Differential Expression of HERV-K (HML-2) Proviruses in Cells and Virions of the Teratocarcinoma Cell Line Tera-1
Abstract
:1. Introduction
2. Materials and Methods
2.1. Cell Culture
2.2. RNA Extraction
2.3. RNASeq Library Preparation
2.4. RNASeq Analysis
2.5. MiSeq In-Silico Simulation
2.6. Phylogenetic Analysis
2.7. LTR Amplification and Cloning
2.8. Transfection and Dual-Luciferase Assay
3. Results
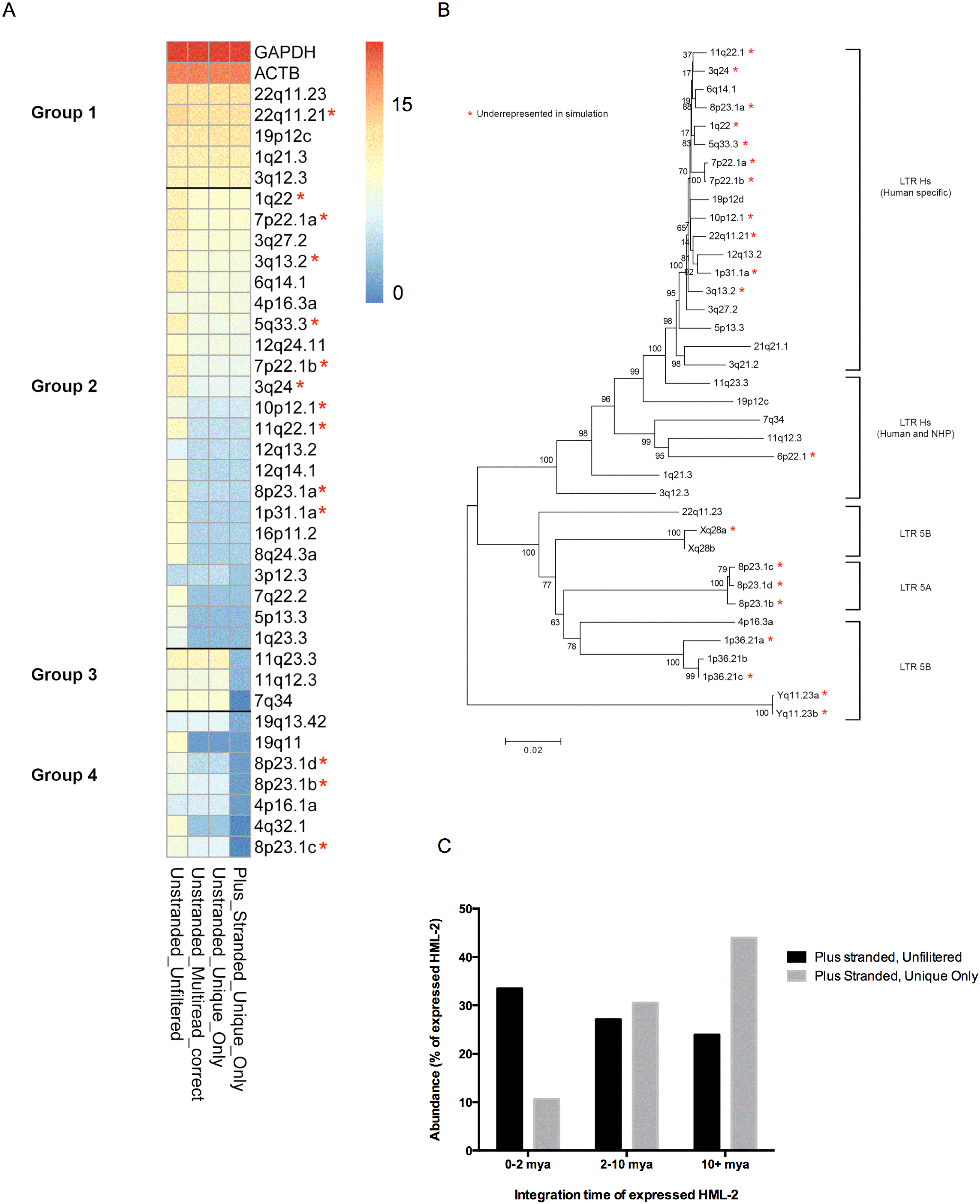
3.1. RNASeq Methodology to Determine HML-2 Provirus Expression
| Provirus | Alias | Chromosomal Location (hg19) |
|---|---|---|
| 1p31.1a | K4, K116, ERV-K1 | chr1: 75842771-75849143 |
| 1p36.21a | N/A | chr1: 12840260-12846364 |
| 1p36.21b | K(OLDAL023753), K6, K76 | chr1: 13458305-13467826 |
| 1p36.21c | K6, K76 | chr1: 13678850-13688242 |
| 1q21.3 | N/A | chr1: 150605284-150608361 |
| 1q22 | K102, K(C1b), K50a, ERVK-7 | chr1: 155596457-155605636 |
| 1q23.3 | K110, K18, 1 (+) K(C1a), ERVK-18 | chr1: 160660575-160669806 |
| 3p12.3 | N/A | chr3: 75600465-75609150 |
| 3q12.3 | K(II), ERVK-5 | chr3: 101410737-101419859 |
| 3q13.2 | K106, K(C3), K68, ERVK-3 | chr3: 112743123-112752282 |
| 3q21.2 | K(I), ERVK-4 | chr3: 125609302-125618416 |
| 3q24 | ERVK-13 | chr3: 148281477-148285396 |
| 3q27.2 | K50b, K117, 3 (-) ERVK-11 | chr3: 185280336-185289515 |
| 4p16.1a | K17b | chr4: 9123515-9133075 |
| 4p16.3a | N/A | chr4: 234989-239459 |
| 4q32.1 | N/A | chr4: 161579938-161582360 |
| 5p13.3 | K104, K50d | chr5: 30487114-30496205 |
| 5q33.3 | K107/K10, K(C5), ERVK-10 | chr5: 156084717-156093896 |
| 6p22.1 | K(OLDAL121932), K69, K20 | chr6: 28650367-28660735 |
| 6q14.1 | K109, K(C6), ERVK-9 | chr6: 78427019-78436083 |
| 7p22.1a | K108L, K(HML.2-HOM), K(C7), ERVK-6 | chr7: 4622057-4631528 |
| 7p22.1b | K108R, ERVK-6 | chr7: 4630561-4640031 |
| 7q22.2 | ERVK-14 | chr7: 104388369-104393266 |
| 7q34 | K(OLDAC004979), ERVK-15 | chr7: 141450926-141455903 |
| 8p23.1a | K115, ERVK-8 | chr8: 7355397-7364859 |
| 8p23.1b | K27 | chr8: 8054700-8064221 |
| 8p23.1c | N/A | chr8: 12073970-12083497 |
| 8p23.1d | KOLD130352 | chr8: 12316492-12326007 |
| 8q24.3a | N/A | chr8: 140472149-140475236 |
| 10p12.1 | K103, K(C10) | chr10: 27182399-27183380 |
| 11q12.3 | K(OLDAC004127) | chr11: 62135963-62150563 |
| 11q22.1 | K(C11c), K36, K118, ERVK-25 | chr11: 101565794-101575259 |
| 11q23.3 | K(C11b), K37, ERVK-20 | chr11: 118591724-118600883 |
| 12q13.2 | N/A | chr12: 55727215-55728183 |
| 12q14.1 | K(C12), K41, K119, ERVK-21 | chr12: 58721242-58730698 |
| 12q24.11 | N/A | chr12: 111007843-111009325 |
| 16p11.2 | N/A | chr16: 34231474-34234142 |
| 19p12a | K52 | chr19: 20387400-20397512 |
| 19p12b | K113 | chr19: 21841536-21841542 (empty site) |
| 19p12c | K51 | chr19: 22757824-22764561 |
| 19p12d | N/A | chr19: 22414379-22414382 (empty site) |
| 19q11 | K(C19), ERVK-19 | chr19: 28128498-28137361 |
