Flavonoids with Anti-Herpes Simplex Virus Properties: Deciphering Their Mechanisms in Disrupting the Viral Life Cycle
Abstract
1. Introduction
2. A Brief Overview of the HSV Life Cycle
3. Bioactive Flavonoids: Nature’s Antiviral Arsenal
4. Blocking HSV-1 Infection via Flavonoids
5. Blocking HSV-2 Infection via Flavonoids
6. Flavonoid-Enhanced Approaches to Elevate HSV Therapy
6.1. Nanoparticles and Gel-Formulation-Based Strategies
6.2. Physical Properties Targeting Approach
6.3. Combination Therapies
7. Clinical Studies
8. Conclusions, Challenges, and Future Prospects
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Tognarelli, E.I.; Palomino, T.F.; Corrales, N.; Bueno, S.M.; Kalergis, A.M.; González, P.A. Herpes Simplex Virus Evasion of Early Host Antiviral Responses. Front. Cell. Infect. Microbiol. 2019, 9, 127. [Google Scholar] [CrossRef] [PubMed]
- Brezáni, V.; Leláková, V.; Hassan, S.T.S.; Berchová-Bímová, K.; Nový, P.; Klouček, P.; Maršík, P.; Dall’Acqua, S.; Hošek, J.; Šmejkal, K. Anti-Infectivity against Herpes Simplex Virus and Selected Microbes and Anti-Inflammatory Activities of Compounds Isolated from Eucalyptus globulus Labill. Viruses 2018, 10, 360. [Google Scholar] [CrossRef] [PubMed]
- Omarova, S.; Cannon, A.; Weiss, W.; Bruccoleri, A.; Puccio, J. Genital Herpes Simplex Virus—An Updated Review. Adv. Pediatr. 2022, 69, 149–162. [Google Scholar] [CrossRef] [PubMed]
- Petti, S.; Lodi, G. The Controversial Natural History of Oral Herpes Simplex Virus Type 1 Infection. Oral Dis. 2019, 25, 1850–1865. [Google Scholar] [CrossRef] [PubMed]
- Hendrickx, D.M.; Sousa, J.D.; Libin, P.J.K.; Delva, W.; Liesenborgs, J.; Hens, N.; Müller, V.; Vandamme, A.-M. Comparison of Two Simulators for Individual Based Models in HIV Epidemiology in a Population with HSV 2 in Yaoundé (Cameroon). Sci. Rep. 2021, 11, 14696. [Google Scholar] [CrossRef] [PubMed]
- Desai, D.V.; Kulkarni, S.S. Herpes Simplex Virus: The Interplay Between HSV, Host, and HIV-1. Viral Immunol. 2015, 28, 546–555. [Google Scholar] [CrossRef] [PubMed]
- Pinninti, S.G.; Kimberlin, D.W. Neonatal Herpes Simplex Virus Infections. Semin. Perinatol. 2018, 42, 168–175. [Google Scholar] [CrossRef]
- Herpes Simplex Virus. Available online: https://www.who.int/news-room/fact-sheets/detail/herpes-simplex-virus (accessed on 5 November 2023).
- Alareeki, A.; Osman, A.M.M.; Khandakji, M.N.; Looker, K.J.; Harfouche, M.; Abu-Raddad, L.J. Epidemiology of Herpes Simplex Virus Type 2 in Europe: Systematic Review, Meta-Analyses, and Meta-Regressions. Lancet Reg. Health—Eur. 2023, 25, 100558. [Google Scholar] [CrossRef]
- Samies, N.L.; James, S.H. Prevention and Treatment of Neonatal Herpes Simplex Virus Infection. Antiviral Res. 2020, 176, 104721. [Google Scholar] [CrossRef]
- Fatahzadeh, M.; Schwartz, R.A. Human Herpes Simplex Virus Infections: Epidemiology, Pathogenesis, Symptomatology, Diagnosis, and Management. J. Am. Acad. Dermatol. 2007, 57, 737–763, quiz 764–766. [Google Scholar] [CrossRef]
- Kurt-Jones, E.A.; Orzalli, M.H.; Knipe, D.M. Innate Immune Mechanisms and Herpes Simplex Virus Infection and Disease. Adv. Anat. Embryol. Cell Biol. 2017, 223, 49–75. [Google Scholar] [CrossRef] [PubMed]
- Poole, C.L.; James, S.H. Antiviral Therapies for Herpesviruses: Current Agents and New Directions. Clin. Ther. 2018, 40, 1282–1298. [Google Scholar] [CrossRef] [PubMed]
- Schalkwijk, H.H.; Snoeck, R.; Andrei, G. Acyclovir Resistance in Herpes Simplex Viruses: Prevalence and Therapeutic Alternatives. Biochem. Pharmacol. 2022, 206, 115322. [Google Scholar] [CrossRef] [PubMed]
