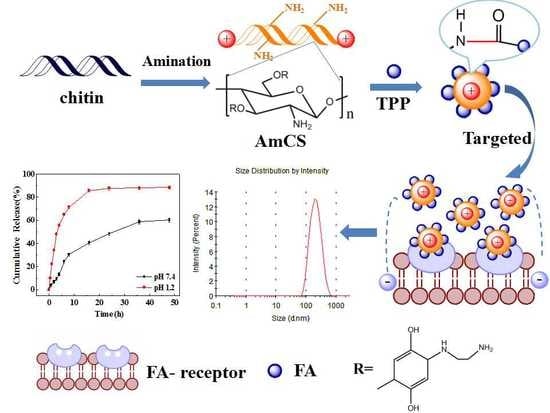
Fabrication of Ion-Crosslinking Aminochitosan Nanoparticles for Encapsulation and Slow Release of Curcumin
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of Aminated Chitosan (AmCS)
2.3. Preparation of Folate-Modified Aminated Chitosan Sodium Tripolyphosphate (FA-AmCS-TPP) Nanoparticles
2.4. Characterization
2.4.1. Particle Size, Polydispersity Index (PDI), and Zeta (ζ)-Potential Measurement
2.4.2. Determination of FA
2.4.3. Drug-Loading Studies
2.4.4. In Vitro Release Studies
2.4.5. Cytotoxicity and Uptake by Tumor Cells
2.4.6. Other Characterization
3. Results and Discussion
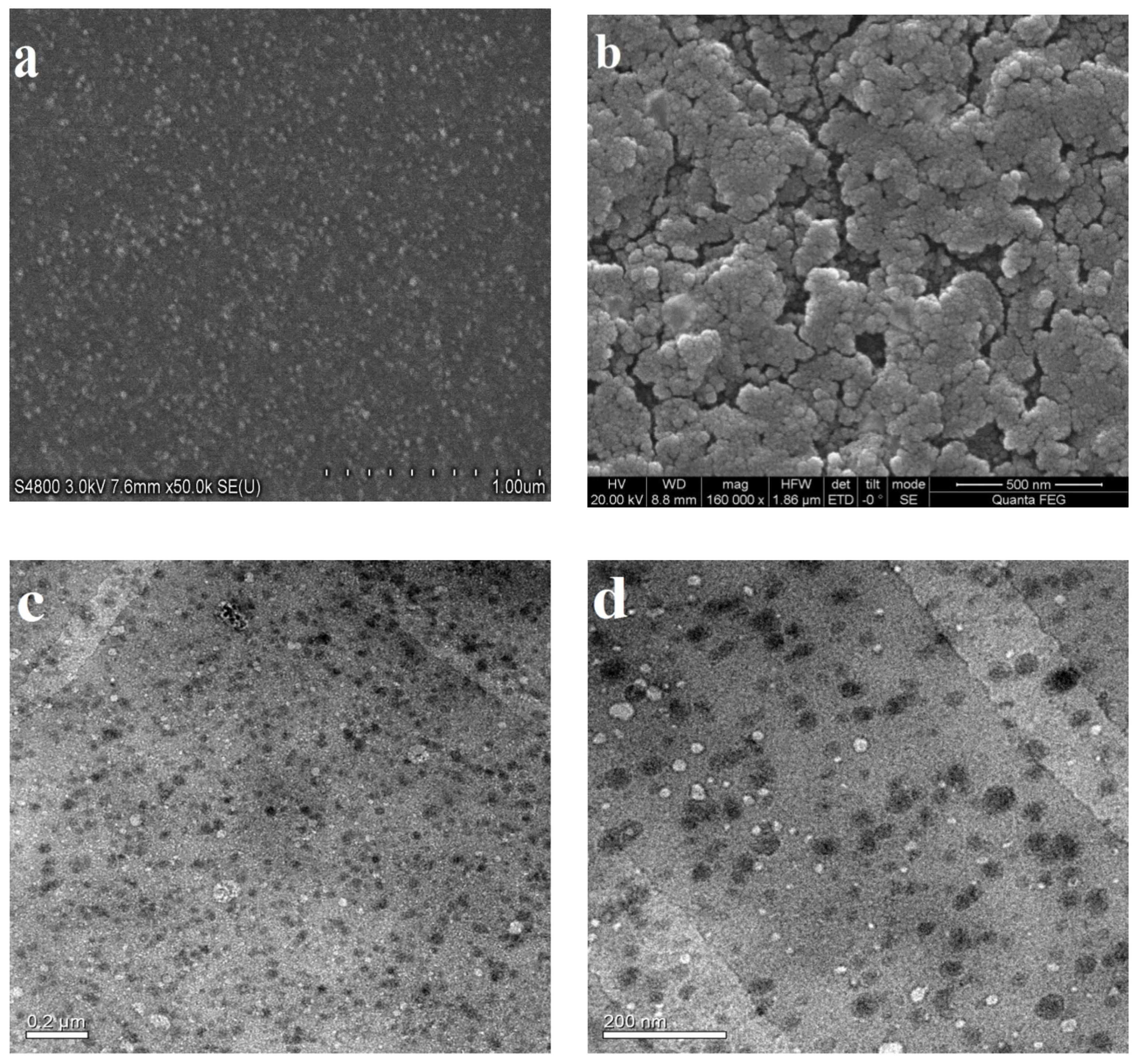
3.1. Analysis of Shape and Appearance
3.2. Fourier Transform Infrared Spectrophotometry (FT-IR)
3.3. Thermogravimetric Analysis (TGA) and Differential Scanning Calorimetry (DSC)
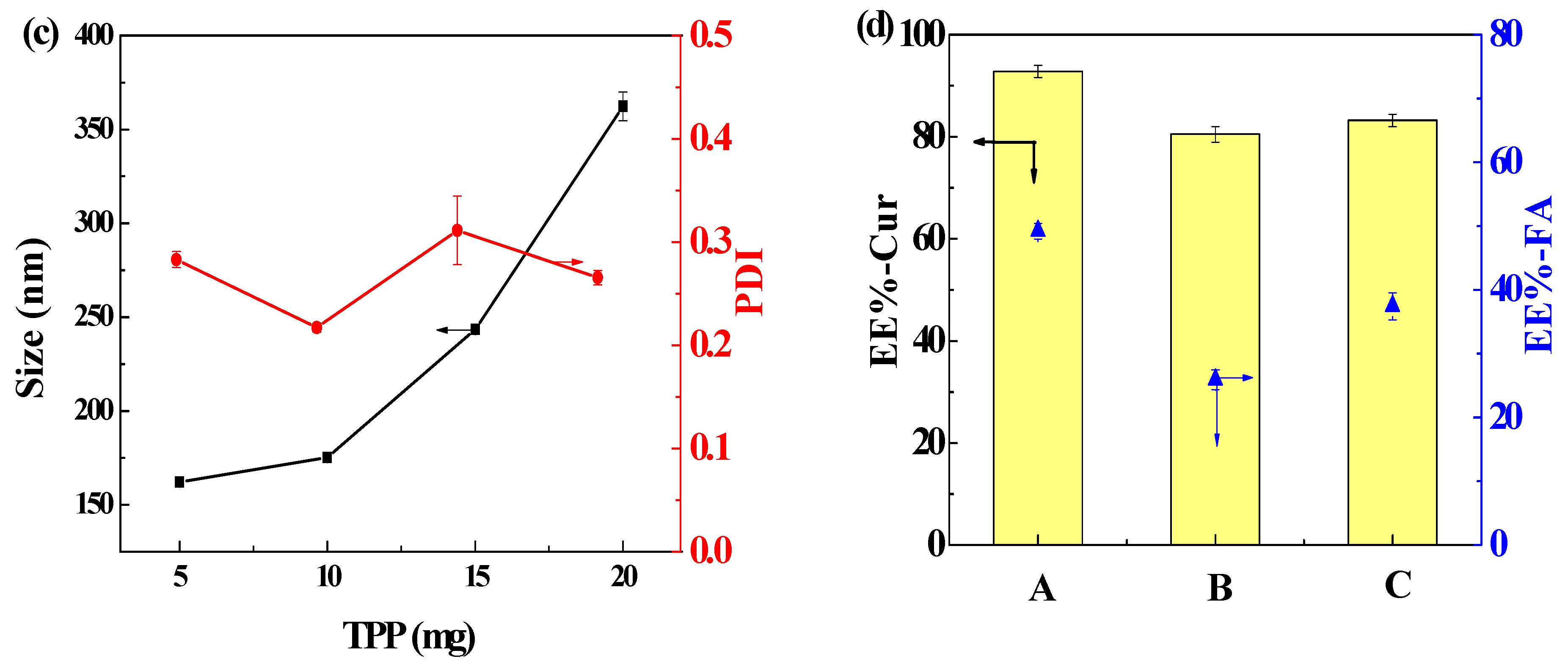
3.4. Particle Size, PDI, and ζ-Potential
3.5. Factors Influencing Cur Encapsulation Efficiency (EE-Cur) and FA Encapsulation Efficiency (EE-FA)
3.5.1. The Effect of Drug Addition Method
3.5.2. The Effect of Drug Dosage
3.5.3. The Effect of Material Concentrations
3.6. In Vitro Release Studies of Cur/FA-AmCS-TPP Nanoparticles
3.7. Cytotoxicity and Uptake by Tumor Cells
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Wang, X.G.; Dong, Z.Y.; Cheng, H.; Wan, S.S.; Chen, W.H.; Zou, M.Z.; Huo, J.W.; Deng, H.X.; Zhang, X.Z. A multifunctional metal–organic framework based tumor targeting drug delivery system for cancer therapy. Nanoscale 2015, 7, 16061–16070. [Google Scholar] [CrossRef]
- Kudgus, R.A.; Walden, C.A.; Mcgovern, R.M.; Reid, J.M.; Robertson, J.D.; Mukherjee, P. Tuning pharmacokinetics and biodistribution of a targeted drug delivery system through incorporation of a passive targeting component. Sci. Rep. 2014, 4, 5669. [Google Scholar] [CrossRef]
- Tamara, F.R.; Lin, C.; Mi, F.L.; Ho, Y.C. Antibacterial effects of chitosan/cationic peptide nanoparticles. Nanomaterials 2018, 8, 88. [Google Scholar] [CrossRef]
- Yoksan, R.; Matsusaki, M.; Akashi, M.; Chirachanchai, S. Controlled hydrophobic/hydrophilic chitosan: Colloidal phenomena and nanosphere formation. Colloid Polym. Sci. 2004, 282, 337–342. [Google Scholar] [CrossRef]
- Liang, X.X.; Wang, N.; Qu, Y.L.; Yang, L.Y.; Wang, Y.G.; Ouyang, X.K. Facile preparation of metal-organic framework (mil-125)/chitosan beads for adsorption of pb(ii) from aqueous solutions. Molecules 2018, 23, 1524. [Google Scholar] [CrossRef]