| 19q13.42 | LTR13 | chr19: 53862348-53868044 |
| 21q21.1 | K60, ERVK-23 | chr21: 19933916-19941962 |
| 22q11.21 | K101, K(C22), ERVK-24 | chr21: 18926187-18935307 |
| 22q11.23 | K(OLDAP000345), KOLD345 | chr21: 23879930-23890615 |
| Xq28a | K63 | chrX: 153817163-153819562 |
| Xq28b | K63 | chrX: 153836675-153844015 |
| Yq11.23a | N/A | chrY: 26397837-26401035 |
| Yq11.23b | N/A | chrY: 27561402-27564601 |
3.2. Expression and Packaging of HML-2 Proviruses in Tera-1 Cells and Virions
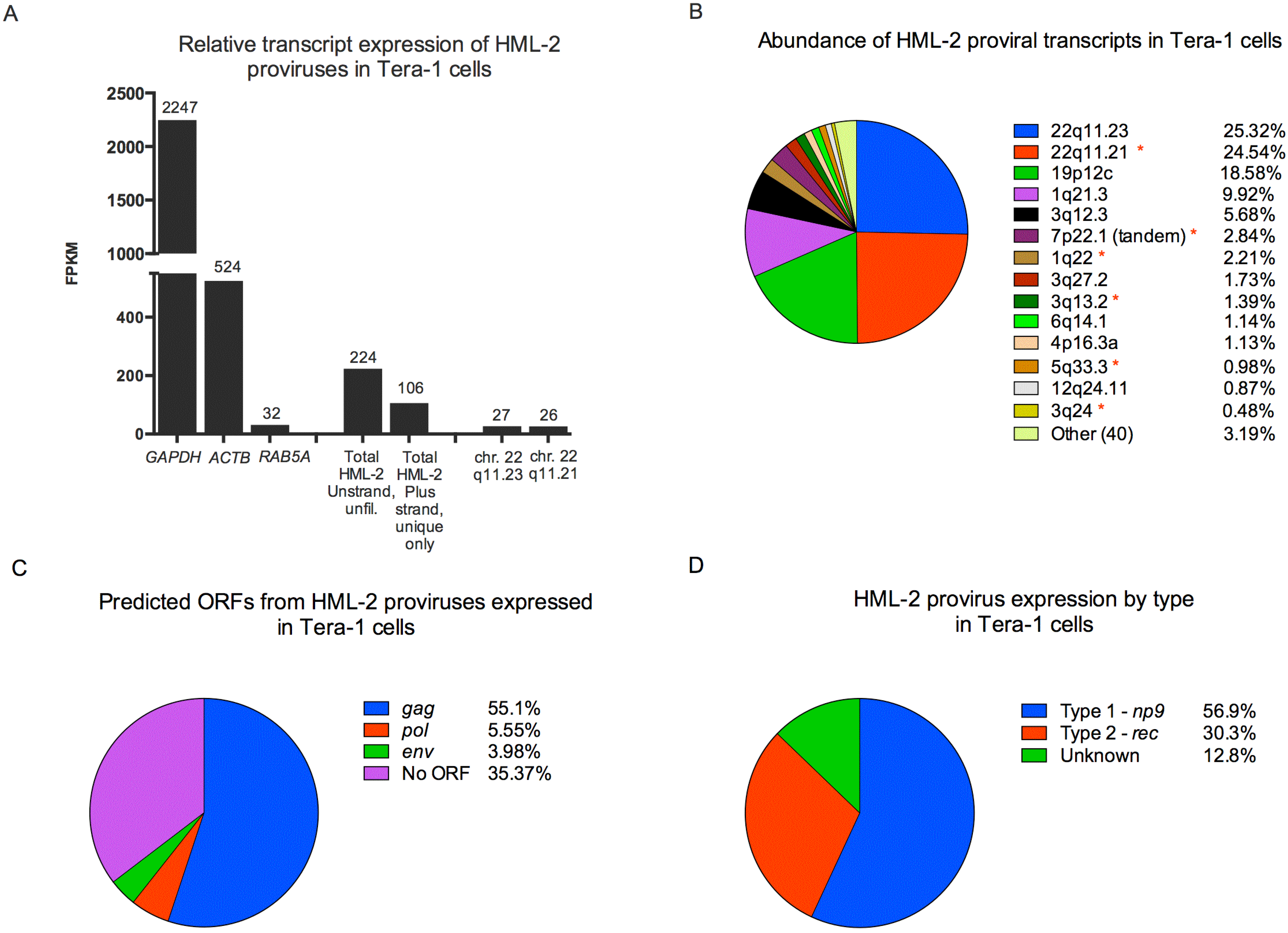
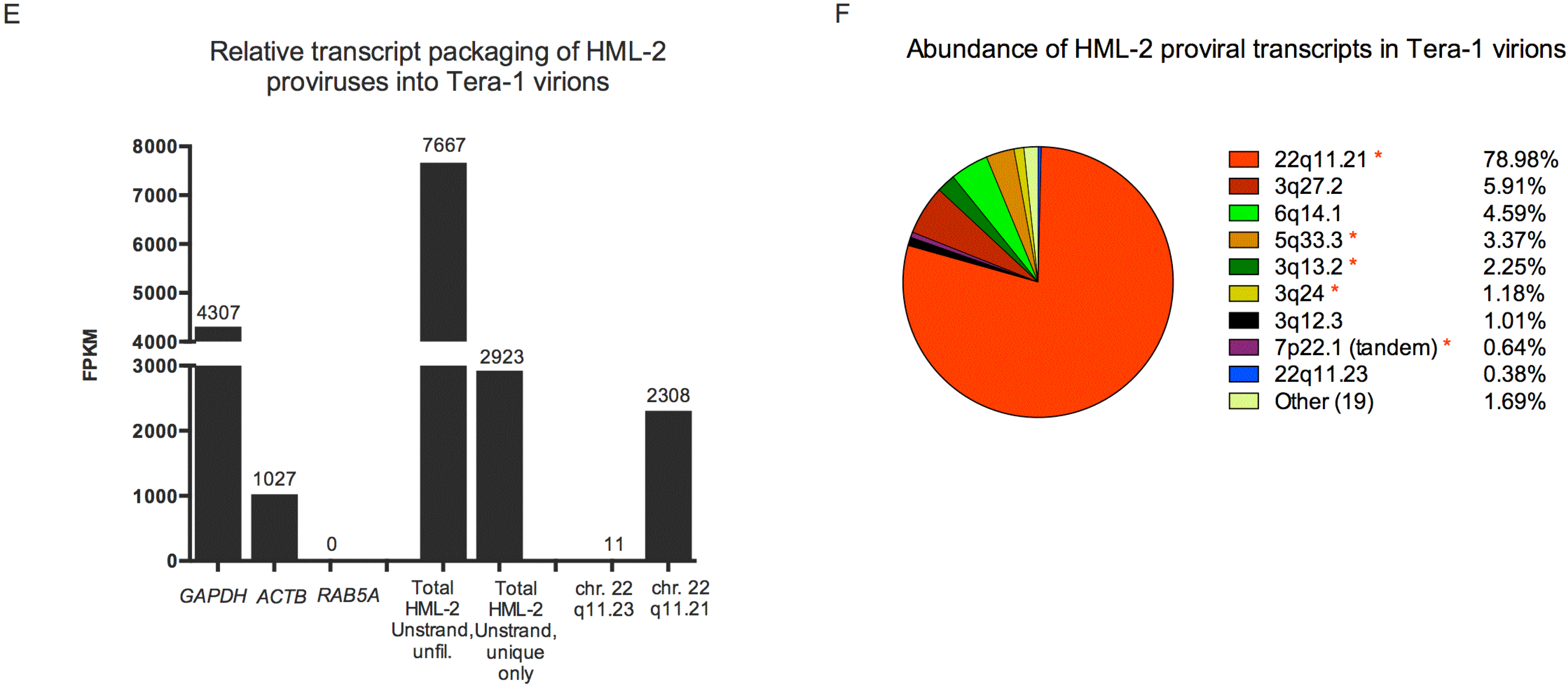
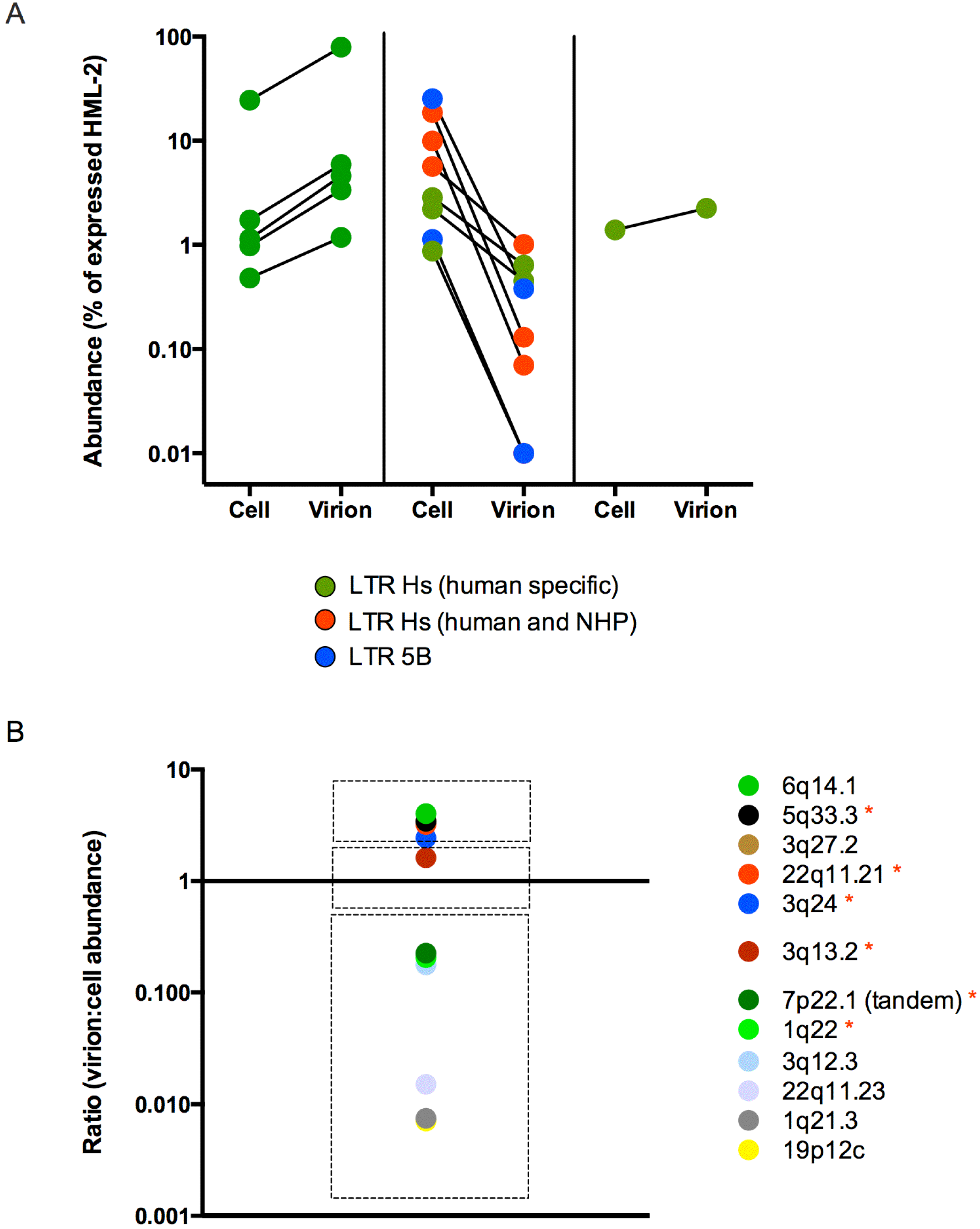
3.3. Human Specific LTR Hs Proviruses were Enriched in HML-2 Virions
3.4. HML-2 Proviruses are Transcribed through a Variety of Mechanisms
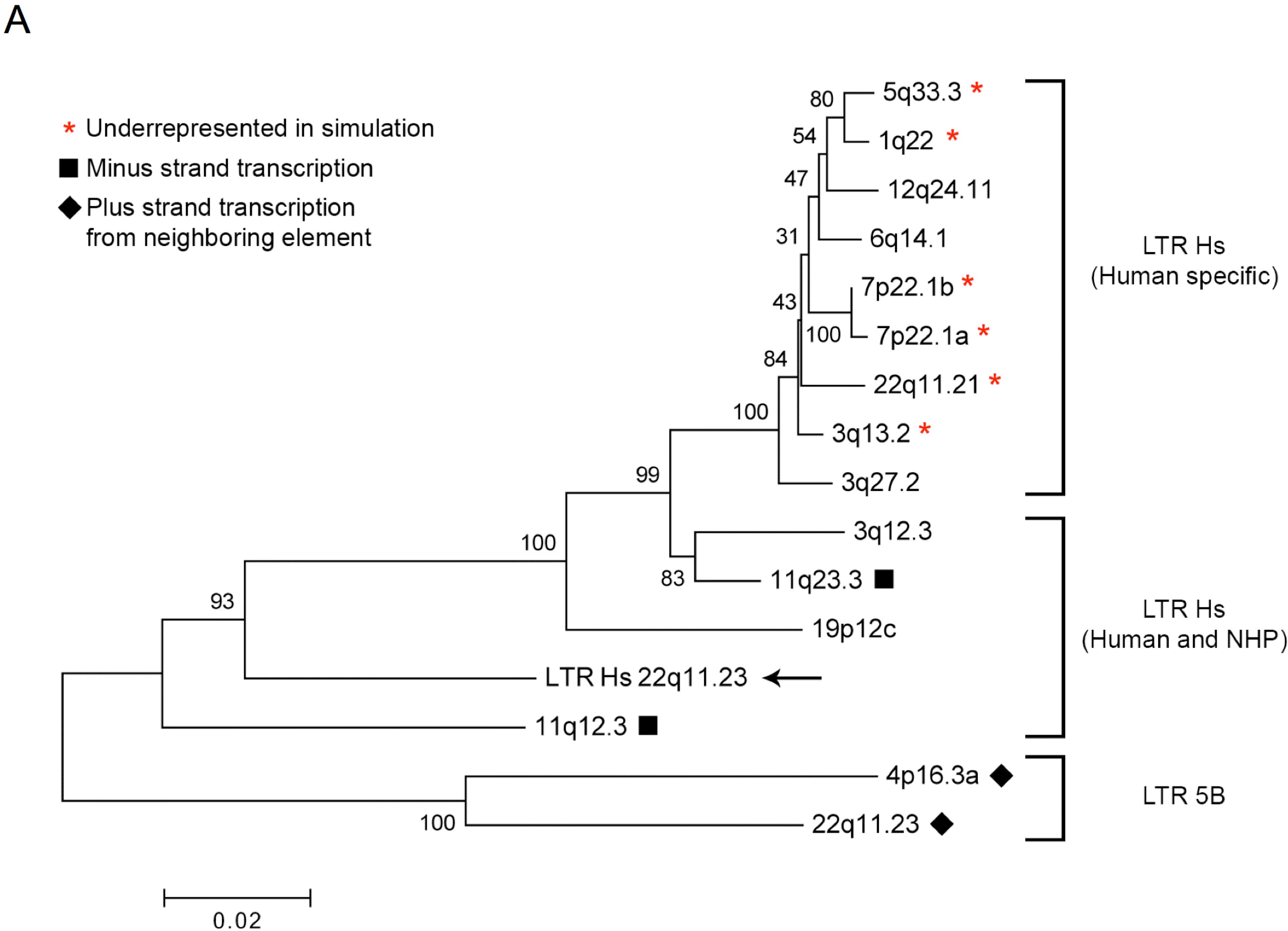
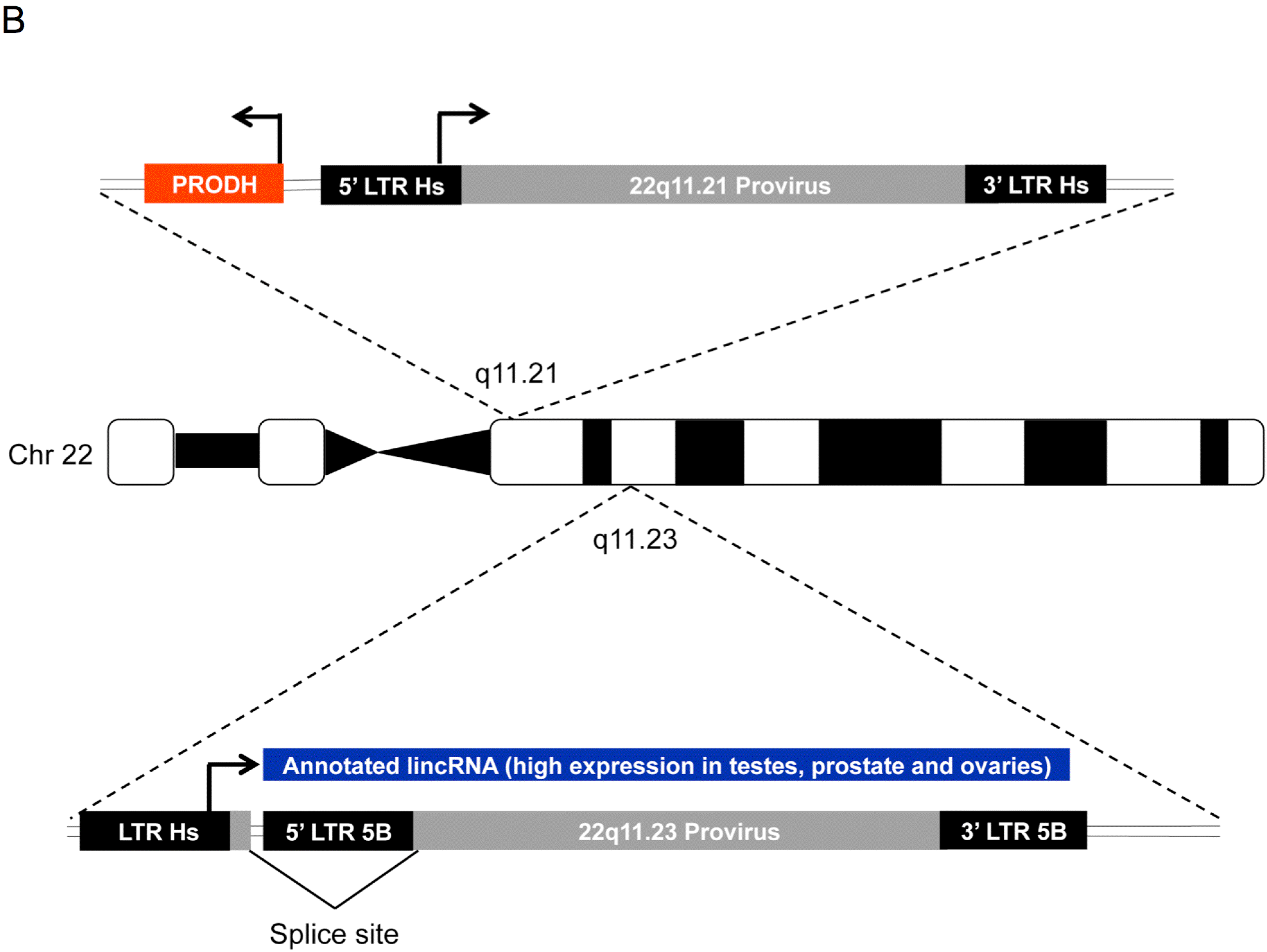
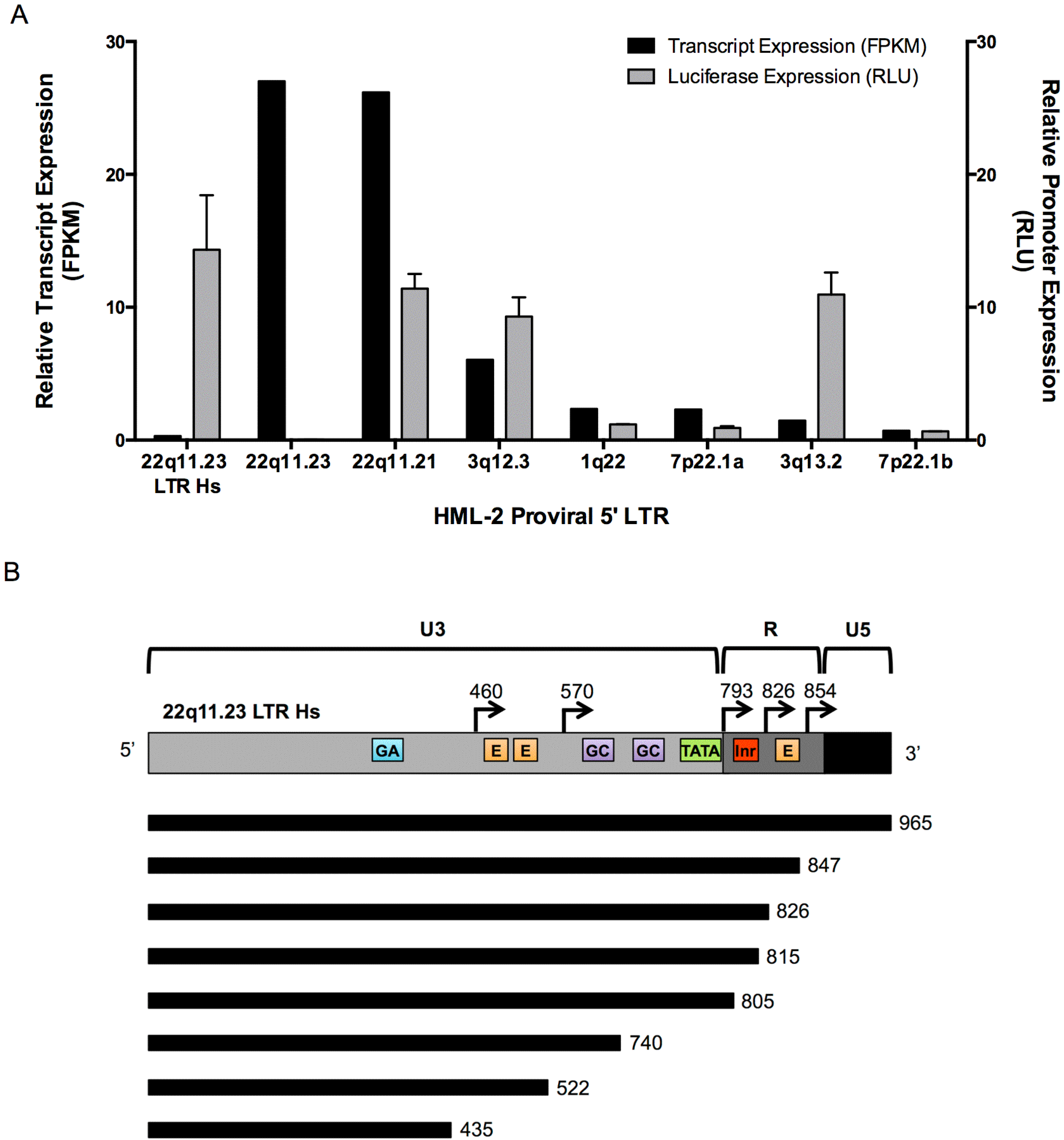
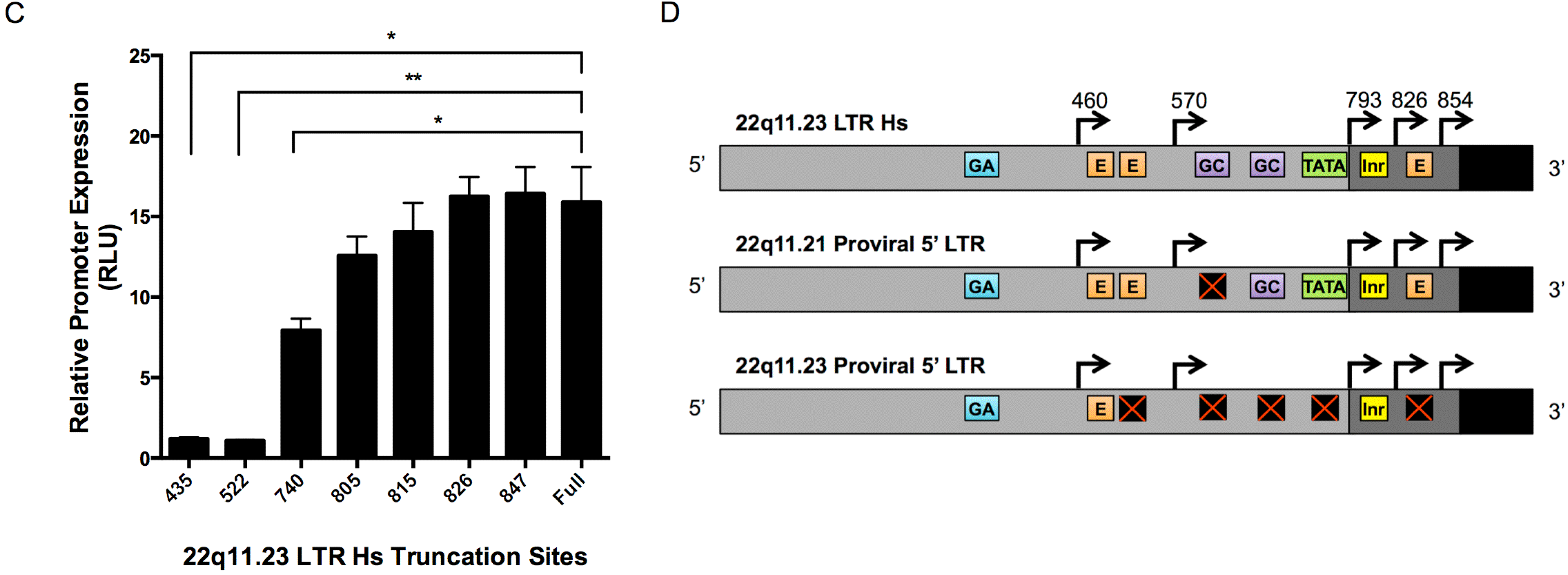
3.5. 5’ LTR Activities of Expressed HML-2 Proviruses Corroborate RNASeq Findings
4. Discussion
Supplementary Files
Supplementary File 1Acknowledgments
Author Contributions
Conflicts of Interest
References
- Boeke, J.D.; Stoye, J.S. Retrotransposons, Endogenous Retroviruses, and the Evolution of Retroelements. In Retroviruses; Coffin, J.M., Hughes, S.H., Varmus, H.E., Eds.; Cold Spring Harbor Laboratory Press: Cold Spring Harbor, NY, USA, 1997; pp. 343–435. [Google Scholar]
- Bannert, N.; Kurth, R. The Evolutionary Dynamics of Human Endogenous Retroviral Families. Annu. Rev. Genomics Hum. Genet. 2006, 7, 149–173. [Google Scholar] [CrossRef] [PubMed]
- Jern, P.; Coffin, J.M. Effects of Retroviruses on Host Genome Function. Annu. Rev. Genet. 2008, 42, 709–732. [Google Scholar] [CrossRef] [PubMed]
- Hughes, J.F.; Coffin, J.M. Human Endogenous Retrovirus K Solo-Ltr Formation and Insertional Polymorphisms: Implications for Human and Viral Evolution. Proc. Natl. Acad. Sci. USA 2004, 101, 1668–1672. [Google Scholar] [CrossRef] [PubMed]
- Subramanian, R.P.; Wildschutte, J.H.; Russo, C.; Coffin, J.M. Identification, Characterization, and Comparative Genomic Distribution of the Herv-K (Hml-2) Group of Human Endogenous Retroviruses. Retrovirology 2011, 8, e90. [Google Scholar] [CrossRef]
- Belshaw, R.; Dawson, A.L.; Woolven-Allen, J.; Redding, J.; Burt, A.; Tristem, M. Genomewide Screening Reveals High Levels of Insertional Polymorphism in the Human Endogenous Retrovirus Family Herv-K(Hml2): Implications for Present-Day Activity. J. Virol. 2005, 79, 12507–12514. [Google Scholar] [CrossRef] [PubMed]
- Marchi, E.; Kanapin, A.; Magiorkinis, G.; Belshaw, R. Unfixed Endogenous Retroviral Insertions in the Human Population. J. Virol. 2014, 88, 9529–9537. [Google Scholar] [CrossRef] [PubMed]
- Callahan, R.; Chiu, I.M.; Wong, J.F.; Tronick, S.R.; Roe, B.A.; Aaronson, S.A.; Schlom, J. A New Class of Endogenous Human Retroviral Genomes. Science 1985, 228, 1208–1211. [Google Scholar] [CrossRef] [PubMed]
