First Expert Elicitation of Knowledge on Possible Drivers of Observed Increasing Human Cases of Tick-Borne Encephalitis in Europe
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Objective
2.2. Questionnaire Design
2.3. Expert Elicitation to Assess Drivers of Emergence or Increasing Incidence of TBE in Humans
2.4. Scoring, Weighting System, and Level of Uncertainty
2.5. Calculation of an Overall Weighted Score for Each Driver and the Ranking Process
2.6. Cluster Analysis
2.7. Sensitivity Analysis to Test the Robustness of the Expert Elicitation
3. Results
3.1. Response Rate and Field of Expertise Mobilized by the Experts
3.2. Estimating the Overall Weighted Score and Ranking of the Observed Emergence or Increasing Incidence of TBE in Humans
3.3. Cluster Analysis
3.4. Sensitivity Analysis of the Impact of Experts on the Final Ranking of the Observed Emergence or Increasing Incidence of the TBE in Humans
3.5. Level of Uncertainty Per Domain of Drivers
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Appendix A
| DOMAIN D1. DISEASE/PATHOGEN CHARACTERISTICS. | ||
| D1-1 | Current knowledge of the pathogen (e.g., transmission routes, incubation period depending of the transmission route, infectious dose, etc.). | |
| Score 0 | ||
| Score 1 | Very high: deep scientific knowledge on the pathogen, extensive scientific literature available on its biology (transmission mode, infectivity, etc.). | |
| Score 2 | High: detailed scientific knowledge on the pathogen but conflicting scientific results; some elements of the pathogen’s biology are still not elucidated. | |
| Score 3 | Moderate: limited scientific knowledge on the pathogen agent because it is still under characterization; pathogen recently discovered/isolated but belonging to a well-known and studied family of pathogens; the pathogen is characterized by multiple variants that are not characterized yet. | |
| Score 4 | Low: lack of scientific knowledge on the pathogen (multiplication, infectivity, incubation period, transmission mode, transmission route, etc.); pathogen agent recently discovered and emerging. | |
| D1-2 | The current domestic species able to excrete the virus into the milk. | |
| Score 0 | ||
| Score 1 | Low: only one species is involved. | |
| Score 2 | Medium: two species involved. | |
| Score 3 | High: three species involved. | |
| Score 4 | Very high: more than three species involved. | |
| D1-3 | Genetic variability with time of the infectious agent. | |
| Score 0 | Negligible: the infectious agent is genetically stable. | |
| Score 1 | Low: the genetic variability is low; therefore, it has a low effect in the (re-)emergence of the pathogen. | |
| Score 2 | Medium: the pathogen is considered to have a medium genetic variability. | |
| Score 3 | High: the pathogen is considered to have a high genetic variability. | |
| Score 4 | Very high: very high genetic variability (e.g., high mutation rate and re-assortment and recombination). | |
| D1-4 | Risk of showing no clinical signs and silent spread during infection and post-infection in humans. | |
| Score 0 | Null: silent spread is not a part of pathogen’s characteristics. | |
| Score 1 | Low: very short incubation period and signs of infections that are easily detected/recognised. | |
| Score 2 | Moderate: very short incubation period and signs of infection that are NOT easily detected/recognised. | |
| Score 3 | Medium: long incubation period, clinical signs that are not characteristic, and therefore specific diagnosis is necessary to detect infection. | |
| Score 4 | Very high: long incubation period. Disease/infection shows not clinical symptoms during the infectious period; chronic shedder. | |
| D1-5 | Existence of susceptible/ vulnerable groups (people at a higher risk of developing symptomatic or severe forms of the disease) in relation to its influence on the prevalence/incidence of the disease in your country. | |
| Score 0 | ||
| Score 1 | Negligible: no known susceptible groups. The disease affects all populations and the increasing prevalence/incidence of vulnerable groups (elderly nd immuno-suppressed people), has no effect on the (re-)emergence or increasing incidence of the disease in your country. | |
| Score 2 | Low: an increase in the prevalence/incidence of vulnerable groups (elderly and immuno-suppressed people) has a low effect on the (re-)emergence or increasing incidence of the disease in your country. | |
| Score 3 | Moderate some populations are at a higher risk of developing symptomatic forms of the disease and the increasing prevalence/incidence of vulnerable groups (elderly and immune suppressed people) have a moderate effect on the (re-)emergence or the increasing incidence of the disease in your country. | |
| Score 4 | High: some populations are at a higher risk of developing severe forms of the disease and the increasing prevalence/incidence of vulnerable groups (elderly and immune suppressed people) have a high effect on the (re-)emergence of the disease in your country. | |
| D1-6 | The number of wildlife compartments/groups (e.g., small mammals, birds, ungulates, etc.) playing roles as reservoirs or amplifying hosts for the pathogen and its potential spread. | |
| Score 0 | Null: no known wildlife reservoir. The pathogen has never been reported in wildlife species (i.e., possible detection but not necessary proof of reservoir or amplification host). | |
| Score 1 | Low: the pathogen has been reported in only one group of wildlife. | |
| Score 2 | Moderate: the pathogen has been reported in two groups of wildlife. | |
| Score 3 | High: the pathogen has been reported in at least three groups of wildlife. | |
| Score 4 | Very high: disease establishes itself in wildlife as a reservoir and is very hard to eradicate from wildlife. Livestock easily infected via wildlife contact; or the pathogen has never been investigated in wildlife species. | |
| D1-7 | Number of tick species vectors of the pathogen and their distribution in the country. | |
| Score 0 | Null: no known tick. | |
| Score 1 | Low: only one species of tick is present in the country but its role in the transmission is presumed low (has not been assessed to date) or has a limited distribution in the country. | |
| Score 2 | Medium: more than one species of tick are present in the country but their role in the transmission are presumed low (has not been assessed to date) or have a limited distribution in the country. | |
| Score 3 | High: only one competent tick is present and can carry and spread the disease and is found spread in most of the territory. | |
| Score 4 | Very high: more than one species of tick can carry and spread the disease and are found spread in most of the territory. | |
| D1-8 | Existence of other vectors, including mechanic vectors (e.g., mosquitoes, midges, and culicoides) and a potential spread. | |
| Score 0 | Null: no other known vector. | |
| Score 1 | Low: only one type of vector is present in the country but its role in the transmission is presumed low (has not been assessed to date). | |
| Score 2 | Moderate: more than one type of vector exists in the country but their role in the transmission is presumed low and has only been suspected as a source and spread of the disease. | |
| Score 3 | High: only one vector is present in the country and its role in transmission is presumed moderate to high. | |
| Score 4 | Very high: more than one type of vector can carry and spread the disease and are found spread in most of the territory. | |
| D1-9 | Transmission of the pathogen to humans. | |
| Score 0 | ||
| Score 1 | Low: humans are infected by direct close contact with other infected humans and by vertical transmission. | |
| Score 2 | Moderate: transmission by direct and indirect contacts only (e.g., through vehicles, foodborne, clothes, and instruments) or non-flying vector (e.g., ticks). | |
| Score 3 | High: vector transmission by flying vectors (e.g., culicoides and mosquitoes). | |
| Score 4 | Very high: more than three modes of transmission and/or airborne transmission. | |
| D1-10 | Environmental persistence. | |
| Score 0 | Null: pathogen does not survive in the environment. | |
| Score 1 | Low: only anecdotal isolation of the pathogen from the environment has been recorded. | |
| Score 2 | Moderate: the survival of the agent in the environment is limited (only temporary) and it is dependent on certain environmental conditions (e.g., humidity, temperature, rainfall, etc.). | |
| Score 3 | High: the survival of the agent in the environment is limited (only temporary) and NOT dependent on certain environmental conditions (e.g., humidity, temperature, rainfall, etc.). | |
| Score 4 | Very high: agent naturally surviving in the environment (soil and water) and organic materials is were it has a long-term survival. | |
| D1-11 | Transmission of the pathogen through food. | |
| Score 0 | ||
| Score 1 | Low: transmission through only one food category (e.g., raw milk). | |
| Score 2 | Moderate: transmission through two food categories (e.g., milk and cheese). | |
| Score 3 | High: transmission through three food categories (e.g., milk, cheese, and meats). | |
| Score 4 | Very high: transmission through more than three food categories. | |
| D1-12 | Virus persistence in food and/ or during food processing. | |
| Score 0 | Null: pathogen does not survive in dairy products. | |
| Score 1 | Low: only one anecdotal isolation of the pathogen from dairy products has been recorded. | |
| Score 2 | Moderate: the survival of the agent in the environment (matrix) is limited (only temporary) and it is dependent on certain environmental conditions, such as humidity, temperature, rainfall, etc. | |
| Score 3 | High: the survival of the agent in dairy products is limited and dependent on the physico-chemical characteristics of the product (e.g., pH and aw) and the food processing technology (e.g., pasteurization, sterilization, and ripening). | |
| Score 4 | Very high: agent naturally resistant to food processing technologies (long term-survival). | |
| Number of drivers = 12, hence 120 points to be distributed across this domain for the intra-domain weighing. | ||
| DOMAIN D2. DISTANCE TO EUROPE AND YOUR COUNTRY. | ||
| D2-1 | Current incidence (cases)/prevalence of the disease in the world, excluding Europe (the case of Europe is considered in the next criteria). | |
| Score 0 | ||
| Score 1 | Pathogen has been reported only in the countries of the Australasia (Australia, New Zealand, New Guinea, and neighbouring Pacific Islands) region. | |
| Score 2 | Disease was reported in countries of the Americas, the Caribbean, and Asia (excluding the Russian Federation). | |
| Score 3 | Disease was reported/present in the African continent. | |
| Score 4 | Disease was reported in countries of the Mediterranean Basin, Middle East, and the Russian Federation. | |
| D2-2 | European geographic proximity of the pathogen/disease to your country. | |
| Score 0 | ||
| Score 1 | Disease has never been present in Europe. | |
| Score 2 | Disease has been reported in Europe in the past but is currently exotic. | |
| Score 3 | Disease is currently present in at least one European country which is NOT bordering your country. | |
| Score 4 | Disease is currently present in at least one of the countries bordering your country. | |
| Score 5 | Disease reported in the country (human cases and/or infections in animals). | |
| D2-3 | To your knowledge when was the disease last reported in Europe. | |
| Score 0 | More than 20 years ago. | |
| Score 1 | More than 10 years ago. | |
| Score 2 | More than 5 years ago. | |
| Score 3 | More than 1 year ago. | |
| Score 4 | Currently present in Europe. | |
| Number of drivers = 3, hence 30 points to be distributed across this domain for the intra-domain weighing. | ||
| DOMAIN D3. ABILITY TO MONITOR, TREAT, AND CONTROL THE DISEASE. | ||
| D3-1 | Ability of preventive/control measures to stop the disease from entering the country or spreading (containment of the epidemic/pandemic), EXCLUDING treatment, vaccination, and vector(s)/reservoir(s) control. | |
| Score 0 | ||
| Score 1 | Very High: sanitary certificate; effective traceability of animals and by-products; effective disinfection measures; no contact between domestic and wild animals; and effective biosecurity measures. | |
| Score 2 | High: no sanitary certificate; effective traceability of animals and by-products; effective disinfection measures; limited or incomplete possibilities to restrict contacts between domestic and wild animals; and effective biosecurity measures. | |
| Score 3 | Low: no sanitary certificate; incomplete traceability of animals and by-products; ineffective disinfection measures; incomplete restriction of contacts between domestic and wild animals; incomplete restriction of wildlife movements; and ineffective biosecurity measures. | |
| Score 4 | Very low: no sanitary certificate; no traceability of animals and by-products; ineffective disinfection measures; impossibility to restrict spread by wild animals; and totally ineffective biosecurity measures. | |
| D3-2 | Vaccine availability for humans. | |
| Score 0 | ||
| Score 1 | Very high: commercialized vaccine available on a global scale (worldwide). | |
| Score 2 | High: local/mono-strain vaccine available at a regional/national scale (not systematically available for a global fight plan). | |
| Score 3 | Low: experimental vaccine, not commercialized to date; severe adverse reaction when applied; limited protector effect. | |
| Score 4 | Very low: absence; no vaccine available on the market for a use in the species considered in the study, no experimental vaccine either. | |
| D3-3 | Control of reservoir(s) and/or vector(s). | |
| Score 0 | Null: no vector-borne transmission and/or no reservoir(s) known to date. | |
