The Role of Nucleotide Excision by Reverse Transcriptase in HIV Drug Resistance
Abstract
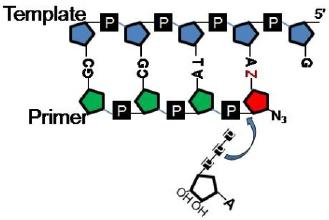
:1. Introduction
| Common name | Abbrev. | Structural name | Natural analog | Year FDA approved | Common resistance mutations selected b | Mechanism of resistance |
|---|---|---|---|---|---|---|
| zidovudine | ZDV, AZT | β-D-(+)-3′-azido-3′-deoxythymidine | dTTP | 1987 | M41L, D67N, K70R, L210W, T215F/Y, K219Q/E | excision |
| didanosine | ddI | β-D-(+)-2′,3-dideoxyinosine | dATP | 1991 | K65R, L74V | discrimination |
| stavudine | d4T | β-D-(+)-2′,3′-didehydro-3′-deoxythymidine | dTTP | 1994 | M41L, D67N, K70R, L210W, T215F/Y, K219Q/E | excision |
| lamivudine | 3TC | β-L-(−)-2′,3′-dideoxy-3′-thiacytidine | dCTP | 1995 | K65R, M184V/I | discrimination |
| abacavir | ABC | (1S,4R)-4-[2-amino-6-(cyclopropyl-amino)-9H-purin-9-yl]-2-cyclopentene-1-methanol succinate | dGTP | 1998 | K65R, L74V, Y115F, M184V | discrimination |
| tenofovir c | TFV | ({[(2R)-1-(6-amino-9H-purin-9-yl)propan-2- yl]oxy}methyl)phosphonic acid | dATP | 2001 | K65R, K70E | discrimination |
| emtricitabine | FTC | β-L-(−)-2′,3′-dideoxy-5-fluoro-3′-thiacytidine | dCTP | 2003 | K65R, M184V/I | discrimination |
2. Mutants of HIV-1 RT that Enhance the Rate of Nucleotide Excision

3. Mutations of HIV-1 RT that Decrease the Rate of Nucleotide Excision and Suppress AZT Resistance
| Location in RT structure | How isolated? | Mutations | AZT sensitivity | Excision activity in vitro |
|---|---|---|---|---|
| dNTP binding site | Site-directed mutagenesis or bacterial library mutagenesis | D113G/E, A114S/G, Y115N/H, Q151H [66,70] R72A [71] | Hypersensitivity shown for D113E & A114S [70] | Reduced for A114S [72] |
| From patients treated with NRTIs | K65R [73–76] | Suppression of AZT resistance [37,77–80] | Reduced for K65R [37,78] | |
| Palm domain between the fingers domain and the template strand | From PFA-treated patients | W88S, W88G, Q161L, H208Y [68] | Suppression of AZT resistance shown for W88G and Q161L [37] No effect for W88S [37] | Reduced for W88G and Q161L [37] Not reduced for W88S [37] |
| Serial passage of HIV-1 in culture | Q161L/H208Y [68] E89K, L92I, S156A [67] W88G, S117T, F160Y, M164I [76] | Suppression of AZT resistance shown for W88G, E89K, Q161L/H208Y, S117T, and M164I [37,67–69] | Reduced for W88G, E89K, Q161L/H208Y, S117T, and M164I [37] | |
| Site-directed or random mutagenesis | K154E, Y183S, G190R [66,70,81] E89G/T/A/D/V[82,83] V90A [84] | AZT sensitivity not determined in infectivity assays | Not done |


4. Additional Mutations of HIV-1 RT that Suppress AZT Resistance (L74V, Y181C, L100I, V75I, M184V and K65R)
5. Indirect Enhancement of Excision due to Mutations in the RNase H and Connection Domains of RT
6. Conclusions
Acknowledgments
References
- Fischl, M.A.; Richman, D.D.; Grieco, M.H.; Gottlieb, M.S.; Volberding, P.A.; Laskin, O.L.; Leedom, J.M.; Groopman, J.E.; Mildvan, D.; Schooley, R.T.; Jackson, G.G.; Durack, D.T.; King, D. The efficacy of azidothymidine (AZT) in the treatment of patients with AIDS and AIDS-related complex. A double-blind placebo-controlled trial. N. Engl. J. Med. 1987, 317, 185–191. [Google Scholar] [PubMed]
- De Clercq, E. Non-nucleoside reverse transcriptase inhibitors (NNRTIs): Past, present, and future. Chem. Biodivers. 2004, 1, 44–64. [Google Scholar] [CrossRef]
- Domaoal, R.A.; Demeter, L.M. Structural and biochemical effects of human immunodeficiency virus mutants resistant to non-nucleoside reverse transcriptase inhibitors. Int. J. Biochem. Cell Biol. 2004, 36, 1735–1751. [Google Scholar] [CrossRef] [PubMed]
- Sluis-Cremer, N.; Temiz, N.A.; Bahar, I. Conformational changes in HIV-1 reverse transcriptase induced by nonnucleoside reverse transcriptase inhibitor binding. Curr. HIV Res. 2004, 2, 323–332. [Google Scholar] [CrossRef] [PubMed]
- Das, K.; Lewi, P.J.; Hughes, S.H.; Arnold, E. Crystallography and the design of anti-AIDS drugs: Conformational flexibility and positional adaptability are important in the design of non-nucleoside HIV-1 reverse transcriptase inhibitors. Prog. Biophys. Mol. Biol. 2005, 88, 209–231. [Google Scholar] [CrossRef] [PubMed]
- Yin, P.D.; Das, D.; Mitsuya, H. Overcoming HIV drug resistance through rational drug design based on molecular, biochemical, and structural profiles of HIV resistance. Cell. Mol. Life Sci. 2006, 63, 1706–1724. [Google Scholar] [CrossRef] [PubMed]
- El Safadi, Y.; Vivet-Boudou, V.; Marquet, R. HIV-1 reverse transcriptase inhibitors. Appl. Microbiol. Biotechnol. 2007, 75, 723–737. [Google Scholar] [CrossRef] [PubMed]
- Sluis-Cremer, N.; Tachedjian, G. Mechanisms of inhibition of HIV replication by non-nucleoside reverse transcriptase inhibitors. Virus Res. 2008, 134, 147–156. [Google Scholar] [CrossRef] [PubMed]
- Sarafianos, S.G.; Marchand, B.; Das, K.; Himmel, D.M.; Parniak, M.A.; Hughes, S.H.; Arnold, E. Structure and function of HIV-1 reverse transcriptase: Molecular mechanisms of polymerization and inhibition. J. Mol. Biol. 2009, 385, 693–713. [Google Scholar] [CrossRef] [PubMed]
- Maga, G.; Radi, M.; Gerard, M.-A.; Botta, M.; Ennifar, E. HIV-1 RT inhibitors with a novel mechanism of action: NNRTIs that are competing with the nucleotide substrate. Viruses submitted for publication.