- Franklin, G.C.; Chretien, S.; Hanson, I.M.; Rochefort, H.; May, F.E.B.; Westley, B.R. Expression of Human Sequences Related to Those of Mouse Mammary Tumor Virus. J. Virol. 1988, 62, 1203–1210. [Google Scholar] [PubMed]
- Bittner, J.J. Some Possible Effects of Nursing on the Mammary Gland Tumor Incidence in Mice. Science 1936, 84, 162. [Google Scholar] [CrossRef] [PubMed]
- Boller, K.; Konig, H.; Sauter, M.; Mueller-Lantzsch, N.; Lower, R.; Lower, J.; Kurth, R. Evidence That Herv-K Is the Endogenous Retrovirus Sequence That Codes for the Human Teratocarcinoma-Derived Retrovirus Htdv. Virology 1993, 196, 349–353. [Google Scholar] [CrossRef] [PubMed]
- Bieda, K.; Hoffmann, A.; Boller, K. Phenotypic Heterogeneity of Human Endogenous Retrovirus Particles Produced by Teratocarcinoma Cell Lines. J. Gen. Virol. 2001, 82, 591–596. [Google Scholar] [PubMed]
- Ruprecht, K.; Ferreira, H.; Flockerzi, A.; Wahl, S.; Sauter, M.; Mayer, J.; Mueller-Lantzsch, N. Human Endogenous Retrovirus Family Herv-K(Hml-2) Rna Transcripts Are Selectively Packaged into Retroviral Particles Produced by the Human Germ Cell Tumor Line Tera-1 and Originate Mainly from a Provirus on Chromosome 22q11.21. J. Virol. 2008, 82, 10008–10016. [Google Scholar] [CrossRef] [PubMed]
- Lower, R.; Lower, J.; Frank, H.; Harzmann, R.; Kurth, R. Human Teratocarcinomas Cultured in Vitro Produce Unique Retrovirus-Like Viruses. J. Gen. Virol. 1984, 65, 887–898. [Google Scholar] [CrossRef] [PubMed]
- Goedert, J.J.; Sauter, M.E.; Jacobson, L.P.; Vessella, R.L.; Hilgartner, M.W.; Leitman, S.F.; Fraser, M.C.; Mueller-Lantzsch, N.G. High Prevalence of Antibodies against Herv-K10 in Patients with Testicular Cancer but Not with Aids. Cancer Epidemiol. Biomark. Prev. 1999, 8, 293–296. [Google Scholar]
- Sauter, M.; Schommer, S.; Kremmer, E.; Remberger, K.; Dölken, G.; Lemm, I.; Buck, M.; Best, B.; Neumann-Haefelin, D.; Mueller-Lantzsch, N. Human Endogenous Retrovirus K10: Expression of Gag Protein and Detection of Antibodies in Patients with Seminomas. J. Virol. 1995, 69, 414–421. [Google Scholar] [PubMed]
- Kleiman, A.; Senyuta, N.; Tryakin, A.; Sauter, M.; Karseladze, A.; Tjulandin, S.; Gurtsevitch, V.; Mueller-Lantzsch, N. Herv-K(Hml-2) Gag/Env Antibodies as Indicator for Therapy Effect in Patients with Germ Cell Tumors. Int. J. Cancer 2004, 110, 459–461. [Google Scholar] [CrossRef] [PubMed]
- Kurth, R.; Bannert, N. Beneficial and Detrimental Effects of Human Endogenous Retroviruses. Int. J. Cancer 2010, 126, 306–314. [Google Scholar] [CrossRef] [PubMed]
- Boese, A.; Sauter, M.; Mueller-Lantzsch, N. A Rev-Like Nes Mediates Cytoplasmic Localization of Herv-K Corf. FEBS Lett. 2000, 468, 65–67. [Google Scholar] [CrossRef] [PubMed]
- Hohn, O.; Hanke, K.; Bannert, N. Herv-K(Hml-2), the Best Preserved Family of Hervs: Endogenization, Expression, and Implications in Health and Disease. Front. Oncol. 2013, 3, 246. [Google Scholar] [CrossRef] [PubMed]
