Soluble IL-2R Levels at Baseline Predict the Development of Severe Respiratory Failure and Mortality in COVID-19 Patients
Abstract
:1. Introduction
2. Materials and Methods
Statistical Analysis
3. Results
3.1. Demographic, Clinical, and Laboratory Characteristics
3.2. Outcome of Patients
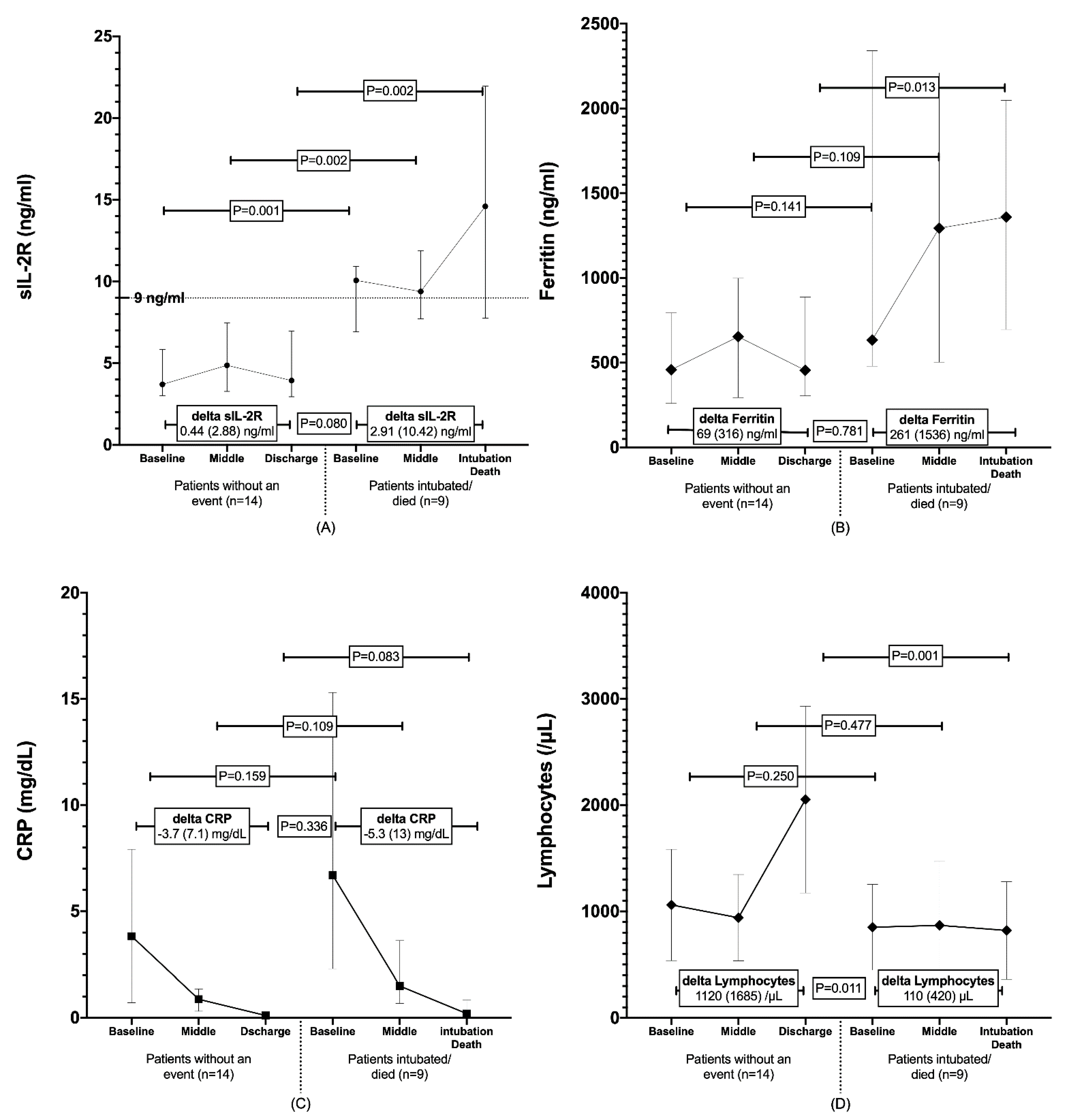
3.3. sIL-2R, Ferritin, CRP, and Lymphocytes Levels Alteration during Hospitalisation and Outcome
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Dalekos, G.N.; Stefos, A.; Georgiadou, S.; Lygoura, V.; Michail, A.; Ntaios, G.; Samakidou, A.; Giannoulis, G.; Gabeta, S.; Vlychou, M.; et al. Lessons from pathophysiology: Use of individualized combination treatments with immune interventional agents to tackle severe respiratory failure in patients with COVID-19. Eur. J. Intern. Med. 2021, 88, 52–62. [Google Scholar] [CrossRef]
- Giamarellos-Bourboulis, E.J.; Netea, M.G.; Rovina, N.; Akinosoglou, K.; Antoniadou, A.; Antonakos, N.; Damoraki, G.; Gkavogianni, T.; Adami, M.E.; Katsaounou, P.; et al. Complex Immune Dysregulation in COVID-19 Patients with Severe Respiratory Failure. Cell Host Microbe 2020, 27, 992–1000.e1003. [Google Scholar] [CrossRef] [PubMed]
- Speletas, M.; Dadouli, K.; Syrakouli, A.; Gatselis, N.; Germanidis, G.; Mouchtouri, V.A.; Koulas, I.; Samakidou, A.; Nikolaidou, A.; Stefos, A.; et al. MBL deficiency-causing B allele (rs1800450) as a risk factor for severe COVID-19. Immunobiology 2021, 226, 152136. [Google Scholar] [CrossRef] [PubMed]
- Berlin, D.A.; Gulick, R.M.; Martinez, F.J. Severe COVID-19. N. Engl. J. Med. 2020, 383, 2451–2460. [Google Scholar] [CrossRef] [PubMed]
- Verity, R.; Okell, L.C.; Dorigatti, I.; Winskill, P.; Whittaker, C.; Imai, N.; Cuomo-Dannenburg, G.; Thompson, H.; Walker, P.G.T.; Fu, H.; et al. Estimates of the severity of coronavirus disease 2019: A model-based analysis. Lancet Infect. Dis. 2020, 20, 669–677. [Google Scholar] [CrossRef]
- Zhou, F.; Yu, T.; Du, R.; Fan, G.; Liu, Y.; Liu, Z.; Xiang, J.; Wang, Y.; Song, B.; Gu, X.; et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study. Lancet 2020, 395, 1054–1062. [Google Scholar] [CrossRef]
- Ruan, Q.; Yang, K.; Wang, W.; Jiang, L.; Song, J. Clinical predictors of mortality due to COVID-19 based on an analysis of data of 150 patients from Wuhan, China. Intensive Care Med. 2020, 46, 846–848. [Google Scholar] [CrossRef] [Green Version]
- Fajgenbaum, D.C.; June, C.H. Cytokine Storm. N. Engl. J. Med. 2020, 383, 2255–2273. [Google Scholar] [CrossRef]
- Osuchowski, M.F.; Winkler, M.S.; Skirecki, T.; Cajander, S.; Shankar-Hari, M.; Lachmann, G.; Monneret, G.; Venet, F.; Bauer, M.; Brunkhorst, F.M.; et al. The COVID-19 puzzle: Deciphering pathophysiology and phenotypes of a new disease entity. Lancet Respir. Med. 2021, 9, 622–642. [Google Scholar] [CrossRef]