- Piret, J.; Boivin, G. Resistance of Herpes Simplex Viruses to Nucleoside Analogues: Mechanisms, Prevalence, and Management. Antimicrob. Agents Chemother. 2011, 55, 459–472. [Google Scholar] [CrossRef]
- Hassan, S.T.S.; Šudomová, M.; Berchová-Bímová, K.; Šmejkal, K.; Echeverría, J. Psoromic Acid, a Lichen-Derived Molecule, Inhibits the Replication of HSV-1 and HSV-2, and Inactivates HSV-1 DNA Polymerase: Shedding Light on Antiherpetic Properties. Molecules 2019, 24, 2912. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.-C.; Feng, H.; Lin, Y.-C.; Guo, X.-R. New Strategies against Drug Resistance to Herpes Simplex Virus. Int. J. Oral Sci. 2016, 8, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Ruchawapol, C.; Yuan, M.; Wang, S.-M.; Fu, W.-W.; Xu, H.-X. Natural Products and Their Derivatives against Human Herpesvirus Infection. Molecules 2021, 26, 6290. [Google Scholar] [CrossRef]
- Cairns, T.M.; Connolly, S.A. Entry of Alphaherpesviruses. Curr. Issues Mol. Biol. 2021, 41, 63–124. [Google Scholar] [CrossRef]
- Agelidis, A.M.; Shukla, D. Cell Entry Mechanisms of HSV: What We Have Learned in Recent Years. Future Virol. 2015, 10, 1145–1154. [Google Scholar] [CrossRef]
- Zhu, S.; Viejo-Borbolla, A. Pathogenesis and Virulence of Herpes Simplex Virus. Virulence 2021, 12, 2670–2702. [Google Scholar] [CrossRef]
- Azab, W.; Osterrieder, K. Initial Contact: The First Steps in Herpesvirus Entry. Adv. Anat. Embryol. Cell Biol. 2017, 223, 1–27. [Google Scholar] [CrossRef]
- Connolly, S.A.; Jardetzky, T.S.; Longnecker, R. The Structural Basis of Herpesvirus Entry. Nat. Rev. Microbiol. 2021, 19, 110–121. [Google Scholar] [CrossRef] [PubMed]
- Arii, J.; Kawaguchi, Y. The Role of HSV Glycoproteins in Mediating Cell Entry. Adv. Exp. Med. Biol. 2018, 1045, 3–21. [Google Scholar] [CrossRef] [PubMed]
- Heming, J.D.; Conway, J.F.; Homa, F.L. Herpesvirus Capsid Assembly and DNA Packaging. Adv. Anat. Embryol. Cell Biol. 2017, 223, 119–142. [Google Scholar] [CrossRef]
- Adlakha, M.; Livingston, C.M.; Bezsonova, I.; Weller, S.K. The Herpes Simplex Virus 1 Immediate Early Protein ICP22 Is a Functional Mimic of a Cellular J Protein. J. Virol. 2020, 94, e01564-19. [Google Scholar] [CrossRef] [PubMed]
- Adler, B.; Sattler, C.; Adler, H. Herpesviruses and Their Host Cells: A Successful Liaison. Trends Microbiol. 2017, 25, 229–241. [Google Scholar] [CrossRef]
- Krawczyk, E.; Kangas, C.; He, B. HSV Replication: Triggering and Repressing STING Functionality. Viruses 2023, 15, 226. [Google Scholar] [CrossRef]
- Rice, S.A. Release of HSV-1 Cell-Free Virions: Mechanisms, Regulation, and Likely Role in Human-Human Transmission. Viruses 2021, 13, 2395. [Google Scholar] [CrossRef]
- Cohen, J.I. Herpesvirus Latency. J. Clin. Investig. 2020, 130, 3361–3369. [Google Scholar] [CrossRef]
- Lomonte, P. Herpesvirus Latency: On the Importance of Positioning Oneself. Adv. Anat. Embryol. Cell Biol. 2017, 223, 95–117. [Google Scholar] [CrossRef]
- Ostler, J.B.; Sawant, L.; Harrison, K.; Jones, C. Regulation of Neurotropic Herpesvirus Productive Infection and Latency-Reactivation Cycle by Glucocorticoid Receptor and Stress-Induced Transcription Factors. Vitam. Horm. 2021, 117, 101–132. [Google Scholar] [CrossRef] [PubMed]
- Reese, T.A. Coinfections: Another Variable in the Herpesvirus Latency-Reactivation Dynamic. J. Virol. 2016, 90, 5534–5537. [Google Scholar] [CrossRef] [PubMed]
- Harrison, K.S.; Jones, C. Regulation of Herpes Simplex Virus Type 1 Latency-Reactivation Cycle and Ocular Disease by Cellular Signaling Pathways. Exp. Eye Res. 2022, 218, 109017. [Google Scholar] [CrossRef] [PubMed]
- Asha, K.; Sharma-Walia, N. Targeting Host Cellular Factors as a Strategy of Therapeutic Intervention for Herpesvirus Infections. Front. Cell. Infect. Microbiol. 2021, 11, 603309. [Google Scholar] [CrossRef] [PubMed]
