Monkeypox, a Literature Review: What Is New and Where Does This concerning Virus Come From?
Abstract
:1. Introduction
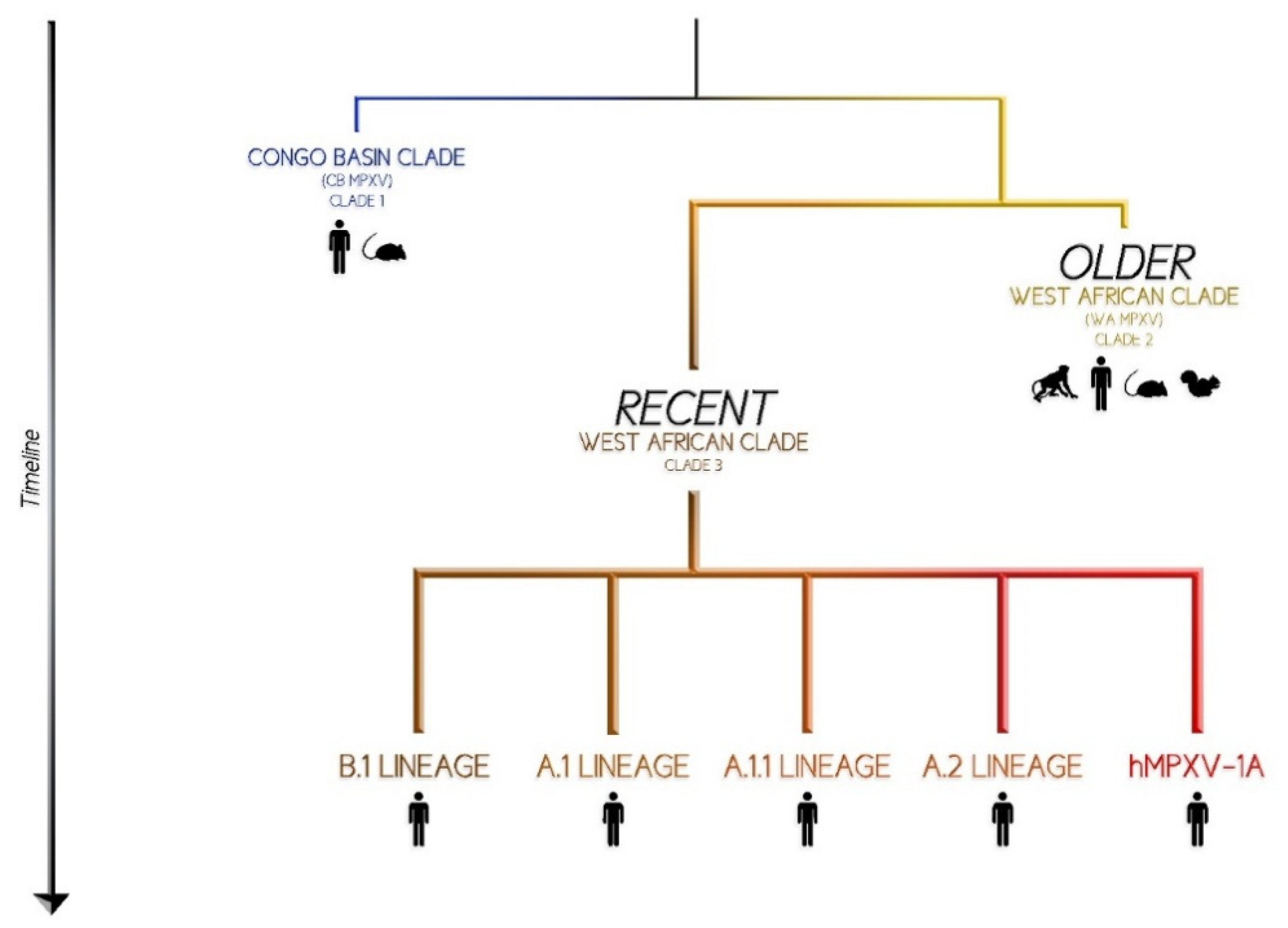
2. Virology and Phylogenesis
3. Epidemiology in Humans
4. Transmission Route and Pathogenesis
5. Clinical Manifestations
6. Diagnosis
7. Prevention
- Pre-exposure Prophylaxis
- Post-exposure Prophylaxis
8. Treatment
- Tecovirimat
- Brincidofovir and Cidofovir
- Vaccinia immune globulin intravenous (VIGIV)
9. Conclusions
10. Research Strategy and Selection Criteria
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Yang, Z.; Gray, M.; Winter, L. Why Do Poxviruses Still Matter? Cell Biosci. 2021, 11, 96. [Google Scholar] [CrossRef] [PubMed]
- Guarner, J.; del Rio, C.; Malani, P.N. Monkeypox in 2022—What Clinicians Need to Know. JAMA 2022, 328, 139–140. [Google Scholar] [CrossRef] [PubMed]
- Shchelkunov, S.N. Orthopoxvirus Genes That Mediate Disease Virulence and Host Tropism. Adv. Virol. 2012, 2012, 1–17. [Google Scholar] [CrossRef]
- Trindade, G.S.; Emerson, G.L.; Carroll, D.S.; Kroon, E.G.; Damon, I.K. Brazilian Vaccinia Viruses and Their Origins. Emerg. Infect. Dis. 2007, 13, 965–972. [Google Scholar] [CrossRef] [PubMed]
- Vorou, R.M.; Papavassiliou, V.G.; Pierroutsakos, I.N. Cowpox Virus Infection: An Emerging Health Threat. Curr. Opin. Infect. Dis. 2008, 21, 153–156. [Google Scholar] [CrossRef]
- Townsend, M.B.; Keckler, M.S.; Patel, N.; Davies, D.H.; Felgner, P.; Damon, I.K.; Karem, K.L. Humoral Immunity to Smallpox Vaccines and Monkeypox Virus Challenge: Proteomic Assessment and Clinical Correlations. J. Virol. 2013, 87, 900–911. [Google Scholar] [CrossRef]
