OCTN2-Mediated Acetyl-l-Carnitine Transport in Human Pulmonary Epithelial Cells In Vitro
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Human Alveolar Epithelial Cell Isolation and Culture
2.3. Cell Line Culture
2.4. Uptake Studies
2.5. Transport Studies
2.6. Data Analysis
3. Results
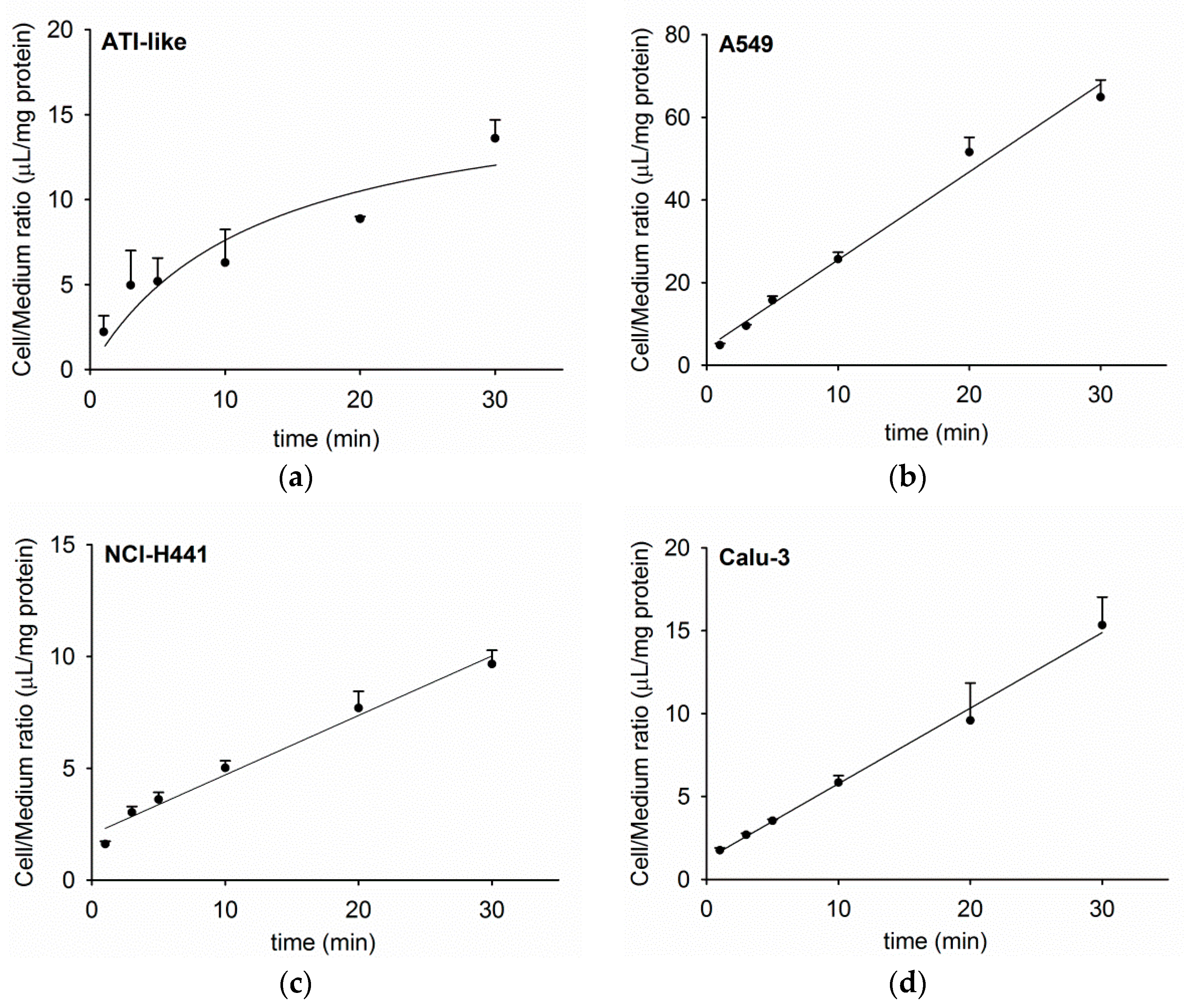
3.1. Time Course of Acetyl-l-Carnitine Uptake by Human Respiratory Epithelial Cells
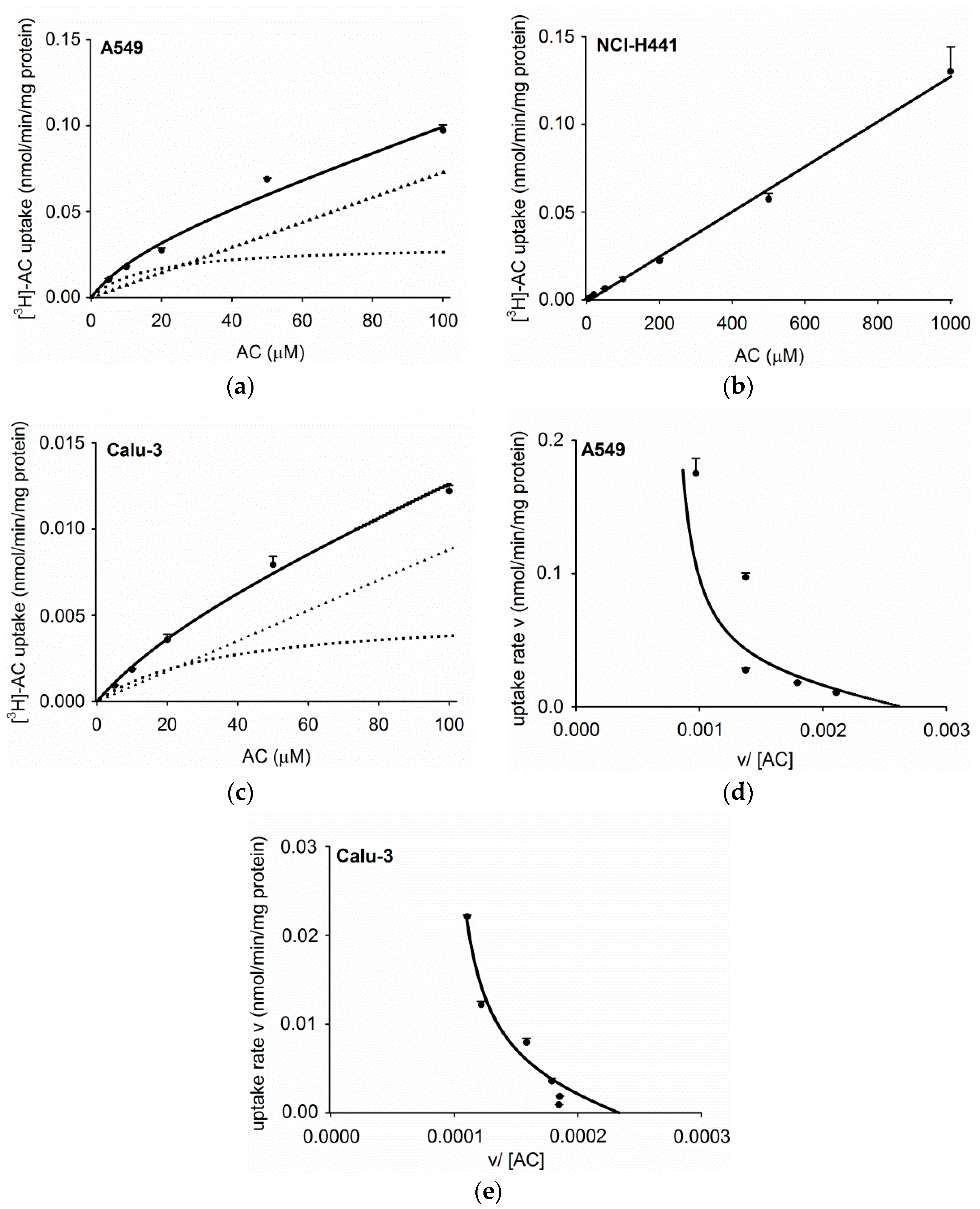
3.2. Concentration Dependence of Acetyl-l-Carnitine Uptake
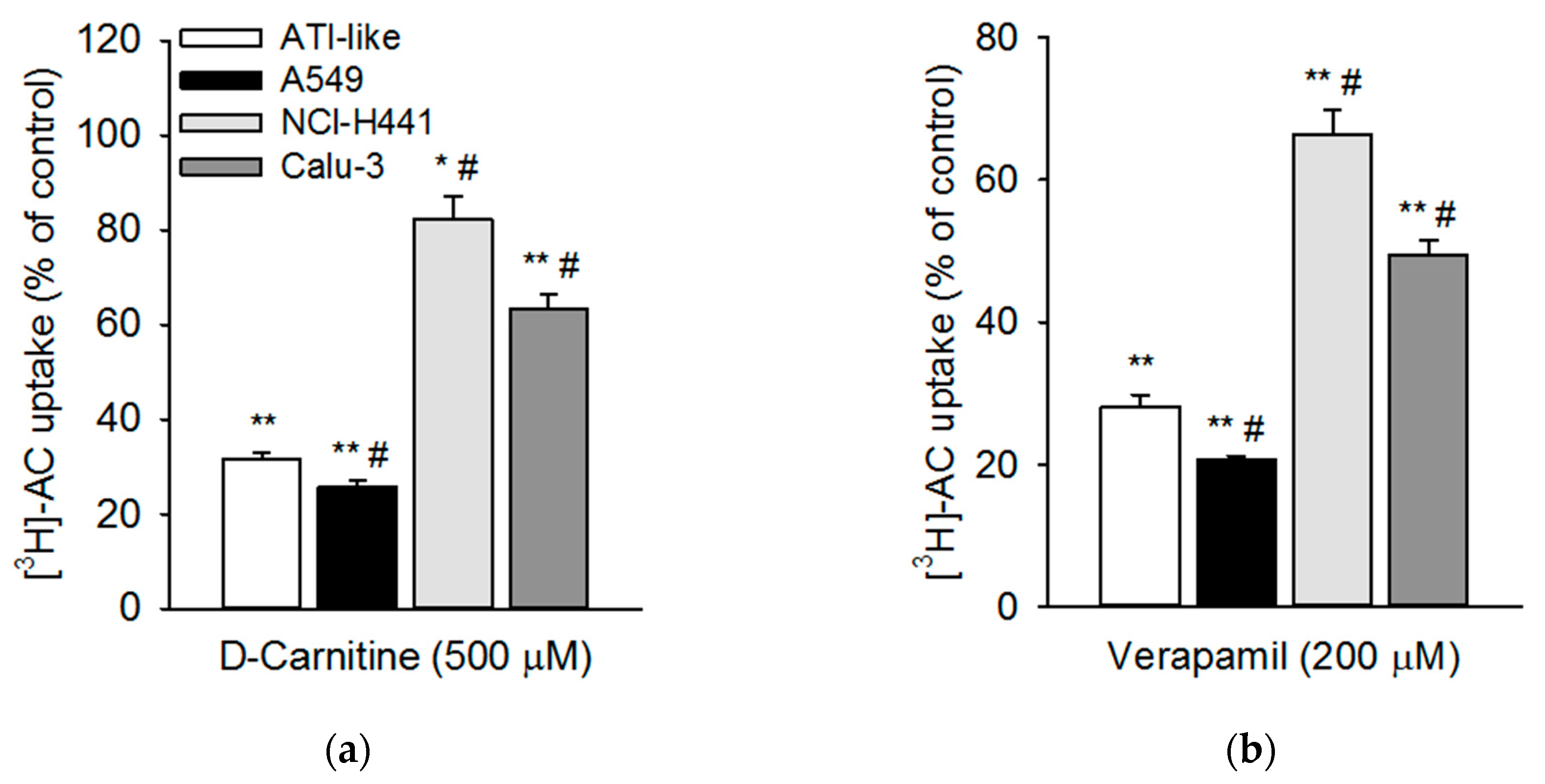
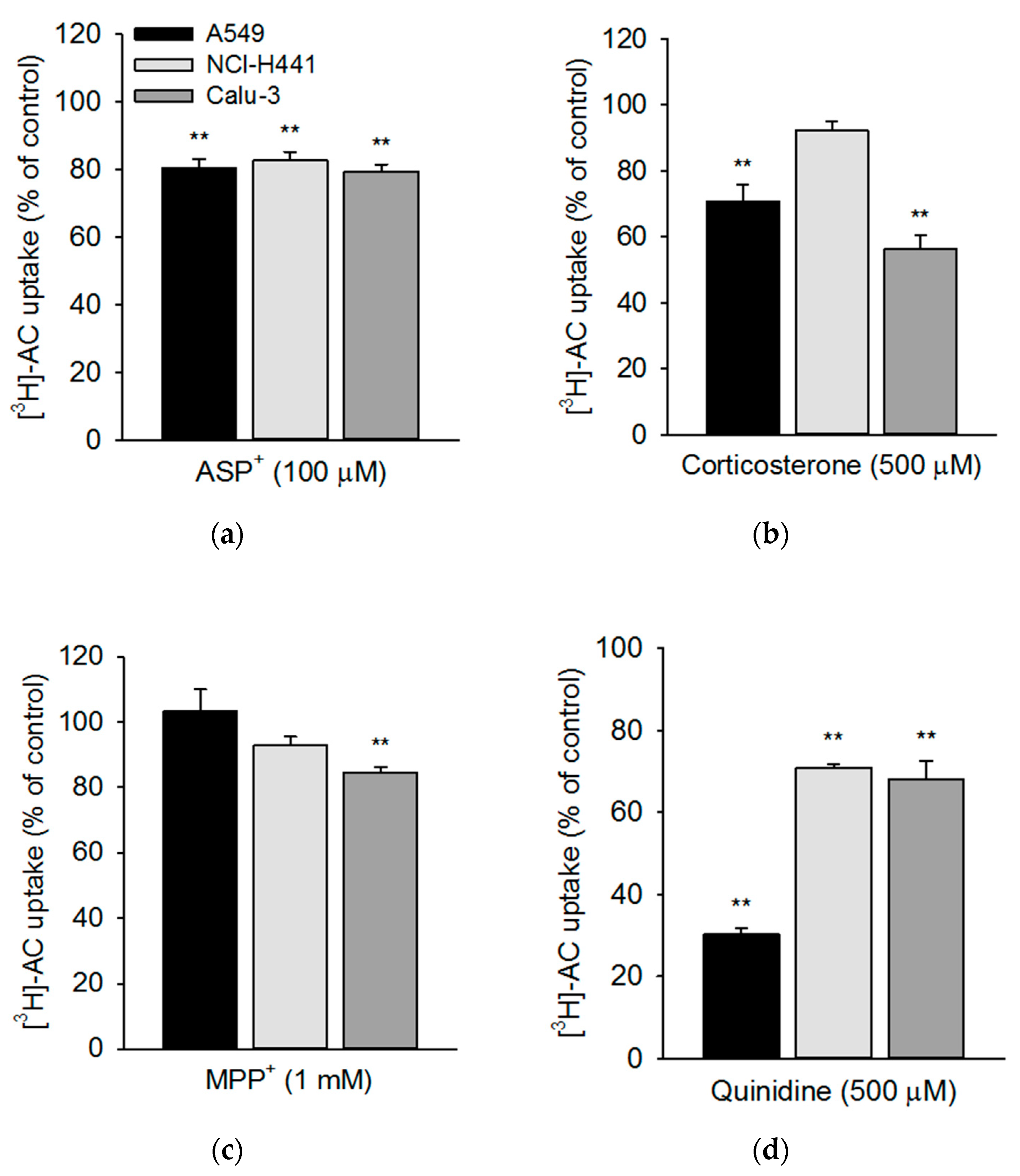
3.3. Influence of Pharmacological Modulators on Acetyl-l-Carnitine Uptake
3.4. Transport of Acetyl-l-Carnitine Uptake across NCl-H441 and Calu-3 Cells
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Koepsell, H. Multiple binding sites in organic cation transporters require sophisticated procedures to identify interactions of novel drugs. Biol. Chem. 2019, 400, 195–207. [Google Scholar] [CrossRef] [PubMed]
- Salomon, J.J.; Ehrhardt, C. Organic cation transporters in the blood-air barrier: Expression and implications for pulmonary drug delivery. Ther. Deliv. 2012, 3, 735–747. [Google Scholar] [CrossRef] [PubMed]
- Salomon, J.J.; Hagos, Y.; Petzke, S.; Kuhne, A.; Gausterer, J.C.; Hosoya, K.; Ehrhardt, C. Beta-2 Adrenergic Agonists Are Substrates and Inhibitors of Human Organic Cation Transporter 1. Mol. Pharm. 2015, 12, 2633–2641. [Google Scholar] [CrossRef] [PubMed]
- Gnadt, M.; Trammer, B.; Kardziev, B.; Bayliss, M.K.; Edwards, C.D.; Schmidt, M.; Hogger, P. Comparison of the bronchodilating effects of inhaled beta(2)-agonists after methacholine challenge in a human lung reperfusion model. Eur. J. Pharm. Biopharm. 2012, 81, 617–626. [Google Scholar] [CrossRef] [PubMed]