- Kukhanova, M.K.; Korovina, A.N.; Kochetkov, S.N. Human Herpes Simplex Virus: Life Cycle and Development of Inhibitors. Biochemistry 2014, 79, 1635–1652. [Google Scholar] [CrossRef] [PubMed]
- Packard, J.E.; Dembowski, J.A. HSV-1 DNA Replication-Coordinated Regulation by Viral and Cellular Factors. Viruses 2021, 13, 2015. [Google Scholar] [CrossRef] [PubMed]
- Wen, L.; Jiang, Y.; Yang, J.; Zhao, Y.; Tian, M.; Yang, B. Structure, Bioactivity, and Synthesis of Methylated Flavonoids. Ann. N. Y. Acad. Sci. 2017, 1398, 120–129. [Google Scholar] [CrossRef]
- Chen, L.; Cao, H.; Huang, Q.; Xiao, J.; Teng, H. Absorption, Metabolism and Bioavailability of Flavonoids: A Review. Crit. Rev. Food Sci. Nutr. 2022, 62, 7730–7742. [Google Scholar] [CrossRef]
- Safe, S.; Jayaraman, A.; Chapkin, R.S.; Howard, M.; Mohankumar, K.; Shrestha, R. Flavonoids: Structure-Function and Mechanisms of Action and Opportunities for Drug Development. Toxicol. Res. 2021, 37, 147–162. [Google Scholar] [CrossRef]
- Liu, W.; Feng, Y.; Yu, S.; Fan, Z.; Li, X.; Li, J.; Yin, H. The Flavonoid Biosynthesis Network in Plants. Int. J. Mol. Sci. 2021, 22, 12824. [Google Scholar] [CrossRef]
- Petrussa, E.; Braidot, E.; Zancani, M.; Peresson, C.; Bertolini, A.; Patui, S.; Vianello, A. Plant Flavonoids--Biosynthesis, Transport and Involvement in Stress Responses. Int. J. Mol. Sci. 2013, 14, 14950–14973. [Google Scholar] [CrossRef]
- Šamec, D.; Karalija, E.; Šola, I.; Vujčić Bok, V.; Salopek-Sondi, B. The Role of Polyphenols in Abiotic Stress Response: The Influence of Molecular Structure. Plants 2021, 10, 118. [Google Scholar] [CrossRef]
- Teng, H.; Chen, L. Polyphenols and Bioavailability: An Update. Crit. Rev. Food Sci. Nutr. 2019, 59, 2040–2051. [Google Scholar] [CrossRef] [PubMed]
- Hassan, S.T.S.; Šudomová, M. Molecular Mechanisms of Flavonoids against Tumor Gamma-Herpesviruses and Their Correlated Cancers—A Focus on EBV and KSHV Life Cycles and Carcinogenesis. Int. J. Mol. Sci. 2022, 24, 247. [Google Scholar] [CrossRef] [PubMed]
- Singh, B.; Kumar, A.; Malik, A.K. Flavonoids Biosynthesis in Plants and Its Further Analysis by Capillary Electrophoresis. Electrophoresis 2017, 38, 820–832. [Google Scholar] [CrossRef] [PubMed]
- Wen, K.; Fang, X.; Yang, J.; Yao, Y.; Nandakumar, K.S.; Salem, M.L.; Cheng, K. Recent Research on Flavonoids and Their Biomedical Applications. Curr. Med. Chem. 2021, 28, 1042–1066. [Google Scholar] [CrossRef] [PubMed]
- Šudomová, M.; Berchová-Bímová, K.; Mazurakova, A.; Šamec, D.; Kubatka, P.; Hassan, S.T.S. Flavonoids Target Human Herpesviruses That Infect the Nervous System: Mechanisms of Action and Therapeutic Insights. Viruses 2022, 14, 592. [Google Scholar] [CrossRef]
- Russo, M.; Moccia, S.; Spagnuolo, C.; Tedesco, I.; Russo, G.L. Roles of Flavonoids against Coronavirus Infection. Chem. Biol. Interact. 2020, 328, 109211. [Google Scholar] [CrossRef]
- Sharma, V.; Sehrawat, N.; Sharma, A.; Yadav, M.; Verma, P.; Sharma, A.K. Multifaceted Antiviral Therapeutic Potential of Dietary Flavonoids: Emerging Trends and Future Perspectives. Biotechnol. Appl. Biochem. 2021, 69, 2028–2045. [Google Scholar] [CrossRef]
- Ninfali, P.; Antonelli, A.; Magnani, M.; Scarpa, E.S. Antiviral Properties of Flavonoids and Delivery Strategies. Nutrients 2020, 12, 2534. [Google Scholar] [CrossRef]
- Hassan, S.T.S.; Masarčíková, R.; Berchová, K. Bioactive Natural Products with Anti-Herpes Simplex Virus Properties. J. Pharm. Pharmacol. 2015, 67, 1325–1336. [Google Scholar] [CrossRef] [PubMed]