| Score 1 | Very high: effective; limited reservoir(s) with limited geographical repartition, easy-to-identify; high scientific knowledge on vector(s)/reservoir(s); effective fighting measures. | |
| Score 2 | High: limited reservoir(s)/vector(s) with limited geographical repartition; easy-to-identify, high scientific knowledge on vector(s)/reservoir(s); effective fighting measures but NOT applicable at a large scale; limited fighting measures. | |
| Score 3 | Low: numerous reservoirs vectors identified with limited geographical repartition; hard to identify; lack of scientific knowledge on vector(s)/reservoir(s); fighting measures are not adequately effective—resistances and/or negative impact on environment. | |
| Score 4 | Very low: numerous vector(s)/reservoir(s)identified with wide geographic distribution; hard to identify, absence of scientific knowledge on vector(s)/reservoir(s); NO effective fighting measure against vector(s) (no active molecule, resistance to measures applied). | |
| D3-4 | Detection of pathogen circulation in humans—e.g., difficulties for the physicians to diagnose the disease or clinical signs that not so evident. | |
| Score 0 | ||
| Score 1 | Very high: disease is easily detected with pathognomonic clinical signs and physicians are aware of the disease and willing to notify it as soon as possible it. | |
| Score 2 | High: disease is easily detected by the clinical signs but physicians do not have sufficient knowledge/awareness nor interest to notify it or there is no central system of notification. | |
| Score 3 | Moderate: disease is not as easily detect by the clinical signs and physicians do not have sufficient knowledge/awareness nor interest to notify or there is no central system of notification. | |
| Score 4 | Low: humans do not show any pathognomonic clinical sign(s); physicians are reluctant to declare/notify or there is no central system of notification. | |
| D3-5 | Detection of pathogen circulation in domestic animals—e.g., difficulties for the farmer/veterinarian to report the disease or clinical signs that are not so evident. | |
| Score 0 | ||
| Score 1 | Very high: disease is easily detected with clinically signs and farmers are aware of the disease and willing to report it as soon as possible. | |
| Score 2 | High: disease is easily detected by the clinical signs but farmers do not have sufficient knowledge/awareness nor interest to report it. | |
| Score 3 | Moderate: disease is not as easily detect by the clinical signs and farmers do not have sufficient knowledge/awareness nor interest to report it. | |
| Score 4 | Low: the infected animal does not show any pathognomonic clinical sign(s); farmer is reluctant to declare/report any abnormality. | |
| D3-6 | Methods for detecting viral agents in human in your country. | |
| Score 0 | ||
| Score 1 | Very High: field tests are available and easy to use, with highly discriminating sensitivity and specificity (including infectivity tests). | |
| Score 2 | High: tests are used in local/regional laboratories but not in the field. | |
| Score 3 | Low: tests are only used in specialized laboratories/national reference laboratories. | |
| Score 4 | Very Low: there are no detection methods available to date. | |
| D3-7 | Methods for detecting viral agent in animals in your country. | |
| Score 0 | ||
| Score 1 | Very High: field tests are available and easy to use, with highly discriminating sensitivity and specificity (including infectivity tests). | |
| Score 2 | High: tests are used in local/regional laboratories but not in the field. | |
| Score 3 | Low: tests are only used in specialized laboratories/national reference laboratories. | |
| Score 4 | Very Low: there are no detection methods available to date. | |
| D3-8 | Methods for detecting viral agent in ticks in your country. | |
| Score 0 | ||
| Score 1 | Very High: field tests are available and easy to use, with highly discriminating sensitivity and specificity (including infectivity tests). | |
| Score 2 | High: tests are used in local/regional laboratories but not in the field. | |
| Score 3 | Low: tests are only used in specialized laboratories/national reference laboratories. | |
| Score 4 | Very Low: there are no detection methods available to date. | |
| D3-9 | Methods for detecting viral agent in food in your country. | |
| Score 0 | ||
| Score 1 | Very High: field tests are available and easy to use, with highly discriminating sensitivity and specificity (including infectivity tests). | |
| Score 2 | High: tests used in local/regional laboratories but not in the field. | |
| Score 3 | Low: tests are only used in specialized laboratories/national reference laboratories. | |
| Score 4 | Very Low: there are no detection methods available to date. | |
| D3-10 | Disease in humans that is currently under surveillance overseas (OMS and EU). | |
| Score 0 | ||
| Score 1 | Very high: generalized surveillance implemented by ALL EU Member States and the worldwide surveillance. | |
| Score 2 | High: surveillance of the pathogen in EU member states only. | |
| Score 3 | Low: surveillance only in some EU member states (because they had cases of the disease) and only in some NON-EU countries (not a disease reported in any international organisations). | |
| Score 4 | Very low: absence of surveillance of the pathogen in ALL EU member countries AND worldwide. | |
| D3-11 | Eradication experience in other countries and/or your country. | |
| Score 0 | ||
| Score 1 | Very high: previous experience on eradication has been applied rapidly and successfully. | |
| Score 2 | High: previous experience on eradicating the disease but with some setbacks in the process. | |
| Score 3 | Low: knowledge on eradication procedures but have never had to implement an eradication program in your country. | |
| Score 4 | Very low: it is an endemic disease with the impossibility of eradication or is a novel disease; first time countries are faced with a new disease to eradicate. | |
| Number of drivers = 11, hence 110 points to be distributed across this domain for the intra-domain weighing. | ||
| DOMAIN D4. Farm/European characteristics. | ||
| D4-1 | Type of farms / productions: dairy / beef (cattle) production, sheep or goat production, mono-species farms—one single farmed animal (e.g., only bovines) or multi-species farms (farms with more than one species, e.g., goats and bovines in the same farm/land/premises). | |
| Score 0 | ||
| Score 1 | Negligible: the type of farm does not influence human disease incidence in your country. | |
| Score 2 | Low: the type of farm has a low effect on human disease incidence in your country. | |
| Score 3 | Moderate: the type or types of farmed animals has a moderate effect on human disease incidence in your country. | |
| Score 4 | High: the type of farmed animals has a high influence on human disease incidence in your country. | |
| D4-2 | Animal density of farms. Extensive (small holders with a few animals) vs. intensive farming—effect on the human contamination through animal products (e.g., consumption of unpasteurised milk products). | |
| Score 0 | ||
| Score 1 | Negligible: animal farm density has a negligible effect on human disease incidence in your country. | |
| Score 2 | Low: farm density (extensive or intensive) of animals has a low effect on human disease incidence in your country. | |
| Score 3 | Moderate: farm density of animals in the farm (extensive v/s intensive) has a moderate effect on human disease incidence in your country. | |
| Score 4 | High: farm density of animals has a high effect on human disease incidence in your country. | |
| D4-3 | Farm management practices (off-ground farming, treatment, feeding practices, etc.) | |
| Score 0 | ||
| Score 1 | Negligible: management practices have a negligible effect on human disease incidence in your country. | |
| Score 2 | Low: management practices have a low effect on human disease incidence in your country. | |
| Score 3 | Moderate: management practices have a moderate effect on human disease incidence in your country. | |
| Score 4 | High: management practices have a high effect on human disease incidence in your country. | |
| D4-4 | The number of animals infected by TBEV in the herd (infection rate). | |
| Score 0 | ||
| Score 1 | Negligible: the number of animals infected by TBEV in farms have a negligible effect on human disease incidence in your country. | |
| Score 2 | Low: the number of animals infected by TBEV in farms have a low effect on human disease incidence in your country. | |
| Score 3 | Moderate: the number of animals infected by TBEV in farms have a moderate effect on human disease incidence in your country. | |
| Score 4 | High: the number of animals infected by TBEV in farms have a high effect on human disease incidence in your country. | |
| D4-5 | The rural(farm)-wildlife interface. | |
| Score 0 | ||
| Score 1 | Negligible: the disease has never (re-)emerged from the narrowing of the farm-wildlife interface. | |
| Score 2 | Low: the disease has a low probability to (re-)emerge via the livestock farm-forest interface. The disease has been known to (re-)emerge from the wild bush, but very rarely. | |
| Score 3 | Moderate: the disease has a moderate probability to occur via the farm/wildlife interface. Barriers (natural or artificial) are needed to keep the disease incidence in livestock. | |
| Score 4 | High: there is a high probability for the disease to (re-)emerge via the farm/forest interface. Barriers (natural or artificial) separating farms from natural forests are ineffective. | |
| Number of drivers = 5, hence 50 points to be distributed across this domain for the intra-domain weighing. | ||
| DOMAIN D5. GLOBAL CHANGES. | ||
| D5-1 | Influence of rainfall on the abundance of vector populations. | |
| Score 0 | ||
| Score 1 | Negligible: abundance of vector populations is not influenced by increased rainfall. | |
| Score 2 | Low: abundance of vector populations is slightly influenced by increased rainfall. | |
| Score 3 | Moderate: abundance of vector populations is moderately influenced by increased rainfall. | |
| Score 4 | High: abundance of vector populations is highly influenced by increased rainfall. | |
| D5-2 | Influence of humidity on the survival and transmission of the pathogen/disease. | |
| Score 0 | ||
| Score 1 | Negligible: abundance of vector populations is not influenced by increased humidity. | |
| Score 2 | Low: abundance of vector populations is slightly influenced by increased humidity. | |
| Score 3 | Moderate: abundance of vector populations is moderately influenced by increased humidity. | |
| Score 4 | High: abundance of vector populations is highly influenced by increased humidity. | |
| D5-3 | Influence of temperature on the survival and transmission of the pathogen/disease. | |
| Score 0 | ||
| Score 1 | Negligible: abundance of vector populations is not influenced by increased temperature. | |
| Score 2 | Low: abundance of vector populations is slightly influenced by increased temperature. | |
| Score 3 | Moderate: abundance of vector populations is moderately influenced by increased temperature. | |
| Score 4 | High: abundance of vector populations is highly influenced by increased temperature. | |
| D4-4 | Changes in landscape, e.g., landscape fragmentation, creation of barriers, and landfill sites. | |
| Score 0 | ||
| Score 1 | Negligible: changes in landscape have a negligible effect on the (re-)emergence/incidence of pathogen/disease. | |
| Score 2 | Low: changes in landscape have a low effect on the (re-)emergence/incidence of the disease/pathogen but need other factors (e.g., land use changes combined with higher winter temperatures). | |
| Score 3 | Moderate: landscape changes increases the density of reservoir hosts, availability of vectors, increases the pathogen’s survival, or increases the contact between humans and vectors. Furthermore, empty land can create a suitable environment for certain wildlife carrying the disease (e.g., migratory birds). | |
| Score 4 | High: landscape changes are one of the main drivers for the pathogen, its vectors, or the contact between humans and vectors. | |
| Number of drivers = 4, hence 40 points to be distributed across this domain for the intra-domain weighing. | ||
| DOMAIN D6. WILDLIFE. | ||
| D6-1 | Potential roles of zoos in the (re-)emergence of the pathogen or increasing human disease incidence. | |
| Score 0 | ||
| Score 1 | Negligible: the disease can be present in zoo animals but it is not known to have been transmitted from zoo animals to humans. | |
| Score 2 | Low: the disease can enter a zoo (e.g., with introduction of an infected exotic animal) but only accidental transmissions of the disease from zoo animals to human have been reported. Hence, zoos have a low effect on the (re-)emergence of the disease or increasing human disease incidence in the country. | |
| Score 3 | Moderate: the disease can enter a zoo and be present in zoo animals but it needs a vector (biological/mechanical) for its transmission to humans. Therefore, zoos have a moderate effect on the (re-)emergence of the disease or increasing human disease incidence in the country. | |
| Score 4 | High: the disease can be introduced into a zoo via an infected imported animal; zoo animals can carry the disease that can be easily transmissed to humans. | |
| D6-2 | Increase of autochthonous (indigenous animal) wild mammals in Europe and neighbouring countries. | |
| Score 0 | Not applicable: the disease has not been reported in wildlife. | |
| Score 1 | Negligible: the increase the autochthonous mammals population does not affect the risk of the human diseases to (re-)emerge. | |
