DRAVP: A Comprehensive Database of Antiviral Peptides and Proteins
Abstract
1. Introduction
2. Materials and Methods
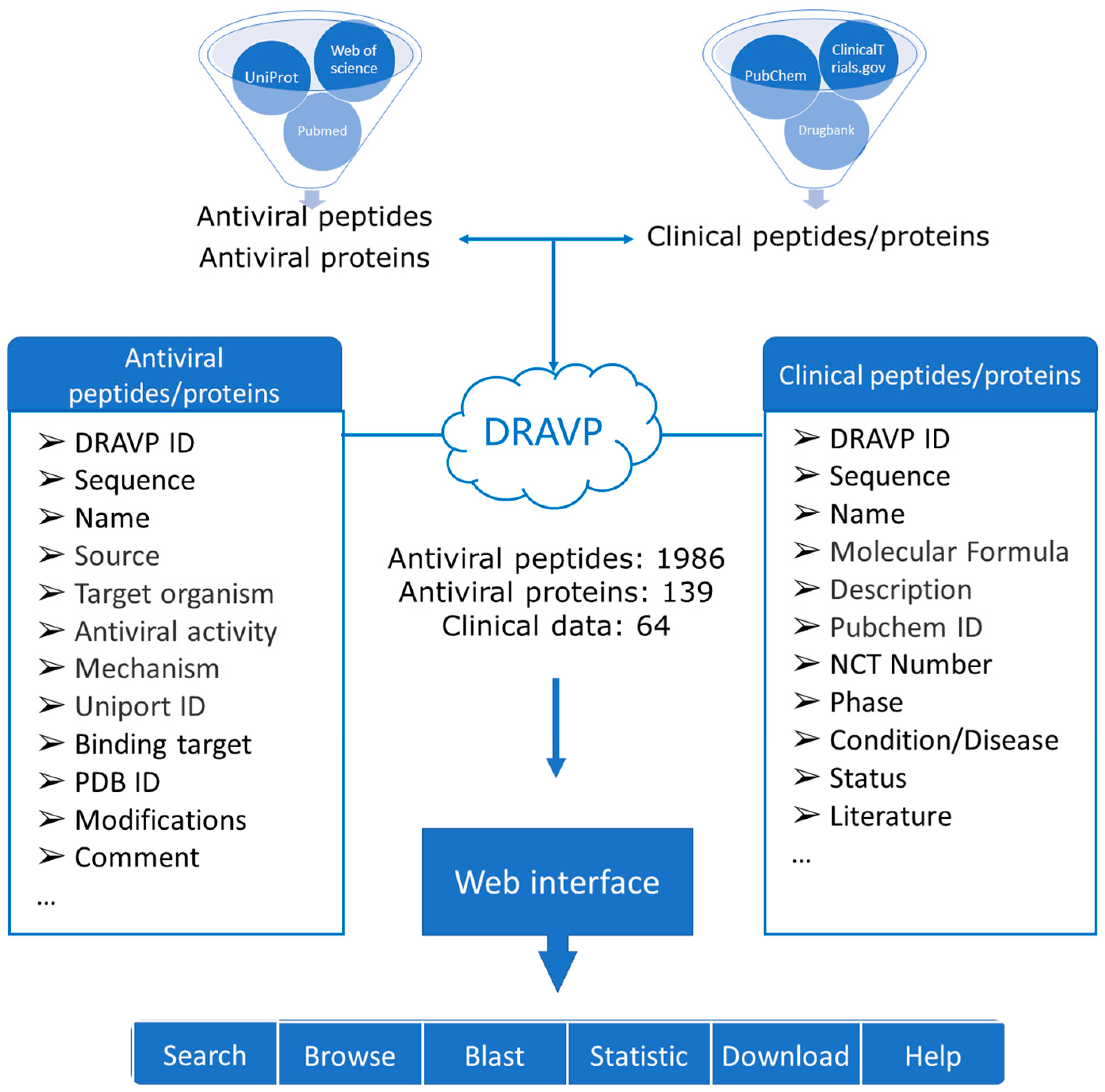
2.1. Data Collection and Extraction
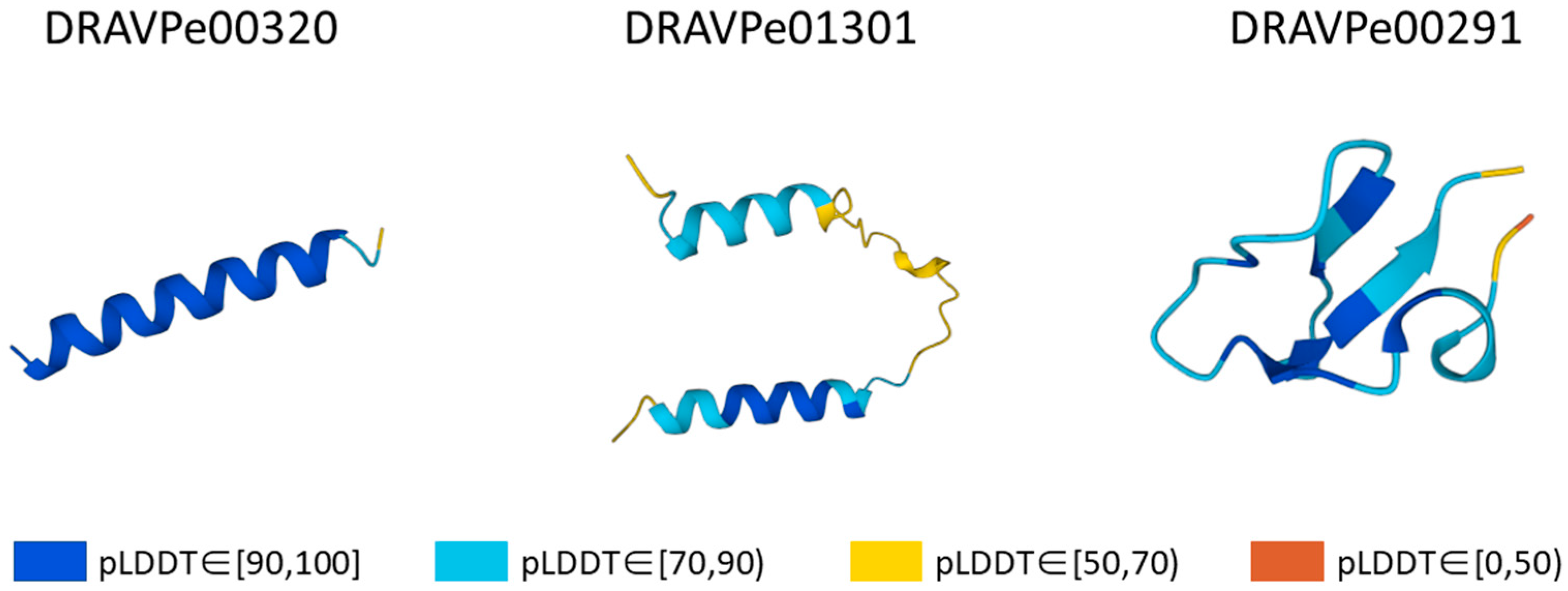
2.2. Structure Prediction and Evaluation
2.3. Database Construction
3. Results and Discussion
3.1. Database
- (i)
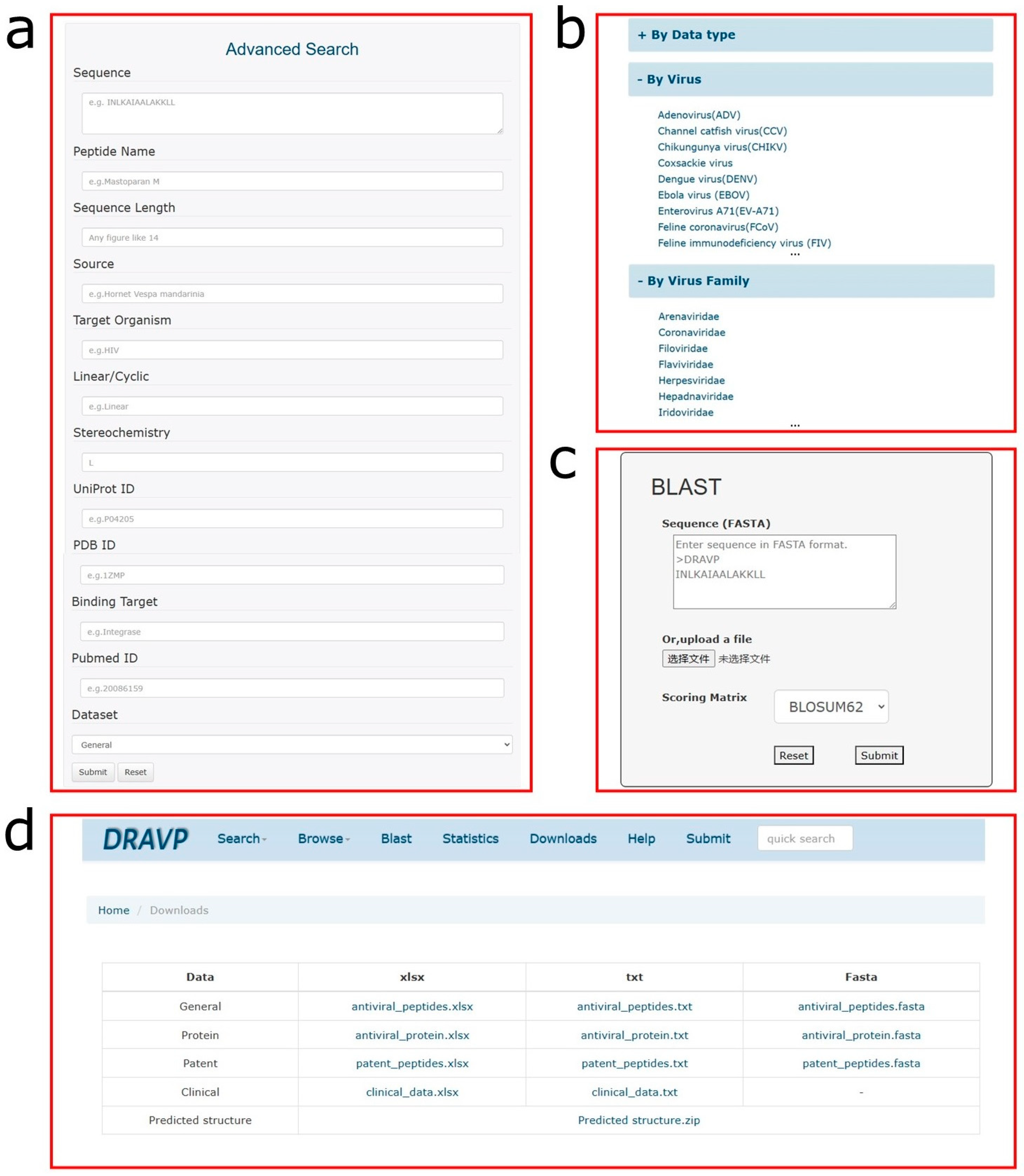
- Search page: This page can help users to more conveniently fetch the desired data. To meet different needs, a simple search and advanced search for data retrieval were set up. The simple search allows users to retrieve data from a specific field from the dropdown menu. The advanced search is carried out through a combination of multiple fields, such as sequence, target virus, or UniProt ID, to achieve more accurate data retrieval.
