Administration of Bacteriophages via Nebulization during Mechanical Ventilation: In Vitro Study and Lung Deposition in Macaques
Abstract
1. Introduction
2. Materials and Methods
2.1. Strains
2.2. Formulation of Phage Anti-Pseudomonas Aeruginosa
2.2.1. Phage Discovery
2.2.2. Selection of Virulent Phage
2.2.3. Phage Titration
- Plaque-forming units (PFUs) can be seen on the bacterial lawn when the phage is diluted with a total lysis of the spot at high phage concentrations, which correspond to confluent PFUs.
- A partial lysis of the spot is observed without any PFU at further dilutions.
- Total absence of lysis, whatever the phage dilution.
2.2.4. Phage Efficiency
2.2.5. Phage Mix Preparation for Nebulization
2.3. Nebulization Parameters Influencing Phage Viability
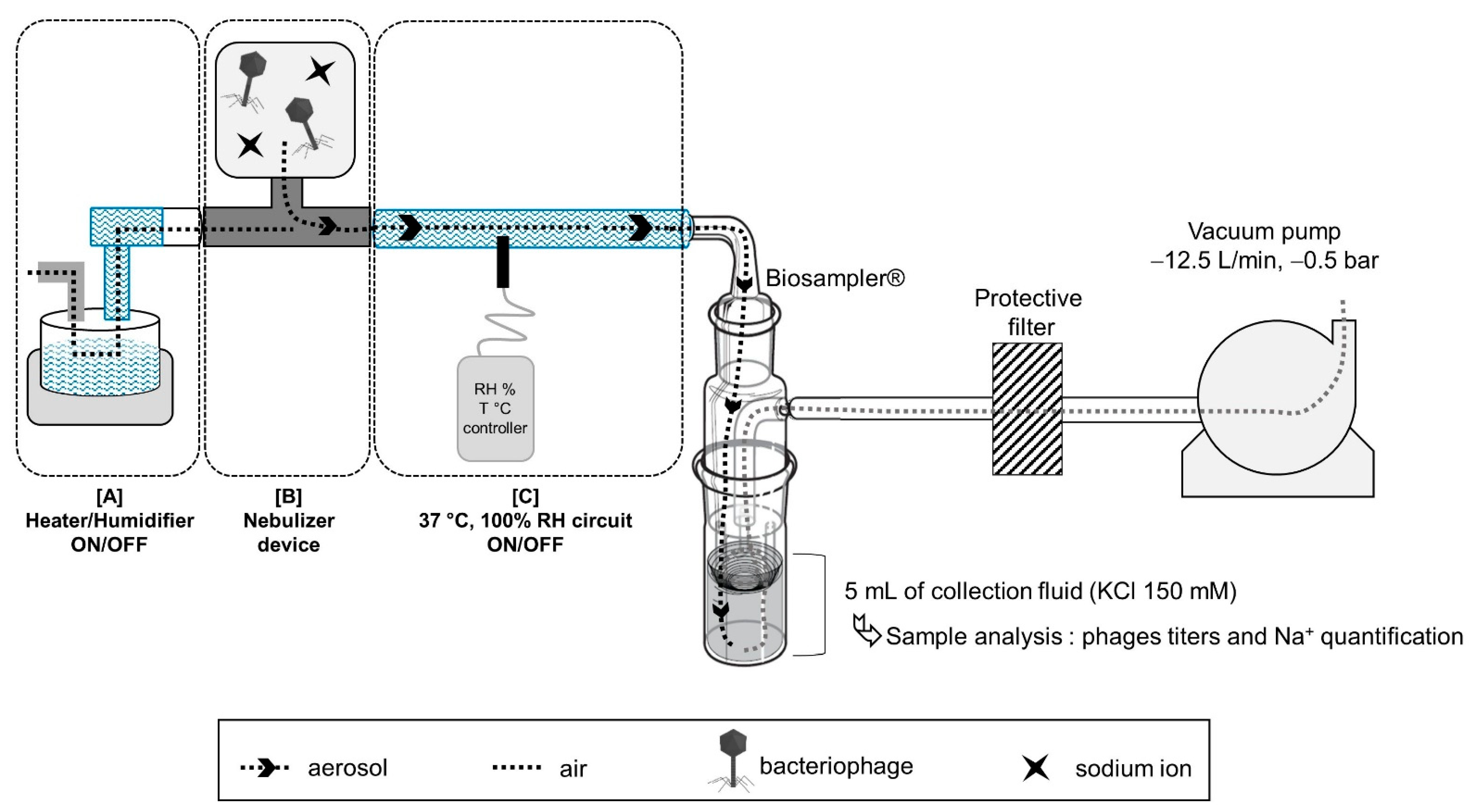
2.3.1. Nebulizer Devices, Phage Aerosol Collections and Viability Characterizations
2.3.2. Phage Aerosol Collections and Viability Characterizations
2.3.3. Influence of Nebulizer Temperature
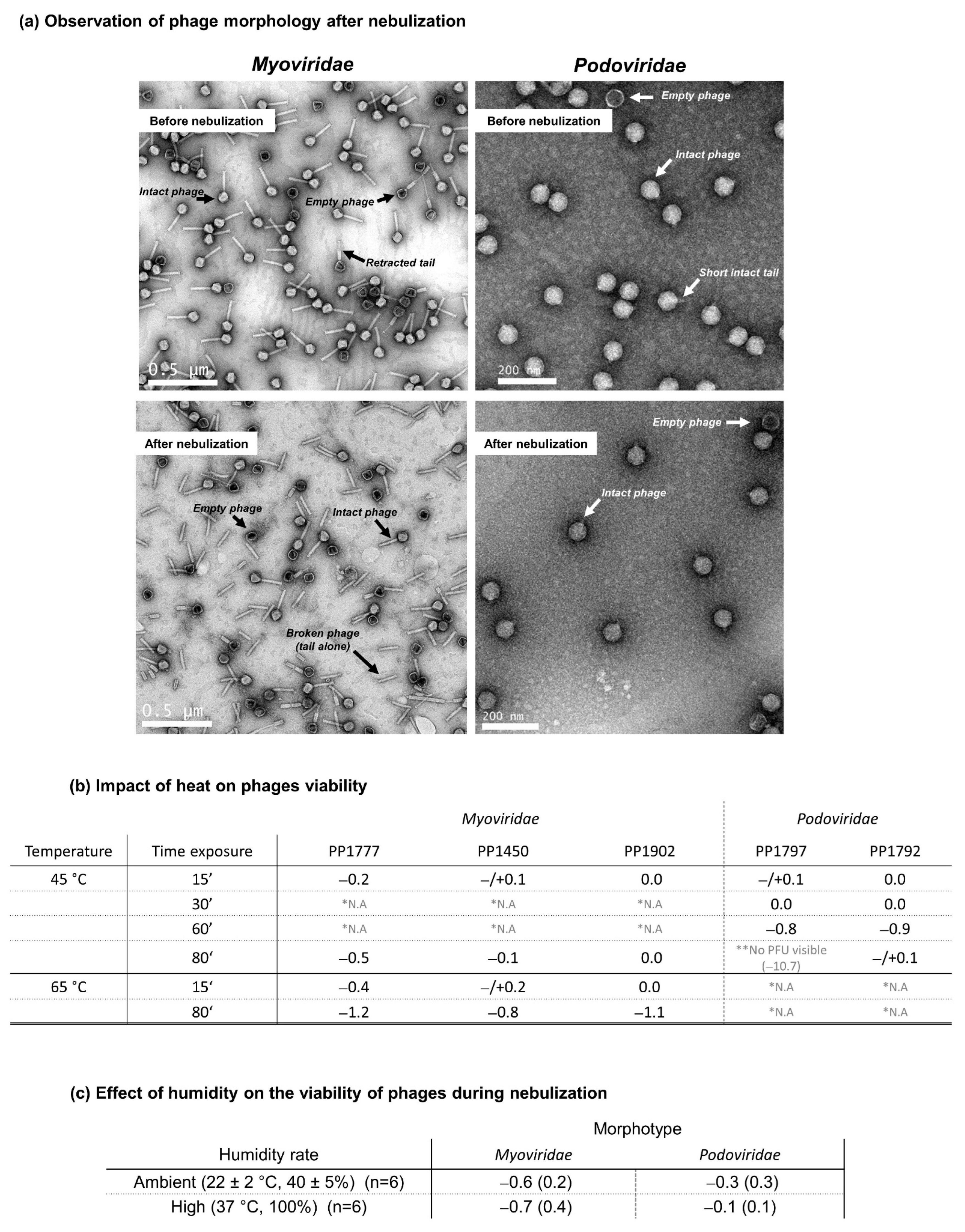
2.3.4. Phage Morphological Analyses before and after Nebulization
2.4. Aerosol Phage Administration in a Human MV Model
2.5. In Vivo Lung Deposition Measurements in Ventilated Non-Human Primate
2.5.1. Animals
2.5.2. Perfusion Scintigraphy
2.5.3. Set-Up for Aerosol Delivery under MV and Scintigraphy Gamma Camera Imaging
2.6. Statistical Analysis
3. Results
3.1. Selection and Evaluation of Phage Mix and Efficacy on Clinical Strains
3.1.1. Phage Discovery
3.1.2. Phage Selection and Host Range
3.2. Nebulization Parameters Influencing Phage Viability
3.3. Phage Morphological Analysis
3.4. Effect of Temperature during Nebulization Process