- Patience, C.; Simpson, G.R.; Colletta, A.A.; Welch, H.M.; Weiss, R.A.; Boyd, M.T. Human Endogenous Retrovirus Expression and Reverse Transcriptase Activity in the T47d Mammary Carcinoma Cell Line. J. Virol. 1996, 70, 2654–2657. [Google Scholar] [PubMed]
- Etkind, P.R.; Lumb, K.; Du, J.; Racevskis, J. Type 1 Herv-K Genome Is Spliced into Subgenomic Transcripts in the Human Breast Tumor Cell Line T47d. Virology 1997, 234, 304–308. [Google Scholar] [CrossRef] [PubMed]
- Wang-Johanning, F.; Frost, A.R.; Johanning, G.L.; Khazaeli, M.B.; LoBuglio, A.F.; Shaw, D.R.; Strong, T.V. Expression of Human Endogenous Retrovirus K Envelope Transcripts in Human Breast Cancer. Clin. Cancer Res. 2001, 7, 1553–1560. [Google Scholar] [PubMed]
- Wang-Johanning, F.; Frost, A.R.; Jian, B.; Epp, L.; Lu, D.W.; Johanning, G.L. Quantitation of Herv-K Env Gene Expression and Splicing in Human Breast Cancer. Oncogene 2003, 22, 1528–1535. [Google Scholar] [CrossRef] [PubMed]
- Wang-Johanning, F.; Radvanyi, L.; Rycaj, K.; Plummer, J.B.; Yan, P.; Sastry, K.J.; Piyathilake, C.J.; Hunt, K.K.; Johanning, G.L. Human Endogenous Retrovirus K Triggers an Antigen-Specific Immune Response in Breast Cancer Patients. Cancer Res. 2008, 68, 5869–5877. [Google Scholar] [CrossRef] [PubMed]
- Wang-Johanning, F.; Rycaj, K.; Plummer, J.B.; Li, M.; Yin, B.; Frerich, K.; Garza, J.G.; Shen, J.; Lin, K.; Yan, P.; et al. Immunotherapeutic Potential of Anti-Human Endogenous Retrovirus-K Envelope Protein Antibodies in Targeting Breast Tumors. J. Natl. Cancer Inst. 2012, 104, 189–210. [Google Scholar] [CrossRef] [PubMed]
- Buscher, K.; Hahn, S.; Hofmann, M.; Trefzer, U.; Ozel, M.; Sterry, W.; Lower, J.; Lower, R.; Kurth, R.; Denner, J. Expression of the Human Endogenous Retrovirus-K Transmembrane Envelope, Rec and Np9 Proteins in Melanomas and Melanoma Cell Lines. Melanoma Res. 2006, 16, 223–234. [Google Scholar] [CrossRef] [PubMed]
- Muster, T.; Waltenberger, A.; Grassauer, A.; Hirschl, S.; Caucig, P.; Romirer, I.; Fodinger, D.; Seppele, H.; Schanab, O.; Magin-Lachmann, C.; et al. An Endogenous Retrovirus Derived from Human Melanoma Cells. Cancer Res. 2003, 63, 8735–8741. [Google Scholar] [PubMed]
- Contreras-Galindo, R.; Gonzalez, M.; Almodovar-Camacho, S.; Gonzalez-Ramirez, S.; Lorenzo, E.; Yamamura, Y. A New Real-Time-Rt-Pcr for Quantitation of Human Endogenous Retroviruses Type K (Herv-K) Rna Load in Plasma Samples: Increased Herv-K Rna Titers in Hiv-1 Patients with Haart Non-Suppressive Regimens. J. Virol. Methods 2006, 136, 51–57. [Google Scholar] [CrossRef] [PubMed]
- Contreras-Galindo, R.; Kaplan, M.H.; Markovitz, D.M.; Lorenzo, E.; Yamamura, Y. Detection of Herv-K(Hml-2) Viral Rna in Plasma of Hiv Type 1-Infected Individuals. AIDS Res. Hum. Retrovir. 2006, 22, 979–984. [Google Scholar] [CrossRef] [PubMed]