- Mangalmurti, N.; Hunter, C.A. Cytokine Storms: Understanding COVID-19. Immunity 2020, 53, 19–25. [Google Scholar] [CrossRef]
- de Bruin, S.; Bos, L.D.; van Roon, M.A.; Tuip-de Boer, A.M.; Schuurman, A.R.; Koel-Simmelinck, M.J.A.; Bogaard, H.J.; Tuinman, P.R.; van Agtmael, M.A.; Hamann, J.; et al. Clinical features and prognostic factors in COVID-19: A prospective cohort study. EBioMedicine 2021, 67, 103378. [Google Scholar] [CrossRef] [PubMed]
- Kyriazopoulou, E.; Poulakou, G.; Milionis, H.; Metallidis, S.; Adamis, G.; Tsiakos, K.; Fragkou, A.; Rapti, A.; Damoulari, C.; Fantoni, M.; et al. Early treatment of COVID-19 with anakinra guided by soluble urokinase plasminogen receptor plasma levels: A double-blind, randomized controlled phase 3 trial. Nat. Med. 2021, 27, 1752–1760. [Google Scholar] [CrossRef] [PubMed]
- Karakike, E.; Dalekos, G.N.; Koutsodimitropoulos, I.; Saridaki, M.; Pourzitaki, C.; Papathanakos, G.; Kotsaki, A.; Chalvatzis, S.; Dimakopoulou, V.; Vechlidis, N.; et al. ESCAPE: An Open-Label Trial of Personalized Immunotherapy in Critically lll COVID-19 Patients. J. Innate Immun. 2021, 71, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Kyriazopoulou, E.; Panagopoulos, P.; Metallidis, S.; Dalekos, G.N.; Poulakou, G.; Gatselis, N.; Karakike, E.; Saridaki, M.; Loli, G.; Stefos, A.; et al. An open label trial of anakinra to prevent respiratory failure in COVID-19. eLife 2021, 10, e66125. [Google Scholar] [CrossRef]
- Chen, G.; Wu, D.; Guo, W.; Cao, Y.; Huang, D.; Wang, H.; Wang, T.; Zhang, X.; Chen, H.; Yu, H.; et al. Clinical and immunological features of severe and moderate coronavirus disease 2019. J. Clin. Investig. 2020, 130, 2620–2629. [Google Scholar] [CrossRef] [Green Version]
- Jang, H.J.; Leem, A.Y.; Chung, K.S.; Ahn, J.Y.; Jung, J.Y.; Kang, Y.A.; Park, M.S.; Kim, Y.S.; Lee, S.H. Soluble IL-2R Levels Predict in-Hospital Mortality in COVID-19 Patients with Respiratory Failure. J. Clin. Med. 2021, 10, 4242. [Google Scholar] [CrossRef]
- Kaya, H.; Kaji, M.; Usuda, D. Soluble interleukin-2 receptor levels on admission associated with mortality in coronavirus disease 2019. Int. J. Infect. Dis. 2021, 105, 522–524. [Google Scholar] [CrossRef]
- Ma, A.; Zhang, L.; Ye, X.; Chen, J.; Yu, J.; Zhuang, L.; Weng, C.; Petersen, F.; Wang, Z.; Yu, X. High Levels of Circulating IL-8 and Soluble IL-2R Are Associated With Prolonged Illness in Patients With Severe COVID-19. Front. Immunol. 2021, 12, 626235. [Google Scholar] [CrossRef]
