The Response of Heterotrophic Prokaryote and Viral Communities to Labile Organic Carbon Inputs Is Controlled by the Predator Food Chain Structure
Abstract
:1. Introduction
2. Materials and Methods
2.1. Model Framework
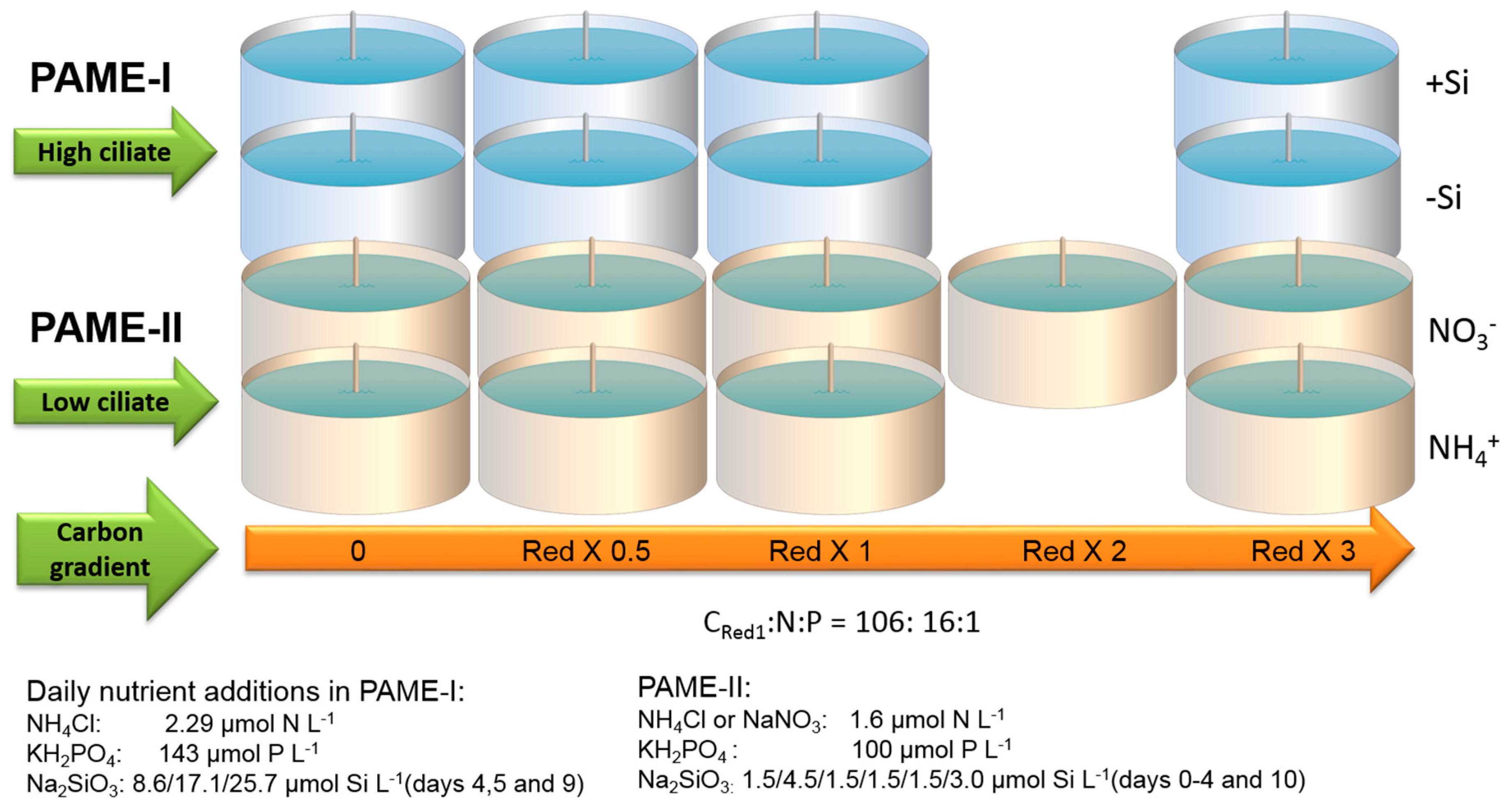
2.2. Experimental Design
2.2.1. Mesocosm Setup
2.2.2. Viral Counts
2.2.3. Viral Concentration and Pulse Field Gel Electrophoresis (PFGE)
2.2.4. DNA Isolation, PCR, and Denaturing Gradient Gel Electrophoresis (DGGE)
2.2.5. Statistical Analysis
3. Results
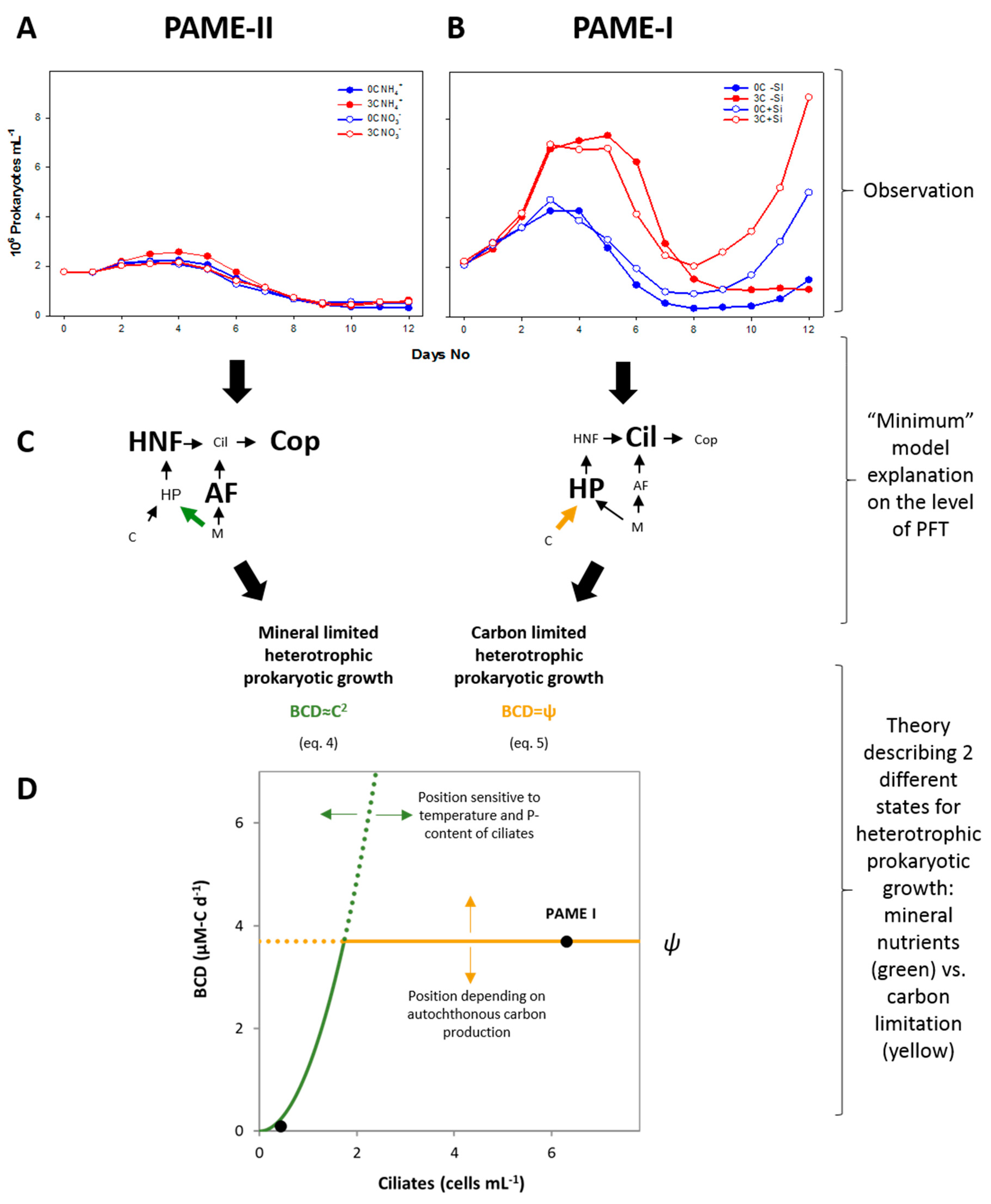
3.1. Theoretical Considerations
3.2. Experimental Results
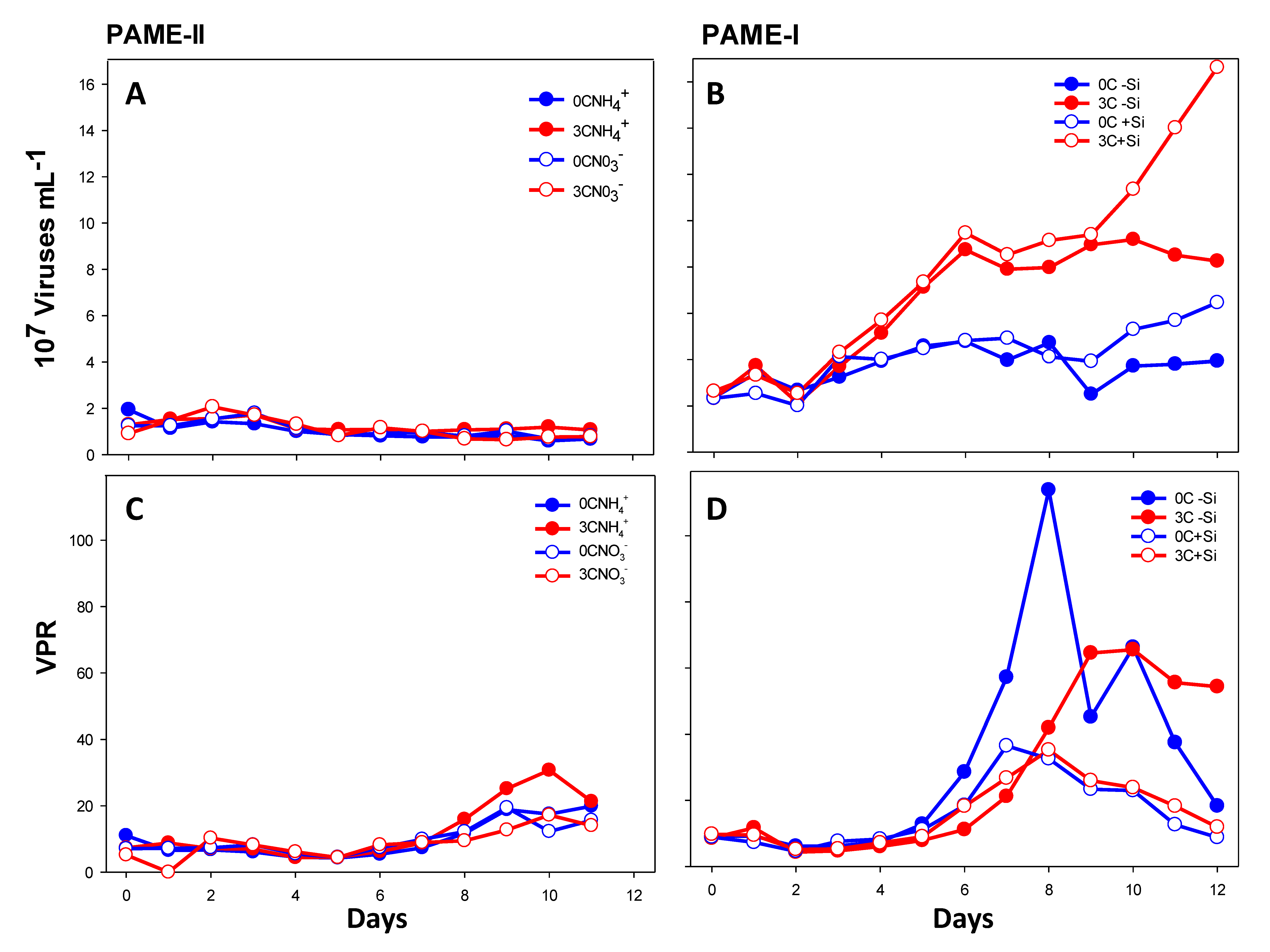
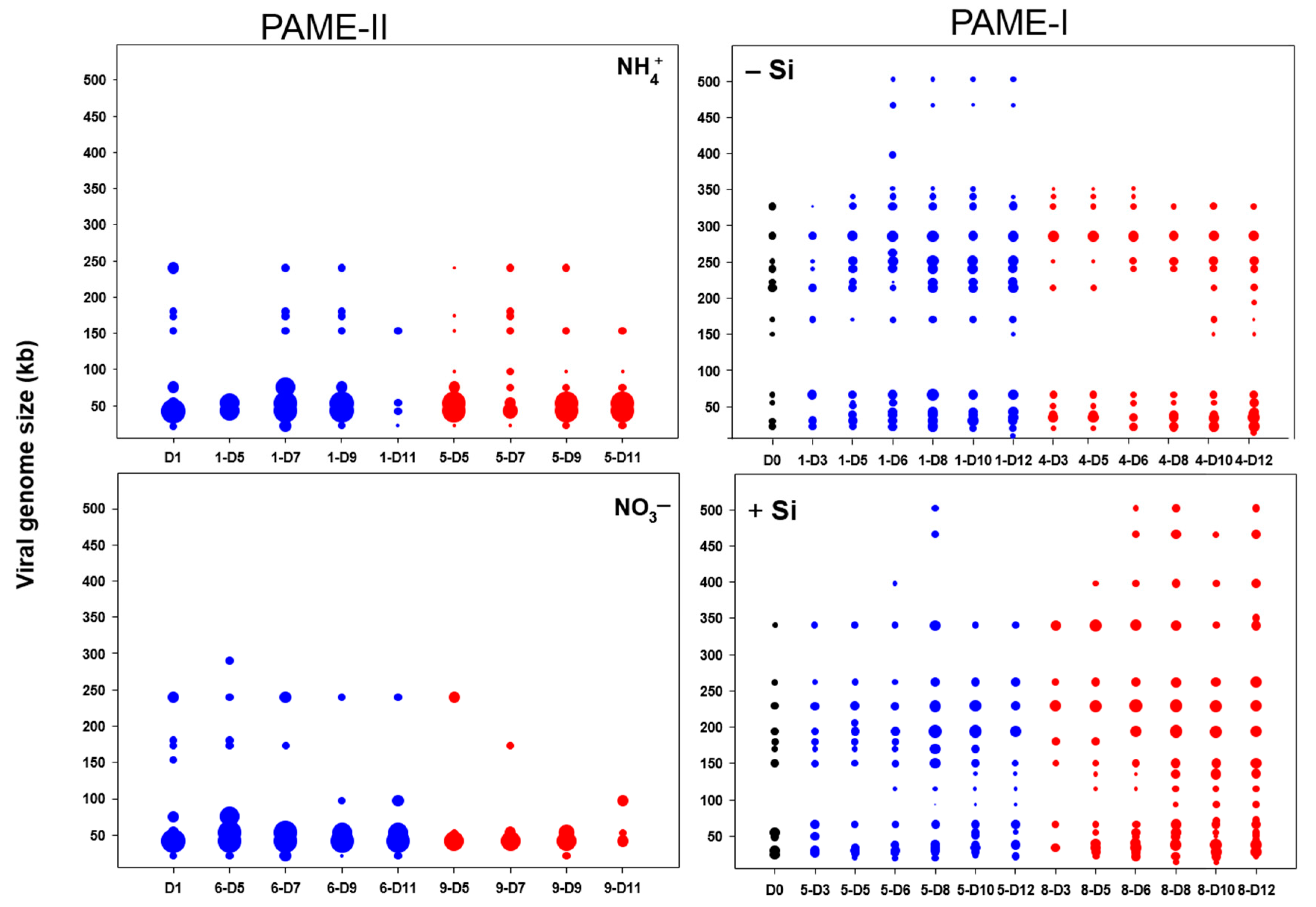
3.2.1. Total Viral Abundance
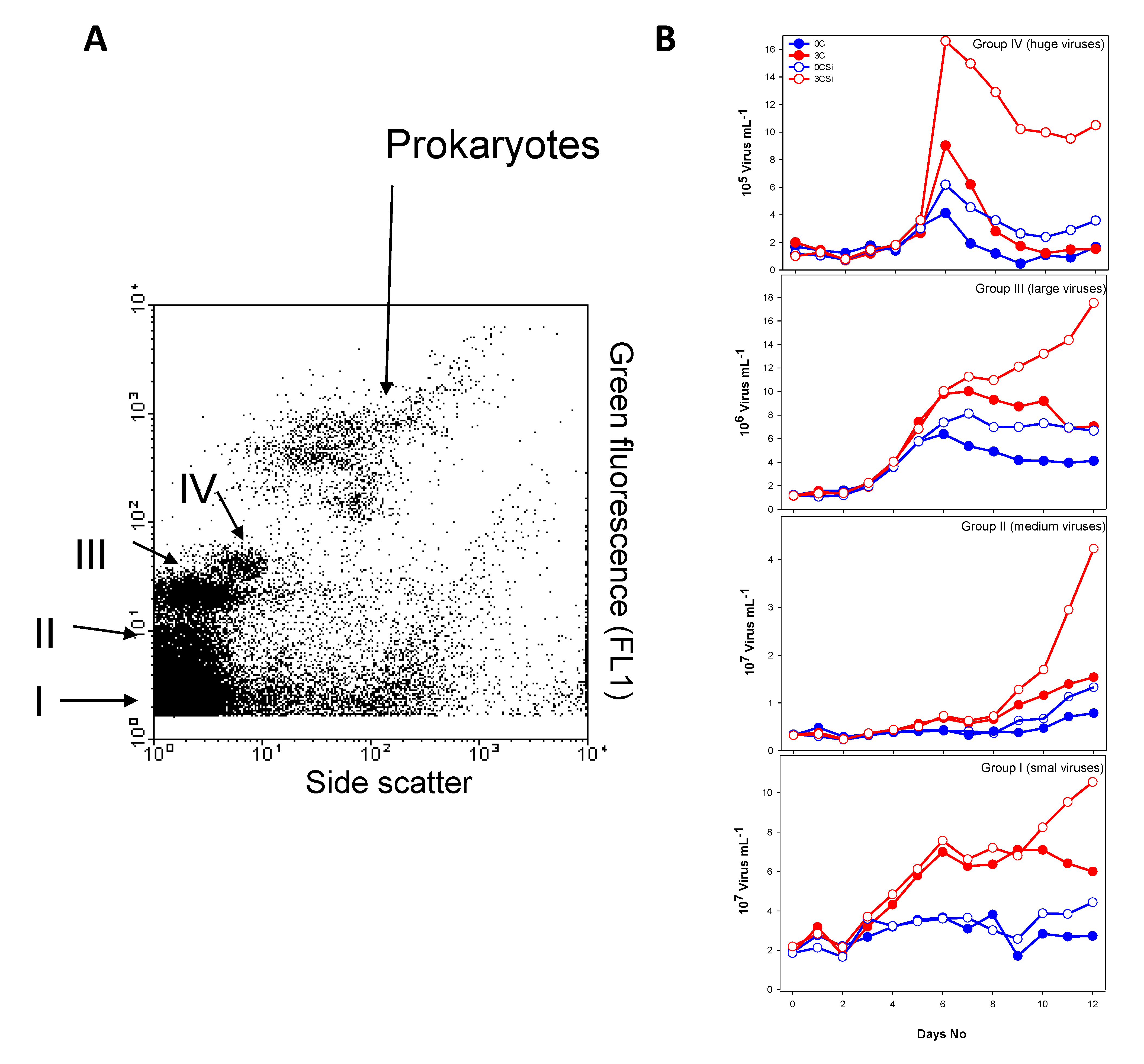
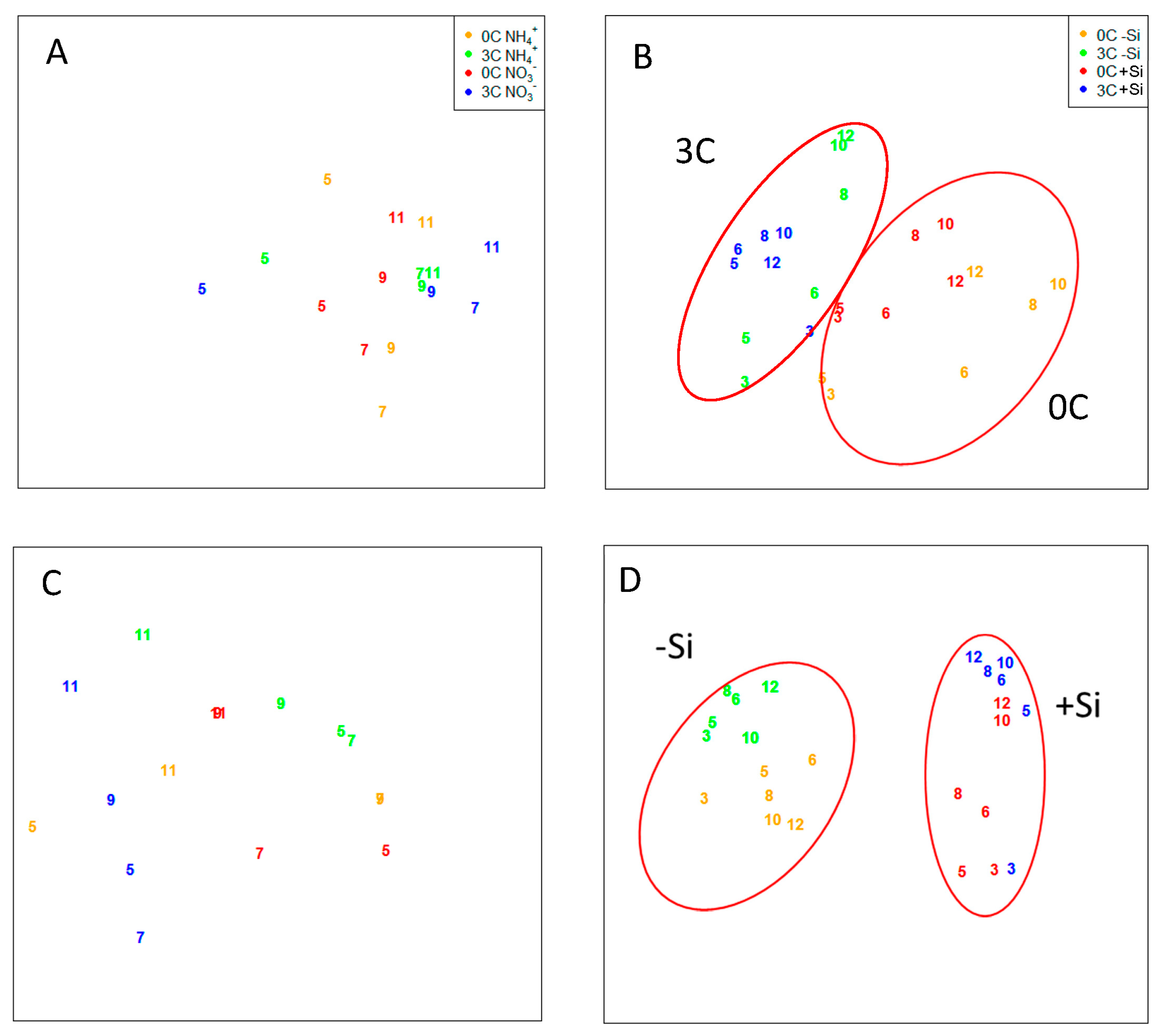
3.2.2. Bacterial and Viral Community Structure