- Backstrom, E.; Lundqvist, A.; Boger, E.; Svanberg, P.; Ewing, P.; Hammarlund-Udenaes, M.; Friden, M. Development of a Novel Lung Slice Methodology for Profiling of Inhaled Compounds. J. Pharm. Sci. 2015, 105, 838–845. [Google Scholar] [CrossRef] [PubMed]
- Nakanishi, T.; Hasegawa, Y.; Haruta, T.; Wakayama, T.; Tamai, I. In vivo evidence of organic cation transporter-mediated tracheal accumulation of the anticholinergic agent ipratropium in mice. J. Pharm. Sci. 2013, 102, 3373–3381. [Google Scholar] [CrossRef]
- Moffatt, M.F.; Gut, I.G.; Demenais, F.; Strachan, D.P.; Bouzigon, E.; Heath, S.; von Mutius, E.; Farrall, M.; Lathrop, M.; Cookson, W.; et al. A large-scale, consortium-based genomewide association study of asthma. N. Engl. J. Med. 2010, 363, 1211–1221. [Google Scholar] [CrossRef]
- Shrine, N.; Portelli, M.A.; John, C.; Soler Artigas, M.; Bennett, N.; Hall, R.; Lewis, J.; Henry, A.P.; Billington, C.K.; Ahmad, A.; et al. Moderate-to-severe asthma in individuals of European ancestry: A genome-wide association study. Lancet Respir. Med. 2019, 7, 20–34. [Google Scholar] [CrossRef]
- Berg, T.; Hegelund-Myrback, T.; Ockinger, J.; Zhou, X.H.; Brannstrom, M.; Hagemann-Jensen, M.; Werkstrom, V.; Seidegard, J.; Grunewald, J.; Nord, M.; et al. Expression of MATE1, P-gp, OCTN1 and OCTN2, in epithelial and immune cells in the lung of COPD and healthy individuals. Respir. Res. 2018, 19, 68. [Google Scholar] [CrossRef]