- Parniak, M.A.; McBurney S.; Oldfield, E.; Mellors, J.W. Bisphosphonate inhibitors of nucleoside reverse transcriptase inhibitor excision. (abstract 26: Tenerife Sur-Costa Adeje: Canary Islands, Spain). Antivir. Ther. 2004, 9, S32. [Google Scholar]
- Cruchaga, C.; Ansó, E.; Rouzaut, A.; Martínez-Irujo, J.J. Selective excision of chain-terminating nucleotides by HIV-1 reverse transcriptase with phosphonoformate as substrate. J. Biol. Chem. 2006, 281, 27744–27752. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.; Chan, J.M.W.; Tovian, Z.; Secrest, A.; Nagy, E.; Krysiak, K.; Bergan, K.; Parniak, M.A.; Oldfield, E. Bisphosphonate inhibitors of ATP-mediated HIV-1 reverse transcriptase catalyzed excision of chain-terminating 3'-azido, 3'-deoxythymidine: A QSAR investigation. Bioorg. Med. Chem. 2008, 16, 8959–8967. [Google Scholar] [CrossRef] [PubMed]
- Shafer, R.W. Genotypic testing for human immunodeficiency virus type 1 drug resistance. Clin. Micro. Revs. 2002, 15, 247–277. [Google Scholar] [CrossRef]
- Goldschmidt, V.; Marquet, R. Primer unblocking by HIV-1 reverse transcriptase and resistance to nucleoside RT inhibitors (NRTIs). Intl. J. Biochem. Cell Biol. 2004, 36, 1687–1705. [Google Scholar] [CrossRef]
- Sarafianos, S.G.; Hughes, S.H.; Arnold, E. Designing anti-AIDS drugs targeting the major mechanism of HIV-1 RT resistance to nucleoside analog drugs. Intl. J. Biochem. Cell Biol. 2004, 36, 1706–1715. [Google Scholar] [CrossRef]
- Menéndez-Arias, L. Mechanisms of resistance to nucleoside analogue inhibitors of HIV-1 reverse transcriptase. Virus Res. 2008, 134, 124–146. [Google Scholar] [CrossRef] [PubMed]
- Menéndez-Arias, L. Molecular basis of human immunodeficiency virus drug resistance: An update. Antivir. Res. 2010, 85, 210–231. [Google Scholar] [CrossRef]
- Johnson, V.A.; Brun-Vézinet, F.; Clotet, B.; Günthard, H.F.; Kuritzkes, D.R.; Pillay, D.; Schapiro, J.M.; Richman, D.D. Update of the drug resistance mutations in HIV-1: December 2008. Top. HIV Med. 2008, 16, 138–145. [Google Scholar] [PubMed]
- Larder, B.A.; Kemp, S.D. Multiple mutations in HIV-1 reverse transcriptase confer high-level resistance to zidovudine (AZT). Science 1989, 246, 1155–1158. [Google Scholar] [PubMed]
- Kellam, P.; Boucher, C.A.B.; Larder, B.A. Fifth mutation in human immunodeficiency virus type 1 reverse transcriptase contributes to the development of high-level resistance to zidovudine. Proc. Natl. Acad. Sci. USA 1992, 89, 1934–1938. [Google Scholar] [CrossRef]
- Harrigan, P.R.; Kinghorn, I.; Bloor, S.; Kemp, S.D.; Nájera, I.; Kohli, A.; Larder, B.A. Significance of amino acid variation at human immunodeficiency virus type 1 reverse transcriptase residue 210 for zidovudine susceptibility. J. Virol. 1996, 70, 5930–5934. [Google Scholar] [PubMed]
- Hooker, D.J.; Tachedjian, G.; Solomon, A.E.; Gurusinghe, A.D.; Land, S.; Birch, C.; Anderson, J.L; Roy, B.M.; Arnold, E.; Deacon, N.J. An in vivo mutation from leucine to tryptophan at position 210 in human immunodeficiency virus type 1 reverse transcriptase contributes to high-level resistance to 3'-azido-3'-deoxythymidine. J. Virol. 1996, 70, 8010–8018. [Google Scholar] [PubMed]
- Scott, W.A. The enzymatic basis for thymidine analogue resistance in HIV-1. AIDS Rev. 2001, 3, 194–200. [Google Scholar]
- Whitcomb, J.M.; Parkin, N.T.; Chappey, C.; Hellmann, N.S.; Petropoulos, C.J. Broad nucleoside reverse-transcriptase inhibitor cross-resistance in human immunodeficiency virus type 1 clinical isolates. J. Infect. Dis. 2003, 188, 992–1000. [Google Scholar] [CrossRef] [PubMed]
- Boucher, C.A.B.; O'Sullivan, E.; Mulder, J.W.; Ramautarsing, C.; Kellam, P.; Darby, G.; Lange, J.M.A.; Goudsmit, J.; Larder, B.A. Ordered appearance of zidovudine resistance mutations during treatment of 18 human immunodeficiency virus-positive subjects. J. Infect. Dis. 1992, 165, 105–110. [Google Scholar] [PubMed]
- Larder, B.A. Interactions between drug resistance mutations in human immunodeficiency virus type 1 reverse transcriptase. J. Gen. Virol. 1994, 75, 951–957. [Google Scholar] [CrossRef] [PubMed]
- Lacey, S.F.; Reardon, J.E.; Furfine, E.S.; Kunkel, T.A.; Bebenek, K.; Eckert, K.A.; Kemp, S.D.; Larder, B.A. Biochemical studies on the reverse transcriptase and RNase H activities from human immunodeficiency virus strains resistant to 3'-azido-3'-deoxythymidine. J. Biol. Chem. 1992, 267, 15789–15794. [Google Scholar] [PubMed]
- Carroll, S.S.; Geib, J.; Olsen, D.B.; Stahlhut, M.; Shafer, J.A.; Kuo, L.C. Sensitivity of HIV-1 reverse transcriptase and its mutants to inhibition by azidothymidine triphosphate. Biochemistry 1994, 33, 2113–2120. [Google Scholar] [CrossRef] [PubMed]
- Krebs, R.; Immendörfer, U.; Thrall, S.H.; Wöhrl, B.M.; Goody, R.S. Single-step kinetics of HIV-1 reverse transcriptase mutants responsible for virus resistance to nucleoside inhibitors zidovudine and 3-TC. Biochemistry 1997, 36, 10292–10300. [Google Scholar] [CrossRef] [PubMed]
- Arion, D.; Kaushik, N.; McCormick, S.; Borkow, G.; Parniak, M.A. Phenotypic mechanism of HIV-1 resistance to 3'-azido-3'-deoxythymidine (AZT): Increased polymerization processivity and enhanced sensitivity to pyrophosphate of the mutant viral reverse transcriptase. Biochemistry 1998, 37, 15908–15917. [Google Scholar] [CrossRef] [PubMed]