4. Discussion
Supplementary Materials
Acknowledgement
Author Contributions
References
- Zöllner, E.; Hopp, H.-G.; Sommer, U.; Juergens, K. Effect of zooplankton-mediated trophic cascades on marine microbial food web components (bacteria, nanoflagellates, ciliates). Limnol. Oceanogr. 2009, 54, 262–275. [Google Scholar] [CrossRef] [Green Version]
- Bouvier, T.; Del Giorgio, P.A. Key role of selective viral-induced mortality in determining marine bacterial community composition. Environ. Microbiol. 2007, 9, 287–297. [Google Scholar] [CrossRef] [PubMed]
- Sandaa, R.-A.; Gomez-Consarnau, L.; Pinhassi, J.; Riemann, L.; Malits, A.; Weinbauer, M.G.; Gasol, J.M.; Thingstad, T.F. Viruses control of bacterial biodiversity—Linkages between viral and bacterial community structure in a nutrient enriched mesocosm experiment. Environ. Microbiol. 2009, 11, 2585–2595. [Google Scholar] [CrossRef] [PubMed]
- Weinbauer, M.G. Ecology of prokaryotic viruses. FEMS Microbiol. Rev. 2004, 28, 127–181. [Google Scholar] [CrossRef] [PubMed]
- Thingstad, T.F.; Havskum, H.; Zweifel, U.L.; Berdalet, E.; Sala, M.M.; Peters, F.; Alcaraz, M.; Scharek, R.; Perez, M.; Jacquet, S.; et al. Ability of a “minimum” microbial food web model to reproduce response patterns observed in mesocosms manipulated with N and P, glucose, and Si. J. Mar. Syst. 2007, 64, 15–34. [Google Scholar] [CrossRef]
- Beninca, E.; Huisman, J.; Heerkloss, R.; Jöhnk, K.D.; Branco, P.; Van Nes, E.H.; Scheffer, M.; Ellner, S.P. Chaos in a long-term experiment with a plankton community. Nature 2008, 451, 822–825. [Google Scholar] [CrossRef] [PubMed]
