Human Papillomavirus Genotype Landscape Across Cervical Cytology Grades and Impact of HIV Among Women of Eastern Cape Province, South Africa
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Setting and Population
2.2. Data Collection
2.3. Laboratory Investigations
2.4. Statistical Analysis
2.5. Ethics Considerations
3. Results
3.1. Study Population Description
3.2. Prevalence of HPV Infection According to Cervical Cytology and HIV Status Among Eastern Cape Women
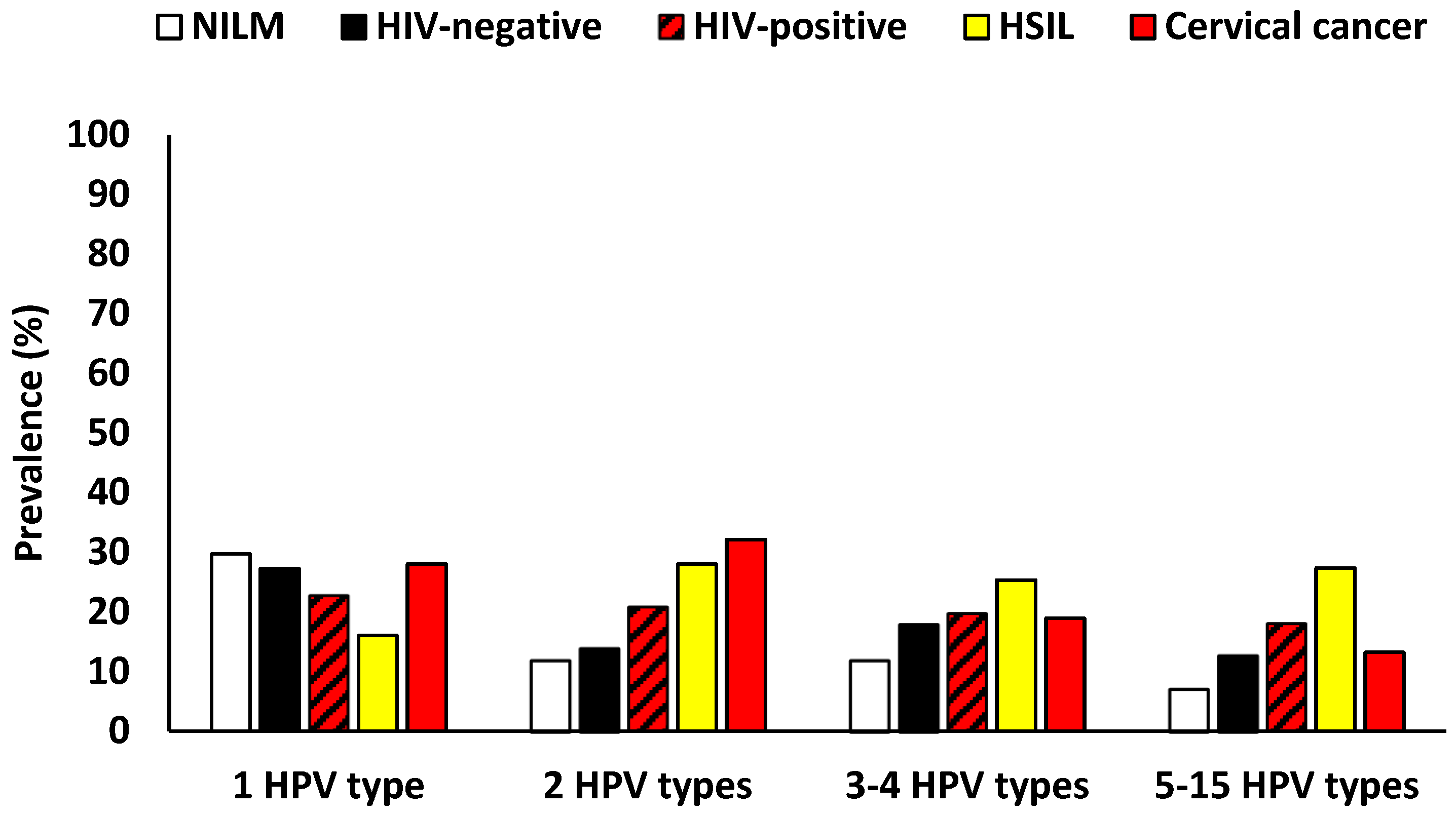
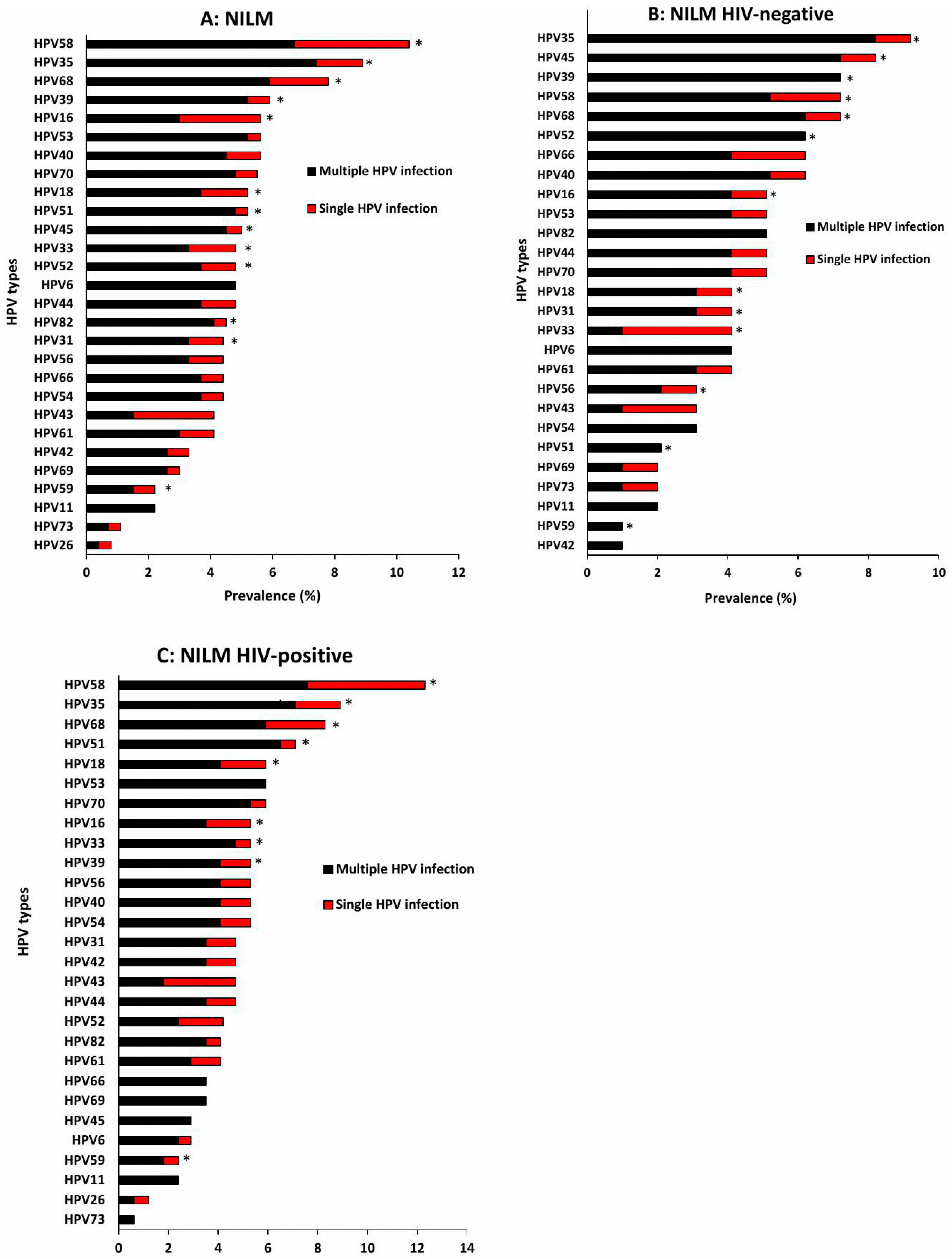
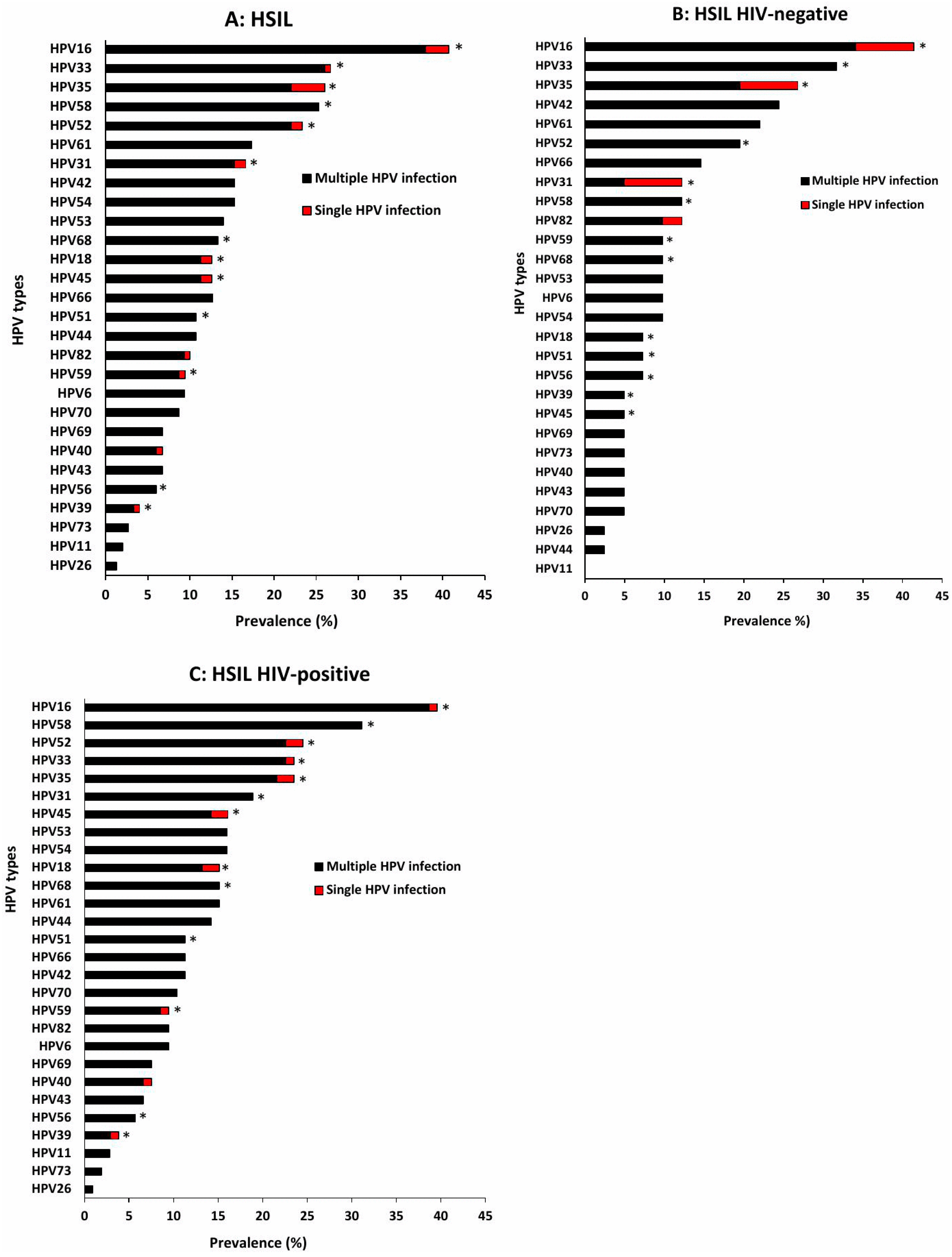
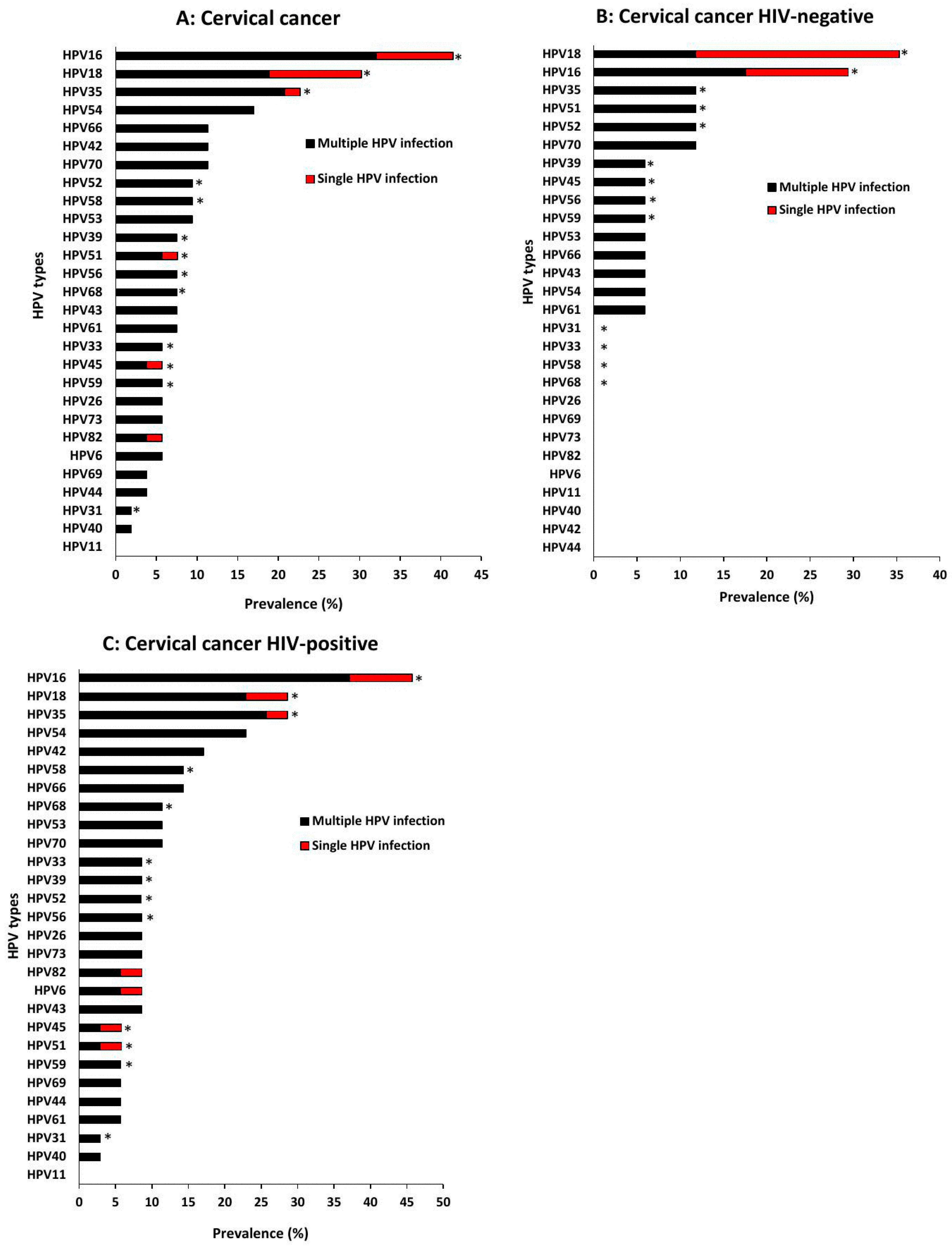
3.3. Distribution of HPV Genotypes Among Women with Different Cervical Cytology Grades in Single and Multiple Infections According to HIV Status
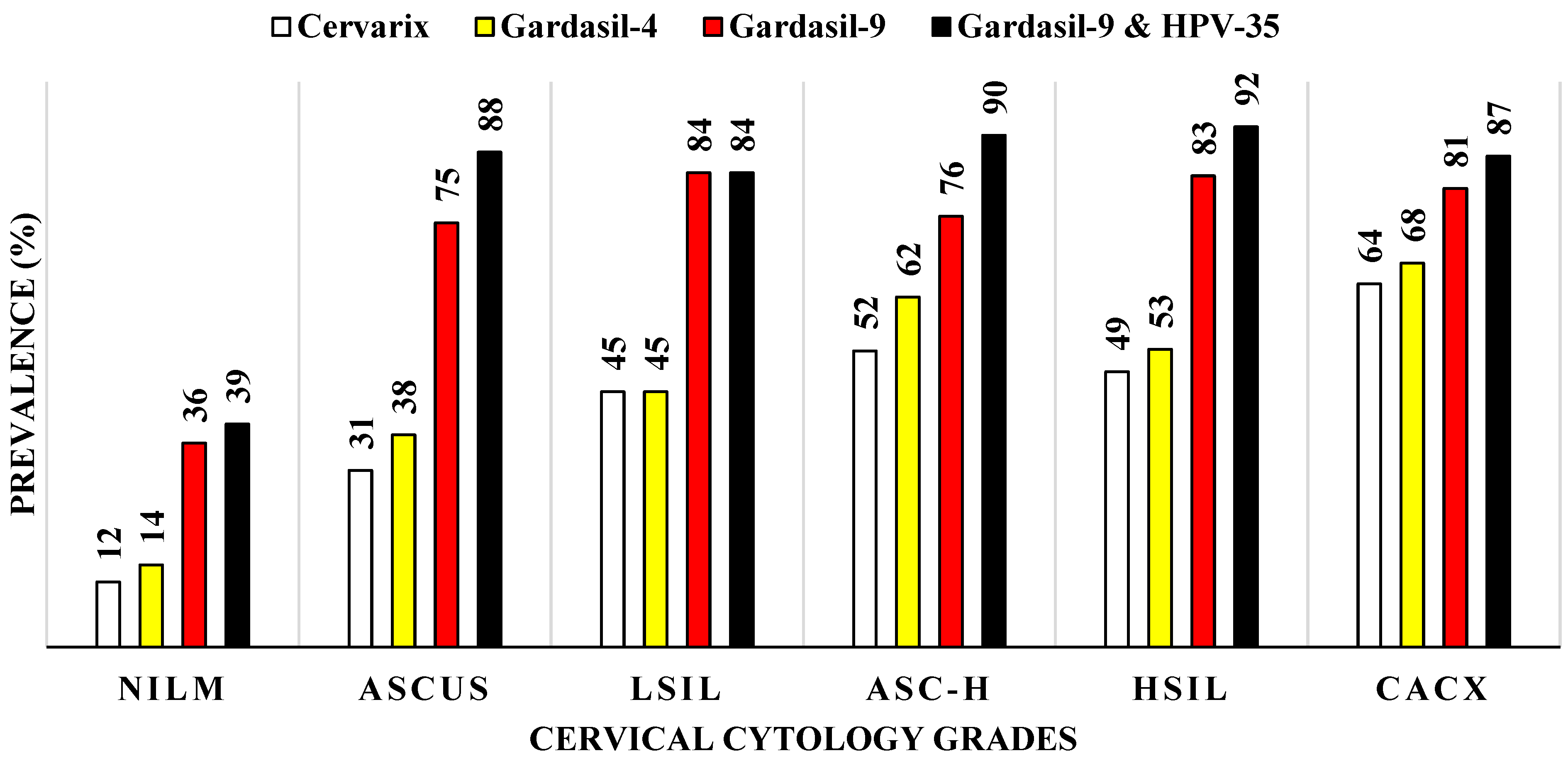
3.4. Prevalence of HPV Types Targeted by Current Commercial HPV Vaccines According to Different Cervical Cytology Grades
3.5. Factors Associated with HPV Infection Among Women with Normal Cervical Cytology in the Eastern Cape Province
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| HPV | Human papillomavirus |
| HIV | Human Immunodeficiency Virus |
| ASC-US | Atypical squamous cells of undetermined significance |
| LSILs | Low-grade intraepithelial lesions |
| ASC-Hs | Atypical squamous cells cannot exclude high-grade squamous intraepithelial lesions |
| HSILs | High-grade squamous intraepithelial lesions |
| HR-HPV | High-risk human papillomavirus |
| LR-HPV | Low-risk human papillomavirus |
| ART | Antiretroviral therapy |
| HREC | Health Research Ethics Committee |
| KSD | King Sabata Dalindyebo Municipality |
| PCR | Polymerase Chain Reaction |
| IQR | Interquartile range |
| CI | Confidence Interval |
| NILM | Negative for intraepithelial lesions or malignancy |