- Meyer, P.R.; Matsuura, S.E.; So, A.G.; Scott, W.A. Unblocking of chain-terminated primer by HIV-1 reverse transcriptase through a nucleotide-dependent mechanism. Proc. Natl. Acad. Sci. USA 1998, 95, 13471–13476. [Google Scholar] [CrossRef]
- Meyer, P.R.; Matsuura, S.E.; Mian, A.M.; So, A.G.; Scott, W.A. A mechanism of AZT resistance: An increase in nucleotide-dependent primer unblocking by mutant HIV-1 reverse transcriptase. Mol. Cell 1999, 4, 35–43. [Google Scholar] [CrossRef]
- Meyer, P.R.; Smith, A.J.; Matsuura, S.E.; Scott, W.A. Chain-terminating dinucleoside tetraphosphates are substrates for DNA polymerization by human immunodeficiency virus type 1 reverse transcriptase with increased activity against thymidine analogue-resistant mutants. Antimicrob. Agents Chemother. 2006, 50, 3607–3614. [Google Scholar] [CrossRef] [PubMed]
- Dharmasena, S.; Pongracz, Z.; Arnold, E.; Sarafianos, S.G.; Parniak, M.A. 3'-Azido-3'-deoxythymidine-(5')-tetraphospho-(5')-adenosine, the product of ATP-mediated excision of chain-terminating AZTMP, is a potent chain-terminating substrate for HIV-1 reverse transcriptase. Biochemistry 2007, 46, 828–836. [Google Scholar] [CrossRef] [PubMed]
- Shao, X.W.; Hjalmarsson, S.; Lennerstrand, J.; Svennerholm, B.; Blomberg, J.; Källander, C.F.R.; Gronowitz, J.S. Application of a colorimetric chain-termination assay for characterization of reverse transcriptase from 3'-azido-2', 3’-deoxythymidine-resistant HIV isolates. Biotechnol. Appl. Biochem. 2002, 35, 155–164. [Google Scholar] [CrossRef] [PubMed]
- Meyer, P.R.; Matsuura, S.E.; Zonarich, D.; Chopra, R.R.; Pendarvis, E.; Bazmi, H.Z.; Mellors, J.W.; Scott, W.A. Relationship between 3'-azido-3'-deoxythymidine resistance and primer unblocking activity in foscarnet-resistant mutants of human immunodeficiency virus type 1 reverse transcriptase. J. Virol. 2003, 77, 6127–6137. [Google Scholar] [CrossRef] [PubMed]
- Boyer, P.L. Selective excision of AZTMP by drug-resistant human immunodeficiency virus reverse transcriptase. J. Virol. 2001, 75, 4832–4842. [Google Scholar] [CrossRef] [PubMed]
- Chamberlain, P.P.; Ren, J.; Nichols, C.E.; Douglas, L.; Lennerstrand, J. Crystal structures of zidovudine- or lamivudine-resistant human immunodeficiency virus type 1 reverse transcriptases containing mutations at codons 41, 184, and 215. J. Virol. 2002, 76, 10015–10019. [Google Scholar] [CrossRef] [PubMed]
- Smith, A.J.; Scott, W.A. The influence of natural substrates and inhibitors on the nucleotide-dependent excision activity of HIV-1 reverse transcriptase in the infected cell. Curr. Pharm. Des. 2006, 12, 1827–1841. [Google Scholar] [CrossRef] [PubMed]
- Smith, A.J.; Meyer, P.R.; Asthana, D.; Ashman, M.R.; Scott, W.A. Intracellular substrates for the primer-unblocking reaction by human immunodeficiency virus type 1 reverse transcriptase: detection and quantitation in extracts from quiescent- and activated-lymphocyte subpopulations. Antimicrob. Agents Chemother. 2005, 49, 1761–1769. [Google Scholar] [CrossRef] [PubMed]
- Ray, A.S.; Murakami, E.; Basavapathruni, A.; Vaccaro, J.A.; Ulrich, D.; Chu, C.K.; Schinazi, R.F.; Anderson, K.S. Probing the molecular mechanisms of AZT drug resistance mediated by HIV-1 reverse transcriptase using a transient kinetic analysis. Biochemistry 2003, 42, 8831–8841. [Google Scholar] [CrossRef] [PubMed]
- Meyer, P.R.; Matsuura, S.E.; Schinazi, R.F.; So, A.G.; Scott, W.A. Differential removal of thymidine nucleotide analogues from blocked DNA chains by human immunodeficiency virus reverse transcriptase in the presence of physiological concentrations of 2'-deoxynucleoside triphosphates. Antimicrob. Agents Chemother. 2000, 44, 3465–3472. [Google Scholar] [CrossRef] [PubMed]
- Tong, W.; Lu, C.D.; Sharma, S.K.; Matsuura, S.; So, A.G.; Scott, W.A. Nucleotide-induced stable complex formation by HIV-1 reverse transcriptase. Biochemistry 1997, 36, 5749–5757. [Google Scholar] [CrossRef] [PubMed]
- Sarafianos, S.G.; Clark, A.D.; Das, K.; Tuske, S.; Birktoft, J.J.; Ilankumaran, P. Structures of HIV-1 reverse transcriptase with pre- and post-translocation AZTMP-terminated DNA. EMBO J. 2002, 21, 6614–6624. [Google Scholar] [CrossRef] [PubMed]
- Marchand, B.; Götte, M. Site-specific footprinting reveals differences in the translocation status of HIV-1 reverse transcriptase. J. Biol. Chem. 2003, 278, 35362–35372. [Google Scholar] [CrossRef] [PubMed]
- Meyer, P.R.; Rutvisuttinunt, W.; Matsuura, S.E.; So, A.G.; Scott, W.A. Stable complexes formed by HIV-1 reverse transcriptase at distinct positions on the primer-template controlled by binding deoxynucleoside triphosphates or foscarnet. J. Mol. Biol. 2007, 369, 41–54. [Google Scholar] [CrossRef] [PubMed]
- Lacey, S.F.; Larder, B.A. Mutagenic study of codons 74 and 215 of the human immunodeficiency virus type 1 reverse transcriptase, which are significant in nucleoside analog resistance. J. Virol. 1994, 68, 3421–3424. [Google Scholar] [PubMed]
