Prevalence of Anti-SARS-CoV-2 Antibodies and Potential Determinants among the Belgian Adult Population: Baseline Results of a Prospective Cohort Study
Abstract
:1. Introduction
2. Materials and Methods
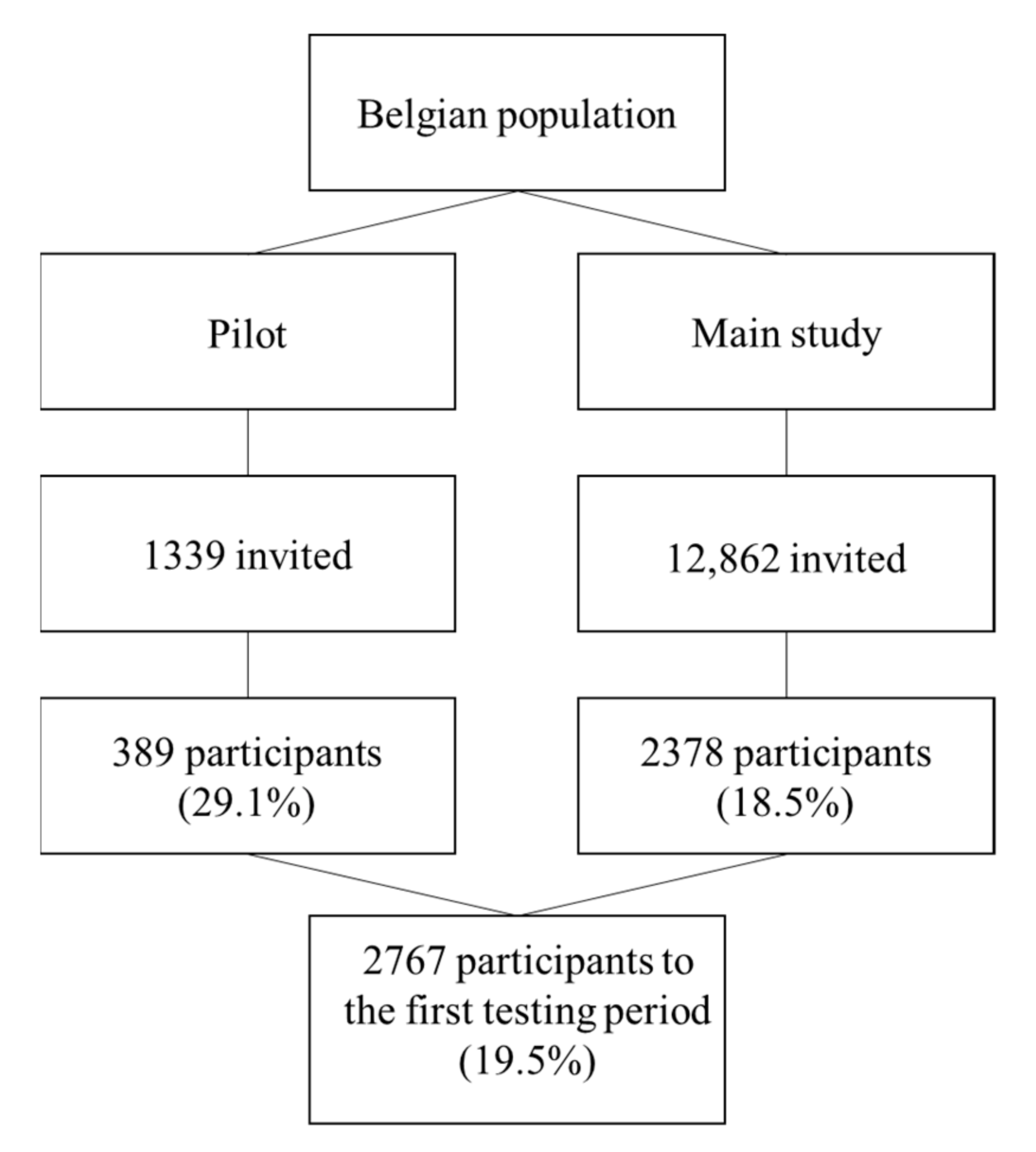
2.1. Design, Setting, and Study Population
2.2. Data Collection
2.3. Outcomes and Potential Determinants
2.4. Sample Size
2.5. Time Periods Considered for Trend Analyses
2.6. Statistical Analyses
3. Results
3.1. Description of the Population
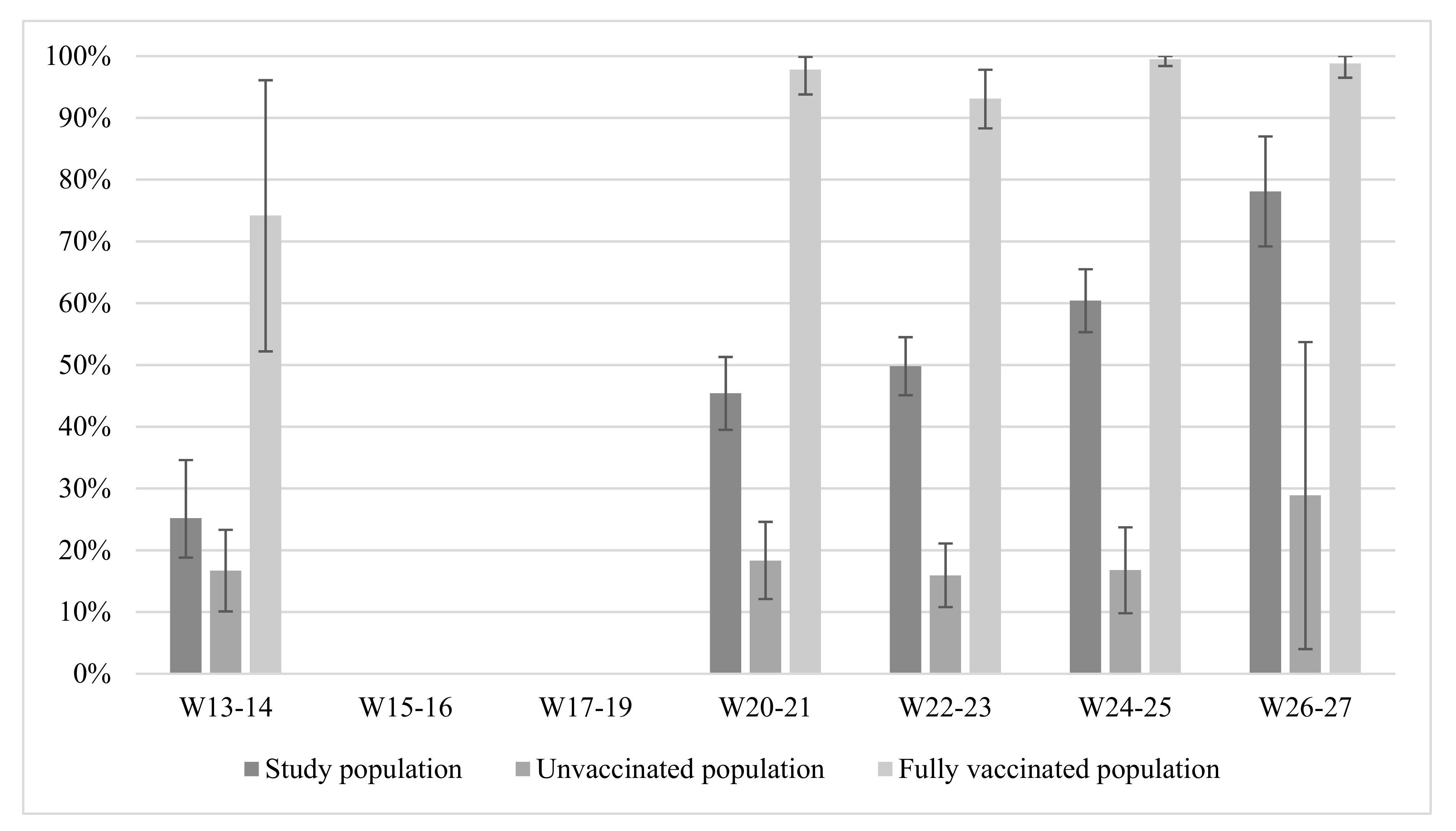
3.2. Prevalence of Anti-SARS-CoV-2 Antibodies over Time among the Study Population and by Vaccination Status
3.3. Determinants of the Prevalence of Antibodies among Vaccinated and Unvaccinated Populations
3.3.1. Vaccinated Population
3.3.2. Unvaccinated Population
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- WHO/Europe | Coronavirus Disease (COVID-19) Outbreak—About the Virus. Available online: https://www.euro.who.int/en/health-topics/health-emergencies/coronavirus-covid-19/novel-coronavirus-2019-ncov (accessed on 24 January 2022).
- Nine Belgians Safely Repatriated from Wuhan in China | Coronavirus COVID-19. Available online: https://web.archive.org/web/20200407125919/https://www.info-coronavirus.be/en/news/nine-belgians-safely-repatriated-from-wuhan-in-chin/ (accessed on 24 January 2022).
- Epistat—COVID-19 Monitoring. Available online: https://epistat.wiv-isp.be/covid/ (accessed on 13 December 2020).
- Advies 9618—Prioritering van Risicogroepen Voor SARS-CoV-2 Vaccinatie (Fase Ib). Available online: https://www.health.belgium.be/nl/advies-9618-prioritering-van-risicogroepen-voor-sars-cov-2-vaccinatie-fase-ib (accessed on 9 February 2021).
- CDC Coronavirus Disease 2019 (COVID-19). Available online: https://www.cdc.gov/coronavirus/2019-ncov/science/science-briefs/vaccine-induced-immunity.html (accessed on 25 January 2022).
- COVID-19 Surveillance. Available online: https://www.healthybelgium.be/en/health-status/covid-19-crisis/covid-19-surveillance (accessed on 7 February 2022).