- Bleasby, K.; Castle, J.C.; Roberts, C.J.; Cheng, C.; Bailey, W.J.; Sina, J.F.; Kulkarni, A.V.; Hafey, M.J.; Evers, R.; Johnson, J.M.; et al. Expression profiles of 50 xenobiotic transporter genes in humans and pre-clinical species: A resource for investigations into drug disposition. Xenobiotica 2006, 36, 963–988. [Google Scholar] [CrossRef]
- Endter, S.; Francombe, D.; Ehrhardt, C.; Gumbleton, M. RT-PCR analysis of ABC, SLC and SLCO drug transporters in human lung epithelial cell models. J. Pharm. Pharmacol. 2009, 61, 583–591. [Google Scholar] [CrossRef] [PubMed]
- Courcot, E.; Leclerc, J.; Lafitte, J.J.; Mensier, E.; Jaillard, S.; Gosset, P.; Shirali, P.; Pottier, N.; Broly, F.; Lo-Guidice, J.M. Xenobiotic metabolism and disposition in human lung cell models: Comparison with in vivo expression profiles. Drug Metab. Dispos. 2012, 40, 1953–1965. [Google Scholar] [CrossRef] [PubMed]
- Sakamoto, A.; Matsumaru, T.; Yamamura, N.; Suzuki, S.; Uchida, Y.; Tachikawa, M.; Terasaki, T. Drug Transporter Protein Quantification of Immortalized Human Lung Cell Lines Derived from Tracheobronchial Epithelial Cells (Calu-3 and BEAS2-B), Bronchiolar-Alveolar Cells (NCI-H292 and NCI-H441), and Alveolar Type II-like Cells (A549) by Liquid Chromatography-Tandem Mass Spectrometry. J. Pharm. Sci. 2015, 104, 3029–3038. [Google Scholar] [CrossRef] [PubMed]
- Salomon, J.J.; Endter, S.; Tachon, G.; Falson, F.; Buckley, S.T.; Ehrhardt, C. Transport of the fluorescent organic cation 4-(4-(dimethylamino)styryl)-N-methylpyridinium iodide (ASP+) in human respiratory epithelial cells. Eur. J. Pharm. Biopharm. 2012, 81, 351–359. [Google Scholar] [CrossRef] [PubMed]
