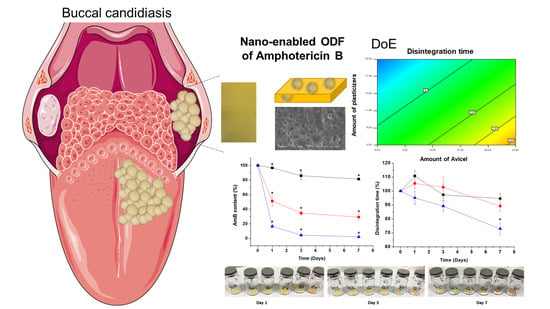
Designing Fast-Dissolving Orodispersible Films of Amphotericin B for Oropharyngeal Candidiasis
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Quality by Design (QbD) Optimisation of ODF
2.2.1. ODF Manufacture and Response Evaluation
2.2.2. ODF Optimisation
2.3. Full Physicochemical Characterisation of Optimised ODF
2.4. Content Uniformity
2.5. Release Studies in Artificial Saliva
2.6. In Vitro Antifungal Assays
2.7. Stability Studies
3. Results
3.1. QbD Studies for Optimisation of AmB-Loaded ODFs
3.2. Manufacturing of Optimised AmB ODF
3.3. Performance and Further Characterisation of the Optimised AmB-Loaded ODF
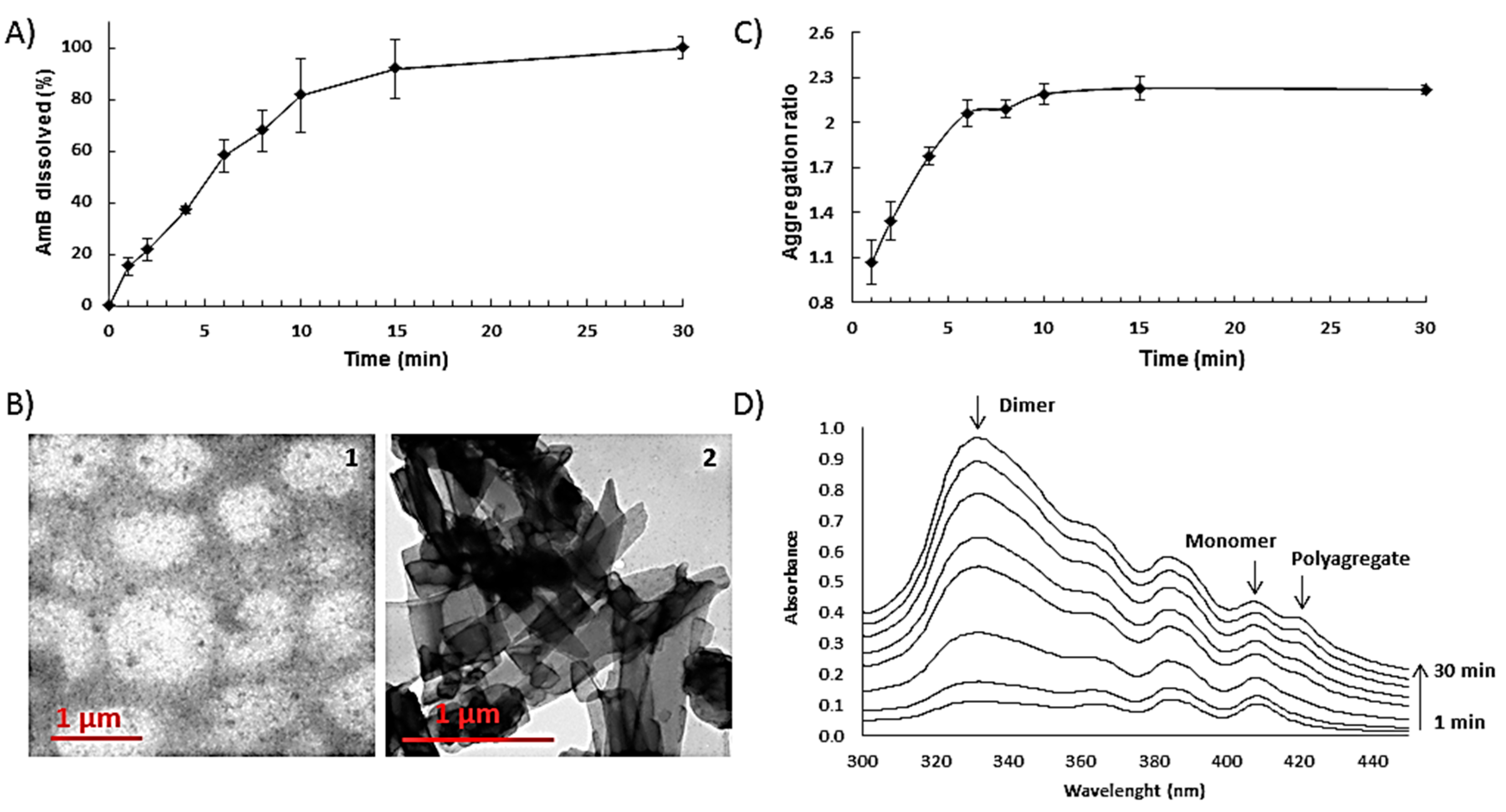
3.4. Release and Aggregation State
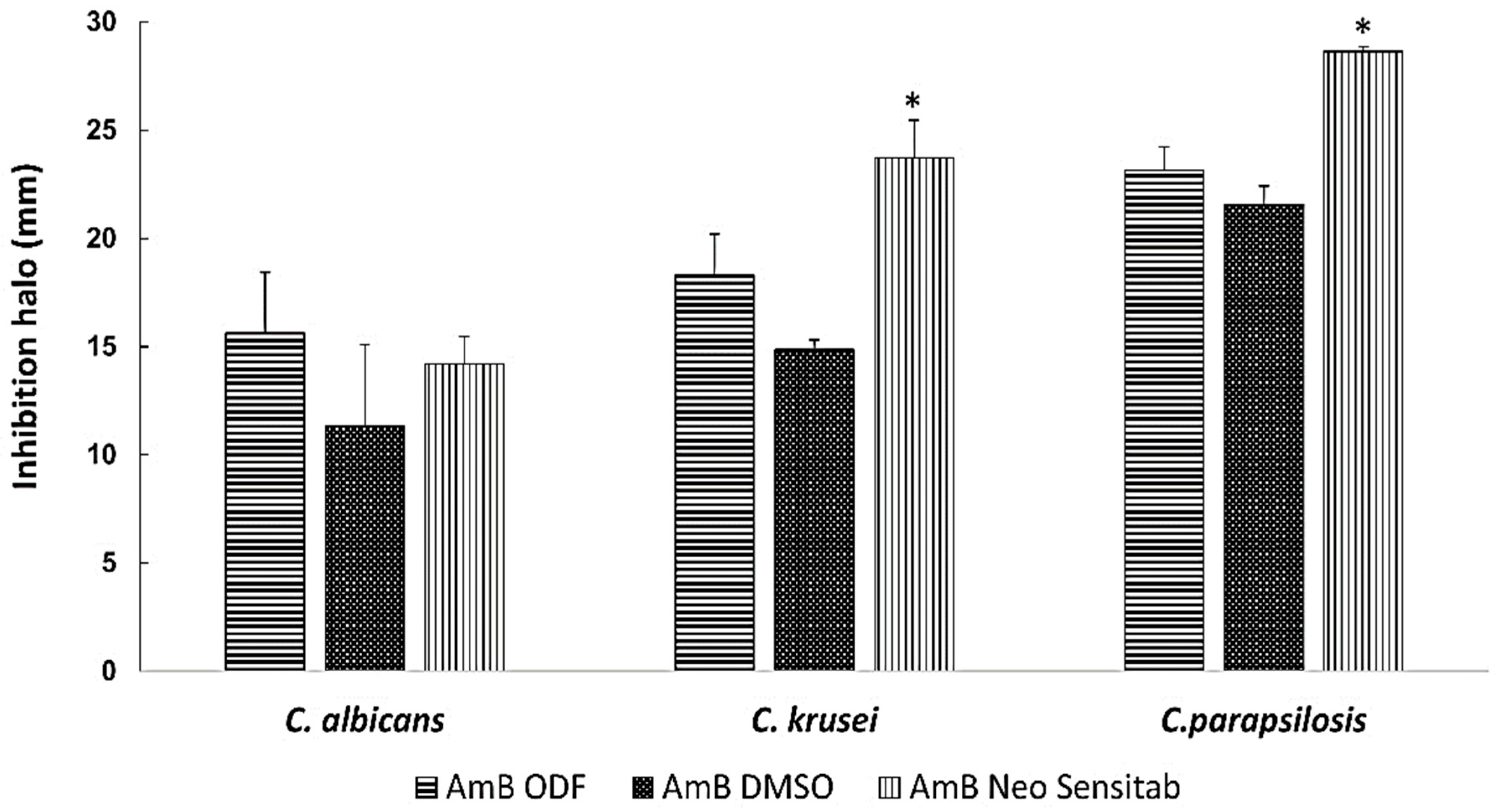
3.5. Antifungal Activity
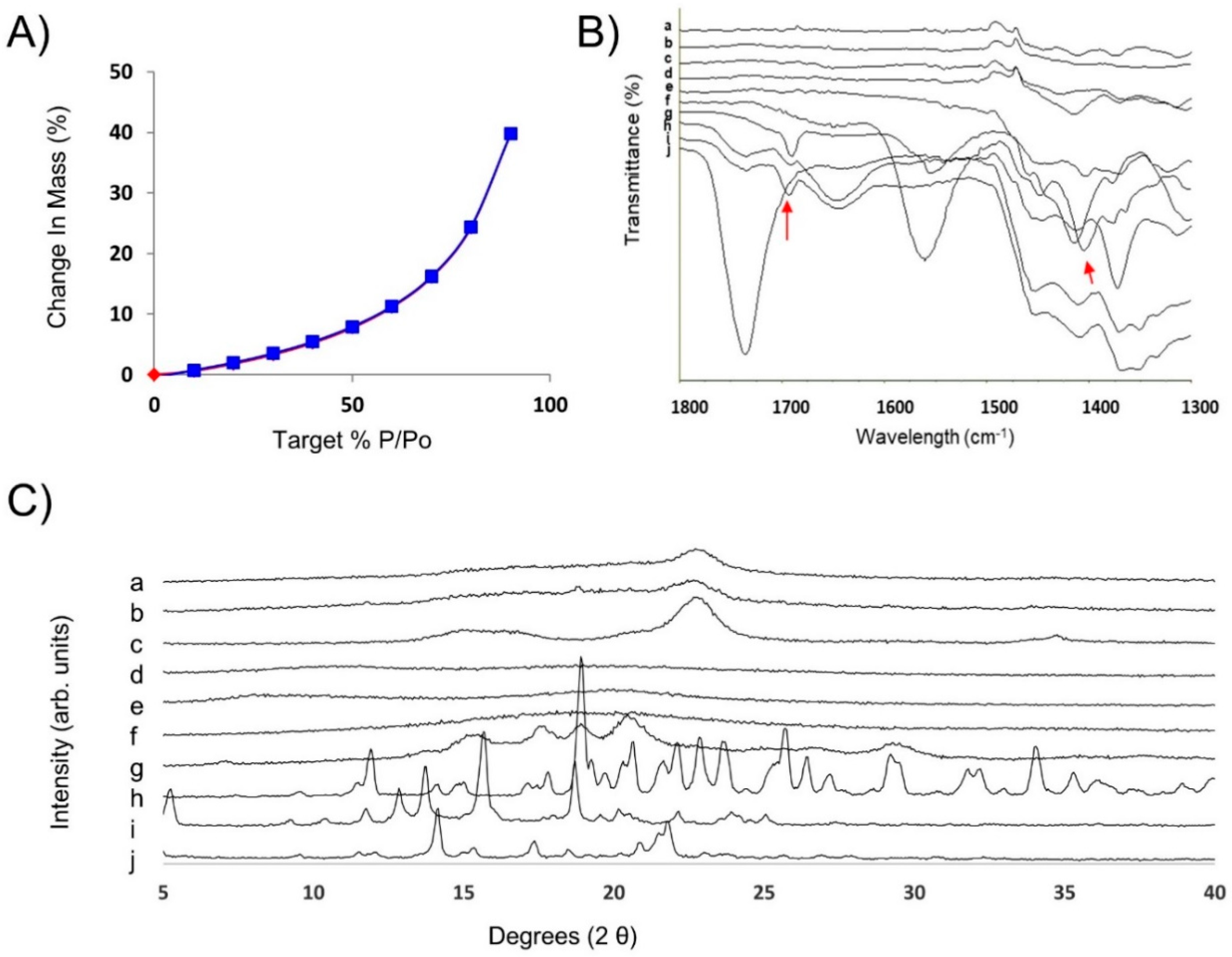
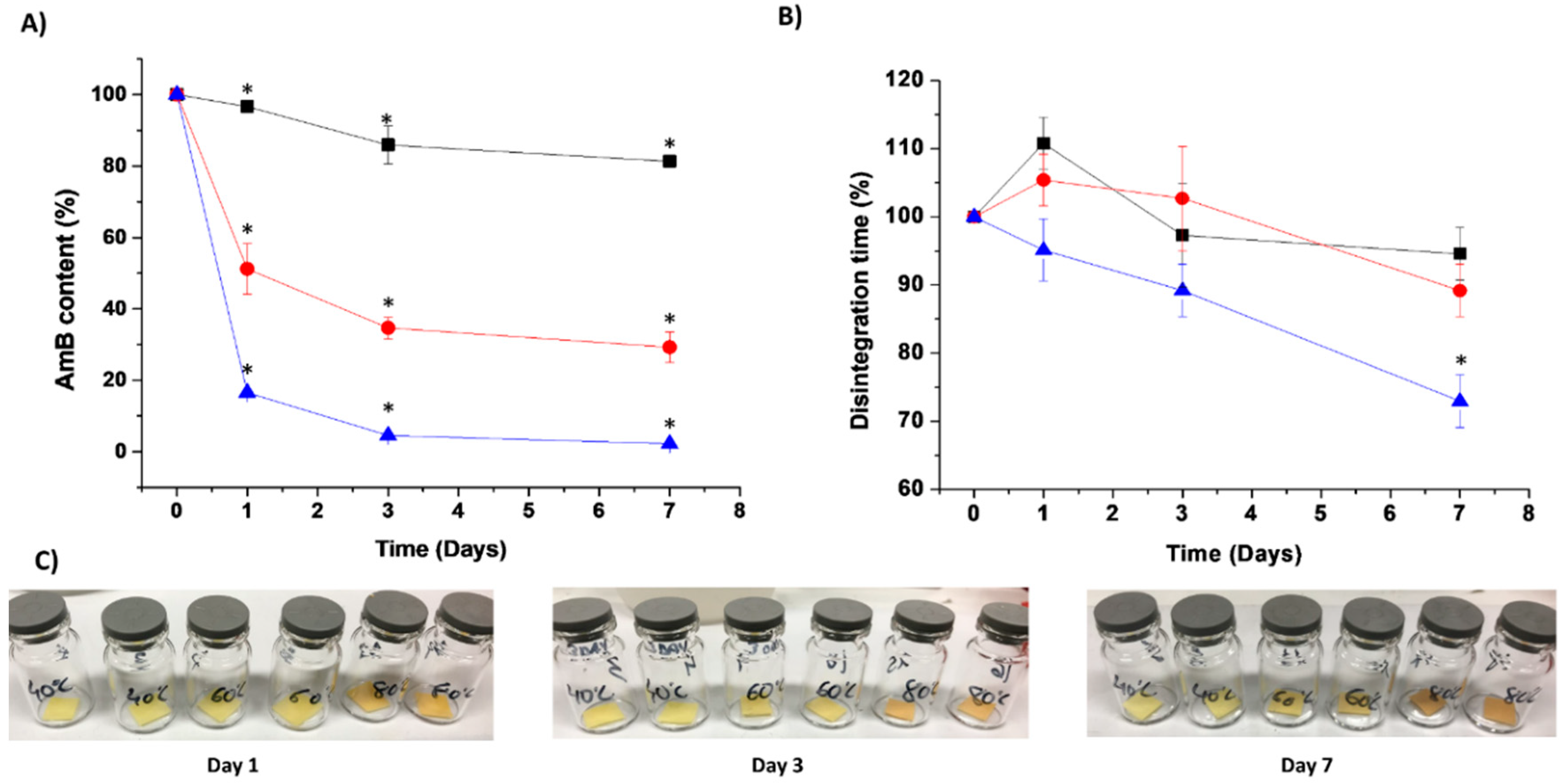
3.6. Physicochemical Stability