| CACX | Cervical cancer |
| NMAH | Nelson Mandela Academic Hospital |
References
- Maswanganye, C.K.; Mkhize, P.P.; Matume, N.D. Mapping the HPV Landscape in South African Women: A Systematic Review and Meta-Analysis of Viral Genotypes, Microbiota, and Immune Signals. Viruses 2024, 16, 1893. [Google Scholar] [CrossRef]
- Bolon, J.; Samson, A.; Irwin, N.; Murray, L.; Mbodi, L.; Stacey, S.; Aikman, N.; Moonsamy, L.; Zamparini, J. An audit of adherence to cervical cancer screening guidelines in a tertiary-level HIV clinic. South. Afr. J. HIV Med. 2023, 24, 1490. [Google Scholar] [CrossRef]
- Somdyala, N.I.M.; Bradshaw, D.; Dhansay, M.; Stefan, D.C. Increasing Cervical Cancer Incidence in Rural Eastern Cape Province of South Africa from 1998 to 2012: A Population-Based Cancer Registry Study. JCO Glob. Oncol. 2020, 6, 8. [Google Scholar] [CrossRef]
- Moosa-Battey, R.M.; Masuku, P.; Mayimele, N. Human papillomavirus (HPV) as the main cause of cervical cancer and other related cancers: A review. SA Pharm. J. 2025, 92, 58–62. [Google Scholar] [CrossRef]
- Tesfaye, E.; Kumbi, B.; Mandefro, B. Prevalence of human papillomavirus infection and associated factors among women attending cervical cancer screening in setting of Addis Ababa, Ethiopia. Sci. Rep. 2024, 14, 4053. [Google Scholar] [CrossRef]
- Fitzpatric, M.B.; Hahn, Z.; Mandishora, R.S.D.; Dao, J.; Weber, J.; Haung, C. Whole-genome analysis of cervical human papillomavirus type 35 from Zimbabwean women. Sci. Rep. 2020, 10, 7001. [Google Scholar] [CrossRef]
- Okunade, K.S. Human papillomavirus and cervical cancer. J. Obs. Gynaecol. 2020, 40, 602–608. [Google Scholar] [CrossRef] [PubMed]
- Skelin, J.; Tomaic, V. Comparative Analysis of Alpha and Beta HPV E6 Oncoproteins: Insights into Functional Distinctions and Divergent Mechanisms of Pathogenesis. Viruses 2023, 15, 2253. [Google Scholar] [CrossRef] [PubMed]