- Salomon, J.J.; Muchitsch, V.E.; Gausterer, J.C.; Schwagerus, E.; Huwer, H.; Daum, N.; Lehr, C.M.; Ehrhardt, C. The cell line NCl-H441 is a useful in vitro model for transport studies of human distal lung epithelial barrier. Mol. Pharm. 2014, 11, 995–1006. [Google Scholar] [CrossRef]
- Nakamura, T.; Nakanishi, T.; Haruta, T.; Shirasaka, Y.; Keogh, J.P.; Tamai, I. Transport of ipratropium, an anti-chronic obstructive pulmonary disease drug, Is mediated by organic cation/carnitine transporters in human bronchial epithelial cells: Implications for carrier-mediated pulmonary absorption. Mol. Pharm. 2010, 7, 187–195. [Google Scholar] [CrossRef] [PubMed]
- Ingoglia, F.; Visigalli, R.; Rotoli, B.M.; Barilli, A.; Riccardi, B.; Puccini, P.; Dall’Asta, V. Functional activity of L-carnitine transporters in human airway epithelial cells. Biochim. Biophys. Acta 2016, 1858, 210–219. [Google Scholar] [CrossRef]
- Macdonald, C.; Shao, D.; Oli, A.; Agu, R.U. Characterization of Calu-3 cell monolayers as a model of bronchial epithelial transport: Organic cation interaction studies. J. Drug Target. 2013, 21, 97–106. [Google Scholar] [CrossRef]
- Sakamoto, A.; Matsumaru, T.; Yamamura, N.; Uchida, Y.; Tachikawa, M.; Ohtsuki, S.; Terasaki, T. Quantitative expression of human drug transporter proteins in lung tissues: Analysis of regional, gender, and interindividual differences by liquid chromatography-tandem mass spectrometry. J. Pharm. Sci. 2013, 102, 3395–3406. [Google Scholar] [CrossRef]
- Lips, K.S.; Volk, C.; Schmitt, B.M.; Pfeil, U.; Arndt, P.; Miska, D.; Ermert, L.; Kummer, W.; Koepsell, H. Polyspecific cation transporters mediate luminal release of acetylcholine from bronchial epithelium. Am. J. Respir. Cell Mol. Biol. 2005, 33, 79–88. [Google Scholar] [CrossRef]