3.5. Aerosol Phage Administration in a Human Mechanical Ventilation Model
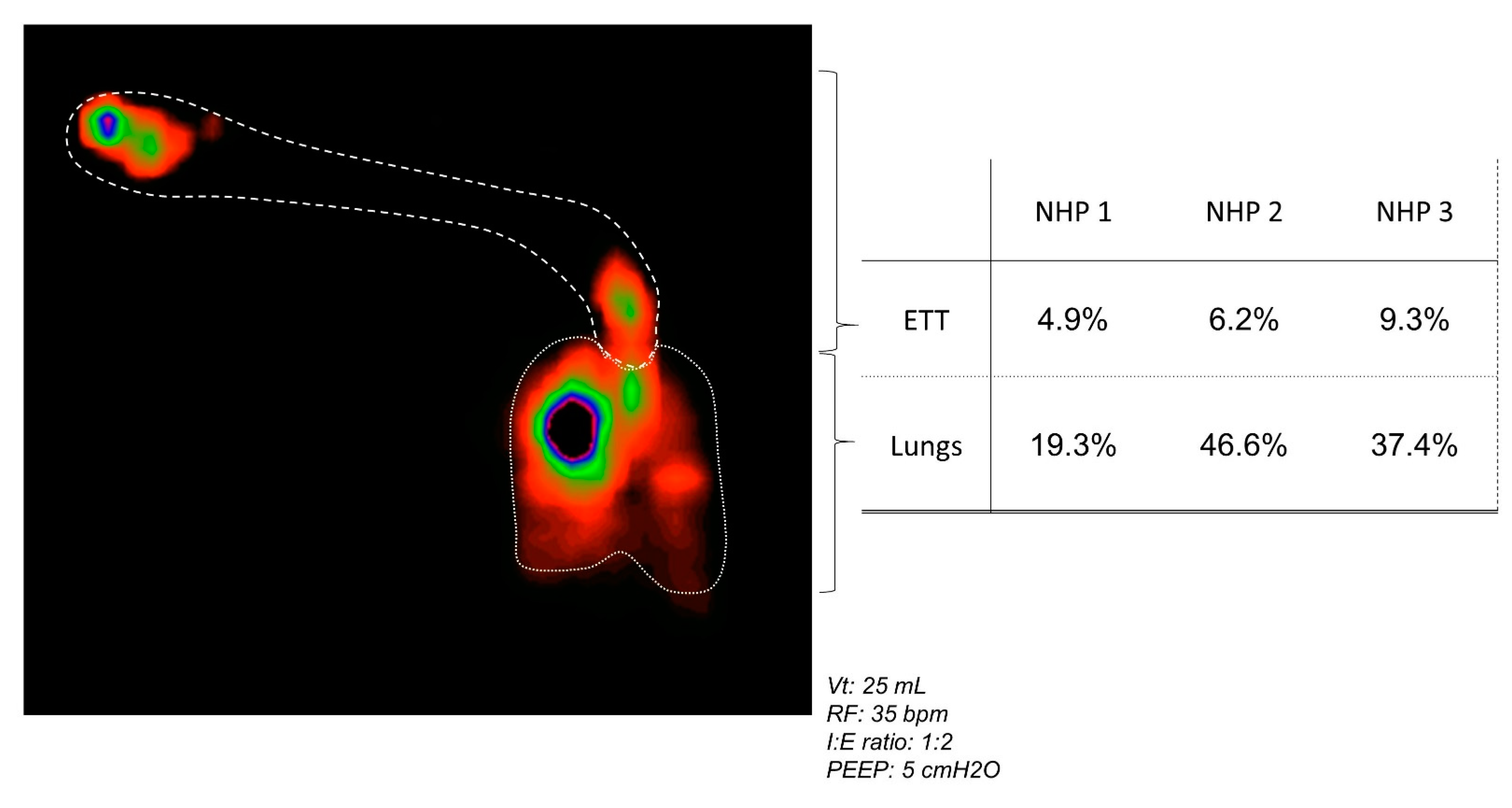
3.6. In Vivo Aerosol Deposition in NHP
4. Discussion
5. Conclusions
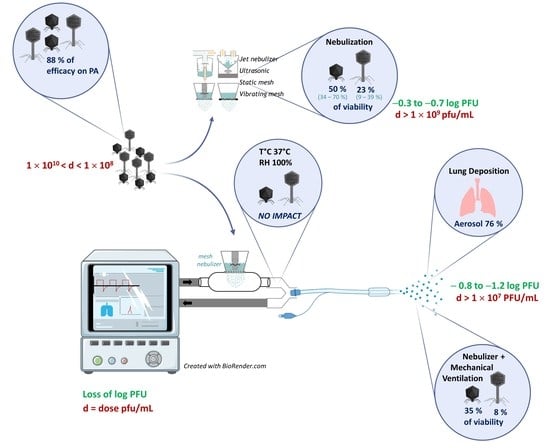
- A set of four anti-PA phages was selected, with a coverage of 87.8% on the international PA reference panel, and produced as a drug pharmaceutical product.
- Five anti-PA phages were tested for nebulization: Myoviridae are significantly more sensitive to nebulization than Podoviridae; their loss of infectivity is linked to structural damage with tail detachment. This damage appeared to be independent of temperature stress but rather due to shearing stress during the nebulization operation.
- The nebulization process alone led to a loss of 0.30–0.65 log of infective phage titers.
- Mesh nebulizers combine a high output delivery with a moderate impact on phage survival.
- There is no effect of humidification on phage viability during mechanical ventilation.
- According to the in vitro model of adult mechanical ventilation, the lung deposition prediction of viable phage particles ranges from 6% to 26% of the phages loaded in the nebulizer.
- According to the intubated NHPs in the in vivo model, the lung deposition prediction ranges from 8% to 15%. In this pediatric model of phages, we observed an asymmetrical deposition in the lungs.
- A phage dose of 1 × 109 PFU/mL is efficient for nebulization via a mesh nebulizer during MV and it shows a good prediction between in vitro and in vivo (lung) concentrations.
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- World Health Organization. Available online: //www.who.int/news/item/27-02-2017-who-publishes-list-of-bacteria-for-which-new-antibiotics-are-urgently-needed (accessed on 13 December 2022).
