Inhaled Dry Powder of Antiviral Agents: A Promising Approach to Treating Respiratory Viral Pathogens
Abstract
1. Introduction
2. A Brief Description of Common Respiratory Viral Pathogens
2.1. Influenza Virus
2.2. Respiratory Syncytial Virus
2.3. SARS-CoV-2
2.4. Rhinovirus
2.5. Adenovirus
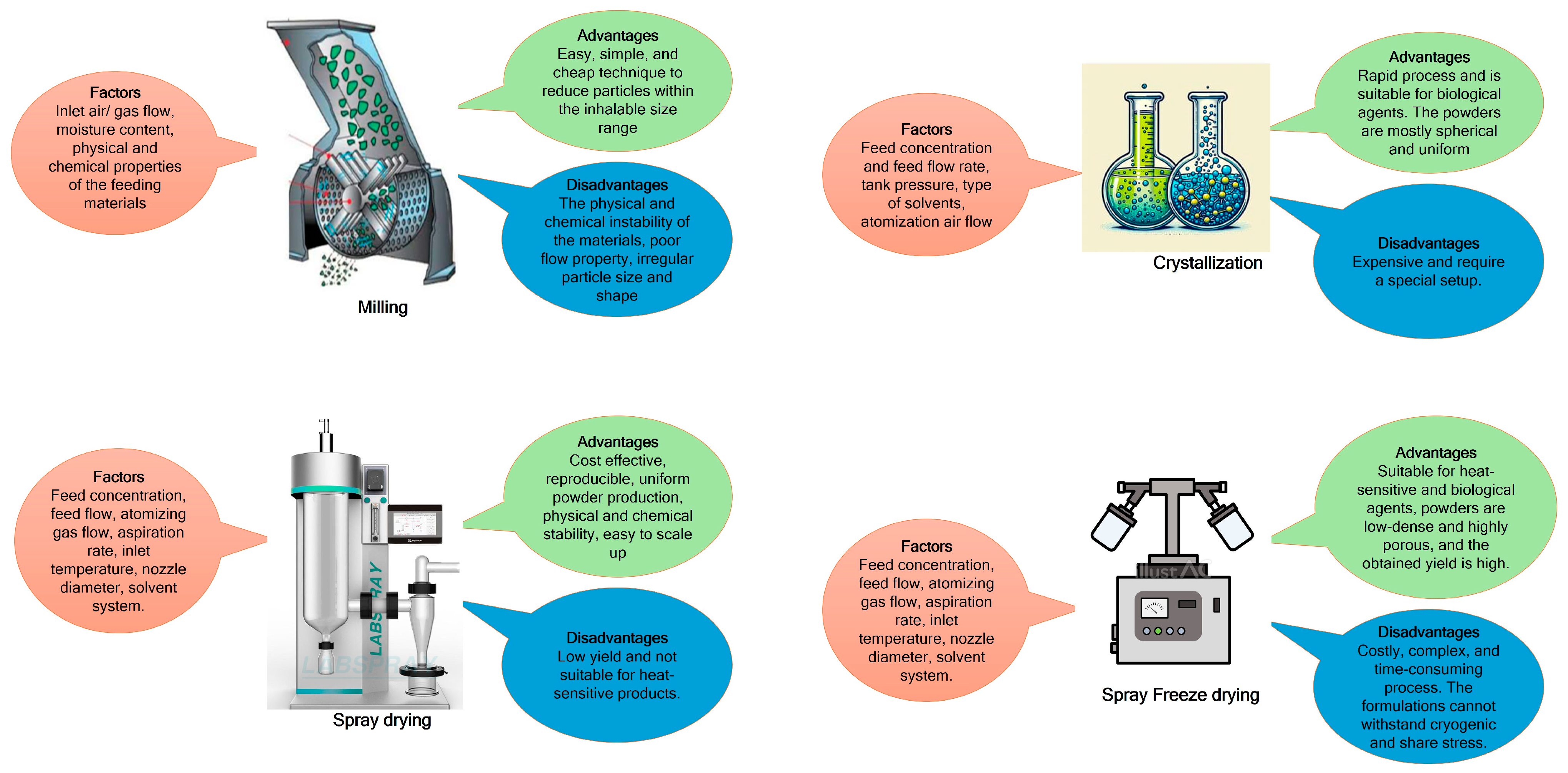
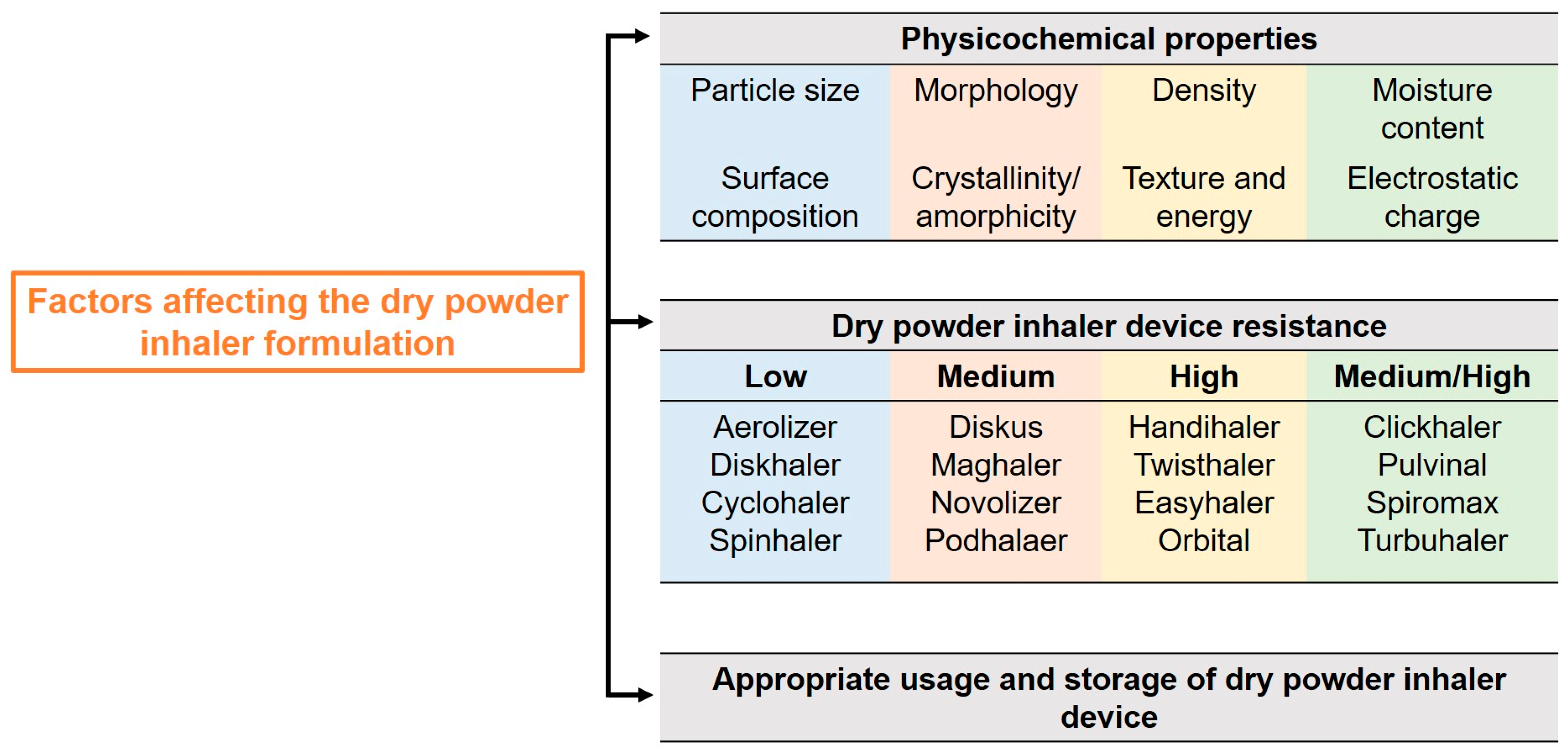
3. Considering Factors for a Successful Dry Powder Formulation Development
4. An Overview of Developed Dry Powder Inhaler of Antiviral Agents
5. An Overview of Marketed Dry Powder Inhaler Containing Antiviral Agents
6. Inhaled Combinational Formulations: An Area to Explore
| Drug A | Mode of Action | Drug B | Mode of Action | Pathogens | Cell Line Used | Ref |
|---|---|---|---|---|---|---|
| Nitazoxanide | Replication inhibitor | Oseltamivir | Neuraminidase inhibitor | Influenza virus | MDCK cell line | [110] |
| Nitazoxanide | Replication inhibitor | Zanamivir | Neuraminidase inhibitor | Influenza virus | MDCK cell line | [110] |
| Oseltamivir | RdRp inhibitor | Rimantadine | Viral replication | Influenza virus | MDCK cell line | [111] |
| Oseltamivir | RdRp inhibitor | Zanamivir | Neuraminidase inhibitor | Influenza virus | MDCK cell line | [112] |
| Oseltamivir | Neuraminidase inhibitor | Favipiravir | RdRp inhibitor | Influenza virus | MDCK cell line | [113] |
| Remdesivir | RdRp inhibitor | Ebselen | Protease inhibitor | SARS-CoV-2 | Vero cell line | [114] |
| Remdesivir | RdRp inhibitor | Disulfiram | Protease inhibitor | SARS-CoV-2 | Vero cell line | [114] |
| Remdesivir | RdRp inhibitor | Ivermectin | Importin α/β1 inhibitor | SARS-CoV-2 | 264.7 murine macrophage cell line | [115] |
| Remdesivir | RdRp inhibitor | Nitazoxanide | Entry inhibitor | SARS-CoV-2 | Vero E6 cell line | [116] |
| Nitazoxanide | Entry inhibitor | Umifenovir | Entry inhibitor | SARS-CoV-2 | Vero E6 cell line | [116] |
| Nitazoxanide | Entry inhibitor | Emetine dihydrochloride hydrate | Replication inhibitor | SARS CoV-2 | Vero E6 cell line | [116] |
| Nitazoxanide | Entry inhibitor | Amodiaquine | Entry inhibitor | SARS-CoV-2 | Vero E6 cell line | [116] |
| Favipiravir | RdRp inhibitor | Ivermectin | Importin α/β1 inhibitor | SARS-CoV-2 | Vero E6 cell line | [117] |
| Otamixaban | Entry inhibitor | Camostat | TMPRSS2 inhibitor | SARS-CoV-2 | Vero E6 cell line | [118] |
| Otamixaban | Entry inhibitor | Nafamostat | TMPRSS2 inhibitor | SARS-CoV-2 | Vero E6 cell line | [118] |
| Remdesivir | RdRp inhibitor | Brequinar | Replication inhibitor | SARS-CoV-2 | Vero E6 cell line | [119] |
| Molnupiravir | RdRp inhibitor | Brequinar | Replication inhibitor | SARS-CoV-2 | Vero E6 cell line | [119] |
| Cepharanthine | Entry inhibitor | Nelfinavir | Replication inhibitor | SARS-CoV-2 | Vero E6 cell line | [120] |
| Lumicitabine (ALS8176) | RdRp inhibitor | RSV604 | RdRp inhibitor | RSV | Hep-2 cells | [121] |
| Lumicitabine (ALS8176) | RdRp inhibitor | BMS433771 | Inhibits fusion protein | RSV | Hep-2 cells | [121] |
| Presatovir (GS5806) | Inhibits fusion protein | BMS433771 | Inhibits fusion protein | RSV | Hep-2 cells | [121] |
| Lumicitabine (ALS8176) | RdRp inhibitor | Ziresovir | Inhibits fusion protein | RSV | Hep-2 cells | [121] |
| Lumicitabine (ALS8176) | RdRp inhibitor | Presatovir (GS5806) | Inhibits fusion protein | RSV | Hep-2 cells | [121] |
| Glycyrrhizic acid | Inhibits entry | Ephedrine | Inhibits entry | RSV | A549 cells | [122] |
| Fluticasone propionate | Inhibits VEGF, FGF-2 production | Salmeterol | Inhibits VEGF, FGF-2 production | Rhinovirus | Bronchial epithelial cells | [123] |
| Rupintrivir | Inhibits replication by targeting 3C protease | Itraconazole | Targets oxysterol-binding protein and inhibits replication | Enterovirus 71 | RD cells | [124] |
| Rupintrivir | Inhibits replication by targeting 3C protease | Favipiravir | Inhibits replication targeting RNA polymerase | Enterovirus 71 | RD cells | [124] |
| Suramin | Prevents attaching to the host cell surface receptors | Favipiravir | Inhibits replication targeting RNA polymerase | Enterovirus 71 | RD cells | [124] |
| Rupintrivir | Inhibits replication by targeting 3C protease | Vemurafenib | Inhibits replication | Enterovirus 1 | A549 cells | [125] |
| Rupintrivir | Inhibits replication by targeting 3C protease | Pleconaril | Inhibits replication by binding to viral capsid | Enterovirus 1 | A549 cells | [125] |
