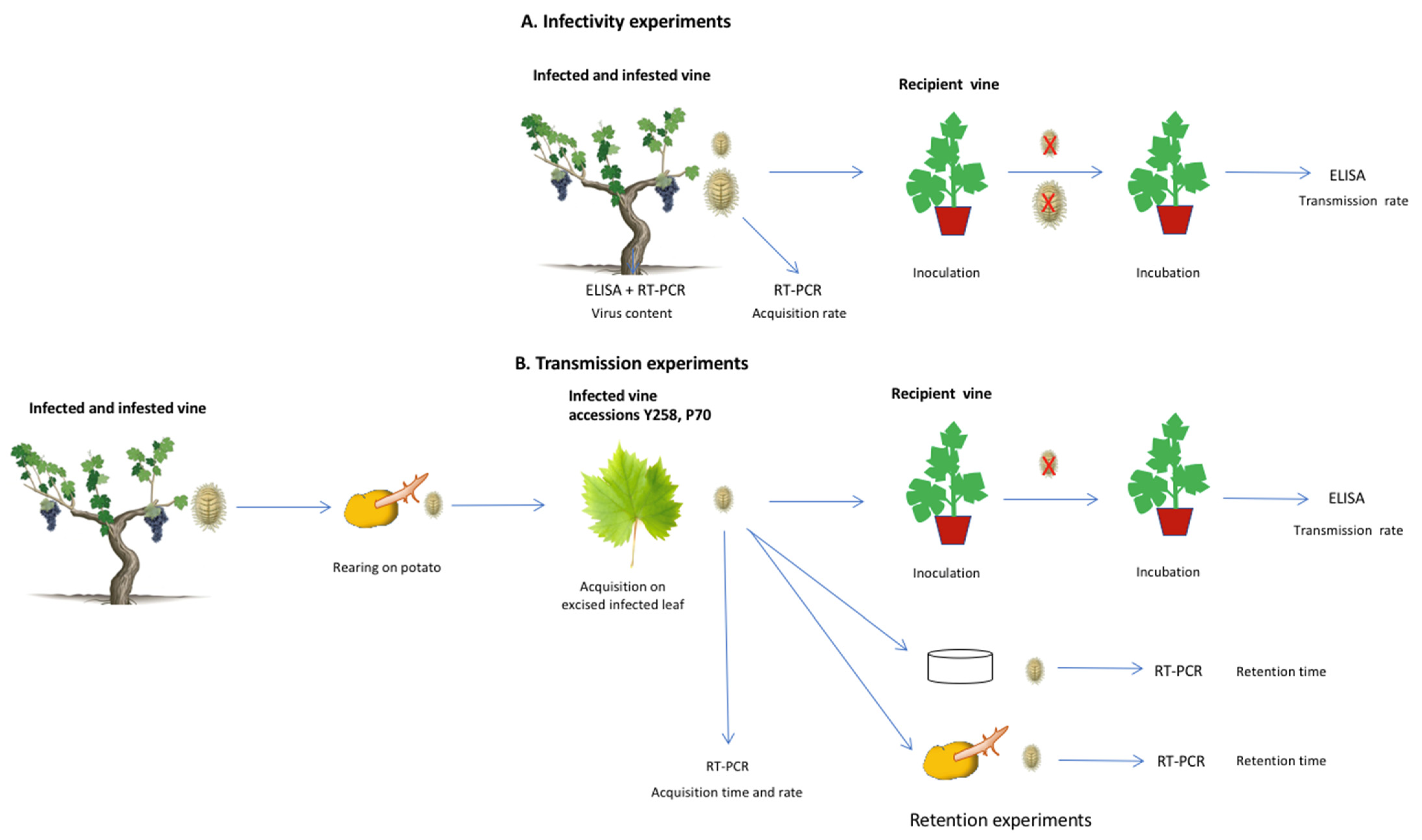
2.1. Infectivity Experiments
Inoculation experiments (
Figure 1A) were conducted to study the transmission ability of natural populations sampled from infected vineyard vines, at different larval stages.