- Chapman, J.L.; Nichols, D.K.; Martinez, M.J.; Raymond, J.W. Animal Models of Orthopoxvirus Infection. Vet. Pathol. 2010, 47, 852–870. [Google Scholar] [CrossRef] [PubMed]
- Babkin, I.V.; Babkina, I.N.; Tikunova, N.V. An Update of Orthopoxvirus Molecular Evolution. Viruses 2022, 14, 388. [Google Scholar] [CrossRef] [PubMed]
- Nuzzo, J.B.; Borio, L.L.; Gostin, L.O. The WHO Declaration of Monkeypox as a Global Public Health Emergency. JAMA 2022. [Google Scholar] [CrossRef]
- Jacobs, B.L.; Langland, J.O.; Kibler, K.V.; Denzler, K.L.; White, S.D.; Holechek, S.A.; Wong, S.; Huynh, T.; Baskin, C.R. Vaccinia Virus Vaccines: Past, Present and Future. Antivir. Res. 2009, 84, 1–13. [Google Scholar] [CrossRef] [Green Version]
- Parker, S.; Buller, R.M. A Review of Experimental and Natural Infections of Animals with Monkeypox Virus between 1958 and 2012. Future Virol. 2013, 8, 129–157. [Google Scholar] [CrossRef] [PubMed]
- Von Magnus, P.; Andersen, E.K.; Petersen, K.B.; Birch-Andersen, A. A Pox-like Disease in Cynomolgus Monkeys. APMIS 2009, 46, 156–176. [Google Scholar] [CrossRef]
- Shchelkunov, S.N.; Totmenin, A.V.; Safronov, P.F.; Mikheev, M.V.; Gutorov, V.V.; Ryazankina, O.I.; Petrov, N.A.; Babkin, I.V.; Uvarova, E.A.; Sandakhchiev, L.S.; et al. Analysis of the Monkeypox Virus Genome. Virology 2002, 297, 172–194. [Google Scholar] [CrossRef]
- Firth, C.; Kitchen, A.; Shapiro, B.; Suchard, M.A.; Holmes, E.C.; Rambaut, A. Using Time-Structured Data to Estimate Evolutionary Rates of Double-Stranded DNA Viruses. Mol. Biol. Evol. 2010, 27, 2038–2051. [Google Scholar] [CrossRef]
- Nextstrain Bioinformatics Genomic Epidemiology of Monkeypox Virus. Available online: https://nextstrain.org/monkeypox/hmpxv1 (accessed on 7 August 2022).
