Molecular Modeling of Prion Transmission to Humans
Abstract
:1. Introduction
2. Materials and Methods
2.1. Seeds and Substrates
| Mouse Line | Reference | Transgene | Expression Rate (as Compared with) | Supplier |
|---|---|---|---|---|
| Tg650 | [18] | HuPrPMet | x6 (human brain) | H. Laude, INRA, Jouy-en-Josas, France |
| Tg340 | [19] | HuPrPMet | x4 (human brain) | J-M. Torres, CISA-INIA, Madrid, Spain |
| Tg152 | [20] | HuPrPVal | x4–8 (human brain) | S. Prusiner, UCSF, San Francisco, USA |
| Tg362 | [21] | HuPrPVal | x8 (human brain) | J-M. Torres, CISA-INIA, Madrid, Spain |
| Tg4092 | [22] | BovPrP | x8–16 (bovine brain) | S. Prusiner, UCSF, San Francisco, USA |
| Tg110 | [23] | BovPrP | x8 (bovine brain) | J-M. Torres, CISA-INIA, Madrid, Spain |
| RIII | [24] | - | endogenous MoPrP | F. Cortade, INRA, Nouzilly, France |
2.2. Brain Homogenate Preparation
2.3. Protein Misfolding Cyclic Amplification (PMCA)
2.4. Western Blotting
2.5. Statistical Analysis
3. Results
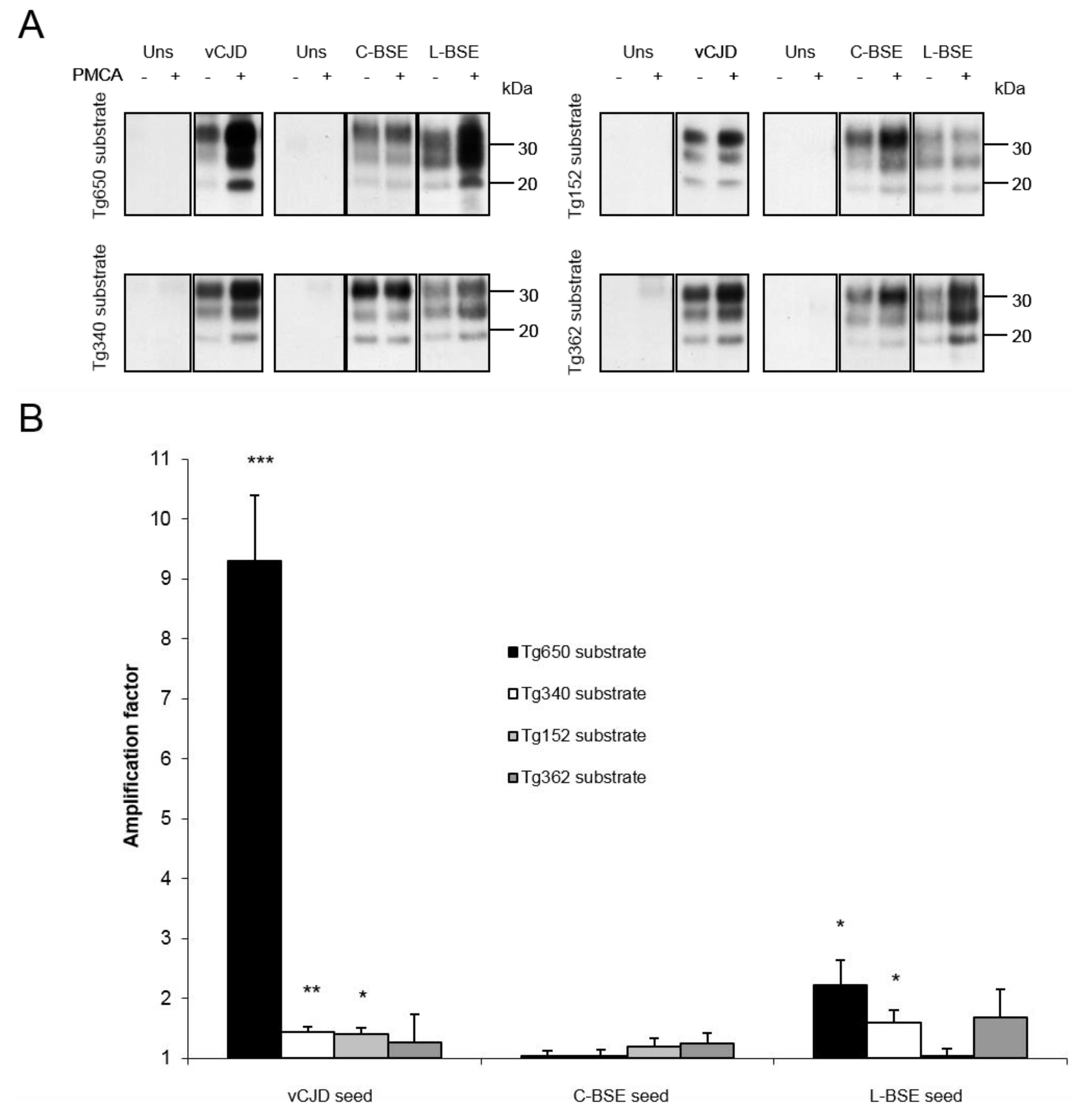
| Inoculum: | vCJD | C-BSE | L-BSE | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mouse line: | Tg650 | Tg340 | Tg152 | Tg362 | Tg650 | Tg340 | Tg152 | Tg362 | Tg650 | Tg340 | Tg152 | Tg362 |
| Attack rate: | 100% | 100% | ~50% | 0% | <33% | <20% | <50% | 0% | 100% | 100% | NA | 0% |
| Reference: | [18] | [19] | [28] | [21] | [13] | [19] | [28] | [21] | [13] | [21] | NA | [21] |
| Amplification factor: | 9.3 | 1.4 | 1.4 | 1.3 | 1 | 1 | 1.2 | 1.3 | 2.2 | 1.6 | 1 | 1.7 |
| Concordance with PMCA results: | + | + | + | + | + | + | + | + | + | + | ? | + |
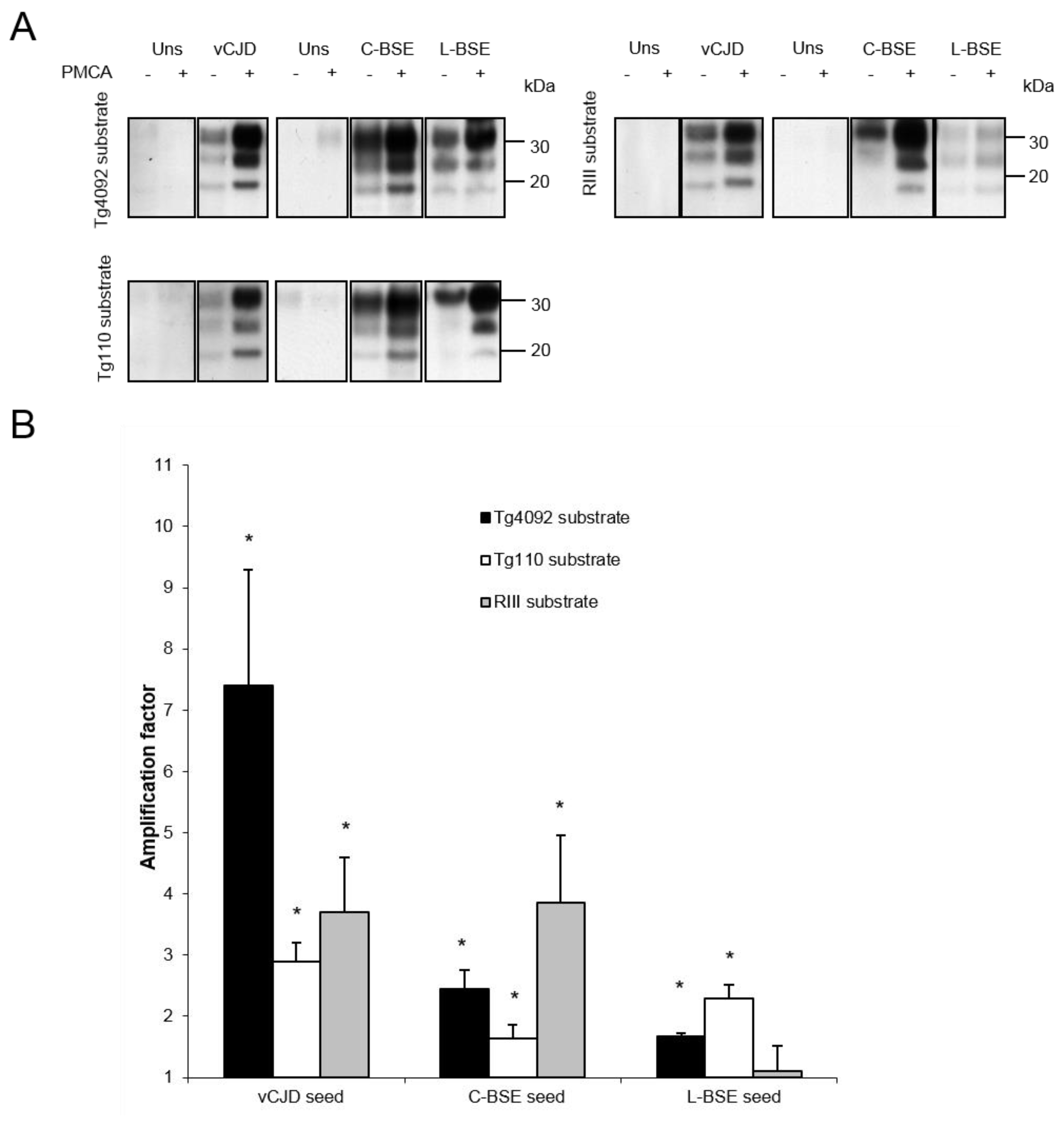
| Inoculum: | vCJD | C-BSE | L-BSE | |||||||||
| Mouse line: | Tg4092 | Tg110 | RIII | Tg4092 | Tg110 | RIII | Tg4092 | Tg110 | RIII | |||