- Meyer, P.R.; Matsuura, S.E.; Tolun, A.A.; Pfeifer, I.; So, A.G.; Mellors, J.W.; Scott, W.A. Effects of specific zidovudine resistance mutations and substrate structure on nucleotide-dependent primer unblocking by human immunodeficiency virus type 1 reverse transcriptase. Antimicrob. Agents Chemother. 2002, 46, 1540–1545. [Google Scholar] [CrossRef] [PubMed]
- Boyer, P.L.; Sarafianos, S.G.; Arnold, E.; Hughes, S.H. Nucleoside analog resistance caused by insertions in the fingers of human immunodeficiency virus type 1 reverse transcriptase involves ATP-mediated excision. J. Virol. 2002, 76, 9143–9151. [Google Scholar] [CrossRef] [PubMed]
- Meyer, P.R.; Lennerstrand, J.; Matsuura, S.E.; Larder, B.A.; Scott, W.A. Effects of dipeptide insertions between codons 69 and 70 of human immunodeficiency virus type 1 reverse transcriptase on primer unblocking, deoxynucleoside triphosphate inhibition, and DNA chain elongation. J. Virol. 2003, 77, 3871–3877. [Google Scholar] [CrossRef] [PubMed]
- Winters, M.A.; Merigan, T.C. Insertions in the human immunodeficiency virus type 1 protease and reverse transcriptase genes: Clinical impact and molecular mechanisms. Antimicrob. Agents Chemother. 2005, 49, 2575–2582. [Google Scholar] [CrossRef] [PubMed]
- Menéndez-Arias, L.; Matamoros, T.; Cases-González, C.E. Insertions and deletions in HIV-1 reverse transcriptase: Consequences for drug resistance and viral fitness. Curr. Pharm. Des. 2006, 12, 1811–1825. [Google Scholar] [CrossRef] [PubMed]
- Eggink, D.; Huigen, M.C.D.G.; Boucher, C.A.B.; Götte, M.; Nijhuis, M. Insertions in the β3-β4 loop of reverse transcriptase of human immunodeficiency virus type 1 and their mechanism of action, influence on drug susceptibility and viral replication capacity. Antivir. Res. 2007, 75, 93–103. [Google Scholar] [CrossRef]
- Arzumanov, A.A.; Semizarov, D.G.; Victorova, L.S.; Dyatkina, N.B.; Krayevsky, A.A. γ-Phosphate-substituted 2'-deoxynucleoside 5'-triphosphates as substrates for DNA polymerases. J. Biol. Chem. 1996, 271, 24389–24394. [Google Scholar] [CrossRef] [PubMed]
- Alexandrova, L.A.; Skoblov, A.Y.; Jasko, M.V.; Victorova, L.S.; Krayevsky, A.A. 2'-Deoxynucleoside 5'-triphosphates modified at α-, β- and γ-phosphates as substrates for DNA polymerases. Nucl. Acids Res. 1998, 26, 778–786. [Google Scholar] [CrossRef]
- Mulder, B.A.; Anaya, S.; Yu, P.; Lee, K.W.; Nguyen, A.; Murphy, J.; Willson, R.; Briggs, J.M.; Gao, X.; Hardin, S.H. Nucleotide modification at the γ-phosphate leads to improved fidelity of HIV-1 reverse transcriptase. Nucl. Acids Res. 2005, 33, 4865–4873. [Google Scholar] [CrossRef]
- Adelfinskaya, O.; Herdewijn, P. Amino acid phosphoramidate nucleotides as alternative substrates for HIV-1 reverse transcriptase. Angew. Chem. Int. Ed. 2007, 46, 4356–4358. [Google Scholar] [CrossRef]
- Adelfinskaya, O.; Terrazas, M.; Froeyen, M.; Marlière, P.; Nauwelaerts, K.; Herdewijn, P. Polymerase-catalyzed synthesis of DNA from phosphoramidate conjugates of deoxynucleotides and amino acids. Nucl. Acids Res. 2007, 35, 5060–5072. [Google Scholar] [CrossRef]
- Terrazas, M.; Marlière, P.; Herdewijn, P. Enzymatically catalyzed DNA synthesis using L-asp-dGMP, L-Asp-dCMP, and L-Asp-dTMP. Chem. Biodivers. 2008, 5, 31–39. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Giraut, A.; Dyubankova, N.; Song, X.P.; Herdewijn, P. Phosphodiester substrates for incorporation of nucleotides in DNA using HIV-1 reverse transcriptase. Chem. Bio. Chem. 2009, 10, 2246–2252. [Google Scholar] [CrossRef] [PubMed]
- Garforth, S.J.; Parniak, M.A.; Prasad, V.R. Utilization of a deoxynucleoside diphosphate substrate by HIV reverse transcriptase. PLoS ONE 2008, 3, e2074. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Victorova, L.; Sosunov, V.; Skoblov, A.; Shipytsin, A.; Krayevsky, A. New substrates of DNA polymerases. FEBS Lett. 1999, 453, 6–10. [Google Scholar] [CrossRef] [PubMed]
- Han, Q.; Sarafianos, S.G.; Arnold, E.; Parniak, M.A.; Gaffney, B.L.; Jones, R.A. Synthesis of AZTpspcx2ppsA and AZTpspcx2ppsAZT: Hydrolysis-resistant potential inhibitors of the AZT excision reaction of HIV-1 RT. Org. Lett. 2007, 9, 5243–5246. [Google Scholar] [CrossRef] [PubMed]
- Selmi, B.; Deval, J.; Alvarez, K.; Boretto, J.; Sarfati, S.; Guerreiro, C.; Canard, B. The Y181C substitution in 3'-azido-3'-deoxythymidine-resistant human immunodeficiency virus, type 1, reverse transcriptase suppresses the ATP-mediated repair of the 3'-azido-3'-deoxythymidine 5'-monophosphate-terminated primer. J. Biol. Chem. 2003, 278, 40464–40472. [Google Scholar] [CrossRef] [PubMed]
- Larder, B.A.; Kemp, S.D.; Purifoy, D.J.M. Infectious potential of human immunodeficiency virus type 1 reverse transcriptase mutants with altered inhibitor sensitivity. Proc. Natl. Acad. Sci. USA 1989, 86, 4803–4807. [Google Scholar] [CrossRef]
- Tachedjian, G.; Hooker, D.J.; Gurusinghe, A.D.; Bazmi, H.; Deacon, N.J.; Mellors, J.; Birch, C.; Mills, J. Characterization of foscarnet-resistant strains of human immunodeficiency virus type 1. Virology 1995, 212, 58–68. [Google Scholar] [CrossRef] [PubMed]