| Pleconaril | Inhibits replication by binding to viral capsid | Vemurafenib | Inhibits replication | Enterovirus 1 | A549 cells | [125] |
| Rupintrivir | Inhibits replication by targeting 3C protease | Interferon | Inhibits replication | Enterovirus 71 | Vero cells | [126] |
| Ribavirin | RdRp inhibitor | Gemcitabine | Inhibits replication | Enterovirus 71 | Vero cells | [127] |
| Rupintrivir | Inhibits replication by targeting 3C protease | Cycloheximide | Inhibits replication | Enterovirus 71 | A549 cells | [125] |
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ong, C.W.M.; Migliori, G.B.; Raviglione, M.; MacGregor-Skinner, G.; Sotgiu, G.; Alffenaar, J.W.; Tiberi, S.; Adlhoch, C.; Alonzi, T.; Archuleta, S.; et al. Epidemic and pandemic viral infections: Impact on tuberculosis and the lung: A consensus by the World Association for Infectious Diseases and Immunological Disorders (WAidid), Global Tuberculosis Network (GTN), and members of the European Society of Clinical Microbiology and Infectious Diseases Study Group for Mycobacterial Infections (ESGMYC). Eur. Respir. J. 2020, 56, 2001727. [Google Scholar] [PubMed]
- Caballero, M.T.; Bianchi, A.M.; Nuño, A.; Ferretti, A.J.P.; Polack, L.M.; Remondino, I.; Rodriguez, M.G.; Orizzonte, L.; Vallone, F.; Bergel, E.; et al. Mortality associated with acute respiratory infections among children at home. J. Infect. Dis. 2019, 219, 358–364. [Google Scholar] [CrossRef] [PubMed]
- Han, S. Clinical vaccine development. Clin. Exp. Vaccine Res. 2015, 4, 46–53. [Google Scholar] [CrossRef] [PubMed]
- Shyr, Z.A.; Cheng, Y.S.; Lo, D.C.; Zheng, W. Drug combination therapy for emerging viral diseases. Drug Discov. Today 2021, 26, 2367–2376. [Google Scholar] [CrossRef] [PubMed]
- Feikin, D.R.; Higdon, M.M.; Abu-Raddad, L.J.; Andrews, N.; Araos, R.; Goldberg, Y.; Groome, M.J.; Huppert, A.; O’Brien, K.L.; Smith, P.G.; et al. Duration of effectiveness of vaccines against SARS-CoV-2 infection and COVID-19 disease: Results of a systematic review and meta-regression. Lancet 2022, 399, 924–944. [Google Scholar] [CrossRef]
- Olliaro, P.; Torreele, E.; Vaillant, M. COVID-19 vaccine efficacy and effectiveness-the elephant (not) in the room. Lancet Microbe 2021, 2, e279–e280. [Google Scholar] [CrossRef]
- Nuwarda, R.F.; Ramzan, I.; Weekes, L.; Kayser, V. Vaccine hesitancy: Contemporary issues and historical background. Vaccines 2022, 10, 1595. [Google Scholar] [CrossRef] [PubMed]
- Pourrazavi, S.; Fathifar, Z.; Sharma, M.; Allahverdipour, H. COVID-19 vaccine hesitancy: A systematic review of cognitive determinants. Health Promot. Perspect. 2023, 13, 21–35. [Google Scholar] [CrossRef] [PubMed]
- Saha, T.; Quiñones-Mateu, M.E.; Das, S.C. Inhaled therapy for COVID-19: Considerations of drugs, formulations and devices. Int. J. Pharm. 2022, 624, 122042. [Google Scholar] [CrossRef]
- Martinez, M.N.; Papich, M.G.; Drusano, G.L. Dosing regimen matters: The importance of early intervention and rapid attainment of the pharmacokinetic/pharmacodynamic target. Antimicrob. Agents Chemother. 2012, 56, 2795–2805. [Google Scholar] [CrossRef] [PubMed]
- Eedara, B.B.; Alabsi, W.; Encinas-Basurto, D.; Polt, R.; Ledford, J.G.; Mansour, H.M. Inhalation delivery for the treatment and prevention of COVID-19 infection. Pharmaceutics 2021, 13, 1077. [Google Scholar] [CrossRef] [PubMed]
- Borghardt, J.M.; Kloft, C.; Sharma, A. Inhaled therapy in respiratory disease: The complex interplay of pulmonary kinetic processes. Can. Respir. J. 2018, 2018, 2732017. [Google Scholar] [CrossRef]
- Vermillion, M.S.; Murakami, E.; Ma, B.; Pitts, J.; Tomkinson, A.; Rautiola, D.; Babusis, D.; Irshad, H.; Seigel, D.; Kim, C.; et al. Inhaled remdesivir reduces viral burden in a nonhuman primate model of SARS-CoV-2 infection. Sci. Transl. Med. 2022, 14, eabl8282. [Google Scholar] [CrossRef]
- Brahim, M.; Verma, R.; Garcia-Contreras, L. Inhalation drug delivery devices: Technology update. Med. Devices (Auckl) 2015, 8, 131–139. [Google Scholar]
- Niven, R. Prospects and challenges: Inhalation delivery systems. Ther. Deliv. 2013, 4, 519–522. [Google Scholar] [CrossRef] [PubMed]
- Nainwal, N. Treatment of respiratory viral infections through inhalation therapeutics: Challenges and opportunities. Pulm. Pharmacol. Ther. 2022, 77, 102170. [Google Scholar] [CrossRef] [PubMed]
- Adhikari, B.R.; Dummer, J.; Gordon, K.C.; Das, S.C. An expert opinion on respiratory delivery of high-dose powders for lung infections. Expert Opin. Drug Deliv. 2022, 19, 795–813. [Google Scholar] [CrossRef] [PubMed]