- Centers for Disease Control and Prevention Website. CDC’s Antibiotic Resistance Threats in the United States. AR Threats Report. 2019. Available online: https://www.cdc.gov/drugresistance/biggest-threats.html (accessed on 13 December 2022).
- Fernández-Barat, L.; Ferrer, M.; De Rosa, F.; Gabarrús, A.; Esperatti, M.; Terraneo, S.; Rinaudo, M.; Li Bassi, G.; Torres, A. Intensive Care Unit-Acquired Pneumonia Due to Pseudomonas aeruginosa with and without Multidrug Resistance. J. Infect. 2017, 74, 142–152. [Google Scholar] [CrossRef]
- Chastre, J.; Fagon, J.-Y. Ventilator-Associated Pneumonia. Am. J. Respir. Crit. Care Med. 2002, 165, 867–903. [Google Scholar] [CrossRef]
- Chastre, J. Antibiotic Prescribing for Ventilator-Associated Pneumonia: Get It Right from the Beginning but Be Able to Rapidly Deescalate. Intensive Care Med. 2005, 31, 1463–1465. [Google Scholar] [CrossRef]
- Porzecanski, I.; Bowton, D.L. Diagnosis and Treatment of Ventilator-Associated Pneumonia. Chest 2006, 130, 597–604. [Google Scholar] [CrossRef]
- Goldstein, I.; Wallet, F.; Nicolas-Robin, A.; Ferrari, F.; Marquette, C.-H.; Rouby, J.-J. Lung Deposition and Efficiency of Nebulized Amikacin during Escherichia Coli Pneumonia in Ventilated Piglets. Am. J. Respir. Crit. Care Med. 2002, 166, 1375–1381. [Google Scholar] [CrossRef]
- Lu, Q.; Yang, J.; Liu, Z.; Gutierrez, C.; Aymard, G.; Rouby, J.-J.; Nebulized Antibiotics Study Group. Nebulized Ceftazidime and Amikacin in Ventilator-Associated Pneumonia Caused by Pseudomonas aeruginosa. Am. J. Respir. Crit. Care Med. 2011, 184, 106–115. [Google Scholar] [CrossRef]