- Bobrovitz, N.; Arora, R.K.; Cao, C.; Boucher, E.; Liu, M.; Donnici, C.; Yanes-Lane, M.; Whelan, M.; Perlman-Arrow, S.; Chen, J.; et al. Global Seroprevalence of SARS-CoV-2 Antibodies: A Systematic Review and Meta-Analysis. PLoS ONE 2021, 16, e0252617. [Google Scholar] [CrossRef] [PubMed]
- Stringhini, S.; Zaballa, M.-E.; Pullen, N.; Perez-Saez, J.; de Mestral, C.; Loizeau, A.J.; Lamour, J.; Pennacchio, F.; Wisniak, A.; Dumont, R.; et al. Seroprevalence of Anti-SARS-CoV-2 Antibodies 6 Months into the Vaccination Campaign in Geneva, Switzerland, 1 June to 7 July 2021. Eurosurveillance 2021, 26, 2100830. [Google Scholar] [CrossRef] [PubMed]
- Canto E Castro, L.; Gomes, A.; Serrano, M.; Pereira, A.H.G.; Ribeiro, R.; Napoleão, P.; Domingues, I.; Silva, C.; Fanczal, J.; Afonso, Â.; et al. Longitudinal SARS-CoV-2 Seroprevalence in Portugal and Antibody Maintenance 12 Months after Infection. Eur. J. Immunol. 2021, 52, 149–160. [Google Scholar] [CrossRef] [PubMed]
- Siller, A.; Seekircher, L.; Wachter, G.A.; Astl, M.; Tschiderer, L.; Pfeifer, B.; Schennach, H.; Willeit, P. Seroprevalence, Waning, and Correlates of Anti-SARS-CoV-2 IgG Antibodies in Tyrol, Austria: Large-Scale Study of 35,193 Blood Donors Conducted between June 2020 and September 2021. Viruses 2022, 14, 568. [Google Scholar] [CrossRef]
- Coronavirus (COVID-19)—Office for National Statistics. Available online: https://www.ons.gov.uk/peoplepopulationandcommunity/healthandsocialcare/conditionsanddiseases (accessed on 19 January 2022).
- Belgium COVID-19 Dashboard—Sciensano. Available online: http://datastudio.google.com/reporting/c14a5cfc-cab7-4812-848c-0369173148ab (accessed on 13 January 2022).
- Mortgat, L.; Verdonck, K.; Hutse, V.; Thomas, I.; Barbezange, C.; Heyndrickx, L.; Fischer, N.; Vuylsteke, B.; Kabouche, I.; Ariën, K.K.; et al. Prevalence and Incidence of Anti-SARS-CoV-2 Antibodies among Healthcare Workers in Belgian Hospitals before Vaccination: A Prospective Cohort Study. BMJ Open 2021, 11, e050824. [Google Scholar] [CrossRef]
- Coenen Prevalence and Incidence of Antibodies against SARS-CoV-2 among Primary Healthcare Providers in Belgium: Results First Testing Point—January 2021. Available online: https://www.sciensano.be/en/biblio/prevalence-and-incidence-antibodies-against-sars-cov-2-among-primary-healthcare-providers-belgium-0 (accessed on 7 February 2022).
- Dusyburgh, E.; Callies, M.; Kabouche, I.; Merckx, J.; Roelants, M.; Vermeulen, M.; Desombere, I. Prevalence and Incidence of Antibodies against Sars-Cov-2 in Children and School Staff Measured between December 2020 and June 2021: An Observational Sero-Prevalence Prospective Cohort Study. In Findings of the First Testing Period; Sciensano: Brussels, Belgium, 2021. [Google Scholar]
- Meyers, E. SARS-CoV-2 Seroprevalence among Vaccinated Nursing Home Residents and Staff in Belgium in August 2021—Brief Communication on Preliminary Results of the Scope Study. Available online: https://www.sciensano.be/en/biblio/sars-cov-2-seroprevalence-among-vaccinated-nursing-home-residents-and-staff-belgium-august-2021 (accessed on 7 February 2022).
- Manenti, A.; Gianchecchi, E.; Dapporto, F.; Leonardi, M.; Cantaloni, P.; Fattorini, F.; Piu, P.; Bollati, V.; Pastorino, U.; Apolone, G.; et al. Evaluation and Correlation between SARS-CoV-2 Neutralizing and Binding Antibodies in Convalescent and Vaccinated Subjects. J. Immunol. Methods 2022, 500, 113197. [Google Scholar] [CrossRef]
- Pisanic, N.; Randad, P.R.; Kruczynski, K.; Manabe, Y.C.; Thomas, D.L.; Pekosz, A.; Klein, S.L.; Betenbaugh, M.J.; Clarke, W.A.; Laeyendecker, O.; et al. COVID-19 Serology at Population Scale: SARS-CoV-2-Specific Antibody Responses in Saliva. J. Clin. Microbiol. 2020, 59, e02204-20. [Google Scholar] [CrossRef]
- Augustine, S.A.J.; Eason, T.N.; Simmons, K.J.; Curioso, C.L.; Griffin, S.M.; Ramudit, M.K.D.; Plunkett, T.R. Developing a Salivary Antibody Multiplex Immunoassay to Measure Human Exposure to Environmental Pathogens. J. Vis. Exp. JoVE 2016, e54415. [Google Scholar] [CrossRef]
- Vandermeulen, C.; Duysburgh, E.; Desombere, I.; Boey, L.; Roelants, M. Seroprevalence of Sars-CoV-2 Antibodies in School Aged Children in Two Regions with Difference of COVID-19 Disease. In Validation Study of Saliva Test for SARS-Cov-2 Antibodies in Children; Interim Report; KULeuven: Leuven, Belgium; Sciensano: Brussels, Belgium, 2021; p. 28. [Google Scholar]
- Von Elm, E.; Altman, D.G.; Egger, M.; Pocock, S.J.; Gøtzsche, P.C.; Vandenbroucke, J.P. STROBE Initiative The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) Statement: Guidelines for Reporting Observational Studies. J. Clin. Epidemiol. 2008, 61, 344–349. [Google Scholar] [CrossRef] [Green Version]
- Herzog, S.; De Bie, J.; Abrams, S.; Wouters, I.; Ekinci, E.; Patteet, L.; Coppens, A.; De Spiegeleer, S.; Beutels, P.; Van Damme, P.; et al. Seroprevalence of IgG Antibodies against SARS Coronavirus 2 in Belgium—A Serial Prospective Cross-Sectional Nationwide Study of Residual Samples. Eurosurveillance 2022, 27, 2100419. [Google Scholar] [CrossRef]
- Nouveau Coronavirus (Maladie: COVID-19, Virus: SARS-CoV-2) | Coronavirus COVID-19. Available online: https://covid-19.sciensano.be/fr (accessed on 3 March 2022).