- Wang, L.; Shang, J.; Weng, S.; Aliyari, S.R.; Ji, C.; Cheng, G.; Wu, A. Genomic Annotation and Molecular Evolution of Monkeypox Virus Outbreak in 2022. J. Med. Virol. 2022. [Google Scholar] [CrossRef] [PubMed]
- Damon, I.K. Status of Human Monkeypox: Clinical Disease, Epidemiology and Research. Vaccine 2011, 29, D54–D59. [Google Scholar] [CrossRef] [PubMed]
- Forni, D.; Molteni, C.; Cagliani, R.; Sironi, M. Geographic Structuring and Divergence Time Frame of Monkeypox Virus in the Endemic Region. J. Infect. Dis. 2022. [Google Scholar] [CrossRef]
- Kaler, J.; Hussain, A.; Flores, G.; Kheiri, S.; Desrosiers, D. Monkeypox: A Comprehensive Review of Transmission, Pathogenesis, and Manifestation. Cureus 2022, 14, e26531. [Google Scholar] [CrossRef]
- Mccollum, A.M.; Damon, I.K. Human Monkeypox. Clin. Infect. Dis. 2014, 58, 260–267. [Google Scholar] [CrossRef]
- Hammarlund, E.; Dasgupta, A.; Pinilla, C.; Norori, P.; Früh, K.; Slifka, M.K. Monkeypox Virus Evades Antiviral CD4 + and CD8 + T Cell Responses by Suppressing Cognate T Cell Activation. Proc. Natl. Acad. Sci. USA 2008, 105, 14567–14572. [Google Scholar] [CrossRef] [Green Version]
- Happi, C.; Adetifa, I.; Mbala, P.; Njouom, R.; Nakoune, E.; Happi, A.; Ndodo, N.; Ayansola, O.; Mboowa, G.; Bedford, T.; et al. Urgent Need for a Non-Discriminatory and Non-Stigmatizing Nomenclature for Monkeypox Virus. Available online: https://virological.org/t/urgent-need-for-a-non-discriminatory-and-non-stigmatizing-nomenclature-for-monkeypox-virus/853 (accessed on 7 August 2022).
- Isidro, J.; Borges, V.; Pinto, M.; Sobral, D.; Santos, J.D.; Nunes, A.; Mixão, V.; Ferreira, R.; Santos, D.; Duarte, S.; et al. Phylogenomic Characterization and Signs of Microevolution in the 2022 Multi-Country Outbreak of Monkeypox Virus. Nat. Med. 2022. [Google Scholar] [CrossRef]
- O’Toole, A.; Rambaut, A. Initial Observations about Putative APOBEC3 Deaminase Editing Driving Short-Term Evolution of MPXV Since 2017. Available online: https://virological.org/t/initial-observations-about-putative-apobec3-deaminase-editing-driving-short-term-evolution-of-mpxv-since-2017/830 (accessed on 7 August 2022).
- Tiecco, G.; Storti, S.; Arsuffi, S.; Degli Antoni, M.; Focà, E.; Castelli, F.; Quiros-Roldan, E. Omicron BA.2 Lineage, the “Stealth” Variant: Is It Truly a Silent Epidemic? A Literature Review. Int. J. Mol. 2022, 23, 7315. [Google Scholar] [CrossRef]
- Luna, N.; Ramírez, A.L.; Muñoz, M.; Ballesteros, N.; Patiño, L.H.; Castañeda, S.A.; Bonilla-Aldana, D.K.; Paniz-Mondolfi, A.; Ramírez, J.D. Phylogenomic Analysis of the Monkeypox Virus (MPXV) 2022 Outbreak: Emergence of a Novel Viral Lineage? Travel Med. Infect. Dis. 2022, 49, 102402. [Google Scholar] [CrossRef] [PubMed]
- Haider, N.; Guitian, J.; Simons, D.; Asogun, D.; Ansumana, R.; Honeyborne, I.; Velavan, T.P.; Ntoumi, F.; Valdoleiros, S.R.; Petersen, E.; et al. Increased Outbreaks of Monkeypox Highlight Gaps in Actual Disease Burden in Sub-Saharan Africa and in Animal Reservoirs. Int. J. Infect. Dis. 2022, 122, 107–111. [Google Scholar] [CrossRef] [PubMed]
- Ogoina, D.; Izibewule, J.H.; Ogunleye, A.; Ederiane, E.; Anebonam, U.; Neni, A.; Oyeyemi, A.; Etebu, E.N.; Ihekweazu, C. The 2017 Human Monkeypox Outbreak in Nigeria—Report of Outbreak Experience and Response in the Niger Delta University Teaching Hospital, Bayelsa State, Nigeria. PLoS ONE 2019, 14, e0214229. [Google Scholar] [CrossRef]
- CDC Update: Multistate Outbreak of Monkeypox—Illinois, Indiana, Kansas, Missouri, Ohio, and Wisconsin. 2003. Available online: https://www.cdc.gov/mmwr/preview/mmwrhtml/mm5227a5.htm#top (accessed on 7 August 2022).