- Liang, X.X.; Omer, A.M.; Hu, Z.-H.; Wang, Y.-G.; Yu, D.; Ouyang, X.-K. Efficient adsorption of diclofenac sodium from aqueous solutions using magnetic amine-functionalized chitosan. Chemosphere 2019, 217, 270–278. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Shen, J.; Yu, D.; Ouyang, X.K. Preparation of ph-sensitive fe3o4@c/carboxymethyl cellulose/chitosan composite beads for diclofenac sodium delivery. Int. J. Biol. Macromol. 2019, 127, 594–605. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Liu, C.; Omer, A.M.; Lu, W.; Zhang, S.; Jiang, X.; Wu, H.; Yu, D.; Ouyang, X.K. Ph-sensitive zno/carboxymethyl cellulose/chitosan bio-nanocomposite beads for colon-specific release of 5-fluorouracil. Int. J. Biol. Macromol. 2019, 128, 468–479. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Liu, C.; Omer, A.M.; Yang, L.Y.; Ouyang, X.K. Dual-layered ph-sensitive alginate/chitosan/kappa-carrageenan microbeads for colon-targeted release of 5-fluorouracil. Int. J. Biol. Macromol. 2019, 132, 487–494. [Google Scholar] [CrossRef]
- Shentu, J.; Wu, J.; Song, W.; Jia, Z. Chitosan microspheres as immobilized dye affinity support for catalase adsorption. Int. J. Biol. Macromol. 2005, 37, 42–46. [Google Scholar] [CrossRef]
- Carmen, A.L.; Angel, C.; Dubovik, A.S.; Grinberg, N.V.; Burova, T.V.; Valerij Ya, G. Temperature-sensitive chitosan-poly(n-isopropylacrylamide) interpenetrated networks with enhanced loading capacity and controlled release properties. J. Control. Release 2005, 102, 629–641. [Google Scholar]
- Raik, S.V.; Poshina, D.N.; Lyalina, T.A.; Polyakov, D.S.; Vasilyev, V.B.; Kritchenkov, A.S.; Skorik, Y.A. N-[4-(n,n,n-trimethylammonium)benzyl] chitosan chloride: Synthesis, interaction with DNA and evaluation of transfection efficiency. Carbohydr. Polym. 2018, 181, 693–700. [Google Scholar] [CrossRef]
- Sahariah, P.; Óskarsson, B.M.; Hjálmarsdóttir, M.Á.; Másson, M. Synthesis of guanidinylated chitosan with the aid of multiple protecting groups and investigation of antibacterial activity. Carbohydr. Polym. 2015, 127, 407–417. [Google Scholar] [CrossRef]
- Chen, Y.; Li, J.; Li, Q.; Shen, Y.; Ge, Z.; Zhang, W.; Chen, S. Enhanced water-solubility, antibacterial activity and biocompatibility upon introducing sulfobetaine and quaternary ammonium to chitosan. Carbohydr. Polym. 2016, 143, 246–253. [Google Scholar] [CrossRef] [PubMed]