- Våge, S.; Pree, B.; Thingstad, T.F. Linking internal and external bacterial community control gives mechanistic framework for pelagic virus-to-bacteria ratios. Environ. Microbiol. 2016, 11, 3932–3948. [Google Scholar] [CrossRef] [PubMed]
- Larsen, A.; Egge, J.K.; Nejstgaard, J.C.; Di Capua, I.; Thyrhaug, R.; Bratbak, G.; Thingstad, T.F. Contrasting response to nutrient manipulation in Arctic mesocosms are reproduced by a minimum microbial food web model. Limnol. Oceanogr. 2015, 60, 360–374. [Google Scholar] [CrossRef] [PubMed]
- Banchetti, R.; Nobili, R.; Esposito, F. An experimentally determined carbon: Volume ratio for marine “oligotrichous” ciliates from estuarine and coastal waters. Limnol. Oceanogr. 1989, 34, 1097–1103. [Google Scholar]
- Thingstad, T.F.; Bellerby, R.G.J.; Bratbak, G.; Borsheim, K.Y.; Egge, J.K.; Heldal, M.; Larsen, A.; Neill, C.; Nejstgaard, J.; Norland, S.; et al. Counterintuitive carbon-to-nutrient coupling in an Arctic pelagic ecosystem. Nature 2008, 455, 387–390. [Google Scholar] [CrossRef] [PubMed]
- Marie, D.; Brussaard, C.P.D.; Thyrhaug, R.; Bratbak, G.; Vaulot, D. Enumeration of marine viruses in culture and natural samples by flow cytometry. Appl. Environ. Microbiol. 1999, 65, 45–52. [Google Scholar] [PubMed]
- Ray, J.; Dondrup, M.; Modha, S.; Steen, I.H.; Sandaa, R.-A.; Clokie, M. Finding a Needle in the Virus Metagenome Haystack-Micro-Metagenome Analysis Captures a Snapshot of the Diversity of a Bacteriophage Armoire. PLoS ONE 2012, 7, e34238. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sandaa, R.-A.; Short, S.M.; Schroeder, D.C. Fingerprinting aquatic virus communities. In Manual of Aquatic Viral Ecology; Wilhelm, S.W., Weinbauer, M.G., Suttle, C.A., Eds.; American Society of Limnology and Oceanography: Wasco, TX, USA, 2010; Volume 2, pp. 9–18. [Google Scholar]
- Töpper, B.; Thingstad, T.F.; Sandaa, R.-A. Effects of differences in organic supply on bacterial diversity subject to viral lysis. FEMS Microbiol. Ecol. 2012, 83, 202–213. [Google Scholar] [CrossRef] [PubMed]
- Töpper, B.; Larsen, A.; Thingstad, T.; Thyrhaug, R.; Sandaa, R.-A. Bacterial community composition in an Arctic phytoplankton mesocosm bloom: The impact of silicate and glucose. Polar Biol. 2010, 33, 1557–1565. [Google Scholar] [CrossRef]
- Oksanen, J.; Blanchet, F.G.; Kindt, R.; Legendre, P.; Minchin, P.R.; O’Hara, R.B.; Simpson, L.G.; Solymos, P.; Stevens, M.H.H.; Wagner, H. Vegan: Community Ecology Package. R Package Version 2.3-0. 2015. Available online: https://cran.r-project.org/package=vegan (accessed on 19 March 2016).
