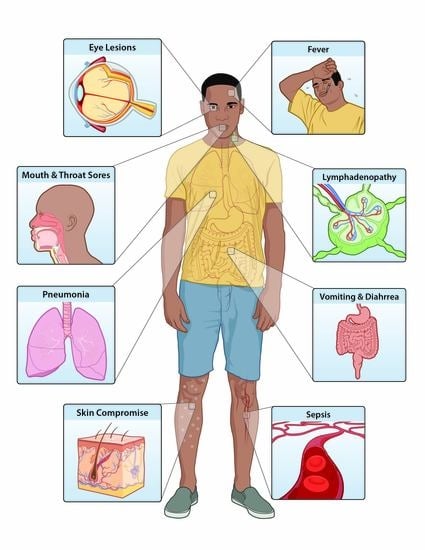
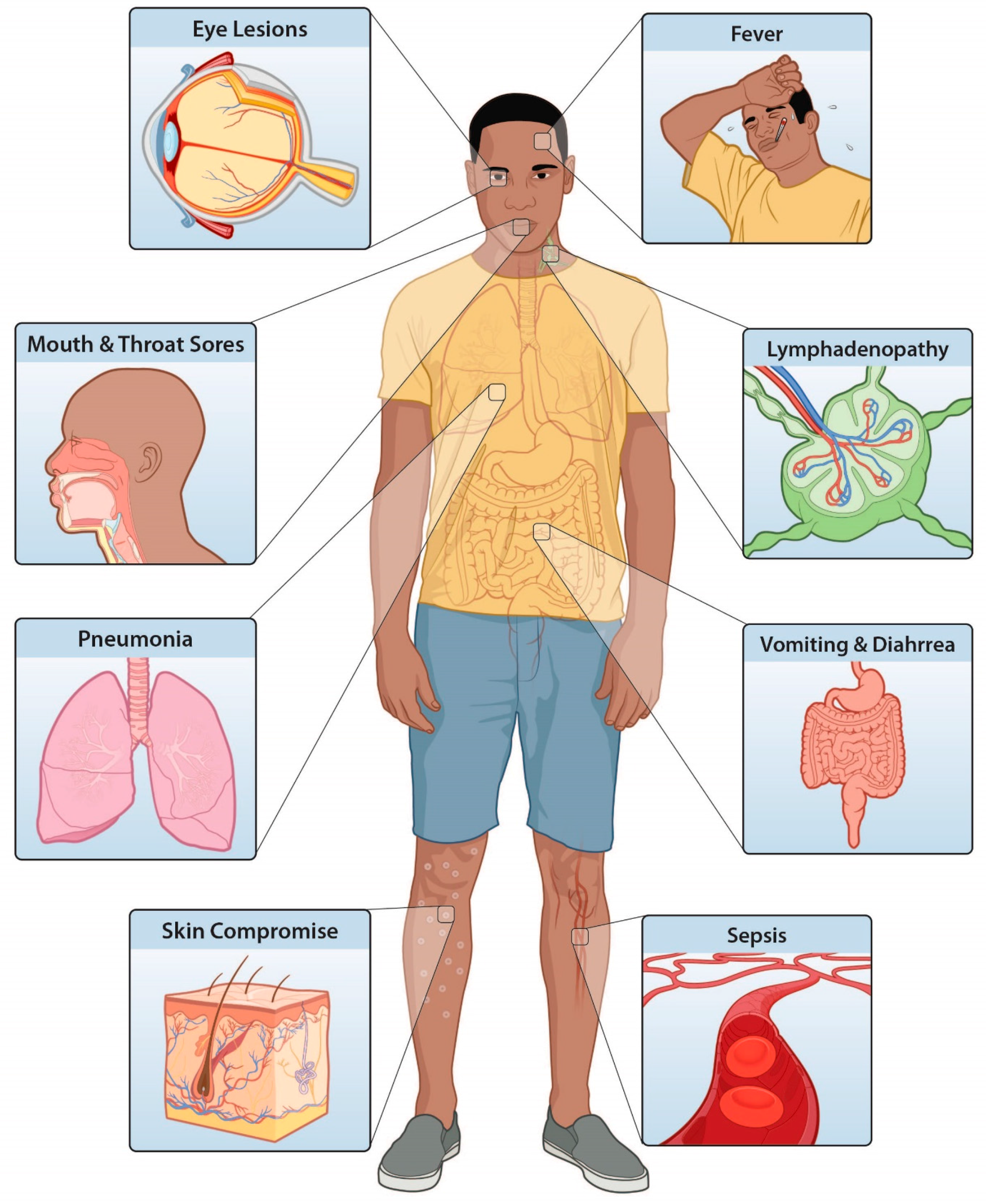
Improving the Care and Treatment of Monkeypox Patients in Low-Resource Settings: Applying Evidence from Contemporary Biomedical and Smallpox Biodefense Research
Abstract
:1. Introduction
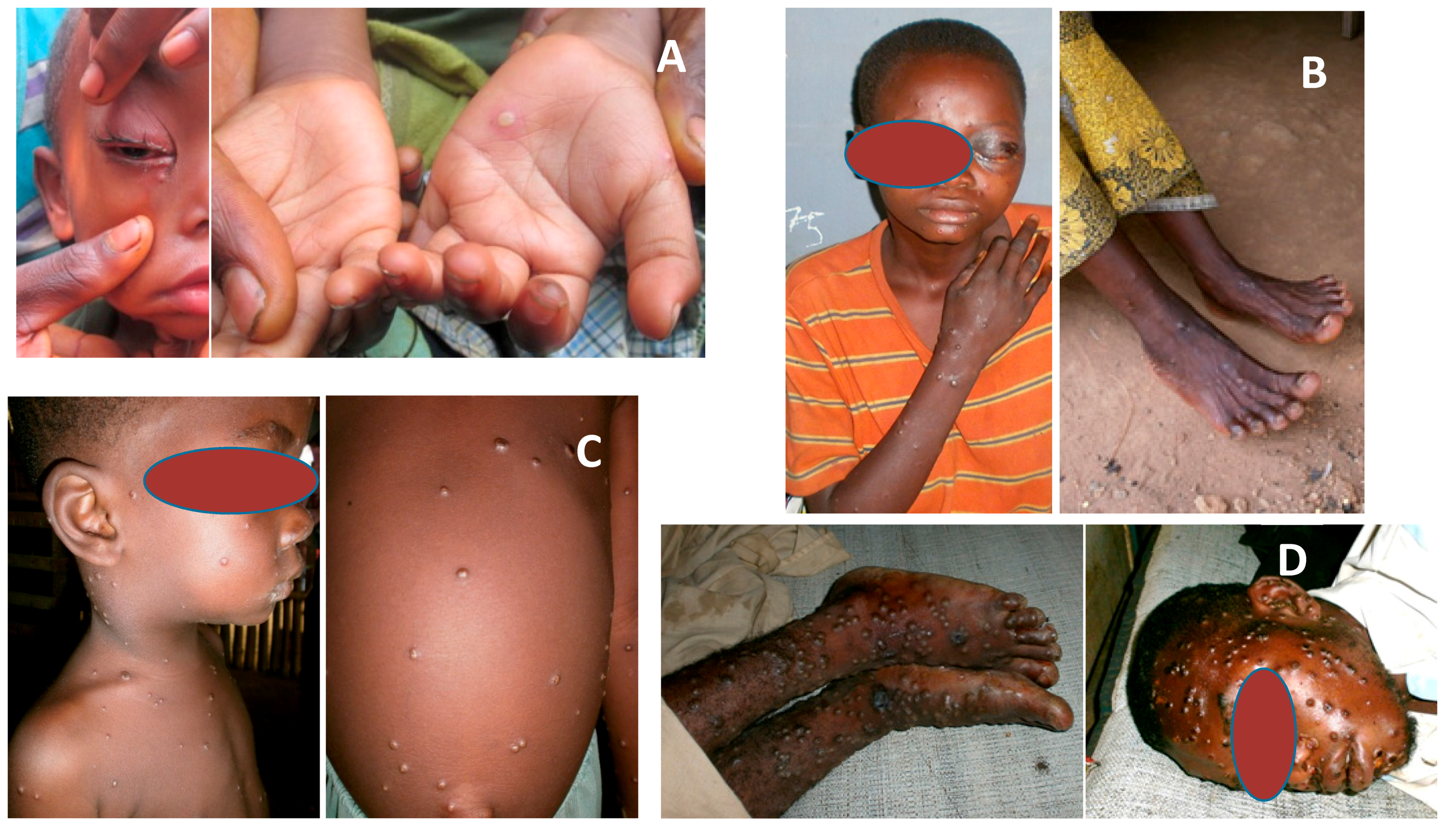
2. Skin
3. Eyes
4. Systemic Illness
5. Bronchopneumonia
6. Optimization of Supportive Care
7. Conclusions
Conflicts of Interest
References
- Khodakevich, L.; Szczeniowski, M.; Nambu, M.D.; Jezek, Z.; Marennikova, S.; Nakano, J.; Meier, F. Monkeypox virus in relation to the ecological features surrounding human settlements in Bumba zone, Zaire. Trop. Geogr. Med. 1987, 39, 56–63. [Google Scholar] [PubMed]
- Khodakevich, L.; Jezek, Z.; Messinger, D. Monkeypox virus: Ecology and public health significance. Bull. World Health Organ. 1988, 66, 747–752. [Google Scholar] [PubMed]
- McCollum, A.M.; Nakazawa, Y.; Ndongala, G.M.; Pukuta, E.; Karhemere, S.; Lushima, R.S.; llunga, B.K.; Kabamba, J.; Wikins, K.; Gao, J.; et al. Human Monkeypox in the Kivus, a Conflict Region of the Democratic Republic of the Congo. Am. J. Trop. Med. Hyg. 2015, 93, 718–721. [Google Scholar] [CrossRef] [PubMed]
- Rimoin, A.W.; Kisalu, N.; Kebela-Ilunga, B.; Mukaba, T.; Wright, L.L.; Formenty, P.; Wolfe, N.D.; Shongo, R.L.; Tshioko, F.; Okitolonda, E.; et al. Endemic human monkeypox, Democratic Republic of Congo, 2001–2004. Emerg. Infect. Dis. 2007, 13, 934–937. [Google Scholar] [CrossRef] [PubMed]
- Rimoin, A.W.; Mulembakani, P.M.; Johnston, S.C.; Lloyd Smith, J.O.; Kisalu, N.K.; Kinkela, T.L.; Blumberg, S.; Thomassen, H.A.; Pike, B.L.; Fair, J.N.; et al. Major increase in human monkeypox incidence 30 years after smallpox vaccination campaigns cease in the Democratic Republic of Congo. Proc. Natl. Acad. Sci. USA 2010, 107, 16262–16267. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, M.G.; Damon, I.K. Outbreaks of human monkeypox after cessation of smallpox vaccination. Trends Microbiol. 2012, 20, 80–87. [Google Scholar] [CrossRef] [PubMed]
- Berthet, N.; Nakoune, E.; Whist, E.; Selekon, B.; Burguiere, A.M.; Manuguerra, J.C.; Gessain, A.; Kazanji, M. Maculopapular lesions in the Central African Republic. Lancet 2011, 378, 1354. [Google Scholar] [CrossRef]
- Kalthan, E.; Dondo-Fongbia, J.P.; Yambele, S.; Dieu-Creer, L.R.; Zepio, R.; Pamatika, C.M. Twelve cases of monkeypox virus outbreak in Bangassou District (Central African Republic) in December 2015. Bull. Soc. Pathol. Exot. 2016, 109, 358–363. [Google Scholar] [CrossRef] [PubMed]