- Hilgenbrink, A.R.; Low, P.S. Folate receptor-mediated drug targeting: From therapeutics to diagnostics. J. Pharm. Sci. 2005, 94, 2135–2146. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Rana, S.; Srivastava, R.S.; Misra, R.D.K. On the chemical synthesis and drug delivery response of folate receptor-activated, polyethylene glycol-functionalized magnetite nanoparticles. Acta Biomater. 2008, 4, 40–48. [Google Scholar] [CrossRef] [PubMed]
- Thomas, T.P.; Majoros, I.J.; Alina, K.; Kukowska-Latallo, J.F.; Anna, B.; Andrzej, M.; Baker, J.R. Targeting and inhibition of cell growth by an engineered dendritic nanodevice. J. Med. Chem. 2005, 48, 3729–3735. [Google Scholar] [CrossRef]
- Leamon, C.P.; Low, P.S. Folate-mediated targeting: From diagnostics to drug and gene delivery. Drug Discov. Today 2001, 6, 44–51. [Google Scholar] [CrossRef]
- Ghaghada, K.B.; Saul, J.; Natarajan, J.V.; Bellamkonda, R.V.; Annapragada, A.V. Folate targeting of drug carriers: A mathematical model. J. Control. Release Off. J. Control. Release Soc. 2005, 104, 113–128. [Google Scholar] [CrossRef] [PubMed]
- Schroeder, J.; Shwek, I.; Shmeeda, H.; Banin, U.; Gabizon, A. Folate-mediated tumor cell uptake of quantum dots entrapped in lipid nanoparticles. J. Control. Release 2007, 124, 28–34. [Google Scholar] [CrossRef] [PubMed]
- Porta, F.; Lamers, G.E.; Morrhayim, J.; Chatzopoulou, A.; Schaaf, M.; Den, D.H.; Backendorf, C.; Zink, J.I.; Kros, A. Folic acid-modified mesoporous silica nanoparticles for cellular and nuclear targeted drug delivery. Adv. Healthc. Mater. 2013, 2, 281–286. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Lin, Y.; Liufu, C.; Tu, J.; Yang, C.; Chen, Z.Y. Research of gene delivery mediated by ultrasound, microbubble and folate-modified chitosan nanoparticles. Curr. Mol. Med. 2018, 18, 383–391. [Google Scholar] [CrossRef] [PubMed]
- Pan, Y.; Li, Y.J.; Zhao, H.Y.; Zheng, J.M.; Xu, H.; Wei, G.; Hao, J.S.; Cui, F.D. Bioadhesive polysaccharide in protein delivery system: Chitosan nanoparticles improve the intestinal absorption of insulin in vivo. Int. J. Pharm. 2002, 249, 139–147. [Google Scholar] [CrossRef]
- Ward, C.M.; Pechar, M.; Oupicky, D.; Ulbrich, K.; Seymour, L.W. Modification of pll/DNA complexes with a multivalent hydrophilic polymer permits folate-mediated targeting in vitro and prolonged plasma circulation in vivo. J. Gene Med. 2002, 4, 536–547. [Google Scholar] [CrossRef]