- CDC 2022 Monkeypox Outbreak Global Map. Available online: https://www.cdc.gov/poxvirus/monkeypox/response/2022/world-map.html (accessed on 7 August 2022).
- ECDC Monkeypox Multi-Country Outbreak. Available online: https://www.ecdc.europa.eu/en/monkeypox-outbreak (accessed on 7 August 2022).
- Fine, P.E.M.; Jezek, Z.; Grab, B.; Dixon, H. The Transmission Potential of Monkeypox Virus in Human Populations. Int. J. Epidemiol. 1988, 17, 643–650. [Google Scholar] [CrossRef] [PubMed]
- WHO Monkeypox. Available online: https://www.who.int/news-room/fact-sheets/detail/monkeypox (accessed on 7 August 2022).
- Reynolds, M.G.; Yorita, K.L.; Kuehnert, M.J.; Davidson, W.B.; Huhn, G.D.; Holman, R.C.; Damon, I.K. Clinical Manifestations of Human Monkeypox Influenced by Route of Infection. J. Infect. Dis. 2006, 194, 773–780. [Google Scholar] [CrossRef]
- CDC Monkeypox: How It Spreads. Available online: https://www.cdc.gov/poxvirus/monkeypox/transmission.html (accessed on 7 August 2022).
- CDC Newsroom CDC and Health Partners Responding to Monkeypox Case in the U.S. Available online: https://www.cdc.gov/media/releases/2022/s0518-monkeypox-case.html (accessed on 7 August 2022).
- Antinori, A.; Mazzotta, V.; Vita, S.; Carletti, F.; Tacconi, D.; Lapini, L.E.; D’Abramo, A.; Cicalini, S.; Lapa, D.; Pittalis, S.; et al. Epidemiological, Clinical and Virological Characteristics of Four Cases of Monkeypox Support Transmission through Sexual Contact, Italy, May 2022. Eurosurveillance 2022, 27, 2200421. [Google Scholar] [CrossRef]
- Nguyen, S.; Dang, A.; Vu, G.; Nguyen, C.; Le, T.; Truong, N.; Hoang, C.; Tran, T.; Tran, T.; Pham, H.; et al. Lack of Knowledge about Sexually Transmitted Diseases (STDs): Implications for STDs Prevention and Care among Dermatology Patients in an Urban City in Vietnam. Int. J. Environ. Res. Public Health 2019, 16, 1080. [Google Scholar] [CrossRef]
- Mbala, P.K.; Huggins, J.W.; Riu-Rovira, T.; Ahuka, S.M.; Mulembakani, P.; Rimoin, A.W.; Martin, J.W.; Muyembe, J.-J.T. Maternal and Fetal Outcomes Among Pregnant Women With Human Monkeypox Infection in the Democratic Republic of Congo. J. Infect. Dis. 2017, 216, 824–828. [Google Scholar] [CrossRef]
- Sookaromdee, P.; Wiwanitkit, V. Neonate, Infected Mother and Monkeypox: The Present Concern. J. Perinat. Med. 2022. [Google Scholar] [CrossRef]
- Atkinson, B.; Burton, C.; Pottage, T.; Thompson, K.; Ngabo, D.; Crook, A.; Pitman, J.; Summers, S.; Lewandowski, K.; Furneaux, J.; et al. Infection-competent Monkeypox Virus Contamination Identified in Domestic Settings Following an Imported Case of Monkeypox into the UK. Environ. Microbiol. 2022. [Google Scholar] [CrossRef]
- CDC Potential Exposure to Person with Confirmed Human Monkeypox Infection—United States. 2021. Available online: https://emergency.cdc.gov/han/2021/han00446.asp (accessed on 7 August 2022).
