Pathogenesis of Noroviruses, Emerging RNA Viruses
Abstract
:1. Introduction
2. Clinical Disease and Epidemiology
2.1. Clinical Disease
2.2. Genetic Diversity
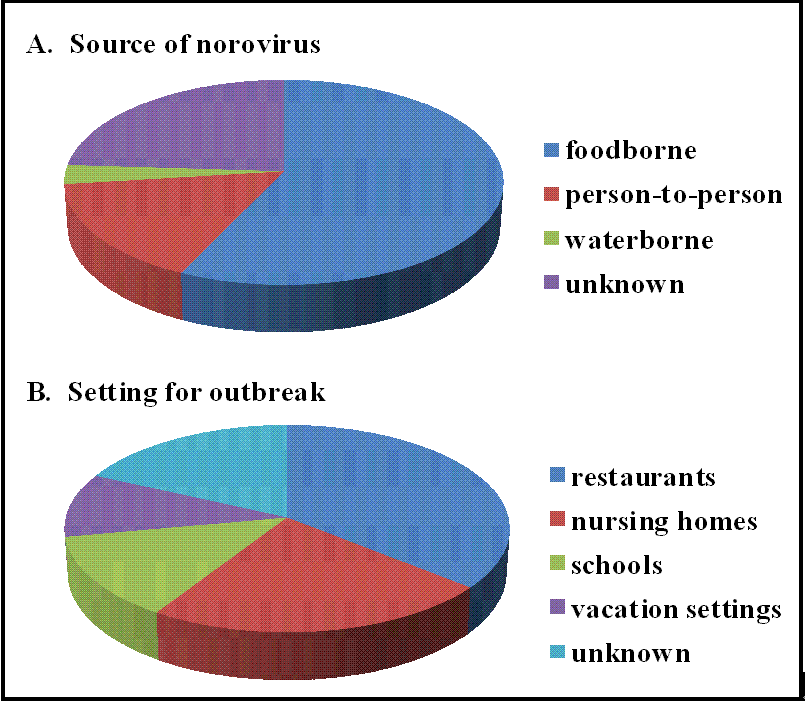
2.3. Epidemiology
3. Pathophysiology
3.1. Histological Alterations in the Intestine
3.2. Physical and Biochemical Manifestations
3.3. Systemic Infection
3.4. The Course of Norovirus Infection
4. Cellular Determinants of Norovirus Pathogenesis
4.1. Recent Advances of In Vitro Human Norovirus Infection Models
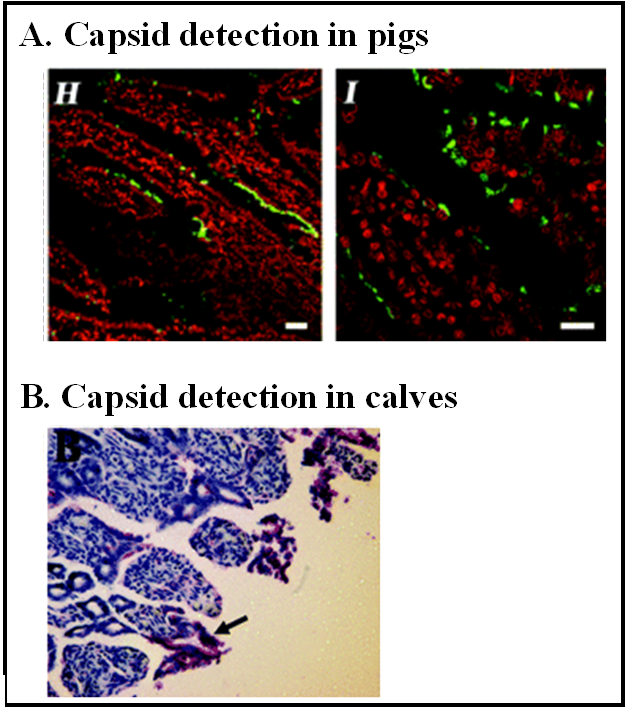
4.2. Human Norovirus Cell Tropism in Animal Models
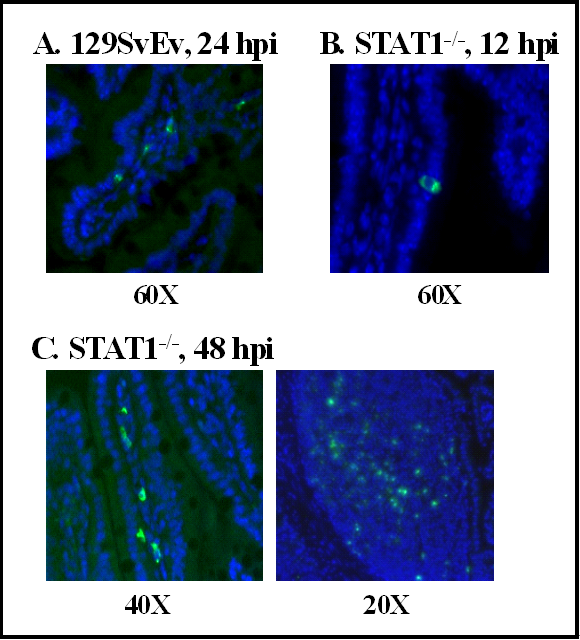
4.3. Murine Norovirus Cell Tropism
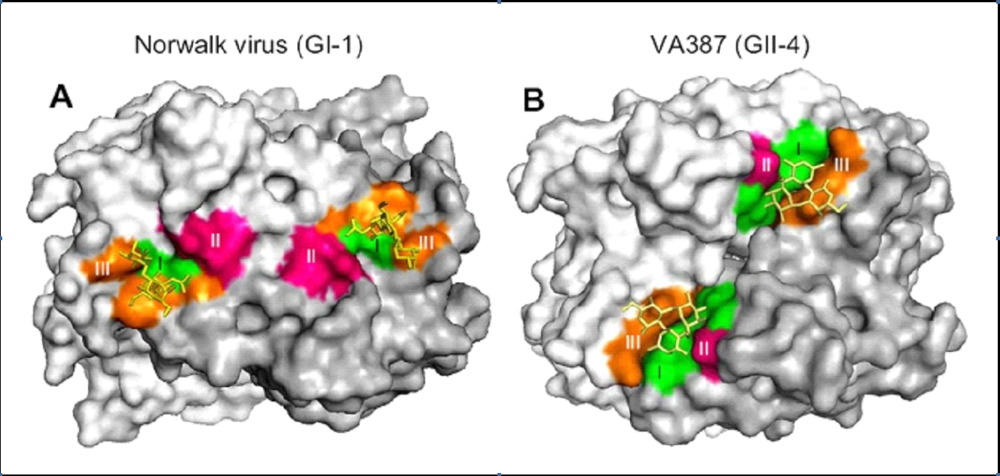
4.4. Norovirus Receptors
5. Molecular Determinants of Norovirus Pathogenesis
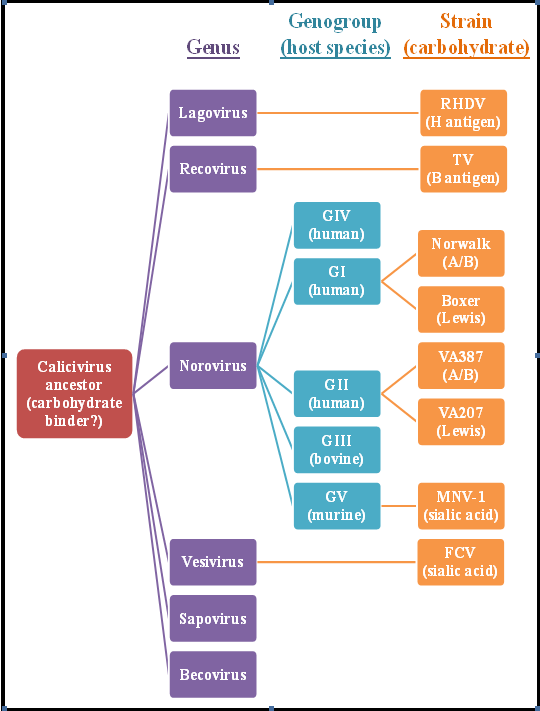
5.1. Norovirus Genomic Structures
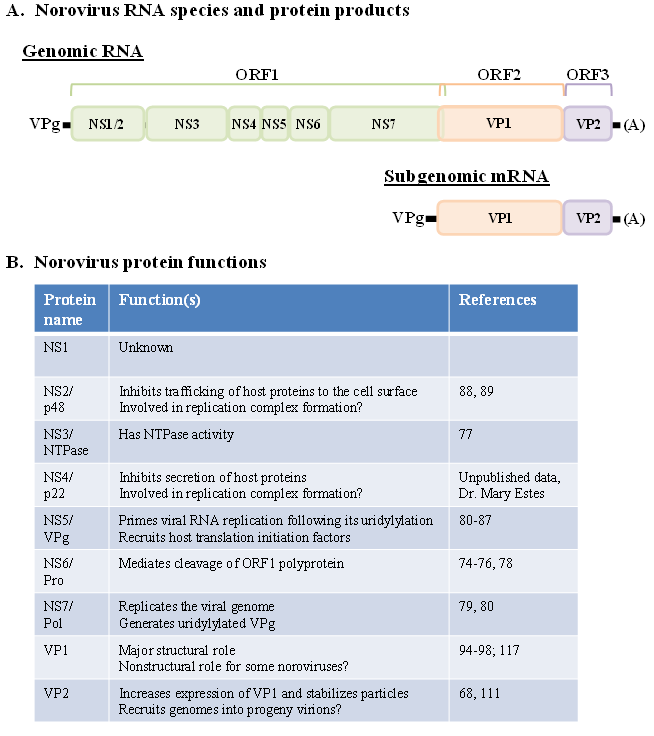
5.2. Norovirus Nonstructural Proteins
5.3. Norovirus Structural Proteins
6. Norovirus Immunity and Vaccination
6.1. Immunity

6.2. Candidate Norovirus Vaccination Strategies
7. Conclusions
Acknowledgments
Referneces
- Mead, P.S.; Slutsker, L.; Dietz, V.; McCaig, L.F.; Bresee, J.S.; Shapiro, C.; Griffin, P.M.; Tauxe, R.V. Food-related illness and death in the United States. Emerg. Infect. Dis. 1999, 5, 607–625. [Google Scholar] [CrossRef] [PubMed]
- Patel, M.M. Systematic Literature Review of Role of Noroviruses in Sporadic Gastroenteritis. Emerg. Infect. Dis. 2008, 14, 1224–1231. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention. Surveillance for foodborne disease outbreaks - United States. 2006 . MMWR 2009, 58, 609–615. [Google Scholar] [PubMed]
- Green, K.Y. Caliciviridae: The Noroviruses. In Fields Virology; 2007; Lippincott Williams and Wilkins: Philadelphia, PA, USA. [Google Scholar]
- Parrino, T.A.; Schreiber, D.S.; Trier, J.S.; Kapikian, A.Z.; Blacklow, N.R. Clinical immunity in acute gastroenteritis caused by Norwalk agent. N. Engl. J. Med. 1977, 297, 86–89. [Google Scholar] [PubMed]
- Johnson, P.C.; Mathewson, J.J.; DuPont, H.L.; Greenberg, H.B. Multiple-challenge study of host susceptibility to Norwalk gastroenteritis in US adults. J. Infect. Dis. 1990, 161, 18–21. [Google Scholar] [PubMed]
- Estes, M.K.; Prasad, B.V.; Atmar, R.L. Noroviruses everywhere: has something changed? Curr. Opin. Infect. Dis. 2006, 19, 467–474. [Google Scholar] [CrossRef] [PubMed]
- Kirkwood, C.D.and Streitberg. Calicivirus shedding in children after recovery from diarrhoeal disease . J. Clin. Virol. 2008, 43, 346–348. [Google Scholar] [CrossRef] [PubMed]
- Murata, T.; Katsushima, N.; Mizuta, K.; Muraki, Y.; Hongo, S.; Matsuzaki, Y. Prolonged Norovirus Shedding in Infants - 6 Months of Age With Gastroenteritis. Pediatr. Infect. Dis. J. 2007, 26, 46–49. [Google Scholar] [CrossRef] [PubMed]
- Sakai, Y.; Nakata, S.; Honma, S.; Tatsumi, M.; Numata-Kinoshita, K.; Chiba, S. Clinical severity of Norwalk virus and Sapporo virus gastroenteritis in children in Hokkaido, Japan. Pediatr. Infect. Dis. J. 2001, 20, 849–853. [Google Scholar] [CrossRef] [PubMed]
