 79, 913–916. [Google Scholar] [CrossRef]
- Macagno, A.; Gilliet, M.; Sallusto, F.; Lanzavecchia, A.; Nestle, F.O.; Groettrup, M. Dendritic cells up-regulate immunoproteasomes and the proteasome regulator PA28 during maturation. Eur. J. Immunol. 1999, 29, 4037–4042. [Google Scholar] [CrossRef]
- López-Albaitero, A.; Mailliard, R.; Hackman, T.; Filho, P.A.A.; Wang, X.; Gooding, W.; Ferrone, S.; Kalinski, P.; Ferris, R.L. Maturation Pathways of Dendritic Cells Determine TAP1 and TAP2 Levels and Cross-presenting Function. J. Immunother. 2009, 32, 465–473. [Google Scholar] [CrossRef]
- Rengachari, S.; Groiss, S.; Devos, J.M.; Caron, E.; Grandvaux, N.; Panne, D. Structural basis of stat2 recognition by irf9 reveals molecular insights into isgf3 function. Proc. Natl. Acad. Sci. USA 2018, 115, E601–E609. [Google Scholar] [CrossRef] [PubMed]
- Kumar, P.; Sweeney, T.R.; Skabkin, M.A.; Skabkina, O.V.; Hellen, C.U.; Pestova, T.V. Inhibition of translation by IFIT family members is determined by their ability to interact selectively with the 5′-terminal regions of cap0-, cap1-and 5′ppp-mrnas. Nucleic Acids Res. 2014, 42, 3228–3245. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.S.; Hwang, Y.S.; Kim, L.K.; Lee, S.; Lee, W.B.; Kim-Ha, J.; Kim, Y.J. OASL1 Traps Viral RNAs in Stress Granules to Promote Antiviral Responses. Mol. Cells 2018, 41, 214–223. [Google Scholar] [PubMed]
- Villarroya-Beltri, C.; Guerra, S.; Sanchez-Madrid, F. Isgylation—A key to lock the cell gates for preventing the spread of threats. J. Cell Sci. 2017, 130, 2961–2969. [Google Scholar] [CrossRef] [PubMed]
- Latz, E. The inflammasomes: Mechanisms of activation and function. Curr. Opin. Immunol. 2010, 22, 28–33. [Google Scholar] [CrossRef] [PubMed]
- O’Neill, L.A.J.; Palsson-McDermott, E.M.; Pålsson-McDermott, E.M. The Warburg effect then and now: From cancer to inflammatory diseases. BioEssays 2013, 35, 965–973. [Google Scholar]
- Krawczyk, C.M.; Holowka, T.; Sun, J.; Blagih, J.; Amiel, E.; DeBerardinis, R.J.; Cross, J.R.; Jung, E.; Thompson, C.B.; Jones, R.G.; et al. Toll-like receptor–induced changes in glycolytic metabolism regulate dendritic cell activation. Blood 2010, 115, 4742–4749. [Google Scholar] [CrossRef]
- Chatterjee, A.; Duerkop, B.A. Beyond Bacteria: Bacteriophage-Eukaryotic Host Interactions Reveal Emerging Paradigms of Health and Disease. Front. Microbiol. 2018, 9, 1394. [Google Scholar] [CrossRef]
- Greber, U.F.; Willetts, M.; Webster, P.; Helenius, A. Stepwise dismantling of adenovirus 2 during entry into cells. Cell 1993, 75, 477–486. [Google Scholar] [CrossRef]
- Pasqualini, R.; Ruoslahti, E. Organ targeting In vivo using phage display peptide libraries. Nature 1996, 380, 364–366. [Google Scholar] [CrossRef]
- Dor-On, E.; Solomon, B. Targeting glioblastoma via intranasal administration of Ff bacteriophages. Front. Microbiol. 2015, 6, 530. [Google Scholar] [CrossRef] [PubMed]
- Delmastro, P. Immunogenicity of filamentous phage displaying peptide mimotopes after oral administration. Vaccine 1997, 15, 1276–1285. [Google Scholar] [CrossRef]