- Ozaydin-Yavuz, G.; Bilgili, S.; Guducuoglu, H.; Yavuz, I.; Elibuyuk-Aksac, S.; Karadag, A. Determinants of high-risk human papillomavirus infection in anogenital warts. Adv. Dermatol. Allergol./Postep. Dermatol. I Alergol. 2019, 36, 76–81. [Google Scholar] [CrossRef] [PubMed]
- Mbulawa, Z.Z.A.; Phohlo, K.; Garcia-Jardon, M.; Williamson, A.L.; Businge, C.B. High-human papillomavirus (HPV)-35 prevalence among South African women with cervical intraepithelial neoplasia warrants attention. PLoS ONE 2022, 17, e0264498. [Google Scholar] [CrossRef]
- Kufa, T.; Mandiriri, A.; Shamu, T.; Dube Mandishora, R.S.; Pascoe, M.J. Prevalence of cervical high-risk human papillomavirus among Zimbabwean women living with HIV. South. Afr. J. HIV Med. 2024, 25, 1633. [Google Scholar] [CrossRef] [PubMed]
- Taku, O.; Mbulawa, Z.Z.A.; Phohlo, K.; Garcia-Jardon, M.; Businge, C.B.; Williamson, A.L. Distribution of Human Papillomavirus (HPV) Genotypes in HIV-Negative and HIV-Positive Women with Cervical Intraepithelial Lesions in the Eastern Cape Province, South Africa. Viruses 2021, 13, 280. [Google Scholar] [CrossRef]
- Clifford, G.M.; De Vuyst, H.; Tenet, V.; Plummer, M.; Tully, S.; Franceschi, S. Effect of HIV infection on Human Papillomavirus Types Causing Invasive Cervical Cancer in Africa. JAIDS J. Acquir. Immune Defic. Syndr. 2016, 73, 332–339. [Google Scholar] [CrossRef] [PubMed]
- Chachage, M.; Parikh, A.P.; Maheng, A.; Bahemana, E.; Mkai, J.; Mbuya, W.; Mcharo, R.; Maganga, L.; Mwamwaja, J.; Gervas, R.; et al. High-risk human papillomavirus genotype distribution among women living with and at risk for HIV in Africa. AIDS 2023, 37, 625–635. [Google Scholar] [CrossRef]