- (ii)
- Browse page: The database provides three cross-linked browse tables based on different criteria: (1) classification based on data type, including antiviral peptides, antiviral proteins, patent data, and clinical data; (2) classification based on targeted virus type, with a total of 47 different virus types; and (3) classification based on virus family, such as coronaviridae, flaviviridae, and retroviridae. Some basic information, such as DRAVP ID, peptide name, source, and target organism, is provided in the overview table.
- (iii)
- Blast: To facilitate the sequence analysis in database, the Blast [43] search tool was integrated into DRAVP. A user can submit sequences in the FASTA format and choose the score matrix to obtain the sequence alignment results, which contain the score, E value, identities, positives, and gaps. The sequence alignment is shown for the peptides or proteins found to be identical or similar in the database.
- (iv)
- Download page: All the data and website source code are freely available for users. Three types of data files are provided on this page to satisfy different requirements. Meanwhile, the predicted structure and installed package of the Blast software are also collected on the page for download.
3.2. Database Content
3.3. Structure Verification
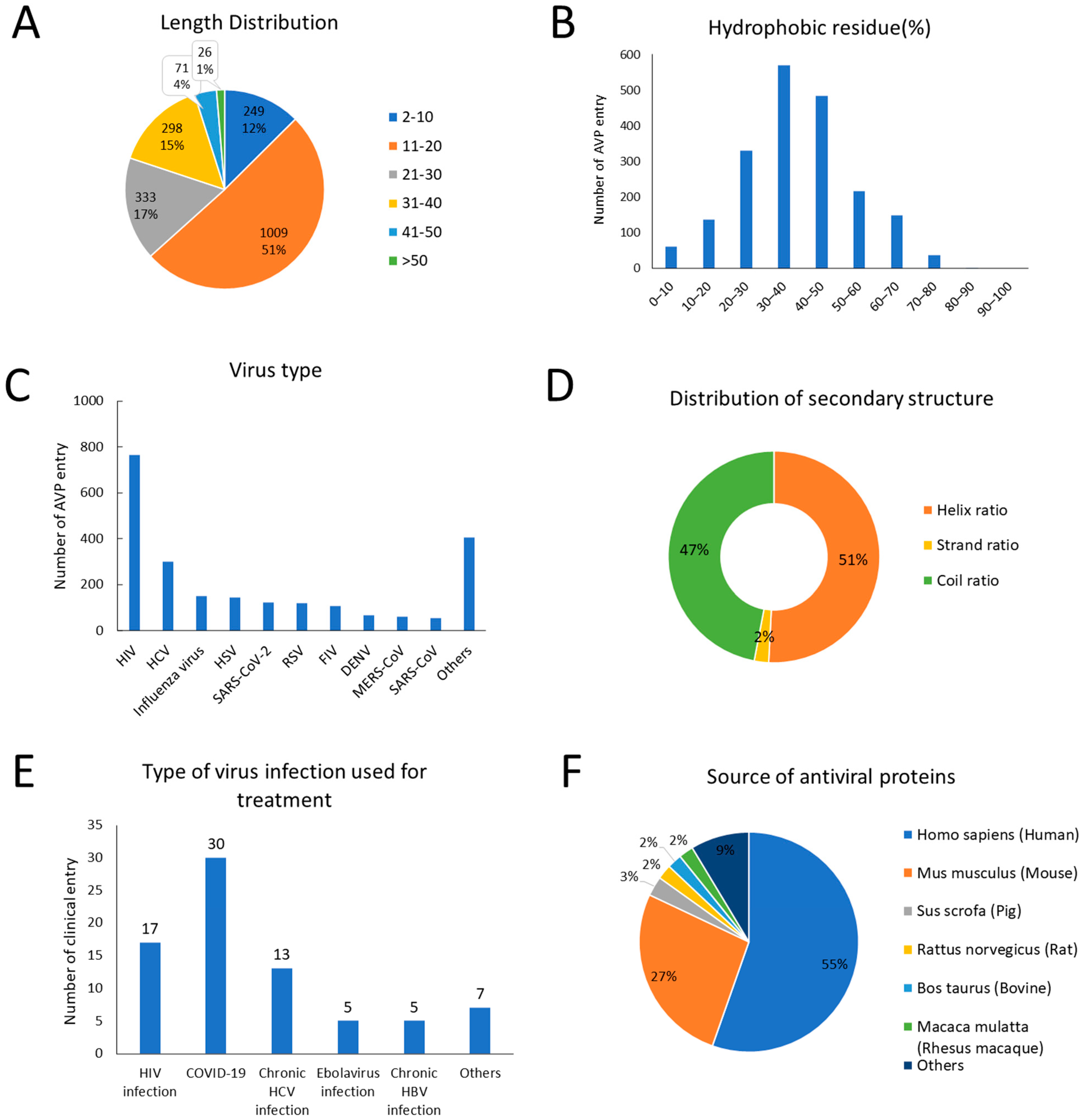
3.4. Statistics
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Marston, B.J.; Dokubo, E.K.; van Steelandt, A.; Martel, L.; Williams, D.; Hersey, S.; Jambai, A.; Keita, S.; Nyenswah, T.G.; Redd, J.T. Ebola Response Impact on Public Health Programs, West Africa, 2014–2017. Emerg. Infect. Dis. 2017, 23, S25–S32. [Google Scholar] [CrossRef]
- Pardy, R.D.; Richer, M.J. Zika Virus Pathogenesis: From Early Case Reports to Epidemics. Viruses 2019, 11, 886. [Google Scholar] [CrossRef]
- Zhong, N.S.; Zheng, B.J.; Li, Y.M.; Poon, L.L.M.; Xie, Z.H.; Chan, K.H.; Li, P.H.; Tan, S.Y.; Chang, Q.; Xie, J.P.; et al. Epidemiology and cause of severe acute respiratory syndrome (SARS) in Guangdong, People’s Republic of China, in February, 2003. Lancet 2003, 362, 1353–1358. [Google Scholar] [CrossRef] [PubMed]
- The Lancet. MERS-CoV: A global challenge. Lancet 2013, 381, 1960. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.; Tian, E.K.; He, B.; Tian, L.; Han, R.; Wang, S.; Xiang, Q.; Zhang, S.; El Arnaout, T.; Cheng, W. Overview of lethal human coronaviruses. Signal. Transduct. Target. Ther. 2020, 5, 89. [Google Scholar] [CrossRef]
- Lou, Z.; Sun, Y.; Rao, Z. Current progress in antiviral strategies. Trends Pharmacol. Sci. 2014, 35, 86–102. [Google Scholar] [CrossRef] [PubMed]
- Heydari, H.; Golmohammadi, R.; Mirnejad, R.; Tebyanian, H.; Fasihi-Ramandi, M.; Moosazadeh Moghaddam, M. Antiviral peptides against Coronaviridae family: A review. Peptides 2021, 139, 170526. [Google Scholar] [CrossRef]
- Agarwal, G.; Gabrani, R. Antiviral Peptides: Identification and Validation. Int. J. Pept. Res. Ther. 2021, 27, 149–168. [Google Scholar] [CrossRef]
- Tonk, M.; Růžek, D.; Vilcinskas, A. Compelling Evidence for the Activity of Antiviral Peptides against SARS-CoV-2. Viruses 2021, 13, 912. [Google Scholar] [CrossRef]
- Wack, A.; Openshaw, P.; O’Garra, A. Contribution of cytokines to pathology and protection in virus infection. Curr. Opin. Virol. 2011, 1, 184–195. [Google Scholar] [CrossRef]
- Breen, E.C. Pro- and anti-inflammatory cytokines in human immunodeficiency virus infection and acquired immunodeficiency syndrome. Pharmacol. Ther. 2002, 95, 295–304. [Google Scholar] [CrossRef] [PubMed]