The
H. bohemicus populations originated from commercial vine plots at six locations in Alsace (north-eastern France): Bennwihr, Colmar, Kientzheim, Nothalten, Ribeauvillé, and Turckheim, where it was the only mealybug species present. Heavily infested grapevines were first tested by enzyme-linked immunosorbent assay (ELISA) for GLRaV-1, -2, and -3, and GVA (
Table 1) before sampling mealybug-bearing leaves for infectivity experiments. Additional ELISA and reverse transcription polymerase chain reaction (RT-PCR) tests were performed for grapevines for which initial results were not clear-cut. Each grapevine tested was identified by its spatial location (row number, stock number). The distribution of viruses among the grapevines tested within each plot is given in
Table 1. We attempted to obtain a representative number of the different virus combinations among source grapevines hosting
H. bohemicus colonies. GLRaV-2 being rare, associations with this virus were few.
For a portion of the plants tested, a multiplex reverse transcription polymerase chain reaction (RT-PCR) was used to detect GLRaV-1, -2, and -3, and GVA in insect samples (1 to 74 L2) before infectivity experiments, using the following primers for GLRaV-1 (‘LR1-H70F1’ GTTGGTGAATTCTCCGTTCGT and ‘LR1-H70R1’ ACTTCGCTTGAACGAGTTATAC), GLRaV-2 (‘P19qtF’ ATGGAGTATTGTTTGAAGCAGGTAC and ‘P24qtR’ AGAATGTCTTCAGCTTCATAAGGAG), GLRaV-3 (‘LR3-POLF1’ ACGTAACGGGGCAGAATATAGT and ‘LR3-POLR1’ TATCAACACCAAGTGTCAAGAGTA), and GVA (‘GVA-CPF1’ GGCTACGACCGAAATATGTAC and ‘GVA-CPR1’ AGAAACGATGGGTCATCCATC), following the protocol developed by Beuve et al. [
28]. Nymphs were stored in 1.5 mL Eppendorf tubes with 150 μL RLT buffer (RNeasy Plant Mini Kit™; Qiagen, Les Ulis, France) containing 1% β-mercaptoethanol, and were kept at −20 °C before total RNA extraction and detection of viruses by RT-PCR. PCR products were visualised under UV light on a 2% agarose gel stained with ethidium bromide. RNA extracts from GLRaV-1, -2, and -3, and GVA infected grapevines were used as virus positive controls, and RNAse-free water (Kit Qiagen™) was used as the negative control.
Virus-free grapevines were obtained from rooted cuttings of Vitis vinifera cv. Pinot noir, frequently planted in vineyards of Alsace, Champagne, and Burgundy (clones P114 and mainly P115), or from germinated pips of Pinot noir and Pinot blanc. Mother plants of the cuttings were tested with ELISA and RT-PCR for the absence of leafroll and rugose wood viruses (GLRaV-1, -2, and -3, and GVA). Plants were grown in individual pots under greenhouse conditions until the 6–12 leaf stage and then used in transmission experiments. They were sprayed bi-monthly with an insecticide (alternatively Confidor™ 0.35 mL/L, Kiros™ 1 mL/L, or Fuoro™ 3 mL/L) to ensure the absence of insects; this spraying was stopped at least one month before insect inoculation. A sub-sample of ten recipient plants was tested to determine their virus-free status by ELISA prior to transmission experiments.