- Contreras-Galindo, R.; Lopez, P.; Velez, R.; Yamamura, Y. Hiv-1 Infection Increases the Expression of Human Endogenous Retroviruses Type K (Herv-K) in Vitro. AIDS Res. Hum. Retrovir. 2007, 23, 116–122. [Google Scholar] [CrossRef] [PubMed]
- Ormsby, C.E.; Sengupta, D.; Tandon, R.; Deeks, S.G.; Martin, J.N.; Jones, R.B.; Ostrowski, M.A.; Garrison, K.E.; Vazquez-Perez, J.A.; Reyes-Teran, G.; et al. Human Endogenous Retrovirus Expression is Inversely Associated with Chronic Immune Activation in Hiv-1 Infection. PLoS One 2012, 7, e41021. [Google Scholar] [CrossRef] [PubMed]
- Contreras-Galindo, R.; Kaplan, M.H.; Contreras-Galindo, A.C.; Gonzalez-Hernandez, M.J.; Ferlenghi, I.; Giusti, F.; Lorenzo, E.; Gitlin, S.D.; Dosik, M.H.; Yamamura, Y.; et al. Characterization of Human Endogenous Retroviral Elements in the Blood of Hiv-1-Infected Individuals. J. Virol. 2012, 86, 262–276. [Google Scholar] [CrossRef] [PubMed]
- Bhardwaj, N.; Maldarelli, F.; Mellors, J.; Coffin, J.M. HIV-1 Infection Leads to Increased Transcription of Herv-K (Hml-2) Proviruses in vivo but Not to Increased Virion Production. J. Virol. 2014, in press. [Google Scholar]
- Palmer, S.; Kearney, M.; Maldarelli, F.; Halvas, E.K.; Bixby, C.J.; Bazmi, H.; Rock, D.; Falloon, J.; Davey, R.T., Jr.; Dewar, R.L.; et al. Multiple, Linked Human Immunodeficiency Virus Type 1 Drug Resistance Mutations in Treatment-Experienced Patients Are Missed by Standard Genotype Analysis. J. Clin. Microbiol. 2005, 43, 406–413. [Google Scholar] [CrossRef] [PubMed]
- Santoni, F.A.; Guerra, J.; Luban, J. Herv-H Rna is Abundant in Human Embryonic Stem Cells and a Precise Marker for Pluripotency. Retrovirology 2012, 9, 111. [Google Scholar] [CrossRef] [PubMed]
- Schmitt, K.; Richter, C.; Backes, C.; Meese, E.; Ruprecht, K.; Mayer, J. Comprehensive Analysis of Human Endogenous Retrovirus Group Herv-W Locus Transcription in Multiple Sclerosis Brain Lesions by High-Throughput Amplicon Sequencing. J. Virol. 2013, 87, 13837–13852. [Google Scholar] [CrossRef] [PubMed]
- Brinzevich, D.; Young, G.R.; Sebra, R.; Ayllon, J.; Maio, S.M.; Deikus, G.; Chen, B.K.; Fernandez-Sesma, A.; Simon, V.; Mulder, L.C. Hiv-1 Interacts with Human Endogenous Retrovirus K (Hml-2) Envelopes Derived from Human Primary Lymphocytes. J. Virol. 2014, 88, 6213–6223. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Hernandez, M.J.; Cavalcoli, J.D.; Sartor, M.A.; Contreras-Galindo, R.; Meng, F.; Dai, M.; Dube, D.; Saha, A.K.; Gitlin, S.D.; Omenn, G.S.; et al. Regulation of the Herv-K (Hml-2) Transcriptome by the HIV-1 Tat Protein. J. Virol. 2014. [Google Scholar] [CrossRef]
- Simpson, G.R.; Patience, C.; Löwer, R.; Tönjes, R.R.; Moore, H.D.M.; Weiss, R.A.; Boyd, M.T. Endogenous D-Type (Herv-K) Related Sequences Are Packaged into Retroviral Particles in the Placenta and Possess Open Reading Frames for Reverse Transcriptase. Virology 1996, 222, 451–456. [Google Scholar] [CrossRef] [PubMed]