- Khodakevich, L.; Widy-Wirski, R.; Arita, I.; Marennikova, S.S.; Nakano, J.; Meunier, D. Monkey pox virus infection in humans in the Central African Republic. Bull. Soc. Pathol. Exot. Fil. 1985, 78, 311–320. [Google Scholar]
- Chen, N.; Li, G.; Liszewski, M.K.; Atkinson, J.P.; Jahrling, P.B.; Feng, Z.; Schriewer, J.; Buck, C.; Wang, C.; Lefkowitz, E.J.; et al. Virulence differences between monkeypox virus isolates from West Africa and the Congo basin. Virology 2005, 340, 46–63. [Google Scholar] [CrossRef] [PubMed]
- Likos, A.M.; Sammons, S.A.; Olson, V.A.; Frace, A.M.; Li, Y.; Olsen-Rasmussen, M.; Davidson, W.; Galloway, R.; Khristova, M.L.; Reynolds, M.G.; et al. A tale of two clades: Monkeypox viruses. J. Gen. Virol. 2005, 86 Pt 10, 2661–2672. [Google Scholar] [CrossRef] [PubMed]
- Huhn, G.D.; Bauer, A.M.; Yorita, K.; Graham, M.B.; Sejvar, J.; Likos, A.; Damon, I.K.; Reynolds, M.G.; Kuehnert, M.J. Clinical characteristics of human monkeypox, and risk factors for severe disease. Clin. Infect. Dis. 2005, 41, 1742–1751. [Google Scholar] [CrossRef] [PubMed]
- Reed, K.D.; Melski, J.W.; Graham, M.B.; Regnery, R.L.; Sotir, M.J.; Wegner, M.V.; Kazmierczak, J.J.; Stratman, E.J.; Li, Y.; Fairley, J.A.; et al. The detection of monkeypox in humans in the Western Hemisphere. N. Engl. J. Med. 2004, 350, 342–350. [Google Scholar] [CrossRef] [PubMed]
- Jezek, Z.; Szczeniowski, M.; Paluku, K.M.; Mutombo, M. Human monkeypox: Clinical features of 282 patients. J. Infect. Dis. 1987, 156, 293–298. [Google Scholar] [CrossRef] [PubMed]
- Jezek, Z.; Szczeniowski, M.; Paluku, K.M.; Mutombo, M.; Grab, B. Human monkeypox: Confusion with chickenpox. Acta Trop. 1988, 45, 297–307. [Google Scholar] [PubMed]
- Jezek, Z.; Grab, B.; Szczeniowski, M.; Paluku, K.M.; Mutombo, M. Clinico-epidemiological features of monkeypox patients with an animal or human source of infection. Bull. World Health Organ. 1988, 66, 459–464. [Google Scholar] [PubMed]
- Learned, L.A.; Reynolds, M.G.; Wassa, D.W.; Li, Y.; Olson, V.A.; Karem, K.; Stempora, L.L.; Braden, Z.H.; Kline, R.; Likos, A.; et al. Extended Interhuman Transmission of Monkeypox in a Hospital Community in the Republic of the Congo, 2003. Am. J. Trop. Med. Hyg. 2005, 73, 428–434. [Google Scholar] [PubMed]