Since viruses are unevenly distributed on grapevines, several leaf pieces were cut from infected grapevines bearing first instars nymphs (L1) (2 to 100 individuals, mean ± sd = 34 ± 33) and attached with hairclips onto the leaves of virus-free recipient grapevines (3–4 leaf pieces per plant). Insects crawled off as the leaf fragments dried. As for the L2 nymphs, these were gently stimulated to move, then transferred with a fine paintbrush inside small cages (h = 8 mm, internal diameter = 13 mm). The cages were then attached with hairclips to leaves of the recipient plants. The number of recipient plants used according to the origin and cultivar of the virus combinations is given in
Table 2. The numbers of nymphs used to settle on recipient plants varied greatly according to the numbers collected from the source grapevine (Bennwihr: 2 to 100 L2, mean ± sd = 32 ± 20; other locations: 1 to 60 L2, mean ± sd = 20 ± 16). Finally, batches of 2 to 70 overwintering L2 (mean ± sd = 33 ± 19) were collected between January to early April under the bark of infected vines from Bennwihr and transferred onto recipient vines at laboratory temperature. Each recipient plant was isolated from the others under a 0.1 mm mesh micro-perforated plastic bag (‘bread bags’, Sealed Air SAS, Épernon, France), firmly secured to the pot using a rubber band to prevent nymphs from migrating between plants. Transmission experiments were conducted at 20–23 °C, 16 h/8 h (L/D) under artificial light. After 5 to 7 days of the inoculation access period (IAP), source leaves or cages were withdrawn. Mealybugs on leaves were removed with a paintbrush, after which grapevines were immediately sprayed with mevinphos (4 mL/l Phosdrin W10™) to kill the remaining insects. After two days, the treated plants were checked for any surviving insects, then transferred into a glasshouse compartment dedicated to recipient plants only. Recipient grapevines with nymphs from uninfected grapevines grown under the same conditions were used as negative controls. In late November, the recipient grapevines were pruned back to two buds and kept under an unheated glasshouse for overwintering. In spring, they were transferred into a heated glasshouse. All recipient plants were periodically sprayed with insecticide and fungicide, and pruned to avoid overgrowth until the end of the study.
The infection of recipient vines was assessed by double-antibody sandwich enzyme-linked immunosorbent assay (DAS-ELISA). The grapevines were checked by ELISA 4–6 and 8–12 months after IAP, and up to 18–24 months for surviving plants that had previously remained negative. The plants inoculated in late September could not be tested before cold storage and were therefore tested first about 6–7 months after IAP. Regardless of the recipient grapevine, these were systematically tested for the presence of GLRaV-1 and -3 and GVA. GLRaV-2 was only tested for when the source grapevine had been infected with this virus. The accession Y258 (Armenian cv. Liali Bidona, also named Vardabuyr), multi-infected by GLRaV-1 and -3, and GVA, was used as a positive control for the three viruses. The accession Chardonnay V38, infected by GLRaV-2 and -3, was used as the positive control for GLRaV-2. Healthy P115 cuttings were used as negative controls. Tissue extracts were obtained from pooled fragments of three leaves. Leaf fragments were ground (1 g leaves for 5 mL buffer) inside extraction bags with a bullet blender (Homex 5™, Bioreba, Reinach, Switzerland). Polyclonal antibodies raised against GLRaV-1, -2, or -3, or GVA produced in the laboratory were used in a biotine-streptavidine procedure [
29]. Absorbance values were recorded at 405 nm using a Multiskan™ microplate reader (Thermo Labsystems, Helsinki, Finland). Values above the mean of six healthy controls (six replicates per plate) plus three times their standard deviation were considered positive.
Chi-square tests were used to compare the detection rates in L2 nymphs, according to virus species and life cycle. A p-value < 0.05 was considered as the threshold for significance. Statistical analyses were performed with R, version 2.10.
2.2. Controlled Transmission Experiments
Firstly, the rate of virus acquisition by L1 nymphs was investigated according to feeding time and to nymph numbers (
Figure 1B). Secondly, the time of virus acquisition was refined with L2 nymphs. Based on the results obtained, optimal times for virus acquisition and inoculation were then investigated for these two stages. Lastly, the virus retention time of L2, either starved or fed on potatoes, was tested after 3 days AAP on infected grapevines (
Figure 1B).