- Alejo, J.L.; Mitchell, J.; Chang, A.; Chiang, T.P.Y.; Massie, A.B.; Segev, D.L.; Makary, M.A. Prevalence and Durability of SARS-CoV-2 Antibodies Among Unvaccinated US Adults by History of COVID-19. JAMA 2022, 327, 1085. [Google Scholar] [CrossRef]
- Duysburgh, E.; Mortgat, L.; Barbezange, C.; Dierick, K.; Fischer, N.; Heyndrickx, L.; Hutse, V.; Thomas, I.; Gucht, S.V.; Vuylsteke, B.; et al. Persistence of IgG Response to SARS-CoV-2. Lancet Infect. Dis. 2021, 21, 163–164. [Google Scholar] [CrossRef]
- Yu, Y.; Esposito, D.; Kang, Z.; Lu, J.; Remaley, A.T.; De Giorgi, V.; Chen, L.N.; West, K.; Cao, L. MRNA Vaccine-Induced Antibodies More Effective than Natural Immunity in Neutralizing SARS-CoV-2 and Its High Affinity Variants. Sci. Rep. 2022, 12, 2628. [Google Scholar] [CrossRef]
- Huang, A.T.; Garcia-Carreras, B.; Hitchings, M.D.T.; Yang, B.; Katzelnick, L.C.; Rattigan, S.M.; Borgert, B.A.; Moreno, C.A.; Solomon, B.D.; Trimmer-Smith, L.; et al. A Systematic Review of Antibody Mediated Immunity to Coronaviruses: Kinetics, Correlates of Protection, and Association with Severity. Nat. Commun. 2020, 11, 4704. [Google Scholar] [CrossRef]
- Marra, A.R.; Kobayashi, T.; Suzuki, H.; Alsuhaibani, M.; Tofaneto, B.M.; Bariani, L.M.; de Auler, M.A.; Salinas, J.L.; Edmond, M.B.; Doll, M.; et al. Short-Term Effectiveness of COVID-19 Vaccines in Immunocompromised Patients: A Systematic Literature Review and Meta-Analysis. J. Infect. 2021, 84, 297–310. [Google Scholar] [CrossRef]
- Amato, M.; Werba, J.P.; Frigerio, B.; Coggi, D.; Sansaro, D.; Ravani, A.; Ferrante, P.; Veglia, F.; Tremoli, E.; Baldassarre, D. Relationship between Influenza Vaccination Coverage Rate and COVID-19 Outbreak: An Italian Ecological Study. Vaccines 2020, 8, 535. [Google Scholar] [CrossRef]
- Fink, G.; Orlova-Fink, N.; Schindler, T.; Grisi, S.; Ferrer, A.P.S.; Daubenberger, C.; Brentani, A. Inactivated Trivalent Influenza Vaccination Is Associated with Lower Mortality among Patients with COVID-19 in Brazil. BMJ Evid.-Based Med. 2021, 26, 192–193. [Google Scholar] [CrossRef]
- Shrotri, M.; van Schalkwyk, M.C.I.; Post, N.; Eddy, D.; Huntley, C.; Leeman, D.; Rigby, S.; Williams, S.V.; Bermingham, W.H.; Kellam, P.; et al. T Cell Response to SARS-CoV-2 Infection in Humans: A Systematic Review. PLoS ONE 2021, 16, e0245532. [Google Scholar] [CrossRef]
- Liu, N.; Zhang, T.; Ma, L.; Zhang, H.; Wang, H.; Wei, W.; Pei, H.; Li, H. The Impact of ABO Blood Group on COVID-19 Infection Risk and Mortality: A Systematic Review and Meta-Analysis. Blood Rev. 2021, 48, 100785. [Google Scholar] [CrossRef]
- Rana, R.; Ranjan, V.; Kumar, N. Association of ABO and Rh Blood Group in Susceptibility, Severity, and Mortality of Coronavirus Disease 2019: A Hospital-Based Study From Delhi, India. Front. Cell. Infect. Microbiol. 2021, 11, 1071. [Google Scholar] [CrossRef]
