HCIV-1 and Other Tailless Icosahedral Internal Membrane-Containing Viruses of the Family Sphaerolipoviridae
Abstract
:1. Introduction
2. Family Sphaerolipoviridae
3. Life Cycles of Viruses in the Genus Alphasphaerolipovirus
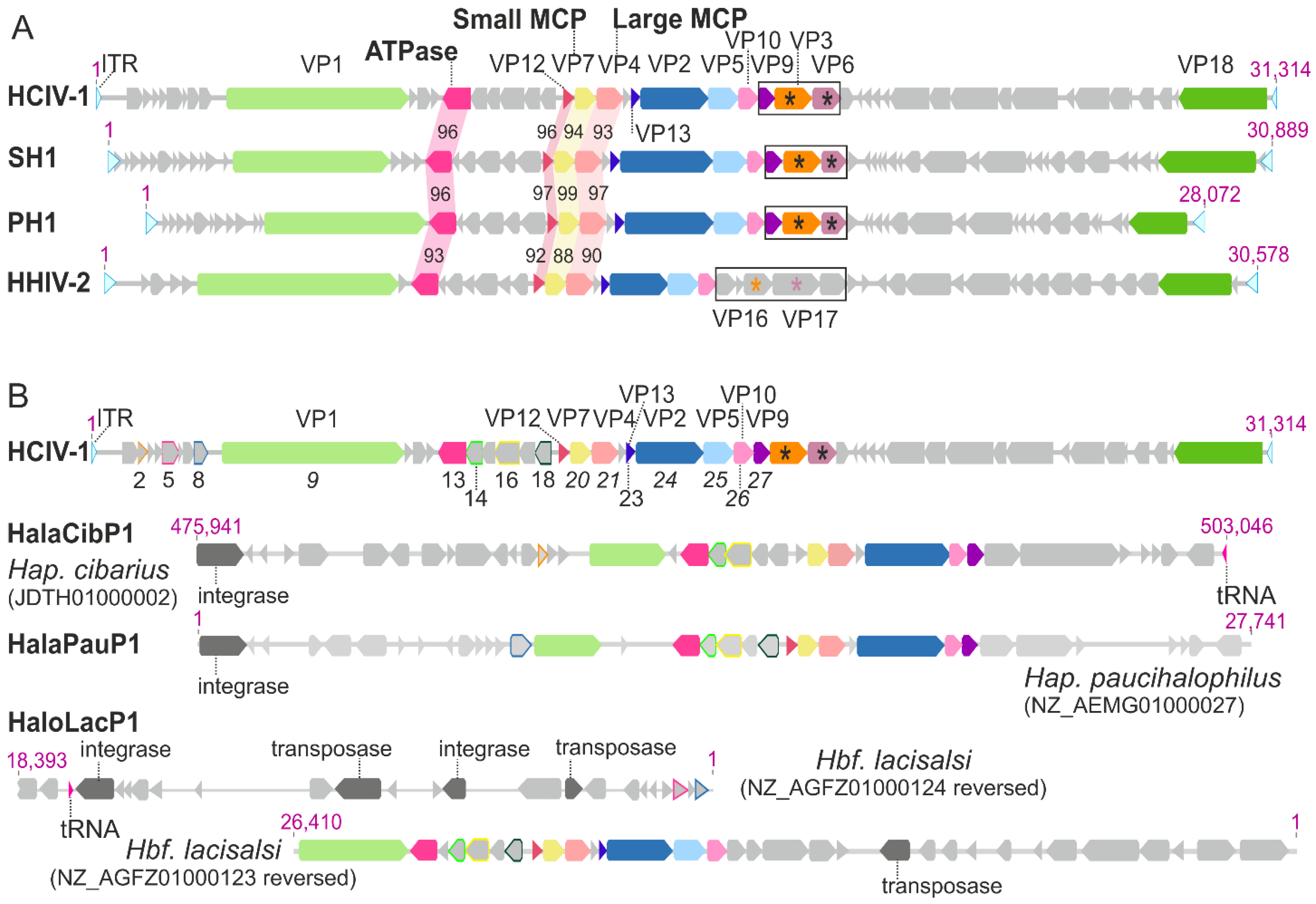
4. Synteny among the Genomes of HCIV-1, SH1, PH1 and HHIV-2
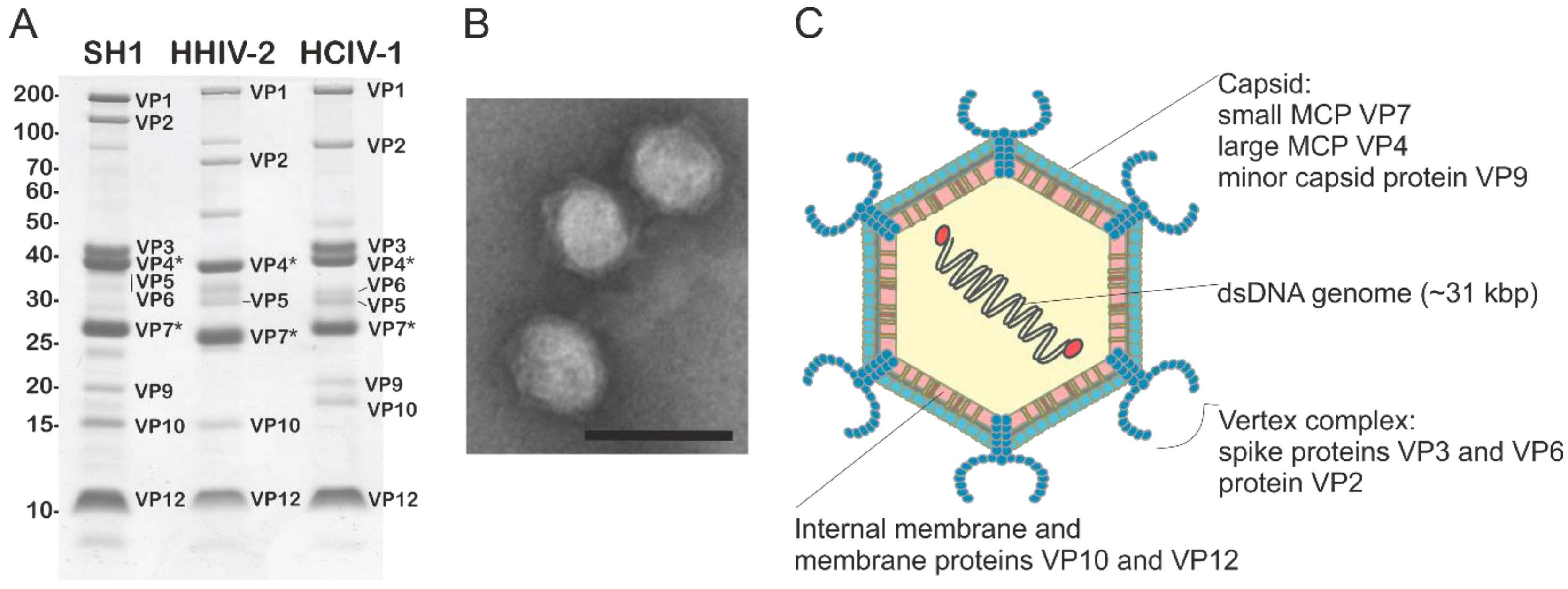
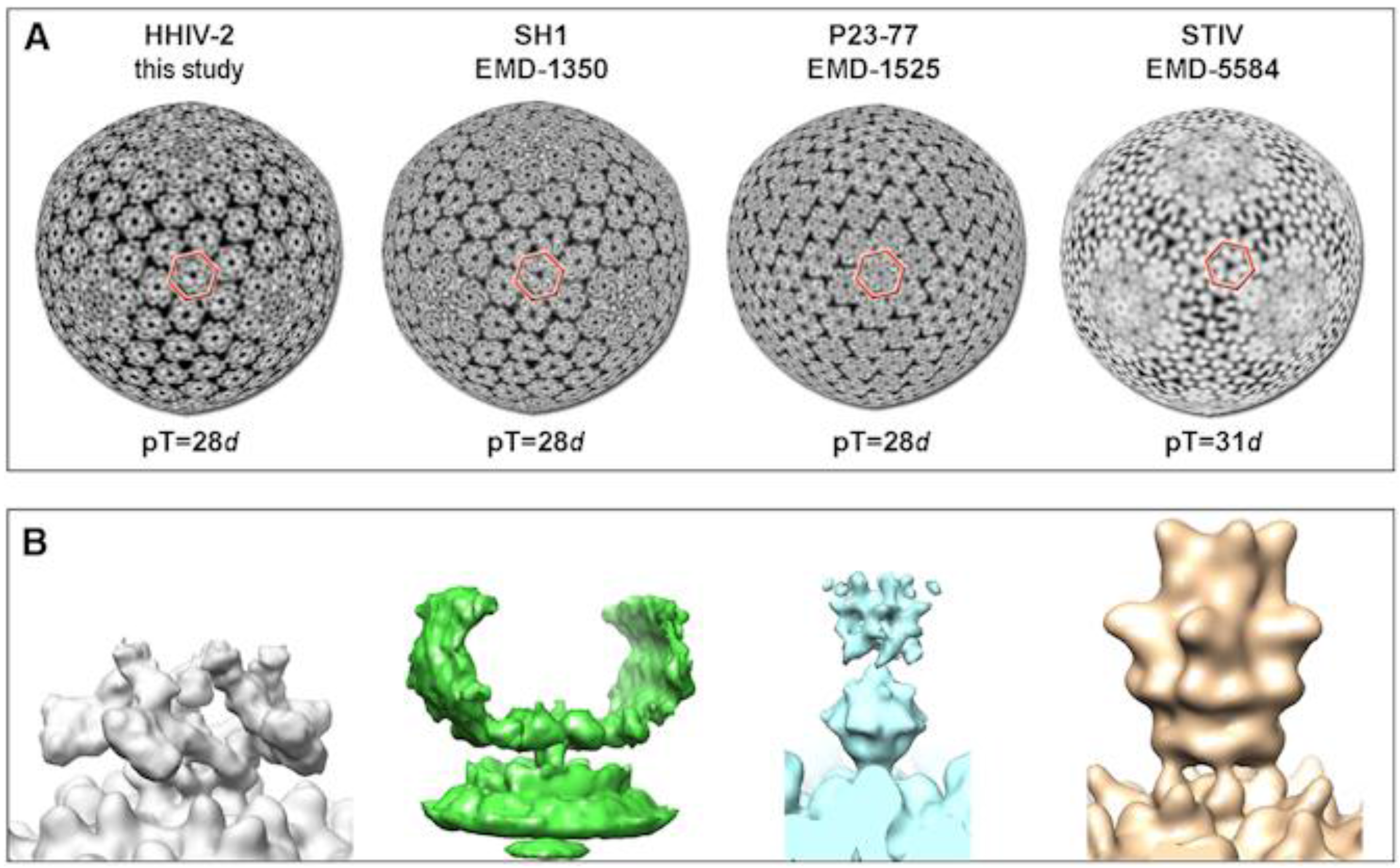
5. Virion Organization of Alphasphaerolipoviruses
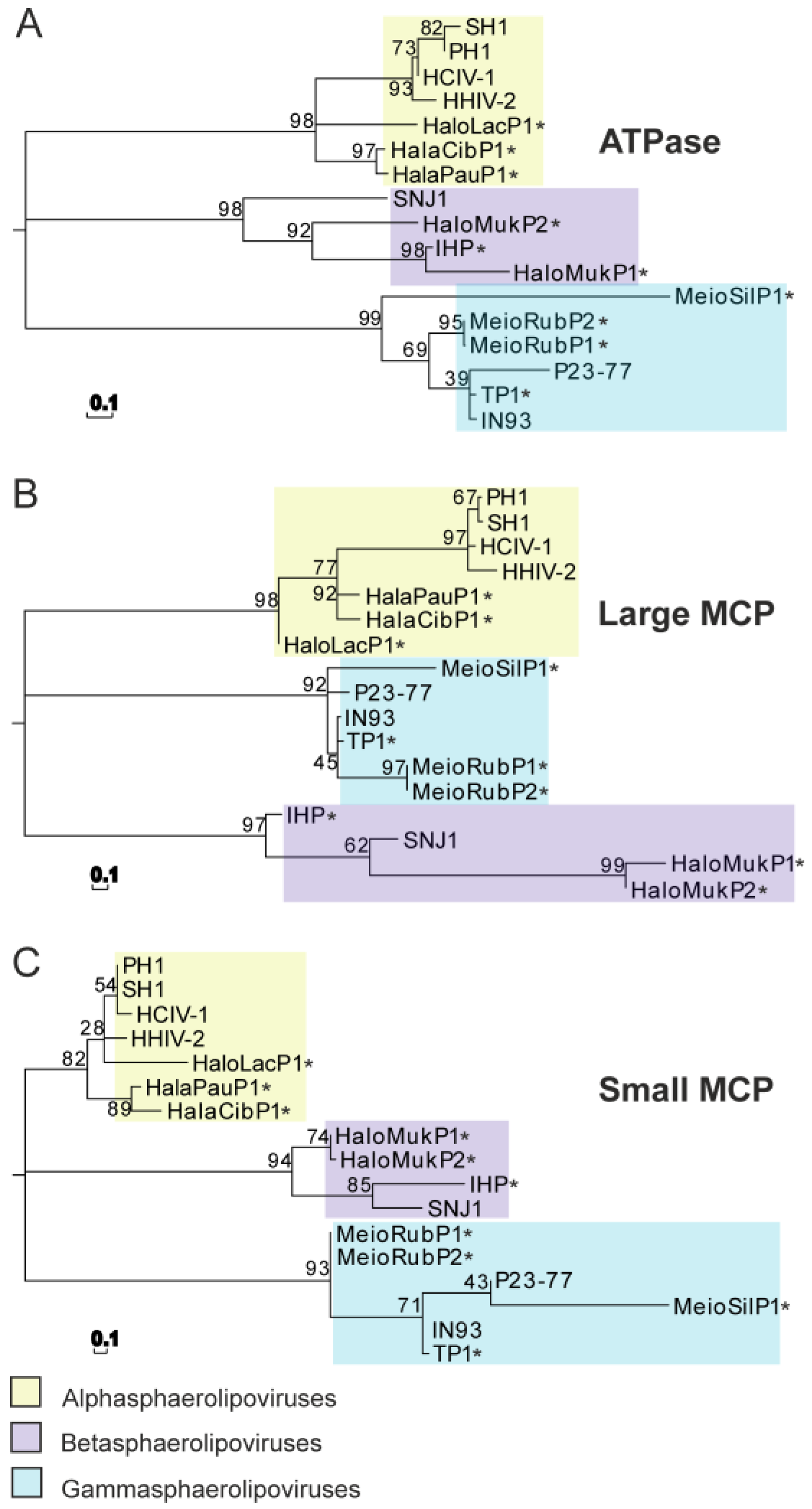
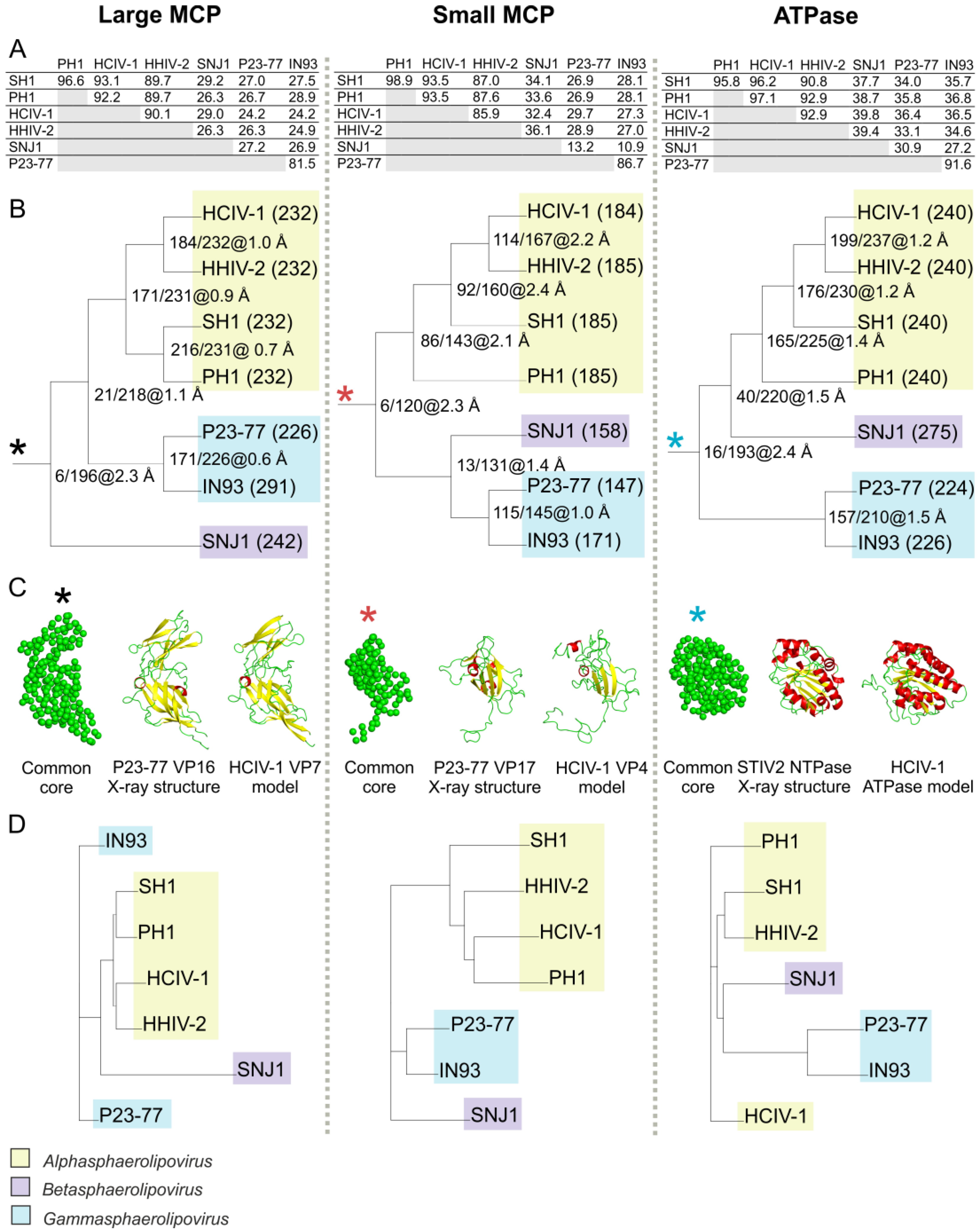
6. Phylogenic Analysis and Structural Clustering of the Virion Core Elements of Sphaerolipoviruses
7. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Disclaimer
References
- Atanasova, N.S.; Bamford, D.H.; Oksanen, H.M. Haloarchaeal Virus Morphotypes. Biochimie 2015, 118, 333–343. [Google Scholar] [CrossRef] [PubMed]
- Prangishvili, D.; Basta, T.; Garrett, R.A.; Krupovič, M. Viruses of the Archaea 2016. In eLS; John Wiley & Sons Ltd.: Chichester; Available online: http://www.els.net (accessed on 1 September 2016). [CrossRef]
- Dellas, N.; Snyder, J.C.; Bolduc, B.; Young, M.J. Archaeal Viruses: Diversity, Replication, and Structure. Annu. Rev. Virol. 2014, 1, 399–426. [Google Scholar] [CrossRef] [PubMed]
- Krupovič, M.; Forterre, P.; Bamford, D.H. Comparative Analysis of the Mosaic Genomes of Tailed Archaeal Viruses and Proviruses Suggests Common Themes for Virion Architecture and Assembly with Tailed Viruses of Bacteria. J. Mol. Biol. 2010, 397, 144–160. [Google Scholar] [CrossRef] [PubMed]
- Krupovič, M.; Bamford, D.H. Archaeal Proviruses TKV4 and MVV Extend the PRD1-Adenovirus Lineage to the Phylum Euryarchaeota. Virology 2008, 375, 292–300. [Google Scholar]
- Gudbergsdóttir, S.R.; Menzel, P.; Krogh, A.; Young, M.; Peng, X. Novel Viral Genomes Identified from Six Metagenomes Reveal Wide Distribution of Archaeal Viruses and High Viral Diversity in Terrestrial Hot Springs. Environ. Microbiol. 2016, 18, 863–874. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Wang, J.; Liu, Y.; Wang, Y.; Zhang, Z.; Oksanen, H.M.; Bamford, D.H.; Chen, X. Identification and Characterization of SNJ2, the First Temperate Pleolipovirus Integrating into the Genome of the SNJ1-Lysogenic Archaeal Strain. Mol. Microbiol. 2015, 98, 1002–1020. [Google Scholar] [CrossRef] [PubMed]