The H. bohemicus culture originated from the Riesling plot at Colmar. L2 nymphs were collected in April from new leaf buds and raised on potato sprouts inside glass jars, closed with a 28 μm mesh tissue to enable airflow, and kept in the dark at 20–23 °C. When they became adults and gave birth to larval clusters, L1 nymphs were collected to determine their rate of virus acquisition according to feeding time and optimal AAP and IAP. L2 nymphs used to evaluate the virus acquisition rate according to feeding time were collected on the same plot and reared for at least one month on potato. Those used for optimal AAP/IAP and retention time were born on potato.
Virus source cuttings from accessions of our reference collection of grapevine viruses [
30] were rooted in a greenhouse. Accession Y258 infected with GLRaV-1 and -3, and GVA was used as the virus source plant to determine minimal AAP and IAP by L1 and the rate of acquisition by L2 nymphs according to time. Accession P70 of Pinot noir infected with GLRaV-1 and GVA [
31] was used as the virus source plant to determine minimal AAP and IAP by L2 nymphs. P70 was chosen because its viruses are well transmitted by
Pa. corni [
32] and
Phenacoccus aceris (Signoret) [
13]. The virus content of the source plants was checked by ELISA and RT-PCR prior to the experiments.
Leaves of virus source grapevines were collected and placed individually into tight and round polystyrene crystal boxes. A small wet cotton piece was wrapped around the cut petiole of each leaf, then tightly swathed inside ParafilmTM to retain the water supply during AAP. Mealybugs were collected with a fine paintbrush from sprouted potatoes and transferred onto the underside of source grapevines leaves for AAP.
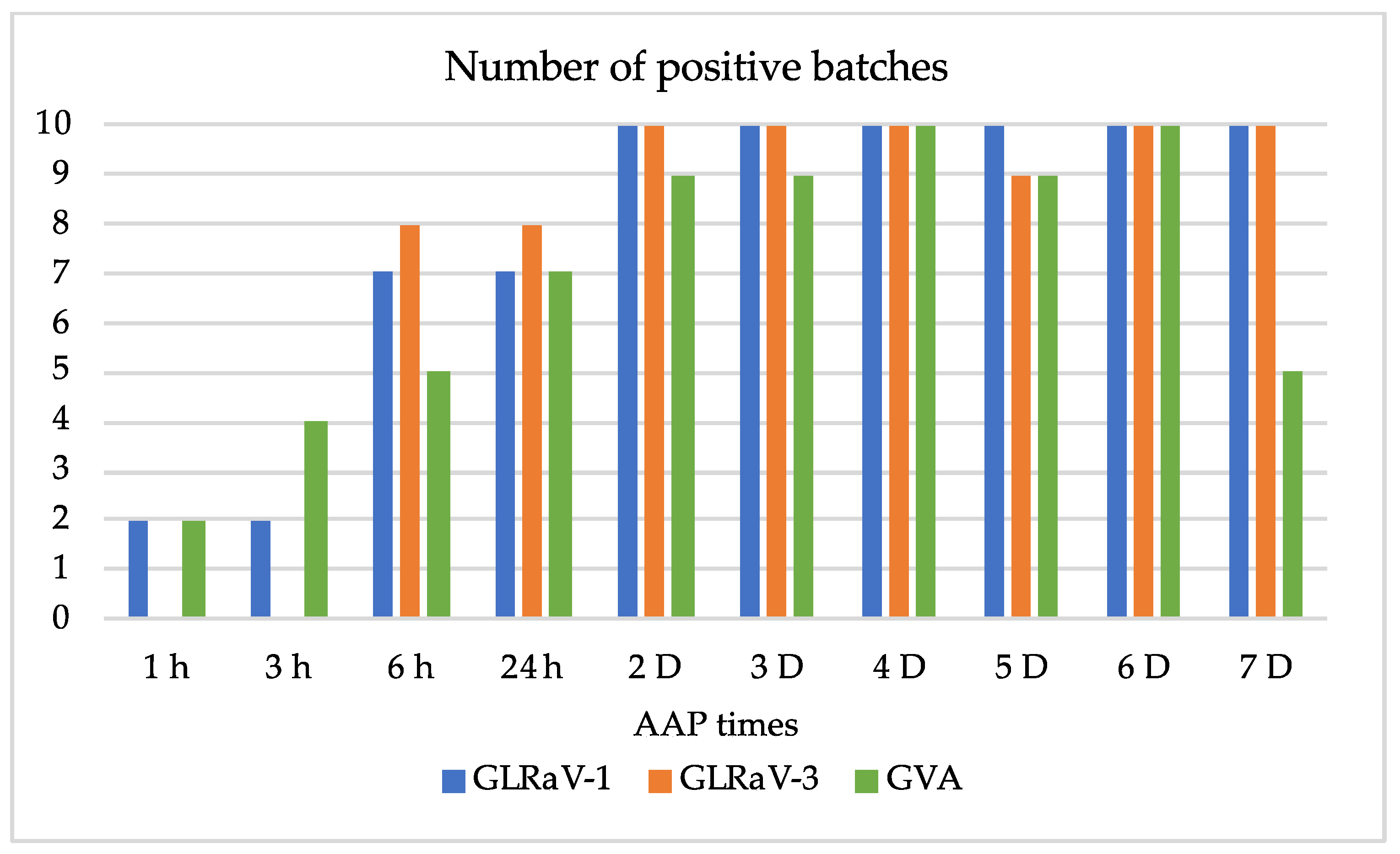
Rate of virus acquisition according to time of feeding: AAP of L1 nymphs on Y258 leaves was tested for 24, 48, and 72 h. For each time, virus detection was conducted by RT-PCR on five samples of 5, 10, and 20 L1. For L2, AAP of 1, 3, 6, 24 h, and from 2 up to 7 days, was tested on leaves of Y258. Ten mealybugs per batch were chosen based on the results obtained with L1, and virus detection by RT-PCR was performed on ten batches of L2 for each AAP. The presence of viruses was checked in fragments of source leaves before AAP. A batch of ten mealybugs collected from potato rearing was used as a healthy control.
