Identification of FactorsInfluencing the Puumala Virus Seroprevalence within Its Reservoir in aMontane Forest Environment
Abstract
:1. Introduction
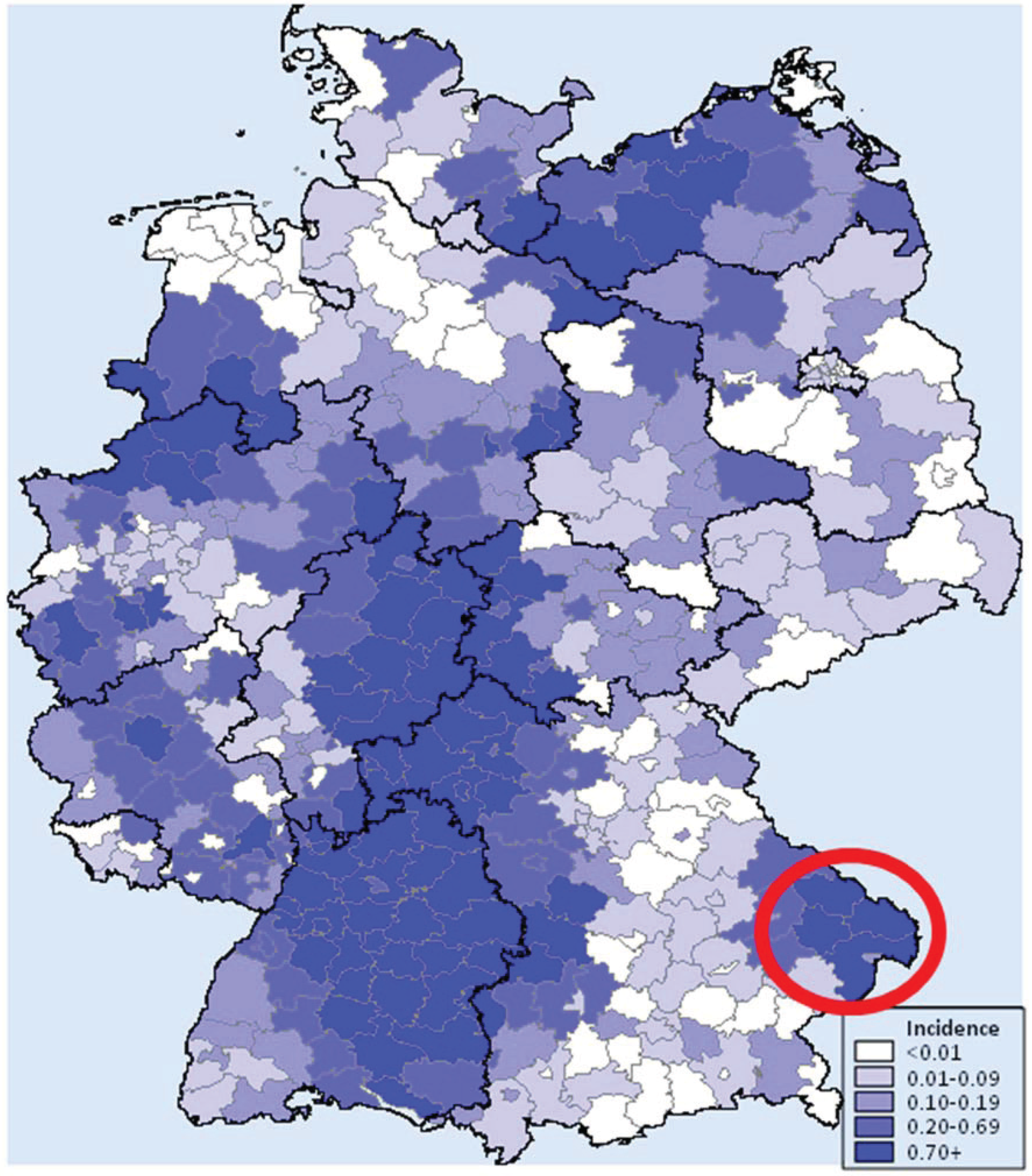
2. Results and Discussion
2.1. Animal Species, Diversity, Abundance and PUUV Seroprevalence
| Parameter | 2008 | 2009 | 2010 |
|---|---|---|---|
| Number of trapped animals (n=674) | 43 | 143 | 488 |
| Number of trapping nights | 9 | 40 | 30 |
| General annual trapping index (calculated for all sites) | 4.11 | 3.36 | 14.06 |
| General annual diversity index of small mammals in Sherman traps (calculated for all sites) | 1.01 | 1.37 | 0.83 |
| Number of trapped Apodemus sp. (A. flavicollis and A. sylvaticus) | 23 | 46 | 193 |
| Number of trapped Microtus agrestis | 4 | 19 | 12 |
| Number of trapped Sorex sp. | 1 | 10 | 4 |
| Number of trapped Glis glis | 0 | 8 | 0 |
| Number of trapped Mustela nivalis | 0 | 1 | 0 |
| Number of trapped bank voles investigated for PUUV(total trapped number) | 15 (19) | 57 (57) | 277 (277) |
| Annual PUUV seroprevalence [%] (number of PUUV-reactive bank voles) | 6.7 (1) | 7.0 (4) | 29.2(81) |
2.2. Multivariate Analysis
| Name of site | Altitude [m asl] | Mean annual temperature [°C] (1980-2007) | Mean annual solar radiation [kwh/m2] | Percent coverage of herb layer (0.02 ha) | Percent coverage of shrub layer (1 ha) | Percent coverage of deadwood layer (0.02 ha) | Percent coverage of beech upper layer (1 ha) |
|---|---|---|---|---|---|---|---|
| Isar32*° | 317 | 8.26 | 3.16 | 80.0 | 30 | 2.0 | 0 |
| Igg 35*° | 379 | 8.03 | 2.68 | 50.0 | 30 | 5.0 | 30 |
| Igg 33*° | 412 | 7.45 | 3.99 | 3.0 | 2 | 7.0 | 45 |
| Sal 27* | 490 | 7.03 | 4.43 | 30.0 | 30 | 10.0 | 40 |
| Els 5* | 510 | 6.61 | 2.35 | 3.0 | 15 | 10.0 | 88 |
| Els 8* | 578 | 6.28 | 2.63 | 20.0 | 15 | 20.0 | 50 |
| NP 37* | 629 | 6.61 | 3.52 | 40.0 | 20 | 10.0 | 0 |
| T4_29 | 670 | 6.34 | 3.37 | 30.0 | 0 | 10.0 | 0 |
| T4_35 | 707 | 6.17 | 3.94 | 90.0 | 20 | 20.0 | 0 |
| T4_39 | 767 | 5.98 | 3.71 | 0.5 | 30 | 5.0 | 15 |
| T2_23 | 827 | 4.98 | 3.49 | 10.0 | 3 | 0.0 | 5 |
| T4_47 | 894 | 5.43 | 4.09 | 20.0 | 30 | 3.0 | 0 |
| T2_38 | 949 | 4.87 | 3.82 | 3.0 | 30 | 10.0 | 45 |
| T2_44 | 990 | 5.20 | 3.70 | 20.0 | 10 | 20.0 | 50 |
| T4_51 | 1007 | 5.02 | 3.55 | 80.0 | 10 | 20.0 | 25 |
| T2_50 | 1082 | 5.31 | 4.25 | 20.0 | 10 | 20.0 | 20 |
| T4_59 | 1150 | 4.62 | 3.08 | 80.0 | 10 | 10.0 | 0 |
| T2_54 | 1184 | 5.07 | 4.33 | 30.0 | 0 | 40.0 | 0 |
| T4_72° | 1220 | 4.21 | 2.99 | 90.0 | 0 | 0.5 | 0 |
