Sophoraflavenone G Restricts Dengue and Zika Virus Infection via RNA Polymerase Interference
Abstract
:1. Introduction
2. Materials and Methods
2.1. Isolation and Identification of Sophoraflavenone G
2.2. Cell Culture
2.3. Virus Production, and Quantification
2.4. MTT Assay
2.5. RNA Dependent RNA Polymerase Activity Assay
2.6. DENV and ZIKV E Staining
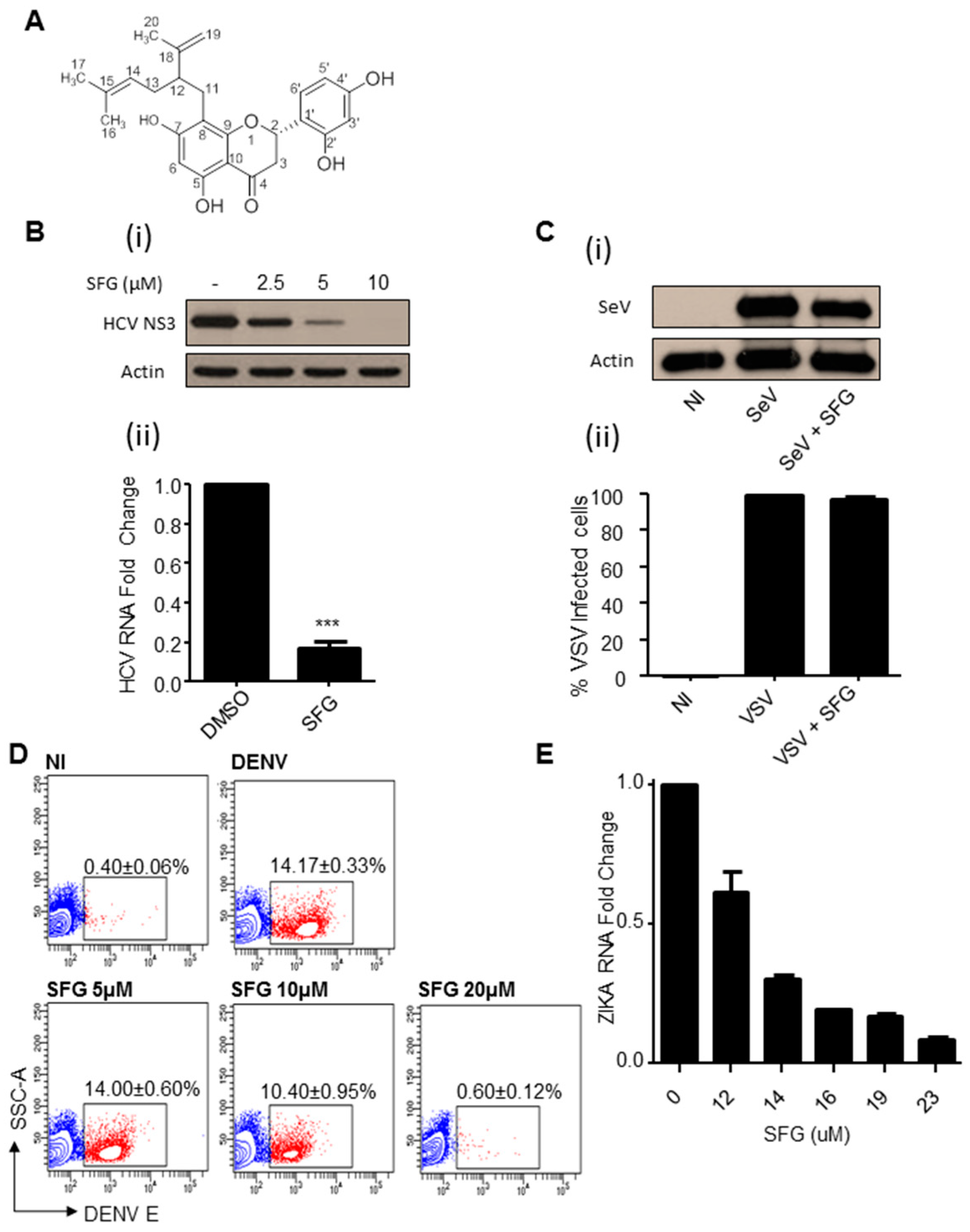
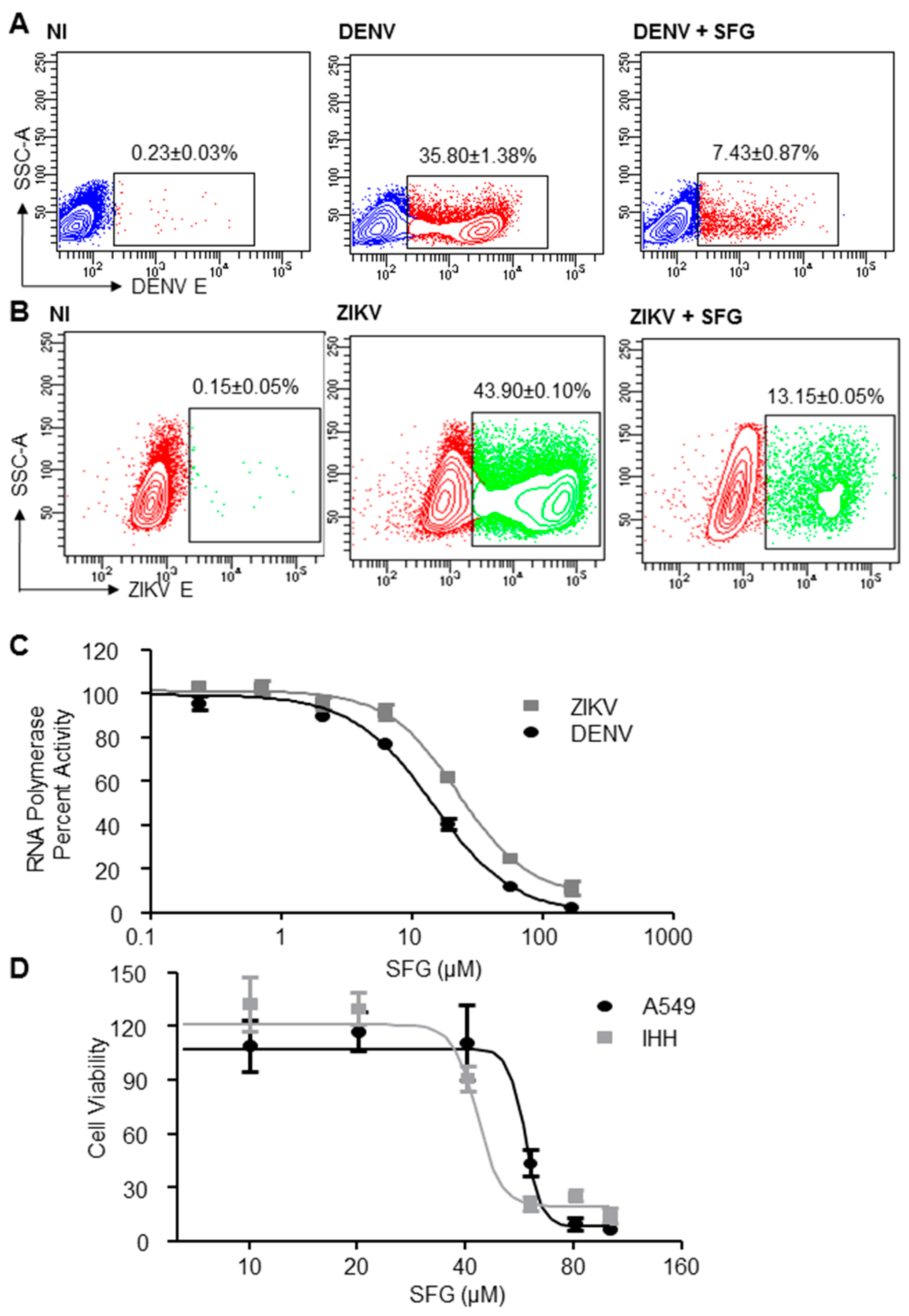
3. Results
4. Discussion
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Vescovo, T.; Refolo, G.; Vitagliano, G.; Fimia, G.M.; Piacentini, M. Molecular mechanisms of hepatitis C virus-induced hepatocellular carcinoma. Clin. Microbiol. Infect. Off. Publ. Eur. Soc. Clin. Microbiol. Infect. Dis. 2016, 22, 853–861. [Google Scholar] [CrossRef] [PubMed]
- Olagnier, D.; Amatore, D.; Castiello, L.; Ferrari, M.; Palermo, E.; Diamond, M.S.; Palamara, A.T.; Hiscott, J. Dengue virus immunopathogenesis: Lessons applicable to the emergence of Zika virus. J. Mol. Biol. 2016, 428, 3429–3448. [Google Scholar] [CrossRef] [PubMed]
- Olagnier, D.; Muscolini, M.; Coyne, C.B.; Diamond, M.S.; Hiscott, J. Mechanisms of Zika virus infection and neuropathogenesis. DNA Cell Biol. 2016, 35, 367–372. [Google Scholar] [CrossRef] [PubMed]
- Bharucha, T.; Breuer, J. Review: A neglected flavivirus: An update on Zika virus in 2016 and the future direction of research. Neuropathol. Appl. Neurobiol. 2016, 42, 317–325. [Google Scholar] [CrossRef] [PubMed]
- Mlakar, J.; Korva, M.; Tul, N.; Popovic, M.; Poljsak-Prijatelj, M.; Mraz, J.; Kolenc, M.; Resman Rus, K.; Vesnaver Vipotnik, T.; Fabjan Vodusek, V.; et al. Zika virus associated with microcephaly. N. Engl. J. Med. 2016, 374, 951–958. [Google Scholar] [CrossRef] [PubMed]
- Cugola, F.R.; Fernandes, I.R.; Russo, F.B.; Freitas, B.C.; Dias, J.L.; Guimaraes, K.P.; Benazzato, C.; Almeida, N.; Pignatari, G.C.; Romero, S.; et al. The brazilian Zika virus strain causes birth defects in experimental models. Nature 2016, 534, 267–271. [Google Scholar] [CrossRef] [PubMed]