- Horvath, G.; Schmid, N.; Fragoso, M.A.; Schmid, A.; Conner, G.E.; Salathe, M.; Wanner, A. Epithelial organic cation transporters ensure pH-dependent drug absorption in the airway. Am. J. Respir. Cell Mol. Biol 2007, 36, 53–60. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, M.; Cingolani, E.; Pritchard, D.I.; Bosquillon, C. Enhanced expression of Organic Cation Transporters in bronchial epithelial cell layers following insults associated with asthma-Impact on salbutamol transport. Eur. J. Pharm. Sci. 2017, 106, 62–70. [Google Scholar] [CrossRef] [PubMed]
- Al-Jayyoussi, G.; Price, D.F.; Kreitmeyr, K.; Keogh, J.P.; Smith, M.W.; Gumbleton, M.; Morris, C.J. Absorption of ipratropium and l-carnitine into the pulmonary circulation of the ex-vivo rat lung is driven by passive processes rather than active uptake by OCT/OCTN transporters. Int. J. Pharm. 2015, 496, 834–841. [Google Scholar] [CrossRef] [PubMed]
- Eaton, D.C.; Chen, J.; Ramosevac, S.; Matalon, S.; Jain, L. Regulation of Na+ channels in lung alveolar type II epithelial cells. Proc. Am. Thorac. Soc. 2004, 1, 10–16. [Google Scholar] [CrossRef] [PubMed]
- Koepsell, H.; Lips, K.; Volk, C. Polyspecific organic cation transporters: Structure, function, physiological roles, and biopharmaceutical implications. Pharm. Res. 2007, 24, 1227–1251. [Google Scholar] [CrossRef] [PubMed]
- Tachikawa, M.; Takeda, Y.; Tomi, M.; Hosoya, K. Involvement of OCTN2 in the transport of acetyl-L-carnitine across the inner blood-retinal barrier. Investig. Ophthalmol. Vis. Sci. 2010, 51, 430–436. [Google Scholar] [CrossRef] [PubMed]