- Zietz, M.; Zucker, J.; Tatonetti, N.P. Associations between Blood Type and COVID-19 Infection, Intubation, and Death. Nat. Commun. 2020, 11, 5761. [Google Scholar] [CrossRef] [PubMed]
- Beaumont, A.; Durand, C.; Ledrans, M.; Schwoebel, V.; Noel, H.; Le Strat, Y.; Diulius, D.; Colombain, L.; Médus, M.; Gueudet, P.; et al. Seroprevalence of Anti-SARS-CoV-2 Antibodies after the First Wave of the COVID-19 Pandemic in a Vulnerable Population in France: A Cross-Sectional Study. BMJ Open 2021, 11, e053201. [Google Scholar] [CrossRef] [PubMed]
- Grzelak, L.; Velay, A.; Madec, Y.; Gallais, F.; Staropoli, I.; Schmidt-Mutter, C.; Wendling, M.-J.; Meyer, N.; Planchais, C.; Rey, D.; et al. Sex Differences in the Evolution of Neutralizing Antibodies to Severe Acute Respiratory Syndrome Coronavirus 2. J. Infect. Dis. 2021, 224, 983–988. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, C.J.; Pade, C.; Gibbons, J.M.; Butler, D.K.; Otter, A.D.; Menacho, K.; Fontana, M.; Smit, A.; Sackville-West, J.E.; Cutino-Moguel, T.; et al. Prior SARS-CoV-2 Infection Rescues B and T Cell Responses to Variants after First Vaccine Dose. Science 2021, 372, 1418–1423. [Google Scholar] [CrossRef]
- Cromer, D.; Juno, J.A.; Khoury, D.; Reynaldi, A.; Wheatley, A.K.; Kent, S.J.; Davenport, M.P. Prospects for Durable Immune Control of SARS-CoV-2 and Prevention of Reinfection. Nat. Rev. Immunol. 2021, 21, 395–404. [Google Scholar] [CrossRef]
- Immune Responses and Immunity to SARS-CoV-2. Available online: https://www.ecdc.europa.eu/en/covid-19/latest-evidence/immune-responses (accessed on 13 January 2022).
- Levin, E.G.; Lustig, Y.; Cohen, C.; Fluss, R.; Indenbaum, V.; Amit, S.; Doolman, R.; Asraf, K.; Mendelson, E.; Ziv, A.; et al. Waning Immune Humoral Response to BNT162b2 COVID-19 Vaccine over 6 Months. N. Engl. J. Med. 2021, 385, e84. [Google Scholar] [CrossRef]
- HIQA Duration of Immunity (Protection from Reinfection) Following SARS-CoV-2 Infection. Available online: https://www.hiqa.ie/reports-and-publications/health-technology-assessment/duration-protective-immunity-protection (accessed on 13 January 2022).
- Catteau, L.; van Loenhout, J.; Stouten, V.; Billuart, M.; Hubin, P.; Haarhuis, F. Wyndham Thomas C Couverture Vaccinale et Impact Épidémiologique de La Campagne de Vaccination COVID-19 En Belgique. In Données Jusqu’au 31 Octobre 2021 Inclus 2021; Sciensano: Brussels, Belgium, 2021. [Google Scholar]
- Tartof, S.Y.; Slezak, J.M.; Fischer, H.; Hong, V.; Ackerson, B.K.; Ranasinghe, O.N.; Frankland, T.B.; Ogun, O.A.; Zamparo, J.M.; Gray, S.; et al. Effectiveness of MRNA BNT162b2 COVID-19 Vaccine up to 6 Months in a Large Integrated Health System in the USA: A Retrospective Cohort Study. Lancet 2021, 398, 1407–1416. [Google Scholar] [CrossRef]
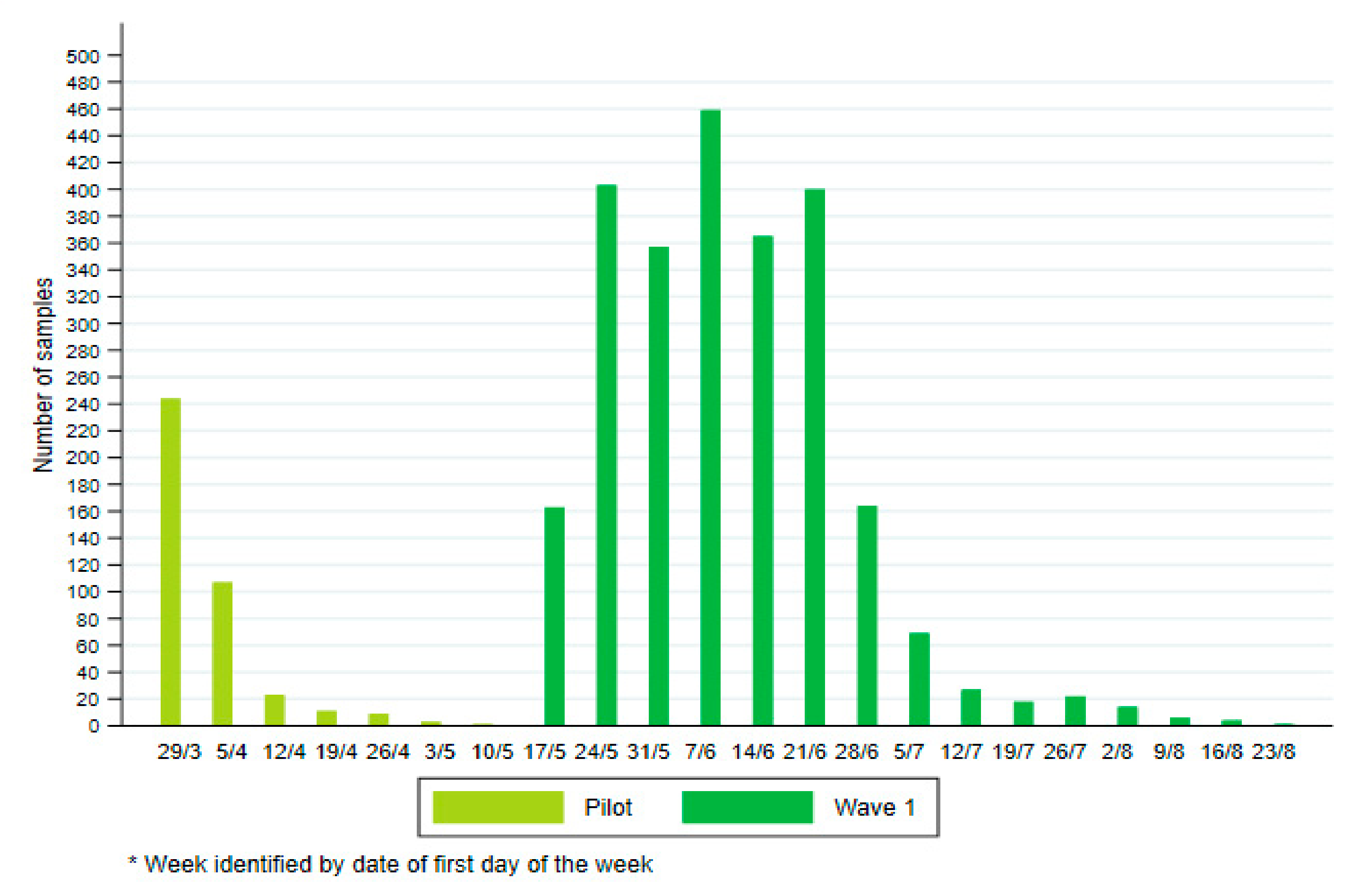
| Periods | Week of References | Dates |
|---|---|---|
| Period 1 | Week 13–14 | 29 March to 11 April 2021 |
| Period 2 | Week 20–21 | 17 May to 30 May 2021 |