| Score 2 | Low: a slight increase of autochthonous mammals can slightly increase the probably of the human disease emerging/incidence. | |
| Score 3 | Moderate: an increase of wild mammals has been associated with the re-emergence/incidence of the human disease. | |
| Score 4 | High: an increase of wild mammals is the only factor associated with outbreaks of the disease in humans. | |
| D6-3 | Increase in endemic/migrating populations of wild birds. | |
| Score 0 | Not applicable: wild/migrating birds are not a reservoir of the disease or responsible for the spread of the pathogen through vector transport. | |
| Score 1 | Negligible: there is a negligible probability of the disease (re-)emerging/increase of incidence in humans because of an increase in populations of endemic/migrating wild birds. | |
| Score 2 | Low: there is a low probability of the disease (re-)emerging/increase of incidence and spreading through increased populations of endemic/migrating wild birds. Disease has spread from the endemic/migrating wild birds accidentally or under exceptional circumstances. | |
| Score 3 | Moderate: there is a moderate probability of a disease being introduced and spread through increased populations of endemic/migrating wild birds. | |
| Score 4 | High: there is a high probability for a disease to (re-)emerge/increase of incidence through increased populations of wild/migrating birds. These are hosts or reservoirs of the disease. | |
| D6-4 | Hunting Activities: hunted animals can be brought back to where livestock is present. | |
| Score 0 | ||
| Score 1 | Negligible: the risk of the disease/pathogen (re-)emerging in livestock due to hunting activities is practically non-existent. | |
| Score 2 | Low: disease is present in hunted wildlife and birds and only accidental cases have been reported in livestock that have (re-)emerged because of hunting. The risk of the disease/pathogen (re-)emerging in livestock due to hunting activities is practically non-existent. | |
| Score 3 | Moderate: disease is present in hunted wildlife and birds but a certain control is established by the hunter. | |
| Score 4 | High: disease is present in hunted wildlife and birds and hunting is one of the main modes of transmitting disease to livestock. | |
| D6-5 | Transboundary movements of terrestrial wildlife from other countries. | |
| Score 0 | Not applicable: pathogen or infected vector is not carried by terrestrial wildlife. | |
| Score 1 | Negligible: (re-)emergence of disease or disease increasing by terrestrial movements of wildlife has only been suspected but never confirmed. | |
| Score 2 | Low: there is a low probability for the disease to (re-)emerge and spread through transboundary movements of terrestrial wildlife. | |
| Score 3 | Moderate: there is a moderate probability for the disease to (re-)emerge and spread through transboundary movements of terrestrial wildlife. | |
| Score 4 | High: there is a high probability for the disease to (re-)emerge and spread through transboundary movements of terrestrial wildlife. These are host and may spread/carry the disease along. | |
| Number of drivers = 5, hence 50 points to be distributed across this domain for the intra-domain weighing. | ||
| DOMAIN D7. HUMAN ACTIVITIES. | ||
| D7-1 | Changes in human behaviour / activities leading to more contact with the pathogen in high risk areas (recreational activities, forest activities, mushroom picking, etc.) | |
| Score 0 | ||
| Score 1 | Negligible: the changes of human behaviour are a negligible driver of the re-emergence/incidence of the disease in humans. | |
| Score 2 | Low: the changes of human behaviour are a low driver of the re-emergence/incidence of the disease in humans. | |
| Score 3 | Moderate: the changes of human behaviour are a moderate driver of the re-emergence/incidence of the disease in humans. | |
| Score 4 | High: the changes of human behaviour are a high driver of the re-emergence/incidence of the disease in humans. | |
| D7-2 | Could changes in eating habits or consumer demands lead to an increased exposure to the hazard through foods? | |
| Score 0 | ||
| Score 1 | Negligible: the changes in eating habits of consumers (e.g., demand for local and natural food such as raw milk cheese) are a negligible driver of the re-emergence/incidence of the disease. | |
| Score 2 | Low: the changes in eating habits of consumers (e.g., demand for local and natural food such as raw milk cheese) are a low driver of the re-emergence/incidence of the disease. | |
| Score 3 | Moderate: the changes in eating habits of consumers (e.g., demand for local and natural food such as raw milk cheese) are a moderate driver of the re-emergence/incidence of the disease. | |
| Score 4 | High: the changes in eating habits of consumers (e.g., demand for local and natural food such as raw milk cheese) are a high driver of the re-emergence/incidence of the disease. | |
| D7-3 | People’s movements linked to tourism (infection of tourists during a visit to an infected country). | |
| Score 0 | ||
| Score 1 | Negligible: tourism is a negligible driver on the emergence, re-emergence, or increasing human disease incidence in the country of interest. | |
| Score 2 | Low: tourism increase has a low driver of the (re-)emergence or increasing human disease incidence in the country of interest. | |
| Score 3 | Moderate: tourism increase has a moderate driver for the (re-)emergence or increasing human disease incidence in the country of interest. Biosecurity measures are enough to stop the entering of the pathogen. | |
| Score 4 | High: tourist movement is a high driver on the (re-)emergence or increasing human disease incidence in the country of interest. Tourists are highly likely to bring the disease into your country in their belongings; biosecurity measures are insufficient to stop the pathogen. | |
| D7-4 | Human immigration. | |
| Score 0 | ||
| Score 1 | Negligible: the immigration movements are a negligible driver of the disease (re-)emergence or increasing human disease incidence in the country of interest. | |
| Score 2 | Low: the immigration movements are a low driver of the disease (re-)emergence or increasing human disease incidence in the country of interest. | |
| Score 3 | Moderate: the immigration movement has a moderate effect as a driver on the (re-)emergence or increasing human disease incidence in the country of interest. Disease is highly likely to emerge using this route but biosecurity measures are enough to avoid (re-)emergence or increasing human disease incidence in the country of interest. | |
| Score 4 | High: the immigration movement has a high effect as a driver on the (re-)emergence or increasing human disease incidence in the country of interest. Disease is highly likely to emerge using this route as biosecurity measures are not enough to avoid (re-)emergence or increasing human disease incidence in the country of interest. | |
| D7-5 | Transport movements: more specifically, commercial flights and commercial transport by ships, cars, or military (EXCLUDING TRANSPORT VEHICLES OF LIVE ANIMALS). | |
| Score 0 | ||
| Score 1 | Negligible: the role of commercial movements as a driver on the (re-)emergence or increasing human disease incidence is negligible. | |
| Score 2 | Low: the role of commercial movements as a driver on the (re-)emergence or increasing human disease incidence is low. It is easily preventable by implementing biosecurity measures. | |
| Score 3 | Moderate: the role of commercial movements as a driver on the (re-)emergence or increasing human disease incidence is moderate. Disease can be prevented if biosecurity measures are tightened. | |
| Score 4 | High: the role of commercial movements as a driver on the (re-)emergence or increasing human disease incidence is high. Disease is hard to control with the current biosecurity measures. | |
| D7-6 | Transport vehicles of live animals. | |
| Score 0 | ||
| Score 1 | Negligible: the role of transport vehicles of live animals as a driver for the (re-)emergence or increasing human disease incidence is negligible. | |
| Score 2 | Low: the role of transport vehicles of live animals as a driver for the (re-)emergence or increasing human disease incidence is low. | |
| Score 3 | Moderate: the role of transport vehicles of live animals as a driver for (re-)emergence or increasing human disease incidence is moderate. | |
| Score 4 | High: the role of transport vehicles of live animals as a driver for (re-)emergence or increasing human disease incidence is high. | |
| D7-7 | Bioterrorism potential. | |
| Score 0 | ||
| Score 1 | Negligible: the role of bioterrorism as a driver for a disease to (re-)emerge is negligible: agent is available but difficult to handle or has a low potential of spread or generates few economic consequences. | |
| Score 2 | Low: the role of bioterrorism as a driver for a disease to (re-)emerge is low: agent is available and easy to handle by professionals and labs but has a low spread. | |
| Score 3 | Moderate: the role of bioterrorism as a driver for a disease to (re-)emerge is moderate: agent available and easy to handle by professionals, labs, and rapidly spreads. | |
| Score 4 | High: the role of bioterrorism as a driver for a disease to (re-)emerge is high. Agent is available and easy to handle by individuals and rapidly spreads. | |
| D7-8 | Inadvertent release of an exotic infectious agent from a containment facility, e.g., laboratory. | |
| Score 0 | ||
| Score 1 | Negligible: the pathogen is not currently present in any laboratory. | |
| Score 2 | Low: the pathogen is present in a containment facility but its release is very unlikely as it is very easily contained. | |
| Score 3 | Moderate: the pathogen is present in a containment facility and its release can occur as it is not easily contained. | |
| Score 4 | High: pathogen is handled in a risk 3 or 4 laboratory (BSL3 or BSL4) in the country. It can leave the facility if the correct biosecurity measures are not implemented correctly and can easily spread to livestock. | |
| Number of drivers = 8, hence 80 points to be distributed across this domain for the intra-domain weighing. | ||
| DOMAIN D8. ECONOMIC AND TRADE ACTIVITIES. | ||
| D8-1 | Decrease of resources allocated to the disease surveillance in human and/or animal and/or environment. | |
| Score 0 | ||
| Score 1 | Negligible: resources allocated to the disease surveillance have no effect on the (re-)emergence/incidence of the disease in your country. Disease has never been under surveillance. | |
| Score 2 | Low: resources allocated to the disease surveillance have a low effect on the (re-)emergence/incidence of the disease in your country. Disease has been under surveillance in the past and no change has happened after surveillance has been stopped. | |
| Score 3 | Medium: resources allocated to the disease surveillance have a moderate effect on the (re-)emergence/incidence of the disease in your country. Disease is under passive surveillance (reported only when observed) but with no need to further increase its surveillance | |
| Score 4 | High: resources allocated to the disease surveillance have a high effect on the (re-)emergence of the disease in your country. Disease needs to be under active and passive surveillance as its (re-)emergence/incidence can easily occur. Therefore, if its surveillance decreases it is highly likely to (re-)emerge. | |
| D8-2 | Modification of the disease status (i.e., reportable disease becoming not reportable) or changes in screening frequency due to a reduced national budget. | |
| Score 0 | ||
| Score 1 | Negligible: modification of the disease status due to a reduced national budget has a negligible effect on the (re-)emergence/incidence of the disease in your country. | |
| Score 2 | Low: modification of the disease status due to a reduced national budget has a low effect on the (re-)emergence/incidence of the disease in your country. | |
| Score 3 | Moderate: modification of the disease status due to a reduced national budget has a moderate effect on the (re-)emergence/incidence of the disease in your country. | |
| Score 4 | High: modification of the disease status due to a reduced national budget has a high effect on the (re-)emergence/incidence of the disease in your country. | |
| D8-3 | Decrease of resources allocated to the implementation of biosecurity measures at border controls (e.g., harbors or airports). | |
| Score 0 | Endemic disease | |
| Score 1 | Negligible: decreasing the resources allocated to the implementation of biosecurity measures has a negligible effect on the (re-)emergence or the human disease incidence in your country. Disease has never been detected in the past in a harbor or an airport. | |
| Score 2 | Low: decreasing the resources allocated to the implementation of biosecurity measures has a low effect on the (re-)emergence or the human disease incidence in your country. The disease has been suspected to have entered other countries because of deficient biosecurity at border controls. | |
| Score 3 | Medium: decreasing the resources allocated to the implementation of biosecurity measures has a moderate effect on the (re-)emergence or the human disease incidence in your country. The disease has been introduced in other countries because of deficient biosecurity at border controls. | |
| Score 4 | High: decreasing the resources allocated to the implementation of biosecurity measures highly increases the risk of (re-)emergence or the human disease incidence in your country. In the past, the disease has been introduced in other countries AND in your country because of deficient biosecurity measures at border controls. | |