- Quartuccio, L.; Fabris, M.; Sonaglia, A.; Peghin, M.; Domenis, R.; Cifu, A.; Curcio, F.; Tascini, C. Interleukin 6, soluble interleukin 2 receptor alpha (CD25), monocyte colony-stimulating factor, and hepatocyte growth factor linked with systemic hyperinflammation, innate immunity hyperactivation, and organ damage in COVID-19 pneumonia. Cytokine 2021, 140, 155438. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, X.; Li, X.; Xi, D.; Mao, R.; Wu, X.; Cheng, S.; Sun, X.; Yi, C.; Ling, Z.; et al. Potential contribution of increased soluble IL-2R to lymphopenia in COVID-19 patients. Cell Mol. Immunol. 2020, 17, 878–880. [Google Scholar] [CrossRef]
- Kovarik, J.J.; Kampf, A.K.; Gasser, F.; Herdina, A.N.; Breuer, M.; Kaltenecker, C.C.; Wahrmann, M.; Haindl, S.; Mayer, F.; Traby, L.; et al. Identification of Immune Activation Markers in the Early Onset of COVID-19 Infection. Front. Cell Infect. Microbiol. 2021, 11, 651484. [Google Scholar] [CrossRef] [PubMed]
- Hou, H.; Zhang, B.; Huang, H.; Luo, Y.; Wu, S.; Tang, G.; Liu, W.; Mao, L.; Mao, L.; Wang, F.; et al. Using IL-2R/lymphocytes for predicting the clinical progression of patients with COVID-19. Clin. Exp. Immunol. 2020, 201, 76–84. [Google Scholar] [CrossRef] [PubMed]
- Gooding, R.; Riches, P.; Dadian, G.; Moore, J.; Gore, M. Increased soluble interleukin-2 receptor concentration in plasma predicts a decreased cellular response to IL-2. Br. J. Cancer 1995, 72, 452–455. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rubin, L.A.; Kurman, C.C.; Fritz, M.E.; Biddison, W.E.; Boutin, B.; Yarchoan, R.; Nelson, D.L. Soluble interleukin 2 receptors are released from activated human lymphoid cells in vitro. J. Immunol. 1985, 135, 3172–3177. [Google Scholar] [PubMed]
- Bien, E.; Balcerska, A. Serum soluble interleukin 2 receptor alpha in human cancer of adults and children: A review. Biomarkers 2008, 13, 1–26. [Google Scholar] [CrossRef] [PubMed]
- Manoussakis, M.N.; Papadopoulos, G.K.; Drosos, A.A.; Moutsopoulos, H.M. Soluble interleukin 2 receptor molecules in the serum of patients with autoimmune diseases. Clin. Immunol. Immunopathol. 1989, 50, 321–332. [Google Scholar] [CrossRef]
- Wagner, D.K.; Kiwanuka, J.; Edwards, B.K.; Rubin, L.A.; Nelson, D.L.; Magrath, I.T. Soluble interleukin-2 receptor levels in patients with undifferentiated and lymphoblastic lymphomas: Correlation with survival. J. Clin. Oncol. 1987, 5, 1262–1274. [Google Scholar] [CrossRef]
- Dalekos, G.N.; Manoussakis, M.N.; Goussia, A.C.; Tsianos, E.V.; Moutsopoulos, H.M. Soluble interleukin-2 receptors, antineutrophil cytoplasmic antibodies, and other autoantibodies in patients with ulcerative colitis. Gut 1993, 34, 658–664. [Google Scholar] [CrossRef] [Green Version]