- Forbes, B.; Ehrhardt, C. Human respiratory epithelial cell culture for drug delivery applications. Eur. J. Pharm. Biopharm. 2005, 60, 193–205. [Google Scholar] [CrossRef] [PubMed]
- Daum, N.; Kuehn, A.; Hein, S.; Schaefer, U.F.; Huwer, H.; Lehr, C.M. Isolation, cultivation, and application of human alveolar epithelial cells. Methods Mol. Biol. 2012, 806, 31–42. [Google Scholar] [CrossRef]
- Demling, N.; Ehrhardt, C.; Kasper, M.; Laue, M.; Knels, L.; Rieber, E.P. Promotion of cell adherence and spreading: A novel function of RAGE, the highly selective differentiation marker of human alveolar epithelial type I cells. Cell Tissue Res. 2006, 323, 475–488. [Google Scholar] [CrossRef]
- Salomon, J.J.; Gausterer, J.C.; Yahara, T.; Hosoya, K.I.; Huwer, H.; Hittinger, M.; Schneider-Daum, N.; Lehr, C.M.; Ehrhardt, C. Organic cation transporter function in different in vitro models of human lung epithelium. Eur. J. Pharm. Sci. 2015. [Google Scholar] [CrossRef]
- Salomon, J.J.; Ehrhardt, C.; Hosoya, K. The verapamil transporter expressed in human alveolar epithelial cells (A549) does not interact with beta2-receptor agonists. Drug Metab. Pharmacokinet. 2014, 29, 101–104. [Google Scholar] [CrossRef]
- Nies, A.T.; Koepsell, H.; Damme, K.; Schwab, M. Organic cation transporters (OCTs, MATEs), in vitro and in vivo evidence for the importance in drug therapy. Handb. Exp. Pharmacol. 2011. [Google Scholar] [CrossRef]
- Torgerson, D.G.; Ampleford, E.J.; Chiu, G.Y.; Gauderman, W.J.; Gignoux, C.R.; Graves, P.E.; Himes, B.E.; Levin, A.M.; Mathias, R.A.; Hancock, D.B.; et al. Meta-analysis of genome-wide association studies of asthma in ethnically diverse North American populations. Nat. Gen. 2011, 43, 887–892. [Google Scholar] [CrossRef]