- Pang, T. Cytokines in infection. Med. J. Malays. 1993, 48, 9–11. [Google Scholar]
- Poynard, T.; Marcellin, P.; Lee, S.S.; Niederau, C.; Minuk, G.S.; Ideo, G.; Bain, V.; Heathcote, J.; Zeuzem, S.; Trepo, C.; et al. Randomised trial of interferon alpha2b plus ribavirin for 48 weeks or for 24 weeks versus interferon alpha2b plus placebo for 48 weeks for treatment of chronic infection with hepatitis C virus. International Hepatitis Interventional Therapy Group (IHIT). Lancet 1998, 352, 1426–1432. [Google Scholar] [CrossRef] [PubMed]
- Wyde, P.R.; Moore, D.K.; Pimentel, D.M.; Gilbert, B.E.; Nimrod, R.; Panet, A. Recombinant superoxide dismutase (SOD) administered by aerosol inhibits respiratory syncytial virus infection in cotton rats. Antivir. Res. 1996, 31, 173–184. [Google Scholar] [CrossRef]
- Stravalaci, M.; Pagani, I.; Paraboschi, E.M.; Pedotti, M.; Doni, A.; Scavello, F.; Mapelli, S.N.; Sironi, M.; Perucchini, C.; Varani, L.; et al. Recognition and inhibition of SARS-CoV-2 by humoral innate immunity pattern recognition molecules. Nat. Immunol. 2022, 23, 275–286. [Google Scholar] [CrossRef] [PubMed]
- Rani, P.; Kapoor, B.; Gulati, M.; Atanasov, A.G.; Alzahrani, Q.; Gupta, R. Antimicrobial peptides: A plausible approach for COVID-19 treatment. Expert. Opin. Drug. Discov. 2022, 17, 473–487. [Google Scholar] [CrossRef]
- Feng, M.; Fei, S.; Xia, J.; Labropoulou, V.; Swevers, L.; Sun, J. Antimicrobial Peptides as Potential Antiviral Factors in Insect Antiviral Immune Response. Front. Immunol. 2020, 11, 2030. [Google Scholar] [CrossRef]
- Zakaryan, H.; Chilingaryan, G.; Arabyan, E.; Serobian, A.; Wang, G. Natural antimicrobial peptides as a source of new antiviral agents. J. Gen. Virol. 2021, 102, 001661. [Google Scholar] [CrossRef]
- Vilas Boas, L.C.P.; Campos, M.L.; Berlanda, R.L.A.; de Carvalho Neves, N.; Franco, O.L. Antiviral peptides as promising therapeutic drugs. Cell. Mol. Life Sci. 2019, 76, 3525–3542. [Google Scholar] [CrossRef]
- Zhou, J.; Xu, W.; Liu, Z.; Wang, C.; Xia, S.; Lan, Q.; Cai, Y.; Su, S.; Pu, J.; Xing, L.; et al. A highly potent and stable pan-coronavirus fusion inhibitor as a candidate prophylactic and therapeutic for COVID-19 and other coronavirus diseases. Acta Pharm. Sin. B 2022, 12, 1652–1661. [Google Scholar] [CrossRef]
- Xia, S.; Chan, J.F.; Wang, L.; Jiao, F.; Chik, K.K.; Chu, H.; Lan, Q.; Xu, W.; Wang, Q.; Wang, C.; et al. Peptide-based pan-CoV fusion inhibitors maintain high potency against SARS-CoV-2 Omicron variant. Cell Res. 2022, 32, 404–406. [Google Scholar] [CrossRef] [PubMed]
- Dando, T.M.; Perry, C.M. Enfuvirtide. Drugs 2003, 63, 2755–2766. [Google Scholar] [CrossRef] [PubMed]
- Ahmadi, K.; Farasat, A.; Rostamian, M.; Johari, B.; Madanchi, H. Enfuvirtide, an HIV-1 fusion inhibitor peptide, can act as a potent SARS-CoV-2 fusion inhibitor: An in silico drug repurposing study. J. Biomol. Struct. Dyn. 2022, 40, 5566–5576. [Google Scholar] [CrossRef]
- Wilby, K.J.; Partovi, N.; Ford, J.A.; Greanya, E.; Yoshida, E.M. Review of boceprevir and telaprevir for the treatment of chronic hepatitis C. Can. J. Gastroenterol. 2012, 26, 205–210. [Google Scholar] [CrossRef]
- Mousavi Maleki, M.S.; Sardari, S.; Ghandehari Alavijeh, A.; Madanchi, H. Recent Patents and FDA-Approved Drugs Based on Antiviral Peptides and Other Peptide-Related Antivirals. Int. J. Pept. Res. Ther. 2023, 29, 5. [Google Scholar] [CrossRef]