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Ruiz, H.K.; Serrano, D.R.; Dea-Ayuela, M.A.; Bilbao-Ramos, P.E.; Bolas-Fernandez, F.; Torrado, J.J.; Molero, G. New amphotericin B-gamma cyclodextrin formulation for topical use with synergistic activity against diverse fungal species and Leishmania spp. Int. J. Pharm. 2014, 473, 148–157. [Google Scholar] [CrossRef] [PubMed]
- Montaga, M.T.; Lovero, G.; Coretti, C.; de Giglio, O.; Mratinelli, D.; Bedini, A.; Delia, M.; Rosato, A.; Codeluppi, M.; Caggiano, C. In vitro activities of amphotericin B deoxycholate and liposomal amphotericin B against 604 clinical yeast isolates. J. Med. Microbiol. 2014, 63, 1638–1643. [Google Scholar] [CrossRef] [PubMed]
- Lagerlof, F.; Dawes, C. The volume of saliva in the mouth before and after swallowing. J. Dent. Res. 1984, 63, 618–621. [Google Scholar] [CrossRef] [PubMed]
- Technical Data Sheet of Fungizone. Available online: https://botplusweb.portalfarma.com/documentos/2003/9/22/F46773%20FUNGIZONA%20IV%20200111.pdf (accessed on 15 January 2019).