- Yang, S.J.; Lin, F.H.; Tsai, H.M.; Lin, C.F.; Chin, H.C.; Wong, J.M.; Shieh, M.J. Alginate-folic acid-modified chitosan nanoparticles for photodynamic detection of intestinal neoplasms. Biomaterials 2011, 32, 2174–2182. [Google Scholar] [CrossRef]
- Baroni, P.; Vieira, R.S.; Meneghetti, E.; Da Silva, M.G.C.; Beppu, M.M. Evaluation of batch adsorption of chromium ions on natural and crosslinked chitosan membranes. J. Hazard. Mater. 2008, 152, 1155–1163. [Google Scholar] [CrossRef]
- Miretzky, P.; Cirelli, A.F. Hg(ii) removal from water by chitosan and chitosan derivatives: A review. J. Hazard. Mater. 2009, 167, 10–23. [Google Scholar] [CrossRef]
- Sau, S.; Tatiparti, K.; Alsaab, H.O.; Kashaw, S.K.; Iyer, A.K. A tumor multicomponent targeting chemoimmune drug delivery system for reprograming the tumor microenvironment and personalized cancer therapy. Drug Discov. Today 2018, 23, 1344–1356. [Google Scholar] [CrossRef]
- Campos, A.M.D.; Sánchez, A.; Alonso, M.A.J. Chitosan nanoparticles: A new vehicle for the improvement of the delivery of drugs to the ocular surface. Application to cyclosporin a. Int. J. Pharm. 2001, 224, 159–168. [Google Scholar] [CrossRef]
- Janes, K.A.; Fresneau, M.P.; Marazuela, A.; Fabra, A.; Alonso, M.J. Chitosan nanoparticles as delivery systems for doxorubicin. J. Control. Release 2001, 73, 255–267. [Google Scholar] [CrossRef]
- Dudhani, A.R.; Kosaraju, S.L. Bioadhesive chitosan nanoparticles: Preparation and characterization. Carbohydr. Polym. 2010, 81, 243–251. [Google Scholar] [CrossRef]
- Gan, Q.; Wang, T.; Cochrane, C.; Mccarron, P. Modulation of surface charge, particle size and morphological properties of chitosan–tpp nanoparticles intended for gene delivery. Colloids Surf. B Biointerfaces 2005, 44, 65–73. [Google Scholar] [CrossRef]
- Zhang, L.; Zhu, W.; Yang, C.; Guo, H.; Yu, A.; Ji, J.; Gao, Y.; Sun, M.; Zhai, G. A novel folate-modified self-microemulsifying drug delivery system of curcumin for colon targeting. Int. J. Nanomed. 2012, 7, 151–162. [Google Scholar] [Green Version]
- Sharma, R.A.; Gescher, A.J.; Steward, W.P. Curcumin: The story so far. Eur. J. Cancer 2005, 41, 1955–1968. [Google Scholar] [CrossRef] [PubMed]
- Hatcher, H.; Planalp, R.; Cho, J.; Torti, F.M.; Torti, S.V. Curcumin: From ancient medicine to current clinical trials. Cell. Mol. Life Sci. 2008, 65, 1631–1652. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.; Wang, T.R.; Hu, Q.B.; Zhou, M.Y.; Xue, J.Y.; Luo, Y.C. Pectin coating improves physicochemical properties of caseinate/zein nanoparticles as oral delivery vehicles for curcumin. Food Hydrocoll. 2017, 70, 143–151. [Google Scholar] [CrossRef]