| Attack rate: | 100% | 100% | 100% | 100% | 100% | 100% | NA | 100% | 0% | |||
| Reference: | [29] | [23] | [30] | [22,29] | [23] | [30,31] | NA | [21] | [31] | |||
| Amplification factor: | 7.4 | 2.9 | 3.7 | 2.5 | 1.6 | 3.9 | 1.7 | 2.3 | 1.1 | |||
| Concordance witd PMCA results: | + | + | + | + | + | + | ? | + | + | |||
4. Discussion
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Prusiner, S.B. Prions. Proc. Natl. Acad. Sci. USA 1998, 95, 13363–13383. [Google Scholar] [CrossRef] [PubMed]
- Ward, H.J.; Everington, D.; Cousens, S.N.; Smith-Bathgate, B.; Leitch, M.; Cooper, S.; Heath, C.; Knight, R.S.; Smith, P.G.; Will, R.G. Risk factors for variant creutzfeldt-jakob disease: A case-control study. Ann. Neurol. 2006, 59, 111–120. [Google Scholar] [CrossRef] [PubMed]
- Brandel, J.P.; Heath, C.A.; Head, M.W.; Levavasseur, E.; Knight, R.; Laplanche, J.L.; Langeveld, J.P.; Ironside, J.W.; Hauw, J.J.; Mackenzie, J.; et al. Variant creutzfeldt-jakob disease in france and the united kingdom: Evidence for the same agent strain. Ann. Neurol. 2009, 65, 249–256. [Google Scholar] [CrossRef] [PubMed]
- Brandel, J.P.; Galanaud, D.; Freeman, L.; Laplanche, J.L.; Haik, S. Variant or sporadic creutzfeldt-jakob disease? Lancet, 2010; 375, 889, author reply 889–890. [Google Scholar]
- Kaski, D.; Mead, S.; Hyare, H.; Cooper, S.; Jampana, R.; Overell, J.; Knight, R.; Collinge, J.; Rudge, P. Variant cjd in an individual heterozygous for prnp codon 129. Lancet 2009, 374, 2128. [Google Scholar] [CrossRef] [PubMed]
- Jones, M.; Peden, A.H.; Prowse, C.V.; Groner, A.; Manson, J.C.; Turner, M.L.; Ironside, J.W.; MacGregor, I.R.; Head, M.W. In vitro amplification and detection of variant creutzfeldt-jakob disease prpsc. J. Pathol. 2007, 213, 21–26. [Google Scholar] [CrossRef] [PubMed]