- Mousavi Maleki, M.S.; Rostamian, M.; Madanchi, H. Antimicrobial peptides and other peptide-like therapeutics as promising candidates to combat SARS-CoV-2. Expert. Rev. Anti Infect. Ther. 2021, 19, 1205–1217. [Google Scholar] [CrossRef]
- Ali, W.; Elsahn, A.; Ting, D.S.J.; Dua, H.S.; Mohammed, I. Host Defence Peptides: A Potent Alternative to Combat Antimicrobial Resistance in the Era of the COVID-19 Pandemic. Antibiotics 2022, 11, 475. [Google Scholar] [CrossRef]
- Li, J.; Pu, Y.; Tang, J.; Zou, Q.; Guo, F. DeepAVP: A Dual-Channel Deep Neural Network for Identifying Variable-Length Antiviral Peptides. IEEE J. Biomed. Health Inform. 2020, 24, 3012–3019. [Google Scholar] [CrossRef] [PubMed]
- Pang, Y.; Yao, L.; Jhong, J.H.; Wang, Z.; Lee, T.Y. AVPIden: A new scheme for identification and functional prediction of antiviral peptides based on machine learning approaches. Brief. Bioinform. 2021, 22, bbab263. [Google Scholar] [CrossRef]
- Pirtskhalava, M.; Amstrong, A.A.; Grigolava, M.; Chubinidze, M.; Alimbarashvili, E.; Vishnepolsky, B.; Gabrielian, A.; Rosenthal, A.; Hurt, D.E.; Tartakovsky, M. DBAASP v3: Database of antimicrobial/cytotoxic activity and structure of peptides as a resource for development of new therapeutics. Nucleic Acids Res. 2021, 49, D288–D297. [Google Scholar] [CrossRef]
- Shi, G.; Kang, X.; Dong, F.; Liu, Y.; Zhu, N.; Hu, Y.; Xu, H.; Lao, X.; Zheng, H. DRAMP 3.0: An enhanced comprehensive data repository of antimicrobial peptides. Nucleic Acids Res. 2022, 50, D488–D496. [Google Scholar] [CrossRef]
- Wang, G.; Li, X.; Wang, Z. APD3: The antimicrobial peptide database as a tool for research and education. Nucleic Acids Res. 2016, 44, D1087–D1093. [Google Scholar] [CrossRef] [PubMed]
- Qureshi, A.; Thakur, N.; Kumar, M. HIPdb: A database of experimentally validated HIV inhibiting peptides. PLoS ONE 2013, 8, e54908. [Google Scholar] [CrossRef]
- Zhang, Q.; Chen, X.; Li, B.; Lu, C.; Yang, S.; Long, J.; Chen, H.; Huang, J.; He, B. A database of anti-coronavirus peptides. Sci. Data 2022, 9, 294. [Google Scholar] [CrossRef]
- Qureshi, A.; Thakur, N.; Tandon, H.; Kumar, M. AVPdb: A database of experimentally validated antiviral peptides targeting medically important viruses. Nucleic Acids Res. 2014, 42, D1147–D1153. [Google Scholar] [CrossRef] [PubMed]
- The UniProt Consortium. UniProt: The universal protein knowledgebase in 2021. Nucleic Acids Res. 2021, 49, D480–D489. [Google Scholar] [CrossRef]
- Hammami, R.; Zouhir, A.; Naghmouchi, K.; Ben Hamida, J.; Fliss, I. SciDBMaker: New software for computer-aided design of specialized biological databases. BMC Bioinform. 2008, 9, 121. [Google Scholar] [CrossRef] [PubMed]
- Burley, S.K.; Berman, H.M.; Kleywegt, G.J.; Markley, J.L.; Nakamura, H.; Velankar, S. Protein Data Bank (PDB): The Single Global Macromolecular Structure Archive. Methods Mol. Biol. 2017, 1607, 627–641. [Google Scholar] [CrossRef] [PubMed]
- Jumper, J.; Evans, R.; Pritzel, A.; Green, T.; Figurnov, M.; Ronneberger, O.; Tunyasuvunakool, K.; Bates, R.; Žídek, A.; Potapenko, A.; et al. Highly accurate protein structure prediction with AlphaFold. Nature 2021, 596, 583–589. [Google Scholar] [CrossRef] [PubMed]
- Sehnal, D.; Bittrich, S.; Deshpande, M.; Svobodová, R.; Berka, K.; Bazgier, V.; Velankar, S.; Burley, S.K.; Koča, J.; Rose, A.S. Mol* Viewer: Modern web app for 3D visualization and analysis of large biomolecular structures. Nucleic Acids Res. 2021, 49, W431–W437. [Google Scholar] [CrossRef]