- Sibum, I.; Hagedoorn, P.; de Boer, A.H.; Frijlink, H.W.; Grasmeijer, F. Challenges for pulmonary delivery of high powder doses. Int. J. Pharm. 2018, 548, 325–336. [Google Scholar] [CrossRef] [PubMed]
- Ari, A. Practical strategies for a safe and effective delivery of aerosolized medications to patients with COVID-19. Respir. Med. 2020, 167, 105987. [Google Scholar] [CrossRef] [PubMed]
- Bountouri, M.; Ntafis, V.; Fragkiadaki, E.; Kanellos, T.; Xylouri, E. Phylogenetic analysis of the five internal genes and evolutionary pathways of the Greek H3N8 equine influenza virus. Nat. Sci. 2012, 4, 839–847. [Google Scholar] [CrossRef]
- Yewdell, J.; García-Sastre, A. Influenza virus still surprises. Curr. Opin. Microbiol. 2002, 5, 414–418. [Google Scholar] [CrossRef] [PubMed]
- Wright, K.E.; Wilson, G.A.; Novosad, D.; Dimock, C.; Tan, D.; Weber, J.M. Typing and subtyping of influenza viruses in clinical samples by PCR. J. Clin. Microbiol. 1995, 33, 1180–1184. [Google Scholar] [CrossRef]
- Cottey, R.; Rowe, C.A.; Bender, B.S. Influenza virus. Curr. Protoc. Immunol. 2001, 42, 19.11.1–19.11.32. [Google Scholar] [CrossRef]
- Taubenberger, J.K.; Morens, D.M. The pathology of influenza virus infections. Annu. Rev. Pathol. Mech. Dis. 2008, 3, 499–522. [Google Scholar] [CrossRef]
- Van den Dool, C.; Hak, E.; Wallinga, J.; Van Loon, A.M.; Lammers, J.W.J.; Bonten, M.J.M. Symptoms of influenza virus infection in hospitalized patients. Infect. Control Hosp. Epidemiol. 2008, 29, 314–319. [Google Scholar] [CrossRef] [PubMed]
- Banning, M. Influenza: Incidence, symptoms, and treatment. Br. J. Nurs. 2005, 14, 1192–1197. [Google Scholar] [CrossRef]
- Eccles, R. Understanding the symptoms of the common cold and influenza. Lancet Infect. Dis. 2005, 5, 718–725. [Google Scholar] [CrossRef] [PubMed]
- Rohwedder, A.; Keminer, O.; Forster, J.; Schneider, K.; Schneider, E.; Werchau, H. Detection of respiratory syncytial virus RNA in blood of neonates by polymerase chain reaction. J. Med. Virol. 1998, 54, 320–327. [Google Scholar] [CrossRef]
- Gardner, P.S.; McQuillin, J. Rapid Virus Diagnosis: Application Of immunofluorescence; Butterworth-Heinemann: Oxford, UK, 2014. [Google Scholar]
- Åkerlind, B.; Norrby, E.; Örvell, C.; Mufson, M.A. Respiratory syncytial virus: Heterogeneity of subgroup B strains. J. Gen. Virol. 1988, 69, 2145–2154. [Google Scholar] [CrossRef] [PubMed]
- Mitra, R.; Baviskar, P.; Duncan-Decocq, R.R.; Patel, D.; Oomens, A.G. The human respiratory syncytial virus matrix protein is required for maturation of viral filaments. J. Virol. 2012, 86, 4432–4443. [Google Scholar] [CrossRef]
- Jha, A.; Jarvis, H.; Fraser, C.; Openshaw, P. Respiratory syncytial virus. SARS MERS Other Viral Lung Infect. 2016. Available online: https://www.ncbi.nlm.nih.gov/books/NBK442240/ (accessed on 14 January 2025).
- Li, Y.; Wang, X.; Blau, D.M.; Caballero, M.T.; Feikin, D.R.; Gill, C.J.; Nair, H. Global, regional, and national disease burden estimates of acute lower respiratory infections due to respiratory syncytial virus in children younger than 5 years in 2019: A systematic analysis. Lancet 2022, 399, 2047–2064. [Google Scholar] [CrossRef] [PubMed]
- Savic, M.; Penders, Y.; Shi, T.; Branche, A.; Pirçon, J.Y. Respiratory syncytial virus disease burden in adults aged 60 years and older in high-income countries: A systematic literature review and meta-analysis. Influenza Other Respir. Viruses 2023, 17, e13031. [Google Scholar] [CrossRef]
- Polak, M.J. Respiratory syncytial virus (RSV): Overview, treatment, and prevention strategies. Newborn Infant Nurs. Rev. 2004, 4, 15–23. [Google Scholar] [CrossRef]
- Walsh, E.E.; Falsey, A.R. Respiratory syncytial virus infection in adult populations. Infect. Disord.-Drug Targets 2012, 12, 98–102. [Google Scholar] [CrossRef]
- Harrison, A.G.; Lin, T.; Wang, P. Mechanisms of SARS-CoV-2 transmission and pathogenesis. Trends Immunol. 2020, 41, 1100–1115. [Google Scholar] [CrossRef]
- Baloch, S.; Baloch, M.A.; Zheng, T.; Pei, X. The coronavirus disease 2019 (COVID-19) pandemic. Tohoku J. Exp. Med. 2020, 250, 271–278. [Google Scholar] [CrossRef]
- Gorbalenya, A.; Baker, S.; Baric, R.S.; de Groot, R.; Drosten, C.; Gulyaeva, A.A.; Ziebuhr, J. The species severe acute respiratory syndrome-related coronavirus: Classifying 2019-nCoV and naming it SARS-CoV-2. Nat. Microbiol. 2020, 5, 536–544. [Google Scholar]