- Jones, M.; Wight, D.; Barron, R.; Jeffrey, M.; Manson, J.; Prowse, C.; Ironside, J.W.; Head, M.W. Molecular model of prion transmission to humans. Emerg. Infect. Dis. 2009, 15, 2013–2016. [Google Scholar] [CrossRef] [PubMed]
- Castilla, J.; Gonzalez-Romero, D.; Saa, P.; Morales, R.; de Castro, J.; Soto, C. Crossing the species barrier by prp(sc) replication in vitro generates unique infectious prions. Cell 2008, 134, 757–768. [Google Scholar] [CrossRef] [PubMed]
- Castilla, J.; Saa, P.; Hetz, C.; Soto, C. In vitro generation of infectious scrapie prions. Cell 2005, 121, 195–206. [Google Scholar] [CrossRef] [PubMed]
- Castilla, J.; Morales, R.; Saa, P.; Barria, M.; Gambetti, P.; Soto, C. Cell-free propagation of prion strains. EMBO J. 2008, 27, 2557–2566. [Google Scholar] [CrossRef] [PubMed]
- Green, K.M.; Castilla, J.; Seward, T.S.; Napier, D.L.; Jewell, J.E.; Soto, C.; Telling, G.C. Accelerated high fidelity prion amplification within and across prion species barriers. PLoS Pathog. 2008, 4, e1000139. [Google Scholar] [CrossRef] [PubMed]
- Klingeborn, M.; Race, B.; Meade-White, K.D.; Chesebro, B. Lower specific infectivity of protease-resistant prion protein generated in cell-free reactions. Proc. Natl. Acad. Sci. USA 2011, 108, E1244–E1253. [Google Scholar] [CrossRef]
- Beringue, V.; Herzog, L.; Reine, F.; le Dur, A.; Casalone, C.; Vilotte, J.L.; Laude, H. Transmission of atypical bovine prions to mice transgenic for human prion protein. Emerg. Infect. Dis. 2008, 14, 1898–1901. [Google Scholar] [CrossRef] [PubMed]