| T4_78 | 1298 | 3.96 | 2.84 | 97.5 | 40 | 0.0 | 0 |
| T2_73° | 1318 | 4.32 | 3.52 | 97.5 | 0 | 10.0 | 0 |
| T2_Wsh | 1360 | 3.63 | 3.78 | 97.5 | 0 | 20.0 | 0 |
| T2_67 | 1420 | 3.63 | 3.16 | 97.5 | 0 | 5.0 | 0 |
| Parameter | Log Odds Ratio† | 95% CI | Z-value | p-value |
|---|---|---|---|---|
| Species diversity | 1.156 | (0.163, 2.148) | 2.282 | 0.022 |
| Deadwood layer coverage | 0.040 | (0.002, 0.077) | 2.075 | 0.038 |
| Mean annual solar radiation | -−0.758 | (−1.449, -(−0.066) | −2.148 | 0.032 |
| Mean annual temperature | -−0.501 | (-(−0.873, -(−0.129) | -−2.641 | 0.008 |
| Herb layer coverage | -−0.018 | (-(−0.029, -(−008) | -−3.368 | 0.001 |
| Shrub layer coverage | 0.011 | (-(−0.022, 0.043) | 0.651 | 0.515 |
| Beech coverage | -−0.002 | (-(−0.024, 0.021) | -−0.153 | 0.878 |
| Trapping index | 0.005 | (-(−0.024, 0.035) | 0.338 | 0.735 |
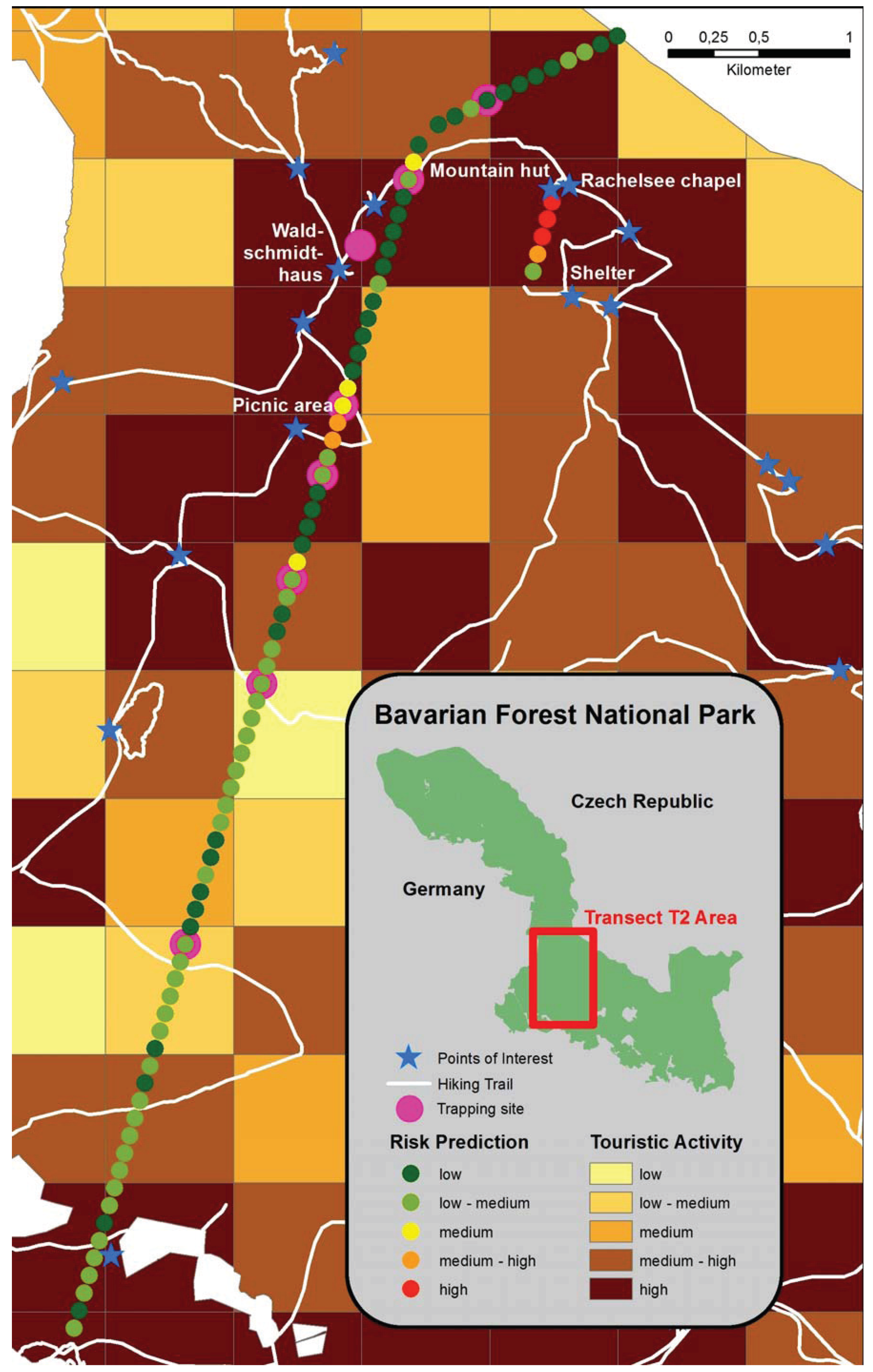
2.3. IllustrativeMap of Puumala Virus Prediction in Bank Voles
3. Experimental Section
3.1. The Study Area
3.2. Trapping Sites and Collection of Small Animals
3.3. Serological Investigation of Bank Voles for PUUV
3.4. Choice of Predictors of PUUV Seroprevalence
| Variable | Explanation |
|---|---|
| Trapping index | Number of bank voles/100 trapping nights (2008–2010, individually calculated for each site for each trapping event) |
| Species diversity | Shannon‘s diversity index of small mammals trapped with Sherman live traps (2008–2010, specifically calculated for each site for each trapping event) |
| Mean annual temperature | Mean annual temperature in °C (1980–2007) |
| Mean annual solar radiation | Mean annual solar radiation in kwh per m2 |
| Herb layer coverage | Percent coverage of herb layer (0.02 ha) recorded on 1 m altitude |
| Shrub layer coverage | Percent coverage of shrub layer (0.02 ha) recorded on 1–5 m altitude |
| Beech layer coverage | Percent coverage of beech upper layer (1 ha) recorded on >15 m altitude |
| Deadwood layer coverage | Percent coverage of deadwood layer (0.1ha) |
3.4.1. Trapping index
3.4.2. Small mammals’ diversity index
3.4.3. Mean Annual Solar Radiation
3.4.4. Mean Annual Temperature
3.4.5. Herblayer
3.4.6. Shrub Layer
3.4.7. Beech Layer Coverage
3.4.8. Dead wood layer coverage
3.5. Data Analyses
4. Conclusions
Supplementary Files
Supplementary File 1Acknowledgments
Conflicts of Interest
References and Notes