- Zakaryan, H.; Arabyan, E.; Oo, A.; Zandi, K. Flavonoids: Promising Natural Compounds against Viral Infections. Arch. Virol. 2017, 162, 2539–2551. [Google Scholar] [CrossRef] [PubMed]
- Ortega, J.T.; Serrano, M.L.; Suárez, A.I.; Baptista, J.; Pujol, F.H.; Cavallaro, L.V.; Campos, H.R.; Rangel, H.R. Antiviral Activity of Flavonoids Present in Aerial Parts of Marcetia Taxifolia against Hepatitis B Virus, Poliovirus, and Herpes Simplex Virus in Vitro. EXCLI J. 2019, 18, 1037–1048. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.I.; Kwon, E.-B.; Oh, Y.-C.; Go, Y.; Choi, J.-G. Mori Ramulus and Its Major Component Morusin Inhibit Herpes Simplex Virus Type 1 Replication and the Virus-Induced Reactive Oxygen Species. Am. J. Chin. Med. 2021, 49, 163–179. [Google Scholar] [CrossRef]
- Čulenová, M.; Sychrová, A.; Hassan, S.T.S.; Berchová-Bímová, K.; Svobodová, P.; Helclová, A.; Michnová, H.; Hošek, J.; Vasilev, H.; Suchý, P.; et al. Multiple In Vitro Biological Effects of Phenolic Compounds from Morus alba Root Bark. J. Ethnopharmacol. 2020, 248, 112296. [Google Scholar] [CrossRef]
- Chu, Y.; Lv, X.; Zhang, L.; Fu, X.; Song, S.; Su, A.; Chen, D.; Xu, L.; Wang, Y.; Wu, Z.; et al. Wogonin Inhibits in Vitro Herpes Simplex Virus Type 1 and 2 Infection by Modulating Cellular NF-κB and MAPK Pathways. BMC Microbiol. 2020, 20, 227. [Google Scholar] [CrossRef]
- Luo, Z.; Kuang, X.-P.; Zhou, Q.-Q.; Yan, C.-Y.; Li, W.; Gong, H.-B.; Kurihara, H.; Li, W.-X.; Li, Y.-F.; He, R.-R. Inhibitory Effects of Baicalein against Herpes Simplex Virus Type 1. Acta Pharm. Sin. B 2020, 10, 2323–2338. [Google Scholar] [CrossRef]
- Fahmy, N.M.; Al-Sayed, E.; Moghannem, S.; Azam, F.; El-Shazly, M.; Singab, A.N. Breaking Down the Barriers to a Natural Antiviral Agent: Antiviral Activity and Molecular Docking of Erythrina Speciosa Extract, Fractions, and the Major Compound. Chem. Biodivers. 2020, 17, e1900511. [Google Scholar] [CrossRef]
- Wang, Y.; Li, F.; Wang, Z.; Song, X.; Ren, Z.; Wang, X.; Wang, Y.; Zheng, K. Luteolin Inhibits Herpes Simplex Virus 1 Infection by Activating Cyclic Guanosine Monophosphate-Adenosine Monophosphate Synthase-Mediated Antiviral Innate Immunity. Phytomedicine 2023, 120, 155020. [Google Scholar] [CrossRef]
- Li, F.; Song, X.; Su, G.; Wang, Y.; Wang, Z.; Jia, J.; Qing, S.; Huang, L.; Wang, Y.; Zheng, K.; et al. Amentoflavone Inhibits HSV-1 and ACV-Resistant Strain Infection by Suppressing Viral Early Infection. Viruses 2019, 11, 466. [Google Scholar] [CrossRef]
- Li, W.; Xu, C.; Hao, C.; Zhang, Y.; Wang, Z.; Wang, S.; Wang, W. Inhibition of Herpes Simplex Virus by Myricetin through Targeting Viral gD Protein and Cellular EGFR/PI3K/Akt Pathway. Antivir. Res. 2020, 177, 104714. [Google Scholar] [CrossRef] [PubMed]
- Yarmolinsky, L.; Nakonechny, F.; Budovsky, A.; Zeigerman, H.; Khalfin, B.; Sharon, E.; Yarmolinsky, L.; Ben-Shabat, S.; Nisnevitch, M. Antimicrobial and Antiviral Compounds of Phlomis Viscosa Poiret. Biomedicines 2023, 11, 441. [Google Scholar] [CrossRef] [PubMed]
- Tarbeeva, D.V.; Krylova, N.V.; Iunikhina, O.V.; Likhatskaya, G.N.; Kalinovskiy, A.I.; Grigorchuk, V.P.; Shchelkanov, M.Y.; Fedoreyev, S.A. Biologically Active Polyphenolic Compounds from Lespedeza bicolor. Fitoterapia 2022, 157, 105121. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.; Wang, F.; Tang, B.; Han, J.; Li, X.; Lian, G.; Li, X.; Hao, S. Anti-Inflammatory Effects of Kaempferol-3-O-Rhamnoside on HSV-1 Encephalitis in Vivo and in Vitro. Neurosci. Lett. 2021, 765, 136172. [Google Scholar] [CrossRef] [PubMed]
- Sochocka, M.; Sobczyński, M.; Ochnik, M.; Zwolińska, K.; Leszek, J. Hampering Herpesviruses HHV-1 and HHV-2 Infection by Extract of Ginkgo Biloba (EGb) and Its Phytochemical Constituents. Front. Microbiol. 2019, 10, 2367. [Google Scholar] [CrossRef]