- AmBisome Technical Data Sheet. Available online: https://www.gilead.com/-/media/files/pdfs/medicines/other/ambisome/ambisome_pi.pdf (accessed on 14 March 2019).
- Reiner, V.; Giarratana, N.; Monti, N.C.; Breitenbach, A.; Klaffenbach, P. Rapidfilm: An innovative pharmaceutical form designed to improve patient compliance. Int. J. Pharm. 2010, 393, 55–60. [Google Scholar] [CrossRef] [PubMed]
- Gijare, C.; Deshpande, A. Orodispersible Films: A Systematic Patent Review. Recent Patents Drug Deliv. Formul. 2018, 12, 110–120. [Google Scholar] [CrossRef] [PubMed]
- Irfan, M.; Rabel, S.; Bukhtar, Q.; Qadir, M.I.; Jabeen, F.; Khan, A. Orally disintegrating films: A modern expansion in drug delivery system. Saudi Pharm. J. 2016, 24, 537–546. [Google Scholar] [CrossRef]
- Choudhary, D.R.; Patel, V.A.; Chhalotiya, U.K.; Patel, H.V.; Kundawala, A.J. Development and characterization of pharmacokinetic parameters of fast-dissolving films containing levocetirizine. Sci. Pharm. 2012, 80, 779–787. [Google Scholar] [CrossRef]
- Dhiman, M.K.; Dhiman, A.; Sawant, K.K. Transbuccal delivery of 5-fluorouracil: Permeation enhancement and pharmacokinetic study. AAPS Pharm. Sci. Technol. 2009, 10, 258–265. [Google Scholar] [CrossRef]
- Rolon, M.; Serrano, D.R.; Lalatsa, A.; de Pablo, E.; Torrado, J.J.; Ballesteros, M.P.; Healy, A.M.; Vega, C.; Coronel, C.; Bolas-Fernandez, F.; et al. Engineering Oral and Parenteral Amorphous Amphotericin B Formulations against Experimental Trypanosoma cruzi Infections. Mol. Pharm. 2017, 14, 1095–1106. [Google Scholar] [CrossRef]
- Okamoto, H.; Taguchi, H.; Iida, K.; Danjo, K. Development of polymer film dosage forms of lidocaine for buccal administration. I. Penetration rate and release rate. J. Control. Release 2001, 77, 253–260. [Google Scholar] [CrossRef]
- Senthilkumar, K.; Vijaya, C. Formulation Development of Mouth Dissolving Film of Etoricoxib for Pain Management. Adv. Pharm. 2015, 2015, 1–11. [Google Scholar] [CrossRef]
- Serrano, D.R.; Walsh, D.; O’Connell, P.; Mugheirbi, N.A.; Worku, Z.A.; Bolas-Fernandez, F.; Galiana, C.; Dea-Ayuela, M.A.; Healy, A.M. Optimising the in vitro and in vivo performance of oral cocrystal formulations via spray coating. Eur. J. Pharm. Biopharm. 2018, 124, 13–27. [Google Scholar] [CrossRef] [PubMed]
- Serrano, D.R.; Persoons, T.; D’Arcy, D.M.; Galiana, C.; Dea-Ayuela, M.A.; Healy, A.M. Modelling and shadowgraph imaging of cocrystal dissolution and assessment of in vitro antimicrobial activity for sulfadimidine/4-aminosalicylic acid cocrystals. Eur. J. Pharm. Sci. 2016, 89, 125–136. [Google Scholar] [CrossRef] [PubMed]
- Serrano, D.R.; Hernandez, L.; Fleire, L.; Gonzalez-Alvarez, I.; Montoya, A.; Ballesteros, M.P.; Dea-Ayuela, M.A.; Miro, G.; Bolas-Fernandez, F.; Torrado, J.J. Hemolytic and pharmacokinetic studies of liposomal and particulate amphotericin B formulations. Int. J. Pharm. 2013, 447, 38–46. [Google Scholar] [CrossRef] [PubMed]
- Torrado, J.J.; Espada, R.; Ballesteros, M.P.; Torrado-Santiago, S. Amphotericin B formulations and drug targeting. J. Pharm. Sci. 2008, 97, 2405–2425. [Google Scholar] [CrossRef] [PubMed]
- Clinical and Laboratory Standards Institute. Method for antifungal disk diffusion susceptibility testing of yeast. In CLSI document M44-A2, 2nd ed.; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2009; Available online: https://clsi.org/media/1634/m44a2_sample.pdf (accessed on 7 July 2019).