| Period 3 | Week 22–23 | 31 May to 21 June 2021 |
| Period 4 | Week 24–25 | 14 June to 27 June 2021 |
| Period 5 | Week 26–27 | 28 June to 11 July 2021 |
| Overall Distribution | Distribution by Vaccination Status | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Study Population N = 2767 | Fully Vaccinated Population Since 2 or More Weeks N = 747 (28.5%) | Partially Vaccinated Population N = 874 (33.4%) | Unvaccinated Population N = 998 (38.1%) | ||||||
| % | Total N | % | Total N | % | Total N | % | Total N | ||
| Gender | Man | 45.1 | 1247 | 39.5 | 295 | 46.5 | 406 | 46.5 | 464 |
| Woman | 54.9 | 1520 | 60.5 | 452 | 53.5 | 468 | 53.5 | 534 | |
| Age | 18–49 yrs | 46.4 | 1285 | 21.6 | 161 | 35.6 | 311 | 75.1 | 749 |
| ≥50 yrs | 53.6 | 1482 | 78.4 | 586 | 64.4 | 563 | 24.9 | 249 | |
| Region | Flanders | 41.9 | 1160 | 34.1 | 255 | 42.8 | 374 | 47.6 | 475 |
| Brussels | 29.6 | 819 | 36.7 | 274 | 27.0 | 236 | 26.1 | 260 | |
| Wallonia | 28.5 | 788 | 29.2 | 218 | 30.2 | 264 | 26.4 | 263 | |
| Education | ≤Secondary | 42.7 | 1138 | 40.2 | 286 | 42.5 | 359 | 44.7 | 437 |
| Bachelor | 27.1 | 722 | 29.4 | 209 | 28.8 | 243 | 24.5 | 239 | |
| ≥Master | 30.2 | 807 | 30.5 | 217 | 28.8 | 243 | 30.8 | 301 | |
| Household size | 1 member | 15.2 | 382 | 21.6 | 153 | 15.4 | 129 | 10.4 | 100 |
| 2 members | 44.8 | 1124 | 54.0 | 383 | 49.4 | 414 | 34.1 | 327 | |
| 3 members | 15.5 | 389 | 9.9 | 70 | 14.8 | 124 | 20.3 | 195 | |
| ≥4 members | 24.4 | 612 | 14.5 | 103 | 20.4 | 171 | 35.2 | 338 | |
| Health care worker | Yes | 8.1 | 214 | 17.8 | 128 | 5.2 | 43 | 3.5 | 34 |
| No | 91.9 | 2423 | 82.2 | 592 | 94.8 | 786 | 96.5 | 924 | |
| Presence of at least one | Yes | 24.3 | 643 | 32.8 | 235 | 25.0 | 207 | 17.1 | 165 |
| chronic disease | No | 75.7 | 2002 | 67.2 | 482 | 75.0 | 622 | 82.9 | 801 |
| Presence of at least two | Yes | 5.7 | 151 | 8.1 | 58 | 6.2 | 51 | 3.8 | 37 |
| chronic diseases | No | 94.3 | 2494 | 91.9 | 659 | 93.8 | 778 | 96.2 | 929 |
| Overweight | Yes | 48.8 | 1299 | 54.4 | 392 | 51.3 | 430 | 41.8 | 407 |
| No | 51.2 | 1365 | 45.6 | 329 | 48.7 | 409 | 58.2 | 566 | |
| Obesity | Yes | 16.6 | 441 | 21.1 | 152 | 16.7 | 140 | 12.9 | 126 |
| No | 83.4 | 2223 | 78.9 | 569 | 83.3 | 699 | 87.1 | 847 | |
| Blood type | O blood type | 56.7 | 1020 | 58.0 | 286 | 58.1 | 349 | 52.8 | 331 |
| Non-O blood type | 43.3 | 780 | 42.0 | 207 | 41.9 | 252 | 47.2 | 296 | |
| Rhesus | Positive | 81.8 | 1411 | 81.4 | 380 | 81.8 | 472 | 82.9 | 503 |
| Negative | 18.2 | 313 | 18.6 | 87 | 18.2 | 105 | 17.1 | 104 | |
| Smokers | Yes | 14.7 | 394 | 10.1 | 73 | 13.4 | 114 | 19.5 | 191 |
| No | 85.3 | 2280 | 89.9 | 647 | 86.6 | 735 | 80.5 | 786 | |
| Influenza vaccine | Vaccinated | 37.4 | 1011 | 62.7 | 458 | 42.2 | 360 | 14.4 | 142 |
| Not vaccinated | 62.6 | 1693 | 37.3 | 273 | 57.8 | 493 | 85.6 | 844 | |