- Okeke, S.U. Fighting cervical cancer in Africa: Taking a closer look at human papillomavirus 35. Afr. J. Lab. Med. 2024, 13, 3. [Google Scholar] [CrossRef]
- Amponsah-Dacosta, E.; Blose, N.; Nkwinika, W.; Chepkurui, V. Human Papillomavirus Vaccination in South Africa: Programmatic challenges and Opportunities for integration With other Adolescent Health services. Front. Public Health 2022, 10, 799984. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Cervical Cancer. Available online: https://www.who.int/news-room/fact-sheets/detail/cervical-cancer (accessed on 20 November 2025).
- Delany-Moretlwe, S.; Kelly, K.F.; James, S.; Scorgie, F.; Subedar, H.; Dlamini, N.R.; Pillay, Y.; Naidoo, N.; Chikandiwa, A.; Rees, H. Human papillomavirus vaccine introduction in South Africa: Implementation lessons from an evaluation of the national school-based vaccination campaign. Glob. Health Sci. Pract. 2018, 6, 425–438. [Google Scholar] [CrossRef]
- Bruni, L.; Albero, G.; Serrano, B.; Mena, M.; Gomez, D.; Munoz, J.; Bosch, F.; de Sanjose, S. ICO/IARC Information Centre on HPV and Cancer. In Human Papillomavirus and Related Diseases in South Africa. 2023. Available online: https://hpvcentre.net/statistics/reports/ZAF.pdf (accessed on 20 November 2025).
- Botha, M.; Mabenge, M.; Makua, M.; Mbodi, M.; Rogers, L.; Sebitloane, M.; Smith, T.; Van der Merwe, F.; Williamson, A.; Whittaker, J. Cervical Cancer Screening Guidelines for South Africa. Afr. J. Obstet. Gynaecol. 2023, 1, 27–31. [Google Scholar]
- Kremer, W.W.; Van Zummeren, M.; Breytenbach, E.; Richter, K.L.; Steenbergen, R.D.; Meijer, C.J.; Dreyer, G. The use of molecular mackers for cervical screening of women living with HIV in South Africa. Aids 2019, 33, 2025–2042. [Google Scholar] [CrossRef]
- UNAIDS. South African Countries. Available online: https://www.unaids.org/en/regionscountries (accessed on 11 November 2025).