- Zintz, C.; Bok, K.; Parada, E.; Barnes-Eley, M.; Berke, T.; Staat, M.A.; Azimi, P.; Jiang, X.; Matson, D.O. Prevalence and genetic characterization of caliciviruses among children hospitalized for acute gastroenteritis in the United States. Infect. Genet. Evol. 2005, 5, 281–290. [Google Scholar] [CrossRef]
- Glass, R.I.; Parashar, U.D.; Estes, M.K. Norovirus Gastroenteritis. N. Engl. J. Med. 2009, 361, 1776–1785. [Google Scholar] [CrossRef] [PubMed]
- Gallimore, C.I.; Lewis, D.; Taylor, C.; Cant, A.; Gennery, A.; Gray, J.J. Chronic excretion of a norovirus in a child with cartilage hair hypoplasia (CHH). J. Clin. Virol. 2004, 30, 196–204. [Google Scholar] [CrossRef] [PubMed]
- Morotti, R.A.; Kaufman, S.S.; Fishbein, T.M.; Chatterjee, N.K.; Fuschino, M.E.; Morse, D.L.; Magid, M.S. Calicivirus infection in pediatric small intestine transplant recipients: Pathological considerations. Hum. Pathol. 2004, 35, 1236–1240. [Google Scholar] [CrossRef] [PubMed]
- Nilsson, M.; Hedlund, K.; Thorhagen, M.; Larson, G.; Johansen, K.; Ekspong, A.; Svensson, L. Evolution of Human Calicivirus RNA In vivo: Accumulation of Mutations in the Protruding P2 Domain of the Capsid Leads to Structural Changes and Possibly a New Phenotype. J. Virol. 2003, 77, 13117–13124. [Google Scholar] [CrossRef] [PubMed]
- Gallimore, C.I.; Taylor, C.; Gennery, A.R.; Cant, A.J.; Galloway, A.; Iturriza-Gomara, M.; Gray, J.J. Environmental Monitoring for Gastroenteric Viruses in a Pediatric Primary Immunodeficiency Unit. J. Clin. Microbiol. 2006, 44, 395–399. [Google Scholar] [CrossRef] [PubMed]
- Kaufman, S.S.; Chatterjee, N.K.; Fuschino, M.E.; Magid, M.S.; Gordon, R.E.; Morse, D.L.; Herold, B.C.; LeLeiko, N.S.; Tschernia, A.; Florman, S.S.; Gondolesi, G.E.; Fishbein, T.M. Calicivirus Enteritis in an Intestinal Transplant Recipient. Am. J. Transplant. 2003, 3, 764–768. [Google Scholar] [CrossRef] [PubMed]
- Kaufman, S.S.; Chatterjee, N.K.; Fuschino, M.E.; Morse, D.L.; Morotti, R.A.; Magid, M.S.; Gondolesi, G.E.; Florman, S.S.; Fishbein, T.M. Characteristics of human calicivirus enteritis in intestinal transplant recipients. J. Pediatr. Gastroenterol. Nutr. 2005, 40, 328–333. [Google Scholar] [CrossRef] [PubMed]
- Westhoff, T.H.; Vergoulidou, M.; Loddenkemper, C.; Schwartz, S.; Hofmann, J.; Schneider, T.; Zidek, W.; van der Giet, M. Chronic norovirus infection in renal transplant recipients. Nephrol. Dial. Transplant. 2009, 24, 1051–1053. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention. Norovirus activity--United States, 2006â2007 . MMWR 2007, 56, 842–846. [Google Scholar] [PubMed]
- Lopman, B.A.; Adak, G.K.; Reacher, M.H.; Brown, D.W.G. Two epidemiologic patterns of norovirus outbreaks: surveillance in England and wales, 1992–2000. Emerg. Infect. Dis. 2003, 9, 71–77. [Google Scholar] [PubMed]
- Lopman, B.A.; Reacher, M.H.; Vipond, I.B.; Sarangi, J.; Brown, D.W.G. Clinical Manifestation of Norovirus Gastroenteritis in Health Care Settings. Clin. Infect. Dis. 2004, 39, 318–324. [Google Scholar] [CrossRef] [PubMed]
- Story from REDORBIT NEWS. Norovirus outbreak kills 3 in Washington . 2006. Available online: http://www.redorbit.com/modules/news/tools.php?tool=print&id=462875 .