- Bolduc, B.; Shaughnessy, D.P.; Wolf, Y.I.; Koonin, E.V.; Roberto, F.F.; Young, M. Identification of Novel Positive-Strand RNA Viruses by Metagenomic Analysis of Archaea-Dominated Yellowstone Hot Springs. J. Virol. 2012, 86, 5562–5573. [Google Scholar] [CrossRef] [PubMed]
- Pietilä, M.K.; Demina, T.A.; Atanasova, N.S.; Oksanen, H.M.; Bamford, D.H. Archaeal Viruses and Bacteriophages: Comparisons and Contrasts. Trends Microbiol. 2014, 22, 334–344. [Google Scholar] [CrossRef] [PubMed]
- Prangishvili, D. The Wonderful World of Archaeal Viruses. Annu. Rev. Microbiol. 2013, 67, 565–585. [Google Scholar] [CrossRef] [PubMed]
- Happonen, L.J.; Redder, P.; Peng, X.; Reigstad, L.J.; Prangishvili, D.; Butcher, S.J. Familial Relationships in Hyperthermo- and Acidophilic Archaeal Viruses. J. Virol. 2010, 84, 4747–4754. [Google Scholar] [CrossRef] [PubMed]
- Rice, G.; Tang, L.; Stedman, K.; Roberto, F.; Spuhler, J.; Gillitzer, E.; Johnson, J.E.; Douglas, T.; Young, M. The Structure of a Thermophilic Archaeal Virus shows a Double-Stranded DNA Viral Capsid Type that Spans all Domains of Life. Proc. Natl. Acad. Sci. USA 2004, 101, 7716–7720. [Google Scholar] [CrossRef] [PubMed]
- Maaty, W.S.; Ortmann, A.C.; Dlakic, M.; Schulstad, K.; Hilmer, J.K.; Liepold, L.; Weidenheft, B.; Khayat, R.; Douglas, T.; Young, M.J.; et al. Characterization of the Archaeal Thermophile Sulfolobus Turreted Icosahedral Virus Validates an Evolutionary Link among Double-Stranded DNA Viruses from all Domains of Life. J. Virol. 2006, 80, 7625–7635. [Google Scholar] [CrossRef] [PubMed]
- Ortmann, A.C.; Brumfield, S.K.; Walther, J.; McInnerney, K.; Brouns, S.J.; van de Werken, H.J.; Bothner, B.; Douglas, T.; van de Oost, J.; Young, M.J. Transcriptome Analysis of Infection of the Archaeon Sulfolobus Solfataricus with Sulfolobus Turreted Icosahedral Virus. J. Virol. 2008, 82, 4874–4883. [Google Scholar] [CrossRef] [PubMed]
- Khayat, R.; Tang, L.; Larson, E.T.; Lawrence, C.M.; Young, M.; Johnson, J.E. Structure of an Archaeal Virus Capsid Protein Reveals a Common Ancestry to Eukaryotic and Bacterial Viruses. Proc. Natl. Acad. Sci. USA 2005, 102, 18944–18949. [Google Scholar] [CrossRef] [PubMed]
- Fulton, J.; Bothner, B.; Lawrence, M.; Johnson, J.E.; Douglas, T.; Young, M. Genetics, Biochemistry and Structure of the Archaeal Virus STIV. Biochem. Soc. Trans. 2009, 37, 114–117. [Google Scholar] [CrossRef] [PubMed]