- Larsen, J.B.; Larsen, A.; Thyrhaug, R.; Bratbak, G.; Sandaa, R.-A. Marine viral populations detected during a nutrient induced phytoplankton bloom at elevated pCO2 levels. Biogeosciences 2008, 5, 523–533. [Google Scholar] [CrossRef]
- Ovreas, L.; Bourne, D.; Sandaa, R.A.; Casamayor, E.O.; Benlloch, S.; Goddard, V.; Smerdon, G.; Heldal, M.; Thingstad, T.F. Response of bacterial and viral communities to nutrient manipulations in seawater mesocosms. Aquat. Microb. Ecol. 2003, 31, 109–121. [Google Scholar] [CrossRef]
- Riemann, L.; Steward, G.F.; Azam, F. Dynamics of bacterial community composition and activity during a mesocosm diatom bloom. Appl. Environ. Microbiol. 2000, 66, 578–587. [Google Scholar] [CrossRef] [PubMed]
- Sandaa, R.-A. Burden or benefit? Virus-host interactions in the marine environment. Res. Microbiol. 2008, 159, 374–381. [Google Scholar] [CrossRef] [PubMed]
- Campillo-Balderas, J.A.; Lazcano, A.; Becerra, A. Viral Genome Size Distribution Does not Correlate with the Antiquity of the Host Lineages. Front. Ecol. Evol. 2015, 3. [Google Scholar] [CrossRef]
- Sandaa, R.A.; Heldal, M.; Castberg, T.; Thyrhaug, R.; Bratbak, G. Isolation and characterization of two viruses with large genome size infecting Chrysochromulina ericina (Prymnesiophyceae) and Pyramimonas orientalis (Prasinophyceae). Virology 2001, 290, 272–280. [Google Scholar] [CrossRef] [PubMed]
- Johannessen, T.V.; Bratbak, G.; Larsen, A.; Ogata, H.; Egge, E.S.; Edvardsen, B.; Eikrem, W.; Sandaa, R.-A. Characterisation of three novel giant viruses reveals huge diversity among viruses infecting Prymnesiales (Haptophyta). Virology 2015, 476, 180–188. [Google Scholar] [CrossRef] [PubMed]
- Fischer, M.G.; Allen, M.J.; Wilson, W.H.; Suttle, C.A. Giant virus with a remarkable complement of genes infects marine zooplankton. Proc. Natl. Acad. Sci. USA 2010, 107, 19508–19513. [Google Scholar] [CrossRef] [PubMed]
- Massana, R.; del Campo, J.; Dinter, C.; Sommaruga, R. Crash of a population of the marine heterotrophic flagellate Cafeteria roenbergensis by viral infection. Environ. Microbiol. 2007, 9, 2660–2669. [Google Scholar] [CrossRef] [PubMed]
- Nagasaki, K. Dinoflagellates, diatoms, and their viruses. J. Microbiol. 2008, 46, 235–243. [Google Scholar] [CrossRef] [PubMed]
- Weinbauer, M.G.; Dolan, J.R.; Šimek, K. A population of giant tailed virus-like particles associated with heterotrophic flagellates in a lake-type reservoir. Aquat. Microb. Ecol. 2015, 76, 111–116. [Google Scholar] [CrossRef]
- Vadstein, O. Large variation in growth-limiting factors for marine heterotrophic bacteria in the Arctic waters of Spitsbergen (78 degrees N). Aquat. Microb. Ecol. 2011, 63, 289–297. [Google Scholar] [CrossRef]
- Matsuoka, A.; Babin, M.; Doxaran, D.; Hooker, S.B.; Mitchell, B.G.; Belanger, S.; Bricaud, A. A synthesis of light absorption properti,es of the Arctic Ocean: Application to semianalytical estimates of dissolved organic carbon concentrations from space. Biogeosciences 2014, 11, 3131–3147. [Google Scholar] [CrossRef] [Green Version]