- Hutin, Y.J.F.; Williams, R.J.; Malfait, P.; Pebody, R.; Loparev, V.N.; Ropp, S.L.; Rodriguez, M.; Knight, J.C.; Tshioko, F.K.; Khan, A.S.; et al. Outbreak of Human Monkeypox, Democratic Republic of Congo, 1996 to 1997. Emerg. Infect. Dis. 2001, 7, 434–438. [Google Scholar] [CrossRef]
- Jezek, Z.; Marennikova, S.S.; Mutumbo, M.; Nakano, J.H.; Paluku, K.M.; Szczeniowski, M. Human Monkeypox: A Study of 2,510 Contacts of 214 Patients. J. Infect. Dis. 1986, 154, 551–555. [Google Scholar] [CrossRef]
- Adler, H.; Gould, S.; Hine, P.; Snell, L.B.; Wong, W.; Houlihan, C.F.; Osborne, J.C.; Rampling, T.; Beadsworth, M.B.; Duncan, C.J.; et al. Clinical Features and Management of Human Monkeypox: A Retrospective Observational Study in the UK. Lancet Infect. Dis. 2022, 22, 1153–1162. [Google Scholar] [CrossRef]
- Girometti, N.; Byrne, R.; Bracchi, M.; Heskin, J.; McOwan, A.; Tittle, V.; Gedela, K.; Scott, C.; Patel, S.; Gohil, J.; et al. Demographic and Clinical Characteristics of Confirmed Human Monkeypox Virus Cases in Individuals Attending a Sexual Health Centre in London, UK: An Observational Analysis. Lancet Infect. Dis. 2022. [Google Scholar] [CrossRef]
- ECDC Factsheet for Health Professionals on Monkeypox. Available online: https://www.ecdc.europa.eu/en/all-topics-z/monkeypox/factsheet-health-professionals (accessed on 7 August 2022).
- CDC Treatment Information for Healthcare Professionals. Available online: https://www.cdc.gov/poxvirus/monkeypox/clinicians/treatment.html (accessed on 7 August 2022).
- Yinka-Ogunleye, A.; Aruna, O.; Ogoina, D.; Aworabhi, N.; Eteng, W.; Badaru, S.; Mohammed, A.; Agenyi, J.; Etebu, E.N.; Numbere, T.-W.; et al. Reemergence of Human Monkeypox in Nigeria, 2017. Emerg. Infect. Dis. 2018, 24, 1149–1151. [Google Scholar] [CrossRef]
- Jezek, Z.; Szczeniowski, M.; Paluku, K.M.; Mutombo, M. Human Monkeypox: Clinical Features of 282 Patients. J. Infect. Dis. 1987, 156, 293–298. [Google Scholar] [CrossRef]
- CDC Monkeypox: Signs and Symptoms. Available online: https://www.cdc.gov/poxvirus/monkeypox/symptoms.html (accessed on 7 August 2022).
- Brown, K.; Leggat, P. Human Monkeypox: Current State of Knowledge and Implications for the Future. Trop. Med. Infect. Dis. 2016, 1, 8. [Google Scholar] [CrossRef]
- Gong, Q.; Wang, C.; Chuai, X.; Chiu, S. Monkeypox Virus: A Re-Emergent Threat to Humans. Virol. Sin. 2022. [Google Scholar] [CrossRef]
- Huhn, G.D.; Bauer, A.M.; Yorita, K.; Graham, M.B.; Sejvar, J.; Likos, A.; Damon, I.K.; Reynolds, M.G.; Kuehnert, M.J. Clinical Characteristics of Human Monkeypox, and Risk Factors for Severe Disease. Clin. Infect. Dis. 2005, 41, 1742–1751. [Google Scholar] [CrossRef] [PubMed]
- Al-Tammemi, A.B.; Albakri, R.; Alabsi, S. The Outbreak of Human Monkeypox in 2022: A Changing Epidemiology or an Impending Aftereffect of Smallpox Eradication? Front. Trop. Dis. 2022, 3, 951380. [Google Scholar] [CrossRef]
- WHO Weekly Bulletin on Outbreaks and Other Emergencies, Week 22: 23–29 May 2022. 2022. Available online: https://apps.who.int/iris/bitstream/handle/10665/354782/OEW22-2329052022.pdf (accessed on 7 August 2022).