- Fu, C.Y.; Johnson, J.E. Structure and Cell Biology of Archaeal Virus STIV. Curr. Opin. Virol. 2012, 2, 122–127. [Google Scholar] [CrossRef] [PubMed]
- Snyder, J.C.; Brumfield, S.K.; Kerchner, K.M.; Quax, T.E.; Prangishvili, D.; Young, M.J. Insights into a Viral Lytic Pathway from an Archaeal Virus-Host System. J. Virol. 2013, 87, 2186–2192. [Google Scholar] [CrossRef] [PubMed]
- Brumfield, S.K.; Ortmann, A.C.; Ruigrok, V.; Suci, P.; Douglas, T.; Young, M.J. Particle Assembly and Ultrastructural Features Associated with Replication of the Lytic Archaeal Virus Sulfolobus Turreted Icosahedral Virus. J. Virol. 2009, 83, 5964–5970. [Google Scholar] [CrossRef] [PubMed]
- Veesler, D.; Ng, T.S.; Sendamarai, A.K.; Eilers, B.J.; Lawrence, C.M.; Lok, S.M.; Young, M.J.; Johnson, J.E.; Fu, C.Y. Atomic Structure of the 75 MDa Extremophile Sulfolobus Turreted Icosahedral Virus Determined by CryoEM and X-Ray Crystallography. Proc. Natl. Acad. Sci. USA 2013, 110, 5504–5509. [Google Scholar] [CrossRef] [PubMed]
- Benson, S.D.; Bamford, J.K.; Bamford, D.H.; Burnett, R.M. Viral Evolution Revealed by Bacteriophage PRD1 and Human Adenovirus Coat Protein Structures. Cell 1999, 98, 825–833. [Google Scholar] [CrossRef]
- Abrescia, N.G.; Grimes, J.M.; Kivelä, H.M.; Assenberg, R.; Sutton, G.C.; Butcher, S.J.; Bamford, J.K.H.; Bamford, D.H.; Stuart, D.I. Insights into Virus Evolution and Membrane Biogenesis from the Structure of the Marine Lipid-Containing Bacteriophage PM2. Mol. Cell 2008, 31, 749–761. [Google Scholar] [CrossRef] [PubMed]
- Rux, J.J.; Kuser, P.R.; Burnett, R.M. Structural and Phylogenetic Analysis of Adenovirus Hexons by use of High-Resolution X-Ray Crystallographic, Molecular Modeling, and Sequence-Based Methods. J. Virol. 2003, 77, 9553–9566. [Google Scholar] [CrossRef] [PubMed]
- Nandhagopal, N.; Simpson, A.A.; Gurnon, J.R.; Yan, X.; Baker, T.S.; Graves, M.V.; Van Etten, J.L.; Rossmann, M.G. The Structure and Evolution of the Major Capsid Protein of a Large, Lipid-Containing DNA Virus. Proc. Natl. Acad. Sci. USA 2002, 99, 14758–14763. [Google Scholar] [CrossRef] [PubMed]