- Miner, J.J.; Cao, B.; Govero, J.; Smith, A.M.; Fernandez, E.; Cabrera, O.H.; Garber, C.; Noll, M.; Klein, R.S.; Noguchi, K.K.; et al. Zika virus infection during pregnancy in mice causes placental damage and fetal demise. Cell 2016, 165, 1081–1091. [Google Scholar] [CrossRef] [PubMed]
- Carteaux, G.; Maquart, M.; Bedet, A.; Contou, D.; Brugieres, P.; Fourati, S.; Cleret de Langavant, L.; de Broucker, T.; Brun-Buisson, C.; Leparc-Goffart, I.; et al. Zika virus associated with meningoencephalitis. N. Engl. J. Med. 2016, 374, 1595–1596. [Google Scholar] [CrossRef] [PubMed]
- Mecharles, S.; Herrmann, C.; Poullain, P.; Tran, T.H.; Deschamps, N.; Mathon, G.; Landais, A.; Breurec, S.; Lannuzel, A. Acute myelitis due to Zika virus infection. Lancet 2016, 387, 1481. [Google Scholar] [CrossRef]
- Eyer, L.; Nencka, R.; Huvarova, I.; Palus, M.; Joao Alves, M.; Gould, E.A.; de Clercq, E.; Ruzek, D. Nucleoside inhibitors of Zika virus. J. Infect. Dis. 2016, 214, 707–711. [Google Scholar] [CrossRef] [PubMed]
- He, X.; Fang, J.; Huang, L.; Wang, J.; Huang, X. Sophora flavescens ait: Traditional usage, phytochemistry and pharmacology of an important traditional Chinese medicine. J. Ethnopharmacol. 2015, 172, 10–29. [Google Scholar] [CrossRef] [PubMed]
- Shi, Z.; Chen, J.; Li, C.; An, N.; Wang, Z.; Yang, S.; Huang, K.; Bao, J. Antitumor effects of concanavalin a and sophora flavescens lectin in vitro and in vivo. Acta Pharmacol. Sin. 2014, 35, 248–256. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.S.; Wang, G.J.; Cai, X.; Yu, H.Y.; Hu, Y.P. Inhibition of hepatitis B virus by oxymatrine in vivo. World J. Gastroenterol. 2001, 7, 49–52. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Yao, L.F.; Zhao, Y.; Wei, L.M.; Guo, P.; Yu, M.; Cao, B.; Li, T.; Chen, H.; Zou, Z.M. The Chinese herbal medicine formula mKG suppresses pulmonary fibrosis of mice induced by bleomycin. Int. J. Mol. Sci. 2016, 17, 238. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.T.; Colby-Germinario, S.P.; Hassounah, S.; Quashie, P.K.; Han, Y.; Oliveira, M.; Stranix, B.R.; Wainberg, M.A. Identification of a pyridoxine-derived small-molecule inhibitor targeting Dengue virus RNA-dependent RNA polymerase. Antimicrob. Agents Chemother. 2015, 60, 600–608. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.T.; Hassounah, S.A.; Colby-Germinario, S.P.; Oliveira, M.; Fogarty, C.; Quan, Y.; Han, Y.; Golubkov, O.; Ibanescu, I.; Brenner, B.; et al. Purification of Zika virus RNA-dependent RNA polymerase and its use to identify small-molecule Zika inhibitors. J. Antimicrob. Chemother. 2017, 72, 727–734. [Google Scholar] [CrossRef] [PubMed]
- Kim, C.S.; Park, S.N.; Ahn, S.J.; Seo, Y.W.; Lee, Y.J.; Lim, Y.K.; Freire, M.O.; Cho, E.; Kook, J.K. Antimicrobial effect of sophoraflavanone G isolated from sophora flavescens against mutans streptococci. Anaerobe 2013, 19, 17–21. [Google Scholar] [CrossRef] [PubMed]
- Azzam, H.S.; Goertz, C.; Fritts, M.; Jonas, W.B. Natural products and chronic hepatitis C virus. Liver Int. Off. J. Int. Assoc. Stud. Liver 2007, 27, 17–25. [Google Scholar] [CrossRef] [PubMed]
- Bullard-Feibelman, K.M.; Govero, J.; Zhu, Z.; Salazar, V.; Veselinovic, M.; Diamond, M.S.; Geiss, B.J. The FDA-approved drug sofosbuvir inhibits Zika virus infection. Antivir. Res. 2017, 137, 134–140. [Google Scholar] [CrossRef] [PubMed]
- Dick, G.W.; Kitchen, S.F.; Haddow, A.J. Zika virus. I. Isolations and serological specificity. Trans. R. Soc. Trop. Med. Hyg. 1952, 46, 509–520. [Google Scholar] [CrossRef]
- Roth, A.; Mercier, A.; Lepers, C.; Hoy, D.; Duituturaga, S.; Benyon, E.; Guillaumot, L.; Souares, Y. Concurrent outbreaks of Dengue, Chikungunya and Zika virus infections—An unprecedented epidemic wave of mosquito-borne viruses in the pacific 2012–2014. Euro. Surveill. Bull. Eur. Mal. Transm. Eur. Commun. Dis. Bull. 2014, 19, 20929. [Google Scholar] [CrossRef]