- Heyman, P.; Ceianu, C.S.; Christova, I.; Tordo, N.; Beersma, M.; João Alves, M.; Lundkvist, A.; Hukic, M.; Papa, A.; Tenorio, A.; et al. A five-year perspective on the situation of haemorrhagic fever with renal syndrome and status of the hantavirus reservoirs in Europe, 2005–2010. Euro Surveill. 2011, 16. [Google Scholar] [PubMed]
- Essbauer, S.; Krautkrämer, E. Hantaviruses —infections, epidemiology and hosts. In Zoonoses: Infections Affecting Men and Animals —A Focus on Public Health Aspects; Sing, A., Ed.; Springer Verlag: Heidelberg, Germany, 2015. [Google Scholar]
- Heyman, P.; Vaheri, A.; ENIVD Members. Situation of hantavirus infections and haemorrhagic fever with renal syndrome in European countries as of December 2006. Euro Surveill. 2008, 13. [Google Scholar] [PubMed]
- Mailles, A.; Sin, M.A.; Ducoffre, G.; Heyman, P.; Koch, J.; Zeller, H. Larger than usual increase in cases of hantavirus infections in Belgium, France and Germany, June 2005. Euro Surveill. 2005, 10. E050721.4. [Google Scholar] [PubMed]
- Heyman, P.; Cochez, C.; Ducoffre, G.; Mailles, A.; Zeller, H.; Abu Sin, M.; Koch, J.; van Doornum, G.; Koopmans, M.; Mossong, J.; Schneider, F. Haemorrhagic Fever with Renal Syndrome: An analysis of the outbreaks in Belgium, France, Germany, the Netherlands and Luxembourg in 2005. Euro Surveill. 2007, 12, E15–E16. [Google Scholar] [PubMed]
- Koch, J.; Brockmann, S.O.; Winter, C.; Kimmig, P.; Stark, K. Significant increase of hantavirus infections in Germany since the beginning of 2007. Euro Surveill. 2007, 12. E070503.1. [Google Scholar] [PubMed]
- Faber, M.S.; Ulrich, R.G.; Frank, C.; Brockmann, S.O.; Pfaff, G.M.; Jacob, J.; Krüger, D.H.; Stark, K. Steep rise in notified hantavirus infections in Germany, April 2010. Euro Surveill. 2010, 15. [Google Scholar] [PubMed]
- Hofmann, J.; Meisel, H.; Klempa, B.; Vesenbeckh, S.M.; Beck, R.; Michel, D.; Schmidt-Chanasit, J.; Ulrich, R.G.; Grund, S.; et al. Hantavirus outbreak, Germany, 2007. Emerging Infect. Dis. 2008, 14, 850–852. [Google Scholar] [CrossRef] [PubMed]
- Ettinger, J.; Hofmann, J.; Enders, M.; Tewald, F.; Oehme, R.M.; Rosenfeld, U.M.; Ali, H.S.; Schlegel, M.; Essbauer, S.; Osterberg, A.; et al. Multiple synchronous outbreaks of Puumala virus, Germany, 2010. Emerging Infect. Dis. 2012, 18, 1461–1464. [Google Scholar] [CrossRef] [PubMed]
- Krautkrämer, E.; Kruger, D.H.; Zeier, M. Recent outbreaks of hantavirus disease in Germany and in the United States. Kidney Int. 2012, 82, 1243–1245. [Google Scholar] [CrossRef] [PubMed]
- SurvStat@RKI – Abfrageergebnis. Available online: http://www3.rki.de/SurvStat/ResultList.aspx (accessed on 30 August 2014).