- Markande, A.; Nezzal, A.; Fitzpatrick, J.J.; Aerts, L. Investigation of the Crystallization Kinetics of Dextrose Monohydrate Using In Situ Particle Size and Supersaturation Monitoring. Part. Sci. Technol. 2009, 27, 373–388. [Google Scholar] [CrossRef]
- Serrano, D.R.; Ballesteros, M.P.; Schatzlein, A.G.; Torrado, J.J.; Uchegbu, I.F. Amphotericin B Formulations —The Possibility of Generic Competition. Pharm. Nanotechnol. 2013, 1, 250–258. [Google Scholar] [CrossRef]
- Rajagopalan, N.; Dicken, C.M.; Ravin, L.J.; Sternson, L.A. A study of the solubility of amphotericin B in nonaqueous solvent systems. J. Parenter. Sci. Technol. Publ. Parenter. Drug Assoc. 1988, 42, 97–102. [Google Scholar]
- Pycia, K.; Gryszkin, A.; Berski, W.; Juszczak, L. The Influence of Chemically Modified Potato Maltodextrins on Stability and Rheological Properties of Model Oil-in-Water Emulsions. Polymers 2018, 10, 67. [Google Scholar] [CrossRef] [PubMed]
- Iorgulescu, G. Saliva between normal and pathological. Important factors in determining systemic and oral health. J. Med. Life 2009, 2, 303–307. [Google Scholar] [PubMed]
- Jun, S.; Zhang, H.; Bechhoefer, J. Nucleation and growth in one dimension. I. The generalized Kolmogorov-Johnson-Mehl-Avrami model. Phys. Rev. E 2005, 71, 011908. [Google Scholar] [CrossRef] [PubMed]
- Poletto, M.; Pistor, V.; Zeni, M.; Zattera, A.J. Crystalline properties and decomposition kinetics of cellulose fibers in wood pulp obtained by two pulping processes. Polym. Degrad. Stab. 2011, 96, 679–685. [Google Scholar] [CrossRef]
- Pistor, V.; Soares, B.G.; Mauler, R.S. Influence of the Polyhedral Oligomeric Silsesquioxane n-Phenylaminopropyl—POSS in the Thermal Stability and the Glass Transition Temperature of Epoxy Resin. Polimeros 2013, 23, 328–331. [Google Scholar] [CrossRef]
- Borges, A.F.; Silva, C.; Coelho, J.F.; Simoes, S. Oral films: Current status and future perspectives II Intellectual property, technologies and market needs. J. Control. Release 2015, 206, 108–121. [Google Scholar] [CrossRef] [PubMed]