- Lu, Q.; Luo, R.; Bodin, L.; Yang, J.; Zahr, N.; Aubry, A.; Golmard, J.-L.; Rouby, J.-J.; Nebulized Antibiotics Study Group. Efficacy of High-Dose Nebulized Colistin in Ventilator-Associated Pneumonia Caused by Multidrug-Resistant Pseudomonas aeruginosa and Acinetobacter baumannii. Anesthesiology 2012, 117, 1335–1347. [Google Scholar] [CrossRef]
- Niederman, M.S.; Chastre, J.; Corkery, K.; Fink, J.B.; Luyt, C.-E.; García, M.S. BAY41-6551 Achieves Bactericidal Tracheal Aspirate Amikacin Concentrations in Mechanically Ventilated Patients with Gram-Negative Pneumonia. Intensive Care Med. 2012, 38, 263–271. [Google Scholar] [CrossRef]
- Kollef, M.H.; Ricard, J.-D.; Roux, D.; Francois, B.; Ischaki, E.; Rozgonyi, Z.; Boulain, T.; Ivanyi, Z.; János, G.; Garot, D.; et al. A Randomized Trial of the Amikacin Fosfomycin Inhalation System for the Adjunctive Therapy of Gram-Negative Ventilator-Associated Pneumonia: IASIS Trial. Chest 2017, 151, 1239–1246. [Google Scholar] [CrossRef]
- Uyttebroek, S.; Chen, B.; Onsea, J.; Ruythooren, F.; Debaveye, Y.; Devolder, D.; Spriet, I.; Depypere, M.; Wagemans, J.; Lavigne, R.; et al. Safety and Efficacy of Phage Therapy in Difficult-to-Treat Infections: A Systematic Review. Lancet Infect. Dis. 2022, 22, e208–e220. [Google Scholar] [CrossRef]
- Harper, D.R.; Parracho, H.M.R.T.; Walker, J.; Sharp, R.; Hughes, G.; Werthén, M.; Lehman, S.; Morales, S. Bacteriophages and Biofilms. Antibiotics 2014, 3, 270–284. [Google Scholar] [CrossRef]
- Alves, D.R.; Perez-Esteban, P.; Kot, W.; Bean, J.E.; Arnot, T.; Hansen, L.H.; Enright, M.C.; Jenkins, A.T.A. A Novel Bacteriophage Cocktail Reduces and Disperses Pseudomonas aeruginosa Biofilms under Static and Flow Conditions. Microb. Biotechnol. 2016, 9, 61–74. [Google Scholar] [CrossRef]