| D8-4 | Most likely influence of (il)legal movements of live animals (livestock, pets, horses, etc.) from neighbouring/European Union member states (MS) for the disease to (re-)emerge in your country. | |
| Score 0 | ||
| Score 1 | Negligible: (il)legal movements of live animals (livestock, pets, horses, etc.) from neighbouring/European Union MS have a negligible influence on the pathogen/disease (re-)emergence/incidence in your country. | |
| Score 2 | Low: (il)legal movements (livestock, pets, horses, etc.) from neighbouring/European Union MS have a low influence on the pathogen/disease (re-)emergence in your country. | |
| Score 3 | Moderate: (il)legal movements (livestock, pets, horses, etc.) from neighbouring/European Union MS have a moderate influence on the pathogen/disease (re-)emergence in your country. | |
| Score 4 | High: (il)legal movements (livestock, pets, horses, etc.) from neighbouring/European Union MS have a high influence on the pathogen/disease (re-)emergence in your country. | |
| D8-5 | Most likely influence of (il)legal EU trade exchanges of food products (e.g., raw milk cheese products) for disease (re-)emergence in your country. | |
| Score 0 | ||
| Score 1 | Negligible: increased imports of dairy products such as raw milk cheese from European countries have a negligible influence on the pathogen/disease (re-)emergence in your country. | |
| Score 2 | Low: increased imports of dairy products such as raw milk cheese from European countries have a low influence on the pathogen/disease (re-)emergence in your country. | |
| Score 3 | Moderate: increased imports of dairy products such as raw milk cheese from European countries have a moderate influence on the pathogen/disease (re-)emergence in your country. | |
| Score 4 | High: increased imports of dairy products such as raw milk cheese from European countries have a high influence on the pathogen/disease (re-)emergence in your country. | |
| D8-6 | Most likely influence of increased (il)legal imports of animal subproducts, such as skin, meat, and edible products from neighbouring/European Union member states (MS) for the disease to (re-)emerge in your country. | |
| Score 0 | ||
| Score 1 | Negligible: Increased imports of animal subproducts, such as skin, meat, and edible products from neighbouring/European Union member states (MS) have a negligible influence on the pathogen/disease (re-)emergence in your country. | |
| Score 2 | Low: Increased imports of animal subproducts, such as skin, meat, and edible products from neighbouring/European Union member states (MS) have a low influence on the pathogen/disease (re-)emergence in your country. | |
| Score 3 | Moderate: Increased imports of animal subproducts, such as skin, meat, and edible products from neighbouring/European Union member states (MS) have a moderate influence on the pathogen/disease (re-)emergence in your country. | |
| Score 4 | High: Increased imports of animal subproducts, such as skin, meat, and edible products from neighbouring/European Union member states (MS) have a high influence on the pathogen/disease (re-)emergence in your country. | |
| D8-7 | Most likely influence of increased (il)legal imports of NON-animal products, such as tires, wood, and furniture from EU member states for the disease/pathogen to (re-)emerge in your country. | |
| Score 0 | ||
| Score 1 | Negligible: (il)legal movements of NON-animal products, such as tires, wood, and furniture from EU member states for the disease/pathogen to (re-)emerge in your country have a negligible influence on the pathogen/disease (re-)emergence/incidence in your country. | |
| Score 2 | Low: (il)legal movements of NON-animal products, such as tires, wood, and furniture from EU member states for the disease/pathogen to (re-)emerge in your country have a low influence on the pathogen/disease (re-)emergence/incidence in your country. | |
| Score 3 | Moderate: (il)legal movements of NON-animal products, such as tires, wood, and furniture from EU member states for the disease/pathogen to (re-)emerge in your country have a moderate influence on the pathogen/disease (re-)emergence/incidence in your country. | |
| Score 4 | High: (il)legal movements of NON-animal products, such as tires, wood, and furniture from EU member states for the disease/pathogen to (re-)emerge in your country have a high influence on the pathogen/disease (re-)emergence/incidence in your country. | |
| D8-8 | Most likely influence of (il)legal movements of live animals (livestock, pets, horses, etc.) from third-world countries for the disease to (re-)emerge in your country. | |
| Score 0 | ||
| Score 1 | Negligible:(il)legal movements of live animals (livestock, pets, horses, etc.) from third-world countries have a negligible influence on the pathogen/disease (re-)emergence/incidence in your country. | |
| Score 2 | Low: (il)legal movements of live animals (livestock, pets, horses, etc.) from third-world countries have a low influence on the pathogen/disease (re-)emergence/incidence in your country. | |
| Score 3 | Moderate: (il)legal movements of live animals (livestock, pets, horses, etc.) from third-world countries have a moderate on the pathogen/disease (re-)emergence/incidence in your country. | |
| Score 4 | High: (il)legal movements of live animals (livestock, pets, horses, etc.) from third-world countries have a high influence on the pathogen/disease (re-)emergence in your country. | |
| D8-9 | Most likely influence of (il)legal third-world countries trade exchanges of food products (dairy raw products) for disease (re-)emergence in your country. | |
| Score 0 | ||
| Score 1 | Negligible: increased imports of dairy products such as raw milk cheese from third-world countries have a negligible influence on the pathogen/disease (re-)emergence/incidence in your country. | |
| Score 2 | Low: increased imports of dairy products such as raw milk cheese from third-world countries have a low influence on the pathogen/disease (re-)emergence/incidence in your country. | |
| Score 3 | Moderate: increased imports of dairy products such as raw milk cheese from third-world countries have a influence on the pathogen/disease (re-)emergence/incidence in your country. | |
| Score 4 | High: increased imports of dairy products such as raw milk cheese from third-world countries have a high influence on the pathogen/disease (re-)emergence/incidence in your country. | |
| D8-10 | Most likely influence of increased imports of animal subproducts, such as skin, meat, and edible products from third-world countries, for the disease to (re-)emerge in your country. | |
| Score 0 | ||
| Score 1 | Negligible: increased imports of animal subproducts, such as skin, meat, and edible products from third-world countries have a negligible influence on the pathogen/disease (re-)emergence/incidence in your country. | |
| Score 2 | Low: increased imports of animal subproducts, such as skin, meat, and edible products from third-world countries have a low influence on the pathogen/disease (re-)emergence/incidence in your country. | |
| Score 3 | Moderate: increased imports of animal subproducts, such as skin, meat, and edible products from third-world countries have a moderate on the pathogen/disease (re-)emergence/incidence in your country. | |
| Score 4 | High: increased imports of animal subproducts, such as skin, meat, and edible products from third-world countries have a high influence on the pathogen/disease (re-)emergence/incidence in your country. | |
| D8-11 | Most likely influence of increased (il)legal imports of NON-animal products, such as tires, wood, and furniture from third-world countries, for the disease to (re-)emerge in your country. | |
| Score 0 | ||
| Score 1 | Negligible: increased (il)legal imports of NON-animal products, such as tires, wood, and furniture from third-world countries have a negligible influence on the pathogen/disease (re-)emergence/incidence in your country. | |
| Score 2 | Low: increased (il)legal imports of NON-animal products, such as tires, wood, and furniture from third-world countries have a low influence on the pathogen/disease (re-)emergence/incidence in your country. | |
| Score 3 | Moderate: increased (il)legal imports of NON-animal products, such as tires, wood, and furniture from third-world countries have a moderate influence on the pathogen/disease (re-)emergence/incidence in your country. | |
| Score 4 | High: increased (il)legal imports of NON-animal products, such as tires, wood, and furniture from third-world countries have a high influence on the pathogen/disease (re-)emergence/incidence in your country. | |
| Number of drivers = 11, hence 110 points to be distributed across this domain for the intra-domain weighing. | ||
| ID | Name | Surname | Gender | ISO | Level | Employment | Competency | Keyword 1 | Keyword 2 | Keyword 3 |
|---|---|---|---|---|---|---|---|---|---|---|
| 1. | Berger | Thomas F.H. | M | CH | N; R; I | GI; RS | FS | Dairy | Food safety | Risk evaluation and mitigation |
| 2. | Bødker | René | M | DK | N | UN | PH; AH | VBD | Epidemiology | Surveiilance |
| 3. | Bonnet | Sarah I. | F | FR | N; R; I | RS | PHAHEH | Ticks | TBP | Tick-host-pathogen interactions |
| 4. | Boulanger | Nathalie | F | FR | N; I | GI; UN; RS | PH; LD | Medical entomologist | Parasitologist | Infectious diseases |
| 5. | Bournez | Laure | F | FR | N | RS | AH; EH | Eco-Epidemiology | Ticks and TBP | TBEV |
| 6. | Brugger | Katharina | F | AT | NI | RS | PH; EH | Climatology | Epidemiology | Vectors and VBD |
| 7. | De Regge | Nick | M | BE | N | GI | AH; LD | Virology | VBD | Diagnostics |
| 8. | Estrada-Peña | Agustín | M | SP | N | UN | PH; AH | Ticks | Ecology, Epidemiology, and Vector hosts | Zoonoses |
| 9. | Fravalo | Philippe | M | FR | N | UN | PH; FS; LD | Biological risk in food | Meat production chain | Pathogen characterization |
| 10. | Geller | Julia | F | EE | N | GI; RS | PH | Research | Monitoring | Surveillance |
| 11. | Gilot-Fromont | Emmanuelle | F | FR | N | UN | AH; EH | Eco-epidemiology | Disease management | |
| 12. | Gonzalez | Gaëlle | F | FR | N | GI | AH | Surveillance | Virulence determinants | Co-infection |
| 13. | Haddad | Nadia | F | FR | N | UN; RS | PH; AH | Zoonoses | TBP | Epidemiology |
| 14. | Heyman | Paul | M | BE | N; I | GI; DL | PH; AH; LD | TBD | Hantaviruses | |
| 15. | Hoch | Thierry | M | FR | N | RS | AH | Matematical modelling | Epidemiology | Ticks |
| 16. | Humblet | Marie-France | F | BE | R | UN | AH | Animal biosecurity | Veterinary epidemiology | One Health |
| 17. | Jourdain | Elsa | F | FR | N | GI | PH; AH; LD | Descriptive epidemiology | Zoonotic diseases | Animal reservoir |
| 18. | Kerlik | Jana | F | SK | R | GI | PH | Epidemiology | Zoonoses | TBE |
| 19. | Klein | Matthias | M | DE | R | UN | PH | Neuroinfectiology | Emergency medicine | Intensive care |
| 20. | Knap | Natasa | F | SI | N | UN; RS; DL | LD | Diagnostics | Ecology | Virology |
| 21. | Kooh | Pauline | F | FR | N | GI | FS | Food Microbiology | Risk assessment | Virology |
| 22. | Lacour | Sandrine A. | F | FR | N | GI | AH; FS | Molecular virology | Virus-host interactions | Milk/cheese-borne TBEV |
| 23. | Leandri | Marc | M | FR | N | UN | EC | Decision | Perceptions | Information |
| 24. | Leib | Stephen L. | M | CH | N; R; I | UN; RS; DL | LD | Infectious Diseases | Microbiology | Brain infections |
| 25. | Lernout | Tinne | F | BE | N | RS | PH | TBD | Epidemiology | Risk Assessement |
| 26. | L’Hostis | Monique | F | FR | N | UN | PH | Parasitologist | Ixodes ricinus | Ecology |
| 27. | Martin-Latil | Sandra | F | FR | N | GI | FS; LD | Virus | Detection | Water / food |
| 28. | Moutailler | Sara | F | FR | N | GI; RS | AH | Ticks and TBP | Vector competence | Molecular epidemiology |
| 29. | Purse | Bethan | F | UK | I | RS | PH; AH; EH | VBD | Ecology | Epidemiological modeling |
| 30. | Quillery | Elsa | F | FR | N | GI | PH | Ecology | Vector diseases | Risk assessment |
| 31. | Raffetin | Alice | F | FR | N; R; I | UN; RS; HO | PH | Lyme borreliosis | TBD | Infectious diseases |
| 32. | Ruiz-Fons | Francisco | M | SP | N; R; I | GI; RS | AH | Epidemiology | Wildlife | VBP |
| 33. | Ruzek | Daniel | M | CZ | N | UN | AH; LD | TBE | Flaviviruses | Virus-host interactions |
| 34. | Solveig | Jore | F | NO | N | RS | PH | Zoonoses | TBD | Climate change |
| 35. | Sprong | Hein | M | NL | NI | GI; UN | PH; EH; LD | TBD | Molecular epidemiology | Disease ecology |
| 36. | Studahl | Marie | F | SE | N; R | UN; RS; HO | PH | Viral CNS infections | Viral diagnostics | Burden of infectious diseases |
| 37. | Thiry | Etienne | M | BE | N | UN | AH; LD | Animal viruses | Veterinary infectiology | Veterinary Public Health |
| 38. | Velay | Aurélie | F | FR | R | UN; RS; DL | PH; LD | Virology | Emerging Diseases | TBE |
| 39. | Zdenek | Hubalek | M | CZ | N; I | RS | PH; AH; EH | Arbovirology | Zoology | Monitoring |
| 40. | Zomer | Tizza | F | NL | R | HO | PH | Lyme borreliosis | Epidemiology | TBD |
Appendix B. Guidance Letter for the Expert Elicitation

- Objectives of the questionnaire
- How to fill out the questionnaire
- Expert information
- Pathogen characteristics: 12 criteria
- Distance to Europe and your country: 3 criteria
- Ability to monitor, treat, and control the disease: 11 criteria
- Farm/European characteristics: 5 criteria
- Global change: 4 criteria
- Wildlife interface: 5 criteria
- Human activities: 8 criteria
- Economy and trade activities: 11 criteria
- Intra-category weighing
- Level of uncertainty per domain of criteria
- Actions to be performed:
- (1)
- Score and balance each driver within each category of drivers:
- ▪
- Please give a score according to what you estimate is the importance of each driver in the (re-)emergence or increasing incidence of tick-borne encephalitis disease in humans across Europe.