- Kong, Q.; Zheng, M.; Casalone, C.; Qing, L.; Huang, S.; Chakraborty, B.; Wang, P.; Chen, F.; Cali, I.; Corona, C.; et al. Evaluation of the human transmission risk of an atypical bovine spongiform encephalopathy prion strain. J. Virol. 2008, 82, 3697–3701. [Google Scholar] [CrossRef] [PubMed]
- Comoy, E.E.; Casalone, C.; Lescoutra-Etchegaray, N.; Zanusso, G.; Freire, S.; Marce, D.; Auvre, F.; Ruchoux, M.M.; Ferrari, S.; Monaco, S.; et al. Atypical bse (base) transmitted from asymptomatic aging cattle to a primate. PLoS One 2008, 3, e3017. [Google Scholar] [CrossRef] [PubMed]
- Mestre-Frances, N.; Nicot, S.; Rouland, S.; Biacabe, A.G.; Quadrio, I.; Perret-Liaudet, A.; Baron, T.; Verdier, J.M. Oral transmission of l-type bovine spongiform encephalopathy in primate model. Emerg. Infect. Dis. 2012, 18, 142–145. [Google Scholar] [CrossRef]
- Casalone, C.; Zanusso, G.; Acutis, P.; Ferrari, S.; Capucci, L.; Tagliavini, F.; Monaco, S.; Caramelli, M. Identification of a second bovine amyloidotic spongiform encephalopathy: Molecular similarities with sporadic creutzfeldt-jakob disease. Proc. Natl. Acad. Sci. USA 2004, 101, 3065–3070. [Google Scholar] [CrossRef] [PubMed]
- Beringue, V.; le Dur, A.; Tixador, P.; Reine, F.; Lepourry, L.; Perret-Liaudet, A.; Haik, S.; Vilotte, J.L.; Fontes, M.; Laude, H. Prominent and persistent extraneural infection in human prp transgenic mice infected with variant cjd. PLoS One 2008, 3, e1419. [Google Scholar] [CrossRef] [PubMed]
- Padilla, D.; Beringue, V.; Espinosa, J.C.; Andreoletti, O.; Jaumain, E.; Reine, F.; Herzog, L.; Gutierrez-Adan, A.; Pintado, B.; Laude, H.; et al. Sheep and goat bse propagate more efficiently than cattle bse in human prp transgenic mice. PLoS Pathog. 2011, 7, e1001319. [Google Scholar] [CrossRef] [PubMed]