- Li, M.D.; Bronson, D.L.; Lemke, T.D.; Faras, A.J. Restricted Expression of New Herv-K Members in Human Teratocarcinoma Cells. Virology 1995, 208, 733–741. [Google Scholar] [CrossRef] [PubMed]
- Bolger, A.M.; Lohse, M.; Usadel, B. Trimmomatic: A Flexible Trimmer for Illumina Sequence Data. Bioinformatics 2014, 30, 2114–2120. [Google Scholar] [CrossRef] [PubMed]
- Langmead, B.; Salzberg, S.L. Fast Gapped-Read Alignment with Bowtie 2. Nat. Methods 2012, 9, 357–359. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.; Pertea, G.; Trapnell, C.; Pimentel, H.; Kelley, R.; Salzberg, S.L. Tophat2: Accurate Alignment of Transcriptomes in the Presence of Insertions, Deletions and Gene Fusions. Genome Biol. 2013, 14, R36. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Handsaker, B.; Wysoker, A.; Fennell, T.; Ruan, J.; Homer, N.; Marth, G.; Abecasis, G.; Durbin, R.; Genome Project Data Processing S. The Sequence Alignment/Map Format and Samtools. Bioinformatics 2009, 25, 2078–2079. [Google Scholar] [CrossRef] [PubMed]
- Roberts, A.; Trapnell, C.; Donaghey, J.; Rinn, J.L.; Pachter, L. Improving Rna-Seq Expression Estimates by Correcting for Fragment Bias. Genome Biol. 2011, 12, R22. [Google Scholar] [CrossRef] [PubMed]
- Mortazavi, A.; Williams, B.A.; McCue, K.; Schaeffer, L.; Wold, B. Mapping and Quantifying Mammalian Transcriptomes by Rna-Seq. Nat. Methods 2008, 5, 621–628. [Google Scholar] [CrossRef] [PubMed]
- Sayers, E.W.; Barrett, T.; Benson, D.A.; Bolton, E.; Bryant, S.H.; Canese, K.; Chetvernin, V.; Church, D.M.; DiCuccio, M.; Federhen, S.; et al. Database Resources of the National Center for Biotechnology Information. Nucleic Acids Res. 2011, 39, D38–D51. [Google Scholar] [CrossRef] [PubMed]
- Thorvaldsdottir, H.; Robinson, J.T.; Mesirov, J.P. Integrative Genomics Viewer (Igv): High-Performance Genomics Data Visualization and Exploration. Brief. Bioinform. 2013, 14, 178–192. [Google Scholar] [CrossRef] [PubMed]
- Kent, W.J.; Sugnet, C.W.; Furey, T.S.; Roskin, K.M.; Pringle, T.H.; Zahler, A.M.; Haussler, D. The Human Genome Browser at Ucsc. Genome Res. 2002, 12, 996–1006. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.; Li, L.; Myers, J.R.; Marth, G.T. Art: A Next-Generation Sequencing Read Simulator. Bioinformatics 2012, 28, 593–594. [Google Scholar] [CrossRef] [PubMed]
- Tamura, K.; Stecher, G.; Peterson, D.; Filipski, A.; Kumar, S. Mega6: Molecular Evolutionary Genetics Analysis Version 6.0. Mol. Biol. Evol. 2013, 30, 2725–2729. [Google Scholar] [CrossRef] [PubMed]
- Karolchik, D.; Hinrichs, A.S.; Furey, T.S.; Roskin, K.M.; Sugnet, C.W.; Haussler, D.; Kent, W.J. The Ucsc Table Browser Data Retrieval Tool. Nucleic Acids Res. 2004, 32, D493–D496. [Google Scholar] [CrossRef] [PubMed]