- Snijder, E.J.; Van Der Meer, Y.; Zevenhoven-Dobbe, J.; Onderwater, J.J.; Van Der Meulen, J.; Koerten, H.K.; Mommaas, A.M. Ultrastructure and origin of membrane vesicles associated with the severe acute respiratory syndrome coronavirus replication complex. J. Virol. 2006, 80, 5927–5940. [Google Scholar] [CrossRef] [PubMed]
- Cortese, M.; Lee, J.Y.; Cerikan, B.; Neufeldt, C.J.; Oorschot, V.M.; Köhrer, S.; Bartenschlager, R. Integrative imaging reveals SARS-CoV-2-induced reshaping of subcellular morphologies. Cell Host Microbe 2020, 28, 853–866. [Google Scholar] [CrossRef]
- Ghosh, S.; Dellibovi-Ragheb, T.A.; Kerviel, A.; Pak, E.; Qiu, Q.; Fisher, M.; Altan-Bonnet, N. β-Coronaviruses use lysosomes for egress instead of the biosynthetic secretory pathway. Cell 2020, 183, 1520–1535. [Google Scholar] [CrossRef] [PubMed]
- Scherer, K.M.; Mascheroni, L.; Carnell, G.W.; Wunderlich, L.C.; Makarchuk, S.; Brockhoff, M.; Kaminski, C.F. SARS-CoV-2 nucleocapsid protein adheres to replication organelles before viral assembly at the Golgi/ERGIC and lysosome-mediated egress. Sci. Adv. 2022, 8, eabl4895. [Google Scholar] [CrossRef] [PubMed]
- Grant, M.C.; Geoghegan, L.; Arbyn, M.; Mohammed, Z.; McGuinness, L.; Clarke, E.L.; Wade, R.G. The prevalence of symptoms in 24,410 adults infected by the novel coronavirus (SARS-CoV-2; COVID-19): A systematic review and meta-analysis of 148 studies from 9 countries. PLoS ONE 2020, 15, e0234765. [Google Scholar] [CrossRef]
- Chung, E.; Chow, E.J.; Wilcox, N.C.; Burstein, R.; Brandstetter, E.; Han, P.D.; Chu, H.Y. Comparison of symptoms and RNA levels in children and adults with SARS-CoV-2 infection in the community setting. JAMA Pediatr. 2021, 175, e212025. [Google Scholar] [CrossRef] [PubMed]
- Nasserie, T.; Hittle, M.; Goodman, S.N. Assessment of the frequency and variety of persistent symptoms among patients with COVID-19: A systematic review. JAMA Netw. Open 2021, 4, e2111417. [Google Scholar] [CrossRef] [PubMed]
- Scharf, R.E.; Anaya, J.M. Post-COVID syndrome in adults—An overview. Viruses 2023, 15, 675. [Google Scholar] [CrossRef]
- Bizot, E.; Bousquet, A.; Charpié, M.; Coquelin, F.; Lefevre, S.; Le Lorier, J.; Basmaci, R. Rhinovirus: A narrative review on its genetic characteristics, pediatric clinical presentations, and pathogenesis. Front. Pediatr. 2021, 9, 643219. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, S.E.; Lamson, D.M.; St. George, K.; Walsh, T.J. Human Rhinoviruses. Clin. Microbiol. Rev. 2013, 26, 135–162. [Google Scholar] [CrossRef]
- Carstens, E.B. Ratification vote on taxonomic proposals to the International Committee on Taxonomy of Viruses (2009). Arch. Virol. 2010, 155, 133–146. [Google Scholar] [CrossRef] [PubMed]
- Ledford, R.M.; Patel, N.R.; Demenczuk, T.M.; Watanyar, A.; Herbertz, T.; Collett, M.S. VP1 sequencing of all human rhinovirus serotypes: Insights into genus phylogeny and susceptibility to antiviral capsid-binding compounds. J. Virol. 2004, 78, 3663–3674. [Google Scholar] [CrossRef] [PubMed]
- Ganjian, H.; Zietz, C.; Mechtcheriakova, D.; Blaas, D.; Fuchs, R. ICAM-1 binding rhinoviruses enter HeLa cells via multiple pathways and travel to distinct intracellular compartments for uncoating. Viruses 2017, 9, 68. [Google Scholar] [CrossRef]
- Esneau, C.; Bartlett, N.; Bochkov, Y.A. Rhinovirus structure, replication, and classification. In Rhinovirus Infections; Academic Press: Cambridge, MA, USA, 2019; pp. 1–23. [Google Scholar]
- Greenberg, S.B. Respiratory consequences of rhinovirus infection. Arch. Intern. Med. 2003, 163, 278–284. [Google Scholar] [CrossRef]
- Peltola, V.; Waris, M.; Österback, R.; Susi, P.; Hyypiä, T.; Ruuskanen, O. Clinical effects of rhinovirus infections. J. Clin. Virol. 2008, 43, 411–414. [Google Scholar] [CrossRef] [PubMed]
- Lynch, J.P.; Fishbein, M.; Echavarria, M. Adenovirus. Semin. Respir. Crit. Care Med. 2011, 32, 494–511. [Google Scholar] [CrossRef] [PubMed]
- Greber, U.F. Adenoviruses–infection, pathogenesis and therapy. FEBS Lett. 2020, 594, 1818–1827. [Google Scholar] [CrossRef]
- Lasswitz, L.; Chandra, N.; Arnberg, N.; Gerold, G. Glycomics and proteomics approaches to investigate early adenovirus–host cell interactions. J. Mol. Biol. 2018, 430, 1863–1882. [Google Scholar] [CrossRef] [PubMed]
- Fleischli, C.; Sirena, D.; Lesage, G.; Havenga, M.J.; Cattaneo, R. Species B adenovirus serotypes 3, 7, 11 and 35 share similar binding sites on the membrane cofactor protein CD46 receptor. J. Gen. Virol. 2007, 88, 2925–2934. [Google Scholar] [CrossRef] [PubMed]