- Hooft, R.W.; Vriend, G.; Sander, C.; Abola, E.E. Errors in protein structures. Nature 1996, 381, 272. [Google Scholar] [CrossRef] [PubMed]
- Laskowski, R.A.; Rullmannn, J.A.; MacArthur, M.W.; Kaptein, R.; Thornton, J.M. AQUA and PROCHECK-NMR: Programs for checking the quality of protein structures solved by NMR. J. Biomol. NMR 1996, 8, 477–486. [Google Scholar] [CrossRef]
- Camacho, C.; Coulouris, G.; Avagyan, V.; Ma, N.; Papadopoulos, J.; Bealer, K.; Madden, T.L. BLAST+: Architecture and applications. BMC Bioinform. 2009, 10, 421. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Chen, J.; Cheng, T.; Gindulyte, A.; He, J.; He, S.; Li, Q.; Shoemaker, B.A.; Thiessen, P.A.; Yu, B.; et al. PubChem in 2021: New data content and improved web interfaces. Nucleic Acids Res. 2021, 49, D1388–D1395. [Google Scholar] [CrossRef]
- Wishart, D.S.; Feunang, Y.D.; Guo, A.C.; Lo, E.J.; Marcu, A.; Grant, J.R.; Sajed, T.; Johnson, D.; Li, C.; Sayeeda, Z.; et al. DrugBank 5.0: A major update to the DrugBank database for 2018. Nucleic Acids Res. 2018, 46, D1074–D1082. [Google Scholar] [CrossRef]
- Mendez, D.; Gaulton, A.; Bento, A.P.; Chambers, J.; De Veij, M.; Félix, E.; Magariños, M.P.; Mosquera, J.F.; Mutowo, P.; Nowotka, M.; et al. ChEMBL: Towards direct deposition of bioassay data. Nucleic Acids Res. 2019, 47, D930–D940. [Google Scholar] [CrossRef]
- McDonald, E.F.; Jones, T.; Plate, L.; Meiler, J.; Gulsevin, A. Benchmarking AlphaFold2 on peptide structure prediction. Structure 2023, 31, 111–119.e112. [Google Scholar] [CrossRef]
- Cao, L.; Goreshnik, I.; Coventry, B.; Case, J.B.; Miller, L.; Kozodoy, L.; Chen, R.E.; Carter, L.; Walls, A.C.; Park, Y.J.; et al. De novo design of picomolar SARS-CoV-2 miniprotein inhibitors. Science 2020, 370, 426–431. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Chong, H.; Yu, D.; Guo, Y.; Zhou, Y.; He, Y. Design and Characterization of Cholesterylated Peptide HIV-1/2 Fusion Inhibitors with Extremely Potent and Long-Lasting Antiviral Activity. J. Virol. 2019, 93, e02312-18. [Google Scholar] [CrossRef] [PubMed]
- Yin, P.; Zhang, L.; Ye, F.; Deng, Y.; Lu, S.; Li, Y.P.; Zhang, L.; Tan, W. A screen for inhibitory peptides of hepatitis C virus identifies a novel entry inhibitor targeting E1 and E2. Sci. Rep. 2017, 7, 3976. [Google Scholar] [CrossRef]
- Schmidt, A.G.; Yang, P.L.; Harrison, S.C. Peptide inhibitors of flavivirus entry derived from the E protein stem. J. Virol. 2010, 84, 12549–12554. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Cooper, C.L.; Wang, G.; Morwitzer, M.J.; Kota, K.; Tran, J.P.; Bradfute, S.B.; Liu, Y.; Shao, J.; Zhang, A.K.; et al. Engineered Human Cathelicidin Antimicrobial Peptides Inhibit Ebola Virus Infection. iScience 2020, 23, 100999. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Z.; Zhang, R.; Hong, W.; Cheng, Y.; Wang, H.; Lang, Y.; Ji, Z.; Wu, Y.; Li, W.; Xie, Y.; et al. Histidine-rich Modification of a Scorpion-derived Peptide Improves Bioavailability and Inhibitory Activity against HSV-1. Theranostics 2018, 8, 199–211. [Google Scholar] [CrossRef] [PubMed]
- Ho, T.Y.; Wu, S.L.; Chen, J.C.; Wei, Y.C.; Cheng, S.E.; Chang, Y.H.; Liu, H.J.; Hsiang, C.Y. Design and biological activities of novel inhibitory peptides for SARS-CoV spike protein and angiotensin-converting enzyme 2 interaction. Antivir. Res. 2006, 69, 70–76. [Google Scholar] [CrossRef]