- Untergasser, A.; Cutcutache, I.; Koressaar, T.; Ye, J.; Faircloth, B.C.; Remm, M.; Rozen, S.G. Primer3—New Capabilities and Interfaces. Nucleic Acids Res. 2012, 40, e115. [Google Scholar] [CrossRef] [PubMed]
- Ono, M.; Kawakami, M.; Takezawa, T. A Novel Human Nonviral Retroposon Derived from an Endogenous Retrovirus. Nucleic Acids Res. 1987, 15, 8725–8737. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.N.; Malim, M.H.; Bieniasz, P.D. Hypermutation of an Ancient Human Retrovirus by Apobec3g. J. Virol. 2008, 82, 8762–8770. [Google Scholar] [CrossRef] [PubMed]
- Armbruester, V.; Sauter, M.; Krautkraemer, E.; Meese, E.; Kleiman, A.; Best, B.; Roemer, K.; Mueller-Lantzsch, N. A Novel Gene from the Human Endogenous Retrovirus K Expressed in Transformed Cells. Clin. Cancer Res. 2002, 8, 1800–1807. [Google Scholar] [PubMed]
- Löwer, R.; Tönjes, R.R.; Korbmacher, C.; Kurth, R.; Löwer, J. Identification of a Rev-Related Protein by Analysis of Spliced Transcripts of the Human Endogenous Retroviruses Htdv/Herv-K. J. Virol. 1995, 69, 141–149. [Google Scholar] [PubMed]
- Cabili, M.N.; Trapnell, C.; Goff, L.; Koziol, M.; Tazon-Vega, B.; Regev, A.; Rinn, J.L. Integrative Annotation of Human Large Intergenic Noncoding Rnas Reveals Global Properties and Specific Subclasses. Genes Dev. 2011, 25, 1915–1927. [Google Scholar] [CrossRef] [PubMed]
- Suntsova, M.; Gogvadze, E.V.; Salozhin, S.; Gaifullin, N.; Eroshkin, F.; Dmitriev, S.E.; Martynova, N.; Kulikov, K.; Malakhova, G.; Tukhbatova, G.; et al. Human-Specific Endogenous Retroviral Insert Serves as an Enhancer for the Schizophrenia-Linked Gene Prodh. Proc. Natl. Acad. Sci. USA 2013, 110, 19472–19477. [Google Scholar] [CrossRef] [PubMed]
- Ruda, V.M.; Akopov, S.B.; Trubetskoy, D.O.; Manuylov, N.L.; Vetchinova, A.S.; Zavalova, L.L.; Nikolaev, L.G.; Sverdlov, E.D. Tissue Specificity of Enhancer and Promoter Activities of a Herv-K(Hml-2) Ltr. Virus Res. 2004, 104, 11–16. [Google Scholar] [CrossRef] [PubMed]
- Manghera, M.; Douville, R.N. Endogenous Retrovirus-K Promoter: A Landing Strip for Inflammatory Transcription Factors? Retrovirology 2013, 10, e16. [Google Scholar] [CrossRef]
- Fuchs, N.V.; Kraft, M.; Tondera, C.; Hanschmann, K.M.; Lower, J.; Lower, R. Expression of the Human Endogenous Retrovirus (Herv) Group Hml-2/Herv-K Does Not Depend on Canonical Promoter Elements but Is Regulated by Transcription Factors Sp1 and Sp3. J. Virol. 2011, 85, 3436–3448. [Google Scholar] [CrossRef] [PubMed]
- Kovalskaya, E.; Buzdin, A.; Gogvadze, E.; Vinogradova, T.; Sverdlov, E. Functional Human Endogenous Retroviral Ltr Transcription Start Sites Are Located between the R and U5 Regions. Virology 2006, 346, 373–378. [Google Scholar] [CrossRef] [PubMed]
- Ruprecht, K.; Mayer, J.; Sauter, M.; Roemer, K.; Mueller-Lantzsch, N. Endogenous Retroviruses and Cancer. Cell. Mol. Life Sci. 2008, 65, 3366–3382. [Google Scholar] [CrossRef] [PubMed]