- Chan, B.K.; Sistrom, M.; Wertz, J.E.; Kortright, K.E.; Narayan, D.; Turner, P.E. Phage Selection Restores Antibiotic Sensitivity in MDR Pseudomonas aeruginosa. Sci. Rep. 2016, 6, 26717. [Google Scholar] [CrossRef]
- Morello, E.; Saussereau, E.; Maura, D.; Huerre, M.; Touqui, L.; Debarbieux, L. Pulmonary Bacteriophage Therapy on Pseudomonas aeruginosa Cystic Fibrosis Strains: First Steps towards Treatment and Prevention. PLoS ONE 2011, 6, e16963. [Google Scholar] [CrossRef]
- Debarbieux, L.; Leduc, D.; Maura, D.; Morello, E.; Criscuolo, A.; Grossi, O.; Balloy, V.; Touqui, L. Bacteriophages Can Treat and Prevent Pseudomonas aeruginosa Lung Infections. J. Infect. Dis. 2010, 201, 1096–1104. [Google Scholar] [CrossRef]
- Bodier-Montagutelli, E.; Morello, E.; L’Hostis, G.; Guillon, A.; Dalloneau, E.; Respaud, R.; Pallaoro, N.; Blois, H.; Vecellio, L.; Gabard, J.; et al. Inhaled Phage Therapy: A Promising and Challenging Approach to Treat Bacterial Respiratory Infections. Expert Opin. Drug Deliv. 2017, 14, 959–972. [Google Scholar] [CrossRef]
- Guillon, A.; Pardessus, J.; L’Hostis, G.; Fevre, C.; Barc, C.; Dalloneau, E.; Jouan, Y.; Bodier-Montagutelli, E.; Perez, Y.; Thorey, C.; et al. Inhaled Bacteriophage Therapy in a Porcine Model of Pneumonia Caused by Pseudomonas aeruginosa during Mechanical Ventilation. Br. J. Pharmacol. 2021, 178, 3829–3842. [Google Scholar] [CrossRef]
- Sahota, J.S.; Smith, C.M.; Radhakrishnan, P.; Winstanley, C.; Goderdzishvili, M.; Chanishvili, N.; Kadioglu, A.; O’Callaghan, C.; Clokie, M.R.J. Bacteriophage Delivery by Nebulization and Efficacy Against Phenotypically Diverse Pseudomonas aeruginosa from Cystic Fibrosis Patients. J. Aerosol Med. Pulm. Drug Deliv. 2015, 28, 353–360. [Google Scholar] [CrossRef]