- McCarthy, M. Who recommends eight week abstinence or safer sex after return from Zika areas. BMJ (Clin. Res. Ed.) 2016, 353, i3097. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Liu, W.; Gong, P. A structural overview of RNA-dependent RNA polymerases from the flaviviridae family. Int. J. Mol. Sci. 2015, 16, 12943–12957. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.Y.; Huang, W.C.; Tu, R.S.; Gu, P.Y.; Lin, C.F.; Liou, C.J. Sophoraflavanone G induces apoptosis in human leukemia cells and blocks mapk activation. Am. J. Chin. Med. 2016, 44, 165–176. [Google Scholar] [CrossRef] [PubMed]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sze, A.; Olagnier, D.; Hadj, S.B.; Han, X.; Tian, X.H.; Xu, H.-T.; Yang, L.; Shi, Q.; Wang, P.; Wainberg, M.A.; et al. Sophoraflavenone G Restricts Dengue and Zika Virus Infection via RNA Polymerase Interference. Viruses 2017, 9, 287. https://doi.org/10.3390/v9100287
Sze A, Olagnier D, Hadj SB, Han X, Tian XH, Xu H-T, Yang L, Shi Q, Wang P, Wainberg MA, et al. Sophoraflavenone G Restricts Dengue and Zika Virus Infection via RNA Polymerase Interference. Viruses. 2017; 9(10):287. https://doi.org/10.3390/v9100287
Chicago/Turabian StyleSze, Alexandre, David Olagnier, Samar Bel Hadj, Xiaoying Han, Xiao Hong Tian, Hong-Tao Xu, Long Yang, Qingwen Shi, Penghua Wang, Mark A. Wainberg, and et al. 2017. "Sophoraflavenone G Restricts Dengue and Zika Virus Infection via RNA Polymerase Interference" Viruses 9, no. 10: 287. https://doi.org/10.3390/v9100287
APA StyleSze, A., Olagnier, D., Hadj, S. B., Han, X., Tian, X. H., Xu, H.-T., Yang, L., Shi, Q., Wang, P., Wainberg, M. A., Wu, J. H., & Lin, R. (2017). Sophoraflavenone G Restricts Dengue and Zika Virus Infection via RNA Polymerase Interference. Viruses, 9(10), 287. https://doi.org/10.3390/v9100287