- ▪
- After scoring, please balance each driver for each category of drivers. Balancing the criteria will depend on the distribution of points among the different criteria in each category. The total number of points to be distributed among the drivers is specified for each category (on each spreadsheet). For example, in the category “pathogen characteristics”: a total of 120 points must be distributed => for “distance to Europe and your country”, a total of 30 points must be distributed. If a driver has no importance related to others, zero points can be attributed to this driver.
- (2)
- Intra-category weighing:
- (3)
- Level of uncertainty:
| Country of Origin | Number of Cases 2020 | Incidence 2020 | Short Description of the Situation | Trend in Number of Cases 2012−2020 (Country Level) | Proposed Level (Increasing Order: A to E) |
|---|---|---|---|---|---|
| Spain | 0 | 0 | No autochthonous cases reported. | - | A |
| France | 46 | 0.1 | Low endemicity, only in few regions | Increase trend | B |
| Belgium | 7 | 0.1 | Sporadic cases (emergence in 2020) | - | |
| The Netherlands | 5 | Sporadic cases (emergence in 2016) | - | ||
| Denmark | Sporadic cases (endemic only on Bornholm island) | No information | |||
| United Kingdom | 2 | 0 | Sporadic cases (emergence in 2019) | - | |
| Norway | 41 | 0.8 | Low to moderate endemicity, only in few regions | Increase trend | C |
| Austria | 250 | 2.8 | Low to moderate endemicity, throughout the country | Increase trend | D |
| Germany | 705 | 0.5 | Low to high endemicity, throughout the country | Increase trend | |
| Sweden | 267 | 2.6 | Low to high endemicity, in most of the country | Increase trend | |
| Slovakia | 185 | 3.4 | Low to high endemicity, throughout the country | No trend | |
| Switzerland | 454 | 5.1 | Moderate to high endemicity, in most of the country | Increase trend | |
| Czechia | 849 | 7.9 | Moderate to high endemicity, throughout the country | Increase trend | E |
| Estonia | 70 | 5.3 | Moderate to high endemicity, throughout the country | Decrease trend | |
| Slovenia | 187 | 8.9 | Low to high endemicity, throughout the country | No trend |
References
- Michelitsch, A.; Wernike, K.; Klaus, C.; Dobler, G.; Beer, M. Exploring the reservoir hosts of tick-borne encephalitis virus. Viruses 2019, 11, 669. [Google Scholar] [CrossRef]
- Leutloff, R.; Nübling, M.; Neumann-Haefelin, D.; Rieger, M.A. Cows as indicators for TBE endemic regions: Suitability of testing for antibodies in serum and milk. Int. J. Med. Microbiol. 2006, 296, 87–88. [Google Scholar]
- Hoogstraal, H. Ticks in relation to human diseases caused by viruses. Annu. Rev. Entomol. 1966, 11, 261–308. [Google Scholar] [CrossRef] [PubMed]
- Elbaz, M.; Gadoth, A.; Shepshelovich, D.; Shasha, D.; Rudoler, N.; Paran, Y. Systematic Review and Meta-analysis of Foodborne Tick-Borne Encephalitis, Europe, 1980–2021. Emerg. Infect. Dis. 2022, 28, 1945–1954. [Google Scholar] [CrossRef]
- Martello, E.; Gillingham, E.L.; Phalkey, R.; Vardavas, C.; Nikitara, K.; Bakonyi, T.; Gossner, C.M.; Leonardi-Bee, J. Systematic review on the non-vectorial transmission of tick-borne encephalitis virus (TBEv). Ticks Tick-Borne Dis. 2022, 13, 102028. [Google Scholar] [CrossRef] [PubMed]
- Kerlik, J.; Avdičová, M.; Štefkovičová, M.; Tarkovská, V.; Pántiková Valachová, M.; Molčányi, T.; Mezencev, R. Slovakia reports highest occurrence of alimentary tick-borne encephalitis in Europe: Analysis of tick-borne encephalitis outbreaks in Slovakia during 2007–2016. Travel Med. Infect. Dis. 2018, 26, 37–42. [Google Scholar] [CrossRef]
- Pfeffer, M.; Dobler, G. Emergence of zoonotic arboviruses by animal trade and migration. Parasites Vectors 2010, 3, 35. [Google Scholar] [CrossRef]
- Lommano, E.; Dvořák, C.; Vallotton, L.; Jenni, L.; Gern, L. Tick-borne pathogens in ticks collected from breeding and migratory birds in Switzerland. Ticks Tick Borne Dis. 2014, 5, 871–882. [Google Scholar] [CrossRef]
- Süss, J. Tick-borne encephalitis in Europe and beyond—The epidemiological situation as of 2007. Eurosurveillance 2008, 13, 717–727. [Google Scholar] [CrossRef]
- Weaver, S.C.; Reisen, W.K. Present and future arboviral threats. Antivir. Res. 2010, 85, 328–345. [Google Scholar] [CrossRef]
- Gubler, D.J. Human arbovirus infections worldwide. Ann. N. Y. Acad. Sci. 2001, 951, 13–24. [Google Scholar] [CrossRef] [PubMed]
- Randolph, S.E. Transmission of tick-borne pathogens between co-feeding ticks: Milan Labuda’s enduring paradigm. Ticks Tick-Borne Dis. 2011, 2, 179–182. [Google Scholar] [CrossRef]
- Matser, A.; Hartemink, N.; Heesterbeek, H.; Galvani, A.; Davis, S. Elasticity analysis in epidemiology: An application to tick-borne infections. Ecol. Lett. 2009, 12, 1298–1305. [Google Scholar] [CrossRef] [PubMed]
- Haydon, D.T.; Cleaveland, S.; Taylor, L.H.; Laurenson, M.K. Identifying reservoirs of infection: A conceptual and practical challenge. Emerg. Infect. Dis. 2002, 8, 1468–1473. [Google Scholar] [CrossRef]
- Taba, P.; Schmutzhard, E.; Forsberg, P.; Lutsar, I.; Ljøstad, U.; Mygland, Å.; Levchenko, I.; Strle, F.; Steiner, I. EAN consensus review on prevention, diagnosis and management of tick-borne encephalitis. Eur. J. Neurol. 2017, 24, 1214-e61. [Google Scholar] [CrossRef]
- Bogovic, P.; Strle, F. Tick-borne encephalitis: A review of epidemiology, clinical characteristics, and management. World J. Clin. Cases 2015, 3, 430–441. [Google Scholar] [CrossRef] [PubMed]
- Valarcher, J.F.; Hägglund, S.; Juremalm, M.; Blomqvist, G.; Renström, L.; Zohari, S.; Leijon, M.; Chirico, J. Tick-borne encephalitis. Rev. Sci. Tech. 2015, 34, 453–466. [Google Scholar] [CrossRef]
- Deviatkin, A.A.; Karganova, G.G.; Vakulenko, Y.A.; Lukashev, A.N. TBEV Subtyping in Terms of Genetic Distance. Viruses 2020, 12, 1240. [Google Scholar] [CrossRef] [PubMed]
- Kutschera, L.; Wolfinger, M.T. Evolutionary traits of Tick-borne encephalitis virus: Pervasive non-coding RNA structure conservation and molecular epidemiology. Virus Evol. 2022, 8, veac051. [Google Scholar] [CrossRef]
- Kunze, M.; Banović, P.; Bogovič, P.; Briciu, V.; Čivljak, R.; Dobler, G.; Hristea, A.; Kerlik, J.; Kuivanen, S.; Kynčl, J.; et al. Recommendations to Improve Tick-Borne Encephalitis Surveillance and Vaccine Uptake in Europe. Microorganisms 2022, 10, 1283. [Google Scholar] [CrossRef]
- Lundkvist, K.; Vene, S.; Golovljova, I.; Mavtchoutko, V.; Forsgren, M.; Kalnina, V.; Plyusnin, A. Characterization of tick-borne encephalitis virus from Latvia: Evidence for co-circulation of three distinct subtypes. J. Med. Virol. 2001, 65, 730–735. [Google Scholar] [CrossRef] [PubMed]
- Kubinski, M.; Beicht, J.; Gerlach, T.; Volz, A.; Sutter, G.; Rimmelzwaan, G.F. Tick-Borne Encephalitis Virus: A Quest for Better Vaccines against a Virus on the Rise. Vaccines 2020, 8, 451. [Google Scholar] [CrossRef] [PubMed]
- Fafangel, M.; Cassini, A.; Colzani, E.; Klavs, I.; Grgič Vitek, M.; Učakar, V.; Muehlen, M.; Vudrag, M.; Kraigher, A. Estimating the annual burden of tick-borne encephalitis to inform vaccination policy, Slovenia, 2009 to 2013. Eurosurveillance 2017, 22, pii=30509. [Google Scholar] [CrossRef] [PubMed]
- Slunge, D.; Boman, A.; Studahl, M. Burden of Tick-Borne Encephalitis, Sweden. Emerg. Infect. Dis. 2022, 28, 314–322. [Google Scholar] [CrossRef]
- Šmit, R.; Postma, M.J. Review of tick-borne encephalitis and vaccines: Clinical and economical aspects. Expert Rev. Vaccines 2015, 14, 737–747. [Google Scholar] [CrossRef] [PubMed]
- Steffen, R.; Erber, W.; Schmitt, H.J. Can the booster interval for the tick-borne encephalitis (TBE) vaccine ‘FSME-IMMUN’ be prolonged? A systematic review. Ticks Tick-Borne Dis. 2021, 12, 101779. [Google Scholar] [CrossRef]
- Zimna, M.; Brzuska, G.; Salát, J.; Svoboda, P.; Baranska, K.; Szewczyk, B.; Růžek, D.; Krol, E. Functional characterization and immunogenicity of a novel vaccine candidate against tick-borne encephalitis virus based on Leishmania-derived virus-like particles. Antivir. Res. 2022, 26, 105511. [Google Scholar] [CrossRef]
- de Graaf, J.A.; Reimerink, J.H.; Voorn, G.P.; Bij de Vaate, E.A.; de Vries, A.; Rockx, B.; Schuitemaker, A.; Hira, V. First human case of tick-borne encephalitis virus infection acquired in the Netherlands, July 2016. Eurosurveillance 2016, 21, 30318. [Google Scholar] [CrossRef]
- Lernout, T.; Van Esbroeck, M. Surveillance Épidémiologique de L’encéphalite à Tiques TBEV—2018. Sciensano, 2018 (Disponible en Ligne à L’adresse Suivante. Available online: https://epidemio.wiv-isp.be/ID/diseases/Documents/Reports%202018/TBEV_2018_fr.pdf (accessed on 15 March 2023).