- Astudillo, A.; Leung, S.S.Y.; Kutter, E.; Morales, S.; Chan, H.-K. Nebulization Effects on Structural Stability of Bacteriophage PEV 44. Eur. J. Pharm. Biopharm. 2018, 125, 124–130. [Google Scholar] [CrossRef]
- Leung, S.S.Y.; Carrigy, N.B.; Vehring, R.; Finlay, W.H.; Morales, S.; Carter, E.A.; Britton, W.J.; Kutter, E.; Chan, H.-K. Jet Nebulization of Bacteriophages with Different Tail Morphologies-Structural Effects. Int. J. Pharm. 2019, 554, 322–326. [Google Scholar] [CrossRef]
- Verreault, D.; Marcoux-Voiselle, M.; Turgeon, N.; Moineau, S.; Duchaine, C. Resistance of Aerosolized Bacterial Viruses to Relative Humidity and Temperature. Appl. Environ. Microbiol. 2015, 81, 7305–7311. [Google Scholar] [CrossRef]
- De Soyza, A.; Hall, A.J.; Mahenthiralingam, E.; Drevinek, P.; Kaca, W.; Drulis-Kawa, Z.; Stoitsova, S.R.; Toth, V.; Coenye, T.; Zlosnik, J.E.A.; et al. Developing an International Pseudomonas aeruginosa Reference Panel. Microbiolopen 2013, 2, 1010–1023. [Google Scholar] [CrossRef]
- Pirnay, J.-P.; Bilocq, F.; Pot, B.; Cornelis, P.; Zizi, M.; Van Eldere, J.; Deschaght, P.; Vaneechoutte, M.; Jennes, S.; Pitt, T.; et al. Pseudomonas aeruginosa Population Structure Revisited. PLoS ONE 2009, 4, e7740. [Google Scholar] [CrossRef]
- Van Twest, R.; Kropinski, A.M. Bacteriophage Enrichment from Water and Soil. Methods Mol. Biol. 2009, 501, 15–21. [Google Scholar] [CrossRef]
- RAST v2.0 Server. Available online: https://web.archive.org/web/20221213144205/https://rast.nmpdr.org/ (accessed on 13 December 2022).