- Dalekos, G.N.; Manoussakis, M.N.; Zervou, E.; Tsianos, E.V.; Moutsopoulos, H.M. Immunologic and viral markers in the circulation of anti-HIV negative heroin addicts. Eur J. Clin. Investig. 1993, 23, 219–225. [Google Scholar] [CrossRef]
- Del Valle, D.M.; Kim-Schulze, S.; Huang, H.H.; Beckmann, N.D.; Nirenberg, S.; Wang, B.; Lavin, Y.; Swartz, T.H.; Madduri, D.; Stock, A.; et al. An inflammatory cytokine signature predicts COVID-19 severity and survival. Nat. Med. 2020, 26, 1636–1643. [Google Scholar] [CrossRef]
- Abers, M.S.; Delmonte, O.M.; Ricotta, E.E.; Fintzi, J.; Fink, D.L.; de Jesus, A.A.A.; Zarember, K.A.; Alehashemi, S.; Oikonomou, V.; Desai, J.V.; et al. An immune-based biomarker signature is associated with mortality in COVID-19 patients. JCI Insight 2021, 6, e144455. [Google Scholar] [CrossRef] [PubMed]
- Bell, L.C.K.; Meydan, C.; Kim, J.; Foox, J.; Butler, D.; Mason, C.E.; Shapira, S.D.; Noursadeghi, M.; Pollara, G. Transcriptional response modules characterize IL-1beta and IL-6 activity in COVID-19. iScience 2021, 24, 101896. [Google Scholar] [CrossRef] [PubMed]
- Ali, N. Elevated level of C-reactive protein may be an early marker to predict risk for severity of COVID-19. J. Med. Virol. 2020, 92, 2409–2411. [Google Scholar] [CrossRef] [PubMed]
- Tavakolpour, S.; Rakhshandehroo, T.; Wei, E.X.; Rashidian, M. Lymphopenia during the COVID-19 infection: What it shows and what can be learned. Immunol. Lett. 2020, 225, 31–32. [Google Scholar] [CrossRef]
- Rovina, N.; Akinosoglou, K.; Eugen-Olsen, J.; Hayek, S.; Reiser, J.; Giamarellos-Bourboulis, E.J. Soluble urokinase plasminogen activator receptor (suPAR) as an early predictor of severe respiratory failure in patients with COVID-19 pneumonia. Crit. Care 2020, 24, 187. [Google Scholar] [CrossRef] [PubMed]
- Giamarellos-Bourboulis, E.J.; Poulakou, G.; de Nooijer, A.; Milionis, H.; Metallidis, S.; Ploumidis, M.; Grigoropoulou, P.; Rapti, A.; Segala, F.V.; Bails, E.; et al. Development and validation of SCOPE score: A clinical score to predict progression of COVID-19 pneumonia to severe respiratory failure. Cell Rep. Med. 2022, 3, 100560. [Google Scholar] [CrossRef]
- Huang, I.; Pranata, R. Lymphopenia in severe coronavirus disease-2019 (COVID-19): Systematic review and meta-analysis. J. Intensive Care 2020, 8, 36. [Google Scholar] [CrossRef]
- Bermejo-Martin, J.F.; Martin-Fernandez, M.; Lopez-Mestanza, C.; Duque, P.; Almansa, R. Shared Features of Endothelial Dysfunction between Sepsis and Its Preceding Risk Factors (Aging and Chronic Disease). J. Clin. Med. 2018, 7, 400. [Google Scholar] [CrossRef] [Green Version]