- Wodrich, H.; Henaff, D.; Jammart, B.; Segura-Morales, C.; Seelmeir, S. A capsid-encoded PPxY-motif facilitates adenovirus entry. PLoS Pathog. 2010, 6, e1000808. [Google Scholar] [CrossRef]
- Trotman, L.C.; Mosberger, N.; Fornerod, M.; Stidwill, R.P.; Greber, U.F. Import of adenovirus DNA involves the nuclear pore complex receptor CAN/Nup214 and histone H1. Nat. Cell Biol. 2001, 3, 1092–1100. [Google Scholar]
- Karen, K.A.; Hearing, P. Adenovirus core protein VII protects the viral genome from a DNA damage response at early times after infection. J. Virol. 2011, 85, 4135–4142. [Google Scholar] [CrossRef] [PubMed]
- Edwards, K.M.; Thompson, J.; Paolini, J.; Wright, P.F. Adenovirus infections in young children. Pediatrics 1985, 76, 420–424. [Google Scholar] [CrossRef] [PubMed]
- Mufson, M.A.; Zollar, L.M.; Mankad, V.N. Adenovirus infection in acute hemorrhagic cystitis: A study in 25 children. Am. J. Dis. Child. 1971, 121, 281–285. [Google Scholar] [CrossRef] [PubMed]
- Chou, S.M.; Roos, R.; Burrell, R.; Gutmann, L.; Harley, J.B. Subacute focal adenovirus encephalitis. J. Neuropathol. Exp. Neurol. 1973, 32, 34–50. [Google Scholar] [CrossRef] [PubMed]
- Henson, D.; Mufson, M.A. Myocarditis and pneumonitis with type 21 adenovirus infection: Association with fatal myocarditis and pneumonitis. Am. J. Dis. Child. 1971, 121, 334–336. [Google Scholar] [CrossRef] [PubMed]
- Zahradnik, J.M.; Spencer, M.J.; Porter, D.D. Adenovirus infection in the immunocompromised patient. Am. J. Med. 1980, 68, 725–732. [Google Scholar] [CrossRef] [PubMed]
- Chaurasiya, B.; Zhao, Y.Y. Dry Powder for Pulmonary Delivery: A Comprehensive Review. Pharmaceutics 2020, 13, 31. [Google Scholar] [CrossRef] [PubMed]
- Abiona, O.; Wyatt, D.; Koner, J.; Mohammed, A. The Optimisation of Carrier Selection in Dry Powder Inhaler Formulation and the Role of Surface Energetics. Biomedicines 2022, 10, 2707. [Google Scholar] [CrossRef]
- Chow, A.H.L.; Tong, H.H.Y.; Chattopadhyay, P.; Shekunov, B.Y. Particle Engineering for Pulmonary Drug Delivery. Pharm. Res. 2007, 24, 411–437. [Google Scholar] [CrossRef]
- Saleem, I.Y.; Smyth, H.D.C. Micronization of a Soft Material: Air-Jet and Micro-Ball Milling. AAPS PharmSciTech 2010, 11, 1642–1649. [Google Scholar] [CrossRef]
- Shetty, N.; Cipolla, D.; Park, H.; Zhou, Q.T. Physical stability of dry powder inhaler formulations. Expert Opin. Drug Deliv. 2020, 17, 77–96. [Google Scholar] [CrossRef] [PubMed]
- Patel, R.P. Spray drying technology: An overview. Indian J. Sci. Technol. 2009, 2, 44–47. [Google Scholar] [CrossRef]
- Patel, B.B.; Patel, J.K.; Chakraborty, S.; Shukla, D. Revealing facts behind spray dried solid dispersion technology used for solubility enhancement. Saudi Pharm. J. 2015, 23, 352–365. [Google Scholar] [CrossRef] [PubMed]
- Maa, Y.-F.; Prestrelski, S.J. Biopharmaceutical Powders Particle Formation and Formulation Considerations. Curr. Pharm. Biotechnol. 2000, 1, 283–302. [Google Scholar] [CrossRef] [PubMed]
- Pardeshi, S.R.; Kole, E.B.; Kapare, H.S.; Chandankar, S.M.; Shinde, P.J.; Boisa, G.S.; Salgaonkar, S.S.; Giram, P.S.; More, M.P.; Kolimi, P.; et al. Progress on Thin Film Freezing Technology for Dry Powder Inhalation Formulations. Pharmaceutics 2022, 14, 2632. [Google Scholar] [CrossRef]
- Dhoble, S.; Kapse, A.; Ghegade, V.; Chogale, M.; Ghodake, V.; Patravale, V.; Vora, L.K. Design, development, and technical considerations for dry powder inhaler devices. Drug Discov. Today 2024, 29, 103954. [Google Scholar] [CrossRef] [PubMed]
- Mehta, P. Dry Powder Inhalers: A Focus on Advancements in Novel Drug Delivery Systems. J. Drug Deliv. 2016, 2016, 8290963. [Google Scholar] [CrossRef]
- Heyder, J. Deposition of inhaled particles in the human respiratory tract and consequences for regional targeting in respiratory drug delivery. Proc. Am. Thorac. Soc. 2004, 1, 315–320. [Google Scholar] [CrossRef] [PubMed]
- Edwards, D.A.; Hanes, J.; Caponetti, G.; Hrkach, J.; Ben-Jebria, A.; Eskew, M.L.; Mintzes, J.; Deaver, D.; Lotan, N.; Langer, R. Large porous particles for pulmonary drug delivery. Science 1997, 276, 1868–1871. [Google Scholar] [CrossRef] [PubMed]
- Hassan, M.S.; Lau, R.W. Effect of particle shape on dry particle inhalation: Study of flowability, aerosolization, and deposition properties. AAPS PharmSciTech 2009, 10, 1252–1262. [Google Scholar] [CrossRef]