- Dąbrowska, K.; Switala-Jelen, K.; Opolski, A.; Weber-Dabrowska, B.; Górski, A.; Switała-Jelen, K.; Weber-Dabrowska, B. Bacteriophage penetration in vertebrates. J. Appl. Microbiol. 2005, 98, 7–13. [Google Scholar] [CrossRef] [PubMed]
- Zou, J.; Dickerson, M.T.; Owen, N.K.; Landon, L.A.; Deutscher, S.L. Biodistribution of filamentous phage peptide libraries in mice. Mol. Biol. Rep. 2004, 31, 121–129. [Google Scholar] [CrossRef] [PubMed]
- Molenaar, T.J.; Michon, I.; De Haas, S.A.; Van Berkel, T.J.; Kuiper, J.; Biessen, E.A. Uptake and Processing of Modified Bacteriophage M13 in Mice: Implications for Phage Display. Virology 2002, 293, 182–191. [Google Scholar] [CrossRef] [PubMed]
- Frenkel, D.; Solomon, B. Filamentous phage as vector-mediated antibody delivery to the brain. Proc. Natl. Acad. Sci. USA 2002, 99, 5675–5679. [Google Scholar] [CrossRef] [PubMed]
- Solomon, B. Filamentous bacteriophage as a novel therapeutic tool for Alzheimer’s disease treatment. J. Alzheimer’s Dis. 2008, 15, 193–198. [Google Scholar] [CrossRef]
- Rakover, I.S.; Zabavnik, N.; Kopel, R.; Paz-Rozner, M.; Solomon, B. Antigen-specific therapy of EAE via intranasal delivery of filamentous phage displaying a myelin immunodominant epitope. J. Neuroimmunol. 2010, 225, 68–76. [Google Scholar] [CrossRef]
- Schluesener, H.J.; Xianglin, T. Selection of recombinant phages binding to pathological endothelial and tumor cells of rat glioblastoma by in-vivo display. J. Neurol. Sci. 2004, 224, 77–82. [Google Scholar] [CrossRef]
- Ksendzovsky, A.; Walbridge, S.; Saunders, R.C.; Asthagiri, A.R.; Heiss, J.D.; Lonser, R.R. Convection-enhanced delivery of M13 bacteriophage to the brain. J. Neurosurg. 2012, 117, 197–203. [Google Scholar] [CrossRef]
- Yip, Y.L.; Hawkins, N.J.; Smith, G.; Ward, R.L. Biodistribution of filamentous phage-Fab in nude mice. J. Immunol. Methods 1999, 225, 171–178. [Google Scholar] [CrossRef]
- Henry, K.A.; Murira, A.; Van Houten, N.E.; Scott, J.K. Developing strategies to enhance and focus humoral immune responses using filamentous phage as a model antigen. Bioeng. Bugs 2011, 2, 275–283. [Google Scholar] [CrossRef] [PubMed]
- Brigati, J.R.; Petrenko, V.A. Thermostability of landscape phage probes. Anal. Bioanal. Chem. 2005, 382, 1346–1350. [Google Scholar] [CrossRef] [PubMed]
- Olofsson, L.; Ankarloo, J.; Andersson, P.O.; Nicholls, I.A. Filamentous bacteriophage stability in non-aqueous media. Chem. Biol. 2001, 8, 661–671. [Google Scholar] [CrossRef]
- Pershad, K.; Kay, B.K. Generating thermal stable variants of protein domains through phage display. Methods 2013, 60, 38–45. [Google Scholar] [CrossRef] [PubMed]
- Smith, G.P.; Scott, J.K. [15] Libraries of peptides and proteins displayed on filamentous phage. Methods Enzymol. 1993, 217, 228–257. [Google Scholar] [PubMed]
- Zhang, Y.; Peng, X.; Zhang, H.; Watts, A.B.; Ghosh, D. Manufacturing and ambient stability of shelf freeze dried bacteriophage powder formulations. Int. J. Pharm. 2018, 542, 1–7. [Google Scholar] [CrossRef]
- Bicalho, M.; Machado, V.; Nydam, D.; Santos, T.; Bicalho, R. Evaluation of oral administration of bacteriophages to neonatal calves: Phage survival and impact on fecal Escherichia coli. Livest. Sci. 2012, 144, 294–299. [Google Scholar] [CrossRef]