- Xu, H.; Zhong, L.; Deng, J.; Peng, J.; Dan, H.; Zeng, X.; Li, T.; Chen, Q. High expression of ACE2 receptor of 2019-nCoV on the epithelial cells of oral mucosa. Int. J. Oral Sci. 2020, 12, 8. [Google Scholar] [CrossRef]
- Lin, L.; Lu, L.; Cao, W.; Li, T. Hypothesis for potential pathogenesis of SARS-CoV-2 infection-a review of immune changes in patients with viral pneumonia. Emerg. Microbes Infect. 2020, 9, 727–732. [Google Scholar] [CrossRef] [Green Version]
- Liao, Y.C.; Liang, W.G.; Chen, F.W.; Hsu, J.H.; Yang, J.J.; Chang, M.S. IL-19 induces production of IL-6 and TNF-alpha and results in cell apoptosis through TNF-alpha. J. Immunol. 2002, 169, 4288–4297. [Google Scholar] [CrossRef] [PubMed] [Green Version]

| Total (n = 197) | Moderate (n = 65) | Severe (n = 132) | p-Value | |
|---|---|---|---|---|
| Age, median (IQR), years | 61 (21) | 57 (20) | 65 (19) | <0.001 |
| Male sex, n (%) | 120 (60.9%) | 37 (56.9%) | 83 (62.9%) | 0.516 |
| BMI, median (IQR), kg/m2 | 27.7 (6) | 26.4 (5.7) | 27.8 (6.2) | 0.018 |
| Diabetes, n (%) | 38 (19.3%) | 8 (12.3%) | 30 (22.7%) | 0.121 |
| COPD, n (%) | 16 (8.1%) | 3 (4.6%) | 13 (9.8%) | 0.440 |
| Cardiovascular disease, n (%) | 104 (52.8%) | 20 (30.8%) | 84 (63.6%) | 0.001 |
| Smoking, n (%) | 73 (37.1%) | 24 (36.9%) | 49 (37.1%) | 1.000 |
| Disease duration, median (IQR), days | 7 (5) | 7 (6) | 7 (4) | 0.392 |
| pO2/FiO2 ratio, median (IQR) | 304 (127) | 376 (91) | 252 (77) | <0.001 |
| Respiratory rate, median (IQR), /min | 22 (10) | 20 (6) | 24 (10) | <0.001 |
| Lymphocytes, median (IQR), /μL | 950 (595) | 1050 (495) | 885 (633) | 0.005 |
| Ferritin, median (IQR), ng/mL | 477 (800) | 290 (324) | 591 (938) | <0.001 |
| CRP, median (IQR), mg/dL | 3.5 (8.2) | 1.7 (3.7) | 4.7 (9.9) | <0.001 |
| sIL-2R, median (IQR), ng/mL | 5.8 (5.2) | 5.2 (3.4) | 6 (6.2) | 0.017 |
| Univariate Analysis | Multivariate Analysis | |||||
|---|---|---|---|---|---|---|
| HR | 95% CI | p | HR | 95% CI | p | |
| Age | 1.056 | 1.031–1.081 | <0.001 | 1.036 | 1.004–1.070 | 0.030 |
| Male sex | 1.339 | 0.705–2.544 | 0.372 | |||
| BMI | 1.022 | 0.966–1.080 | 0.453 | |||
| Diabetes | 1.983 | 1.031–3.815 | 0.040 | 0.562 | 0.256–1.238 | 0.153 |
| COPD | 0.536 | 0.130–2.218 | 0.390 | |||
| Cardiovascular disease | 3.222 | 1.583–6.557 | 0.001 | 1.706 | 0.745–3.903 | 0.206 |
| Smoking (previous/active) | 1.179 | 0.637–2.183 | 0.601 | |||
| Disease duration | 1.024 | 0.941–1.114 | 0.576 | |||
| pO2/FiO2 ratio | 0.987 | 0.984–0.990 | <0.001 | 0.985 | 0.979–0.990 | <0.001 |
| Lymphocytes | 0.428 | 0.238–0.768 | 0.004 | 0.746 | 0.387–1.438 | 0.382 |
| Ferritin | 1.374 | 1.035–1.824 | 0.028 | 0.833 | 0.584–1.188 | 0.312 |
| CRP | 1.569 | 1.209–2.035 | 0.001 | 0.730 | 0.509–1.048 | 0.088 |