- Hockenberry, A.J.; Wilke, C.O. BACPHLIP: Predicting Bacteriophage Lifestyle from Conserved Protein Domains. PeerJ 2021, 9, e11396. [Google Scholar] [CrossRef]
- Alcock, B.P.; Huynh, W.; Chalil, R.; Smith, K.W.; Raphenya, A.R.; Wlodarski, M.A.; Edalatmand, A.; Petkau, A.; Syed, S.A.; Tsang, K.K.; et al. CARD 2023: Expanded Curation, Support for Machine Learning, and Resistome Prediction at the Comprehensive Antibiotic Resistance Database. Nucleic Acids Res. 2023, 51, D690–D699. [Google Scholar] [CrossRef]
- Liu, B.; Zheng, D.; Zhou, S.; Chen, L.; Yang, J. VFDB 2022: A General Classification Scheme for Bacterial Virulence Factors. Nucleic Acids Res. 2022, 50, D912–D917. [Google Scholar] [CrossRef]
- Arndt, D.; Grant, J.R.; Marcu, A.; Sajed, T.; Pon, A.; Liang, Y.; Wishart, D.S. PHASTER: A Better, Faster Version of the PHAST Phage Search Tool. Nucleic Acids Res. 2016, 44, W16–W21. [Google Scholar] [CrossRef]
- Zhou, Y.; Liang, Y.; Lynch, K.H.; Dennis, J.J.; Wishart, D.S. PHAST: A Fast Phage Search Tool. Nucleic Acids Res. 2011, 39, W347–W352. [Google Scholar] [CrossRef]
- Kutter, E. Phage Host Range and Efficiency of Plating. Methods Mol. Biol. 2009, 501, 141–149. [Google Scholar] [CrossRef]
- Beck-Broichsitter, M.; Kleimann, P.; Schmehl, T.; Betz, T.; Bakowsky, U.; Kissel, T.; Seeger, W. Impact of Lyoprotectants for the Stabilization of Biodegradable Nanoparticles on the Performance of Air-Jet, Ultrasonic, and Vibrating-Mesh Nebulizers. Eur. J. Pharm. Biopharm. 2012, 82, 272–280. [Google Scholar] [CrossRef]
- Boulanger, P. Purification of Bacteriophages and SDS-PAGE Analysis of Phage Structural Proteins from Ghost Particles. Methods Mol. Biol. 2009, 502, 227–238. [Google Scholar] [CrossRef]
- Boukhettala, N.; Porée, T.; Diot, P.; Vecellio, L. In Vitro Performance of Spacers for Aerosol Delivery during Adult Mechanical Ventilation. J. Aerosol Med. Pulm. Drug Deliv. 2015, 28, 130–136. [Google Scholar] [CrossRef]
- Newman, S.P. Scintigraphic Assessment of Therapeutic Aerosols. Crit. Rev. Ther. Drug Carr. Syst. 1993, 10, 65–109. [Google Scholar]
- Dubus, J.C.; Vecellio, L.; De Monte, M.; Fink, J.B.; Grimbert, D.; Montharu, J.; Valat, C.; Behan, N.; Diot, P. Aerosol Deposition in Neonatal Ventilation. Pediatr. Res. 2005, 58, 10–14. [Google Scholar] [CrossRef]
- Green, S.I.; Kaelber, J.T.; Ma, L.; Trautner, B.W.; Ramig, R.F.; Maresso, A.W. Bacteriophages from ExPEC Reservoirs Kill Pandemic Multidrug-Resistant Strains of Clonal Group ST131 in Animal Models of Bacteremia. Sci. Rep. 2017, 7, 46151. [Google Scholar] [CrossRef]
- Antimicrobial Resistance Collaborators. Global Burden of Bacterial Antimicrobial Resistance in 2019: A Systematic Analysis. Lancet 2022, 399, 629–655. [Google Scholar] [CrossRef]
- Tumbarello, M.; De Pascale, G.; Trecarichi, E.M.; Spanu, T.; Antonicelli, F.; Maviglia, R.; Pennisi, M.A.; Bello, G.; Antonelli, M. Clinical Outcomes of Pseudomonas aeruginosa Pneumonia in Intensive Care Unit Patients. Intensive Care Med. 2013, 39, 682–692. [Google Scholar] [CrossRef]
- Loganathan, A.; Manohar, P.; Eniyan, K.; VinodKumar, C.S.; Leptihn, S.; Nachimuthu, R. Phage Therapy as a Revolutionary Medicine against Gram-Positive Bacterial Infections. Beni Suef Univ. J. Basic Appl. Sci. 2021, 10, 49. [Google Scholar] [CrossRef]
- Wang, X.; Xie, Z.; Zhao, J.; Zhu, Z.; Yang, C.; Liu, Y. Prospects of Inhaled Phage Therapy for Combatting Pulmonary Infections. Front. Cell. Infect. Microbiol. 2021, 11, 758392. [Google Scholar] [CrossRef]
- Zhang, Y.; Meng, B.; Wei, X.; Li, Y.; Wang, X.; Zheng, Y.; Wang, C.; Cui, L.; Zhao, X. Evaluation of Phage Therapy for Pulmonary Infection of Mouse by Liquid Aerosol-Exposure Pseudomonas aeruginosa. Infect. Drug Resist. 2021, 14, 4457–4469. [Google Scholar] [CrossRef]
- Geagea, H.; Labrie, S.J.; Subirade, M.; Moineau, S. The Tape Measure Protein Is Involved in the Heat Stability of Lactococcus Lactis Phages. Appl. Environ. Microbiol. 2018, 84, e02082-17. [Google Scholar] [CrossRef]
- Solomita, M.; Daroowalla, F.; Leblanc, D.S.; Smaldone, G.C. Y-Piece Temperature and Humidification during Mechanical Ventilation. Respir. Care 2009, 54, 480–486. [Google Scholar]
- Majoral, C.; Coates, A.L.; Le Pape, A.; Vecellio, L. Humidified and Heated Cascade Impactor for Aerosol Sizing. Front. Bioeng. Biotechnol. 2020, 8, 589782. [Google Scholar] [CrossRef]
- Analytical Methods, Subchapter “2.9.44. Characterization of Nebulization Preparations”. In European Pharmacopoeia 7.3; Council Of Europe: Strasbourg, France, 2012; pp. 4067–4070.