- Nobrega, F.L.; Costa, A.R.; Santos, J.F.; Siliakus, M.F.; Van Lent, J.W.M.; Kengen, S.W.M.; Azeredo, J.; Kluskens, L.D. Genetically manipulated phages with improved pH resistance for oral administration in veterinary medicine. Sci. Rep. 2016, 6, 39235. [Google Scholar] [CrossRef]
- Ma, Y.; Pacan, J.C.; Wang, Q.; Xu, Y.; Huang, X.; Korenevsky, A.; Sabour, P.M. Microencapsulation of Bacteriophage Felix O1 into Chitosan-Alginate Microspheres for Oral Delivery. Appl. Environ. Microbiol. 2008, 74, 4799–4805. [Google Scholar] [CrossRef]
- Vinner, G.K.; Vladisavljevic, G.T.; Clokie, M.R.J.; Malik, D.J. Microencapsulation of Clostridium difficile specific bacteriophages using microfluidic glass capillary devices for colon delivery using pH triggered release. PLoS ONE 2017, 12, e0186239. [Google Scholar] [CrossRef] [PubMed]
- Malik, D.J.; Sokolov, I.J.; Vinner, G.K.; Mancuso, F.; Cinquerrui, S.; Vladisavljevic, G.T.; Clokie, M.R.; Garton, N.J.; Stapley, A.G.; Kirpichnikova, A. Formulation, stabilisation and encapsulation of bacteriophage for phage therapy. Adv. Colloid Interface Sci. 2017, 249, 100–133. [Google Scholar] [CrossRef] [PubMed]
- Dowds, C.M.; Kornell, S.C.; Blumberg, R.S.; Zeissig, S. Lipid antigens in immunity. Biol. Chem. 2014, 395, 61–81. [Google Scholar] [CrossRef] [PubMed]
- Manzo, E.; Gallo, C.; Sartorius, R.; Nuzzo, G.; Sardo, A.; De Berardinis, P.; Fontana, A.; Cutignano, A. Immunostimulatory Phosphatidylmonogalactosyldiacylglycerols (PGDG) from the Marine Diatom Thalassiosira weissflogii: Inspiration for a Novel Synthetic Toll-Like Receptor 4 Agonist. Mar. Drugs 2019, 17, 103. [Google Scholar] [CrossRef] [PubMed]
- Kharkwal, S.S.; Arora, P.; Porcelli, S.A. Glycolipid activators of invariant NKT cells as vaccine adjuvants. Immunogenetics 2016, 68, 597–610. [Google Scholar] [CrossRef] [PubMed]
- Morales, J.; Martínez, J.J.; Manoutcharian, K.; Hernández, M.; Fleury, A.; Gevorkian, G.; Acero, G.; Blancas, A.; Toledo, A.; Cervantes, J.; et al. Inexpensive anti-cysticercosis vaccine: S3Pvac expressed in heat inactivated M13 filamentous phage proves effective against naturally acquired Taenia solium porcine cysticercosis. Vaccine 2008, 26, 2899–2905. [Google Scholar] [CrossRef]
- Samoylova, T.I.; Norris, M.D.; Samoylov, A.M.; Cochran, A.M.; Wolfe, K.G.; Petrenko, V.A.; Cox, N.R. Infective and inactivated filamentous phage as carriers for immunogenic peptides. J. Virol. Methods 2012, 183, 63–68. [Google Scholar] [CrossRef]
- Sattar, S.; Bennett, N.J.; Wen, W.X.; Guthrie, J.M.; Blackwell, L.F.; Conway, J.F.; Rakonjac, J. Ff-nano, short functionalized nanorods derived from Ff (f1, fd, or M13) filamentous bacteriophage. Front. Microbiol. 2015, 6, 316. [Google Scholar] [CrossRef]
| Epitope | Protein | Sequence | Reference |
|---|---|---|---|
| Fba1 | Fructose-bisphosphate aldolase 1 of C. albicans | YGKDVKDLFDYAQE | [24] |
| SE-CA-HSP90 | Heat shock protein 90 of C. albicans | DEPAGE | [25] |
| HSP90386–391 | Heat shock protein 90 of C. albicans | LKVIRK | [42] |