- Jezek, Z.; Grab, B.; Paluku, K.M.; Szczeniowski, M.V. Human monkeypox: Disease pattern, incidence and attack rates in a rural area of northern Zaire. Trop. Geogr. Med. 1988, 40, 73–83. [Google Scholar] [PubMed]
- Reynolds, M.G.; Emerson, G.L.; Pukuta, E.; Karhemere, S.; Muyembe, J.J.; Bikindou, A.; McCollum, A.M.; Moses, C.; Wilkins, K.; Zhao, H.; et al. Detection of human monkeypox in the Republic of the Congo following intensive community education. Am. J. Trop. Med. Hyg. 2013, 88, 982–985. [Google Scholar] [CrossRef] [PubMed]
- Bass, J.; Tack, D.M.; McCollum, A.M.; Kabamba, J.; Pakuta, E.; Malekani, J.; Nguete, B.; Monroe, B.P.; Doty, J.B.; Karhemere, S.; et al. Enhancing health care worker ability to detect and care for patients with monkeypox in the Democratic Republic of the Congo. Int. Health 2013, 5, 237–243. [Google Scholar] [CrossRef] [PubMed]
- Chapman, J.L.; Nichols, D.K.; Martinez, M.J.; Raymond, J.W. Animal models of orthopoxvirus infection. Vet. Pathol. 2010, 47, 852–870. [Google Scholar] [CrossRef] [PubMed]
- Nalca, A.; Livingston, V.A.; Garza, N.L.; Zumbrun, E.E.; Frick, O.M.; Chapman, J.L.; Hartings, J.M. Experimental infection of cynomolgus macaques (Macaca fascicularis) with aerosolized monkeypox virus. PLoS ONE 2010, 5, e12880. [Google Scholar] [CrossRef] [PubMed]
- Zaucha, G.M.; Jahrling, P.B.; Geisbert, T.W.; Swearengen, J.R.; Hensley, L. The pathology of experimental aerosolized monkeypox virus infection in cynomolgus monkeys (Macaca fascicularis). Lab. Investig. 2001, 81, 1581–1600. [Google Scholar] [CrossRef] [PubMed]
- Goff, A.; Mucker, E.; Raymond, J.; Fisher, R.; Bray, M.; Hensley, L.; Paragas, J. Infection of cynomolgus macaques with a recombinant monkeypox virus encoding green fluorescent protein. Arch. Virol. 2011, 156, 1877–1881. [Google Scholar] [CrossRef] [PubMed]
- Stagles, M.J.; Watson, A.A.; Boyd, J.F.; More, I.A.; McSeveney, D. The histopathology and electron microscopy of a human monkeypox lesion. Trans. R. Soc. Trop. Med. Hyg. 1985, 79, 192–202. [Google Scholar] [CrossRef]
- Bayer-Garner, I.B. Monkeypox virus: Histologic, immunohistochemical and electron-microscopic findings. J. Cutan. Pathol. 2005, 32, 28–34. [Google Scholar] [CrossRef] [PubMed]
- Patel, A.R.; Romanelli, P.; Roberts, B.; Kirsner, R.S. Herpes simplex virus: A histopathologic study of the depth of herpetic wounds. Int. J. Dermatol. 2009, 48, 36–40. [Google Scholar] [CrossRef] [PubMed]