- Telling, G.C.; Scott, M.; Mastrianni, J.; Gabizon, R.; Torchia, M.; Cohen, F.E.; DeArmond, S.J.; Prusiner, S.B. Prion propagation in mice expressing human and chimeric prp transgenes implicates the interaction of cellular prp with another protein. Cell 1995, 83, 79–90. [Google Scholar] [CrossRef] [PubMed]
- Torres, J.M. Unpublished.
- Scott, M.R.; Safar, J.; Telling, G.; Nguyen, O.; Groth, D.; Torchia, M.; Koehler, R.; Tremblay, P.; Walther, D.; Cohen, F.E.; et al. Identification of a prion protein epitope modulating transmission of bovine spongiform encephalopathy prions to transgenic mice. Proc. Natl. Acad. Sci. USA 1997, 94, 14279–14284. [Google Scholar] [CrossRef] [PubMed]
- Castilla, J.; Gutierrez Adan, A.; Brun, A.; Pintado, B.; Ramirez, M.A.; Parra, B.; Doyle, D.; Rogers, M.; Salguero, F.J.; Sanchez, C.; et al. Early detection of prpres in bse-infected bovine prp transgenic mice. Arch. Virol. 2003, 148, 677–691. [Google Scholar] [CrossRef] [PubMed]
- Moreno, C.R.; Lantier, F.; Lantier, I.; Sarradin, P.; Elsen, J.M. Detection of new quantitative trait loci for susceptibility to transmissible spongiform encephalopathies in mice. Genetics 2003, 165, 2085–2091. [Google Scholar] [PubMed]
- Bucalossi, C.; Cosseddu, G.; D’Agostino, C.; di Bari, M.A.; Chiappini, B.; Conte, M.; Rosone, F.; de Grossi, L.; Scavia, G.; Agrimi, U.; et al. Assessment of the genetic susceptibility of sheep to scrapie by protein misfolding cyclic amplification and comparison with experimental scrapie transmission studies. J. Virol. 2011, 85, 8386–8392. [Google Scholar] [CrossRef] [PubMed]
- Statview, version 5 for Windows; SAS Institute Inc.: Cary, NC, USA, 1998.
- XLSSTAT, version 2014.4.09; Addinsoft: Paris, France, 2014.