- Ntuli, S.T.; Maimela, E.; Skaal, L.; Mogale, M.; Lekota, P. Abnormal cervical cytology amongst women infected with human immunodeficiency virus in Limpopo province, South Africa. Afr. J. Prim. Health Care Fam. Med. 2020, 12, a2215. [Google Scholar] [CrossRef]
- Marembo, T.; Mandishora, R.D.; Borok, M. Use of Multiplex Polymerase Chain Reaction for Detection of High-Risk Human Papillomavirus Genotypes in Women Attending Routine Cervical Cancer Screening in Harare. Intervirology 2019, 62, 90–95. [Google Scholar] [CrossRef]
- Swase, T.D.; Fasogbon, I.V.; Eseoghene, I.J. The impact of HPV/HIV co-infection on immunosuppression, HPV genotype, and cervical cancer biomarkers. BMC Cancer 2025, 25, 1–15. [Google Scholar] [CrossRef]
- Pavone, G.; Marino, A.; Fisicaro, V.; Motta, L.; Spata, A.; Martorana, F.; Spampinato, S.; Celesia, B.M.; Cacopardo, B.; Vigneri, P. Entangled Connections: HIV and HPV Interplay in Cervical Cancer-A Comprehensive Review. Int. J. Mol. Sci. 2024, 25, 10358. [Google Scholar] [CrossRef]
- Naegele, K.; Bubendorf, L.; Hirsch, H.H.; Leuzinger, K. Comparative evaluation of Anyplex II HPV28 and Allplex HPV28 molecular assays for human papillomavirus detection and genotyping in anogenital cancer screening. J. Med. Virol. 2024, 96, e29649. [Google Scholar] [CrossRef] [PubMed]
- Mafi, S.; Theuillon, F.; Meyer, S.; Woillard, J.B.; Dupont, M.; Rogez, S.; Alain, S.; Hantz, S. Comparative evaluation of Allplex HPV28 and Anyplex II HPV28 assays for high-risk HPV genotyping in cervical samples. PLoS ONE 2025, 20, e0320978. [Google Scholar] [CrossRef]
- National Cancer Institute. Cervical Cancer Causes, Risk Factors, and Prevention. Available online: https://www.cancer.gov/types/cervical/causes-risk-prevention (accessed on 10 November 2025).
- Rikhotso, R.R.; Mitchell, E.M.; Wilson, D.T.; Doede, A.; Matume, N.D.; Bessong, P.O. Prevalence and distribution of selected cervical human papillomavirus types in HIV infected and HIV uninfected women in South Africa, 1989–2021: A narrative review. South. Afr. J. Infect. Dis. 2022, 37, 10. [Google Scholar] [CrossRef] [PubMed]
- Annunziata, C.; Stellato, G.; Greggi, S.; Sanna, V.; Curcio, M.P.; Losito, S.; Botti, G.; Buonaguro, L.; Tornesello, M.L. Prevalence of “unclassified” HPV genotypes among women with abnormal cytology. Infect. Agents Cancer 2018, 13, 26. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.-Q.; Hao, J.-Q.; Mendez, M.J.; Bangura Mohamed, S.; Fu, S.-L.; Zhao, F.; Qiao, Y.-L. The prevalence of cervical HPV infection and genotype distribution in 856,535 chinese women with normal and abnormal cervical lesions: A systematic review. J. Cytol. 2022, 39, 137–147. [Google Scholar] [CrossRef]
- Omoyeni, O.M.; Tsoka-Gwegweni, J.M. Prevalence of cervical abnormalities among rural women in KwaZulu-Natal, South Africa. Pan Afr. Med. J. 2022, 41, 110. [Google Scholar] [CrossRef]
- Tabibzadeh, A.; Panahi, M.; Bouzari, B.; Ashtiana, M.T.H.; Zamani, F.; Arzati, H.T. Niya MHKJIJoM: Distribution of human papillomavirus in suspected women cytological specimens from Tehran, Iran. Iran. J. Microbiol. 2022, 14, 112–118. [Google Scholar] [CrossRef]
- Mojarrad, S.; Najmafshar, M.; Jahromi, Z.K.; Salahi Ardekani, O.; Shahraki, H.R.; Jalvand, M.; Sana, F.A. Prevalence and genotype distribution of human papillomavirus in individuals referred to a laboratory in urmia, Iran. Infect. Agents Cancer 2025, 20, 1–9. [Google Scholar] [CrossRef]
- Aslanimehr, M.; Nemati, S.; Sadeghi, H.; Samiee-Rad, F.; Ghafari, S.; Naserpour-Farivar, T. High-risk HPV genotypes in women with abnormal cytology: A 12-year retrospective study. Infect. Agents Cancer 2025, 20, 1–7. [Google Scholar] [CrossRef]
- Damgaard, R.K.; Jenkins, D.; Stoler, M.H.; Lycke, K.D.; Quint, W.G.; Steiniche, T.; Peterson, L.K.; Hammer, A. High prevalence of HPV16 and high-grade cytology in women undergoing active surveillance for cervical intraephithelial neoplasia grade 2. Acta Obstet. Et Gynecol. Scand. 2023, 102, 1227–1235. [Google Scholar] [CrossRef]
- Adebamowo, S.N.; Oluwatoyisi, O.; Ayotunde, F.; Dareng, E.O.; Richard, O.; Adebamowo, C.A. Persistent Low-Risk and High-Risk Human Papillomavirus infections of the Uterine Cervix in HIV-negative and HIV-positive Women. Front. Public Health 2017, 5, 178. [Google Scholar] [CrossRef] [PubMed]