- Graef, S.; Kurth, A.; Auw-Haedrich, C.; Plange, N.; Kern, W.V.; Nitsche, A.; Reinhard, T. Clinicopathological findings in persistent corneal cowpox infection. JAMA Ophthalmol. 2013, 131, 1089–1091. [Google Scholar] [CrossRef] [PubMed]
- Pahlitzsch, R.; Hammarin, A.L.; Widell, A. A Case of Facial Cellulitis and Necrotizing Lymphadenitis due to Cowpox Virus Infection. Clin. Infect. Dis. 2006, 43, 737–742. [Google Scholar] [CrossRef] [PubMed]
- Patel, A.R.; Romanelli, P.; Roberts, B.; Kirsner, R.S. Treatment of herpes simplex virus infection: Rationale for occlusion. Adv. Skin Wound Care 2007, 20, 408–412. [Google Scholar] [CrossRef] [PubMed]
- Varman, K.M.; Namias, N.; Schulman, C.I.; Pizano, L.R. Acute generalized pustular psoriasis, von Zumbusch type, treated in the burn unit. A review of clinical features and new therapeutics. Burns 2014, 40, e35–e39. [Google Scholar] [CrossRef] [PubMed]
- Johnston, S.C.; Johnson, J.C.; Stonier, S.W.; Lin, K.L.; Kisalu, N.K.; Hensley, L.E.; Rimoin, A.W. Cytokine modulation correlates with severity of monkeypox disease in humans. J. Clin. Virol. 2015, 63, 42–45. [Google Scholar] [CrossRef] [PubMed]
- Wise, L.M.; Stuart, G.S.; Real, N.C.; Fleming, S.B.; Mercer, A.A. Orf virus IL-10 accelerates wound healing while limiting inflammation and scarring. Wound Repair Regen. 2014, 22, 356–367. [Google Scholar] [CrossRef] [PubMed]
- Zevin, A.S.; Xie, I.Y.; Birse, K.; Arnold, K.; Romas, L.; Westmacott, G.; Novak, R.M.; McCorrister, S.; McKinnon, L.R.; Cohen, C.R.; et al. Microbiome Composition and Function Drives Wound-Healing Impairment in the Female Genital Tract. PLoS Pathog. 2016, 12, e1005889. [Google Scholar] [CrossRef] [PubMed]
- Bras, G. Observations on the formation of smallpox scars. AMA Arch. Pathol. 1952, 54, 149–156. [Google Scholar] [PubMed]
- Regan, T.D.; Norton, S.A. The scarring mechanism of smallpox. J. Am. Acad. Dermatol. 2004, 50, 591–594. [Google Scholar] [CrossRef] [PubMed]
- Semba, R.D. The ocular complications of smallpox and smallpox immunization. Arch. Ophthalmol. 2003, 121, 715–719. [Google Scholar] [CrossRef] [PubMed]
- Hu, G.; Wang, M.J.; Miller, M.J.; Holland, G.N.; Bruckner, D.A.; Civen, R.; Bornstein, L.A.; Mascola, L.; Lovett, M.A.; Mondino, B.J.; et al. Ocular vaccinia following exposure to a smallpox vaccinee. Am. J. Ophthalmol. 2004, 137, 554–556. [Google Scholar] [CrossRef] [PubMed]