- Zhou, H.-Y.; Gao, S.-Q.; Gong, Y.-S.; Lin, T.; Tong, S.; Xiong, W.; Shi, C.-Y.; Wang, W.-Q.; Fang, J.-G. Anti-HSV-1 Effect of Dihydromyricetin from Ampelopsis grossedentata via the TLR9-Dependent Anti-Inflammatory Pathway. J. Glob. Antimicrob. Resist. 2020, 23, 370–376. [Google Scholar] [CrossRef]
- Zhou, N.; Zheng, D.; You, Q.; Chen, T.; Jiang, J.; Shen, W.; Zhang, D.; Liu, J.; Chen, D.; Hu, K. Therapeutic Potential of Biochanin A in Herpes Simplex Keratitis. Pharmaceuticals 2023, 16, 1240. [Google Scholar] [CrossRef]
- Pradhan, P.; Nguyen, M.L. Herpes Simplex Virus Virucidal Activity of MST-312 and Epigallocatechin Gallate. Virus Res. 2018, 249, 93–98. [Google Scholar] [CrossRef]
- Wu, C.-Y.; Yu, Z.-Y.; Chen, Y.-C.; Hung, S.-L. Effects of Epigallocatechin-3-Gallate and Acyclovir on Herpes Simplex Virus Type 1 Infection in Oral Epithelial Cells. J. Formos. Med. Assoc. 2021, 120, 2136–2143. [Google Scholar] [CrossRef]
- Wang, H.; Jia, X.; Zhang, M.; Cheng, C.; Liang, X.; Wang, X.; Xie, F.; Wang, J.; Yu, Y.; He, Y.; et al. Isoliquiritigenin Inhibits Virus Replication and Virus-Mediated Inflammation via NRF2 Signaling. Phytomedicine 2023, 114, 154786. [Google Scholar] [CrossRef]
- Vicente, J.; Benedetti, M.; Martelliti, P.; Vázquez, L.; Gentilini, M.V.; Peñaranda Figueredo, F.A.; Nabaes Jodar, M.S.; Viegas, M.; Barquero, A.A.; Bueno, C.A. The Flavonoid Cyanidin Shows Immunomodulatory and Broad-Spectrum Antiviral Properties, Including SARS-CoV-2. Viruses 2023, 15, 989. [Google Scholar] [CrossRef] [PubMed]
- Sivarajan, R.; Oberwinkler, H.; Roll, V.; König, E.-M.; Steinke, M.; Bodem, J. A Defined Anthocyanin Mixture Sourced from Bilberry and Black Currant Inhibits Measles Virus and Various Herpesviruses. BMC Complement. Med. Ther. 2022, 22, 181. [Google Scholar] [CrossRef] [PubMed]
- Guo, H.; Wan, X.; Niu, F.; Sun, J.; Shi, C.; Ye, J.M.; Zhou, C. Evaluation of Antiviral Effect and Toxicity of Total Flavonoids Extracted from Robinia pseudoacacia Cv. Idaho. Biomed. Pharmacother. 2019, 118, 109335. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.-Q.; Cai, L.; Zhang, N.; Zhang, J.; Wang, H.-H.; Zhu, W. Protective Effect of Total Flavonoids from Ixeris Sonchifolia on Herpes Simplex Virus Keratitis in Mice. BMC Complement. Med. Ther. 2020, 20, 113. [Google Scholar] [CrossRef] [PubMed]
- Rittà, M.; Marengo, A.; Civra, A.; Lembo, D.; Cagliero, C.; Kant, K.; Lal, U.R.; Rubiolo, P.; Ghosh, M.; Donalisio, M. Antiviral Activity of a Arisaema tortuosum Leaf Extract and Some of Its Constituents against Herpes Simplex Virus Type 2. Planta Med. 2020, 86, 267–275. [Google Scholar] [CrossRef]
- Stamos, J.D.; Lee, L.H.; Taylor, C.; Elias, T.; Adams, S.D. In Vitro and In Silico Analysis of the Inhibitory Activity of EGCG-Stearate against Herpes Simplex Virus-2. Microorganisms 2022, 10, 1462. [Google Scholar] [CrossRef]
- Obisesan, O.; Katata-Seru, L.; Mufamadi, S.; Mufhandu, H. Applications of Nanoparticles for Herpes Simplex Virus (HSV) and Human Immunodeficiency Virus (HIV) Treatment. J. Biomed. Nanotechnol. 2021, 17, 793–808. [Google Scholar] [CrossRef]
- Treml, J.; Gazdová, M.; Šmejkal, K.; Šudomová, M.; Kubatka, P.; Hassan, S.T.S. Natural Products-Derived Chemicals: Breaking Barriers to Novel Anti-HSV Drug Development. Viruses 2020, 12, 154. [Google Scholar] [CrossRef]
- Tomaszewska, E.; Ranoszek-Soliwoda, K.; Bednarczyk, K.; Lech, A.; Janicka, M.; Chodkowski, M.; Psarski, M.; Celichowski, G.; Krzyzowska, M.; Grobelny, J. Anti-HSV Activity of Metallic Nanoparticles Functionalized with Sulfonates vs. Polyphenols. Int. J. Mol. Sci. 2022, 23, 13104. [Google Scholar] [CrossRef]