- Pan, K.; Luo, Y.; Gan, Y.; Baek, S.J.; Zhong, Q. Ph-driven encapsulation of curcumin in self-assembled casein nanoparticles for enhanced dispersibility and bioactivity. Soft Matter 2014, 10, 6820–6830. [Google Scholar] [CrossRef] [PubMed]
- Hu, S.; Wang, T.; Fernandez, M.L.; Luo, Y. Development of tannic acid cross-linked hollow zein nanoparticles as potential oral delivery vehicles for curcumin. Food Hydrocoll. 2016, 61, 821–831. [Google Scholar] [CrossRef]
- Basniwal, R.K.; Buttar, H.S.; Jain, V.K.; Jain, N. Curcumin nanoparticles: Preparation, characterization, and antimicrobial study. J. Agric. Food Chem. 2011, 59, 2056–2061. [Google Scholar]
- Manconi, M.; Manca, M.L.; Valenti, D.; Escribano, E.; Hillaireau, H.; Fadda, A.M.; Fattal, E. Chitosan and hyaluronan coated liposomes for pulmonary administration of curcumin. Int. J. Pharm. 2017, 525, 203–210. [Google Scholar] [CrossRef]
- Eldin, M.S.M.; Soliman, E.A.; Hashem, A.I.; Tamer, T.M. Antimicrobial activity of novel aminated chitosan derivatives for biomedical applications. Adv. Polym. Technol. 2012, 31, 414–428. [Google Scholar] [CrossRef]
- Mansouri, S.; Cuie, Y.; Winnik, F.; Shi, Q.; Lavigne, P.; Benderdour, M.; Beaumont, E.; Fernandes, J.C. Characterization of folate-chitosan-DNA nanoparticles for gene therapy. Biomaterials 2006, 27, 2060–2065. [Google Scholar] [CrossRef] [PubMed]
- Dash, M.; Chiellini, F.; Ottenbrite, R.M.; Chiellini, E. Chitosan—A versatile semi-synthetic polymer in biomedical applications. Prog. Polym. Sci. 2011, 36, 981–1014. [Google Scholar] [CrossRef]
- Shu, X.Z.; Zhu, K.J. Controlled drug release properties of ionically cross-linked chitosan beads: The influence of anion structure. Int. J. Pharm. 2002, 233, 217–225. [Google Scholar] [CrossRef]
- Park, C.E.; Park, D.J.; Kim, B.K. Effects of a chitosan coating on properties of retinol-encapsulated zein nanoparticles. Food Sci. Biotechnol. 2015, 24, 1725–1733. [Google Scholar] [CrossRef]
- Luo, Y.; Teng, Z.; Li, Y.; Wang, Q. Solid lipid nanoparticles for oral drug delivery: Chitosan coating improves stability, controlled delivery, mucoadhesion and cellular uptake. Carbohydr. Polym. 2015, 122, 221–229. [Google Scholar] [CrossRef]