- Stoefs, A.; Heyndrickx, L.; De Winter, J.; Coeckelbergh, E.; Willekens, B.; Alonso-Jiménez, A.; Tuttino, A.-M.; Geerts, Y.; Ariën, K.K.; Van Esbroeck, M. Autochthonous Cases of Tick-Borne Encephalitis, Belgium, 2020. Emerg. Infect. Dis. 2021, 27, 2179–2182. [Google Scholar] [CrossRef]
- Mansbridge, C.T.; Osborne, J.; Holding, M.; Dryden, M.; Aram, M.; Brown, K.; Sutton, J. Autochthonous tick-borne encephalitis in the United Kingdom: A second probable human case and local eco-epidemiological findings. Ticks Tick Borne Dis. 2022, 13, 101853. [Google Scholar] [CrossRef]
- ECDC. Tick-Borne Encephalitis—Annual Epidemiological Report for 2020. Surveillance Report, 28 Oct 2022, Annual Epidemiological Report on Communicable Diseases in Europe. European Centre for Disease Prevention and Control, Stockholm, Sweden. 2022. Available online: https://www.ecdc.europa.eu/en/publications-data/tick-borne-encephalitis-annual-epidemiological-report-2020 (accessed on 15 March 2023).
- Šmit, R.; Postma, M.J. The Burden of Tick-Borne Encephalitis in Disability-Adjusted Life Years (DALYs) for Slovenia. PLoS ONE 2015, 10, e0144988. [Google Scholar] [CrossRef]
- Mailles, A.; Argemi, X.; Biron, C.; Fillatre, P.; De Broucker, T.; Buzelé, R.; Gagneux-Brunon, A.; Gueit, I.; Henry, C.; Patrat-Delon, S.; et al. Changing profile of encephalitis: Results of a 4-year study in France. Infect Dis. Now 2022, 52, 1–6. [Google Scholar] [CrossRef]
- WHO. Chapter 6—Vaccine-Preventable Diseases and Vaccines; International Travel and Health, WHO: Geneva, Switzerland, 2019; 53p, ISBN 978-92-4-158047-2. [Google Scholar]
- Sumilo, D.; Asokliene, L.; Bormane, A.; Vasilenko, V.; Golovljova, I.; Randolph, S.E. Climate Change Cannot Explain the Upsurge of Tick-Borne Encephalitis in the Baltics. PLoS ONE 2007, 2, e500. [Google Scholar] [CrossRef] [PubMed]
- Cox, R.; Sánchez, J.; Revie, C.W. Multi-Criteria Decision Analysis Tools for Prioritising Emerging or Re-Emerging Infectious Diseases Associated with Climate Change in Canada. PLoS ONE 2013, 8, e68338. [Google Scholar] [CrossRef]
- ECDC. Best Practices in Ranking Emerging Infectious Disease Threats. A Literature Review. European Centre for Disease Prevention and Control. 2015. Available online: https://ecdc.europa.eu/sites/portal/files/media/en/publications/Publications/emerging-infectious-disease-threats-best-practices-ranking.pdf (accessed on 1 December 2022).
- Bianchini, J.; Humblet, M.F.; Cargnel, M.; Van der Stede, Y.; Koenen, F.; de Clercq, K.; Saegerman, C. Prioritization of livestock transboundary diseases in Belgium using a multicriteria decision analysis tool based on drivers of emergence. Transbound. Emerg. Dis. 2020, 67, 344–376. [Google Scholar] [CrossRef] [PubMed]
- Saegerman, C.; Bianchini, J.; Snoeck, C.J.; Moreno, A.; Chiapponi, C.; Zohari, S.; Ducatez, M.F. First expert elicitation of knowledge on drivers of emergence of influenza D in Europe. Transbound. Emerg. Dis. 2021, 68, 3349–3359. [Google Scholar] [CrossRef]
- Saegerman, C.; Evrard, J.; Houtain, J.-Y.; Alzieu, J.-P.; Bianchini, J.; Mpouam, S.E.; Schares, G.; Liénard, E.; Jacquiet, P.; Villa, L.; et al. First Expert Elicitation of Knowledge on Drivers of Emergence of Bovine Besnoitiosis in Europe. Pathogens 2022, 11, 753. [Google Scholar] [CrossRef]
- Gore, S.M. Biostatistics and the Medical Research Council. MRC News 1987, 35, 19–20. [Google Scholar]
- Vanwambeke, S.O.; Sumilo, D.; Bormane, A.; Lambin, E.F.; Randolph, S.E. Landscape predictors of tick-borne encephalitis in Latvia: Land cover, land use, and land ownership. Vector Borne Zoonotic Dis. 2010, 10, 497–506. [Google Scholar] [CrossRef]
- Petit, K.; Dunoyer, C.; Fischer, C.; Hars, J.; Baubet, E.; Ramón López-Olvera, J.; Rossi, S.; Collin, E.; Le Potier, M.-F.; Belloc, C.; et al. Assessment of the impact of forestry and leisure activities on wild boar spatial disturbance with a potential application to ASF risk of spread. Transbound. Emerg. Dis. 2020, 67, 1164–1176. [Google Scholar] [CrossRef]
- Asaaga, F.A.; Rahman, M.; Kalegowda, S.D.; Mathapati, J.; Savanur, I.; Srinivas, P.N.; Seshadri, T.; Narayanswamy, D.; Kiran, S.K.; Oommen, M.A.; et al. ‘None of my ancestors ever discussed this disease before!’ How disease information shapes adaptive capacity of marginalised rural populations in India. PLoS Negl. Trop. Dis. 2021, 15, e0009265. [Google Scholar] [CrossRef]
- Colonna, A.; Durham, C.; Meunier-Goddik, L. Factors affecting consumers’ preferences for and purchasing decisions regarding pasteurized and raw milk specialty cheeses. J. Dairy Sci. 2011, 94, 5217–5226. [Google Scholar] [CrossRef]
- Lengard Almli, V.; Naes, T.; Enderli, G.; Sulmont-Rossé, C.; Issanchou, S.; Hersleth, M. Consumers’ acceptance of innovations in traditional cheese. A comparative study in France and Norway. Appetite 2011, 57, 110–120. [Google Scholar] [CrossRef]
- Bachmann, H.P.; Fröhlich, M.T.; Bisig, W. Raw Milk and Raw-Milk Products Affect our Health [English/French abstract on website, Rohmilch und Rohmilchprodukte beeinflussen unsere Gesundheit]. Swiss Agr. Res. 2020, 11, 124–130. [Google Scholar] [CrossRef]
- Mutius, E.; Schmid, S.; The PASTURE Study Group. The PASTURE project: EU support for the improvement of knowledge about risk factors and preventive factors for atopy in Europe. Allergy 2006, 61, 407–413. [Google Scholar] [CrossRef]
- Nicolau, M.; Esquivel, L.; Schmidt, I.; Fedato, C.; Leimann, L.; Samoggia, A.; Monticone, F.; Prete, D.M.; Ghelfi, R.; Saviolidis, M.N.; et al. Food consumption behaviours in Europe. Mapping drivers, trends and pathways towards sustainability. VALUMICS Proj. “Understanding Food Value Chains and Network Dynamics”, funded by European Union’s Horizon 2020 research and innovation programme GA No 727243. Deliverable: D6.1, CSCP, Germany, 87 pages. 2021. [Google Scholar] [CrossRef]
- Zeimes, C.B.; Olsson, G.E.; Hjertqvist, M.; Vanwambeke, S.O. Shaping zoonosis risk: Landscape ecology vs. landscape attractiveness for people, the case of tick-borne encephalitis in Sweden. Parasites Vectors 2014, 7, 370. [Google Scholar] [CrossRef]
- Uusitalo, R.; Siljander, M.; Lindén, A.; Sormunen, J.J.; Aalto, J.; Hendrickx, G.; Kallio, E.; Vajda, A.; Gregow, H.; Henttonen, H.; et al. Predicting habitat suitability for Ixodes ricinus and Ixodes persulcatus ticks in Finland. Parasit Vectors 2022, 15, 310. [Google Scholar] [CrossRef]
- Requena-García, F.; Cabrero-Sañudo, F.; Olmeda-García, S.; González, J.; Valcárcel, F. Influence of environmental temperature and humidity on questing ticks in central Spain. Exp. Appl. Acarol. 2017, 71, 277–290. [Google Scholar] [CrossRef] [PubMed]
- Stafford, K.C. Survival of immature Ixodes scapularis (Acari, Ixodidae) at different relative humidities. J. Med. Entomol. 1994, 31, 310–314. [Google Scholar] [CrossRef] [PubMed]
- Vail, S.G.; Smith, G. Vertical movement and posture of blacklegged tick (Acari: Ixodidae) nymphs as a function of temperature and relative humidity in laboratory experiments. J. Med. Entomol. 2002, 39, 842–846. [Google Scholar] [CrossRef]
- Ogden, N.H.; Lindsay, L.R.; Beauchamp, G.; Charron, D.; Maarouf, A.; O’Callaghan, C.J.; Waltner-Toews, D.; Barker, I.K. Investigation of relationships between temperature and developmental rates of tick Ixodes scapularis (Acari: Ixodidae) in the laboratory and field. J. Med. Entomol. 2004, 41, 622–633. [Google Scholar] [CrossRef] [PubMed]
- Gray, J.S. The ecology of ticks transmitting Lyme borreliosis. Exp. Appl. Acarol. 1998, 22, 249–258. [Google Scholar] [CrossRef]
- Kahl, O.; Gray, J.S. The biology of Ixodes ricinus with emphasis on its ecology. Ticks Tick Borne Dis. 2023, 14, 102114. [Google Scholar] [CrossRef]
- Tabachnick, W.J. Challenges in predicting climate and environmental effects on vector-borne disease episystems in a changing world. J. Exp. Biol. 2010, 213, 946–954. [Google Scholar] [CrossRef]
- Haider, N.; Kirkeby, C.; Kristensen, B.; Kjær, L.J.; Sørensen, J.H.; Bødker, R. Microclimatic temperatures increase the potential for vector-borne disease transmission in the Scandinavian climate. Sci. Rep. 2017, 7, 8175. [Google Scholar] [CrossRef]
- Borde, J.P.; Glaser, R.; Braun, K.; Riach, N.; Hologa, R.; Kaier, K.; Chitimia-Dobler, L.; Dobler, G. Decoding the Geography of Natural TBEV Microfoci in Germany: A Geostatistical Approach Based on Land-Use Patterns and Climatological Conditions. Int. J. Environ. Res. Public Health 2022, 19, 11830. [Google Scholar] [CrossRef] [PubMed]
- Nah, K.; Bede-Fazekas, Á.; János Trájer, A.; Wu, J. The potential impact of climate change on the transmission risk of tick-borne encephalitis in Hungary. BMC Infect. Dis. 2020, 20, 34. [Google Scholar] [CrossRef]
- Chávez-Larrea, M.A.; Cholota-Iza, C.; Medina-Naranjo, V.; Yugcha-Díaz, M.; Ron-Román, J.; Martin-Solano, S.; Gómez-Mendoza, G.; Saegerman, C.; Reyna-Bello, A. Detection of Babesia spp. in High Altitude Cattle in Ecuador, Possible Evidence of the Adaptation of Vectors and Diseases to New Climatic Conditions. Pathogens 2021, 10, 1593. [Google Scholar] [CrossRef]
- Cunze, S.; Glock, G.; Kochmann, J.; Klimpel, S. Ticks on the move-climate change- induced range shifts of three tick species in Europe: Current and future habitat suitability for Ixodes ricinus in comparison with Dermacentor reticulatus and Dermacentor marginatus. Parasitol Res. 2013, 121, 2241–2252. [Google Scholar] [CrossRef]
- Randolph, S.E.; Asokliene, L.; Avsic-Zupanc, T.; Bormane, A.; Burri, C.; Gern, L.; Golovljova, I.; Hubalek, Z.; Knap, N.; Kondrusik, M.; et al. Variable spikes in tick-borne encephalitis incidence in 2006 independent of variable tick abundance but related to weather. Parasit Vectors 2008, 1, 44. [Google Scholar] [CrossRef]
- Voyiatzaki, C.; Papailia, S.I.; Venetikou, M.S.; Pouris, J.; Tsoumani, M.E.; Papageorgiou, E.G. Climate Changes Exacerbate the Spread of Ixodes ricinus and the Occurrence of Lyme Borreliosis and Tick-Borne Encephalitis in Europe-How Climate Models Are Used as a Risk Assessment Approach for Tick-Borne Diseases. Int. J. Environ. Res. Public Health 2022, 19, 6516. [Google Scholar] [CrossRef] [PubMed]
- Zannou, O.M.; Ouedraogo, A.S.; Biguezoton, A.S.; Lempereur, L.; Yao, K.P.; Abatih, E.; Zoungrana, S.; Lenaert, M.; Toe, P.; Farougou, S.; et al. First digital characterization of the transhumance corridors through Benin used by cattle herds from Burkina Faso and associated risk scoring regarding the invasion of Rhipicephalus (Boophilus) microplus. Transbound. Emerg. Dis. 2021, 68, 2079–2093. [Google Scholar] [CrossRef] [PubMed]
- Zannou, O.M.; Ouedraogo, A.S.; Biguezoton, A.S.; Abatih, E.; Coral-Almeida, M.; Farougou, S.; Yao, K.P.; Lempereur, L.; Saegerman, C. Models for studying the distribution of ticks and tick-borne diseases in animals: A systematic review and a meta-Analysis with a Focus on Africa. Pathogens 2021, 10, 893. [Google Scholar] [CrossRef] [PubMed]
- Lippi, C.A.; Ryan, S.J.; White, A.L.; Gaff, H.D.; Carlson, C.J. Trends and opportunities in tick-borne disease geography. J. Med. Entomol. 2021, 58, 2021–2029. [Google Scholar] [CrossRef]
- Tischhauser, W.; Gründer, J. Prévention Contre les Tiques: Nouvelle Application Mobile. Bulletin OFSP 2015, 12, 207. Available online: www.bag.admin.ch/dam/bag/fr/dokumente/mt/infektionskrankheiten/fsme/zecken-app.pdf.download.pdf/zeckenapp-f.pdf (accessed on 15 March 2023).