- Lewis, F.S.; Norton, S.A.; Bradshaw, R.D.; Lapa, J.; Grabenstein, J.D. Analysis of cases reported as generalized vaccinia during the US military smallpox vaccination program, December 2002 to December 2004. J. Am. Acad. Dermatol. 2006, 55, 23–31. [Google Scholar] [CrossRef] [PubMed]
- Montgomery, J.R.; Carroll, R.B.; McCollum, A.M. Ocular vaccinia: A consequence of unrecognized contact transmission. Mil. Med. 2011, 176, 699–701. [Google Scholar] [CrossRef] [PubMed]
- Altmann, S.; Brandt, C.R.; Murphy, C.J.; Patnaikuni, R.; Takla, T.; Toomey, M.; Nesbit, B.; McIntyre, K.; Covert, J.; Dubielzig, R.; et al. Evaluation of therapeutic interventions for vaccinia virus keratitis. J. Infect. Dis. 2011, 203, 683–690. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, M.G.; Yorita, K.L.; Kuehnert, M.J.; Davidson, W.B.; Huhn, G.D.; Holman, R.C.; Damon, I.K. Clinical manifestations of human monkeypox influenced by route of infection. J. Infect. Dis. 2006, 194, 773–780. [Google Scholar] [CrossRef] [PubMed]
- Goff, A.J.; Chapman, J.; Foster, C.; Wlazlowski, C.; Shamblin, J.; Lin, K.; Kreiselmeier, N.; Mucker, E.; Paragas, J.; Lawler, J.; et al. A novel respiratory model of infection with monkeypox virus in cynomolgus macaques. J. Virol. 2011, 85, 4898–4909. [Google Scholar] [CrossRef] [PubMed]
- Dyall, J.; Johnson, R.F.; Chen, D.Y.; Huzella, L.; Ragland, D.R.; Mollura, D.J.; Byrum, R.; Reba, R.C.; Jennings, G.; Jahrling, P.B.; et al. Evaluation of Monkeypox Disease Progression by Molecular Imaging. J. Infect. Dis. 2011, 204, 1902–1911. [Google Scholar] [CrossRef] [PubMed]
- Bras, G. The morbid anatomy of smallpox. Doc. Med. Geogr. Trop. 1952, 4, 303–351. [Google Scholar] [PubMed]
- McCullers, J.A. The co-pathogenesis of influenza viruses with bacteria in the lung. Nat. Rev. Microbiol. 2014, 12, 252–262. [Google Scholar] [CrossRef] [PubMed]
- Nipp, R.D.; Kelley, M.J.; Williams, C.D.; Kamal, A.H. Evolution of the Quality Oncology Practice Initiative supportive care quality measures portfolio and conformance at a Veterans Affairs medical center. J. Oncol. Pract. 2013, 9, e86–e89. [Google Scholar] [CrossRef] [PubMed]
- Kielstein, J.T.; Beutel, G.; Fleig, S.; Steinhoff, J.; Meyer, T.N.; Hafer, C.; Kuhlmann, U.; Bramstedt, J.; Panzer, U.; Vischedyk, M.; et al. Best supportive care and therapeutic plasma exchange with or without eculizumab in Shiga-toxin-producing E. coli O104:H4 induced haemolytic-uraemic syndrome: An analysis of the German STEC-HUS registry. Nephrol. Dial. Transplant. 2012, 27, 3807–3815. [Google Scholar] [CrossRef] [PubMed]