- Xia, S.; Liu, M.; Wang, C.; Xu, W.; Lan, Q.; Feng, S.; Qi, F.; Bao, L.; Du, L.; Liu, S.; et al. Inhibition of SARS-CoV-2 (previously 2019-nCoV) infection by a highly potent pan-coronavirus fusion inhibitor targeting its spike protein that harbors a high capacity to mediate membrane fusion. Cell Res. 2020, 30, 343–355. [Google Scholar] [CrossRef] [PubMed]
| Column | Fields | Description |
|---|---|---|
| General information | DRAVP ID | The unique ID linking to the corresponding DRAVP entry |
| Peptide name | Name of each peptide/protein | |
| Sequence | Amino acid composition of peptide/protein | |
| Sequence length | The length of peptide/protein sequence | |
| UniProt ID | The accessing link directing to external UniProt entry | |
| Source | The organism where the peptide/protein is extracted or isolated | |
| Activity information | Target organism | Type of targeted virus |
| Assay | Methods for determining the antiviral activity | |
| Activity | Antiviral activity information against viruses; contains EC50, IC50, etc. | |
| Hemolytic activity | Hemolytic activity against red blood cells | |
| Cytotoxicity | Cytotoxicity against cells except for RBCs | |
| Binding target | Peptide active action site | |
| Mechanism | Mechanism of peptides exhibit antiviral activity | |
| Structure information | PDB ID | Provide accessing link directing to the corresponding PDB entry |
| Predicted structure download | Provide the predicted structure file | |
| Linear/cyclic | Peptide linear or cyclic structure | |
| N/C-terminal modification | The modifications of N/C-terminal according to the references | |
| Other modification | All bonds and special amino acids | |
| Stereochemistry | The L/D amino acids consist of peptides | |
| Physicochemical information | Formula, mass, pI, net charge, absent amino acids, common amino acids, basic residues, acidic residues, hydrophobic residues, Boman index, hydrophobicity, aliphatic index, half-life, extinction coefficient cystines, absorbance 280 nm, and polar residues | |
| Literature information | Title, PubMed ID, journal information, and doi of reference articles | |
| Field | Description |
|---|---|
| DRAVP ID | The unique ID linking to the corresponding DRAVP entry |
| Name | Name of the entry |
| Sequence | Amino acid composition of peptide |
| Molecular formula | Molecular formula of the item |
| Condition/disease | Diseases used for treatment |
| Group | Clinical trials status |
| Type | Peptide, protein, or other types of drugs |
| PubChem ID | The accessing link directing to external PubChem entry |
| DrugBank accession number | The accessing number in DrugBank database |
| Description | Some information related to the entry, such as antiviral mechanism, structure description, etc. |
| Active sequence/structure | The chemical structure or modified sequence |
| CHEMBL ID | The accessing link directing to CHEMBL database |
| UNII | The accessing link directing to Global Substance Registration System |
| CAS | The accessing link directing to CAS common chemistry |
| Reference | The PubMed ID of reference articles |
| Clinical trials information | Including NCT number, study title, condition/disease, status, sponsor, and phase of clinical trials |
| DRAVP ID | PDB ID | Sequence Length | RMSD(Å) |
|---|---|---|---|
| DRAVPe00279 | 2ERI | 31 | 0.486 |
| DRAVPe01560 | 2LZI | 18 | 1.327 |
| DRAVPe00303 | 1ZMQ | 32 | 0.409 |
| DRAVPe01598 | 1ULL | 17 | 1.772 |
| DRAVPe00373 | 2JOS | 22 | 2.158 |