- Bahar, M.W.; Graham, S.C.; Stuart, D.I.; Grimes, J.M. Insights into the Evolution of a Complex Virus from the Crystal Structure of Vaccinia Virus D13. Structure 2011, 19, 1011–1020. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Sun, S.; Xiang, Y.; Wong, J.; Klose, T.; Raoult, D.; Rossmann, M.G. Structure of Sputnik, a Virophage, at 3.5-Å Resolution. Proc. Natl. Acad. Sci. USA 2012, 109, 18431–18436. [Google Scholar] [CrossRef] [PubMed]
- Benson, S.D.; Bamford, J.K.H.; Bamford, D.H.; Burnett, R.M. Does Common Architecture Reveal a Viral Lineage Spanning all Three Domains of Life? Mol. Cell 2004, 16, 673–685. [Google Scholar] [CrossRef] [PubMed]
- Bamford, D.H.; Burnett, R.M.; Stuart, D.I. Evolution of Viral Structure. Theor. Popul. Biol. 2002, 61, 461–470. [Google Scholar] [CrossRef] [PubMed]
- Bamford, D.H. Do Viruses Form Lineages Across Different Domains of Life? Res. Microbiol. 2003, 154, 231–236. [Google Scholar] [CrossRef]
- Abrescia, N.G.; Bamford, D.H.; Grimes, J.M.; Stuart, D.I. Structure Unifies the Viral Universe. Annu. Rev. Biochem. 2012, 81, 795–822. [Google Scholar] [CrossRef] [PubMed]
- Bamford, D.H.; Grimes, J.M.; Stuart, D.I. What does Structure Tell Us about Virus Evolution? Curr. Opin. Struct. Biol. 2005, 15, 655–663. [Google Scholar] [CrossRef] [PubMed]
- Yan, X.; Chipman, P.R.; Castberg, T.; Bratbak, G.; Baker, T.S. The Marine Algal Virus PpV01 has an Icosahedral Capsid with T=219 Quasisymmetry. J. Virol. 2005, 79, 9236–9243. [Google Scholar] [CrossRef] [PubMed]
- Yan, X.; Yu, Z.; Zhang, P.; Battisti, A.J.; Holdaway, H.A.; Chipman, P.R.; Bajaj, C.; Bergoin, M.; Rossmann, M.G.; Baker, T.S. The Capsid Proteins of a Large, Icosahedral dsDNA Virus. J. Mol. Biol. 2009, 385, 1287–1299. [Google Scholar] [CrossRef] [PubMed]
- Oksanen, H.M.; Pietilä, M.K.; Senčilo, A.; Atanasova, N.S.; Roine, E.; Bamford, D.H. Virus universe: Can it be constructed from a limited number of viral architectures. In Viruses: Essential Agents of Life; Witzany, G., Ed.; Springer Science & Business Media: Dordrecht, 2012; pp. 83–105. [Google Scholar]
- Bamford, D.H.; Ravantti, J.J.; Rönnholm, G.; Laurinavičius, S.; Kukkaro, P.; Dyall-Smith, M.; Somerharju, P.; Kalkkinen, N.; Bamford, J.K. Constituents of SH1, a Novel Lipid-Containing Virus Infecting the Halophilic Euryarchaeon Haloarcula Hispanica. J. Virol. 2005, 79, 9097–9107. [Google Scholar] [CrossRef] [PubMed]