- Chittick, G.; Morrison, M.; Brundage, T.; Nichols, W.G. Short-term clinical safety profile of brincidofovir: A favorable benefit-risk proposition in the treatment of smallpox. Antivir. Res. 2017, 143, 269–277. [Google Scholar] [CrossRef] [PubMed]
- Jordan, R.; Chinsangaram, J.; Bolken, T.C.; Tyavanagimatt, S.R.; Tien, D.; Jones, K.F.; Frimm, A.; Corrado, M.L.; Pickens, M.; Landis, P.; et al. Safety and Pharmacokinetics of the Anti-Orthopoxvirus Compound ST-246(R) following Repeat Oral Dosing in Healthy Adult Subjects. Antimicrob. Agents Chemother. 2010, 54, 2560–2566. [Google Scholar] [CrossRef] [PubMed]
- Jordan, R.; Leeds, J.M.; Tyavanagimatt, S.; Hruby, D.E. Development of ST-246(R) for Treatment of Poxvirus Infections. Viruses 2010, 2, 2409–2435. [Google Scholar] [CrossRef] [PubMed]
- Lonnroth, K.; Castro, K.G.; Chakaya, J.M.; Chauhan, L.S.; Floyd, K.; Glaziou, P.; Raviglione, M.C. Tuberculosis control and elimination 2010–50: Cure, care, and social development. Lancet 2010, 375, 1814–1829. [Google Scholar] [CrossRef]
- World Health Organization. Leprosy Elimination Monitoring (LEM); Guidelines for Monitors; Report No. WHO/CDS/CPE/2000.17; World Health Organization: Geneva, Switzerland, 2000. [Google Scholar]
- World Health Organization Regional Office of Africa Disease Prevention and Control Custer, US Centers for Disease Control and Prevention Center for Global Health. Technical Guidelines for Integrated Disease Surveillance and Response in the African Region, 2nd ed.; Kasolo, F., Raungou, J., Eds.; World Health Organization and Centers for Disease Control: Atlanta, GA, USA, 2010.
- Mucker, E.M.; Chapman, J.; Huzella, L.M.; Huggins, J.W.; Shamblin, J.; Robinson, C.G.; Hensley, L.E. Susceptibility of Marmosets (Callithrix jacchus) to Monkeypox Virus: A Low Dose Prospective Model for Monkeypox and Smallpox Disease. PLoS ONE 2015, 10, e0131742. [Google Scholar] [CrossRef] [PubMed]
- Tree, J.A.; Hall, G.; Pearson, G.; Rayner, E.; Graham, V.A.; Steeds, K.; Bewley, K.R.; Hatch, G.J.; Dennis, M.; Taylor, I.; et al. Sequence of pathogenic events in cynomolgus macaques infected with aerosolized monkeypox virus. J. Virol. 2015, 89, 4335–4344. [Google Scholar] [CrossRef] [PubMed]
| System Affected/Syndrome | Treatment Objective | Therapeutic Considerations/Clinical Setting | Follow-up/Monitoring | |
|---|---|---|---|---|
| Developed | Low-Resource | |||