| Sap2386–390 | Secreted aspartyl protease of C. albicans | VKYTS | [26,42] |
| Sap2382–394 | Secreted aspartyl protease of C. albicans | SLAQVKYTSASSI | [43] |
| Gp70 | Glycoprotein Gp70 of S. globosa | KPVGHALLTPLGLDR | [27] |
| Aβ 3–6 | beta-amyloid peptide | EFRH | [31,32] |
| Aβ 2–6 | beta-amyloid peptide | AEFRH | [33] |
| Pep23 | Reverse transcriptase (RTase) of HIV-1 | KDSWTVNDIQKLVGK | [39,40,44,45] |
| RT2 | Reverse transcriptase (RTase) of HIV-1 | ILKEPVHGV | [39,44,45] |
| p128, p30 | Pp65 protein of Human Cytomegalovirus | EFFWDANDIYRIF, PLKMLNIPSINVHHY | [41] |
| S28–39 | Hepatitis B surface antigen (HBsAg) | IPQSLDSWWTSL | [46] |
| OVA257–264 | Chicken egg ovalbumin | SIINFEKL | [47,48,62,64] |
| MAGE-A1161–169 | Melanoma-associated antigen (MAGE)-A1 | EADPTGHSY | [51] |
| MAGE-A3271–279 | Melanoma-associated antigen (MAGE)-A3 | FLWGPRALV | [53] |
| MAGE A10254–262 | Melanoma-associated antigen (MAGE)-A10 | GLYDGMEHL | [53] |
| P1A35–43 | P1A of murine mastocytoma tumor P815 | LPYLGVLVF | [52] |
| PA8 | Amastigote surface protein 2 of T.cruzi | VNHRFTLV | [61] |
| TSKB20 | Trans-sialidase of T.cruzi | ANYKFTLV | [61] |
| Transcription Factors | |
| Stat1 | Signal transducer and activator of transcription 1 STAT family members form homo- or heterodimers when phosphorylated by the receptor-associated kinases in response to cytokines. then they translocate to the cell nucleus acting as transcription activators. |
| Stat2 | Signal transducer and activator of transcription 2 STAT family members forms homo- or heterodimers when phosphorylated by the receptor-associated kinases in response to cytokines. then they translocate to the cell nucleus acting as transcription activators. |
| Irf7 | Interferon regulatory factor 7 Plays a role in the transcriptional activation of virus-inducible cellular genes, including interferon beta chain genes. |
| Irf9 | Interferon regulatory factor 9 Transcription factor. IRF9 associates with the phosphorylated STAT1:STAT2 dimer to form a complex termed ISGF3 transcription factor. ISGF3 binds to the ISRE to activate the transcription of IFN-stimulated genes. |
| Hif1alpha | Hypoxia inducible factor 1, alpha subunit This gene encodes the alpha subunit which, along with the beta subunit, forms a heterodimeric transcription factor that regulates the cellular and developmental response to reduced oxygen tension. |
| Nucleic Acid Binding Proteins | |
| Tmem | Transmembrane protein 173 STING STING induces type I interferon production when cells are infected with intracellular pathogens, such as viruses, mycobacteria and intracellular parasites. |
| Mb21d1 | Cyclic GMP-AMP Synthase Coding for cGAS protein: catalyzes the synthesis of cyclic guanosine monophosphate-adenosine monophosphate [cGAMP] after binding DNA in the cytoplasm. |
| Nod1 | Nucleotide-binding oligomerization domain containing 1 Intracellular sensor system component. |
| Ddx58 | DEAD [Asp-Glu-Ala-Asp] box polypeptide 58 RNA helicase It is involved in viral double-stranded [ds] RNA recognition and the regulation of immune response. |