- Mellors, J.W.; Bazmi, H.Z.; Schinazi, R.F.; Roy, B.M.; Hsiou, Y.; Arnold, E.; Weir, J.; Mayers, D.L. Novel mutations in reverse transcriptase of human immunodeficiency virus type 1 reduce susceptibility to foscarnet in laboratory and clinical isolates. Antimicob. Agents Chemother. 1995, 39, 1087–1092. [Google Scholar] [CrossRef]
- Tachedjian, G.; Mellors, J.; Bazmi, H.; Birch, C.; Mills, J. Zidovudine resistance is suppressed by mutations conferring resistance of human immunodeficiency virus type 1 to foscarnet. J. Virol. 1996, 70, 7171–7181. [Google Scholar] [PubMed]
- Larder, B.A.; Purifoy, D.J.M.; Powell, K.L.; Darby G. Site-specific mutagenesis of AIDS virus reverse transcriptase. Nature 1987, 327, 716–717. [Google Scholar] [CrossRef] [PubMed]
- Sarafianos, S.G.; Pandey, V.N.; Kaushik, N.; Modak, M.J. Site-directed mutagenesis of arginine 72 of HIV-1 reverse transcriptase. Catalytic role and inhibitor sensitivity. J. Biol. Chem. 1995, 270, 19729–19735. [Google Scholar] [CrossRef] [PubMed]
- Arion, D.; Sluis-Cremer, N.; Parniak, M.A. Mechanism by which phosphonoformic acid resistance mutations restore 3'-azido-3'-deoxythymidine (AZT) sensitivity to AZT-resistant HIV-1 reverse transcriptase. J. Biol. Chem. 2000, 275, 9251–9255. [Google Scholar] [CrossRef] [PubMed]
- Gu, Z.; Gao, Q.; Fang, H.; Salomon, H.; Parniak, M.A.; Goldberg, E.; Cameron, J.; Wainberg, M.A. Identification of a mutation at codon 65 in the IKKK motif of reverse transcriptase that encodes human immunodeficiency virus resistance to 2',3'-dideoxycytidine and 2',3'-dideoxy-3'-thiacytidine. Antimicrob. Agents Chemother. 1994, 38, 275–281. [Google Scholar] [PubMed]
- Zhang, D.; Caliendo, A.M.; Eron, J.J.; DeVore, K.M.; Kaplan, J.C.; Hirsch, M.S.; D'Aquila, R.T. Resistance to 2',3'-dideoxycytidine conferred by a mutation in codon 65 of the human immunodeficiency virus type 1 reverse transcriptase. Antimicrob. Agents Chemother. 1994, 38, 282–287. [Google Scholar] [PubMed]
- Sluis-Cremer, N.; Arion, D.; Kaushik, N.; Lim, H.; Parniak, M.A. Mutational analysis of Lys65 of HIV-1 reverse transcriptase. Biochem. J. 2000, 348, 77–82. [Google Scholar] [CrossRef] [PubMed]
- Hammond, J.L.; Koontz, D.L.; Bazmi, H.Z.; Beadle, J.R.; Hostetler, S.E.; Kini, G.D.; Aldern, K.A.; Richman, D.D.; Hostetler, K.Y.; Mellors, J.W. Alkylglycerol prodrugs of phosphonoformate are potent in vitro inhibitors of nucleoside-resistant human immunodeficiency virus type 1 and select for resistance mutations that suppress zidovudine resistance. Antimicrob. Agents Chemother. 2001, 45, 1621–1628. [Google Scholar] [CrossRef] [PubMed]
- Bazmi, H.Z.; Hammond, J.L.; Cavalcanti, S.C.H.; Chu, C.K.; Schinazi, R.F.; Mellors, J.W. In vitro selection of mutations in the human immunodeficiency virus type 1 reverse transcriptase that decrease susceptibility to (-)-β-D-dioxolane-guanosine and suppress resistance to 3'-azido-3'-deoxythymidine. Antimicrob. Agents Chemother. 2000, 44, 1783–1788. [Google Scholar] [CrossRef] [PubMed]
- White, K.L.; Chen, J.M.; Feng, J.Y.; Margot, N.A.; Ly, J.K.; Ray, A.S.; MacArthur, H.L.; McDermott, M.J.; Swaminathan, S.; Miller, M.D. The K65R reverse transcriptase mutation in HIV-1 reverses the excision phenotype of zidovudine resistance mutations. Antivir. Ther. 2006, 11, 155–163. [Google Scholar] [PubMed]
- Parikh, U.M.; Barnas, D.C.; Faruki, H.; Mellors, J.W. Antagonism between the HIV-1 reverse-transcriptase mutation K65R and thymidine-analogue mutations at the genomic level. J. Infect. Dis. 2006, 194, 651–660. [Google Scholar] [CrossRef] [PubMed]
- Parikh, U.M.; Bacheler, L.; Koontz, D.; Mellors, J.W. The K65R mutation in human immunodeficiency virus type 1 reverse transcriptase exhibits bidirectional phenotypic antagonism with thymidine analog mutations. J. Virol. 2006, 80, 4971–4977. [Google Scholar] [CrossRef] [PubMed]
- Lowe, D.M.; Parmar, V.; Kemp, S.D.; Larder, B.A. Mutational analysis of two conserved sequence motifs in HIV-1 reverse transcriptase. FEBS Lett 1991, 282, 231–234. [Google Scholar] [CrossRef] [PubMed]
- Prasad, V.R.; Lowy, I.; De Los Santos, T.; Chiang, L.; Goff, S.P. Isolation and characterization of a dideoxyguanosine triphosphate-resistant mutant of human immunodeficiency virus reverse transcriptase. Proc. Natl. Acad. Sci. USA 1991, 88, 11363–11367. [Google Scholar] [CrossRef]
- Song, Q.; Yang, G.; Goff, S.P.; Prasad, V.R. Mutagenesis of the Glu-89 residue in human immunodeficiency virus type 1 (HIV-1) and HIV-2 reverse transcriptases: Effects on nucleoside analog resistance. J. Virol. 1992, 66, 7568–7571. [Google Scholar] [PubMed]
- Im, G.J.; Tramontano, E.; Gonzalez, C.J.; Cheng, Y.C. Identification of the amino acid in the human immunodeficiency virus type 1 reverse transcriptase involved in pyrophosphate binding of antiviral nucleoside triphosphate analogs and phosphonoformate. Implications for multiple drug resistance. Biochem. Pharmacol. 1993, 46, 2307–2313. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.; Chopra, R.; Verdine, G.L.; Harrison, S.C. Structure of a covalently trapped catalytic complex of HIV-1 reverse transcriptase: Implications for drug resistance. Science 1998, 282, 1669–1675. [Google Scholar] [CrossRef] [PubMed]