| Close contact with COVID-19 positive person | Yes | 27.2 | 737 | 23.3 | 170 | 22.7 | 194 | 34.1 | 337 |
| No | 56.3 | 1523 | 61.5 | 449 | 61.7 | 528 | 47.6 | 470 | |
| I do not know | 16.5 | 447 | 15.2 | 111 | 15.7 | 134 | 18.2 | 180 | |
| One or more positive COVID-19 test results | Yes | 11.8 | 306 | 11.0 | 78 | 10.7 | 87 | 12.8 | 121 |
| No | 88.2 | 2289 | 89.0 | 633 | 89.3 | 723 | 87.2 | 822 | |
| Preventive measures | Strictly followed the measures | 30.3 | 555 | 38.9 | 189 | 33.6 | 192 | 21.3 | 148 |
| Moderately followed the measures | 45.4 | 831 | 47.1 | 229 | 46.0 | 263 | 44.2 | 307 | |
| Insufficiently followed the measures | 24.3 | 444 | 14.0 | 68 | 20.5 | 117 | 34.4 | 239 | |
| Vaccinated Population (N = 593) | Unvaccinated Population (N = 838) | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Determinants | Categories | % | Total N | Unadjusted ° OR (95% CI) | Adjusted $ OR (95% CI) | % | Total N | Unadjusted ° OR (95% CI) | Adjusted $ OR (95% CI) |
| Gender | Man | 94.2 | 250 | Ref | Ref | 15.3 | 398 | Ref | Ref |
| Woman | 95.6 | 343 | 1.32 (0.57–3.12) | 1.09 (0.45–2.68) | 21.0 | 440 | 1.46 (1.01–2.12)※ | 1.33 (0.72–2.49) | |
| Age | 18–49 yrs | 97.4 | 137 | Ref | Ref | 18.5 | 638 | Ref | Ref |
| ≥50 yrs | 94.1 | 456 | 0.43 (0.13–1.39) | 0.34 (0.08–1.45) | 16.1 | 200 | 0.96 (0.72–1.28) | 0.87 (0.45–1.67) | |
| Region | Flanders | 97.7 | 196 | Ref | Ref | 16.4 | 388 | Ref | Ref |
| Brussels | 97.8 | 228 | 2.27 (0.78–6.60) | 2.07 (0.59–7.30) | 22.9 | 225 | 1.52 (0.97–2.37) | 0.76 (0.34–1.69) | |
| Wallonia | 93.9 | 169 | 0.81 (0.30–2.15) | 1.25 (0.40–3.90) | 19.6 | 225 | 1.25 (0.74–2.10) | 0.84 (0.34–2.05) | |
| Education | ≤Secondary | 93.8 | 219 | Ref | 17.2 | 358 | Ref | ||
| Bachelor | 95.9 | 169 | 1.54 (0.51–4.70) | 20.9 | 206 | 1.27 (0.80–2.02) | |||
| ≥Master | 96.4 | 177 | 1.78 (0.50–6.32) | 16.8 | 254 | 0.97 (0.61–1.56) | |||
| Household size | 1 member | 95.6 | 117 | Ref | 20.0 | 82 | Ref | ||
| 2 members | 95.1 | 303 | 0.89 (0.22–3.54) | 12.5 | 269 | 0.57 (0.27–1.22) | |||
| 3 members | 88.2 | 61 | 0.34 (0.07–1.65) | 13.5 | 174 | 0.62 (0.27–1.44) | |||
| ≥4 members | 97.9 | 84 | 2.13 (0.29–15.84) | 25.0 | 282 | 1.33 (0.64–2.77) | |||
| Health care worker | Yes | 95.2 | 103 | 0.99 (0.31–3.13) | 17.4 | 33 | 0.99 (0.38–2.57) | ||
| No | 95.2 | 470 | Ref | 17.6 | 770 | Ref | |||
| Presence of at least one chronic disease | Yes | 90.4 | 173 | 0.30 (0.12–0.76) ※ | 0.22 (0.08–0.62) ※ | 21.0 | 125 | 1.29 (0.78–2.14) | |
| No | 97.0 | 400 | Ref | Ref | 17.1 | 684 | Ref | ||
| Presence of at least two chronic diseases | Yes | 93.9 | 41 | 0.81 (0.18–3.73) | 19.7 | 27 | 1.15 (0.42–3.11) | ||
| No | 95.0 | 532 | Ref | Ref | 17.6 | 782 | Ref | ||
| BMI 25–30 kg/m2 | Yes | 93.8 | 306 | 0.59 (0.23–1.48) | 18.5 | 335 | 1.12 (0.75–1.66) | ||
| No | 96.3 | 271 | Ref | 16.8 | 481 | Ref | |||
| BMI > 30 kg/m2 | Yes | 95.0 | 108 | 1.02 (0.36–2.88) | 17.2 | 103 | 0.97 (0.55–1.72) | ||