- Heyman, P.; Thoma, B.R.; Marié, J.-L.; Cochez, C.; Essbauer, S.S. In Search for Factors that Drive Hantavirus Epidemics. Front. Physiol. 2012, 3, 237. [Google Scholar] [CrossRef] [PubMed]
- Pilaski, J.; Ellerich, C.; Kreutzer, T.; Lang, A.; Benik, W.; Pohl-Koppe, A.; Bode, L.; Vanek, E.; Autenrieth, I.B.; Bigos, K. Haemorrhagic fever with renal syndrome in Germany. Lancet 1991, 337, 111. [Google Scholar] [CrossRef] [PubMed]
- Essbauer, S.; Schmidt, J.; Conraths, F.J.; Friedrich, R.; Koch, J.; Hautmann, W.; Pfeffer, M.; Wölfel, R.; Finke, J.; Dobler, G.; et al. A new Puumala hantavirus subtype in rodents associated with an outbreak of Nephropathia epidemica in South-East Germany in 2004. Epidemiol. Infect. 2006, 134, 1333–1344. [Google Scholar] [CrossRef] [PubMed]
- Schilling, S.; Emmerich, P.; Klempa, B.; Auste, B.; Schnaith, E.; Schmitz, H.; Krüger, D.H.; Günther, S.; Meisel, H. Hantavirus disease outbreak in Germany: Limitations of routine serological diagnostics and clustering of virus sequences of human and rodent origin. J. Clin. Microbiol. 2007, 45, 3008–3014. [Google Scholar] [CrossRef] [PubMed]
- Faber, M.; Wollny, T.; Schlegel, M.; Wanka, K.M.; Thiel, J.; Frank, C.; Rimek, D.; Ulrich, R.G.; Stark, K. Puumala Virus Outbreak in Western Thuringia, Germany, 2010: Epidemiology and Strain Identification. Zoonoses Public Health 2013, 60, 549–554. [Google Scholar] [CrossRef] [PubMed]
- Mertens, M.; Wölfel, R.; Ullrich, K.; Yoshimatsu, K.; Blumhardt, J.; Römer, I.; Esser, J.; Schmidt-Chanasit, J.; Groschup, M.H.; Dobler, G.; et al. Seroepidemiological study in a Puumala virus outbreak area in South-East Germany. Med. Microbiol. Immunol. 2009, 198, 83–91. [Google Scholar] [CrossRef] [PubMed]
- Mertens, M.; Kindler, E.; Emmerich, P.; Esser, J.; Wagner-Wiening, C.; Wölfel, R.; Petraityte-Burneikiene, R.; Schmidt-Chanasit, J.; Zvirbliene, A.; Groschup, M.H.; et al. Phylogenetic analysis of Puumala virus subtype Bavaria, characterization and diagnostic use of its recombinant nucleocapsid protein. Virus Genes 2011, 43, 177–191. [Google Scholar] [CrossRef] [PubMed]
- Escutenaire, S.; Chalon, P.; De Jaegere, F.; Karelle-Bui, L.; Mees, G.; Brochier, B.; Rozenfeld, F.; Pastoret, P.-P. Behavioral, physiologic, and habitat influences on the dynamics of Puumala virus infection in bank voles (Clethrionomys glareolus). Emerging Infect. Dis. 2002, 8, 930–936. [Google Scholar] [CrossRef] [PubMed]
- Olsson, G.E.; Ahlm, C.; Elgh, F.; Verlemyr, A.-C.; White, N.; Juto, P.; Palo, R.T. Hantavirus antibody occurrence in bank voles (Clethrionomys glareolus) during a vole population cycle. J. Wildl. Dis. 2003, 39, 299–305. [Google Scholar] [CrossRef] [PubMed]
- Olsson, G.E.; Leirs, H.; Henttonen, H. Hantaviruses and their hosts in Europe: Reservoirs here and there, but not everywhere? Vector Borne Zoonotic Dis. 2010, 10, 549–561. [Google Scholar] [CrossRef] [PubMed]
- Bernshtein, A.D.; Apekina, N.S.; Mikhailova, T.V.; Myasnikov, Y.A.; Khlyap, L.A.; Korotkov, Y.S.; Gavrilovskaya, I.N. Dynamics of Puumala hantavirus infection in naturally infected bank voles (Clethrinomys glareolus). Arch. Virol. 1999, 144, 2415–2428. [Google Scholar] [CrossRef] [PubMed]
- Olsson, G.E.; White, N.; Ahlm, C.; Elgh, F.; Verlemyr, A.-C.; Juto, P.; Palo, R.T. Demographic factors associated with hantavirus infection in bank voles (Clethrionomys glareolus). Emerging Infect. Dis. 2002, 8, 924–929. [Google Scholar] [CrossRef] [PubMed]
- Deter, J.; Chaval, Y.; Galan, M.; Gauffre, B.; Morand, S.; Henttonen, H.; Laakkonen, J.; Voutilainen, L.; Charbonnel, N.; Cosson, J.-F. Kinship, dispersal and hantavirus transmission in bank and common voles. Arch. Virol. 2008, 153, 435–444. [Google Scholar] [CrossRef] [PubMed]
- Augot, D.; Sauvage, F.; Boue, F.; Bouloy, M.; Artois, M.; Demerson, J.M.; Combes, B.; Coudrier, D.; Zeller, H.; Cliquet, F.; et al. Spatial and temporal patterning of bank vole demography and the epidemiology of the Puumala hantavirus in northeastern France. Epidemiol. Infect. 2008, 136, 1638–1643. [Google Scholar] [CrossRef]
- Tersago, K.; Verhagen, R.; Leirs, H. Temporal variation in individual factors associated with hantavirus infection in bank voles during an epizootic: Implications for Puumala virus transmission dynamics. Vector Borne Zoonotic Dis. 2011, 11, 715–721. [Google Scholar] [CrossRef] [PubMed]
- Hanski I., H.H. Temporal Variability and Geographical Patterns in the Population Density of Microtine Rodents: A Reply to Xia and Boonstra. American Naturalist 1994, 144, 329–342. [Google Scholar] [CrossRef]
- Davis, S.; Calvet, E.; Leirs, H. Fluctuating rodent populations and risk to humans from rodent-borne zoonoses. Vector Borne Zoonotic Dis. 2005, 5, 305–314. [Google Scholar] [CrossRef] [PubMed]
- Maillard, J.-C.; Sparagano, O.A.E. Animal biodiversity and emerging diseases prediction and prevention. Introduction. Ann. N. Y. Acad. Sci. 2008, 1149, xvii–xix. [Google Scholar] [CrossRef] [PubMed]
- Jones, K.E.; Patel, N.G.; Levy, M.A.; Storeygard, A.; Balk, D.; Gittleman, J.L.; Daszak, P. Global trends in emerging infectious diseases. Nature 2008, 451, 990–993. [Google Scholar] [CrossRef] [PubMed]
- Aguirre, A.A.; Tabor, G.M. Global factors driving emerging infectious diseases. Ann. N. Y. Acad. Sci. 2008, 1149, 1–3. [Google Scholar] [CrossRef] [PubMed]