| Zbp1 | Z-DNA binding protein 1 Involved in the innate immune response. Binds foreign DNA and induces type-I interferon production. |
| Eif2ak2 | Eukaryotic translation initiation factor 2-alpha kinase 2 dsRNA-dependent serine/threonine-protein kinase, induced by interferons. Involved in the innate immune response to viral infections. |
| Antiviral Proteins | |
| Ifit1 | Interferon-induced protein with tetratricopeptide repeats 1 Involved in the cellular response to cytokine, bacteria, and virus |
| Ifit3 | Interferon-induced protein with tetratricopeptide repeats 3 Inhibitor of cellular as well as viral processes |
| Ifi203 | Interferon activated gene 203 Involved in the cellular response to interferon-beta. |
| Ifi205 | Interferon activated gene 205 Involved the cellular response to interferon-beta and bacteria. |
| Oas2 | 2’-5’ oligoadenylate synthetase 2 Induced by interferons. Synthesize 2’,5’-oligoadenylates [2-5As] starting from adenosine triphosphate. |
| Oas3 | 2′-5′ Oligoadenylate Syntase Induced by interferons: catalyzes the 2’, 5’ oligomers of adenosine to bind and activate RNase L. |
| Oas1g | 2’-5’ oligoadenylate synthetase 1G Involved in the defence response to viruses, the negative regulation of viral genome replication, and the regulation of ribonuclease activity. |
| Trex1 | Three prime repair exonuclease 1 Metabolize DNA fragments of retroviral origin, including L1, LTR and SINE elements. |
| TLR Pathway | |
| Tlr9 | Toll-like receptor 9 Binds DNA present in bacteria and viruses, and triggers signaling cascades, leading to a pro-inflammatory cytokine response. |
| Unc93B | Unc-93 homolog B1 TLR signaling regulator, involved in the innate immune response. Defects in the protein predispose to hypersensitivity to infections. |
| MyD88 | Myeloid Differentiation primary response protein Adapter involved in the Toll-like receptor and IL-1 receptor signaling pathway in the innate immune response. |
| Cytokine and Chemokine Ligands | |
| Il1b | IL-1 b Proinflammatory cytokine. |
| Cxcl10 | Chemokine [C-X-C motif] ligand 10 Involved in the defence response to viruses, the negative regulation of cell differentiation, and response to bacteria. |
| Ccl7 | Chemokine [C-C motif] ligand 7 Involved in several processes, including the G protein-coupled receptor signaling pathway, the cellular response to cytokines, and leukocyte chemotaxis. |
| Cxcl12 | Chemokine [C-X-C motif] ligand 12 Ligand for the G-protein coupled chemokine [C-X-C motif] receptor 4. |
| Proteasome activator | |
| Psme1 | Proteasome activator subunit 1 (PA28 alpha) Proteasome activator that strongly increases the maximal velocity of the hydrolytic reaction and decreases the concentration of substrate required for cleavage |
| Psme2 | Proteasome activator subunit 2 (PA28 beta) Proteasome activator that strongly increases the maximal velocity of the hydrolytic reaction and decreases the concentration of substrate required for cleavage |
| ISGilation and Ubiquitination | |
| Isg15 | ISG15 ubiquitin-like modifier Ubiquitin-like protein conjugated to intracellular target proteins upon activation by interferon-alpha and interferon-beta |