- Paradowska, E.; Studzińska, M.; Jabłońska, A.; Lozovski, V.; Rusinchuk, N.; Mukha, I.; Vitiuk, N.; Leśnikowski, Z.J. Antiviral Effect of Nonfunctionalized Gold Nanoparticles against Herpes Simplex Virus Type-1 (HSV-1) and Possible Contribution of Near-Field Interaction Mechanism. Molecules 2021, 26, 5960. [Google Scholar] [CrossRef]
- Elste, J.; Kumari, S.; Sharma, N.; Razo, E.P.; Azhar, E.; Gao, F.; Nunez, M.C.; Anwar, W.; Mitchell, J.C.; Tiwari, V.; et al. Plant Cell-Engineered Gold Nanoparticles Conjugated to Quercetin Inhibit SARS-CoV-2 and HSV-1 Entry. Int. J. Mol. Sci. 2023, 24, 14792. [Google Scholar] [CrossRef] [PubMed]
- Krzyzowska, M.; Janicka, M.; Chodkowski, M.; Patrycy, M.; Obuch-Woszczatyńska, O.; Tomaszewska, E.; Ranoszek-Soliwoda, K.; Celichowski, G.; Grobelny, J. Epigallocatechin Gallate-Modified Silver Nanoparticles Show Antiviral Activity against Herpes Simplex Type 1 and 2. Viruses 2023, 15, 2024. [Google Scholar] [CrossRef]
- Caldas Dos Santos, T.; Rescignano, N.; Boff, L.; Reginatto, F.H.; Simões, C.M.O.; de Campos, A.M.; Mijangos, C. In Vitro Antiherpes Effect of C-Glycosyl Flavonoid Enriched Fraction of Cecropia glaziovii Encapsulated in PLGA Nanoparticles. Mater. Sci. Eng. C Mater. Biol. Appl. 2017, 75, 1214–1220. [Google Scholar] [CrossRef] [PubMed]
- Sicurella, M.; Sguizzato, M.; Mariani, P.; Pepe, A.; Baldisserotto, A.; Buzzi, R.; Huang, N.; Simelière, F.; Burholt, S.; Marconi, P.; et al. Natural Polyphenol-Containing Gels against HSV-1 Infection: A Comparative Study. Nanomaterials 2022, 12, 227. [Google Scholar] [CrossRef] [PubMed]
- Dickinson, D.; Marsh, B.; Shao, X.; Liu, E.; Sampath, L.; Yao, B.; Jiang, X.; Hsu, S. Virucidal Activities of Novel Hand Hygiene and Surface Disinfectant Formulations Containing EGCG-Palmitates (EC16). Am. J. Infect. Control 2022, 50, 1212–1219. [Google Scholar] [CrossRef] [PubMed]
- Brandariz-Nuñez, A.; Liu, T.; Du, T.; Evilevitch, A. Pressure-Driven Release of Viral Genome into a Host Nucleus Is a Mechanism Leading to Herpes Infection. Elife 2019, 8, e47212. [Google Scholar] [CrossRef] [PubMed]
- Bauer, D.W.; Huffman, J.B.; Homa, F.L.; Evilevitch, A. Herpes Virus Genome, the Pressure Is on. J. Am. Chem. Soc. 2013, 135, 11216–11221. [Google Scholar] [CrossRef] [PubMed]
- Brandariz-Nuñez, A.; Robinson, S.J.; Evilevitch, A. Pressurized DNA State inside Herpes Capsids—A Novel Antiviral Target. PLoS Pathog. 2020, 16, e1008604. [Google Scholar] [CrossRef]
- Šudomová, M.; Hassan, S.T.S. Nutraceutical Curcumin with Promising Protection against Herpesvirus Infections and Their Associated Inflammation: Mechanisms and Pathways. Microorganisms 2021, 9, 292. [Google Scholar] [CrossRef]
- Polansky, H.; Javaherian, A.; Itzkovitz, E. Clinical Trial of Herbal Treatment Gene-Eden-VIR/Novirin in Oral Herpes. J. Evid. Based Integr. Med. 2018, 23, 2515690X18806269. [Google Scholar] [CrossRef]
- Polansky, H.; Javaherian, A.; Itzkovitz, E. Clinical Study in Genital Herpes: Natural Gene-Eden-VIR/Novirin versus Acyclovir, Valacyclovir, and Famciclovir. Drug Des. Devel Ther. 2016, 10, 2713–2722. [Google Scholar] [CrossRef] [PubMed]
- Polansky, H.; Itzkovitz, E.; Javaherian, A. Clinical Study of Gene-Eden-VIR/Novirin in Genital Herpes: Suppressive Treatment Safely Decreases the Duration of Outbreaks in Both Severe and Mild Cases. Clin. Transl. Med. 2016, 5, 40. [Google Scholar] [CrossRef] [PubMed]
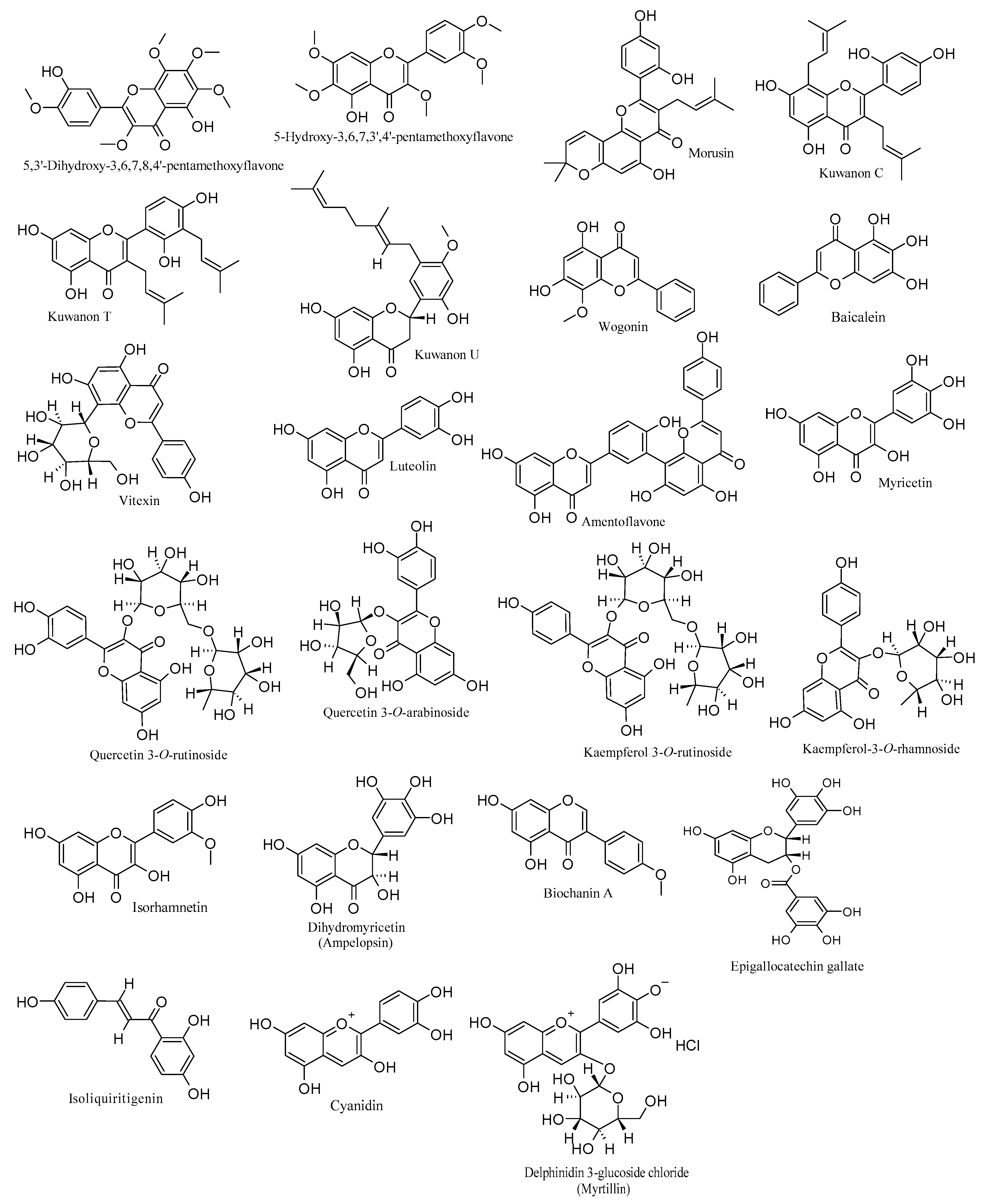

| Compound, Chemical Classification, Concentration/Dose, and Source | Study Type, Strain, and Cell/Animal Models | Mechanisms of Action (Inhibition) | Reference |
|---|---|---|---|