- Khan, R.R.; Lawson, A.D.; Minnich, L.L.; Martin, K.; Nasir, A.; Emmett, M.K.; Welch, C.A.; Udall, J.N. Gastrointestinal Norovirus Infection Associated With Exacerbation of Inflammatory Bowel Disease. J. Pediatr. Gastroenterol. Nutr. 2009, 48, 328–333. [Google Scholar] [CrossRef] [PubMed]
- Koopmans, M. Progress in understanding norovirus epidemiology. Curr. Opin. Infect. Dis. 2008, 21, 544–552. [Google Scholar] [CrossRef] [PubMed]
- Ito, S.; Takeshita, S.; Nezu, A.; Aihara, Y.; Usuku, S.; Noguchi, Y.; Yokota, S. Norovirus-associated encephalopathy. Pediatr. Infect. Dis. J. 2006, 25, 651–652. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention. Outbreak of acute gastroenteritis associated with Norwalk-like viruses among British military personnel--Afghanistan, May 2002 . MMWR 2002, 51, 477–479. [Google Scholar] [PubMed]
- Turcios-Ruiz, R.M.; Axelrod, P.; John, K. St.; Bullitt, E.; Donahue, J.; Robinson, N.; Friss, H.E. Outbreak of Necrotizing Enterocolitis Caused by Norovirus in a Neonatal Intensive Care Unit. J. Pediatr. 2008, 153, 339–344. [Google Scholar] [CrossRef] [PubMed]
- Marshall, J.K.; Thabane, M.; Borgaonkar, M.R.; James, C. Postinfectious Irritable Bowel Syndrome After a Food-Borne Outbreak of Acute Gastroenteritis Attributed to a Viral Pathogen. Clin. Gastroenterol. Hepatol. 2007, 5, 457–460. [Google Scholar] [CrossRef] [PubMed]
- Kawano, G.; Oshige, K.; Syutou, S.; Koteda, Y.; Yokoyama, T.; Kim, B.; Mizuochi, T.; Nagai, K.; Matsuda, K.; Ohbu, K.; Matsuishi, T. Benign infantile convulsions associated with mild gastroenteritis: A retrospective study of 39 cases including virological tests and efficacy of anticonvulsants. Brain Dev. 2007, 29, 617–622. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Tsai, C.; Lai, M.; Chen, C.; Lin, K.; Lin, T.; Chiu, C. Norovirus infection as a cause of diarrhea-associated benign infantile seizures. Clin. Infect. Dis. 2009, 48, 849–855. [Google Scholar] [CrossRef] [PubMed]
- Zheng, D.; Ando, T.; Fankhauser, R.L.; Beard, R.S.; Glass, R.I.; Monroe, S.S. Norovirus classification and proposed strain nomenclature. Virology 2006, 346, 312–323. [Google Scholar] [CrossRef] [PubMed]
- Fankhauser, R.L.; Noel, J.S.; Monroe, S.S.; Ando, T.; Glass, R.I. Molecular Epidemiology of “Norwalk‐like Viruses” in Outbreaks of Gastroenteritis in the United States. J. Infect. Dis. 1998, 178, 1571–1578. [Google Scholar] [CrossRef] [PubMed]
- Sugieda, M.; Nagaoka, H.; Kakishima, Y.; Ohshita, T.; Nakamura, S.; Nakajima, S. Detection of Norwalk-like virus genes in the caecum contents of pigs. Arch. Virol. 1998, 143, 1215–1221. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Han, M.G.; Cheetham, S.; Souza, M.; Funk, J.A.; Saif, L.J. Porcine noroviruses related to human noroviruses. Emerg. Infect. Dis. 2005, 11, 1874–1881. [Google Scholar] [PubMed]
- Liu, B.L.; Lambden, P.R.; Gunther, H.; Otto, P.; Elschner, M.; Clarke, I.N. Molecular Characterization of a Bovine Enteric Calicivirus: Relationship to the Norwalk-Like Viruses. J. Virol. 1999, 73, 819–825. [Google Scholar] [PubMed]
- Oliver, S.L.; Dastjerdi, A.M.; Wong, S.; El-Attar, L.; Gallimore, C.; Brown, D.W.G.; Green, J.; Bridger, J.C. Molecular Characterization of Bovine Enteric Caliciviruses: a Distinct Third Genogroup of Noroviruses (Norwalk-Like Viruses) Unlikely To Be of Risk to Humans. J. Virol. 2003, 77, 2789–2798. [Google Scholar] [CrossRef] [PubMed]
- Hsu, C.; Riley, L.; Livingston, R. Molecular characterization of three novel murine noroviruses. Virus Genes. 2007, 34, 147–155. [Google Scholar] [CrossRef] [PubMed]
- Karst, S.M.; Wobus, C.E.; Lay, M.; Davidson, J.; Virgin, H.W. STAT1-Dependent Innate Immunity to a Norwalk-Like Virus. Science 2003, 299, 1575–1578. [Google Scholar] [CrossRef] [PubMed]
- Müller, B.; Klemm, U.; Mas Marques, A.; Schreier, E. Genetic diversity and recombination of murine noroviruses in immunocompromised mice. Arch. Virol. 2007, 152, 1709–1719. [Google Scholar] [CrossRef] [PubMed]
- Thackray, L.B.; Wobus, C.E.; Chachu, K.A.; Liu, B.; Alegre, E.R.; Henderson, K.S.; Kelley, S.T.; Virgin, H.W. Murine Noroviruses Comprising a Single Genogroup Exhibit Biological Diversity despite Limited Sequence Divergence. J. Virol. 2007, 81, 10460–10473. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.; Lee, H.; Chang, K.; Ko, G. Molecular Characterization of Murine Norovirus Isolates from South Korea. Virus Res. 2010, 147, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Widdowson, M.; Cramer, E.H.; Hadley, L.; Bresee, J.S.; Beard, R.S.; Bulens, S.N.; Charles, M.; Chege, W.; Isakbaeva, E.; Wright, J.G.; Mintz, E.; Forney, D.; Massey, J.; Glass, R.I.; Monroe, S.S. Outbreaks of Acute Gastroenteritis on Cruise Ships and on Land: Identification of a Predominant Circulating Strain of Norovirus—United States, 2002. J. Infect. Dis. 2004, 190, 27–36. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention. Infectious disease and dermatologic conditions in evacuees and rescue workers after Hurricane Katrina--multiple states, August-September, 2005 . MMWR 2005, 54, 961–964. [Google Scholar] [PubMed]
- Centers for Disease Control and Prevention. Norovirus outbreak among evacuees from hurricane Katrina-Houston, Texas, September 2005 . MMWR 2005, 54, 1016–1018. [Google Scholar] [PubMed]
- McCarthy, M.; Estes, M.K.; Hyams, K.C. Norwalkâlike Virus Infection in Military Forces: Epidemic Potential, Sporadic Disease, and the Future Direction of Prevention and Control Efforts . J. Infect. Dis. 2000, 181, S387–S391. [Google Scholar] [CrossRef] [PubMed]
- Thornton, S.A.; Sherman, S.S.; Farkas, T.; Zhong, W.; Torres, P.; Jiang, X. Gastroenteritis in US Marines during Operation Iraqi Freedom. Clin. Infect. Dis. 2005, 40, 519–525. [Google Scholar] [CrossRef] [PubMed]
- Hansen, S.; Stamm-Balderjahn, S.; Zuschneid, I.; Behnke, M.; Rüden, H.; Vonberg, R.; Gastmeier, P. Closure of medical departments during nosocomial outbreaks: data from a systematic analysis of the literature. J. Hosp. Infect. 2007, 65, 348–353. [Google Scholar] [CrossRef] [PubMed]
- Rohayem, J. Norovirus seasonality and the potential impact of climate change. Clin. Microbiol. Infect. 2009, 15, 524–527. [Google Scholar] [CrossRef] [PubMed]
- Teunis, P.F.; Moe, C.L.; Liu, P.; Miller, S.E.; Lindesmith, L.; Baric, R.S.; Pendu, J.L.; Calderon, R.L. Norwalk virus: How infectious is it? J. Med. Virol. 2008, 80, 1468–1476. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention. Outbreaks of gastroenteritis associated with noroviruses on cruise ships--United States, 2002 . MMWR 2002, 51, 1112–1115. [Google Scholar] [PubMed]
- Centers for Disease Control and Prevention. Norovirus outbreak associated with ill food-service workers--Michigan, January-February 2006 . MMWR 2006, 56, 1212–1216. [Google Scholar]
- Centers for Disease Control and Prevention. Norovirus outbreak in an elementary school--District of Columbia, February 2007 . MMWR 2008, 56, 1340–1343. [Google Scholar] [PubMed]
- Atmar, R.L.; Opekun, A.R.; Gilger, M.A.; Estes, M.K.; Crawford, S.E.; Neill, F.H.; Graham, D.Y. Norwalk virus shedding after experimental human infection. Emerg. Infect. Dis. 2008, 14, 1553–1557. [Google Scholar] [CrossRef] [PubMed]