| Respiratory tract | Maintain patent airways, prevent respiratory infection, atelectasis, and respiratory compromise | Suctioning of the nasopharynx and airways, incentive spirometry, chest physiotherapy, bronchodilation, oral/intravenous antibiotics for prophylaxis/treatment, nebulizer treatments, bronchoscopy, noninvasive ventilation (e.g., BiPAP or CPAP) 1, intubation/ventilation | Suctioning of the nasopharynx and airways, incentive spirometry, chest physiotherapy, bronchodilation, oral/intravenous antibiotics for prophylaxis/treatment | Respiratory rate, pulse oximetry |
| Sepsis | Hemodynamic stabilization | Oral/intravenous antibiotics, hemodynamic (e.g., intravenous fluid hydration and vasopressors), supplemental oxygen, corticosteroids, insulin | Oral/intravenous antibiotics, intravenous fluid hydration | Hemodynamic monitoring (e.g., pulse rate, blood pressure) |
| Gastrointestinal/mouth & throat sores | Minimize mucosal pain and disruption of food intake, promote lesion healing | Oral/topical analgesic medications | Oral/topical analgesic medications | Lesion burden, pain scale, food/fluid intake |
| Gastrointestinal/vomiting, diarrhea | Minimize gastrointestinal fluid losses | Oral/intravenous antiemetic and antidiarrheal medications, oral/intravenous rehydration | Oral/intravenous antiemetic and antidiarrheal medications, oral/intravenous rehydration | Frequency and volume of emesis and diarrhea, body weight, fluid intake/ouput |
| Fever | Prevent and treat episodes of fever | Antipyretic medications, external cooling | Antipyretic medications, external cooling | Routine temperature monitoring |
| Exfoliation, skin compromise | Minimize insensible fluid loss, promote lesion healing | Wash with soap and water or dilute water povidone-iodine solution, moisturized dressings, topical antibiotics (e.g., silver sulfadiazine), surgical debridement, skin grafts | Wash with soap and water or dilute water povidone-iodine solution, moisturized dressings, topical antibiotics (e.g., silver sulfadiazine) | Lesion count/rash burden, body weight, fluid intake/ouput |
| Superinfection skin | Prevention/treatment of secondary bacterial infections, promote lesion healing | Oral/intravenous antibiotics, incision and drainage, advanced wound management (e.g., negative pressure wound therapy) | Oral/intravenous antibiotics, incision and drainage | Fever, pain/tenderness, erythema, edema, exudate, warmth |