- Ehrmann, S.; Chastre, J.; Diot, P.; Lu, Q. Nebulized Antibiotics in Mechanically Ventilated Patients: A Challenge for Translational Research from Technology to Clinical Care. Ann. Intensive Care 2017, 7, 78. [Google Scholar] [CrossRef]
- Szychowiak, P.; Desgrouas, M.; Ehrmann, S. Inhaled Antibiotics in Critical Care: State of the Art and Future Perspectives. Infect. Dis. Now 2022, 52, 327–333. [Google Scholar] [CrossRef]
- Gelman, D.; Yerushalmy, O.; Alkalay-Oren, S.; Rakov, C.; Ben-Porat, S.; Khalifa, L.; Adler, K.; Abdalrhman, M.; Coppenhagen-Glazer, S.; Aslam, S.; et al. Clinical Phage Microbiology: A Suggested Framework and Recommendations for the in-Vitro Matching Steps of Phage Therapy. Lancet Microbe 2021, 2, e555–e563. [Google Scholar] [CrossRef] [PubMed]
- Daubie, V.; Chalhoub, H.; Blasdel, B.; Dahma, H.; Merabishvili, M.; Glonti, T.; De Vos, N.; Quintens, J.; Pirnay, J.-P.; Hallin, M.; et al. Determination of Phage Susceptibility as a Clinical Diagnostic Tool: A Routine Perspective. Front. Cell. Infect. Microbiol. 2022, 12, 1000721. [Google Scholar] [CrossRef]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Le Guellec, S.; Pardessus, J.; Bodier-Montagutelli, E.; L’Hostis, G.; Dalloneau, E.; Piel, D.; Samaï, H.C.; Guillon, A.; Mujic, E.; Guillot-Combe, E.; et al. Administration of Bacteriophages via Nebulization during Mechanical Ventilation: In Vitro Study and Lung Deposition in Macaques. Viruses 2023, 15, 602. https://doi.org/10.3390/v15030602
Le Guellec S, Pardessus J, Bodier-Montagutelli E, L’Hostis G, Dalloneau E, Piel D, Samaï HC, Guillon A, Mujic E, Guillot-Combe E, et al. Administration of Bacteriophages via Nebulization during Mechanical Ventilation: In Vitro Study and Lung Deposition in Macaques. Viruses. 2023; 15(3):602. https://doi.org/10.3390/v15030602
Chicago/Turabian StyleLe Guellec, Sandrine, Jeoffrey Pardessus, Elsa Bodier-Montagutelli, Guillaume L’Hostis, Emilie Dalloneau, Damien Piel, Hakim Chouky Samaï, Antoine Guillon, Elvir Mujic, Emmanuelle Guillot-Combe, and et al. 2023. "Administration of Bacteriophages via Nebulization during Mechanical Ventilation: In Vitro Study and Lung Deposition in Macaques" Viruses 15, no. 3: 602. https://doi.org/10.3390/v15030602
APA StyleLe Guellec, S., Pardessus, J., Bodier-Montagutelli, E., L’Hostis, G., Dalloneau, E., Piel, D., Samaï, H. C., Guillon, A., Mujic, E., Guillot-Combe, E., Ehrmann, S., Morello, E., Gabard, J., Heuzé-Vourc’h, N., Fevre, C., & Vecellio, L. (2023). Administration of Bacteriophages via Nebulization during Mechanical Ventilation: In Vitro Study and Lung Deposition in Macaques. Viruses, 15(3), 602. https://doi.org/10.3390/v15030602