- Jäälinoja, H.T.; Roine, E.; Laurinmäki, P.; Kivelä, H.M.; Bamford, D.H.; Butcher, S.J. Structure and Host-Cell Interaction of SH1, a Membrane-Containing, Halophilic Euryarchaeal Virus. Proc. Natl. Acad. Sci. USA 2008, 105, 8008–8013. [Google Scholar] [CrossRef] [PubMed]
- Porter, K.; Kukkaro, P.; Bamford, J.K.H.; Bath, C.; Kivelä, H.M.; Dyall-Smith, M.L.; Bamford, D.H. SH1: A Novel, Spherical Halovirus Isolated from an Australian Hypersaline Lake. Virology 2005, 335, 22–33. [Google Scholar] [CrossRef] [PubMed]
- Jaakkola, S.T.; Penttinen, R.K.; Vilen, S.T.; Jalasvuori, M.; Rönnholm, G.; Bamford, J.K.H.; Bamford, D.H.; Oksanen, H.M. Closely Related Archaeal Haloarcula Hispanica Icosahedral Viruses HHIV-2 and SH1 have Nonhomologous Genes Encoding Host Recognition Functions. J. Virol. 2012, 86, 4734–4742. [Google Scholar] [CrossRef] [PubMed]
- Gil-Carton, D.; Jaakkola, S.T.; Charro, D.; Peralta, B.; Castano-Diez, D.; Oksanen, H.M.; Bamford, D.H.; Abrescia, N.G. Insight into the Assembly of Viruses with Vertical Single Beta-Barrel Major Capsid Proteins. Structure 2015, 23, 1866–1877. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Liu, Y.; Wang, S.; Yang, D.; Cheng, Y.; Hu, J.; Chen, J.; Mei, Y.; Shen, P.; Bamford, D.H.; et al. Temperate Membrane-Containing Halophilic Archaeal Virus SNJ1 has a Circular dsDNA Genome Identical to that of Plasmid pHH205. Virology 2012, 434, 233–241. [Google Scholar] [CrossRef] [PubMed]
- Porter, K.; Tang, S.L.; Chen, C.P.; Chiang, P.W.; Hong, M.J.; Dyall-Smith, M. PH1: An Archaeovirus of Haloarcula Hispanica Related to SH1 and HHIV-2. Archaea 2013, 2013, 456318. [Google Scholar] [CrossRef] [PubMed]
- Demina, T.A.; Pietilä, M.K.; Svirskaite, J.; Ravantti, J.J.; Atanasova, N.S.; Bamford, D.H.; Oksanen, H.M. Archaeal Haloarcula Californiae Icosahedral Virus 1 Highlights Conserved Elements in Icosahedral Membrane-Containing DNA Viruses from Extreme Environments. mBio. 2016, 7, e00699-16. [Google Scholar] [CrossRef] [PubMed]
- Available online: https://talk.ictvonline.org/files/ictv_official_taxonomy_updates_since_the_8th_report/m/prokaryote-official/5210 (accessed on 10 January 2017).
- Pawlowski, A.; Rissanen, I.; Bamford, J.K.H.; Krupovič, M.; Jalasvuori, M. Gammasphaerolipovirus, a Newly Proposed Bacteriophage Genus, Unifies Viruses of Halophilic Archaea and Thermophilic Bacteria within the Novel Family Sphaerolipoviridae. Arch. Virol. 2014, 159, 1541–1554. [Google Scholar] [CrossRef] [PubMed]