- Badenoch, J.B.; Conti, I.; Rengasamy, E.R.; Watson, C.J.; Butler, M.; Hussain, Z.; Rooney, A.G.; Zandi, M.S.; Lewis, G.; David, A.S.; et al. Neurological and Psychiatric Presentations Associated with Human Monkeypox Virus Infection: A Systematic Review and Meta-Analysis. Medrxiv 2022. [Google Scholar] [CrossRef]
- Pastula, D.M.; Tyler, K.L. An Overview of Monkeypox Virus and Its Neuroinvasive Potential. Ann. Neurol. 2022. [Google Scholar] [CrossRef]
- Ogoina, D.; Mohammed, A.; Yinka-Ogunleye, A.; Ihekweazu, C. A Case of Suicide during the 2017 Monkeypox Outbreak in Nigeria. IJID Reg. 2022, 3, 226–227. [Google Scholar] [CrossRef] [PubMed]
- Otu, A.; Ebenso, B.; Walley, J.; Barceló, J.M.; Ochu, C.L. Global Human Monkeypox Outbreak: Atypical Presentation Demanding Urgent Public Health Action. Lancet Microbe 2022, 3, e554–e555. [Google Scholar] [CrossRef] [PubMed]
- Davido, B.; D’anglejan, E.; Jourdan, J.; Robinault, A.; Davido, G. Monkeypox 2022 Outbreak: Cases with Exclusive Genital Lesions. J. Travel Med. 2022. [Google Scholar] [CrossRef]
- Thornhill, J.P.; Barkati, S.; Walmsley, S.; Rockstroh, J.; Antinori, A.; Harrison, L.B.; Palich, R.; Nori, A.; Reeves, I.; Habibi, M.S.; et al. Monkeypox Virus Infection in Humans across 16 Countries—April–June 2022. N. Engl. J. Med. 2022. [Google Scholar] [CrossRef]
- Wise, J. Monkeypox: New Clinical Symptoms Are Identified in Confirmed Cases. BMJ 2022, o1845. [Google Scholar] [CrossRef]
- Ortiz-Martínez, Y.; Zambrano-Sanchez, G.; Rodríguez-Morales, A.J. Monkeypox and HIV/AIDS: When the Outbreak Faces the Epidemic. Int. J. STD AIDS 2022, 095646242211141. [Google Scholar] [CrossRef]
- de Sousa, D.; Patrocínio, J.; Frade, J.; Correia, C.; Borges-Costa, J.; Filipe, P. Human Monkeypox Coinfection with Acute HIV: An Exuberant Presentation. Int. J. STD AIDS 2022, 095646242211149. [Google Scholar] [CrossRef]
- Orba, Y.; Sasaki, M.; Yamaguchi, H.; Ishii, A.; Thomas, Y.; Ogawa, H.; Hang’ombe, B.M.; Mweene, A.S.; Morikawa, S.; Saijo, M.; et al. Orthopoxvirus Infection among Wildlife in Zambia. J. Gen. Virol. 2015, 96, 390–394. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Olson, V.A.; Laue, T.; Laker, M.T.; Damon, I.K. Detection of Monkeypox Virus with Real-Time PCR Assays. J. Clin. Virol. 2006, 36, 194–203. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Zhao, H.; Wilkins, K.; Hughes, C.; Damon, I.K. Real-Time PCR Assays for the Specific Detection of Monkeypox Virus West African and Congo Basin Strain DNA. J. Virol. Methods 2010, 169, 223–227. [Google Scholar] [CrossRef]
- Kulesh, D.A.; Loveless, B.M.; Norwood, D.; Garrison, J.; Whitehouse, C.A.; Hartmann, C.; Mucker, E.; Miller, D.; Wasieloski, L.P.; Huggins, J.; et al. Monkeypox Virus Detection in Rodents Using Real-Time 3′-Minor Groove Binder TaqMan® Assays on the Roche LightCycler. Lab. Investig. 2004, 84, 1200–1208. [Google Scholar] [CrossRef]
- Doellinger, J.; Schaade, L.; Nitsche, A. Comparison of the Cowpox Virus and Vaccinia Virus Mature Virion Proteome: Analysis of the Species- and Strain-Specific Proteome. PLoS ONE 2015, 10, e0141527. [Google Scholar] [CrossRef]
- Alakunle, E.; Moens, U.; Nchinda, G.; Okeke, M.I. Monkeypox Virus in Nigeria: Infection Biology, Epidemiology, and Evolution. Viruses 2020, 12, 1257. [Google Scholar] [CrossRef]
- CDC Monkeypox and Smallpox Vaccine Guidance. Available online: https://www.cdc.gov/poxvirus/monkeypox/clinicians/smallpox-vaccine.html (accessed on 7 August 2022).