Minimal time for virus acquisition and inoculation: After 1, 6, or 24 h AAP on Y258 leaves for L1, and on P70 leaves for L2, nymphs were transferred into small cages, which were clipped onto the leaves of five virus-free recipient cuttings (P115) for 1, 6, or 24 h for IAP (30 nymphs per plant distributed on two leaves), allowing for each growth stage a total of 9 AAP × IAP pairs. Each recipient plant was enclosed in a bread bag, as previously described. Plants were placed at 20–23 °C, 16 h/8 h (L/D), under artificial light. After IAP, the recipient plants were immediately sprayed with an insecticide. Grapevine cuttings of the same age, without nymphs and kept in the same conditions under a glasshouse, were used as negative controls.
Leaves of the recipient vines were tested by ELISA, as described above. First detection tests were conducted on leaves ca. 4 months after the transmission experiments. After a dormancy period under an unheated glasshouse, plants were tested again 10–12 months after IAP. Virus source grapevines and healthy cuttings were used as positive and negative controls, respectively.
To compare inoculation capacities between H. bohemicus populations, L2 nymphs were collected in April from three plots located at Bennwihr, Turckheim, and Ribeauvillé, and reared separately on potato tubers. Inoculation experiments were conducted with these mealybugs (after a latent period of ≥two months on potato, a non-host for leafroll viruses, thus being considered to be non-viruliferous) and their progeny (virus-free, as leafroll viruses are not transovarially transmitted) in June–July and October. Mealybugs from Bennwihr were placed on leaves of infected grapevines from Nothalten (see Virus acquisition above), whereas those from Turckheim and Ribeauvillé were placed on leaves of infected grapevines from Bennwihr for an AAP of 7 days (Nothalten and Colmar populations were too low to allow testing). After AAP, mealybugs were transferred onto healthy P115 cuttings for an IAP of 7 days.