- Jaatinen, S.T.; Happonen, L.J.; Laurinmäki, P.; Butcher, S.J.; Bamford, D.H. Biochemical and Structural Characterisation of Membrane-Containing Icosahedral dsDNA Bacteriophages Infecting Thermophilic Thermus Thermophilus. Virology 2008, 379, 10–19. [Google Scholar] [CrossRef] [PubMed]
- Jalasvuori, M.; Jaatinen, S.T.; Laurinavičius, S.; Ahola-Iivarinen, E.; Kalkkinen, N.; Bamford, D.H.; Bamford, J.K.H. The Closest Relatives of Icosahedral Viruses of Thermophilic Bacteria are among Viruses and Plasmids of the Halophilic Archaea. J. Virol. 2009, 83, 9388–9397. [Google Scholar] [CrossRef] [PubMed]
- Matsushita, I.; Yamashita, N.; Yokota, A. Isolation and Characterization of Bacteriophage Induced from a New Isolate of Thermus Aquaticus. Microbiol. Cult. Coll. 1995, 11, 133–138. [Google Scholar]
- Matsushita, I.; Yanase, H. The Genomic Structure of Thermus bacteriophage ϕIN93. J. Biochem. 2009, 146, 775–785. [Google Scholar] [CrossRef] [PubMed]
- Rissanen, I.; Grimes, J.M.; Pawlowski, A.; Mäntynen, S.; Harlos, K.; Bamford, J.K.H.; Stuart, D.I. Bacteriophage P23-77 Capsid Protein Structures Reveal the Archetype of an Ancient Branch from a Major Virus Lineage. Structure 2013, 21, 718–726. [Google Scholar] [CrossRef] [PubMed]
- Yu, M.; Slater, M.; Ackermann, H. Isolation and Characterization of Thermus Bacteriophages. Arch. Virol. 2006, 151, 663–679. [Google Scholar] [CrossRef] [PubMed]
- Kivelä, H.M.; Roine, E.; Kukkaro, P.; Laurinavičius, S.; Somerharju, P.; Bamford, D.H. Quantitative Dissociation of Archaeal Virus SH1 Reveals Distinct Capsid Proteins and a Lipid Core. Virology 2006, 356, 4–11. [Google Scholar] [CrossRef] [PubMed]
- Atanasova, N.S.; Demina, T.A.; Buivydas, A.; Bamford, D.H.; Oksanen, H.M. Archaeal Viruses Multiply: Temporal Screening in a Solar Saltern. Viruses 2015, 7, 1902–1926. [Google Scholar] [CrossRef] [PubMed]
- Atanasova, N.S.; Roine, E.; Oren, A.; Bamford, D.H.; Oksanen, H.M. Global Network of Specific Virus-Host Interactions in Hypersaline Environments. Environ. Microbiol. 2012, 14, 426–440. [Google Scholar] [CrossRef] [PubMed]
- Available online: http://www.freepik.com/free-vector/blue-globes-and-world-map_712559.htm#term=world%20map&page=1&position=17 (accessed on 19 September 2016).