- Eldin, M.M.; Kenawy, E.R.; Abdel-Hay, F.I.; Tamer, T.M.; Ibrahim, A.E. Novel aminated chitosan-aromatic aldehydes schiff bases: Synthesis, characterization and bio-evaluation. Int. J. Adv. Res. 2015, 3, 563–572. [Google Scholar]
- Anissian, D.; Ghasemi-Kasman, M.; Khalili-Fomeshi, M.; Akbari, A.; Hashemian, M.; Kazemi, S.; Moghadamnia, A. Piperine-loaded chitosan-stpp nanoparticles reduce neuronal loss and astrocytes activation in chemical kindling model of epilepsy. Int. J. Biol. Macromol. 2018, 107, 973–983. [Google Scholar] [CrossRef] [PubMed]
- Fakhreddin, H.S.; Reza, S.M.; Maryam, N. Chitosan/sodium tripolyphosphate nanoparticles as efficient vehicles for antioxidant peptidic fraction from common kilka. Int. J. Biol. Macromol. 2018, 111, 730–737. [Google Scholar]
- Helene, J.; Anna-Lena, K.N.; Marianne, H. Stability of chitosan nanoparticles cross-linked with tripolyphosphate. Biomacromolecules 2012, 13, 3747–3756. [Google Scholar]
- Sun, C.; Liu, F.; Jie, Y.; Wei, Y.; Fang, Y.; Gao, Y. Physical, structural, thermal and morphological characteristics of zeinquercetagetin composite colloidal nanoparticles. Ind. Crops Prod. 2015, 77, 476–483. [Google Scholar] [CrossRef]
- Ouerghemmi, S.; Dimassi, S.; Tabary, N.; Leclercq, L.; Degoutin, S.; Chai, F.; Pierlot, C.; Cazaux, F.; Ung, A.; Staelens, J.N. Synthesis and characterization of polyampholytic aryl-sulfonated chitosans and their in vitro anticoagulant activity. Carbohydr. Polym. 2018, 196, 8–17. [Google Scholar] [CrossRef] [PubMed]
- Dai, L.; Li, R.; Wei, Y.; Sun, C.; Mao, L.; Gao, Y. Fabrication of zein and rhamnolipid complex nanoparticles to enhance the stability and in vitro release of curcumin. Food Hydrocoll. 2018, 77, 617–628. [Google Scholar] [CrossRef]
- Boksebeld, M.; Kilin, V.; Géloën, A.; Ceccone, G.; Monnier, V. Folate-modified silicon carbide nanoparticles as multiphoton imaging nanoprobes for cancer-cell-specific labeling. RSC Adv. 2018, 7, 27361–27369. [Google Scholar] [CrossRef]
- Das, R.P.; Gandhi, V.V.; Singh, B.G.; Kunwar, A.; Kumar, N.N.; Priyadarsini, K.I. Preparation of albumin nanoparticles: Optimum size for cellular uptake of entrapped drug (curcumin). Colloids Surf. A Physicochem. Eng. Asp. 2019, 567, 86–95. [Google Scholar] [CrossRef]
- Silva, H.D.; Beldíková, E.; Poejo, J.; Abrunhosa, L.; Serra, A.T.; Duarte, C.M.M.; Brányik, T.; Cerqueira, M.A.; Pinheiro, A.C.; Vicente, A.A. Evaluating the effect of chitosan layer on bioaccessibility and cellular uptake of curcumin nanoemulsions. J. Food Eng. 2019, 243, 89–100. [Google Scholar] [CrossRef]