- Falk-Petersen, S.; Mayzaud, P.; Kattner, G.; Sargent, J.R. Lipids and life strategy of Arctic Calanus. Mar. Biol. Res. 2009, 5, 18–39. [Google Scholar] [CrossRef]
| Symbol | Meaning | Numerical Value | Unit |
|---|---|---|---|
| Biomasses | |||
| B | Heterotrophic prokaryotes | nmol-P L−1 | |
| H | Heterotrophic flagellates | nmol-P L−1 | |
| C | Ciliates | nmol-P L−1 | |
| A | Autotrophic flagellates | nmol-P L−1 | |
| P | Free phosphate | nmol-P L−1 | |
| Affinities/clearance rates | Value at 17 °C | ||
| αB | Heterotrophic prokaryote affinity for phosphate | 0.08 | L nmol-P−1h−1 |
| αA | Autotrophic flagellate affinity for phosphate | 0.04 | L nmol-P−1h−1 |
| αH | Heterotrophic flagellate clearance rate for bacteria | 0.0015 | L nmol-P−1h−1 |
| αC | Ciliate clearance rate for flagellates | 0.0005 | L nmol-P−1h−1 |
| Yields | |||
| YH | Heterotrophic flagellate yield on heterotrophic prokaryotes | 0.3 | nmol-P nmol-P−1 |
| YBC | Heterotrophic prokaryote yield on DOC | nmol-P nmol-C−1 | |
| Conversion factor | |||
| α | P per ciliate | 0.00043 | nmol-P cell−1 |
| Temperature sensitivity of α-parameters | |||
| Q10 | 1.3 | dimensionless |
| PAME-I | PAME-II | |||
|---|---|---|---|---|
| 0 × C | 3 × C | 0 × C | 3 × C | |
| Persons coefficient (ρ) | −0.175 | 0.835 | 0.630 | 0.783 |
| p Value | p = 0.678 | p = 0.00982 | p = 0.0281 | p = 0.00258 |
| Treatment | Bacteria Community | Viral Community | |||||
|---|---|---|---|---|---|---|---|
| Experiments | F | R2 | p | F | R2 | p | |
| PAME-I | Glucose | 11.86 | 0.31 | <0.001 | 7.33 | 0.11 | 0.002 |
| Silicate | 5.17 | 0.14 | 0.003 | 38.16 | 0.56 | <0.001 | |
| PAME-II | Glucose | 2.23 | 0.13 | 0.07 | 1.32 | 0.06 | 0.3 |
| Nitrogen | 0.63 | 0.04 | 0.67 | 3.53 | 0.17 | 0.048 | |
| PAME-I | PAME-II | |||||||
|---|---|---|---|---|---|---|---|---|
| Statistics | 0C | 3C | 0CSi | 3CSi | 0CNH4 | 3CNH4 | 0CNO3 | 3CNO3 |
| median p-value * | 0.013 | 0.01 | 0.007 | 0.08 | 0.083 | 0.542 | 0.085 | 0.125 |
| p-values < 0.05 [%] * | 100 | 97.1 | 99.6 | 27.5 | 0 | 0 | 0 | 0 |
| median Procrustes correlation * | 0.852 | 0.878 | 0.906 | 0.723 | 0.963 | 0.491 | 0.858 | 0.945 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sandaa, R.-A.; Pree, B.; Larsen, A.; Våge, S.; Töpper, B.; Töpper, J.P.; Thyrhaug, R.; Thingstad, T.F. The Response of Heterotrophic Prokaryote and Viral Communities to Labile Organic Carbon Inputs Is Controlled by the Predator Food Chain Structure. Viruses 2017, 9, 238. https://doi.org/10.3390/v9090238
Sandaa R-A, Pree B, Larsen A, Våge S, Töpper B, Töpper JP, Thyrhaug R, Thingstad TF. The Response of Heterotrophic Prokaryote and Viral Communities to Labile Organic Carbon Inputs Is Controlled by the Predator Food Chain Structure. Viruses. 2017; 9(9):238. https://doi.org/10.3390/v9090238
Chicago/Turabian StyleSandaa, Ruth-Anne, Bernadette Pree, Aud Larsen, Selina Våge, Birte Töpper, Joachim P. Töpper, Runar Thyrhaug, and Tron Frede Thingstad. 2017. "The Response of Heterotrophic Prokaryote and Viral Communities to Labile Organic Carbon Inputs Is Controlled by the Predator Food Chain Structure" Viruses 9, no. 9: 238. https://doi.org/10.3390/v9090238