- Das, K.; Bandwar, R.P.; White, K.L.; Feng, J.Y.; Sarafianos, S.G.; Tuske, S.; Tu, X.; Clark, A.D.; Boyer, P.L.; Hou, X.; Gaffney, B.L.; Jones, R.A.; Miller, M.D.; Hughes, S.H.; Arnold, E. Structural basis for the role of the K65R mutation in HIV-1 reverse transcriptase polymerization, excision antagonism, and tenofovir resistance. J. Biol. Chem. 2009, 284, 35092–35100. [Google Scholar] [CrossRef] [PubMed]
- Götte, M. Effects of nucleotides and nucleotide analogue inhibitors of HIV-1 reverse transcriptase in a ratchet model of polymerase translocation. Curr. Pharm. Des. 2006, 12, 1867–1877. [Google Scholar] [CrossRef] [PubMed]
- Marchand, B.; Tchesnokov, E.P.; Götte, M. The pyrophosphate analogue foscarnet traps the pre-translocational state of HIV-1 reverse transcriptase in a Brownian ratchet model of polymerase translocation. J. Biol. Chem. 2007, 282, 3337–3346. [Google Scholar] [CrossRef] [PubMed]
- Rutvisuttinunt, W.; Meyer, P.R.; Scott, W.A. Interactions between HIV-1 reverse transcriptase and the downstream template strand in stable complexes with primer-template. PLoS ONE 2008, 3, e3561. [Google Scholar] [CrossRef] [PubMed]
- Tuske, S.; Sarafianos, S.G.; Clark, A.D.; Ding, J.; Naeger, L.K.; White, K.L.; Miller, M.D.; Gibbs, C.S.; Boyer, P.L.; Clark, P.; Wang, G.; Gaffney, B.L.; Jones, R.A.; Jerina, D.M.; Hughes, S.H.; Arnold, E. Structures of HIV-1 RT-DNA complexes before and after incorporation of the anti-AIDS drug tenofovir. Nat. Struct. Mol. Biol. 2004, 11, 469–474. [Google Scholar] [CrossRef] [PubMed]
- St. Clair, M.H.; Martin, J.L.; Tudor-Williams, G.; Bach, M.C.; Vavro, C.L.; King, D.M.; Kellam, P.; Kemp, S.D.; Larder, B.A. Resistance to ddI and sensitivity to AZT induced by a mutation in HIV-1 reverse transcriptase. Science 1991, 253, 1557–1559. [Google Scholar] [PubMed]
- Miranda, L.R.; Götte, M.; Liang, F.; Kuritzkes, D.R. The L74V mutation in human immunodeficiency virus type 1 reverse transcriptase counteracts enhanced excision of zidovudine monophosphate associated with thymidine analog resistance mutations. Antimicrob. Agents Chemother. 2005, 49, 2648–2656. [Google Scholar] [CrossRef] [PubMed]
- Frankel, F.A.; Marchand, B.; Turner, D.; Götte, M.; Wainberg, M.A. Impaired rescue of chain-terminated DNA synthesis associated with the L74V mutation in human immunodeficiency virus type 1 reverse transcriptase. Antimicrob. Agents Chemother. 2005, 49, 2657–2664. [Google Scholar] [CrossRef] [PubMed]
- Larder, B.A. 3'-Azido-3'-deoxythymidine resistance suppressed by a mutation conferring human immunodeficiency virus type 1 resistance to nonnucleoside reverse transcriptase inhibitors. Antimicrob. Agents Chemother. 1992, 36, 2664–2669. [Google Scholar] [PubMed]
- Byrnes, V.W.; Emini, E.A.; Schleif, W.A.; Condra, J.H.; Schneider, C.L.; Long, W.L.; Wolfgang, J.A.; Graham, D.J.; Gotlib, L.; Schlabach, A.J.; Wolanski, B.S.; Blahy, O.M.; Quintero, J.C.; Rhodes, A.; Roth, E.; Titus, D.L.; Sardana, V.V. Susceptibilities of human immunodeficiency virus type 1 enzyme and viral variants expressing multiple resistance-engendering amino acid substitutions to reverse transcriptase inhibitors. Antimicrob. Agents Chemother. 1994, 38, 1404–1407. [Google Scholar] [PubMed]
- Matamoros, T.; Nevot, M.; Martínez, M.A.; Menéndez-Arias, L. Thymidine analogue resistance suppression by V75I of HIV-1 reverse transcriptase: Effects of substituting valine 75 on stavudine excision and discrimination. J. Biol. Chem. 2009, 284, 32792–32802. [Google Scholar] [CrossRef] [PubMed]
- Schinazi, R.F.; Lloyd, R.M.; Nguyen, M.H.; Cannon, D.L.; McMillan, A.; Ilksoy, N.; Chu, C.K.; Liotta, D.C.; Bazmi, H.Z.; Mellors, J.W. Characterization of human immunodeficiency viruses resistant to oxathiolane-cytosine nucleosides. Antimicrob. Agents Chemother. 1993, 37, 875–881. [Google Scholar] [PubMed]
- Gao, Q.; Gu, Z; Parniak, M.A.; Cameron, J.; Cammack, N.; Boucher, C.; Wainberg, M.A. The same mutation that encodes low-level human immunodeficiency virus type 1 resistance to 2',3'-dideoxyinosine and 2',3'-dideoxycytidine confers high-level resistance to the (-) enantiomer of 2',3'-dideoxy-3'-thiacytidine. Antimicrob. Agents Chemother. 1993, 37, 1390–1392. [Google Scholar] [PubMed]
- Boucher, C.A.B.; Cammack, N.; Schipper, P.; Schuurman, R.; Rouse, P.; Wainberg, M.A.; Cameron, J.M. High-level resistance to (-) enantiomeric 2'-deoxy-3'-thiacytidine in vitro is due to one amino acid substitution in the catalytic site of human immunodeficiency virus type 1 reverse transcriptase. Antimicrob. Agents Chemother. 1993, 37, 2231–2234. [Google Scholar] [PubMed]
- Tisdale, M.; Kemp, S.D.; Parry, N.R.; Larder, B.A. Rapid in vitro selection of human immunodeficiency virus type 1 resistant to 3'-thiacytidine inhibitors due to a mutation in the YMDD region of reverse transcriptase. Proc. Natl. Acad. Sci. USA 1993, 90, 5653–5656. [Google Scholar] [CrossRef]
- Larder, B.A.; Kemp, S.D.; Harrigan, P.R. Potential mechanism for sustained antiretroviral efficacy of AZT-3TC combination therapy. Science 1995, 269, 696–699. [Google Scholar] [PubMed]
- Sarafianos, S.G.; Das, K.; Clark, A.D.; Ding, J.; Boyer, P.L.; Hughes, S.H.; Arnold, E. Lamivudine (3TC) resistance in HIV-1 reverse transcriptase involves steric hindrance with β-branched amino acids. Proc. Natl. Acad. Sci. USA 1999, 96, 10027–10032. [Google Scholar] [CrossRef]