- Lyu, X.; Zhao, C.; Yan, Z.M.; Hua, H. Efficacy of nystatin for the treatment of oral candidiasis: A systematic review and meta-analysis. Drug Des. Dev. Ther. 2016, 10, 1161–1171. [Google Scholar] [CrossRef] [PubMed]
- Johnson, E.M.; Ojwang, J.O.; Szekely, A.; Wallace, T.L.; Warnock, D.W. Comparison of in vitro antifungal activities of free and liposome-encapsulated nystatin with those of four amphotericin B formulations. Antimicrob. Agents Chemother. 1998, 42, 1412–1416. [Google Scholar] [CrossRef] [PubMed]
- Espada, R.; Valdespina, S.; Alfonso, C.; Rivas, G.; Ballesteros, M.P.; Torrado, J.J. Effect of aggregation state on the toxicity of different amphotericin B preparations. Int. J. Pharm. 2008, 361, 64–69. [Google Scholar] [CrossRef] [PubMed]
- Espada, R.; Valdespina, S.; Molero, G.; Dea, M.A.; Ballesteros, M.P.; Torrado, J.J. Efficacy of alternative dosing regimens of poly-aggregated amphotericin B. Int. J. Antimicrob. Agents 2008, 32, 55–61. [Google Scholar] [CrossRef]
- Singh, S.; Jain, S.; Muthu, M.S.; Tiwari, S.; Tilak, R. Preparation and evaluation of buccal bioadhesive films containing clotrimazole. AAPS Pharm. Sci. Technol. 2008, 9, 660–667. [Google Scholar] [CrossRef]
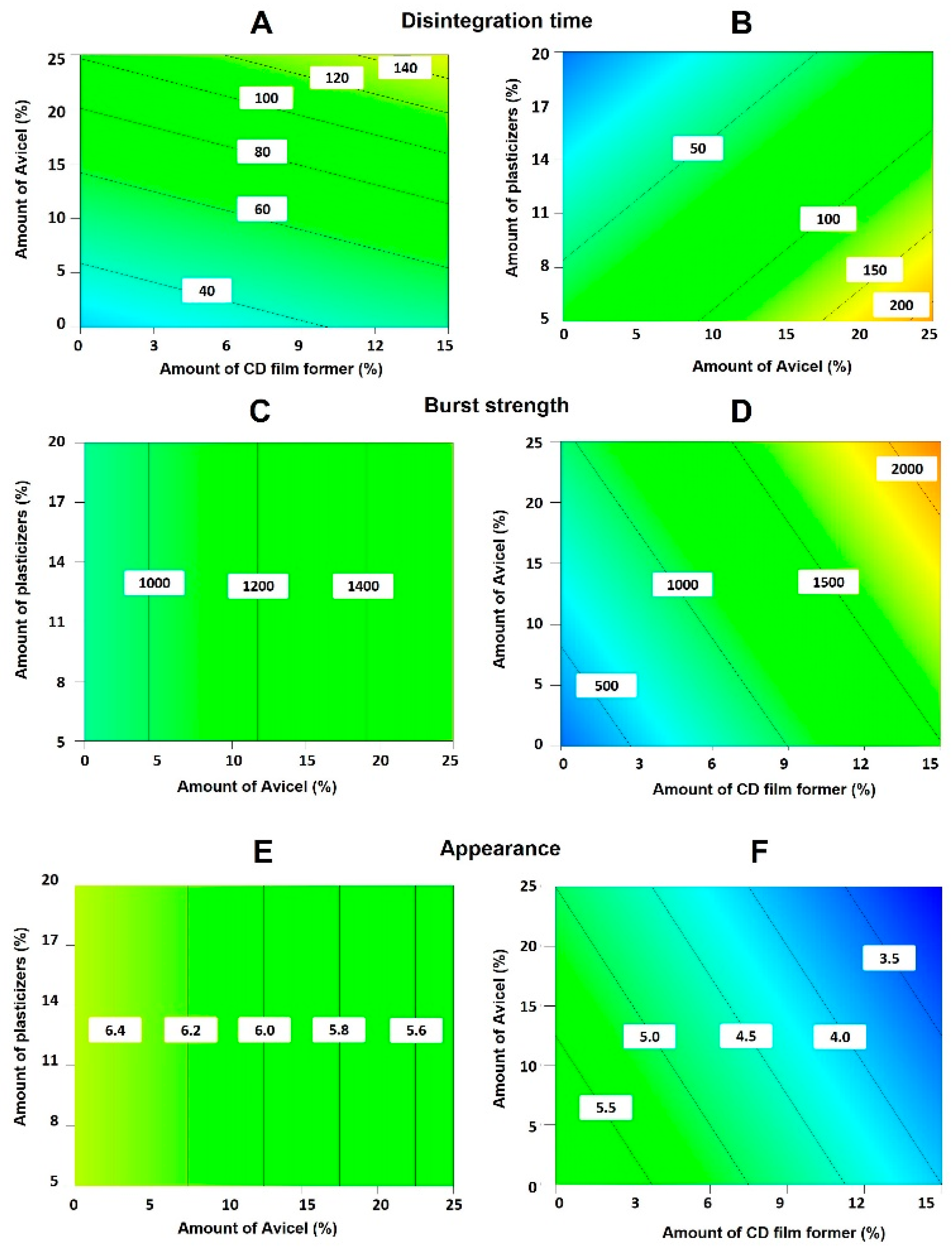

| Variables | Levels | |
|---|---|---|
| Low (−1) | High (+1) | |
| Type of dextrose-derived-polymer film former | Maltodextrin | Dextran |
| Taste masking agent (5%) | Sorbitol | Dextrose |
| Type of binder/suspending agent | Avicel 200 | Avicel CL-611 |
| Amount of Avicel polymers (%) | 0 | 25 |
| Amount of total plasticisers (PEG 400: glycerol, 1:1 w/w) (%) | 5 | 20 |
| Methanol | Low (1–2 mL) | High (>3 mL) |
| Amount of cellulose-derived film formers (HPMC 912 and HPC in a 1:4 weight ratio) (%) | 0 | 15 |
| Run | Type of Dextrose-Derived-Polymer Film Former | Taste Masking Excipient | Type of Avicel | Amount of Avicel (%) | Amount of Total Plasticisers (%) | Amount of Methanol | Amount of Cellulose-Derived Film Formers (%) | Disintegration Time (Seconds) | Flexibi- lity | Adhesion | Appea-rance | Burst Strength (Mn mm) |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Dextran | Sorbitol | 200 | 25 | 5 | 1 | 15 | 270 | 2 | 1 | 4 | 1300.9 |