- Momin, M.A.M.; Tucker, I.G.; Das, S.C. High dose dry powder inhalers to overcome the challenges of tuberculosis treatment. Int. J. Pharm. 2018, 550, 398–417. [Google Scholar] [CrossRef] [PubMed]
- Young, P.M.; Price, R.; Tobyn, M.J.; Buttrum, M.; Dey, F. Effect of humidity on aerosolization of micronized drugs. Drug Dev. Ind. Pharm. 2003, 29, 959–966. [Google Scholar] [CrossRef]
- Vehring, R. Pharmaceutical particle engineering via spray drying. Pharm. Res. 2008, 25, 999–1022. [Google Scholar] [CrossRef] [PubMed]
- Thomas, M.; Williams, A.E. Are outcomes the same with all dry powder inhalers? Int. J. Clin. Pract. Suppl. 2005, 149, 33–35. [Google Scholar] [CrossRef] [PubMed]
- Molimard, M.; Raherison, C.; Lignot, S.; Depont, F.; Abouelfath, A.; Moore, N. Assessment of handling of inhaler devices in real life: An observational study in 3811 patients in primary care. J. Aerosol Med. 2003, 16, 249–254. [Google Scholar] [CrossRef] [PubMed]
- Singh, T.U.; Parida, S.; Lingaraju, M.C.; Kesavan, M.; Kumar, D.; Singh, R.K. Drug repurposing approach to fight COVID-19. Pharmacol. Rep. 2020, 72, 1479–1508. [Google Scholar] [CrossRef]
- Hua, Y.; Dai, X.; Xu, Y.; Xing, G.; Liu, H.; Lu, T.; Chen, Y.; Zhang, Y. Drug repositioning: Progress and challenges in drug discovery for various diseases. Eur. J. Med. Chem. 2022, 234, 114239. [Google Scholar] [CrossRef] [PubMed]
- Sahakijpijarn, S.; Moon, C.; Koleng, J.J.; Christensen, D.J.; Williams, R.O., III. Development of Remdesivir as a Dry Powder for Inhalation by Thin Film Freezing. Pharmaceutics 2020, 12, 1002. [Google Scholar] [CrossRef] [PubMed]
- Saha, T.; Sinha, S.; Harfoot, R.; Quiñones-Mateu, M.E.; Das, S.C. Inhalable dry powder containing remdesivir and disulfiram: Preparation and in vitro characterization. Int. J. Pharm. 2023, 645, 123411. [Google Scholar] [CrossRef]
- Wong, S.N.; Weng, J.; Ip, I.; Chen, R.; Lakerveld, R.; Telford, R.; Blagden, N.; Scowen, I.J.; Chow, S.F. Rational development of a carrier-free dry powder inhalation formulation for respiratory viral infections via quality by design: A drug-drug cocrystal of favipiravir and theophylline. Pharmaceutics 2022, 14, 300. [Google Scholar] [CrossRef]
- Zhang, S.; Yan, S.; Lu, K.; Qiu, S.; Chen, X.D.; Wu, W.D. Spray freeze dried niclosamide nanocrystals embedded dry powder for high dose pulmonary delivery. Powder Technol. 2023, 415, 118168. [Google Scholar] [CrossRef]
- Saha, T.; Sinha, S.; Harfoot, R.; Quiñones-Mateu, M.E.; Das, S.C. Manipulation of spray-drying conditions to develop an inhalable ivermectin dry powder. Pharmaceutics 2022, 14, 1432. [Google Scholar] [CrossRef]
- Panozzo, J.; Oh, D.Y.; Margo, K.; Morton, D.A.; Piedrafita, D.; Mosse, J.; Hurt, A.C. Evaluation of a dry powder delivery system for laninamivir in a ferret model of influenza infection. Antivir. Res. 2015, 120, 66–71. [Google Scholar] [CrossRef] [PubMed]
- Aziz, S.; Scherlieβ, R.; Steckel, H. Development of high dose oseltamivir phosphate dry powder for inhalation therapy in viral pneumonia. Pharmaceutics 2020, 12, 1154. [Google Scholar] [CrossRef]
- Seow, H.C.; Liao, Q.; Lau, A.T.Y.; Leung, S.W.S.; Yuan, S.; Lam, J.K.W. Dual targeting powder formulation of antiviral agent for customizable nasal and lung deposition profile through single intranasal administration. Int. J. Pharm. 2022, 619, 121704. [Google Scholar] [CrossRef] [PubMed]
- Leung, S.S.Y.; Parumasivam, T.; Tang, P.; Chan, H.K. A proof-of-principle setup for delivery of Relenza® (Zanamivir) inhalation powder to intubated patients. J. Aerosol Med. Pulm. Drug Deliv. 2016, 30, 30–35. [Google Scholar] [CrossRef] [PubMed]
- Gaikwad, S.S.; Pathare, S.R.; More, M.A.; Waykhinde, N.A.; Laddha, U.D.; Salunkhe, K.S.; Kshirsagar, S.J.; Patil, S.S.; Ramteke, K.H. Dry powder inhaler with the technical and practical obstacles, and forthcoming platform strategies. J. Control Release 2023, 355, 292–311. [Google Scholar] [CrossRef] [PubMed]
- Muralidharan, P.; Hayes, D., Jr.; Mansour, H.M. Dry powder inhalers in COPD, lung inflammation and pulmonary infections. Expert Opin. Drug Deliv. 2015, 12, 947–962. [Google Scholar] [CrossRef] [PubMed]
- Laborda, P.; Wang, S.Y.; Voglmeir, J. Influenza neuraminidase inhibitors: Synthetic approaches, derivatives and biological activity. Molecules 2016, 21, 1513. [Google Scholar] [CrossRef] [PubMed]