- Porter, K.; Russ, B.E.; Yang, J.; Dyall-Smith, M.L. The Transcription Programme of the Protein-Primed Halovirus SH1. Microbiology 2008, 154, 3599–3608. [Google Scholar] [CrossRef] [PubMed]
- Porter, K.; Dyall-Smith, M.L. Transfection of Haloarchaea by the DNAs of Spindle and Round Haloviruses and the use of Transposon Mutagenesis to Identify Non-Essential Regions. Mol. Microbiol. 2008, 70, 1236–1245. [Google Scholar] [CrossRef] [PubMed]
- Kukkaro, P.; Bamford, D.H. Virus-Host Interactions in Environments with a Wide Range of Ionic Strengths. Environ. Microbiol. Rep. 2009, 1, 71–77. [Google Scholar] [CrossRef] [PubMed]
- Peralta, B.; Gil-Carton, D.; Castano-Diez, D.; Bertin, A.; Boulogne, C.; Oksanen, H.M.; Bamford, D.H.; Abrescia, N.G. Mechanism of Membranous Tunnelling Nanotube Formation in Viral Genome Delivery. PLoS Biol. 2013, 11, e1001667. [Google Scholar] [CrossRef] [PubMed]
- Svirskaitė, J.; Oksanen, H.M.; Daugelavičius, R.; Bamford, D.H. Monitoring Physiological Changes in Haloarchaeal Cell during Virus Release. Viruses 2016, 8. [Google Scholar] [CrossRef]
- Daugelavičius, R.; Bakieṅ, E.; Beržinskieṅ, J.; Bamford, D.H. Binding of Lipophilic Anions to Microbial Cells. Bioelectrochem. Bioenerget. 1997, 42, 263–274. [Google Scholar] [CrossRef]
- Salas, M. Protein-Priming of DNA Replication. Annu. Rev. Biochem. 1991, 60, 39–71. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Yan, R.; Roy, A.; Xu, D.; Poisson, J.; Zhang, Y. The I-TASSER Suite: Protein Structure and Function Prediction. Nat. Methods 2015, 12, 7–8. [Google Scholar] [CrossRef] [PubMed]
- Ravantti, J.; Bamford, D.; Stuart, D.I. Automatic Comparison and Classification of Protein Structures. J. Struct. Biol. 2013, 183, 47–56. [Google Scholar] [CrossRef] [PubMed]
- Rice, P.; Longden, I.; Bleasby, A. EMBOSS: The European Molecular Biology Open Software Suite. Trends Genet. 2000, 16, 276–277. [Google Scholar] [CrossRef]
- Huson, D.H.; Richter, D.C.; Rausch, C.; Dezulian, T.; Franz, M.; Rupp, R. Dendroscope: An Interactive Viewer for Large Phylogenetic Trees. BMC Bioinform. 2007, 8, 1. [Google Scholar] [CrossRef] [PubMed]
- Aalto, A.P.; Bitto, D.; Ravantti, J.J.; Bamford, D.H.; Huiskonen, J.T.; Oksanen, H.M. Snapshot of Virus Evolution in Hypersaline Environments from the Characterization of a Membrane-Containing Salisaeta Icosahedral Phage 1. Proc. Natl. Acad. Sci. USA 2012, 109, 7079–7084. [Google Scholar] [CrossRef] [PubMed]
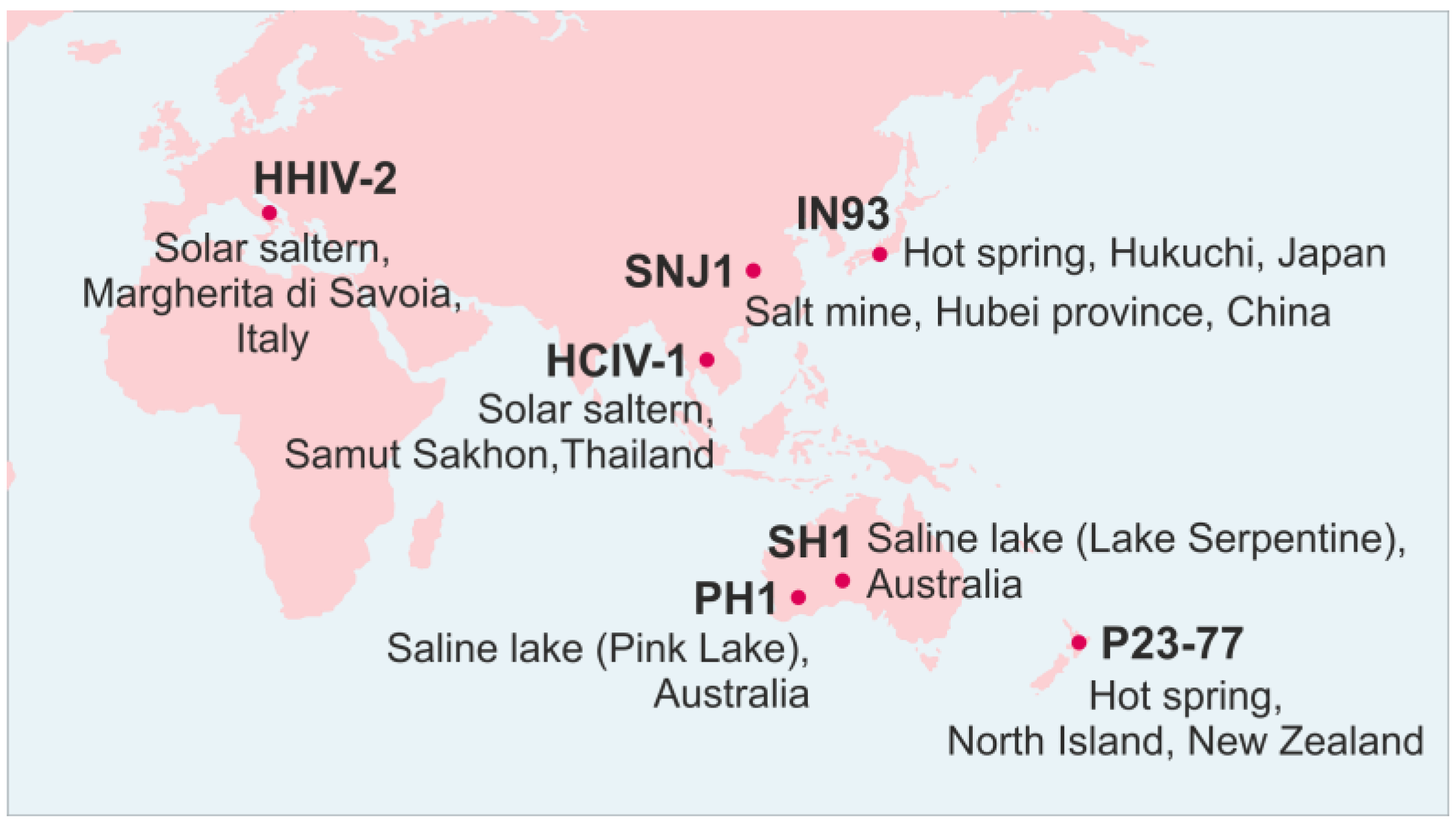
| Genus | Virus species | Virus isolate | Isolation source | Isolation host | Life cycle | Virion diameter (nm) | T number 2 | Internal membrane 3 | dsDNA genome 4 | Reference |