- Nickel, S.; Clerkin, C.G.; Selo, M.A.; Ehrhardt, C. Transport mechanisms at the pulmonary mucosa: Implications for drug delivery. Expert Opin. Drug Deliv. 2016, 13, 667–690. [Google Scholar] [CrossRef]
- Mo, J.; Lim, L.Y.; Zhang, Z.R. L-Carnitine ester of prednisolone: Pharmacokinetic and pharmacodynamic evaluation of a type I prodrug. Int. J. Pharm. 2014, 475, 123–129. [Google Scholar] [CrossRef]
- Ohashi, R.; Tamai, I.; Yabuuchi, H.; Nezu, J.I.; Oku, A.; Sai, Y.; Shimane, M.; Tsuji, A. Na(+)-dependent carnitine transport by organic cation transporter (OCTN2): Its pharmacological and toxicological relevance. J. Pharmacol. Exp. Ther. 1999, 291, 778–784. [Google Scholar]
- Ohashi, R.; Tamai, I.; Nezu Ji, J.; Nikaido, H.; Hashimoto, N.; Oku, A.; Sai, Y.; Shimane, M.; Tsuji, A. Molecular and physiological evidence for multifunctionality of carnitine/organic cation transporter OCTN2. Mol. Pharm. 2001, 59, 358–366. [Google Scholar] [CrossRef]
- Longo, N.; Frigeni, M.; Pasquali, M. Carnitine transport and fatty acid oxidation. Biochim. Biophys. Acta 2016, 1863, 2422–2435. [Google Scholar] [CrossRef]
- Tamai, I. Pharmacological and pathophysiological roles of carnitine/organic cation transporters (OCTNs: SLC22A4, SLC22A5 and Slc22a21). Biopharm. Drug Dispos. 2013, 34, 29–44. [Google Scholar] [CrossRef]
- Otsubo, C.; Bharathi, S.; Uppala, R.; Ilkayeva, O.R.; Wang, D.; McHugh, K.; Zou, Y.; Wang, J.; Alcorn, J.F.; Zuo, Y.Y.; et al. Long-chain Acylcarnitines Reduce Lung Function by Inhibiting Pulmonary Surfactant. J. Biol. Chem. 2015, 290, 23897–23904. [Google Scholar] [CrossRef]