- Gao, H.Q.; Boyer, P.L. The role of steric hindrance in 3TC resistance of human immunodeficiency virus type-1 reverse transcriptase. J. Mol. Biol. 2000, 300, 403–418. [Google Scholar] [CrossRef] [PubMed]
- Diallo, K.; Götte, M.; Wainberg, M.A. Molecular impact of the M184V mutation in human immunodeficiency virus type 1 reverse transcriptase. Antimicrob. Agents Chemother. 2003, 47, 3377–3383. [Google Scholar] [CrossRef] [PubMed]
- Staszewski, S.; Hill, A.M.; Bartlett, J.; Eron, J.J.; Katlama, C.; Johnson, J.; Sawyer, W.; McDade, H. Reductions in HIV-1 disease progression for zidovudine/lamivudine relative to control treatments: A meta-analysis of controlled trials. AIDS 1997, 11, 477–483. [Google Scholar] [CrossRef] [PubMed]
- Petrella, M.; Wainberg, M.A. Might the M184V substitution in HIV-1 RT confer clinical benefit? AIDS Rev. 2002, 4, 224–232. [Google Scholar] [PubMed]
- Foudraine, N.A.; de Jong, J.J.; Weverling, G.J.; van Benthem, B.H.B.; Maas, J.; Keet, I.P.M.; Jurriaans, S.; Roos, M.T.L.; Vandermeulen, K.; de Wolf, F.; Lange, J.M.A. An open randomized controlled trial of zidovudine plus lamivudine versus stavudine plus lamivudine. AIDS 1998, 12, 1513–1519. [Google Scholar] [CrossRef] [PubMed]
- Harrigan, P.R.; Miller, M.D.; McKenna, P.; Brumme, Z.L.; Larder, B.A. Phenotypic susceptibilities to tenofovir in a large panel of clinically derived human immunodeficiency virus type I isolates. Antimicrob. Agents Chemother. 2002, 46, 1067–1072. [Google Scholar] [CrossRef] [PubMed]
- Wolf, K.; Walter, H.; Beerenwinkel, N.; Keulen, W.; Kaiser, R.; Hoffmann, D.; Lengauer, T.; Selbig, J.; Vandamme, A.M.; Korn, K.; Schmidt, B. Tenofovir resistance and resensitization. Antimicrob. Agents Chemother. 2003, 47, 3478–3484. [Google Scholar] [CrossRef] [PubMed]
- Götte, M.; Arion, D.; Parniak, M.A.; Wainberg, M.A. The M184V mutation in the reverse transcriptase of human immunodeficiency virus type 1 impairs rescue of chain-terminated DNA synthesis. J. Virol. 2000, 74, 3579–3585. [Google Scholar] [CrossRef] [PubMed]
- Naeger, L.K.; Margot, N.A.; Miller, M.D. Increased drug susceptibility of HIV-1 reverse transcriptase mutants containing M184V and zidovudine-associated mutations: Analysis of enzyme processivity, chain-terminator removal and viral replication. Antivir. Ther. 2001, 6, 115–126. [Google Scholar] [PubMed]
- Lennerstrand, J.; Hertogs, K.; Stammers, D.K.; Larder, B.A. Correlation between viral resistance to zidovudine and resistance at the reverse transcriptase level for a panel of human immunodeficiency virus type 1 mutants. J. Virol. 2001, 75, 7202–7205. [Google Scholar] [CrossRef] [PubMed]
- Ray, A.S.; Murakami, E.; Peterson, C.N.; Shi, J.; Schinazi, R.F.; Anderson, K.S. Interactions of enantiomers of 2',3'-didehydro-2',3'-dideoxy-fluorocytidine with wild type and M184V mutant HIV-1 reverse transcriptase. Antiviral Res. 2002, 56, 189–205. [Google Scholar] [CrossRef] [PubMed]
- Boyer, P.L.; Sarafianos, S.G.; Arnold, E.; Hughes, S.H. The M184V mutation reduces the selective excision of zidovudine 5'-monophosphate (AZTMP) by the reverse transcriptase of human immunodeficiency virus type 1. J. Virol. 2002, 76, 3248–3256. [Google Scholar] [CrossRef] [PubMed]
- Frankel, F.A.; Invernizzi, C.F.; Oliveira, M.; Wainberg, M.A. Diminished efficiency of HIV-1 reverse transcriptase containing K65R and M184V drug resistance mutations. AIDS 2007, 21, 665–675. [Google Scholar] [CrossRef] [PubMed]
- Tisdale, M.; Alnadaf, T.; Cousens, D. Combination of mutations in human immunodeficiency virus type 1 reverse transcriptase required for resistance to the carbocyclic nucleoside 1592U89. Antimicrob. Agents Chemother. 1997, 41, 1094–1098. [Google Scholar] [PubMed]
- Harrigan, P.R.; Stone, C.; Griffin, P.; Nájera, I.; Bloor, S.; Kemp, S.; Tisdale, M.; Larder, B.; the CNA2001 Investigative Group. Resistance profile of the human immunodeficiency virus type 1 reverse transcriptase inhibitor abacavir (1592U89) after monotherapy and combination therapy. J. Infect. Dis. 2000, 181, 912–920. [Google Scholar] [CrossRef] [PubMed]
- Wainberg, M.A.; Miller, M.D.; Quan, Y.; Salomon, H.; Mulato, A.S.; Lamy, P.D.; Margot, N.A.; Anton, K.E.; Cherrington, J.M. In vitro selection and characterization of HIV-1 with reduced susceptibility to PMPA. Antivir. Ther. 1999, 4, 87–94. [Google Scholar] [PubMed]
- García-Lerma, J.G.; MacInnes, H.; Bennett, D.; Reid, P.; Nidtha, S.; Weinstock, H.; Kaplan, J.E.; Heneine, W. A novel genetic pathway of human immunodeficiency virus type 1 resistance to stavudine mediated by the K65R mutation. J. Virol. 2003, 77, 5685–5693. [Google Scholar] [CrossRef] [PubMed]
- Winters, M.A.; Shafer, R.W.; Jellinger, R.A.; Mamtora, G.; Gingeras, T.; Merigan, T.C. Human immunodeficiency virus type 1 reverse transcriptase genotype and drug susceptibility changes in infected individuals receiving dideoxyinosine monotherapy for 1 to 2 years. Antimicrob. Agents Chemother. 1997, 41, 757–762. [Google Scholar] [PubMed]