- Feng, E.; Ye, D.; Li, J.; Zhang, D.; Wang, J.; Zhao, F.; Hilgenfeld, R.; Zheng, M.; Jiang, H.; Liu, H. Recent Advances in Neuraminidase Inhibitor Development as Anti-influenza Drugs. ChemMedChem 2012, 7, 1527–1536. [Google Scholar] [CrossRef]
- Tian, J.S.; Zhong, J.K.; Li, Y.S.; Ma, D.W. Organocatalytic and Scalable Synthesis of the Anti-Influenza Drugs Zanamivir, Laninamivir, and CS-8958. Angew. Chem. Int. Ed. 2014, 53, 13885–13888. [Google Scholar] [CrossRef] [PubMed]
- White, J.M.; Schiffer, J.T.; Bender Ignacio, R.A.; Xu, S.; Kainov, D.; Ianevski, A.; Aittokallio, T.; Frieman, M.; Olinger, G.G.; Polyak, S.J. Drug Combinations as a First Line of Defense against Coronaviruses and Other Emerging Viruses. mBio 2021, 12, e0334721. [Google Scholar] [CrossRef] [PubMed]
- Lehár, J.; Krueger, A.S.; Avery, W.; Heilbut, A.M.; Johansen, L.M.; Price, E.R.; Rickles, R.J.; Short, G.F., III; Staunton, J.E.; Jin, X.; et al. Synergistic Drug Combinations Tend to Improve Therapeutically Relevant Selectivity. Nat. Biotechnol. 2009, 27, 659–666. [Google Scholar] [CrossRef] [PubMed]
- Poduri, R.; Joshi, G.; Jagadeesh, G. Drugs Targeting Various Stages of the SARS-CoV-2 Life Cycle: Exploring Promising Drugs for the Treatment of COVID-19. Cell Signal. 2020, 74, 109721. [Google Scholar] [CrossRef] [PubMed]
- Wagoner, J.; Herring, S.; Hsiang, T.Y.; Ianevski, A.; Biering, S.B.; Xu, S.; Hoffmann, M.; Pöhlmann, S.; Gale, M., Jr.; Aittokallio, T.; et al. Combinations of Host- and Virus-Targeting Antiviral Drugs Confer Synergistic Suppression of SARS-CoV-2. Microbiol. Spectr. 2022, 10, e0333122. [Google Scholar] [CrossRef] [PubMed]
- Dybul, M.; Fauci, A.S.; Bartlett, J.G.; Kaplan, J.E.; Pau, A.K. Guidelines for Using Antiretroviral Agents among HIV-Infected Adults and Adolescents. MMWR Recomm. Rep. 2002, 51, 1–55. [Google Scholar]
- Babawale, P.I.; Guerrero-Plata, A. Respiratory Viral Coinfections: Insights into Epidemiology, Immune Response, Pathology, and Clinical Outcomes. Pathogens 2024, 13, 316. [Google Scholar] [CrossRef]
- Georgakopoulou, V.E. Insights from Respiratory Virus Co-infections. World J. Virol. 2024, 13, 98600. [Google Scholar] [CrossRef]
- Belardo, G.; Cenciarelli, O.; La Frazia, S.; Rossignol, J.F.; Santoro, M.G. Synergistic Effect of Nitazoxanide with Neuraminidase Inhibitors against Influenza A Viruses in Vitro. Antimicrob. Agents Chemother. 2015, 59, 1061–1069. [Google Scholar] [CrossRef]
- Galabov, A.S.; Simeonova, L.; Gegova, G. Rimantadine and Oseltamivir Demonstrate Synergistic Combination Effect in an Experimental Infection with Type A (H3N2) Influenza Virus in Mice. Antivir. Chem. Chemother. 2006, 17, 251–258. [Google Scholar] [CrossRef] [PubMed]
- de Mello, C.P.P.; Drusano, G.L.; Adams, J.R.; Shudt, M.; Kulawy, R.; Brown, A.N. Oseltamivir-Zanamivir Combination Therapy Suppresses Drug-Resistant H1N1 Influenza A Viruses in the Hollow Fiber Infection Model (HFIM) System. Eur. J. Pharm. Sci. 2018, 111, 443–449. [Google Scholar] [CrossRef] [PubMed]
- Sleeman, K.; Mishin, V.P.; Deyde, V.M.; Furuta, Y.; Klimov, A.I.; Gubareva, L.V. In Vitro Antiviral Activity of Favipiravir (T-705) against Drug-Resistant Influenza and 2009 A (H1N1) Viruses. Antimicrob. Agents Chemother. 2010, 54, 2517–2524. [Google Scholar] [CrossRef]
- Chen, T.; Fei, C.Y.; Chen, Y.P.; Sargsyan, K.; Chang, C.P.; Yuan, H.S.; Lim, C. Synergistic Inhibition of SARS-CoV-2 Replication Using Disulfiram/Ebselen and Remdesivir. ACS Pharmacol. Transl. Sci. 2021, 4, 898–907. [Google Scholar] [CrossRef] [PubMed]
- Tan, Y.L.; Tan, K.S.; Chu, J.J.H.; Chow, V.T. Combination Treatment with Remdesivir and Ivermectin Exerts Highly Synergistic and Potent Antiviral Activity against Murine Coronavirus Infection. Front. Cell. Infect. Microbiol. 2021, 11, 700502. [Google Scholar] [CrossRef] [PubMed]
- Bobrowski, T.; Chen, L.; Eastman, R.T.; Itkin, Z.; Shinn, P.; Chen, C.Z.; Guo, H.; Zheng, W.; Michael, S.; Simeonov, A.; et al. Synergistic and Antagonistic Drug Combinations against SARS-CoV-2. Mol. Ther. 2021, 29, 873–885. [Google Scholar] [CrossRef]
- Jitobaom, K.; Boonarkart, C.; Manopwisedjaroen, S.; Punyadee, N.; Borwornpinyo, S.; Thitithanyanont, A.; Auewarakul, P. Synergistic anti-SARS-CoV-2 activity of repurposed anti-parasitic drug combinations. BMC Pharmacol. Toxicol. 2022, 23, 41. [Google Scholar] [CrossRef] [PubMed]
- Hempel, T.; Elez, K.; Krüger, N.; Raich, L.; Shrimp, J.H.; Danov, O.; Noé, F. Synergistic inhibition of SARS-CoV-2 cell entry by otamixaban and covalent protease inhibitors: Pre-clinical assessment of pharmacological and molecular properties. Chem. Sci. 2021, 12, 12600–12609. [Google Scholar] [CrossRef] [PubMed]