- Patterson, T.; Hutchings, P.; Palmer, S. Outbreak of SRSV gastroenteritis at an international conference traced to food handled by a post-symptomatic caterer. Epidemiol. Infect. 1993, 111, 157–162. [Google Scholar] [CrossRef] [PubMed]
- Rockx, B.; de Wit, M.; Vennema, H.; Vinjé, J.; de Bruin, E.; van Duynhoven, Y.; Koopmans, M. Natural History of Human Calicivirus Infection: A Prospective Cohort Study. Clin. Infect. Dis. 2002, 35, 246–253. [Google Scholar] [CrossRef] [PubMed]
- Lopman, B.; Vennema, H.; Kohli, E.; Pothier, P.; Sanchez, A.; Negredo, A.; Buesa, J.; Schreier, E.; Reacher, M.; Brown, D.; Gray, J.; Iturriza, M.; Gallimore, C.; Bottiger, B.; Hedlund, K.; Torvén, M.; von Bonsdorff, C.; Maunula, L.; Poljsak-Prijatelj, M.; Zimsek, J.; Reuter, G.; Szücs, G.; Melegh, B.; Svennson, L.; van Duijnhoven, Y.; Koopmans, M. Increase in viral gastroenteritis outbreaks in Europe and epidemic spread of new norovirus variant. Lancet 2004, 363, 682–688. [Google Scholar] [CrossRef] [PubMed]
- Noel, J.S.; Fankhauser, R.L.; Ando, T.; Monroe, S.S.; Glass, R.I. Identification of a Distinct Common Strain of "Norwalk‐like Viruses" Having a Global Distribution. J. Infect. Dis. 1999, 179, 1334–1344. [Google Scholar] [CrossRef] [PubMed]
- White, P.A.; Hansman, G.S.; Li, A.; Dable, J.; Isaacs, M.; Ferson, M.; McIver, C.J.; Rawlinson, W.D. Norwalk-like virus 95/96-US strain is a major cause of gastroenteritis outbreaks in Australia. J. Med. Virol. 2002, 68, 113–118. [Google Scholar] [CrossRef] [PubMed]
- Bull, R.A.; Tu, E.T.V.; McIver, C.J.; Rawlinson, W.D.; White, P.A. Emergence of a New Norovirus Genotype II.4 Variant Associated with Global Outbreaks of Gastroenteritis . J. Clin. Microbiol. 2006, 44, 327–333. [Google Scholar] [CrossRef] [PubMed]
- Tu, E.T.; Bull, R.A.; Greening, G.E.; Hewitt, J.; Lyon, M.J.; Marshall, J.A.; McIver, C.J.; Rawlinson, W.D.; White, P.A. Epidemics of Gastroenteritis during 2006 Were Associated with the Spread of Norovirus GII.4 Variants 2006a and 2006b . Clin. Infect. Dis. 2008, 46, 413–420. [Google Scholar] [CrossRef] [PubMed]
- Siebenga, J.J.; Vennema, H.; Zheng, D.; Vinjé, J.; Lee, B.E.; Pang, X.; Ho, E.C.M.; Lim, W.; Choudekar, A.; Broor, S.; Halperin, T.; Rasool, N.B.G.; Hewitt, J.; Greening, G.E.; Jin, M.; Duan, Z.; Lucero, Y.; O’Ryan, M.; Hoehne, M.; Schreier, E.; Ratcliff, R.M.; White, P.A.; Iritani, N.; Reuter, G.; Koopmans, G. Norovirus Illness Is a Global Problem: Emergence and Spread of Norovirus GII.4 Variants, 2001â2007 . J. Infect. Dis. 2009, 200, 802–812. [Google Scholar] [CrossRef] [PubMed]
- Bok, K.; Abente, E.J.; Realpe-Quintero, M.; Mitra, T.; Sosnovtsev, S.V.; Kapikian, A.Z.; Green, K.Y. Evolutionary Dynamics of GII.4 Noroviruses over a Thirty-four Year Period . J. Virol. 2009, 83, 11890–11901. [Google Scholar] [PubMed]
- Friesema, I.; Vennema, H.; Heijne, J.; de Jager, C.; Teunis, P.; van der Linde, R.; Duizer, E.; van Duynhoven, Y. Differences in clinical presentation between norovirus genotypes in nursing homes. J. Clin. Virol. 2009, 46, 341–344. [Google Scholar] [CrossRef] [PubMed]
- Patel, M.M.; Hall, A.J.; Vinjé, J.; Parashar, U.D. Noroviruses: A comprehensive review. J. Clin. Virol. 2009, 44, 1–8. [Google Scholar] [CrossRef]
- Ko, G.; Garcia, C.; Jiang, Z.; Okhuysen, P.C.; Belkind-Gerson, J.; Glass, R.I.; DuPont, H.L. Noroviruses as a Cause of Traveler's Diarrhea among Students from the United States Visiting Mexico. J. Clin. Microbiol. 2005, 43, 6126–6129. [Google Scholar] [CrossRef] [PubMed]
- Chapin, A.R.; Carpenter, C.M.; Dudley, W.C.; Gibson, L.C.; Pratdesaba, R.; Torres, O.; Sanchez, D.; Belkind-Gerson, J.; Nyquist, I.; Karnell, A.; Gustafsson, B.; Halpern, J.L.; Bourgeois, A.L.; Schwab, K.J. Prevalence of Norovirus among Visitors from the United States to Mexico and Guatemala Who Experience Traveler's Diarrhea. J. Clin. Microbiol. 2005, 43, 1112–1117. [Google Scholar] [CrossRef] [PubMed]
- Ko, G.; Jiang, Z.; Okhuysen, P.C.; DuPont, H.L. Fecal cytokines and markers of intestinal inflammation in international travelers with diarrhea due to Noroviruses. J. Med. Virol. 2006, 78, 825–828. [Google Scholar] [CrossRef] [PubMed]
- Acute infectious nonbacterial gastroenteritis: etiology and pathogenesis. Ann. Intern. Med. 1972, 76, 993–1008. [PubMed]
- Dolin, R.; Levy, A.G.; Wyatt, R.G.; Thornhill, T.S.; Gardner, J.D. Viral gastroenteritis induced by the Hawaii agent. Jejunal histopathology and serologic response. Am. J. Med. 1975, 59, 761–768. [Google Scholar] [CrossRef] [PubMed]
- Schreiber, D.S.; Blacklow, N.R.; Trier, J.S. The mucosal lesion of the proximal small intestine in acute infectious nonbacterial gastroenteritis. N. Engl. J. Med. 1973, 288, 1318–1323. [Google Scholar] [PubMed]
- Schreiber, D.S.; Blacklow, N.R.; Trier, J.S. The small intestinal lesion induced by Hawaii agent acute infectious nonbacterial gastroenteritis. J. Infect. Dis. 1974, 129, 705–708. [Google Scholar] [PubMed]
- Troeger, H.; Loddenkemper, C.; Schneider, T.; Schreier, E.; Epple, H.; Zeitz, M.; Fromm, M.; Schulzke, J. Structural and functional changes of the duodenum in human norovirus infection. Gut 2009, 58, 1070–1077. [Google Scholar] [CrossRef] [PubMed]
- Souza, M.; Azevedo, M.S.P.; Jung, K.; Cheetham, S.; Saif, L.J. Pathogenesis and Immune Responses in Gnotobiotic Calves after Infection with the Genogroup II.4-HS66 Strain of Human Norovirus . J. Virol. 2008, 82, 1777–1786. [Google Scholar] [CrossRef] [PubMed]
- Mumphrey, S.M.; Changotra, H.; Moore, T.N.; Heimann-Nichols, E.R.; Wobus, C.E.; Reilly, M.J.; Moghadamfalahi, M.; Shukla, D.; Karst, S.M. Murine Norovirus 1 Infection Is Associated with Histopathological Changes in Immunocompetent Hosts, but Clinical Disease Is Prevented by STAT1-Dependent Interferon Responses. J. Virol. 2007, 81, 3251–3263. [Google Scholar] [CrossRef] [PubMed]
- Cheetham, S.; Souza, M.; Meulia, T.; Grimes, S.; Han, M.G.; Saif, L.J. Pathogenesis of a Genogroup II Human Norovirus in Gnotobiotic Pigs. J. Virol. 2006, 80, 10372–10381. [Google Scholar] [CrossRef] [PubMed]
- Natoni, A.; Kass, G.E.N.; Carter, M.J.; Roberts, L.O. The mitochondrial pathway of apoptosis is triggered during feline calicivirus infection. J. Gen. Virol. 2006, 87, 357–361. [Google Scholar] [CrossRef] [PubMed]
- Bok, K.; Prikhodko, V.G.; Green, K.Y.; Sosnovtsev, S.V. Apoptosis in Murine Norovirus-Infected RAW264.7 Cells Is Associated with Downregulation of Survivin . J. Virol. 2009, 83, 3647–3656. [Google Scholar] [PubMed]
- Roberts, L.O.; Al-Molawi, N.; Carter, M.J.; Kass, G.E.N. Apoptosis in Cultured Cells Infected with Feline Calicivirus. Ann. N. Y. Acad. Sci. 2003, 1010, 587–590. [Google Scholar] [CrossRef] [PubMed]
- Sosnovtsev, S.V.; Prikhod'ko, E.A.; Belliot, G.; Cohen, J.I.; Green, K.Y. Feline calicivirus replication induces apoptosis in cultured cells. Virus Res. 2003, 94, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Furman, L.; Maaty, W.; Petersen, L.; Ettayebi, K.; Hardy, M.; Bothner, B. Cysteine protease activation and apoptosis in Murine norovirus infection. Virol. J. 2009, 6, 139. [Google Scholar] [CrossRef]
- Meeroff, J.C.; Schreiber, D.S.; Trier, J.S.; Blacklow, N.R. Abnormal gastric motor function in viral gastroenteritis. Ann. Intern. Med. 1980, 92, 370–373. [Google Scholar] [PubMed]
- Agus, S.G.; Dolin, R.; Wyatt, R.G.; Tousimis, A.J.; Northrup, R.S. Acute infectious nonbacterial gastroenteritis: intestinal histopathology. Histologic and enzymatic alterations during illness produced by the Norwalk agent in man. Ann. Intern. Medicine 1973, 79, 18–25. [Google Scholar]