- Deval, J.; White, K.L.; Miller, M.D.; Parkin, N.T.; Courcambeck, J.; Halfon, P.; Selmi, B.; Boretto, J.; Canard, B. Mechanistic basis for reduced viral and enzymatic fitness of HIV-1 reverse transcriptase containing both K65R and M184V mutations. J. Biol. Chem. 2004, 279, 509–516. [Google Scholar] [CrossRef] [PubMed]
- Sluis-Cremer, N.; Sheen, C.W.; Zelina, S.; Torres, P.S.A.; Parikh, U.M.; Mellors, J.W. Molecular mechanism by which the K70E mutation in human immunodeficiency virus type 1 reverse transcriptase confers resistance to nucleoside reverse transcriptase inhibitors. Antimicrob. Agents Chemother. 2007, 51, 48–53. [Google Scholar] [CrossRef] [PubMed]
- Svarovskaia, E.S.; Feng, J.Y.; Margot, N.A.; Myrick, F.; Goodman, D.; Ly, J.K.; White, K.L.; Kutty, N.; Wang, R.; Borroto-Esoda, K.; Miller, M.D. The A62V and S68G mutations in HIV-1 reverse transcriptase partially restore the replication defect associated with the K65R mutation. J. Aquir. Immune Defic. Syndr. 2008, 48, 428–436. [Google Scholar] [CrossRef]
- Rezende, L.F.; Prasad, V.R. Nucleoside-analog resistance mutations in HIV-1 reverse transcriptase and their influence on polymerase fidelity and viral mutation rates. Int. J. Biochem. Cell Biol. 2004, 36, 1716–1734. [Google Scholar] [CrossRef] [PubMed]
- Menéndez-Arias, L. Mutation rates and intrinsic fidelity of retroviral reverse transcriptases. Viruses 2009, 1, 1137–1165. [Google Scholar] [CrossRef]
- Nikolenko, G.N.; Svarovskaia, E.S.; Delviks, K.A.; Pathak, V.K. Antiretroviral drug resistance mutations in human immunodeficiency virus type 1 reverse transcriptase increase template-switching frequency. J. Virol. 2004, 78, 8761–8770. [Google Scholar] [CrossRef] [PubMed]
- Nikolenko, G.N.; Palmer, S.; Maldarelli, F.; Mellors, J.W.; Coffin, J.M.; Pathak, V.K. Mechanism for nucleoside analog-mediated abrogation of HIV-1 replication: Balance between RNase H activity and nucleotide excision. Proc. Natl. Acad. Sci. USA 2005, 102, 2093–2098. [Google Scholar] [CrossRef]
- Götte, M. Should we include connection domain mutations of HIV-1 reverse transcriptase in HIV resistance testing? PLoS Med. 2007, 4, e346. [Google Scholar] [CrossRef] [PubMed]
- Roquebert, B.; Marcelin, A.G. The involvement of HIV-1 RNAse H in resistance to nucleoside analogues. J. Antimicrob. Chemother. 2008, 61, 973–975. [Google Scholar] [CrossRef] [PubMed]
- Ehteshami, M.; Götte, M. Effects of mutations in the connection and RNase H domains of HIV-1 reverse transcriptase on drug susceptibility. AIDS Rev. 2008, 10, 224–235. [Google Scholar] [PubMed]
- Yap, S.H.; Sheen, C.W.; Fahey, J.; Zanin, M.; Tyssen, D.; Lima, V.D.; Wynhoven, B.; Kuiper, M.; Sluis-Cremer, N.; Harrigan, P.R.; Tachedjian, G. N348I in the connection domain of HIV-1 reverse transcriptase confers zidovudine and nevirapine resistance. PLoS Med. 2007, 4, e335. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Radzio, J.; Sluis-Cremer, N. Efavirenz accelerates HIV-1 reverse transcriptase ribonuclease H cleavage, leading to diminished zidovudine excision. Molec. Pharm. 2008, 73, 601–606. [Google Scholar] [CrossRef]
- Ehteshami, M.; Beilhartz, G.L.; Scarth, B.J; Tchesnokov, E.P.; McCormick, S.; Wynhoven, B.; Harrigan, P.R.; Götte, M. Connection domain mutations N348I and A360V in HIV-1 reverse transcriptase enhance resistance to 3'-azido-3'-deoxythymidine through both RNase H-dependent and -independent mechanisms. J. Biol. Chem. 2008, 283, 22222–22232. [Google Scholar] [CrossRef] [PubMed]
- Brehm, J.H.; Mellors, J.W.; Sluis-Cremer, N. Mechanism by which a glutamine to leucine substitution at residue 509 in the ribonuclease H domain of HIV-1 reverse transcriptase confers zidovudine resistance. Biochemistry 2008, 47, 14020–14027. [Google Scholar] [CrossRef] [PubMed]
- Delviks-Frankenberry, K.A.; Nikolenko, G.N.; Boyer, P.L.; Hughes, S.H.; Coffin, J.M.; Jere, A.; Pathak, V.K. HIV-1 reverse transcriptase connection subdomain mutations reduce template RNA degradation and enhance AZT excision. Proc. Natl. Acad. Sci. USA 2008, 105, 10943–10948. [Google Scholar] [CrossRef]
- Delviks-Frankenberry, K.A.; Nikolenko, G.N.; Maldarelli, F.; Hase, S.; Takebe, Y.; Pathak, V.K. Subtype-specific differences in the human immunodeficiency virus type1 reverse transcriptase connection subdomain of CRF01_AE are associated with higher levels of resistance to 3'-azido-3'-deoxythymidine. J. Virol. 2009, 83, 8502–8513. [Google Scholar] [CrossRef] [PubMed]
- Delviks-Frankenberry, K.A.; Pathak, V.K. The "connection" between HIV-1 RNase H activity and drug resistance. Viruses to be submitted for publication.
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Share and Cite
Acosta-Hoyos, A.J.; Scott, W.A. The Role of Nucleotide Excision by Reverse Transcriptase in HIV Drug Resistance. Viruses 2010, 2, 372-394. https://doi.org/10.3390/v2020372
Acosta-Hoyos AJ, Scott WA. The Role of Nucleotide Excision by Reverse Transcriptase in HIV Drug Resistance. Viruses. 2010; 2(2):372-394. https://doi.org/10.3390/v2020372
Chicago/Turabian StyleAcosta-Hoyos, Antonio J., and Walter A. Scott. 2010. "The Role of Nucleotide Excision by Reverse Transcriptase in HIV Drug Resistance" Viruses 2, no. 2: 372-394. https://doi.org/10.3390/v2020372