- Süss, J. Epidemiology and ecology of TBE relevant to the production of effective vaccines. Vaccine 2003, 21 (Suppl. 1), S19–S35. [Google Scholar] [CrossRef]
- Bakhvalova, V.N.; Dobrotvorsky, A.K.; Panov, V.V.; Matveeva, V.A.; Tkachev, S.E.; Morozova, O.V. Natural tick-borne encephalitis virus infection among wild small mammals in the southeastern part of western Siberia, Russia. Vector Borne Zoonotic Dis. 2006, 6, 32–41. [Google Scholar] [CrossRef]
- Bakhvalova, V.N.; Potapova, O.F.; Panov, V.V.; Morozova, O.V. Vertical transmission of tick-borne encephalitis virus between generations of adapted reservoir small rodents. Virus Res. 2009, 140, 172–178. [Google Scholar] [CrossRef]
- Tonteri, E.; Kipar, A.; Voutilainen, L.; Vene, S.; Vaheri, A.; Vapalahti, O.; Lundkvist, A. The three subtypes of tick-borne encephalitis virus induce encephalitis in a natural host, the bank vole (Myodes glareolus). PLoS ONE 2013, 8, e81214. [Google Scholar] [CrossRef]
- Tonteri, E.; Jaaskelainen, A.E.; Tikkakoski, T.; Voutilainen, L.; Niemimaa, J.; Henttonen, H.; Vaheri, A.; Vapalahti, O. Tick-borne encephalitis virus in wild rodents in winter, Finland, 2008–2009. Emerg. Infect. Dis. 2011, 17, 72–75. [Google Scholar] [CrossRef] [PubMed]
- Radda, A. Die Zeckenenzephalitis in Europa. Z. Fuer Angew. Zool. 1973, 60, 409–461. [Google Scholar]
- Hinaidy, H.K. Die Parasitenfauna des Rotfuchses, Vulpes vulpes (L.), in Österreich. Zent. Vet. B. 1971, 18, 21–32. [Google Scholar] [CrossRef]
- Jemeršić, L.; Dežđek, D.; Brnić, D.; Prpić, J.; Janicki, Z.; Keros, T.; Roić, B.; Slavica, A.; Terzić, S.; Konjević, D.; et al. Detection and genetic characterization of tick-borne encephalitis virus (TBEV) derived from ticks removed from red foxes (Vulpes vulpes) and isolated from spleen samples of red deer (Cervus elaphus) in Croatia. Ticks Tick Borne Dis. 2014, 5, 7–13. [Google Scholar] [CrossRef]
- Da Rold, G.; Obber, F.; Monne, I.; Milani, A.; Ravagnan, S.; Toniolo, F.; Sgubin, S.; Zamperin, G.; Foiani, G.; Vascellari, M.; et al. Clinical Tick-Borne Encephalitis in a Roe Deer (Capreolus capreolus L.). Viruses 2022, 14, 300. [Google Scholar] [CrossRef]
- Fouché, N.; Oesch, S.; Ziegler, U.; Gerber, V. Clinical presentation and laboratory diagnostic work-up of a horse with tick-borne encephalitis in Switzerland. Viruses 2021, 13, 1474. [Google Scholar] [CrossRef]
- Eisen, L.; Stafford, K.C. Barriers to Effective Tick Management and Tick-Bite Prevention in the United States (Acari: Ixodidae). J. Med. Entomol. 2021, 58, 1588–1600. [Google Scholar] [CrossRef] [PubMed]
- Wilhelmsson, P.; Jaenson, T.G.T.; Olsen, B.; Waldenström, J.; Lindgren, P.E. Migratory birds as disseminators of ticks and the tick-borne pathogens Borrelia bacteria and tick-borne encephalitis (TBE) virus: A seasonal study at Ottenby Bird Observatory in South-eastern Sweden. Parasit Vectors 2020, 13, 607. [Google Scholar] [CrossRef]
- Deinet, S.; Ieronymidou, C.; McRae, L.; Burfield, I.J.; Foppen, R.P.; Collen, B.; Böhm, M. Wildlife comeback in Europe: The recovery of selected mammal and bird species. In Final Report to Rewilding Europe by ZSL, BirdLife International and the European Bird Census Council; ZSL: London, UK, 2013. [Google Scholar]
- Ledger, S.E.H.; Rutherford, C.A.; Benham, C.; Burfield, I.J.; Deinet, S.; Eaton, M.; Freeman, R.; Gray, C.; Herrando, S.; Puleston, H.; et al. Wildlife Comeback in Europe: Opportunities and challenges for species recovery. In Final Report to Rewilding Europe by the Zoological Society of London, BirdLife International and the European Bird Census Council; ZSL: London, UK, 2022. [Google Scholar]
- Hofmeester, T.R.; Sprong, H.; Jansen, P.A.; Prins, H.H.T.; van Wieren, S.E. Deer presence rather than abundance determines the population density of the sheep tick, Ixodes ricinus, in Dutch forests. Parasites Vectors 2017, 10, 433. [Google Scholar] [CrossRef]
- Knap, N.; Avšič-Županc, T. Correlation of TBE incidence with red deer and roe deer abundance in Slovenia. PLoS ONE 2013, 8, e66380. [Google Scholar] [CrossRef]
- Haemig, P.D.; Lithner, S.; Sjöstedt de Luna, S.; Lundqvist, A.; Waldenström, J.; Hansson, L.; Arneborn, M.; Olsen, B. Red fox tick-borne encephalitis (TBE) in humans: Can predators influence public health? Scand. J. Infect. Dis. 2008, 40, 527–532. [Google Scholar] [CrossRef]
- Jaenson, T.G.T.; Hjertqvist, M.; Bergström, T.; Lundkvist, A. Why is tick-borne encephalitis increasing? A review of the key factors causing the increasing incidence of human TBE in Sweden. Parasit Vectors 2012, 5, 184. [Google Scholar] [CrossRef] [PubMed]
- Jaenson, T.G.T.; Petersson, E.H.; Jaenson, D.G.E.; Kindberg, J.; Pettersson, J.H.; Hjertqvist, M.; Medlock, J.M.; Bengtsson, H. The importance of wildlife in the ecology and epidemiology of the TBE virus in Sweden: Incidence of human TBE correlates with abundance of deer and hares. Parasites Vectors 2018, 11, 477. [Google Scholar] [CrossRef] [PubMed]
- Kiffner, C.; Zucchini, W.; Schomaker, P.; Vor, T.; Hagedorn, P.; Niedrig, M.; Rühe, F. Determinants of tick-borne encephalitis in counties of southern Germany, 2001–2008. Int. J. Health Geogr. 2010, 9, 42. [Google Scholar] [CrossRef] [PubMed]
- Johnsen, K.; Boonstra, R.; Boutin, S.; Devineau, O.; Krebs, C.J.; Andreassen, H.P. Surviving winter: Food, but not habitat structure, prevents crashes in cyclic vole populations. Ecol. Evol. 2017, 7, 115–124. [Google Scholar] [CrossRef]
- Heyman, P.; Cochez, C.; Simons, L.; Smets, L.; Saegerman, C. Breeding success of barn owls reflects risk of hantavirus infection. Vet. Rec. 2013, 172, 290. [Google Scholar] [CrossRef]
- Dumpis, U.; Crook, D.; Oksi, J. Tick-borne encephalitis. Clin. Infect. Dis. 1999, 28, 882–890. [Google Scholar] [CrossRef]
- Ličková, M.; Fumačová Havlíková, S.; Sláviková, M.; Mirko Slovák, L.; Drexler, J.F.; Klempa, B. Dermacentor reticulatus is a vector of tick-borne encephalitis virus. Ticks Tick Borne Dis. 2020, 11, 101414. [Google Scholar] [CrossRef] [PubMed]
- Chitimia-Dobler, L.; Lemhöfer, G.; Król, N.; Bestehorn, M.; Dobler, G.; Pfeffer, M. Repeated isolation of tick-borne encephalitis virus from adult Dermacentor reticulatus ticks in an endemic area in Germany. Parasit Vectors 2019, 12, 90. [Google Scholar] [CrossRef]
- Claerebout, E.; Losson, B.; Cochez, C.; Casaert, S.; Dalemans, A.-C.; De Cat, A.; Madder, M.; Saegerman, C.; Heyman, P.; Lempereur, L. Ticks and associated pathogens collected from dogs and cats in Belgium. Parasit Vectors 2013, 6, 183. [Google Scholar] [CrossRef]
- Springer, A.; Lindau, A.; Probst, J.; Drehmann, M.; Fachet, K.; Thoma, D.; Vineer, H.R.; Noll, M.; Dobler, G.; Mackenstedt, U.; et al. Update and prognosis of Dermacentor distribution in Germany: Nationwide occurrence of Dermacentor reticulatus. Front. Vet. Sci. 2022, 9, 1044597. [Google Scholar] [CrossRef] [PubMed]
- Kooyman, F.N.J.; Zweerus, H.; Nijsse, E.R.; Jongejan, F.; Wagenaar, J.A.; Broens, E.M. Monitoring of ticks and their pathogens from companion animals obtained by the "tekenscanner" application in The Netherlands. Parasitol Res. 2022, 121, 1887–1893. [Google Scholar] [CrossRef] [PubMed]
- Kiewra, D.; Szymanowski, M.; Czułowska, A.; Kolanek, A. The local-scale expansion of Dermacentor reticulatus ticks in Lower Silesia, SW Poland. Ticks Tick Borne Dis. 2021, 12, 101599. [Google Scholar] [CrossRef] [PubMed]
- Drehmann, M.; Springer, A.; Lindau, A.; Fachet, K.; Mai, S.; Thoma, D.; Schneider, C.R.; Chitimia-Dobler, L.; Bröker, M.; Dobler, G.; et al. The Spatial Distribution of Dermacentor Ticks (Ixodidae) in Germany-Evidence of a Continuing Spread of Dermacentor reticulatus. Front. Vet. Sci. 2020, 7, 578220. [Google Scholar] [CrossRef] [PubMed]
- Mierzejewska, E.J.; Estrada-Peña, A.; Alsarraf, M.; Kowalec, M.; Bajer, A. Mapping of Dermacentor reticulatus expansion in Poland in 2012–2014. Ticks Tick Borne Dis. 2016, 7, 94–106. [Google Scholar] [CrossRef]
- Földvári, G.; Široký, P.; Szekeres, S.; Majoros, G.; Sprong, H. Dermacentor reticulatus: A vector on the rise. Parasit Vectors. 2016, 9, 314. [Google Scholar] [CrossRef]
- ECDC-EFSA Tick Maps [Internet]. Stockholm: ECDC. 2022. Available online: https://ecdc.europa.eu/en/disease-vectors/surveillance-and-disease-data/tick-maps (accessed on 15 March 2023).