- Schultz, D.C.; Johnson, R.M.; Ayyanathan, K.; Miller, J.; Whig, K.; Kamalia, B.; Cherry, S. Pyrimidine inhibitors synergize with nucleoside analogues to block SARS-CoV-2. Nature 2022, 604, 134–140. [Google Scholar] [CrossRef] [PubMed]
- Ohashi, H.; Watashi, K.; Saso, W.; Shionoya, K.; Iwanami, S.; Hirokawa, T.; Shirai, T.; Kanaya, S.; Ito, Y.; Kim, K.S.; et al. Potential anti-COVID-19 agents, cepharanthine and nelfinavir, and their usage for combination treatment. iScience 2021, 24, 102367. [Google Scholar] [CrossRef]
- Gao, Y.; Cao, J.; Xing, P.; Altmeyer, R.; Zhang, Y. Evaluation of small molecule combinations against respiratory syncytial virus in vitro. Molecules 2021, 26, 2607. [Google Scholar] [CrossRef] [PubMed]
- Song, D.; Lu, C.; Chang, C.; Ji, J.; Lin, L.; Liu, Y.; Chen, R. Natural binary herbal small molecules self-assembled nanogel for synergistic inhibition of respiratory syncytial virus. ACS Biomater. Sci. Eng. 2024, 10, 6648–6660. [Google Scholar] [CrossRef] [PubMed]
- Volonaki, E.; Psarras, S.; Xepapadaki, P.; Psomali, D.; Gourgiotis, D.; Papadopoulos, N.G. Synergistic effects of fluticasone propionate and salmeterol on inhibiting rhinovirus-induced epithelial production of remodelling-associated growth factors. Clin. Exp. Allergy 2006, 36, 1268–1273. [Google Scholar] [CrossRef]
- Wang, Y.; Li, G.; Yuan, S.; Gao, Q.; Lan, K.; Altmeyer, R.; Zou, G. In vitro assessment of combinations of enterovirus inhibitors against enterovirus 71. Antimicrob. Agents Chemother. 2016, 60, 5357–5367. [Google Scholar] [CrossRef]
- Ianevski, A.; Zusinaite, E.; Tenson, T.; Oksenych, V.; Wang, W.; Afset, J.E.; Bjørås, M.; Kainov, D.E. Novel synergistic anti-enteroviral drug combinations. Viruses 2022, 14, 1866. [Google Scholar] [CrossRef]
- Hung, H.C.; Wang, H.C.; Shih, S.R.; Teng, I.F.; Tseng, C.P.; Hsu, J.T.A. Synergistic inhibition of enterovirus 71 replication by interferon and rupintrivir. J. Infect. Dis. 2011, 203, 1784–1790. [Google Scholar] [CrossRef] [PubMed]
- Kang, H.; Kim, C.; Kim, D.E.; Song, J.H.; Choi, M.; Choi, K.; Cho, S. Synergistic antiviral activity of gemcitabine and ribavirin against enteroviruses. Antivir. Res. 2015, 124, 1–10. [Google Scholar] [CrossRef]
| Dry Powder Inhaler | Pressurized Metered Dose Inhaler | Nebulizer | |
|---|---|---|---|
| Medication type | Powder | Liquid | Liquid |
| Physical and chemical stability | More | Less than powder | Less than powder |
| Activation | Breath-activated | Pressurized canister | Through compressed gas/electricity |
| Inhalation and device coordination | Required | Required | Not required |
| Portability | Highly portable | Highly portable | Less portable and requires set up |
| Maintenance | Minimal | Requires cleaning of inhaler and canister | Requires cleaning of nebulizers and replacement of nebulizer parts if needed |
| High-dose delivery | Yes | No | Yes |
| Transmission | No/Less | No/Less | High |
| Product | Active Ingredient | Reported Excipients | Indication | Dose per Day | Manufacturer |
|---|---|---|---|---|---|
| Relenza Diskhaler | Zanamivir | Lactose monohydrate | Influenza | 5 mg/twice per day | Galxo SmithKline, UK |
| Inavir | Laninamivir octanoate | Lactose monohydrate | Influenza | 40 mg/once a day | Daiichi Sankyo, Japan |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Saha, T.; Masum, Z.U.; Biswas, A.; Mou, M.A.; Ahmed, S.; Saha, T. Inhaled Dry Powder of Antiviral Agents: A Promising Approach to Treating Respiratory Viral Pathogens. Viruses 2025, 17, 252. https://doi.org/10.3390/v17020252
Saha T, Masum ZU, Biswas A, Mou MA, Ahmed S, Saha T. Inhaled Dry Powder of Antiviral Agents: A Promising Approach to Treating Respiratory Viral Pathogens. Viruses. 2025; 17(2):252. https://doi.org/10.3390/v17020252
Chicago/Turabian StyleSaha, Tushar, Zia Uddin Masum, Anik Biswas, Moushumi Afroza Mou, Sohag Ahmed, and Tamal Saha. 2025. "Inhaled Dry Powder of Antiviral Agents: A Promising Approach to Treating Respiratory Viral Pathogens" Viruses 17, no. 2: 252. https://doi.org/10.3390/v17020252
APA StyleSaha, T., Masum, Z. U., Biswas, A., Mou, M. A., Ahmed, S., & Saha, T. (2025). Inhaled Dry Powder of Antiviral Agents: A Promising Approach to Treating Respiratory Viral Pathogens. Viruses, 17(2), 252. https://doi.org/10.3390/v17020252