- Boshuizen, J.A.; Reimerink, J.H.J.; Korteland-van Male, A.M.; van Ham, V.J.J.; Koopmans, M.P.G.; Buller, H.A.; Dekker, J.; Einerhand, A.W.C. Changes in Small Intestinal Homeostasis, Morphology, and Gene Expression during Rotavirus Infection of Infant Mice. J. Virol. 2003, 77, 13005–13016. [Google Scholar] [CrossRef] [PubMed]
- Takanashi, S.; Hashira, S.; Matsunaga, T.; Yoshida, A.; Shiota, T.; Tung, P.G.; Khamrin, P.; Okitsu, S.; Mizuguchi, M.; Igarashi, T.; Ushijima, H. Detection, genetic characterization, and quantification of norovirus RNA from sera of children with gastroenteritis. J. Clin. Virol. 2009, 44, 161–163. [Google Scholar] [CrossRef] [PubMed]
- Hsu, C.C.; Riley, L.K.; Wills, H.M.; Livingston, R.S. Persistent Infection with and Serologic Crossreactivity of Three Novel Murine Noroviruses. Comp. Med. 2006, 56, 247–251. [Google Scholar] [PubMed]
- Hsu, C.C.; Wobus, C.E.; Steffen, C.E.; Riley, L.K.; Livingston, R.S. “Development of a Microsphere-Based Serologic Multiplexed Fluorescent Immunoassay and a Reverse Transcriptase PCR Assay To Detect Murine Norovirus 1 Infection in Mice. Clin. Diagn. Lab. Immunol. 2005, 12, 1145–1151. [Google Scholar] [PubMed]
- Liu, G.; Kahan, S.M.; Jia, Y.; Karst, S.M. Primary High-Dose Murine Norovirus 1 Infection Fails To Protect from Secondary Challenge with Homologous Virus. J. Virol. 2009, 83, 6963–6968. [Google Scholar] [CrossRef] [PubMed]
- Ward, J.M.; Wobus, C.E.; Thackray, L.B.; Erexson, C.R.; Faucette, L.G.; Belliot, G.; Barron, E.L.; Sosnovtsev, S.V.; Green, K.Y. Pathology of Immunodeficient Mice With Naturally Occurring Murine Norovirus Infection. Toxicol. Pathol. 2006, 34, 708–715. [Google Scholar] [CrossRef] [PubMed]
- Blutt, S.E.; Fenaux, M.; Warfield, K.L.; Greenberg, H.B.; Conner, M.E. Active Viremia in Rotavirus-Infected Mice. J. Virol. 2006, 80, 6702–6705. [Google Scholar] [CrossRef] [PubMed]
- Crawford, S.E.; Patel, D.G.; Cheng, E.; Berkova, Z.; Hyser, J.M.; Ciarlet, M.; Finegold, M.J.; Conner, M.E.; Estes, M.K. Rotavirus Viremia and Extraintestinal Viral Infection in the Neonatal Rat Model. J. Virol. 2006, 80, 4820–4832. [Google Scholar] [CrossRef] [PubMed]
- Fenaux, M.; Cuadras, M.A.; Feng, N.; Jaimes, M.; Greenberg, H.B. Extraintestinal Spread and Replication of a Homologous EC Rotavirus Strain and a Heterologous Rhesus Rotavirus in BALB/c Mice. J. Virol. 2006, 80, 5219–5232. [Google Scholar] [CrossRef] [PubMed]
- Graham, D.Y.; Jiang, X.; Tanaka, T.; Opekun, A.R.; Madore, H.P.; Estes, M.K. Norwalk virus infection of volunteers: new insights based on improved assays. J. Infect. Dis. 1994, 170, 34–43. [Google Scholar] [PubMed]
- Subekti, D.; Tjaniadi, P.; Lesmana, M.; McArdle, J.; Iskandriati, D.; Budiarsa, I.; Walujo, P.; Suparto, I.; Winoto, I.; Campbell, J.; Porter, K.; Sajuthi, D.; Ansari, A.; Oyofo, B. Experimental infection of Macaca nemestrina with a Toronto Norwalk-like virus of epidemic viral gastroenteritis. J. Med. Virol. 2002, 66, 400–406. [Google Scholar] [CrossRef] [PubMed]
- Radford, A.D.; Dawson, S.; Coyne, K.P.; Porter, C.J.; Gaskell, R.M. The challenge for the next generation of feline calicivirus vaccines. Vet. Microbiol. 2006, 117, 14–18. [Google Scholar] [CrossRef] [PubMed]
- Povey, R.C.; Wardley, R.C.; Jessen, H. Feline picornavirus infection: the in vivo carrier state. Vet. Rec. 1973, 92, 224–229. [Google Scholar] [PubMed]
- Johnson, R.P.; Povey, R.C. Feline calicivirus infection in kittens borne by cats persistently infected with the virus. Res. Vet. Sci. 1984, 37, 114–119. [Google Scholar] [PubMed]
- Oldstone, M.B. Viral persistence: Parameters, mechanisms and future predictions. Virology 2006, 344, 111–118. [Google Scholar] [CrossRef] [PubMed]
- Chachu, K.A.; LoBue, A.D.; Strong, D.W.; Baric, R.S.; Virgin, H.W. Immune Mechanisms Responsible for Vaccination against and Clearance of Mucosal and Lymphatic Norovirus Infection . PLoS Pathog. 2008, 4, e1000236. [Google Scholar] [CrossRef] [PubMed]
- Johnson, R.P. Antigenic change in feline calicivirus during persistent infection. Can. J. Vet. Res. 1992, 56, 326–330. [Google Scholar] [PubMed]
- Radford, A.D.; Turner, P.C.; Bennett, M.; McArdle, F.; Dawson, S.; Glenn, M.A.; Williams, R.A.; Gaskell, R.M. Quasispecies evolution of a hypervariable region of the feline calicivirus capsid gene in cell culture and in persistently infected cats. J. Gen. Virol. 1998, 79, 1–10. [Google Scholar] [PubMed]
- Coyne, K.P.; Gaskell, R.M.; Dawson, S.; Porter, C.J.; Radford, A.D. Evolutionary Mechanisms of Persistence and Diversification of a Calicivirus within Endemically Infected Natural Host Populations. J. Virol. 2007, 81, 1961–1971. [Google Scholar] [CrossRef] [PubMed]
- Wobus, C.E.; Karst, S.M.; Thackray, L.B.; Chang, K.; Sosnovtsev, S.V.; Belliot, G.; Krug, A.; Mackenzie, J.M.; Green, K.Y.; Virgin, H.W. Replication of Norovirus in Cell Culture Reveals a Tropism for Dendritic Cells and Macrophages . PLoS Biol. 2004, 2, e432. [Google Scholar] [CrossRef] [PubMed]
- Duizer, E.; Schwab, K.J.; Neill, F.H.; Atmar, R.L.; Koopmans, M.P.G.; Estes, M.K. Laboratory efforts to cultivate noroviruses. J. Gen. Virol. 2004, 85, 79–87. [Google Scholar] [CrossRef] [PubMed]
- Straub, T.; Honer zu, B.; Orosz-Coghlan, P.; Dohnalkova, A.; Mayer, B.; Bartholomew, R.; Valdez, C.; Bruckner-Lea, C.; Gerba, C.; Abbaszadegan, M.; Nickerson, C. In vitro cell culture infectivity assay for human noroviruses. Emerg. Infect. Dis. 2007, 13, 396–403. [Google Scholar] [CrossRef] [PubMed]
- Papafragkou, E.; Hewitt, J.; Park, G.; Straub, T.; Greening, G.; Vinje, J. Challenges of Culturing Human Norovirus in a 3-D Organoid Cell Culture Model . In Proceedings of the ASM. 109th General Meeting; Philadephia, PA, USA, 2009; May. [Google Scholar]
- Hao, X.; Kim, T.S.; Braciale, T.J. Differential Response of Respiratory Dendritic Cell Subsets to Influenza Virus Infection. J. Virol. 2008, 82, 4908–4919. [Google Scholar] [CrossRef] [PubMed]
- Henri, S.; Curtis, J.; Hochrein, H.; Vremec, D.; Shortman, K.; Handman, E. Hierarchy of Susceptibility of Dendritic Cell Subsets to Infection by Leishmania major: Inverse Relationship to Interleukin-12 Production. Infect. Immun. 2002, 70, 3874–3880. [Google Scholar] [CrossRef] [PubMed]
- Latham, P.S.; Sepelak, S.B. Effect of macrophage source and activation on susceptibility in an age-dependent model of murine hepatitis caused by a phlebovirus, Punta Toro. Arch. Virol. 1992, 122, 175–185. [Google Scholar] [CrossRef] [PubMed]
- Guix, S.; Asanaka, M.; Katayama, K.; Crawford, S.E.; Neill, F.H.; Atmar, R.L.; Estes, M.K. Norwalk Virus RNA Is Infectious in Mammalian Cells. J. Virol. 2007, 81, 12238–12248. [Google Scholar] [CrossRef] [PubMed]
- Asanaka, M.; Atmar, R.L.; Ruvolo, V.; Crawford, S.E.; Neill, F.H.; Estes, M.K. Replication and packaging of Norwalk virus RNA in cultured mammalian cells. Proc. Natl. Acad. Sci. USA 2005, 102, 10327–10332. [Google Scholar] [CrossRef]
- Chaudhry, Y.; Skinner, M.A.; Goodfellow, I.G. Recovery of genetically defined murine norovirus in tissue culture by using a fowlpox virus expressing T7 RNA polymerase. J. Gen. Virol. 2007, 88, 2091–2100. [Google Scholar] [CrossRef] [PubMed]