- Kraus, A.A.; Priemer, C.; Heider, H.; Kruger, D.H.; Ulrich, R. Inactivation of Hantaan virus-containing samples for subsequent investigations outside biosafety level 3 facilities. Intervirology 2005, 48, 255–261. [Google Scholar] [CrossRef] [PubMed]
- Kallio, E.R.; Klingström, J.; Gustafsson, E.; Manni, T.; Vaheri, A.; Henttonen, H.; Vapalahti, O.; Lundkvist, A. Prolonged survival of Puumala hantavirus outside the host: Evidence for indirect transmission via the environment. J. Gen. Virol 2006, 87, 2127–2134. [Google Scholar] [CrossRef] [PubMed]
- Piechotowski, I.; Brockmann, S.O.; Schwarz, C.; Winter, C.H.; Ranft, U.; Pfaff, G. Emergence of hantavirus in South Germany: rodents, climate and human infections. Parasitol. Res. 2008, 103, S131–S137. [Google Scholar] [CrossRef] [PubMed]
- Schwarz, A.C.; Ranft, U.; Piechotowski, I.; Childs, J.E.; Brockmann, S.O. Risk Factors for Human Infection with Puumala Virus, Southwestern Germany. Emerg. Infect. Dis. 2009, 15, 1032–1039. [Google Scholar] [CrossRef]
- Suchomel, J. Populations of Clethrionomys glareolus in three isolated forest complexes in rural southern Moravia (Czech Republic). Hystrix 2006, 17, 155–160. [Google Scholar]
- Haapakoski, M.; Ylönen, H. Effects of fragmented breeding habitat and resource distribution on behavior and survival of the bank vole (Myodes glareolus ). Popul. Ecol. 2010, 52, 427–435. [Google Scholar] [CrossRef]
- Fey, K.; Banks, P.B.; Korpimäki, E. Different microhabitat preferences of field and bank voles under manipulated predation risk from an alien predator. Annales zoologici Fennici 2013, 43, 9–16. [Google Scholar]
- Bujalska, G.; Grüm, L. Social organization of the bank vole (Clethrionomys glareolus, Schreber 1780) and its demographic consequences: A model. Oecologia 1989, 80, 70–81. [Google Scholar] [CrossRef] [PubMed]
- Linard, C.; Tersago, K.; Leirs, H.; Lambin, E.F. Environmental conditions and Puumala virus transmission in Belgium. Int. J. Health Geogr. 2007, 6, 55. [Google Scholar] [CrossRef] [PubMed]
- Tersago, K.; Schreurs, A.; Linard, C.; Verhagen, R.; Van Dongen, S.; Leirs, H. Population, environmental, and community effects on local bank vole (Myodes glareolus ) Puumala virus infection in an area with low human incidence. Vector Borne Zoonotic Dis. 2008, 8, 235–244. [Google Scholar] [CrossRef] [PubMed]
- Olsson, G.E.; White, N.; Hjältén, J.; Ahlm, C. Habitat factors associated with bank voles (Clethrionomys glareolus) and concomitant hantavirus in northern Sweden. Vector Borne Zoonotic Dis. 2005, 5, 315–323. [Google Scholar] [CrossRef] [PubMed]
- Siitonen, J. Forest management, coarse woodydebris and saproxylic organisms: Fennoscandian boreal forests as an example. Ecol. Bull. 2001, 49, 11–41. [Google Scholar]
- Maguire, C. Dead Wood and the Richness of Small Terrestrial Vertebrates in Southwestern Oregon. USDA Forest Service Gen. Tech. Rep. 2002, PSW-GTR-181, 333–345. [Google Scholar]
- Clement, J.; Vercauteren, J.; Verstraeten, W.W.; Ducoffre, G.; Barrios, J.M.; Vandamme, A.-M.; Maes, P.; Ranst, M.V. Relating increasing hantavirus incidences to the changing climate: The mast connection. Int. J. Health Geogr. 2009, 8, 1. [Google Scholar] [CrossRef] [PubMed]
- Tersago, K.; Verhagen, R.; Servais, A.; Heyman, P.; Ducoffre, G.; Leirs, H. Hantavirus disease (nephropathia epidemica) in Belgium: Effects of tree seed production and climate. Epidemiol. Infect. 2009, 137, 250–256. [Google Scholar] [CrossRef] [PubMed]
- Clement, J.; Maes, P.; van Ypserle deStrihou, C.; van derGroen, G.; Barrios, J.M.; Verstraeten, W.W.; van Ranst, M. Beechnuts and outbreaks of nephropathia epidemica (NE): Of mast, mice and men. Nephrol. Dial. Transplant. 2010, 25, 1740–1746. [Google Scholar] [CrossRef] [PubMed]
- Haredasht, S.A.; Taylor, C.J.; Maes, P.; Verstraeten, W.W.; Clement, J.; Barrios, M.; Lagrou, K.; Van Ranst, M.; Coppin, P.; Berckmans, D.; Aerts, J.-M. Model-based prediction of nephropathia epidemica outbreaks based on climatological and vegetation data and bank vole population dynamics. Zoonoses Public Health 2013, 60, 461–477. [Google Scholar] [CrossRef] [PubMed]
- Zeimes, C.B.; Olsson, G.E.; Ahlm, C.; Vanwambeke, S.O. Modelling zoonotic diseases in humans: Comparison of methods for hantavirus in Sweden. Int. J. Health Geogr. 2012, 11, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Zejda, J. Ecology and control of Clethrionomys glareolus Schreber in Czechoslovakia. EPPO Publ. Set. A. 1970, 58, 101–105. [Google Scholar]
- Andrzejewski, R.; Mazurkiewicz, M. Abundance of food supply and size of the bank vole’s home range. Acta theriol. 1976, 21, 237–253. [Google Scholar] [CrossRef]
- Korn, H. Changes in home range size during growth and maturation of the wood mouse (Apodemus sylvaticus) and the bank vole (Clethrionomys glareolus). Oecologica 1986, 68, 623–628. [Google Scholar] [CrossRef]
- Bäumler, D.W. Die Verbreitung von Mäusen in verschiedenen Waldgebieten Bayerns. Anz. Schadlingskde., Pflanzenschutz, Umweltschutz 1981, 54, 99–104. [Google Scholar] [CrossRef]
- Bäumler, D.W.; Brunner, M. Einfluß des Nahrungsangebots auf die Konkurrenz sympatrischer Mäusearten in Forstkulturen. Anz. Schadlingskde., Pflanzenschutz, Umweltschutz 1988, 61, 3–5. [Google Scholar] [CrossRef]