- Knottnerus, S.J.G.; Bleeker, J.C.; Wust, R.C.I.; Ferdinandusse, S.; IJlst, L.; Wijburg, F.A.; Wanders, R.J.A.; Visser, G.; Houtkooper, R.H. Disorders of mitochondrial long-chain fatty acid oxidation and the carnitine shuttle. Rev. Endocr. Metab. Disord. 2018, 19, 93–106. [Google Scholar] [CrossRef]
- Lin, L.; Yee, S.W.; Kim, R.B.; Giacomini, K.M. SLC transporters as therapeutic targets: Emerging opportunities. Nat. Rev. Drug Discov. 2015, 14, 543–560. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Salomon, J.J.; Gausterer, J.C.; Selo, M.A.; Hosoya, K.-i.; Huwer, H.; Schneider-Daum, N.; Lehr, C.-M.; Ehrhardt, C. OCTN2-Mediated Acetyl-l-Carnitine Transport in Human Pulmonary Epithelial Cells In Vitro. Pharmaceutics 2019, 11, 396. https://doi.org/10.3390/pharmaceutics11080396
Salomon JJ, Gausterer JC, Selo MA, Hosoya K-i, Huwer H, Schneider-Daum N, Lehr C-M, Ehrhardt C. OCTN2-Mediated Acetyl-l-Carnitine Transport in Human Pulmonary Epithelial Cells In Vitro. Pharmaceutics. 2019; 11(8):396. https://doi.org/10.3390/pharmaceutics11080396
Chicago/Turabian StyleSalomon, Johanna J., Julia C. Gausterer, Mohammed Ali Selo, Ken-ichi Hosoya, Hanno Huwer, Nicole Schneider-Daum, Claus-Michael Lehr, and Carsten Ehrhardt. 2019. "OCTN2-Mediated Acetyl-l-Carnitine Transport in Human Pulmonary Epithelial Cells In Vitro" Pharmaceutics 11, no. 8: 396. https://doi.org/10.3390/pharmaceutics11080396
APA StyleSalomon, J. J., Gausterer, J. C., Selo, M. A., Hosoya, K.-i., Huwer, H., Schneider-Daum, N., Lehr, C.-M., & Ehrhardt, C. (2019). OCTN2-Mediated Acetyl-l-Carnitine Transport in Human Pulmonary Epithelial Cells In Vitro. Pharmaceutics, 11(8), 396. https://doi.org/10.3390/pharmaceutics11080396