- Rizk, J.G.; Lippi, G.; Henry, B.M.; Forthal, D.N.; Rizk, Y. Prevention and Treatment of Monkeypox. Drugs 2022, 82, 957–963. [Google Scholar] [CrossRef]
- Rao, A.K.; Petersen, B.W.; Whitehill, F.; Razeq, J.H.; Isaacs, S.N.; Merchlinsky, M.J.; Campos-Outcalt, D.; Morgan, R.L.; Damon, I.; Sánchez, P.J.; et al. Use of JYNNEOS (Smallpox and Monkeypox Vaccine, Live, Nonreplicating) for Preexposure Vaccination of Persons at Risk for Occupational Exposure to Orthopoxviruses: Recommendations of the Advisory Committee on Immunization Practices—United States, 2022. MMWR. Morb. Mortal. Wkly. Rep. 2022, 71, 734–742. [Google Scholar] [CrossRef]
- Overton, E.T.; Lawrence, S.J.; Stapleton, J.T.; Weidenthaler, H.; Schmidt, D.; Koenen, B.; Silbernagl, G.; Nopora, K.; Chaplin, P. A Randomized Phase II Trial to Compare Safety and Immunogenicity of the MVA-BN Smallpox Vaccine at Various Doses in Adults with a History of AIDS. Vaccine 2020, 38, 2600–2607. [Google Scholar] [CrossRef]
- Khalil, A.; Samara, A.; O’Brien, P.; Morris, E.; Draycott, T.; Lees, C.; Ladhani, S. Monkeypox Vaccines in Pregnancy: Lessons Must Be Learned from COVID-19. Lancet Glob. Health 2022. [Google Scholar] [CrossRef]
- Cono, J.; Cragan, J.; Jamieson, D.; Rasmussen, S. Prophylaxis and Treatment of Pregnant Women for Emerging Infections and Bioterrorism Emergencies. Emerg. Infect. Dis. 2006, 12, 1631–1637. [Google Scholar] [CrossRef]
- Petersen, B.W.; Harms, T.J.; Reynolds, M.G.; Harrison, L.H. Use of Vaccinia Virus Smallpox Vaccine in Laboratory and Health Care Personnel at Risk for Occupational Exposure to Orthopoxviruses — Recommendations of the Advisory Committee on Immunization Practices (ACIP), 2015. MMWR. Morb. Mortal. Wkly. Rep. 2016, 65, 257–262. [Google Scholar] [CrossRef]
- Minhaj, F.S.; Ogale, Y.P.; Whitehill, F.; Schultz, J.; Foote, M.; Davidson, W.; Hughes, C.M.; Wilkins, K.; Bachmann, L.; Chatelain, R.; et al. Monkeypox Outbreak—Nine States, May 2022. MMWR. Morb. Mortal. Wkly. Rep. 2022, 71, 764–769. [Google Scholar] [CrossRef]
- Kozlov, M. Monkeypox Vaccination Begins—Can the Global Outbreaks Be Contained? Nature 2022, 606, 444–445. [Google Scholar] [CrossRef]
- Reynolds, M.; McCollum, A.; Nguete, B.; Shongo Lushima, R.; Petersen, B. Improving the Care and Treatment of Monkeypox Patients in Low-Resource Settings: Applying Evidence from Contemporary Biomedical and Smallpox Biodefense Research. Viruses 2017, 9, 380. [Google Scholar] [CrossRef] [PubMed]
- Siegrist, E.A.; Sassine, J. Antivirals with Activity Against Monkeypox: A Clinically Oriented Review. Clin. Infect. Dis. 2022. [Google Scholar] [CrossRef] [PubMed]
- SIGA Technologies, Inc. 2018 TPOXX (Tecovirimat) Label Information. 2018. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/label/2018/208627s000lbl.pdf (accessed on 7 August 2022).