- Mappes, T.; Koskela, E.; Ylonen, H. Reproductive costs and litter size in the bank vole. Proc Biol Sci. 1995, 261, 19–24. [Google Scholar] [CrossRef] [PubMed]
- Koskela, E.; Mappes, T.; Ylonen, H. Territorial behaviour and reproductive success of bank vole Clethrionomys glareolus females. J. Anim. Ecol. 1997, 66, 341. [Google Scholar]
- Haredasht, S.A.; Barrios, M.; Farifteh, J.; Maes, P.; Clement, J.; Verstraeten, W.W.; Tersago, K.; Van Ranst, M.; Coppin, P.; Berckmans, D.; et al. Ecological niche modelling of bank voles in Western Europe. Int. J. Environ. Res. Public Health 2013, 10, 499–514. [Google Scholar]
- Barrios, J.M.; Verstraeten, W.W.; Maes, P.; Aerts, J.M.; Farifteh, J.; Coppin, P. Relating land cover and spatial distribution of nephropathia epidemica and Lyme borreliosis in Belgium. Int. J. Environ. Health Res. 2013, 23, 132–154. [Google Scholar] [CrossRef] [PubMed]
- Glaser, R.; Gebhardt, H.; Schenk, W. Geographie Deutschlands; Wissenschaftliche Buchgesellschaft: Darmstadt, Germay, 2007. [Google Scholar]
- Bässler, C.; Förster, B.; Moning, C.; Müller, J. Biodiversity Research between Climate Change and Wilding in a temperate montane forest – The conceptual framework. For. Landsc. Res. 2009, 7, 21–33. [Google Scholar]
- Schex, S.; Dobler, G.; Riehm, J.; Müller, J.; Essbauer, S. Rickettsia spp. in wild small mammals in Lower Bavaria, South-Eastern Germany. Vector Borne Zoonotic Dis. 2011, 11, 493–502. [Google Scholar] [CrossRef] [PubMed]
- Essbauer, S.; Osterberg, A.; Schex, S.; Müller, J. Molecular epidemiology of Puumala virus in the Bavarian Forest National Park. Unpublished work. 2014. [Google Scholar]
- Müller, J.; Bässler, C.; Essbauer, S.; Schex, S.; Müller, D.; Opgenoorth, L.; Brandl, R. Relative heart size but not body size increases with elevation within the populations of two rodent species. J. Biogeog. 2014, in press. [Google Scholar]
- Essbauer, S.; Dobler, G.; Zöller, L.; Lüke, P.; Friedewald, S.; Wölfel, R. Hantavirus-Herdanalyse zur Abschätzung des Infektionsrisikos am Truppenübungsplatz Heuberg. Wehrmedizinische Monatsschrift 2010, 54, 84. [Google Scholar]
- Boone, I.; Wagner-Wiening, C.; Reil, D.; Jacob, J.; Rosenfeld, U.M.; Ulrich, R.G.; Lohr, D.; Pfaff, G. Rise in the number of notified human hantavirus infections since October 2011 in Baden-Wurttemberg, Germany. Euro Surveill. 2012, 17. [Google Scholar] [PubMed]
- Yahnke, C.J.; Meserve, P.L.; Ksiazek, T.G.; Mills, J.N. Patterns of infection with Laguna Negra virus in wild populations of Calomys laucha in the central Paraguayan chaco. Am. J. Trop. Med. Hyg. 2001, 65, 768–776. [Google Scholar] [PubMed]
- Ruedas, L.A.; Salazar-Bravo, J.; Tinnin, D.S.; Armién, B.; Cáceres, L.; García, A.; Díaz, M.A.; Gracia, F.; Suzán, G.; Peters, C.J.; et al. Community ecology of small mammal populations in Panamá following an outbreak of Hantavirus pulmonary syndrome. J. Vector Ecol. 2004, 29, 177–191. [Google Scholar] [PubMed]
- Peixoto, I.D.; Abramson, G. The effect of biodiversity on the hantavirus epizootic. Ecology 2006, 87, 873–879. [Google Scholar] [CrossRef] [PubMed]
- Mills, J.N. Biodiversity loss and emerging infectious disease: An example from the rodent-borne hemorrhagic fevers. Biodiversity 2006, 7, 9–17. [Google Scholar] [CrossRef]
- Keesing, F.; Holt, R.D.; Ostfeld, R.S. Effects of species diversity on disease risk. Ecol. Lett. 2006, 9, 485–498. [Google Scholar] [CrossRef] [PubMed]
- Dizney, L.J.; Ruedas, L.A. Increased host species diversity and decreased prevalence of Sin Nombre virus. Emerging Infect. Dis. 2009, 15, 1012–1018. [Google Scholar] [CrossRef] [PubMed]
- Suzán, G.; Marcé, E.; Giermakowski, J.T.; Mills, J.N.; Ceballos, G.; Ostfeld, R.S.; Armién, B.; Pascale, J.M.; Yates, T.L. Experimental evidence for reduced rodent diversity causing increased hantavirus prevalence. PLoS One 2009, 4, e5461. [Google Scholar] [CrossRef] [PubMed]
- Derne, B.T.; Fearnley, E.J.; Lau, C.L.; Paynter, S.; Weinstein, P. Biodiversity and leptospirosis risk: A case of pathogen regulation? Med. Hypotheses 2011, 77, 339–344. [Google Scholar] [CrossRef]
- Clay, C.A.; Lehmer, E.M.; Jeor, S.S.; Dearing, M.D. Sin nombre virus and rodent species diversity: A test of the dilution and amplification hypotheses. PLoS One 2009, 4, e6467. [Google Scholar] [CrossRef] [PubMed]
- Keesing, F.; Belden, L.K.; Daszak, P.; Dobson, A.; Harvell, C.D.; Holt, R.D.; Hudson, P.; Jolles, A.; Jones, K.E.; Mitchell, C.E.; et al. Impacts of biodiversity on the emergence and transmission of infectious diseases. Nature 2010, 468, 647–652. [Google Scholar] [CrossRef] [PubMed]
- Klingström, J.; Heyman, P.; Escutenaire, S.; Sjölander, K.B.; De Jaegere, F.; Henttonen, H.; Lundkvist, A. Rodent host specificity of European hantaviruses: Evidence of Puumala virus interspecific spillover. J. Med. Virol. 2002, 68, 581–588. [Google Scholar] [CrossRef] [PubMed]
- Holsomback, T.S.; McIntyre, N.E.; Nisbett, R.A.; Strauss, R.E.; Chu, Y.-K.; Abuzeineh, A.A.; de la Sancha, N.; Dick, C.W.; Jonsson, C.B.; Morris, B.E.L. Bayou virus detected in non-oryzomyine rodent hosts: An assessment of habitat composition, reservoir community structure, and marsh rice rat social dynamics. J. Vector Ecol. 2009, 34, 9–21. [Google Scholar] [CrossRef] [PubMed]