| DRAVPe00837 | 2MLT | 26 | 1.507 |
| DRAVPe01303 | 1ZMP | 32 | 0.519 |
| DRAVPe00295 | 1ZA8 | 31 | 0.680 |
| DRAVPe01958 | 1FD4 | 41 | 0.460 |
| DRAVPe00376 | 6GS5 | 13 | 2.305 |
| DRAVP ID | Whatcheck | Procheck | ||||
|---|---|---|---|---|---|---|
| Total Metrics | Error | Error Rate | Core Regions | Additional and Generous Allowed Regions | Disallowed Regions | |
| DRAVPe00279 | 45 | 5 | 11.11% | 80.0% | 20.0% | 0.0% |
| DRAVPe01560 | 37 | 6 | 16.22% | 92.9% | 7.1% | 0.0% |
| DRAVPe00303 | 45 | 5 | 11.11% | 100.0% | 0.0% | 0.0% |
| DRAVPe01598 | 39 | 5 | 12.82% | 100.0% | 0.0% | 0.0% |
| DRAVPe00373 | 43 | 4 | 9.30% | 100.0% | 0.0% | 0.0% |
| DRAVPe00837 | 42 | 4 | 9.52% | 100.0% | 0.0% | 0.0% |
| DRAVPe01303 | 42 | 5 | 11.90% | 96.3% | 3.7% | 0.0% |
| DRAVPe00295 | 43 | 5 | 11.63% | 88.9% | 11.1% | 0.0% |
| DRAVPe01958 | 46 | 5 | 10.87% | 96.7% | 0.0% | 3.3% |
| DRAVPe00376 | 41 | 4 | 9.76% | 100.0% | 0.0% | 0.0% |
| Average | 11.42% | 95.48% | 4.19% | 0.33% | ||
| DRAVP_ID | Virus | Target | Mechanism | IC50/EC50 | Reference |
|---|---|---|---|---|---|
| DRAVPe00482 | SARS-CoV-2 | S protein | Prevent viral entry by binding to the spike receptor-binding domain (RBD) and blocking it from binding to ACE2 | 23.54 pM | Cao et al. [48] |
| DRAVPe00788 | Human immunodeficiency virus (HIV-1 and HIV-2) | Envelope protein | Inhibit viral entry by binding to the prehairpin intermediate (PHI) conformation and competitively prevent its transition to 6-HB | 0.43 pM ~0.026 nM | Zhu et al. [49] |
| DRAVPe01747 | Hepatitis C virus (HCV) | Envelope protein | Inhibit viral entry at the postattachment step and block cell-to-cell transmission | 1–5 nM | Yin et al. [50] |
| DRAVPe01288 | Dengue virus (DENV1, DENV2, DENV3, DENV4) | - | Interact with the viral membrane bringing peptides into the low-pH endosome, where full exposure of the peptide site on the E conformational intermediate leads to the tight, specific binding that blocks membrane fusion | IC90 = 0.1–4 μM | Schmidt et al. [51] |
| DRAVPe01806 | Ebola virus (EBOV) | - | Impair cathepsin B-mediated processing of EBOV glycoprotein, thus preventing virus entry | 0.99 µM | Yu et al. [52] |
| DRAVPe00757 | Herpes simplex virus (HSV-1) | - | Inhibit the viral attachment and viral entry of HSV-1 | 0.67–2.88 µg/mL | Zeng et al. [53] |
| DRAVPe01977 | SARS-CoV | ACE2 | Inhibit viral entry by blocking the binding of S protein to ACE2 | 1.88 nM | Ho et al. [54] |
| DRAVPe00407 | MERS-CoV | S protein | Inhibit viral six-helical bundle (6-HB) formation, thereby preventing viral fusion and entry into host cells | 4.2 nM | Xia et al. [55] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, Y.; Zhu, Y.; Sun, X.; Ma, T.; Lao, X.; Zheng, H. DRAVP: A Comprehensive Database of Antiviral Peptides and Proteins. Viruses 2023, 15, 820. https://doi.org/10.3390/v15040820
Liu Y, Zhu Y, Sun X, Ma T, Lao X, Zheng H. DRAVP: A Comprehensive Database of Antiviral Peptides and Proteins. Viruses. 2023; 15(4):820. https://doi.org/10.3390/v15040820
Chicago/Turabian StyleLiu, Yanchao, Youzhuo Zhu, Xin Sun, Tianyue Ma, Xingzhen Lao, and Heng Zheng. 2023. "DRAVP: A Comprehensive Database of Antiviral Peptides and Proteins" Viruses 15, no. 4: 820. https://doi.org/10.3390/v15040820
APA StyleLiu, Y., Zhu, Y., Sun, X., Ma, T., Lao, X., & Zheng, H. (2023). DRAVP: A Comprehensive Database of Antiviral Peptides and Proteins. Viruses, 15(4), 820. https://doi.org/10.3390/v15040820