| Inflammation/lymphadenopathy | Minimize pain and decrease size of lymphadenopathy | Oral/intravenous anti-inflammatory/analgesic medications | Oral/intravenous anti-inflammatory/analgesic medications | Size of lymphadenopathy, pain/tenderness |
| Ocular infection | Prevent corneal scarring and vision impairment | Ophthalmic antibiotics/antivirals and corticosteroids; slit lamp examination | Ophthalmic antibiotics/antivirals and corticosteroids | Vision testing; repeat examination to assess recrudescence |
| Performance Indicator | Clinical Metric | Bench Mark/Target |
|---|---|---|
| Reduced mortality | % fatal cases | <5% |
| Reduced morbidity | % patients provided with supportive care regimen | >50% (good) 1–50% (basic) <1% (inadequate) |
| Reduced syndrome severity | % patients treated for syndromes/complications (respiratory, epidermal, gastrointestinal, inflammatory) | >50% (good) 1–50% (basic) <1% (inadequate) |
| Prevention of sequelae | % ocular complications treated with triflouridine | >50% (good) 1–50% (basic) <1% (inadequate) |
| Prevention of secondary transmission | % patients placed in isolation | >80% (good) 30–80% (basic) <30% (inadequate) |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Reynolds, M.G.; McCollum, A.M.; Nguete, B.; Shongo Lushima, R.; Petersen, B.W. Improving the Care and Treatment of Monkeypox Patients in Low-Resource Settings: Applying Evidence from Contemporary Biomedical and Smallpox Biodefense Research. Viruses 2017, 9, 380. https://doi.org/10.3390/v9120380
Reynolds MG, McCollum AM, Nguete B, Shongo Lushima R, Petersen BW. Improving the Care and Treatment of Monkeypox Patients in Low-Resource Settings: Applying Evidence from Contemporary Biomedical and Smallpox Biodefense Research. Viruses. 2017; 9(12):380. https://doi.org/10.3390/v9120380
Chicago/Turabian StyleReynolds, Mary G., Andrea M. McCollum, Beatrice Nguete, Robert Shongo Lushima, and Brett W. Petersen. 2017. "Improving the Care and Treatment of Monkeypox Patients in Low-Resource Settings: Applying Evidence from Contemporary Biomedical and Smallpox Biodefense Research" Viruses 9, no. 12: 380. https://doi.org/10.3390/v9120380
APA StyleReynolds, M. G., McCollum, A. M., Nguete, B., Shongo Lushima, R., & Petersen, B. W. (2017). Improving the Care and Treatment of Monkeypox Patients in Low-Resource Settings: Applying Evidence from Contemporary Biomedical and Smallpox Biodefense Research. Viruses, 9(12), 380. https://doi.org/10.3390/v9120380