- Estrada-Peña, A.; de la Fuente, J. Species interactions in occurrence data for a community of tick-transmitted pathogens. Sci. Data 2016, 3, 160056. [Google Scholar] [CrossRef]
- Morgan, M.G. Use (and abuse) of expert elicitation in support of decision making for public policy. Proc. Natl. Acad. Sci. USA 2014, 111, 7176–7184. [Google Scholar] [CrossRef]

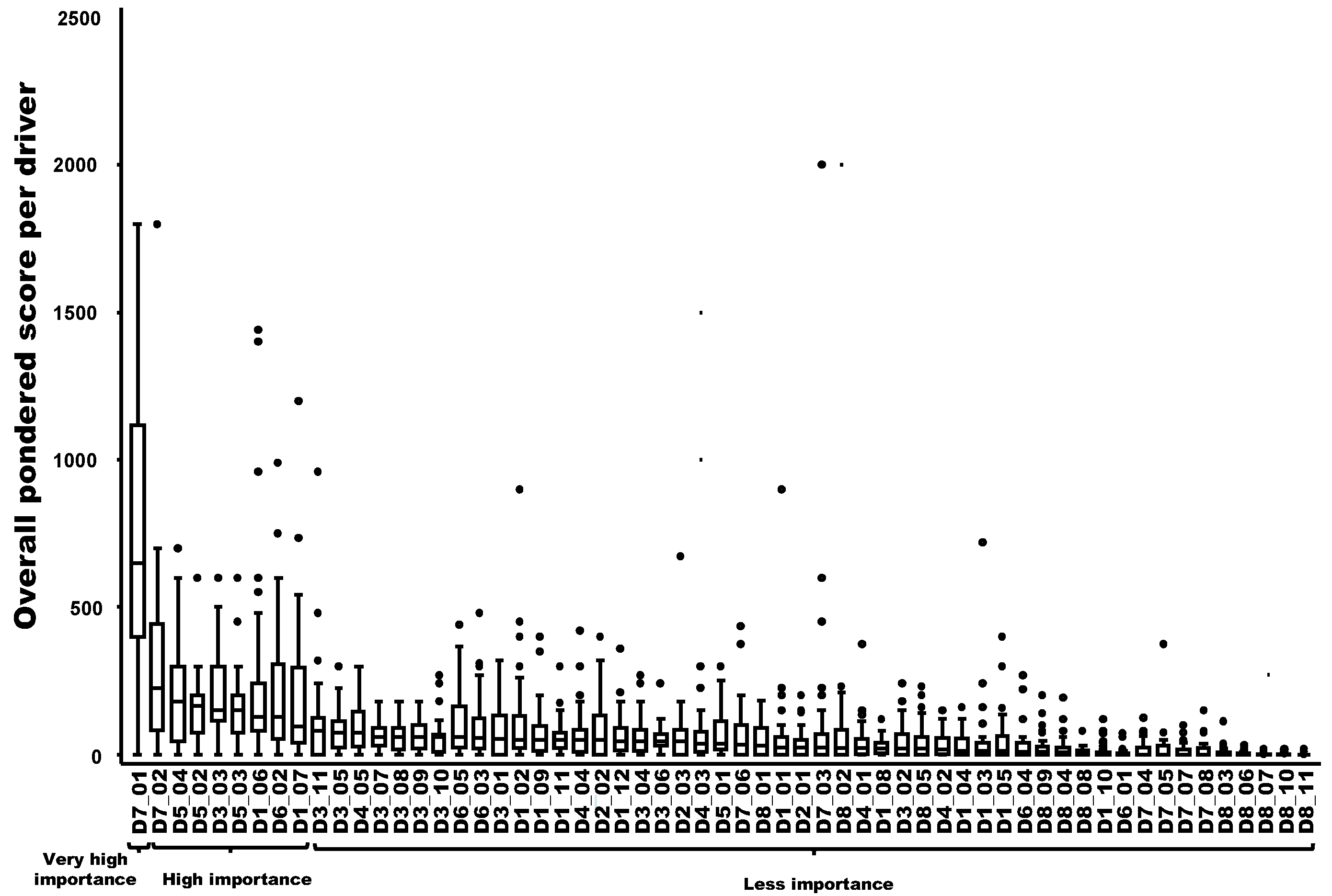
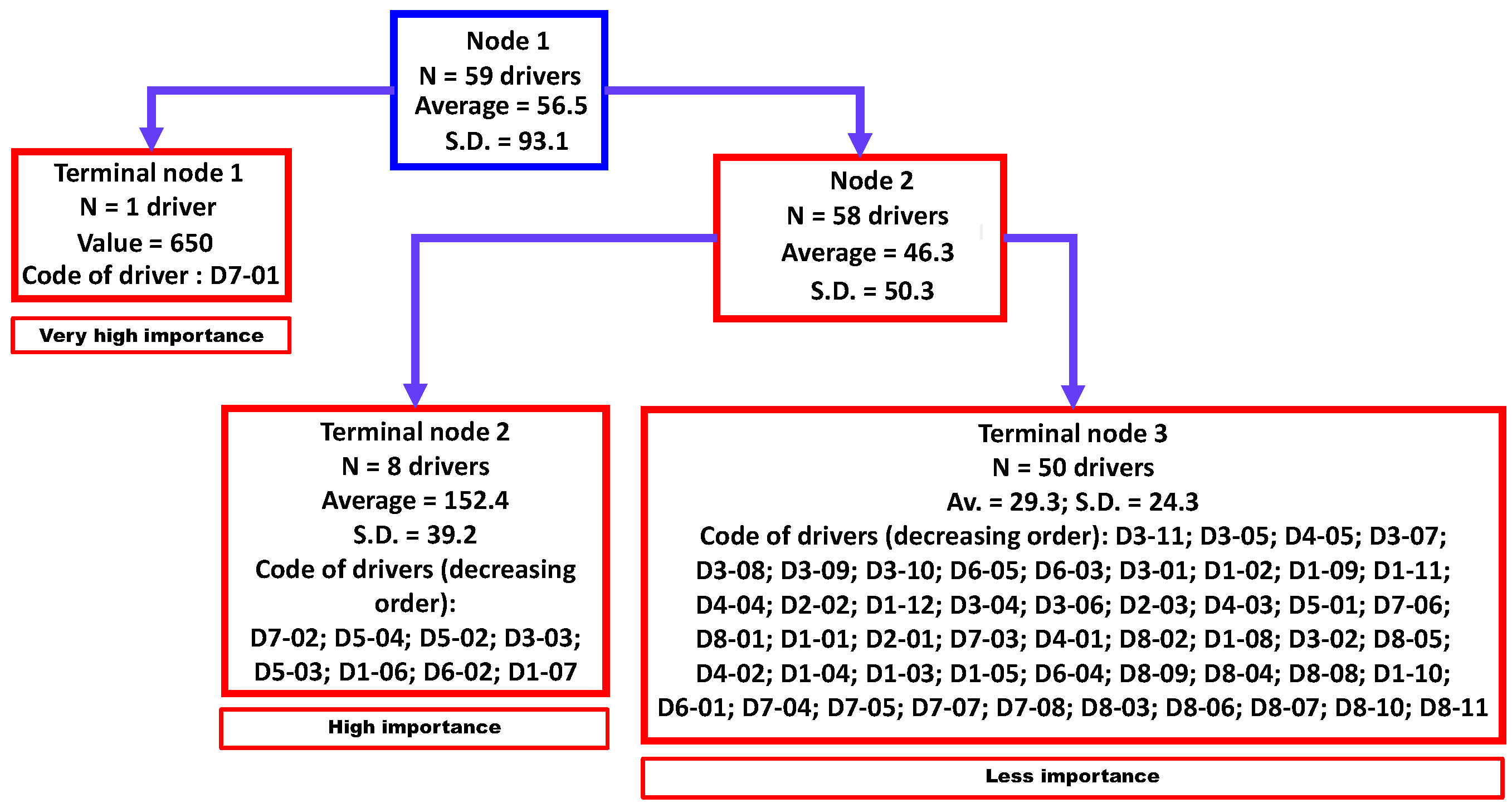

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Saegerman, C.; Humblet, M.-F.; Leandri, M.; Gonzalez, G.; Heyman, P.; Sprong, H.; L’Hostis, M.; Moutailler, S.; Bonnet, S.I.; Haddad, N.; et al. First Expert Elicitation of Knowledge on Possible Drivers of Observed Increasing Human Cases of Tick-Borne Encephalitis in Europe. Viruses 2023, 15, 791. https://doi.org/10.3390/v15030791
Saegerman C, Humblet M-F, Leandri M, Gonzalez G, Heyman P, Sprong H, L’Hostis M, Moutailler S, Bonnet SI, Haddad N, et al. First Expert Elicitation of Knowledge on Possible Drivers of Observed Increasing Human Cases of Tick-Borne Encephalitis in Europe. Viruses. 2023; 15(3):791. https://doi.org/10.3390/v15030791
Chicago/Turabian StyleSaegerman, Claude, Marie-France Humblet, Marc Leandri, Gaëlle Gonzalez, Paul Heyman, Hein Sprong, Monique L’Hostis, Sara Moutailler, Sarah I. Bonnet, Nadia Haddad, and et al. 2023. "First Expert Elicitation of Knowledge on Possible Drivers of Observed Increasing Human Cases of Tick-Borne Encephalitis in Europe" Viruses 15, no. 3: 791. https://doi.org/10.3390/v15030791
APA StyleSaegerman, C., Humblet, M.-F., Leandri, M., Gonzalez, G., Heyman, P., Sprong, H., L’Hostis, M., Moutailler, S., Bonnet, S. I., Haddad, N., Boulanger, N., Leib, S. L., Hoch, T., Thiry, E., Bournez, L., Kerlik, J., Velay, A., Jore, S., Jourdain, E., ... Purse, B. (2023). First Expert Elicitation of Knowledge on Possible Drivers of Observed Increasing Human Cases of Tick-Borne Encephalitis in Europe. Viruses, 15(3), 791. https://doi.org/10.3390/v15030791