- Lehmer, E.M.; Korb, J.; Bombaci, S.; McLean, N.; Ghachu, J.; Hart, L.; Kelly, A.; Jara-Molinar, E.; O’Brien, C.; Wright, K. The interplay of plant and animal disease in a changing landscape: The role of sudden aspen decline in moderating Sin Nombre virus prevalence in natural deer mouse populations. Ecohealth 2012, 9, 205–216. [Google Scholar] [CrossRef] [PubMed]
- Bunnell, F.; Houde, I.; Johnston, B.; Wind, E. How dead trees sustain live organisms in western forests. USDA Forest Service Gen. Tech. Rep. 2002, PSW-GTR-181, 291–318. [Google Scholar]
- Dobly, A.; Yzoard, C.; Cochez, C.; Ducoffre, G.; Aerts, M.; Roels, S.; Heyman, P. Spatiotemporal dynamics of Puumala hantavirus in suburban reservoir rodent populations. J. Vector Ecol. 2012, 37, 276–283. [Google Scholar] [CrossRef] [PubMed]
- Ulrich, R.G.; Schmidt-Chanasit, J.; Schlegel, M.; Jacob, J.; Pelz, H.-J.; Mertens, M.; Wenk, M.; Büchner, T.; Masur, D.; Sevke, K.; et al. Network “Rodent-borne pathogens” in Germany: Longitudinal studies on the geographical distribution and prevalence of hantavirus infections. Parasitol. Res. 2008, 103, S121–S129. [Google Scholar] [CrossRef] [PubMed]
- Heyman, P.; Vervoort, T.; Escutenaire, S.; Degrave, E.; Konings, J.; Vandenvelde, C.; Verhagen, R. Incidence of hantavirus infections in Belgium. Virus Res. 2001, 77, 71–80. [Google Scholar] [CrossRef] [PubMed]
- Sauvage, F.; Langlais, M.; Yoccoz, N.G.; Pontier, D. Modelling Hantavirus in Fluctuating Populations of Bank Voles: The Role of Indirect Transmission on Virus Persistence. J. Anim. Ecol. 2003, 72, 1–13. [Google Scholar]
- Balciauskiene, L.; Balciauskas, L.; Cepukiene, A. Growth of the bank vole Myodes glareolus in the non-vegetative period in NE Lithuania. Est. J. Ecol. 2009, 58, 86–93. [Google Scholar] [CrossRef]
- Evander, M.; Ahlm, C. Milder winters in northern Scandinavia may contribute to larger outbreaks of haemorrhagic fever virus. Global Health Action 2009, 2. [Google Scholar] [CrossRef]
- Guivier, E.; Galan, M.; Chaval, Y.; Xuéreb, A.; Ribas Salvador, A.; Poulle, M.-L.; Voutilainen, L.; Henttonen, H.; Charbonnel, N.; Cosson, J.F. Landscape genetics highlights the role of bank vole metapopulation dynamics in the epidemiology of Puumala hantavirus. Mol. Ecol. 2011, 20, 3569–3583. [Google Scholar] [PubMed]
- Anderson, L.S.; Sinclair, F.L. Ecological interactions in agroforestry systems. Agroforestry Abstracts 1993, 6, 57–91. [Google Scholar]
- Schmidt, W. Herb layer species as indicators of biodiversity of managed and unmanaged beech forests. For. Snow Landsc. 2005, 79, 111–126. [Google Scholar]
- Viro, P.; Niethammer, J. Clethrionomys glareolus (Schreber, 1780) - Rötelmaus. In Handbuch der Säugetiere Europas; Nagetiere; Aula Verlag: Wiesbaden, 1982. [Google Scholar]
- Olsson, G.E.; White, N.; Hjältén, J.; Ahlm, C. Habitat factors associated with bank voles (Clethrionomys glareolus) and concomitant hantavirus in northern Sweden. Vector Borne Zoonotic Dis. 2005, 5, 315–323. [Google Scholar] [CrossRef] [PubMed]
- Heyman, P.; Mele, R.V.; Smajlovic, L.; Dobly, A.; Cochez, C.; Vandenvelde, C. Association between habitat and prevalence of hantavirus infections in bank voles (Myodes glareolus ) and wood mice (Apodemus sylvaticus). Vector Borne Zoonotic Dis. 2009, 9, 141–146. [Google Scholar] [CrossRef] [PubMed]
- Engelthaler, D.M.; Mosley, D.G.; Cheek, J.E.; Levy, C.E.; Komatsu, K.K.; Ettestad, P.; Davis, T.; Tanda, D.T.; Miller, L.; Frampton, J.W.; et al. Climatic and environmental patterns associated with hantavirus pulmonary syndrome, Four Corners region, United States. Emerging Infect. Dis. 1999, 5, 87–94. [Google Scholar] [CrossRef] [PubMed]
- Glass, G.E.; Shields, T.; Cai, B.; Yates, T.L.; Parmenter, R. Persistently highest risk areas for hantavirus pulmonary syndrome: Potential sites for refugia. Ecol. Appl. 2007, 17, 129–139. [Google Scholar] [CrossRef] [PubMed]
- Palo, R.T. Time series analysis performed on nephropathia epidemica in humans of northern Sweden in relation to bank vole population dynamic and the NAO index. Zoonoses Public Health 2009, 56, 150–156. [Google Scholar] [CrossRef] [PubMed]
- Ostfeld, R.; Keesing, F. The function of biodiversity in the ecology of vector-borne zoonotic diseases. Can. J. Zool. 2000, 78, 2061–2078. [Google Scholar] [CrossRef]
- LoGiudice, K.; Ostfeld, R.S.; Schmidt, K.A.; Keesing, F. The ecology of infectious disease: effects of host diversity and community composition on Lyme disease risk. Proc. Natl. Acad. Sci. USA 2003, 100, 567–571. [Google Scholar] [CrossRef] [PubMed]
- Bässler, C.; Müller, J.; Dziock, F. Identification of climate sensitive zones for plants in montane forests. Folia Geobotanica 2010, 45, 163–182. [Google Scholar] [CrossRef]
- Kühn, K.; Reil, D.; Imholt, C.; Mattes, H.; Jacob, J. Zusammenhang zwischen Mikrohabitatstrukturen, Nahrungsverfügbarkeit und Abundanz von Waldnagern. Julius-Kühn-Archiv 2011, 430, 62. [Google Scholar]
- Rösner, S.; Mussard-Forster, E.; Lorenc, T.; Müller, J. Recreation shapes a “landscape of fear” for a threatened forest bird species in Central Europe. Landscape Ecol. 2014, 29, 55–66. [Google Scholar] [CrossRef]
- Franklin, J.F. Preserving Biodiversity: Species, Ecosystems, or Landscapes? Ecological Applications 1993, 3, 202. [Google Scholar] [CrossRef]
- Vapalahti, K.; Virtala, A.-M.; Vaheri, A.; Vapalahti, O. Case-control study on Puumala virus infection: smoking is a risk factor. Epidemiol. Infect. 2010, 138, 576–584. [Google Scholar] [CrossRef] [PubMed]