- Lai, W.F.; Shum, H.C. Hypromellose-graft-chitosan and its polyelectrolyte complex as novel systems for sustained drug delivery. ACS Appl. Mater. Interfaces 2015, 7, 10501–10510. [Google Scholar] [CrossRef] [PubMed]
- Varshosaz, J.; Hassanzadeh, F.; Aliabadi, H.S.; Banitalebi, M.; Rostami, M.; Nayebsadrian, M. Novel worm-like amphiphilic micelles of folate-targeted cyclodextrin/retinoic acid for delivery of doxorubicin in kg−1 cells. Colloid Polym. Sci. 2014, 292, 2647–2662. [Google Scholar] [CrossRef]
- Hua, S.; Yang, H.; Wang, W.; Wang, A. Controlled release of ofloxacin from chitosan–montmorillonite hydrogel. Appl. Clay Sci. 2010, 50, 112–117. [Google Scholar] [CrossRef]
- Jhi-Joung, W.; Kuo-Sheng, L.; Sung, K.C.; Chia-Yin, T.; Jia-You, F. Lipid nanoparticles with different oil/fatty ester ratios as carriers of buprenorphine and its prodrugs for injection. Eur. J. Pharm. Sci. 2009, 38, 138–146. [Google Scholar]
- Morgen, M.; Tung, D.; Boras, B.; Miller, W.; Malfait, A.M.; Tortorella, M. Nanoparticles for improved local retention after intra-articular injection into the knee joint. Pharm. Res. 2013, 30, 257–268. [Google Scholar] [CrossRef] [PubMed]
- Chu, X.Y.; Huang, W.; Wang, Y.L.; Meng, L.W.; Chen, L.Q.; Jin, M.J.; Chen, L.; Gao, C.H.; Ge, C.; Gao, Z.G. Improving antitumor outcomes for palliative intratumoral injection therapy through lecithin- chitosan nanoparticles loading paclitaxel-cholesterol complex. Int. J. Nanomed. 2019, 14, 689–705. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Jiahui, Y.U.; Gong, P.; Dongmei, X.U.; Hong, J.; Zhang, C.; Yao, S. Novel core-shell magnetic nanogels synthesized in an emulsion-free aqueous system under uv irradiation for targeted radiopharmaceutical applications. J. Magn. Magn. Mater. 2006, 3, 273–280. [Google Scholar] [CrossRef]
- Atta, A.M.; Al-Lohedan, H.A.; Al-Hussain, S.A. Functionalization of magnetite nanoparticles as oil spill collector. Int. J. Mol. Sci. 2015, 16, 6911–6931. [Google Scholar] [CrossRef]
- Wang, H.; Zheng, L.; Peng, C.; Shen, M.; Shi, X.; Zhang, G. Folic acid-modified dendrimer-entrapped gold nanoparticles as nanoprobes for targeted ct imaging of human lung adencarcinoma. Biomaterials 2013, 34, 470–480. [Google Scholar] [CrossRef]