| PMF and PMF-OH. Flavones. EC50 = 6.8 and 5.9 µM, respectively. Marcetia taxifolia. | In vitro. HSV-1. Vero cells. | DNA replication. | [54] |
| Morusin. Prenylated flavone. 20 µM. Morus alba L. | In vitro. HSV-1. Vero cells. | DNA replication. gD expression. HSV-1-induced ROS. | [55] |
| Kuwanon C and kuwanon T (prenylated flavones) and kuwanon U (prenylated flavanone). IC50 = 0.91, 0.64, and 1.93 µg/mL, respectively. Morus alba L. | In vitro and in silico. HSV-1. Vero cells. | DNA replication (in vitro). DNA polymerase (in silico). | [56] |
| Wogonin. Flavone. Various concentrations in µM. Scutellaria baicalensis Georgi. | In vitro. HSV-1. Vero cells. | DNA replication. IE genes expressions and gD mRNA transcription. | [57] |
| Baicalein. Flavone. Different concentrations in µM (in vitro). 200 mg/kg/day (in vivo). Scutellaria baicalensis Georgi. | In vitro and in vivo. HSV-1 and HSV-1 ACV-resistant strains. Vero and HaCat cells. BALB/c mice. | DNA replication, viral particles, IKK-β, and NF-κB (in vitro). Viral loads, inflammation, and mortality (in vivo). | [58] |
| Vitexin. Flavone. EC50 = 18 µg/mL. Erythrina speciosa. | In vitro and in silico. HSV-1 (clinical strain). Vero cells. | DNA replication (in vitro). Viral DNA polymerase (in silico). | [59] |
| Luteolin. Flavone. Different concentrations in µM (in vitro). 50 mg/kg (in vivo). Various fruits, vegetables, and medicinal plants. | In vitro and in vivo. HSV-1 and HSV-1 ACV-resistant strains. HaCaT, BV2, and Vero cells. HSE mouse models. | Viral entry and DNA replication (in vitro). Viral post-entry by activating the cGAS-STING pathway and IFN (in vivo). | [60] |
| Amentoflavone. Biflavonoid. EC50 values (22.1 to 25.7 µM). Ginkgo biloba L., Biophytum sensitivum, and Garcinia species. | In vitro. HSV-1 (F strain) and ACV-resistant strains (HSV-1/106, HSV-1/153, and HSV-1/Blue). | DNA replication. UL54, UL52, and UL27 expressions. ICP0 expression. Nuclear import of HSV-1. | [61] |
| Myricetin. Flavonol. (2.2–40 µM; in vitro) and (2.5 and 5 mg/kg; in vivo). Various vegetables and fruits. | In vitro and in vivo. HSV-1. Vero, HeLa, and Hep-2 cells. BALB/c mice. | Virus adsorption, membrane fusion, DNA replication, and gD synthesis (in vitro). Cellular EGFR/PI3K/Akt pathway (in vitro). Virus titers and DNA replication (in vivo). | [62] |
| Quercetin 3-O-rutinoside and quercetin 3-O-arabinoside. Flavonols. 2 µM and IC50 = 8.6 µM. Phlomis viscosa Poiret and Lespedeza bicolor. | In vitro. HSV-1 (KOS- and ACV-resistant strains) Vero cells. | Plaque formation. DNA replication. Virucidal effect. | [63,64] |
| Kaempferol 3-O-rutinoside. Flavonol. IC50 = 12.2 µM. Lespedeza bicolor. | In vitro. HSV-1. Vero cells. | DNA replication. Virucidal effect. | [64] |
| Kaempferol-3-O-rhamnoside. Flavonol. Various concentrations in µM (in vitro). Brain, liver, blood, and muscle samples with diverse treatments in µM (in vivo). Various medicinal herbs. | In vitro and in vivo. HSV-1. Vero cells. HSE mouse models. | Viral infection (in vitro). Viral-induced brain injury in HSE animal models (in vivo). | [65] |
| Isorhamnetin. Flavonol. IC50 = 8.37 µg/mL. Ginkgo biloba. | In vitro. HSV-1. A549 cell. | Initial infection. DNA replication. | [66] |
| Dihydromyricetin (ampelopsin). Dihydroflavonol. EC50 = 12.56 µM. 16–32 µM (gene expressions) Ampelopsis grossedentata. | In vitro. HSV-1. Vero cells. | Plaque formation and progeny virus production. DNA replication. IE genes (ICP4 and ICP22), early genes (ICP8 and UL42), and late genes (gB and VP1/2). mRNA-TLR9. NF-κB and TNFα pathways. | [67] |