- Winter, C.H.; Brockmann, S.O.; Piechotowski, I.; Alpers, K.; an der Heiden, M.; Koch, J.; Stark, K.; Pfaff, G. Survey and case-control study during epidemics of Puumala virus infection. Epidemiol. Infect. 2009, 137, 1479–1485. [Google Scholar] [CrossRef] [PubMed]
- Barrios, J.M.; Verstraeten, W.W.; Maes, P.; Clement, J.; Aerts, J.-M.; Haredasht, S.A.; Wambacq, J.; Lagrou, K.; Ducoffre, G.; Van Ranst, M.; et al. Satellite derived forest phenology and its relation with nephropathia epidemica in Belgium. Int. J. Environ. Res. Public Health 2010, 7, 2486–2500. [Google Scholar] [CrossRef] [PubMed]
- Salvador, A.R.; Guivier, E.; Xuéreb, A.; Chaval, Y.; Cadet, P.; Poulle, M.-L.; Sironen, T.; Voutilainen, L.; Henttonen, H.; Cosson, J.-F.; et al. Concomitant influence of helminth infection and landscape on the distribution of Puumala hantavirus in its reservoir, Myodes glareolus. BMC Microbiol. 2011, 11, 30. [Google Scholar] [CrossRef] [PubMed]
- Crowcroft, N.S.; Infuso, A.; Ilef, D.; Le Guenno, B.; Desenclos, J.C.; Van Loock, F.; Clement, J. Risk factors for human hantavirus infection: Franco-Belgian collaborative case-control study during 1995-6 epidemic. Brit. Med. J. 1999, 318, 1737–1738. [Google Scholar] [CrossRef] [PubMed]
- Zöller, L.; Faulde, M.; Meisel, H.; Ruh, B.; Kimmig, P.; Schelling, U.; Zeier, M.; Kulzer, P.; Becker, C.; Roggendorf, M. Seroprevalence of hantavirus antibodies in Germany as determined by a new recombinant enzyme immunoassay. Eur. J. Clin. Microbiol. Infect. Dis. 1995, 14, 305–313. [Google Scholar] [CrossRef] [PubMed]
- Mertens, M.; Hofmann, J.; Petraityte-Burneikiene, R.; Ziller, M.; Sasnauskas, K.; Friedrich, R.; Niederstrasser, O.; Krüger, D.H.; Groschup, M.H.; Petri, E.; Werdermann, S.; Ulrich, R.G. Seroprevalence study in forestry workers of a non-endemic region in eastern Germany reveals infections by Tula and Dobrava-Belgrade hantaviruses. Med. Microbiol. Immunol. 2011, 200, 263–268. [Google Scholar] [CrossRef] [PubMed]
- Elling, W.; Bauer, E.; Klemm, G.; Koch, H. Klima und Böden. Bayerisches Staatsministerium für Ernährung, Landwirtschaft und Forsten; Nationalparkverwaltung Bayerischer Wald, Wissenschaftliche Schriftenreihe Heft 1: Grafenau, Germany, 1987. [Google Scholar]
- Noack, E. Witterung und Klima im Bayerischen Wald; Bayerisches Staatsministerium für Ernährung, Landwirtschaft und Forsten; Nationalparkverwaltung Bayerischer Wald, Wissenschaftliche Schriftenreihe Heft 5: Grafenau, Germany, 1979. [Google Scholar]
- Bässler, C. Das Klima im Nationalpark Bayerischer Wald - Darstellung, Entwicklung und Auswirkung; Bayerisches Staatsministerium für Ernährung, Landwirtschaft und Forsten; Nationalparkverwaltung Bayerischer Wald: Grafenau, Germany, 2004. [Google Scholar]
- Walentowski, H.; Ewald, J.; Fischer, A.; Kölling, C.; Türk, W. Handbuch der natürlichen Waldgesellschaften in Bayern; Geobotanica: Freising, Coutnry, 2004. [Google Scholar]
- Shannon, C. A mathematical theory of communication. Bell Syst. Tech. J. 1948, 27, 623–656. [Google Scholar] [CrossRef]
- Rosenzweig, M.L. Species diversity in space and time; Cambridge University Press: Cambridge, UK, 1995. [Google Scholar]
- Böhner, J.; McCloy, K.; Strobl, J. SAGA-Analysis and Modelling Applications. 2006. [Google Scholar]
- Hydrologisches Modellierungssystem ArcEGMO. Available online: http://www.arcegmo.de (accessed on 15 July 2011).
- R Core Team R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2013.
- The R Project for Statistical Computing. version 2.15.2. Available online: http://www.r-project.org (accessed on 15 July 2011).
- ArcGISTM, version 10.1; ESRI: Redlands, State Abbreviation, USA, 2010.
- Mills, J.N. Regulation of rodent-borne viruses in the natural host: Implications for human disease. Arch. Virol. Suppl. 2005, 19, 45–57. [Google Scholar] [PubMed]
- Schmaljohn, C.S. Vaccines for hantaviruses: Progress and issues. Expert. Rev. Vaccines 2012, 11, 511–513. [Google Scholar] [CrossRef] [PubMed]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Thoma, B.R.; Müller, J.; Bässler, C.; Georgi, E.; Osterberg, A.; Schex, S.; Bottomley, C.; Essbauer, S.S. Identification of FactorsInfluencing the Puumala Virus Seroprevalence within Its Reservoir in aMontane Forest Environment. Viruses 2014, 6, 3944-3967. https://doi.org/10.3390/v6103944
Thoma BR, Müller J, Bässler C, Georgi E, Osterberg A, Schex S, Bottomley C, Essbauer SS. Identification of FactorsInfluencing the Puumala Virus Seroprevalence within Its Reservoir in aMontane Forest Environment. Viruses. 2014; 6(10):3944-3967. https://doi.org/10.3390/v6103944
Chicago/Turabian StyleThoma, Bryan R., Jörg Müller, Claus Bässler, Enrico Georgi, Anja Osterberg, Susanne Schex, Christian Bottomley, and Sandra S. Essbauer. 2014. "Identification of FactorsInfluencing the Puumala Virus Seroprevalence within Its Reservoir in aMontane Forest Environment" Viruses 6, no. 10: 3944-3967. https://doi.org/10.3390/v6103944
APA StyleThoma, B. R., Müller, J., Bässler, C., Georgi, E., Osterberg, A., Schex, S., Bottomley, C., & Essbauer, S. S. (2014). Identification of FactorsInfluencing the Puumala Virus Seroprevalence within Its Reservoir in aMontane Forest Environment. Viruses, 6(10), 3944-3967. https://doi.org/10.3390/v6103944