| Isg20 | Interferon stimulated gene 20 RNA binding activity and nuclease activity |
| Transmembrane Receptor | |
| CD69 | Calcium dependent lectin superfamily type II transmembrane receptors |
| Cargo | Display Format | Immune Response | Species | References |
|---|---|---|---|---|
| Cathepsin L mimotope fasciola hepatica | Recombinant pIII | Humoral response | Capra hircus | [12] |
| H5N1 influenza virus | Recombinant pIII | Humoral response | Homo sapiens | [16] |
| Fba1 Candida albicans | Recombinant pIII/Recombinant pVIII | Humoral/Cellular response | Mus musculus | [24] |
| HSP90 Candida albicans | Recombinant pVIII | Humoral/Cellular response | Mus musculus | [25,42] |
| Sap2 Candida albicans | Recombinant pVIII | Humoral/Cellular response | Mus musculus | [26,43] |
| Gp70 Sporothrix globosa | Recombinant pIII | Humoral/Cellular response | Mus musculus | [27] |
| beta-amyloid | Recombinant pVIII | Humoral response | Mus musculus | [30,31,32,33] |
| Reverse transcriptase HIV-1 | Recombinant pVIII | Cellular response | Homo sapiens/Mus musculus | [39,44,45] |
| pp65 CMV | Recombinant pVIII | Cellular response | Homo sapiens | [41] |
| Ovalbumin | Recombinant pVIII | Cellular response | Homo sapiens | [48,61,62] |
| MAGE-A1/MAGE-A3/MAGE-A10 melanoma antigen | Recombinant pVIII | Cellular response | Mus musculus | [51,52,53] |
| Multiple myeloma idiotype | Chemical link | Humoral/Cellular response | Homo sapiens | [54] |
| P1A mastocytoma antigen | Recombinant pVIII | Cellular response | Mus musculus | [52] |
| Her2-Neu cancer antigen | Recombinant pVIII | Cellular response | Mus musculus | [55] |
| scFv anti-Carcinoembryonic antigen (CEA) | Recombinant pVIII | Cellular response | Mus musculus | [57] |
| PA8, TSKB20 Trypanosoma cruzi | Recombinant pVIII | Cellular response | Mus musculus | [61] |
| scFv anti- DEC205 receptor | Recombinant pIII | Innate immune response | Mus musculus | [62,64] |
| alpha-galactosylceramide | Chemical link | Cellular response | Mus musculus | [75] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sartorius, R.; D’Apice, L.; Prisco, A.; De Berardinis, P. Arming Filamentous Bacteriophage, a Nature-Made Nanoparticle, for New Vaccine and Immunotherapeutic Strategies. Pharmaceutics 2019, 11, 437. https://doi.org/10.3390/pharmaceutics11090437
Sartorius R, D’Apice L, Prisco A, De Berardinis P. Arming Filamentous Bacteriophage, a Nature-Made Nanoparticle, for New Vaccine and Immunotherapeutic Strategies. Pharmaceutics. 2019; 11(9):437. https://doi.org/10.3390/pharmaceutics11090437
Chicago/Turabian StyleSartorius, Rossella, Luciana D’Apice, Antonella Prisco, and Piergiuseppe De Berardinis. 2019. "Arming Filamentous Bacteriophage, a Nature-Made Nanoparticle, for New Vaccine and Immunotherapeutic Strategies" Pharmaceutics 11, no. 9: 437. https://doi.org/10.3390/pharmaceutics11090437
APA StyleSartorius, R., D’Apice, L., Prisco, A., & De Berardinis, P. (2019). Arming Filamentous Bacteriophage, a Nature-Made Nanoparticle, for New Vaccine and Immunotherapeutic Strategies. Pharmaceutics, 11(9), 437. https://doi.org/10.3390/pharmaceutics11090437