- Katayama, K.; Hansman, G.S.; Oka, T.; Ogawa, S.; Takeda, N. Investigation of norovirus replication in a human cell line. Arch.Virol. 2006, 151, 1291–1308. [Google Scholar] [CrossRef] [PubMed]
- Ward, V.K.; McCormick, C.J.; Clarke, I.N.; Salim, O.; Wobus, C.E.; Thackray, L.B.; Virgin, H.W.; Lambden, P.R. Recovery of infectious murine norovirus using pol II-driven expression of full-length cDNA. Proc. Natl. Acad. Sci. USA 2007, 104, 11050–11055. [Google Scholar] [CrossRef]
- Chang, K.; Sosnovtsev, S.V.; Belliot, G.; King, A.D.; Green, K.Y. Stable expression of a Norwalk virus RNA replicon in a human hepatoma cell line. Virology 2006, 353, 463–473. [Google Scholar] [CrossRef] [PubMed]
- Wobus, C.E.; Thackray, L.B.; Virgin, H.W. Murine Norovirus: a Model System To Study Norovirus Biology and Pathogenesis. J. Virol. 2006, 80, 5104–5112. [Google Scholar] [CrossRef] [PubMed]
- Perry, J.W.; Taube, S.; Wobus, C.E. Murine norovirus-1 entry into permissive macrophages and dendritic cells is pH-independent. Virus Res. 2009, 143, 125–129. [Google Scholar] [CrossRef] [PubMed]
- Perdue, K.A.; Green, K.Y.; Copeland, M.; Barron, E.; Mandel, M.; Faucette, L.J.; Williams, E.M.; Sosnovtsev, S.V.; Elkins, W.R.; Ward, J.M. Naturally Occurring Murine Norovirus Infection in a Large Research Institution. J. Am. Assoc. Lab. Anim. Sci. 2007, 46, 39–45. [Google Scholar] [PubMed]
- Ryman, K.D.; Klimstra, W.B.; Nguyen, K.B.; Biron, C.A.; Johnston, R.E. Alpha/Beta Interferon Protects Adult Mice from Fatal Sindbis Virus Infection and Is an Important Determinant of Cell and Tissue Tropism. J. Virol. 2000, 74, 3366–3378. [Google Scholar] [CrossRef] [PubMed]
- Samuel, M.A.; Diamond, M.S. Alpha/Beta Interferon Protects against Lethal West Nile Virus Infection by Restricting Cellular Tropism and Enhancing Neuronal Survival. J. Virol. 2005, 79, 13350–13361. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Ma, Y.; Barrett, J.W.; Gao, X.; Loh, J.; Barton, E.; Virgin, H.W.; McFadden, G. Disruption of Erk-dependent type I interferon induction breaks the myxoma virus species barrier. Nat. Immunol. 2004, 5, 1266–1274. [Google Scholar] [CrossRef] [PubMed]
- Tan, M.; Jiang, X. Norovirus and its histo-blood group antigen receptors: an answer to a historical puzzle. Trends Microbiol. 2005, 13, 285–293. [Google Scholar] [CrossRef] [PubMed]
- Lindesmith, L.; Moe, C.; Marionneau, S.; Ruvoen, N.; Jiang, X.; Lindblad, L.; Stewart, P.; LePendu, J.; Baric, R. Human susceptibility and resistance to Norwalk virus infection. Nat. Med. 2003, 9, 548–553. [Google Scholar] [CrossRef] [PubMed]
- Huang, P.; Farkas, T.; Marionneau, S.; Zhong, W.; Ruvoën‐Clouet, N.; Morrow, A.L.; Altaye, M.; Pickering, L.K.; Newburg, D.S.; LePendu, J.; Jiang, X. Noroviruses Bind to Human ABO, Lewis, and Secretor Histo–Blood Group Antigens: Identification of 4 Distinct Strain‐Specific Patterns. J. Infect. Dis. 2003, 188, 19–31. [Google Scholar] [CrossRef] [PubMed]
- Huang, P.; Farkas, T.; Zhong, W.; Tan, M.; Thornton, S.; Morrow, A.L.; Jiang, X. Norovirus and Histo-Blood Group Antigens: Demonstration of a Wide Spectrum of Strain Specificities and Classification of Two Major Binding Groups among Multiple Binding Patterns. J. Virol. 2005, 79, 6714–6722. [Google Scholar] [CrossRef] [PubMed]
- Shirato, H.; Ogawa, S.; Ito, H.; Sato, T.; Kameyama, A.; Narimatsu, H.; Xiaofan, Z.; Miyamura, T.; Wakita, T.; Ishii, K.; Takeda, N. Noroviruses Distinguish between Type 1 and Type 2 Histo-Blood Group Antigens for Binding. J. Virol. 2008, 82, 10756–10767. [Google Scholar] [CrossRef] [PubMed]
- Tan, M.; Xia, M.; Chen, Y.; Bu, W.; Hegde, R.S.; Meller, J.; Li, X.; Jiang, X. Conservation of Carbohydrate Binding Interfaces â Evidence of Human HBGA Selection in Norovirus Evolution . PLoS ONE 2009, 4, e5058. [Google Scholar] [CrossRef] [PubMed]
- Lindesmith, L.C.; Donaldson, E.F.; LoBue, A.D.; Cannon, J.L.; Zheng, D.; Vinje, J.; Baric, R.S. Mechanisms of GII.4 Norovirus Persistence in Human Populations . PLoS Med. 2008, 5, e31. [Google Scholar] [CrossRef] [PubMed]
- Cannon, J.L.; Lindesmith, L.C.; Donaldson, E.F.; Saxe, L.; Baric, R.S.; Vinje, J. Herd Immunity to GII.4 Noroviruses Is Supported by Outbreak Patient Sera . J. Virol. 2009, 83, 5363–5374. [Google Scholar] [CrossRef] [PubMed]
- Le Pendu, J.; Ruvoën-Clouet, N.; Kindberg, E.; Svensson, L. Mendelian resistance to human norovirus infections. Sem. Immunol. 2006, 18, 375–386. [Google Scholar] [CrossRef]
- Ruvoen-Clouet, N.; Ganiere, J.P.; Andre-Fontaine, G.; Blanchard, D.; Le Pendu, J. Binding of Rabbit Hemorrhagic Disease Virus to Antigens of the ABH Histo-Blood Group Family. J. Virol. 2000, 74, 11950–11954. [Google Scholar] [CrossRef] [PubMed]
- Farkas, T.; Sestak, K.; Wei, C.; Jiang, X. Characterization of a Rhesus Monkey Calicivirus Representing a New Genus of Caliciviridae. J. Virol. 2008, 82, 5408–5416. [Google Scholar] [CrossRef] [PubMed]
- Stuart, A.D.; Brown, T.D.K. {alpha}2,6-Linked sialic acid acts as a receptor for Feline calicivirus. J. Gen. Virol. 2007, 88, 177–186. [Google Scholar] [CrossRef] [PubMed]
- Taube, S.; Perry, J.W.; Yetming, K.; Patel, S.P.; Auble, H.; Shu, L.; Nawar, H.F.; Lee, C.H.; Connell, T.D.; Shayman, J.A.; Wobus, C.E. Ganglioside-Linked Terminal Sialic Acid Moieties on Murine Macrophages Function as Attachment Receptors for Murine Noroviruses. J. Virol. 2009, 83, 4092–4101. [Google Scholar] [CrossRef] [PubMed]
- Tamura, M.; Natori, K.; Kobayashi, M.; Miyamura, T.; Takeda, N. Genogroup II Noroviruses Efficiently Bind to Heparan Sulfate Proteoglycan Associated with the Cellular Membrane. J. Virol. 2004, 78, 3817–3826. [Google Scholar] [CrossRef] [PubMed]
- Rydell, G.E.; Nilsson, J.; Rodriguez-Diaz, J.; Ruvoen-Clouet, N.; Svensson, L.; Le Pendu, J.; Larson, G. Human noroviruses recognize sialyl Lewis x neoglycoprotein. Glycobiology 2009, 19, 309–320. [Google Scholar] [CrossRef] [PubMed]
- Makino, A.; Shimojima, M.; Miyazawa, T.; Kato, K.; Tohya, Y.; Akashi, H. Junctional Adhesion Molecule 1 Is a Functional Receptor for Feline Calicivirus. J. Virol. 2006, 80, 4482–4490. [Google Scholar] [CrossRef] [PubMed]
- Glass, P.J.; White, L.J.; Ball, J.M.; Leparc-Goffart, I.; Hardy, M.E.; Estes, M.K. Norwalk Virus Open Reading Frame 3 Encodes a Minor Structural Protein. J. Virol. 2000, 74, 6581–6591. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.; Wang, M.; Wang, K.; Estes, M.K. Sequence and Genomic Organization of Norwalk Virus. Virology 1993, 195, 51–61. [Google Scholar] [CrossRef] [PubMed]
- Lambden, P.; Caul, E.; Ashley, C.; Clarke, I. Sequence and genome organization of a human small round-structured (Norwalk-like) virus. Science 1993, 259, 516–519. [Google Scholar] [PubMed]
- Crowder, S.; Kirkegaard, K. Trans-dominant inhibition of RNA viral replication can slow growth of drug-resistant viruses. Nat. Genet. 2005, 37, 701–709. [Google Scholar] [CrossRef] [PubMed]
- Simmonds, P.; Karakasiliotis, I.; Bailey, D.; Chaudhry, Y.; Evans, D.J.; Goodfellow, I.G. Bioinformatic and functional analysis of RNA secondary structure elements among different genera of human and animal caliciviruses. Nucleic Acids Res. 2008, 36, 2530–2546. [Google Scholar] [CrossRef] [PubMed]
- Victoria, M.; Colina, R.; Miagostovich, M.; Leite, J.; Cristina, J. Phylogenetic prediction of cis-acting elements: a cre-like sequence in Norovirus genome? BMC Res. Notes 2009, 2, 176. [Google Scholar] [CrossRef]