- Van der Kuyl, A.C. Hiv Infection and Herv Expression: A Review. Retrovirology 2012, 9, e6. [Google Scholar]
- Seifarth, W.; Frank, O.; Zeilfelder, U.; Spiess, B.; Greenwood, A.D.; Hehlmann, R.; Leib-Mosch, C. Comprehensive Analysis of Human Endogenous Retrovirus Transcriptional Activity in Human Tissues with a Retrovirus-Specific Microarray. J. Virol. 2005, 79, 341–352. [Google Scholar] [CrossRef] [PubMed]
- Flockerzi, A.; Ruggieri, A.; Frank, O.; Sauter, M.; Maldener, E.; Kopper, B.; Wullich, B.; Seifarth, W.; Muller-Lantzsch, N.; Leib-Mosch, C.; et al. Expression Patterns of Transcribed Human Endogenous Retrovirus Herv-K(Hml-2) Loci in Human Tissues and the Need for a Herv Transcriptome Project. BMC Genomics 2008, 9, e354. [Google Scholar] [CrossRef]
- Lavie, L.; Kitova, M.; Maldener, E.; Meese, E.; Mayer, J. Cpg Methylation Directly Regulates Transcriptional Activity of the Human Endogenous Retrovirus Family Herv-K(Hml-2). J. Virol. 2005, 79, 876–883. [Google Scholar] [CrossRef] [PubMed]
- George, M.; Schwecke, T.; Beimforde, N.; Hohn, O.; Chudak, C.; Zimmermann, A.; Kurth, R.; Naumann, D.; Bannert, N. Identification of the Protease Cleavage Sites in a Reconstituted Gag Polyprotein of an Herv-K(Hml-2) Element. Retrovirology 2011, 8, e30. [Google Scholar] [CrossRef]
- Dewannieux, M.; Blaise, S.; Heidmann, T. Identification of a Functional Envelope Protein from the Herv-K Family of Human Endogenous Retroviruses. J. Virol. 2005, 79, 15573–15577. [Google Scholar] [CrossRef] [PubMed]
- D’Souza, V.; Summers, M.F. How Retroviruses Select Their Genomes. Nat. Rev. Microbiol. 2005, 3, 643–655. [Google Scholar] [CrossRef] [PubMed]
- Rulli, S.J., Jr.; Hibbert, C.S.; Mirro, J.; Pederson, T.; Biswal, S.; Rein, A. Selective and Nonselective Packaging of Cellular Rnas in Retrovirus Particles. J. Virol. 2007, 81, 6623–6631. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bhardwaj, N.; Montesion, M.; Roy, F.; Coffin, J.M. Differential Expression of HERV-K (HML-2) Proviruses in Cells and Virions of the Teratocarcinoma Cell Line Tera-1. Viruses 2015, 7, 939-968. https://doi.org/10.3390/v7030939
Bhardwaj N, Montesion M, Roy F, Coffin JM. Differential Expression of HERV-K (HML-2) Proviruses in Cells and Virions of the Teratocarcinoma Cell Line Tera-1. Viruses. 2015; 7(3):939-968. https://doi.org/10.3390/v7030939
Chicago/Turabian StyleBhardwaj, Neeru, Meagan Montesion, Farrah Roy, and John M. Coffin. 2015. "Differential Expression of HERV-K (HML-2) Proviruses in Cells and Virions of the Teratocarcinoma Cell Line Tera-1" Viruses 7, no. 3: 939-968. https://doi.org/10.3390/v7030939
APA StyleBhardwaj, N., Montesion, M., Roy, F., & Coffin, J. M. (2015). Differential Expression of HERV-K (HML-2) Proviruses in Cells and Virions of the Teratocarcinoma Cell Line Tera-1. Viruses, 7(3), 939-968. https://doi.org/10.3390/v7030939