| sIL-2R | 3.251 | 2.171–4.869 | <0.001 | 1.749 | 1.041–2.939 | 0.035 |
| Total (n = 197) | sIL-2R ≥ 9 ng/mL (n = 50) | sIL-2R < 9 ng/mL (n = 147) | p-Value | |
|---|---|---|---|---|
| Age, median (IQR), years | 61 (21) | 70 (16) | 59 (21) | <0.001 |
| Male sex, n (%) | 120 (60.9%) | 18 (36%) | 59 (40.1%) | 0.726 |
| BMI, median (IQR), kg/m2 | 27.7 (6) | 27.9 (5.4) | 27.7 (6.2) | 0.873 |
| Diabetes, n (%) | 38 (19.3%) | 8 (16%) | 30 (20.4%) | 0.635 |
| COPD, n (%) | 16 (8.1%) | 5 (10%) | 11 (7.5%) | 0.558 |
| Cardiovascular disease, n (%) | 104 (52.8%) | 32 (64%) | 72 (49%) | 0.094 |
| Smoking, n (%) | 73 (37.1%) | 20 (40%) | 53 (36.1%) | 0.742 |
| Disease duration, median (IQR), days | 7 (5) | 7 (4) | 7 (5) | 0.391 |
| pO2/FiO2 ratio, median (IQR) | 304 (127) | 238 (147) | 328 (119) | <0.001 |
| Respiratory rate, median (IQR), /min | 22 (10) | 26 (10) | 22 (8) | 0.008 |
| Lymphocytes, median (IQR), /μL | 950 (595) | 800 (453) | 990 (640) | 0.007 |
| Ferritin, median (IQR), ng/mL | 477 (800) | 547 (1261) | 454 (644) | 0.094 |
| CRP, median (IQR), mg/dL | 3.5 (8.2) | 6.8 (12.4) | 2.6 (6.8) | 0.003 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gatselis, N.K.; Lygoura, V.; Lyberopoulou, A.; Giannoulis, G.; Samakidou, A.; Vaiou, A.; Vatidis, G.; Antoniou, K.; Stefos, A.; Georgiadou, S.; et al. Soluble IL-2R Levels at Baseline Predict the Development of Severe Respiratory Failure and Mortality in COVID-19 Patients. Viruses 2022, 14, 787. https://doi.org/10.3390/v14040787
Gatselis NK, Lygoura V, Lyberopoulou A, Giannoulis G, Samakidou A, Vaiou A, Vatidis G, Antoniou K, Stefos A, Georgiadou S, et al. Soluble IL-2R Levels at Baseline Predict the Development of Severe Respiratory Failure and Mortality in COVID-19 Patients. Viruses. 2022; 14(4):787. https://doi.org/10.3390/v14040787
Chicago/Turabian StyleGatselis, Nikolaos K., Vasiliki Lygoura, Aggeliki Lyberopoulou, George Giannoulis, Anna Samakidou, Antonia Vaiou, George Vatidis, Katerina Antoniou, Aggelos Stefos, Sarah Georgiadou, and et al. 2022. "Soluble IL-2R Levels at Baseline Predict the Development of Severe Respiratory Failure and Mortality in COVID-19 Patients" Viruses 14, no. 4: 787. https://doi.org/10.3390/v14040787
APA StyleGatselis, N. K., Lygoura, V., Lyberopoulou, A., Giannoulis, G., Samakidou, A., Vaiou, A., Vatidis, G., Antoniou, K., Stefos, A., Georgiadou, S., Sagris, D., Sveroni, D., Stergioula, D., Gabeta, S., Ntaios, G., & Dalekos, G. N. (2022). Soluble IL-2R Levels at Baseline Predict the Development of Severe Respiratory Failure and Mortality in COVID-19 Patients. Viruses, 14(4), 787. https://doi.org/10.3390/v14040787