- Sosnovtsev, S.V.; Belliot, G.; Chang, K.; Prikhodko, V.G.; Thackray, L.B.; Wobus, C.E.; Karst, S.M.; Virgin, H.W.; Green, K.Y. Cleavage Map and Proteolytic Processing of the Murine Norovirus Nonstructural Polyprotein in Infected Cells. J. Virol. 2006, 80, 7816–7831. [Google Scholar] [CrossRef] [PubMed]
- Belliot, G.; Sosnovtsev, S.V.; Mitra, T.; Hammer, C.; Garfield, M.; Green, K.Y. In vitro Proteolytic Processing of the MD145 Norovirus ORF1 Nonstructural Polyprotein Yields Stable Precursors and Products Similar to Those Detected in Calicivirus-Infected Cells. J. Virol. 2003, 77, 10957–10974. [Google Scholar] [CrossRef] [PubMed]
- Seah, E.L.; Marshall, J.A.; Wright, P.J. trans Activity of the Norovirus Camberwell Proteinase and Cleavage of the N-Terminal Protein Encoded by ORF1. J. Virol. 2003, 77, 7150–7155. [Google Scholar] [CrossRef] [PubMed]
- Pfister, T.; Wimmer, E. Polypeptide p41 of a Norwalk-Like Virus Is a Nucleic Acid-Independent Nucleoside Triphosphatase. J. Virol. 2001, 75, 1611–1619. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Clarke, I.; Lambden, P. Polyprotein processing in Southampton virus: identification of 3C-like protease cleavage sites by in vitro mutagenesis. J. Virol. 1996, 70, 2605–2610. [Google Scholar] [PubMed]
- Fukushi, S.; Kojima, S.; Takai, R.; Hoshino, F.B.; Oka, T.; Takeda, N.; Katayama, K.; Kageyama, T. Poly(A)- and Primer-Independent RNA Polymerase of Norovirus. J. Virol. 2004, 78, 3889–3896. [Google Scholar] [CrossRef] [PubMed]
- Rohayem, J.; Robel, I.; Jager, K.; Scheffler, U.; Rudolph, W. Protein-Primed and De Novo Initiation of RNA Synthesis by Norovirus 3Dpol. J. Virol. 2006, 80, 7060–7069. [Google Scholar] [CrossRef] [PubMed]
- Burroughs, J.N.; Brown, F. Presence of a covalently linked protein on calicivirus RNA. J. Gen. Virol. 1978, 41, 443–446. [Google Scholar] [CrossRef] [PubMed]
- Herbert, T.P.; Brierley, I.; Brown, T.D. Identification of a protein linked to the genomic and subgenomic mRNAs of feline calicivirus and its role in translation. J. Gen. Virol. 1997, 78, 1033–1040. [Google Scholar] [PubMed]
- Schaffer, F.L.; Ehresmann, D.W.; Fretz, M.K.; Soergel, M.I. A protein, VPg, covalently linked to 36S calicivirus RNA. J. Gen. Virol. 1980, 47, 215–220. [Google Scholar] [CrossRef] [PubMed]
- Belliot, G.; Sosnovtsev, S.V.; Chang, K.; McPhie, P.; Green, K.Y. Nucleotidylylation of the VPg Protein of a Human Norovirus by its Proteinase-Polymerase Precursor Protein. Virology 2008, 374, 33–49. [Google Scholar] [CrossRef] [PubMed]
- Chaudhry, Y.; Nayak, A.; Bordeleau, M.; Tanaka, J.; Pelletier, J.; Belsham, G.J.; Roberts, L.O.; Goodfellow, I.G. Caliciviruses Differ in Their Functional Requirements for eIF4F Components. J. Biol. Chem. 2006, 281, 25315–25325. [Google Scholar] [CrossRef] [PubMed]
- Daughenbaugh, K.F.; Fraser, C.S.; Hershey, J.W.; Hardy, M.E. The genome-linked protein VPg of the Norwalk virus binds eIF3, suggesting its role in translation initiation complex recruitment. EMBO J. 2003, 22, 2852–2859. [Google Scholar] [CrossRef] [PubMed]
- Daughenbaugh, K.; Wobus, C.; Hardy, M. VPg of murine norovirus binds translation initiation factors in infected cells. Virol. J. 2006, 3, 33. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Vega, V.; Sosnovtsev, S.V.; Belliot, G.; King, A.D.; Mitra, T.; Gorbalenya, A.; Green, K.Y. Norwalk Virus N-Terminal Nonstructural Protein Is Associated with Disassembly of the Golgi Complex in Transfected Cells. J. Virol. 2004, 78, 4827–4837. [Google Scholar] [CrossRef] [PubMed]
- Ettayebi, K.; Hardy, M.E. Norwalk Virus Nonstructural Protein p48 Forms a Complex with the SNARE Regulator VAP-A and Prevents Cell Surface Expression of Vesicular Stomatitis Virus G Protein. J. Virol. 2003, 77, 11790–11797. [Google Scholar] [CrossRef] [PubMed]
- Hyde, J.L.; Sosnovtsev, S.V.; Green, K.Y.; Wobus, C.; Virgin, H.W.; Mackenzie, J.M. Mouse Norovirus Replication Is Associated with Virus-Induced Vesicle Clusters Originating from Membranes Derived from the Secretory Pathway. J. Virol. 2009, 83, 9709–9719. [Google Scholar] [CrossRef] [PubMed]
- Changotra, H.; Jia, Y.; Moore, T.N.; Liu, G.; Kahan, S.M.; Sosnovtsev, S.V.; Karst, S.M. Type I and Type II Interferons Inhibit the Translation of Murine Norovirus Proteins. J. Virol. 2009, 83, 5683–5692. [Google Scholar] [CrossRef] [PubMed]
- Chang, K.; George, D.W. Interferons and Ribavirin Effectively Inhibit Norwalk Virus Replication in Replicon-Bearing Cells. J. Virol. 2007, 81, 12111–12118. [Google Scholar] [CrossRef] [PubMed]
- Chen, R.; Neill, J.D.; Noel, J.S.; Hutson, A.M.; Glass, R.I.; Estes, M.K.; Prasad, B.V.V. Inter- and Intragenus Structural Variations in Caliciviruses and Their Functional Implications. J. Virol. 2004, 78, 6469–6479. [Google Scholar] [CrossRef] [PubMed]
- Prasad, B.V.; Rothnagel, R.; Jiang, X.; Estes, M.K. Three-dimensional structure of baculovirus-expressed Norwalk virus capsids. J. Virol. 1994, 68, 5117–5125. [Google Scholar] [PubMed]
- Prasad, B.V.V.; Hardy, M.E.; Dokland, T.; Bella, J.; Rossmann, M.G.; Estes, M.K. X-ray Crystallographic Structure of the Norwalk Virus Capsid. Science 1999, 286, 287–290. [Google Scholar] [CrossRef] [PubMed]
- Prasad, B.V.; Crawford, S.; Lawton, J.A.; Pesavento, J.; Hardy, M.; Estes, M.K. Structural studies on gastroenteritis viruses . Novartis Found. Symp. 2001, 238 discussion 37â46, 26–37. [Google Scholar] [PubMed]
- Katpally, U.; Wobus, C.E.; Dryden, K.; Virgin, H.W.; Smith, T.J. Structure of Antibody-Neutralized Murine Norovirus and Unexpected Differences from Viruslike Particles. J. Virol. 2008, 82, 2079–2088. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.; Wang, M.; Graham, D.Y.; Estes, M.K. Expression, self-assembly, and antigenicity of the Norwalk virus capsid protein. J. Virol. 1992, 66, 6527–6532. [Google Scholar] [PubMed]
- Tan, M.; Fang, P.; Chachiyo, T.; Xia, M.; Huang, P.; Fang, Z.; Jiang, W.; Jiang, X. Noroviral P particle: Structure, function and applications in virus-host interaction. Virology 2008, 382, 115–123. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.; Wilton, N.; Zhong, W.M.; Farkas, T.; Huang, P.W.; Barrett, E.; Guerrero, M.; Ruiz‐Palacios, G.; Green, K.Y.; Green, J.; Hale, A.D.; Estes, M.K.; Pickering, L.K.; Matson, D.O. Diagnosis of Human Caliciviruses by Use of Enzyme Immunoassays . J. Infect. Dis. 2000, 181, S349–S359. [Google Scholar] [CrossRef] [PubMed]
- Bu, W.; Mamedova, A.; Tan, M.; Xia, M.; Jiang, X.; Hegde, R.S. Structural Basis for the Receptor Binding Specificity of Norwalk Virus. J. Virol. 2008, 82, 5340–5347. [Google Scholar] [CrossRef] [PubMed]
- Cao, S.; Lou, Z.; Tan, M.; Chen, Y.; Liu, Y.; Zhang, Z.; Zhang, X.C.; Jiang, X.; Li, X.; Rao, Z. Structural Basis for the Recognition of Blood Group Trisaccharides by Norovirus. J. Virol. 2007, 81, 5949–5957. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.; Hutson, A.M.; Estes, M.K.; Prasad, B.V.V. Atomic resolution structural characterization of recognition of histo-blood group antigens by Norwalk virus. Proc. Natl. Acad. Sci. USA 2008, 105, 9175–9180. [Google Scholar] [CrossRef]
- Tan, M.; Huang, P.; Meller, J.; Zhong, W.; Farkas, T.; Jiang, X. Mutations within the P2 Domain of Norovirus Capsid Affect Binding to Human Histo-Blood Group Antigens: Evidence for a Binding Pocket. J. Virol. 2003, 77, 12562–12571. [Google Scholar] [CrossRef] [PubMed]