- Khatik, R.; Dwivedi, P.; Upadhyay, M.; Patel, V.K.; Paliwal, S.K.; Dwivedi, A.K. Toxicological evaluation and targeting tumor cells through folic acid modified guar gum nanoparticles of curcumin. J. Biomater. Tissue Eng. 2014, 4, 143–149. [Google Scholar] [CrossRef]
- Yang, B.; Gao, G.; Liu, L.; Li, Y.; Zhao, L.; Xing, H. Ir-780 loaded folic acid modified chitosan nanoparticles (fasoc-ir780 nps) as a targeting and theranostic agent for breast cancer therapy. J. Biomater. Tissue Eng. 2017, 7, 605–613. [Google Scholar] [CrossRef]
- Fan, W.; Xu, Y.; Li, Z.; Li, Q. Folic acid-modified β-cyclodextrin nanoparticles as drug delivery to load dox for liver cancer therapeutics. Soft Mater. 2019, 17, 437–447. [Google Scholar] [CrossRef]
- Sun, X.; Wang, N.; Yang, L.Y.; Ouyang, X.K.; Huang, F. Folic acid and pei modified mesoporous silica for targeted delivery of curcumin. Pharmaceutics 2019, 11, 430. [Google Scholar] [CrossRef] [PubMed]









| Samples | AmCS (g) | FA (mg) | TPP (mg) |
|---|---|---|---|
| S1 | 0.05 | 10 | 10 |
| S2 | 0.10 | 10 | 10 |
| S3 | 0.15 | 10 | 10 |
| S4 | 0.20 | 10 | 10 |
| S5 | 0.10 | 0 | 10 |
| S6 | 0.10 | 5 | 10 |
| S7 | 0.10 | 15 | 10 |
| S8 | 0.10 | 10 | 5 |
| S9 | 0.10 | 10 | 15 |
| S10 | 0.10 | 10 | 20 |
| Groups | Cur (mg) | EE-Cur (%) | |
|---|---|---|---|
| FA = 5 mg | FA = 10 mg | ||
| 1 | 2 | 73.99 ± 0.84 | 64.30 ± 1.65 |
| 2 | 5 | 90.06 ± 0.93 | 88.23 ± 0.25 |
| 3 | 8 | 93.34 ± 0.14 | 89.79 ± 0.64 |
| 4 | 10 | 94.25 ± 0.68 | 91.36 ± 1.21 |
| 5 | 15 | 97.02 ± 0.65 | 95.82 ± 0.94 |
| 6 | 30 | 98.10 ± 0.55 | 97.71 ± 0.84 |
| 7 | 35 | 91.68 ± 1.11 | 90.25 ± 1.08 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sun, X.; Yu, D.; Ying, Z.; Pan, C.; Wang, N.; Huang, F.; Ling, J.; Ouyang, X.-k. Fabrication of Ion-Crosslinking Aminochitosan Nanoparticles for Encapsulation and Slow Release of Curcumin. Pharmaceutics 2019, 11, 584. https://doi.org/10.3390/pharmaceutics11110584
Sun X, Yu D, Ying Z, Pan C, Wang N, Huang F, Ling J, Ouyang X-k. Fabrication of Ion-Crosslinking Aminochitosan Nanoparticles for Encapsulation and Slow Release of Curcumin. Pharmaceutics. 2019; 11(11):584. https://doi.org/10.3390/pharmaceutics11110584
Chicago/Turabian StyleSun, Xiaoxiao, Dongyan Yu, Zhuoyang Ying, Chuqiao Pan, Nan Wang, Fangfang Huang, Junhong Ling, and Xiao-kun Ouyang. 2019. "Fabrication of Ion-Crosslinking Aminochitosan Nanoparticles for Encapsulation and Slow Release of Curcumin" Pharmaceutics 11, no. 11: 584. https://doi.org/10.3390/pharmaceutics11110584