| 2 | Maltodextrin | Dextrose | 200 | 25 | 20 | 2 | 15 | 90 | 1 | 1 | 3 | 2468 |
| 3 | Maltodextrin | Dextrose | 200 | 0 | 5 | 1 | 0 | 60 | 2 | 3 | 6 | 0 |
| 4 | Dextran | Dextrose | CL611 | 25 | 5 | 2 | 0 | 360 | 3 | 2 | 8 | 0 |
| 5 | Maltodextrin | Sorbitol | CL611 | 25 | 20 | 1 | 0 | 165 | 2 | 3 | 6 | 0 |
| 6 | Maltodextrin | Sorbitol | CL611 | 0 | 5 | 2 | 15 | 150 | 2 | 1 | 4 | 1061 |
| 7 | Dextran | Sorbitol | 200 | 0 | 20 | 2 | 0 | 15 | 3 | 1 | 7 | 0 |
| 8 | Dextran | Dextrose | CL611 | 0 | 5 | 1 | 15 | 240 | 3 | 2 | 8 | 0 |
| Composition | |
| Dextran | 25% (0.74 g) |
| Maltodextrin | 25% (0.74 g) |
| Sorbitol | 5% (0.15 g) |
| Avicel 200 | 10% (0.3 g) |
| Plasticisers (PEG 400: Glycerol, 1:1 w/w) | 20% (0.3 g + 0.3 g) |
| Simil Fungizone (9) | 3 mL (=30 mg AmB + 24.6 mg sodium deoxycholate) |
| Cellulose-derived film formers | 15% (0.09 g HMPC AS + 0.36 g HPC) |
| Methanol | 1.5 mL |
| Properties | |
| Disintegration time | 60 ± 3 s |
| Burst strength | 2190 ± 140 mN mm |
| Overall score of appearance | 4 = Flexibility (1), tackiness (2) and homogeneity (1) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Serrano, D.R.; Fernandez-Garcia, R.; Mele, M.; Healy, A.M.; Lalatsa, A. Designing Fast-Dissolving Orodispersible Films of Amphotericin B for Oropharyngeal Candidiasis. Pharmaceutics 2019, 11, 369. https://doi.org/10.3390/pharmaceutics11080369
Serrano DR, Fernandez-Garcia R, Mele M, Healy AM, Lalatsa A. Designing Fast-Dissolving Orodispersible Films of Amphotericin B for Oropharyngeal Candidiasis. Pharmaceutics. 2019; 11(8):369. https://doi.org/10.3390/pharmaceutics11080369
Chicago/Turabian StyleSerrano, Dolores R., Raquel Fernandez-Garcia, Marta Mele, Anne Marie Healy, and Aikaterini Lalatsa. 2019. "Designing Fast-Dissolving Orodispersible Films of Amphotericin B for Oropharyngeal Candidiasis" Pharmaceutics 11, no. 8: 369. https://doi.org/10.3390/pharmaceutics11080369
APA StyleSerrano, D. R., Fernandez-Garcia, R., Mele, M., Healy, A. M., & Lalatsa, A. (2019). Designing Fast-Dissolving Orodispersible Films of Amphotericin B for Oropharyngeal Candidiasis. Pharmaceutics, 11(8), 369. https://doi.org/10.3390/pharmaceutics11080369