- Tan, M.; Xia, M.; Cao, S.; Huang, P.; Farkas, T.; Meller, J.; Hegde, R.S.; Li, X.; Rao, Z.; Jiang, X. Elucidation of strain-specific interaction of a GII-4 norovirus with HBGA receptors by site-directed mutagenesis study. Virology 2008, 379, 324–334. [Google Scholar] [CrossRef] [PubMed]
- Lochridge, V.P.; Jutila, K.L.; Graff, J.W.; Hardy, M.E. Epitopes in the P2 domain of norovirus VP1 recognized by monoclonal antibodies that block cell interactions. J. Gen. Virol. 2005, 86, 2799–2806. [Google Scholar] [CrossRef] [PubMed]
- Allen, D.J.; Noad, R.; Samuel, D.; Gray, J.J.; Roy, P.; Iturriza-Gomara, M. Characterisation of a GII-4 norovirus variant-specific surface-exposed site involved in antibody binding. Virol. J. 2009, 6, 150. [Google Scholar] [CrossRef]
- Bailey, D.; Thackray, L.B.; Goodfellow, I.G. A Single Amino Acid Substitution in the Murine Norovirus Capsid Protein Is Sufficient for Attenuation In vivo. J. Virol. 2008, 82, 7725–7728. [Google Scholar] [CrossRef] [PubMed]
- Glass, P.J.; Zeng, C.Q.; Estes, M.K. Two Nonoverlapping Domains on the Norwalk Virus Open Reading Frame 3 (ORF3) Protein Are Involved in the Formation of the Phosphorylated 35K Protein and in ORF3-Capsid Protein Interactions. J. Virol. 2003, 77, 3569–3577. [Google Scholar] [CrossRef] [PubMed]
- Bertolotti-Ciarlet, A.; Crawford, S.E.; Hutson, A.M.; Estes, M.K. The 3' End of Norwalk Virus mRNA Contains Determinants That Regulate the Expression and Stability of the Viral Capsid Protein VP1: a Novel Function for the VP2 Protein. J. Virol. 2003, 77, 11603–11615. [Google Scholar] [CrossRef] [PubMed]
- Luttermann, C.; Meyers, G. A Bipartite Sequence Motif Induces Translation Reinitiation in Feline Calicivirus RNA. J. Biol. Chem. 2007, 282, 7056–7065. [Google Scholar] [CrossRef] [PubMed]
- Meyers, G. Characterization of the Sequence Element Directing Translation Reinitiation in RNA of the Calicivirus Rabbit Hemorrhagic Disease Virus. J. Virol. 2007, 81, 9623–9632. [Google Scholar] [CrossRef] [PubMed]
- Meyers, G. Translation of the Minor Capsid Protein of a Calicivirus Is Initiated by a Novel Termination-dependent Reinitiation Mechanism. J. Biol. Chem. 2003, 278, 34051–34060. [Google Scholar] [CrossRef] [PubMed]
- Luttermann, C.; Meyers, G. The importance of inter- and intramolecular base pairing for translation reinitiation on a eukaryotic bicistronic mRNA. Genes Develop. 2009, 23, 331–344. [Google Scholar] [CrossRef]
- McCormick, C.J.; Salim, O.; Lambden, P.R.; Clarke, I.N. Translation Termination Reinitiation between Open Reading Frame 1 (ORF1) and ORF2 Enables Capsid Expression in a Bovine Norovirus without the Need for Production of Viral Subgenomic RNA. J. Virol. 2008, 82, 8917–8921. [Google Scholar] [CrossRef] [PubMed]
- Baron, R.C.; Greenberg, H.B.; Cukor, G.; Blacklow, N.R. Serological responses among teenagers after natural exposure to Norwalk virus. J. Infect. Dis. 1984, 150, 531–534. [Google Scholar] [PubMed]
- Blacklow, N.R.; Cukor, G.; Bedigian, M.K.; Echeverria, P.; Greenberg, H.B.; Schreiber, D.S.; Trier, J.S. Immune response and prevalence of antibody to Norwalk enteritis virus as determined by radioimmunoassay. J. Clin. Microbiol. 1979, 10, 903–909. [Google Scholar] [PubMed]
- Cukor, G.; Nowak, N.A.; Blacklow, N.R. Immunoglobulin M responses to the Norwalk virus of gastroenteritis. Infect. Immun. 1982, 37, 463–468. [Google Scholar] [PubMed]
- Johnson, P.C.; Hoy, J.; Mathewson, J.J.; Ericsson, C.D.; DuPont, H.L. Occurrence of Norwalk virus infections among adults in Mexico. J. Infect. Dis. 1990, 162, 389–393. [Google Scholar] [PubMed]
- Agus, S.G.; Falchuk, Z.M.; Sessoms, C.S.; Wyatt, R.G.; Dolin, R. Increased jejunal IgA synthesis in vitro during acute infectious nonbacterial gastroenteritis. Am. J. Dig. Dis. 1974, 19, 127–131. [Google Scholar] [CrossRef] [PubMed]
- Harrington, P.R.; Lindesmith, L.; Yount, B.; Moe, C.L.; Baric, R.S. Binding of Norwalk Virus-Like Particles to ABH Histo-Blood Group Antigens Is Blocked by Antisera from Infected Human Volunteers or Experimentally Vaccinated Mice. J. Virol. 2002, 76, 12335–12343. [Google Scholar] [CrossRef] [PubMed]
- Rockx, B.; Baric, R.S.; Grijs, I.D.; Duizer, E.; Koopmans, M.P. Characterization of the homo- and heterotypic immune responses after natural norovirus infection. J. Med. Virol. 2005, 77, 439–446. [Google Scholar] [CrossRef] [PubMed]
- Lindesmith, L.; Moe, C.; LePendu, J.; Frelinger, J.A.; Treanor, J.; Baric, R.S. Cellular and Humoral Immunity following Snow Mountain Virus Challenge. J. Virol. 2005, 79, 2900–2909. [Google Scholar] [CrossRef] [PubMed]
- Souza, M.; Cheetham, S.M.; Azevedo, M.S.P.; Costantini, V.; Saif, L.J. Cytokine and Antibody Responses in Gnotobiotic Pigs after Infection with Human Norovirus Genogroup II.4 (HS66 Strain) . J. Virol. 2007, 81, 9183–9192. [Google Scholar] [CrossRef] [PubMed]
- Donaldson, E.F.; Lindesmith, L.C.; Lobue, A.D.; Baric, R.S. Norovirus pathogenesis: mechanisms of persistence and immune evasion in human populations. Immunol. Rev. 2008, 225, 190–211. [Google Scholar] [CrossRef] [PubMed]
- Han, M.G.; Cheetham, S.; Azevedo, M.; Thomas, C.; Saif, L.J. Immune responses to bovine norovirus-like particles with various adjuvants and analysis of protection in gnotobiotic calves. Vaccine. 2006, 24, 317–326. [Google Scholar] [CrossRef] [PubMed]
- Souza, M.; Costantini, V.; Azevedo, M.S.; Saif, L.J. A human norovirus-like particle vaccine adjuvanted with ISCOM or mLT induces cytokine and antibody responses and protection to the homologous GII.4 human norovirus in a gnotobiotic pig disease model . Vaccine 2007, 25, 8448–8459. [Google Scholar] [CrossRef] [PubMed]
- LoBue, A.D.; Thompson, J.M.; Lindesmith, L.; Johnston, R.E.; Baric, R.S. Alphavirus-Adjuvanted Norovirus-Like Particle Vaccines: Heterologous, Humoral, and Mucosal Immune Responses Protect against Murine Norovirus Challenge. J. Virol. 2009, 83, 3212–3227. [Google Scholar] [CrossRef] [PubMed]
- Mackinnon, M.J.; Read, A.F. Immunity Promotes Virulence Evolution in a Malaria Model . PLoS Biol. 2004, 2, e230. [Google Scholar] [CrossRef] [PubMed]
- Hurley, K.E.; Pesavento, P.A.; Pedersen, N.C.; Poland, A.M.; Wilson, E.; Foley, J.E. An outbreak of virulent systemic feline calicivirus disease. J. Am. Vet. Med. Assoc. 2004, 224, 241–249. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, N.C.; Elliott, J.B.; Glasgow, A.; Poland, A.; Keel, K. An isolated epizootic of hemorrhagic-like fever in cats caused by a novel and highly virulent strain of feline calicivirus. Vet. Microbiol. 2000, 73, 281–300. [Google Scholar] [CrossRef] [PubMed]
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Share and Cite
Karst, S.M. Pathogenesis of Noroviruses, Emerging RNA Viruses. Viruses 2010, 2, 748-781. https://doi.org/10.3390/v2030748
Karst SM. Pathogenesis of Noroviruses, Emerging RNA Viruses. Viruses. 2010; 2(3):748-781. https://doi.org/10.3390/v2030748
Chicago/Turabian StyleKarst, Stephanie M. 2010. "Pathogenesis of Noroviruses, Emerging RNA Viruses" Viruses 2, no. 3: 748-781. https://doi.org/10.3390/v2030748
APA StyleKarst, S. M. (2010). Pathogenesis of Noroviruses, Emerging RNA Viruses. Viruses, 2(3), 748-781. https://doi.org/10.3390/v2030748