- Russo, A.T.; Grosenbach, D.W.; Chinsangaram, J.; Honeychurch, K.M.; Long, P.G.; Lovejoy, C.; Maiti, B.; Meara, I.; Hruby, D.E. An Overview of Tecovirimat for Smallpox Treatment and Expanded Anti-Orthopoxvirus Applications. Expert Rev. Anti-Infect. Ther. 2021, 19, 331–344. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Liu, Y.; Li, K.; Zhang, L. Targeting F13 from Monkeypox Virus and Variola Virus by Tecovirimat: Molecular Simulation Analysis. J. Infect. 2022. [Google Scholar] [CrossRef] [PubMed]
- Grosenbach, D.W.; Honeychurch, K.; Rose, E.A.; Chinsangaram, J.; Frimm, A.; Maiti, B.; Lovejoy, C.; Meara, I.; Long, P.; Hruby, D.E. Oral Tecovirimat for the Treatment of Smallpox. N. Engl. J. Med. 2018, 379, 44–53. [Google Scholar] [CrossRef]
- Rao, A.K.; Schulte, J.; Chen, T.-H.; Hughes, C.M.; Davidson, W.; Neff, J.M.; Markarian, M.; Delea, K.C.; Wada, S.; Liddell, A.; et al. Monkeypox in a Traveler Returning from Nigeria—Dallas, Texas, July 2021. MMWR. Morb. Mortal. Wkly. Rep. 2022, 71, 509–516. [Google Scholar] [CrossRef] [PubMed]
- Hutson, C.L.; Kondas, A.V.; Mauldin, M.R.; Doty, J.B.; Grossi, I.M.; Morgan, C.N.; Ostergaard, S.D.; Hughes, C.M.; Nakazawa, Y.; Kling, C.; et al. Pharmacokinetics and Efficacy of a Potential Smallpox Therapeutic, Brincidofovir, in a Lethal Monkeypox Virus Animal Model. Msphere 2021, 6, e00927-20. [Google Scholar] [CrossRef] [PubMed]
- ClinicalTrials.gov Ongoing Clinical Trials on Monkeypox. Available online: https://clinicaltrials.gov/ct2/results?cond=Monkeypox&term=&cntry=&state=&city=&dist= (accessed on 7 August 2022).
| n. Trial and Title | Type | Description |
|---|---|---|
| NCT05443867 Monkeypox Asymptomatic Shedding: Evaluation by Self-Sampling MPX-ASSESS | observational cohort prospective | Close follow-up study of close contacts of MPX confirmed cases. Evaluation of secondary attack rate of MPXV infection in contacts, defined by PCR positivity on any sample; clinical and serological evaluation. |
| NCT05058898 A One Health Study of Monkeypox Human Infection | observational case-control prospective | Proportion of monkeypox cases occurring following interhuman exposures through a quantitative case-control study with an odds ratio of >3 for an exposure factor for human-to-human transmission. |
| NCT05438953 Follow-up of Contact at Risk of Monkeypox Infection: a Prospective Cohort Study | Prospective Interventional | Estimation the failure rate of a post-exposure vaccination by the VMA vaccine in MPX contact case participants at risk (within 14 days after the last contact) after one dose. |
| NCT02977715 IMVAMUNE® Smallpox Vaccine in Adult Healthcare Personnel at Risk for Monkeypox in the Democratic Republic of the Congo | prospective interventional | Description of monkeypox exposure and infection in eligible healthcare workers at risk of monkeypox infection, evaluating the immunogenicity and safety MVA vaccine. Evaluation of proportion of participants who after being vaccinated develop suspected or confirmed monkeypox infection, and experience exposure to monkeypox virus. |
| NCT02080767 Tecovirimat (ST-246) Treatment for Orthopox Virus Exposure | prospective interventional | Evaluation of efficacy and safety of tecovirimat in personnel (including US civilian employees, contractors and other US personnel and dependents, as well as allied military forces and local nationals) of any age exposed to or infected with orthopoxviruses or developed serious complications from vaccinia vaccination. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tiecco, G.; Degli Antoni, M.; Storti, S.; Tomasoni, L.R.; Castelli, F.; Quiros-Roldan, E. Monkeypox, a Literature Review: What Is New and Where Does This concerning Virus Come From? Viruses 2022, 14, 1894. https://doi.org/10.3390/v14091894
Tiecco G, Degli Antoni M, Storti S, Tomasoni LR, Castelli F, Quiros-Roldan E. Monkeypox, a Literature Review: What Is New and Where Does This concerning Virus Come From? Viruses. 2022; 14(9):1894. https://doi.org/10.3390/v14091894
Chicago/Turabian StyleTiecco, Giorgio, Melania Degli Antoni, Samuele Storti, Lina Rachele Tomasoni, Francesco Castelli, and Eugenia Quiros-Roldan. 2022. "Monkeypox, a Literature Review: What Is New and Where Does This concerning Virus Come From?" Viruses 14, no. 9: 1894. https://doi.org/10.3390/v14091894
APA StyleTiecco, G., Degli Antoni, M., Storti, S., Tomasoni, L. R., Castelli, F., & Quiros-Roldan, E. (2022). Monkeypox, a Literature Review: What Is New and Where Does This concerning Virus Come From? Viruses, 14(9), 1894. https://doi.org/10.3390/v14091894