| Biochanin A. Isoflavone. 50 to 150 µM. Trifolium pratense L. | In vitro and in vivo. HSV-1. Vero and HCECs cells. Male C57BL/6 mice | DNA replication (in vitro). IE, E, and L genes (in vitro). Apoptosis of the corneal epithelium of HSK-infected mice (in vivo). | [68] |
| Epigallocatechin gallate Flavanol (catechin). 1–2 µM at 25–37 °C and 25 µg/mL. Camellia sinensis. | In vitro. HSV-1. Vero and oral epithelial cells. | DNA replication in the viral entry phase. Viral particles. IE and ICP0 expressions. | [69,70] |
| Isoliquiritigenin. Chalcone. 25 and 50 µM. Glycyrrhiza uralensis. | In vitro. HSV-1. A549 cells. | DNA replication via a mechanism that links with its NRF2 agonistic action. | [71] |
| Cyanidin. Anthocyanin. EC50 = 4.6 µg/mL. Various types of berries. | In vitro. HSV-1 ACV-resistant strain. HCLE cells. | Viral adsorption. DNA replication. | [72] |
| Delphinidin-3-glucoside chloride (myrtillin). Anthocyanin. 150 µg/mL. | In vitro. HSV-1. Vero cells. | DNA replication. | [73] |
| Total flavonoids. 0.46 g/mL (in vitro). 0.3 g/day/4 weeks (in vivo). Robinia pseudoacacia cv. Idaho. | In vitro and in vivo. HSV-1. Vero cells. Wistar rats. | DNA replication. | [74] |
| Total flavonoids. 50, 100, and 200 mg/kg (twice a day for 14 days). Ixeris Sonchifolia (Bae.) Hance. | In vivo. HSV-1. HSK-BALB/c mice. | Viral infection. IL-4 levels in the serum of mice. | [75] |
| Compound, Chemical Classification, Concentration/Dose, and Source | Study Type, Strain, and Cell/Animal Models | Mechanisms of Action (Inhibition) | Reference |
|---|---|---|---|
| Wogonin. Flavone. Various concentrations in µM. Scutellaria baicalensis Georgi. | In vitro. HSV-2. Vero and HEC-1-A cells. | DNA replication. Viral protein synthesis. HSV-2 virions. IE and gD expressions. mRNA transcription. Cellular NF-κB and JNK/p38 MAPK pathways. | [57] |
| Apigenin and luteolin. Flavones. EC50 = 0.05 and 0.41 µg/mL, respectively. Arisaema tortuosum. | In vitro. HSV-2 and ACV-resistant HSV-2. Vero cells. | DNA replication. Viral progeny production. Cell-to-cell virus spread. | [76] |
| Myricetin. Flavonol. 2.2–40 µM. Various vegetables and fruits. | In vitro and in silico. HSV-2. Vero, HeLa, and Hep-2 cells. | Virus adsorption, membrane fusion, and DNA replication (in vitro). Cellular EGFR/PI3K/Akt pathway. HSV-2 gD (in silico). | [62] |
| Isorhamnetin. Flavonol. IC50 = 7.08 µg/mL. Ginkgo biloba. | In vitro. HSV-2. A549 cell. | Primary infection. DNA replication. | [66] |
| Kuwanon E. Prenylated flavanone. EC50 = 1.61 µg/mL. Morus alba L. | In vitro and in silico. HSV-2. Vero cells. | DNA replication (in vitro). HSV-2 protease (in silico). | [56] |
| Epigallocatechin gallate. Flavanol (catechin). 75 µM (99.9% inhibition). Camellia sinensis. | In vitro. HSV-2. Vero cells. | Viral attachment. DNA replication. gD expression. | [77] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Šudomová, M.; Hassan, S.T.S. Flavonoids with Anti-Herpes Simplex Virus Properties: Deciphering Their Mechanisms in Disrupting the Viral Life Cycle. Viruses 2023, 15, 2340. https://doi.org/10.3390/v15122340
Šudomová M, Hassan STS. Flavonoids with Anti-Herpes Simplex Virus Properties: Deciphering Their Mechanisms in Disrupting the Viral Life Cycle. Viruses. 2023; 15(12):2340. https://doi.org/10.3390/v15122340
Chicago/Turabian StyleŠudomová, Miroslava, and Sherif T. S. Hassan. 2023. "Flavonoids with Anti-Herpes Simplex Virus Properties: Deciphering Their Mechanisms in Disrupting the Viral Life Cycle" Viruses 15, no. 12: 2340. https://doi.org/10.3390/v15122340
APA StyleŠudomová, M., & Hassan, S. T. S. (2023). Flavonoids with Anti-Herpes Simplex Virus Properties: Deciphering Their Mechanisms in Disrupting the Viral Life Cycle. Viruses, 15(12), 2340. https://doi.org/10.3390/v15122340