|---|---|---|---|---|---|---|---|---|---|---|
| Alpha-sphaero-lipovirus | Haloarcula hispanica virus SH1 (type species) | SH1 | Saline lake, Australia | Haloarcula hispanica | Lytic | ~80 | T = 28, dextro | Yes | Linear 30,889 bp AY950802 | [35,36,37] |
| Haloarcula hispanica virus HHIV2 | HHIV-2 | Solar saltern, Italy | Haloarcula hispanica | Lytic | ~80 | T = 28, dextro | Yes | Linear 30,578 bp JN968479 | [38,39] | |
| Haloarcula hispanica virus PH1 | PH1 | Saline lake, Australia | Haloarcula hispanica | Lytic | ~50 | nd | nd | Linear 28,072 bp KC252997 | [41] | |
| Haloarcula virus HCIV1 1 | HCIV-1 | Solar saltern, Thailand | Haloarcula californiae | Lytic | ~70 | nd | Yes | Linear 31,314 bp KT809302 | [42,52] | |
| Beta-sphaero-lipovirus | Natrinema virus SNJ1 (type species) | SNJ1 | Salt mine (Natrinema sp. J7-1) | Natrinema sp. J7-2 | Lysogenic | ~70–75 | nd | Yes | Circular 16,341 bp AY048850.1 | [7,40] |
| Gamma-sphaero-lipovirus | Thermus virus P23-77 (type species) | P23-77 | Alkaline hot spring, New Zealand | Thermus thermophilus | Lytic | ~78 | T = 28, dextro | Yes | Circular 17,036 bp GQ403789 | [45,46,49] |
| Thermus virus IN93 | IN93 | Hot spring soil, Japan (Thermus aquaticus TZ2) | Thermus thermophilus | Lysogenic | ~130 | nd | nd | Circular 19,604 bp AB063393 | [47,48] |
| SH1 | PH1 | HHIV-2 | |
|---|---|---|---|
| HCIV-1 | 63.0 | 58.1 | 56.5 |
| SH1 | 100 | 75.5 | 57.9 |
| PH1 | 75.5 | 100 | 56.4 |
| HHIV-2 | 57.9 | 56.4 | 100 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Demina, T.A.; Pietilä, M.K.; Svirskaitė, J.; Ravantti, J.J.; Atanasova, N.S.; Bamford, D.H.; Oksanen, H.M. HCIV-1 and Other Tailless Icosahedral Internal Membrane-Containing Viruses of the Family Sphaerolipoviridae. Viruses 2017, 9, 32. https://doi.org/10.3390/v9020032
Demina TA, Pietilä MK, Svirskaitė J, Ravantti JJ, Atanasova NS, Bamford DH, Oksanen HM. HCIV-1 and Other Tailless Icosahedral Internal Membrane-Containing Viruses of the Family Sphaerolipoviridae. Viruses. 2017; 9(2):32. https://doi.org/10.3390/v9020032
Chicago/Turabian StyleDemina, Tatiana A., Maija K. Pietilä, Julija Svirskaitė, Janne J. Ravantti, Nina S. Atanasova, Dennis H. Bamford, and Hanna M. Oksanen. 2017. "HCIV-1 and Other Tailless Icosahedral Internal Membrane-Containing Viruses of the Family Sphaerolipoviridae" Viruses 9, no. 2: 32. https://doi.org/10.3390/v9020032
APA StyleDemina, T. A., Pietilä, M. K., Svirskaitė, J., Ravantti, J. J., Atanasova, N. S., Bamford, D. H., & Oksanen, H. M. (2017). HCIV-1 and Other Tailless Icosahedral Internal Membrane-Containing Viruses of the Family Sphaerolipoviridae. Viruses, 9(2), 32. https://doi.org/10.3390/v9020032