- Ogembo, R.K.; Gona, P.N.; Seymour, A.J.; Park, H.S.M.; Bain, P.A.; Maranda, L.; Ogembo, J.G. Prevalence of human papillomavirus genotypes among African women with normal cervical cytology and neoplasia: A sytematic review and meta-analysis. PLoS ONE 2015, 10, e0122488. [Google Scholar] [CrossRef] [PubMed]
- Seyoum, A.; Assefa, N.; Gure, T.; Seyoum, B.; Mulu, A.; Mihret, A. Prevalence and genotype distribution of high-risk human papillomavirus infection among Sub-Saharan African women: A systematic review and meta-analysis. Front. Public Health 2022, 10, 890880. [Google Scholar] [CrossRef]
- Wei, F.; Georges, D.; Man, I.; Baussano, I.; Clifford, G.M. Casual attribution of human papillomavirus genotypes to ivasive cervical cancer worldwide: A systematic analysis of the global literature. Lancet 2024, 404, 435–444. [Google Scholar] [CrossRef] [PubMed]
- Mrema, A.; Ngoma, M.; Lugina, E.L.; Kahakwa, A.; Mwita, C.J.; Iddy, S. Genotype distribution of human papillomavirus among women with cervical cancer stratified by HIV status in Tanzania. PLoS ONE 2025, 25, e0322311. [Google Scholar] [CrossRef]
- Uuskula, A.; Tisler, A.; DeHovitz, J.; Murenzi, G.; Castle, P.E.; Clifford, G. Prevention and control of HPV-related cancers in people living with HIV. Lancet HIV 2025, 12, 293–302. [Google Scholar] [CrossRef]
- Kuguyo, O.; Matimba, A.; Tsikai, N.; Madziyire, M.; Magwali, T.; Dandara, C. A association between human papillomavirus and behaviour, clinocopathology, and cervical cancer outcome in Zimbabwean women: A cross-sectional study. Reprod. Health 2025, 22, 1–9. [Google Scholar] [CrossRef]
- Mbulawa, Z.Z.A.; Somdyala, N.I.; Mabunda, S.A.; Williamson, A.L. High human papillomavirus prevalence among females attending high school in the Eastern Cape Province of South Africa. PLoS ONE 2021, 16, e0253074. [Google Scholar] [CrossRef]
- Zhou, Z.W.; Long, H.Z.; Cheng, Y.; Lou, H.Y.; Wen, D.D.; Gao, L.C. From Microbiome to Inflamation: The Key Drivers of Cervical Cancer. Front. Microbiol. 2021, 12, 767931. [Google Scholar] [CrossRef]
- Sun, L.; Li, L.; Xu, W.; Ma, C. The Role Immunomodulation Of Vaginal Microenvironment On Human Papillomavirus Infection. Galen. Med. J. 2023, 12, e2991. [Google Scholar] [CrossRef]
- Tekalegn, K.B.; Aseffa, T.; Gezahegn, H.; Kene, C. High parity is associated with increased risk of cervical cancer: Systematic review and meta-analysis of case-control studies. Women’s Health 2022, 18. [Google Scholar] [CrossRef]
- Bowden, S.J.; Doulgeraki, T.; Bouras, E. Risk factors for human papillomavirus infection, cervical intraepithelial lesions and cervical cancer: An umbrella review and follow-up Mendelian randomisation studies. BMC Med. 2023, 21, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Simelane, M.S.; Chemhaka, G.B.; Shabalala, F.S.; Simelane, P.T.; Vilakati, Z. Prevalence and determinants of inconsistent condom use among unmarried sexually active youth. A secondary analysis of the 2016–2017 Eswatini HIV incidence measurement survey. Afr. Health Sci. 2023, 23, 400–409. [Google Scholar] [CrossRef] [PubMed]
- Rositch, A.F.; Patel, E.U.; Petersen, M.R.; Quinn, T.C.; Gravitt, P.E.; Tobian, A.A.R. Importance of Lifetime Sexual History on the Prevalence of Genital Human Papillomavirus (HPV) Among Unvaccinated Adults in the National Health and Nutrition Examination Surveys: Implications for Adult HPV Vaccination. Clin. Infect. Dis. 2021, 72, 272–279. [Google Scholar] [CrossRef] [PubMed]
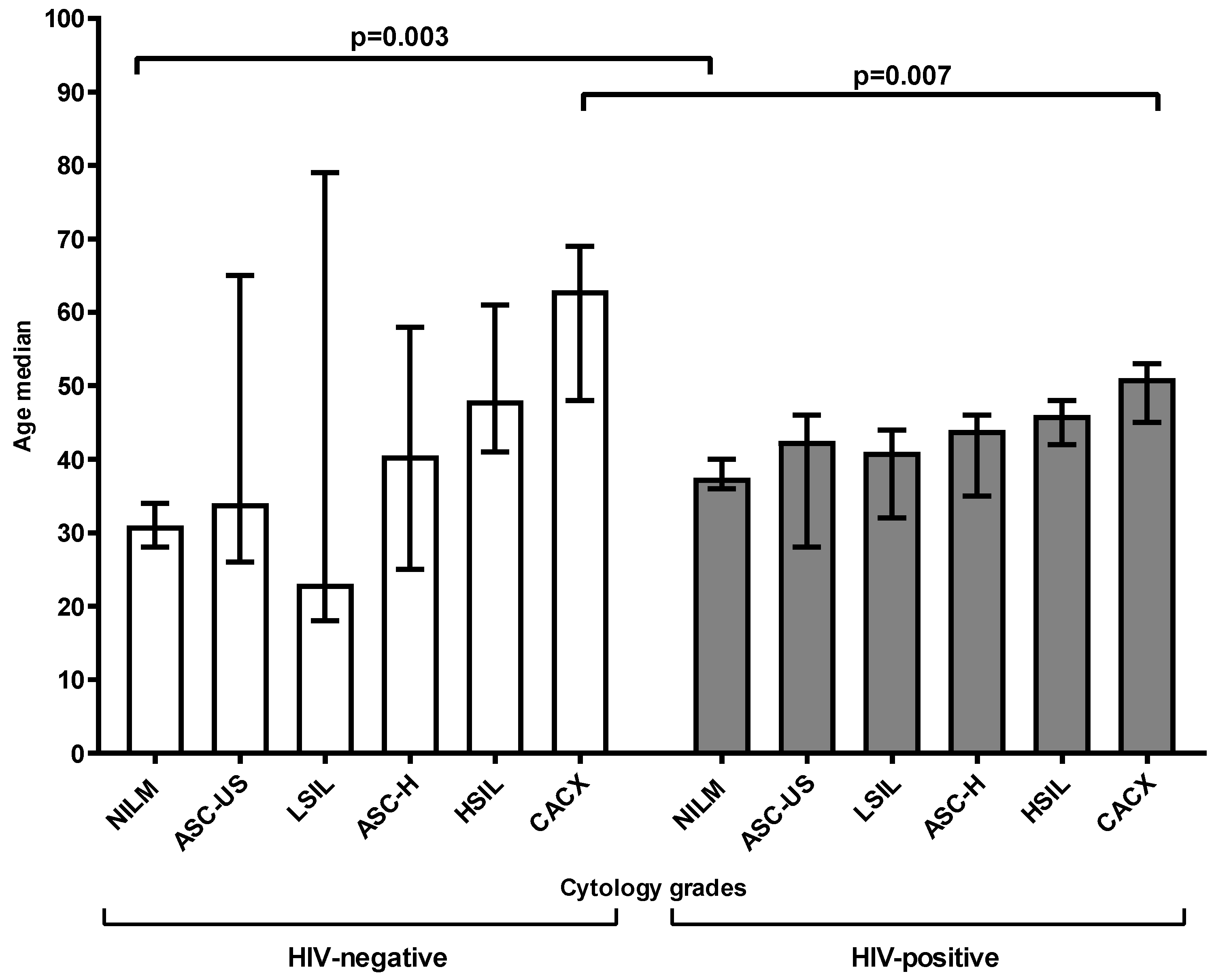
| Characteristics | NILM | ASC-US | LSIL | ASC-H | HSIL | Cervical Cancer | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| n/N | % | n/N | % | n/N | % | n/N | % | n/N | % | n/N | % | |
| Age ∑ | ||||||||||||
| 18–29 years | 74/268 | 27.6 | 3/16 | 18.8 | 10/31 | 32.3 | 2/21 | 9.5 | 4/150 | 2.7 | 1/53 | 1.9 |
| 30–39 years | 84/268 | 31.3 | 4/16 | 25.0 | 7/31 | 22.6 | 6/21 | 28.6 | 37/150 | 24.7 | 7/53 | 13.2 |
| 40–49 years | 68/268 | 25.4 | 7/16 | 43.8 | 10/31 | 32.3 | 10/21 | 47.6 | 55/150 | 36.7 | 13/53 | 24.5 |
| 50–98 years | 42/268 | 15.7 | 2/16 | 12.5 | 4/31 | 12.9 | 3/21 | 14.3 | 54/150 | 36.0 | 32/53 | 60.4 |