- Wadsworth, J.D.; Asante, E.A.; Desbruslais, M.; Linehan, J.M.; Joiner, S.; Gowland, I.; Welch, J.; Stone, L.; Lloyd, S.E.; Hill, A.F.; et al. Human prion protein with valine 129 prevents expression of variant cjd phenotype. Science 2004, 306, 1793–1796. [Google Scholar] [CrossRef] [PubMed]
- Scott, M.R.; Peretz, D.; Nguyen, H.O.; Dearmond, S.J.; Prusiner, S.B. Transmission barriers for bovine, ovine, and human prions in transgenic mice. J. Virol. 2005, 79, 5259–5271. [Google Scholar] [CrossRef] [PubMed]
- Bruce, M.E.; Will, R.G.; Ironside, J.W.; McConnell, I.; Drummond, D.; Suttie, A.; McCardle, L.; Chree, A.; Hope, J.; Birkett, C.; et al. Transmissions to mice indicate that “new variant” cjd is caused by the bse agent. Nature 1997, 389, 498–501. [Google Scholar] [CrossRef] [PubMed]
- Capobianco, R.; Casalone, C.; Suardi, S.; Mangieri, M.; Miccolo, C.; Limido, L.; Catania, M.; Rossi, G.; di Fede, G.; Giaccone, G.; et al. Conversion of the base prion strain into the bse strain: The origin of bse? PLoS Pathog. 2007, 3, e31. [Google Scholar]
- Beringue, V.; Andreoletti, O.; le Dur, A.; Essalmani, R.; Vilotte, J.L.; Lacroux, C.; Reine, F.; Herzog, L.; Biacabe, A.G.; Baron, T.; et al. A bovine prion acquires an epidemic bovine spongiform encephalopathy strain-like phenotype on interspecies transmission. J. Neurosci. 2007, 27, 6965–6971. [Google Scholar] [CrossRef] [PubMed]
- Bossers, A.; Belt, P.; Raymond, G.J.; Caughey, B.; de Vries, R.; Smits, M.A. Scrapie susceptibility-linked polymorphisms modulate the in vitro conversion of sheep prion protein to protease-resistant forms. Proc. Natl. Acad. Sci. USA 1997, 94, 4931–4936. [Google Scholar] [CrossRef] [PubMed]
- Bossers, A.; de Vries, R.; Smits, M.A. Susceptibility of sheep for scrapie as assessed by in vitro conversion of nine naturally occurring variants of prp. J. Virol. 2000, 74, 1407–1414. [Google Scholar] [CrossRef] [PubMed]
- Raymond, G.J.; Hope, J.; Kocisko, D.A.; Priola, S.A.; Raymond, L.D.; Bossers, A.; Ironside, J.; Will, R.G.; Chen, S.G.; Petersen, R.B.; et al. Molecular assessment of the potential transmissibilities of bse and scrapie to humans. Nature 1997, 388, 285–288. [Google Scholar] [CrossRef] [PubMed]
- Piening, N.; Nonno, R.; di Bari, M.; Walter, S.; Windl, O.; Agrimi, U.; Kretzschmar, H.A.; Bertsch, U. Conversion efficiency of bank vole prion protein in vitro is determined by residues 155 and 170, but does not correlate with the high susceptibility of bank voles to sheep scrapie in vivo. J. Biol. Chem. 2006, 281, 9373–9384. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Levavasseur, E.; Privat, N.; Martin, J.-C.E.; Simoneau, S.; Baron, T.; Flan, B.; Torres, J.-M.; Haïk, S. Molecular Modeling of Prion Transmission to Humans. Viruses 2014, 6, 3766-3777. https://doi.org/10.3390/v6103766
Levavasseur E, Privat N, Martin J-CE, Simoneau S, Baron T, Flan B, Torres J-M, Haïk S. Molecular Modeling of Prion Transmission to Humans. Viruses. 2014; 6(10):3766-3777. https://doi.org/10.3390/v6103766
Chicago/Turabian StyleLevavasseur, Etienne, Nicolas Privat, Juan-Carlos Espinosa Martin, Steve Simoneau, Thierry Baron, Benoit Flan, Juan-Maria Torres, and Stéphane Haïk. 2014. "Molecular Modeling of Prion Transmission to Humans" Viruses 6, no. 10: 3766-3777. https://doi.org/10.3390/v6103766
APA StyleLevavasseur, E., Privat, N., Martin, J.-C. E., Simoneau, S., Baron, T., Flan, B., Torres, J.-M., & Haïk, S. (2014). Molecular Modeling of Prion Transmission to Humans. Viruses, 6(10), 3766-3777. https://doi.org/10.3390/v6103766