| No | 94.9 | 469 | Ref | 17.6 | 713 | Ref | |||
| Blood type | O blood type | 13.7 | 291 | Ref | Ref | ||||
| Non-O blood type | 22.5 | 236 | 1.83 (1.11–3.01) ※ | 2.00 (1.09–3.67) ※ | |||||
| Rhesus | Positive | 15.6 | 422 | 0.56 (0.32–1.00) | |||||
| Negative | 24.9 | 90 | Ref | ||||||
| Smokers | Yes | 8.5 | 161 | 0.36 (0.18–0.72) ※ | 0.44 (0.14–1.33) | ||||
| No | 20.2 | 658 | Ref | Ref | |||||
| Close contact with COVID-19 positive person | Yes | 29.9 | 282 | 3.28 (2.08–5.17) * | 1.44 (0.71–2.92) | ||||
| No | 11.5 | 388 | Ref | Ref | |||||
| I do not know | 14.2 | 159 | 1.27 (0.69–2.31) | 0.64 (0.27–1.52) | |||||
| One or more positive COVID-19 test results | Yes | 68.7 | 103 | 17.62 (10.16–30.57) * | 11.04 (4.69–26.02) * | ||||
| No | 11.1 | 690 | Ref | Ref | |||||
| Preventive measures | Strictly followed the measures | 16.1 | 118 | Ref | |||||
| Moderately followed the measures | 19.4 | 261 | 1.26 (0.69–2.31) | ||||||
| Insufficiently followed the measures | 22.3 | 212 | 1.50 (0.80–2.81) | ||||||
| Influenza vaccine | Vaccinated | 96.8 | 361 | 2.62 (1.06–6.49) ※ | 3.79 (1.30–11.07) ※ | 15.5 | 104 | 0.85 (0.46–1.57) | |
| Not vaccinated | 92.0 | 222 | Ref | Ref | 17.8 | 722 | Ref | ||
| Type of vaccination | mRNA vaccine | 96.5 | 461 | 5.10 (1.89–13.79) * | 5.38 (1.72–16.80) ※ | ||||
| adenoviral-vectored vaccine | 84.3 | 58 | Ref | Ref | |||||
| Number of days since the last vaccination | 567 | 1.00 (0.99–1.02) | |||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Leclercq, V.; Van den Houte, N.; Gisle, L.; Roukaerts, I.; Barbezange, C.; Desombere, I.; Duysburgh, E.; Van der Heyden, J. Prevalence of Anti-SARS-CoV-2 Antibodies and Potential Determinants among the Belgian Adult Population: Baseline Results of a Prospective Cohort Study. Viruses 2022, 14, 920. https://doi.org/10.3390/v14050920
Leclercq V, Van den Houte N, Gisle L, Roukaerts I, Barbezange C, Desombere I, Duysburgh E, Van der Heyden J. Prevalence of Anti-SARS-CoV-2 Antibodies and Potential Determinants among the Belgian Adult Population: Baseline Results of a Prospective Cohort Study. Viruses. 2022; 14(5):920. https://doi.org/10.3390/v14050920
Chicago/Turabian StyleLeclercq, Victoria, Nayema Van den Houte, Lydia Gisle, Inge Roukaerts, Cyril Barbezange, Isabelle Desombere, Els Duysburgh, and Johan Van der Heyden. 2022. "Prevalence of Anti-SARS-CoV-2 Antibodies and Potential Determinants among the Belgian Adult Population: Baseline Results of a Prospective Cohort Study" Viruses 14, no. 5: 920. https://doi.org/10.3390/v14050920
APA StyleLeclercq, V., Van den Houte, N., Gisle, L., Roukaerts, I., Barbezange, C., Desombere, I., Duysburgh, E., & Van der Heyden, J. (2022). Prevalence of Anti-SARS-CoV-2 Antibodies and Potential Determinants among the Belgian Adult Population: Baseline Results of a Prospective Cohort Study. Viruses, 14(5), 920. https://doi.org/10.3390/v14050920