| Marital status ∑ | ||||||||||||
| Single | 195/267 | 73.0 | 13/16 | 81.3 | 26/31 | 83.9 | 10/21 | 47.6 | 81/148 | 54.7 | 26/52 | 50.0 |
| Married | 53/267 | 19.9 | 1/16 | 6.3 | 5/31 | 16.1 | 9/21 | 42.9 | 46/148 | 31.1 | 20/52 | 38.5 |
| Widowed & Separated | 19/267 | 7.1 | 2/16 | 12.5 | 0/31 | 0.0 | 2/21 | 9.5 | 21/148 | 14.2 | 6/52 | 11.5 |
| Education ∑ | ||||||||||||
| None | 0/267 | 0.0 | 1/16 | 6.3 | 1/31 | 3.2 | 1/21 | 4.8 | 9/149 | 6.0 | 4/52 | 7.7 |
| Primary | 17/267 | 6.4 | 2/16 | 12.5 | 1/31 | 3.2 | 3/21 | 14.3 | 36/149 | 24.2 | 17/52 | 32.7 |
| Secondary | 141/267 | 52.8 | 7/16 | 43.8 | 20/31 | 64.5 | 10/21 | 47.6 | 61/149 | 40.9 | 23/52 | 44.2 |
| Tertiary | 109/267 | 40.8 | 6/16 | 37.5 | 9/31 | 29.0 | 7/21 | 33.3 | 43/149 | 28.9 | 8/52 | 15.4 |
| Currently working ∑ | ||||||||||||
| Yes | 104/267 | 39.0 | 2/16 | 12.5 | 8/31 | 25.8 | 2/21 | 9.5 | 30/149 | 20.1 | 3/52 | 5.8 |
| No | 163/267 | 61.0 | 14/16 | 87.5 | 23/31 | 74.2 | 19/21 | 90.5 | 119/149 | 79.9 | 49/52 | 94.2 |
| Currently Smoking ∑ | ||||||||||||
| Yes | 30/267 | 11.2 | 4/16 | 25.0 | 9/31 | 29.0 | 7/21 | 33.3 | 20/148 | 13.5 | 7/52 | 13.5 |
| No | 210/267 | 78.7 | 8/16 | 50.0 | 8/31 | 25.8 | 5/21 | 23.8 | 116/148 | 78.4 | 42/52 | 80.8 |
| Ex-smoker | 27/267 | 10.1 | 4/16 | 25.0 | 14/31 | 45.2 | 9/21 | 42.9 | 12/148 | 8.1 | 3/52 | 5.8 |
| Alcohol use ∑ | ||||||||||||
| Yes | 41/267 | 15.4 | 0/16 | 0.0 | 7/31 | 22.6 | 4/21 | 19.0 | 9/149 | 6.0 | 3/51 | 5.9 |
| No | 111/267 | 41.6 | 8/16 | 50.0 | 11/31 | 35.5 | 12/21 | 57.1 | 104/149 | 69.8 | 40/51 | 78.4 |
| Sometimes | 70/267 | 26.2 | 7/16 | 43.8 | 11/31 | 35.5 | 2/21 | 9.5 | 17/149 | 11.4 | 3/51 | 5.9 |
| Past | 45/267 | 16.9 | 1/16 | 6.3 | 2/31 | 6.5 | 3/21 | 14.3 | 19/149 | 12.8 | 5/51 | 9.8 |
| Sexual debut age ∑ | ||||||||||||
| ≤16 years | 57/267 | 21.3 | 4/16 | 25.0 | 12/31 | 38.7 | 7/21 | 33.3 | 55/146 | 37.7 | 13/52 | 25.0 |
| 17–18 years | 105/267 | 39.3 | 4/16 | 25.0 | 7/31 | 22.6 | 5/21 | 23.8 | 46/146 | 31.5 | 18/52 | 34.6 |
| 19–20 years | 56/267 | 21.0 | 2/16 | 12.5 | 5/31 | 16.1 | 4/21 | 19.0 | 27/146 | 18.5 | 12/52 | 23.1 |
| ≥21 years | 49/267 | 18.4 | 6/16 | 37.5 | 7/31 | 22.6 | 5/21 | 23.8 | 18/146 | 12.3 | 9/52 | 17.3 |
| Once Pregnant ∑ | ||||||||||||
| Yes | 226/266 | 85.0 | 15/16 | 93.8 | 28/31 | 90.3 | 21/21 | 100.0 | 145/148 | 98.0 | 52/52 | 100.0 |
| No | 40/266 | 15.0 | 1/16 | 6.3 | 3/31 | 9.7 | 0/21 | 0.0 | 3/148 | 2.0 | 0/52 | 0.0 |
| HIV status ∑ | ||||||||||||
| Negative | 97/267 | 36.3 | 6/16 | 37.5 | 8/31 | 25.8 | 4/21 | 19.0 | 41/147 | 27.9 | 17/52 | 32.7 |
| Positive | 170/267 | 63.7 | 10/16 | 62.5 | 23/31 | 74.2 | 17/21 | 81.0 | 106/147 | 72.1 | 35/52 | 67.3 |
| Number of lifetime sexual partners ∑ | ||||||||||||
| 1 | 17/267 | 6.4 | 1/16 | 6.3 | 2/31 | 6.5 | 2/21 | 9.5 | 20/146 | 13.7 | 5/52 | 9.6 |
| 2 | 39/267 | 14.0 | 4/16 | 25.0 | 7/31 | 22.6 | 4/21 | 19.0 | 29/146 | 19.9 | 14/52 | 26.9 |
| 3–4 | 117/267 | 43.8 | 7/16 | 43.8 | 12/31 | 38.7 | 6/21 | 28.6 | 52/146 | 35.6 | 23/52 | 44.2 |
| ≥5 | 94/267 | 35.2 | 4/16 | 25.0 | 10/31 | 32.3 | 9/21 | 42.9 | 45/146 | 30.8 | 10/52 | 19.2 |
| Variables | Normal | ASC-US | LSIL | ASC-H | HSIL | CaCx |
|---|---|---|---|---|---|---|
| % (n/269) | % (n/16) | % (n/31) | % (n/21) | % (n/150) | % (n/53) | |
| Any type | 60.6 (163) | 93.8 (15) | 100.0 (31) | 95.2 (20) | 93.7 (145) | 92.5 (49) |
| Single infection | 29.7 (80) | 12.5 (2) | 22.6 (7) | 19.0 (4) | 14.7 (22) | 28.3 (15) |
| Multiple infection | 30.9 (83) | 81.3 (13) | 77.4 (24) | 76.2 (16) | 80.7 (121) | 64.2 (34) |
| HR-HPV | 47.2 (127) | 87.5 (14) | 100.0 (31) | 95.2 (20) | 95.3 (143) | 90.6 (48) |
| Probable HR-HPV | 14.5 (39) | 25.5 (4) | 41.7 (13) | 23.8 (5) | 34.0 (51) | 28.3 (15) |
| LR-HPV | 27.9 (75) | 37.5 (6) | 54.8 (17) | 52.4 (11) | 52.0 (78) | 35.8 (19) |
| Variable | HPV Prevalence %, n/N | RR (95% CI) | p-Value | |
|---|---|---|---|---|
| Age | ||||
| 18–29 years | 77.0%, 57/74 | Reference | ||
| 30–39 years | 64.3%, 54/84 | 0.83 (0.68–1.02) | 0.085 | |
| 40–49 years | 47.1%, 32/68 | 0.61 (0.45–0.80) | <0.0001 | |
| 50–60 years | 45.2%, 19/42 | 0.59 (0.40–0.81) | 0.0001 | |
| Sexual debut age | ||||
| ≤16 years | 61.4%, 35/57 | Reference | ||
| 17 years | 66.7%, 30/45 | 1.09 (0.80–1.46) | 0.680 | |
| 18 years | 56.7%, 34/60 | 0.92 (0.68–1.25) | 0.707 | |
| 19–20 years | 50.0%, 28/56 | 0.81 (0.58–1.13) | 0.258 | |
| ≥21 years | 69.4%, 34/49 | 1.13 (0.85–1.50) | 0.420 | |
| HIV status | ||||
| Negative | 56.7%, 55/97 | Reference | ||
| Positive | 62.4%, 106/170 | 1.10 (0.90–1.37) | 0.367 | |
| Education | ||||
| Primary | 70.6%, 12/17 | Reference | ||
| Secondary | 55.3%, 78/141 | 0.78 (0.60–1.21) | 0.303 | |
| Tertiary | 65.1%, 71/109 | 0.92 (0.71–1.42) | 0.787 | |
| Ever consumed alcohol | ||||
| No | 47.7%, 53/111 | Reference | ||
| Yes | 68.3%, 28/41 | 1.43 (1.05–1.88) | 0.029 | |
| Sometimes | 70.1%, 49/70 | 1.47 (1.14–1.88) | 0.004 | |
| Past | 68.9%, 31/45 | 1.44 (1.08–1.88) | 0.021 | |
| Smoker | ||||
| No | 57.1%, 120/210 | Reference | ||
| Yes | 63.3%, 19/30 | 1.11 (0.78–1.42) | 0.559 | |
| Past | 81.5%, 22/27 | 1.43 (1.09–1.71) | 0.020 | |
| Currently working | ||||
| No | 62.0%, 101/163 | Reference | ||
| Yes | 57.7%, 60/104 | 0.93 (0.75–1.14) | 0.522 | |
| Marital status | ||||
| Single | 67.2%, 131/195 | Reference | ||
| Married/cohabiting | 45.8%, 27/59 | 0.68 (0.49–0.89) | 0.004 | |
| Separated/widowed | 23.1%, 3/13 | 0.34 (0.12–0.75) | 0.002 | |
| Number of lifetime sexual partners | ||||
| 1–2 | 55.4%, 31/56 | Reference | ||
| 3 | 62.2%, 46/74 | 1.12 (0.84–1.53) | 0.474 | |
| 4 | 65.1%, 28/43 | 1.18 (0.84–1.63) | 0.410 | |
| ≥5 | 59.6%, 56/94 | 1.08 (0.82–1.46) | 0.733 | |
| Number of sexual partners in the past year | ||||
| 0 | 45.8%, 22/48 | Reference | ||
| 1 | 63.6%, 110/173 | 1.39 (1.04–1.98) | 0.031 | |
| 2–4 | 63.0%, 29/46 | 1.38 (0.95–2.04) | 0.103 | |
| Number of sexual partners in the past month | ||||
| 0 | 55.8%, 43/77 | Reference | ||
| 1–2 | 62.1%, 118/190 | 1.11 (0.90–1.42) | 0.408 | |
| Condom use during last sexual intercourse | ||||
| No | 62.9%, 105/167 | Reference | ||
| Yes | 55.6%, 55/99 | 0.88 (0.71–1.08) | 0.247 | |
| I don’t remember | 100.0%, 1/1 | 1.59 (0.33–3.57) | 1.000 | |
| Ever pregnant | ||||
| No | 72.5%, 29/40 | Reference | ||
| Yes | 58.0%, 131/226 | 0.80 (0.66–1.04) | 0.114 | |
| Parity | ||||
| 0 | 87.5%, 14/16 | Reference | ||
| 1 | 66.7%, 40/60 | 0.76 (0.60–1.08) | 0.129 | |
| 2 | 60.7%, 34/56 | 0.68 (0.53–0.99) | 0.070 | |
| 3–4 | 62.1%, 41/66 | 0.71 (0.55–1.01) | 0.075 | |
| ≥5 | 39.4%, 15/38 | 0.45 (0.29–0.70) | 0.002 | |
| No response | 54.8%, 17/31 | 0.63 (0.42–0.93) | 0.049 | |
| Contraceptives | ||||
| None | 51.6%, 49/95 | Reference | ||
| Pills | 50.0%, 7/14 | 0.97 (0.51–1.52) | 1.000 | |
| Condoms | 73.5%, 25/34 | 1.43 (1.05–1.87) | 0.028 | |
| Intrauterine device | 66.7%, 8/12 | 1.29 (0.74–1.84) | 0.373 | |
| Depoprovera | 64.7%, 55/85 | 1.26 (0.98–1.62) | 0.096 | |
| Nur-Isterate | 69.2%, 9/13 | 1.34 (0.80–1.87) | 0.255 | |
| Withdrawal or other | 53.8%, 7/13 | 1.04 (0.55–1.60) | 1.000 | |
| Vaginal discharge | ||||
| No | 50.4%, 68/135 | Reference | ||
| Yes | 70.5%, 93/132 | 1.40 (1.15–1.72) | 0.0001 | |
| Genital warts/blisters | ||||
| No | 59.6%, 84/141 | Reference | ||
| Yes | 61.1%, 77/126 | 1.03 (0.84–1.25) | 0.804 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Kondlo, S.; Giyose, N.; Businge, C.B.; Mbulawa, Z.Z.A. Human Papillomavirus Genotype Landscape Across Cervical Cytology Grades and Impact of HIV Among Women of Eastern Cape Province, South Africa. Viruses 2026, 18, 65. https://doi.org/10.3390/v18010065
Kondlo S, Giyose N, Businge CB, Mbulawa ZZA. Human Papillomavirus Genotype Landscape Across Cervical Cytology Grades and Impact of HIV Among Women of Eastern Cape Province, South Africa. Viruses. 2026; 18(1):65. https://doi.org/10.3390/v18010065
Chicago/Turabian StyleKondlo, Sinazo, Nwabisa Giyose, Charles B. Businge, and Zizipho Z. A. Mbulawa. 2026. "Human Papillomavirus Genotype Landscape Across Cervical Cytology Grades and Impact of HIV Among Women of Eastern Cape Province, South Africa" Viruses 18, no. 1: 65. https://doi.org/10.3390/v18010065
APA StyleKondlo, S., Giyose, N., Businge, C. B., & Mbulawa, Z. Z. A. (2026). Human Papillomavirus Genotype Landscape Across Cervical Cytology Grades and Impact of HIV Among Women of Eastern Cape Province, South Africa. Viruses, 18(1), 65. https://doi.org/10.3390/v18010065






