VZV Infection of Primary Human Adrenal Cortical Cells Produces a Proinflammatory Environment without Cell Death
Abstract
:1. Introduction
2. Materials and Methods
2.1. Cells and Virus
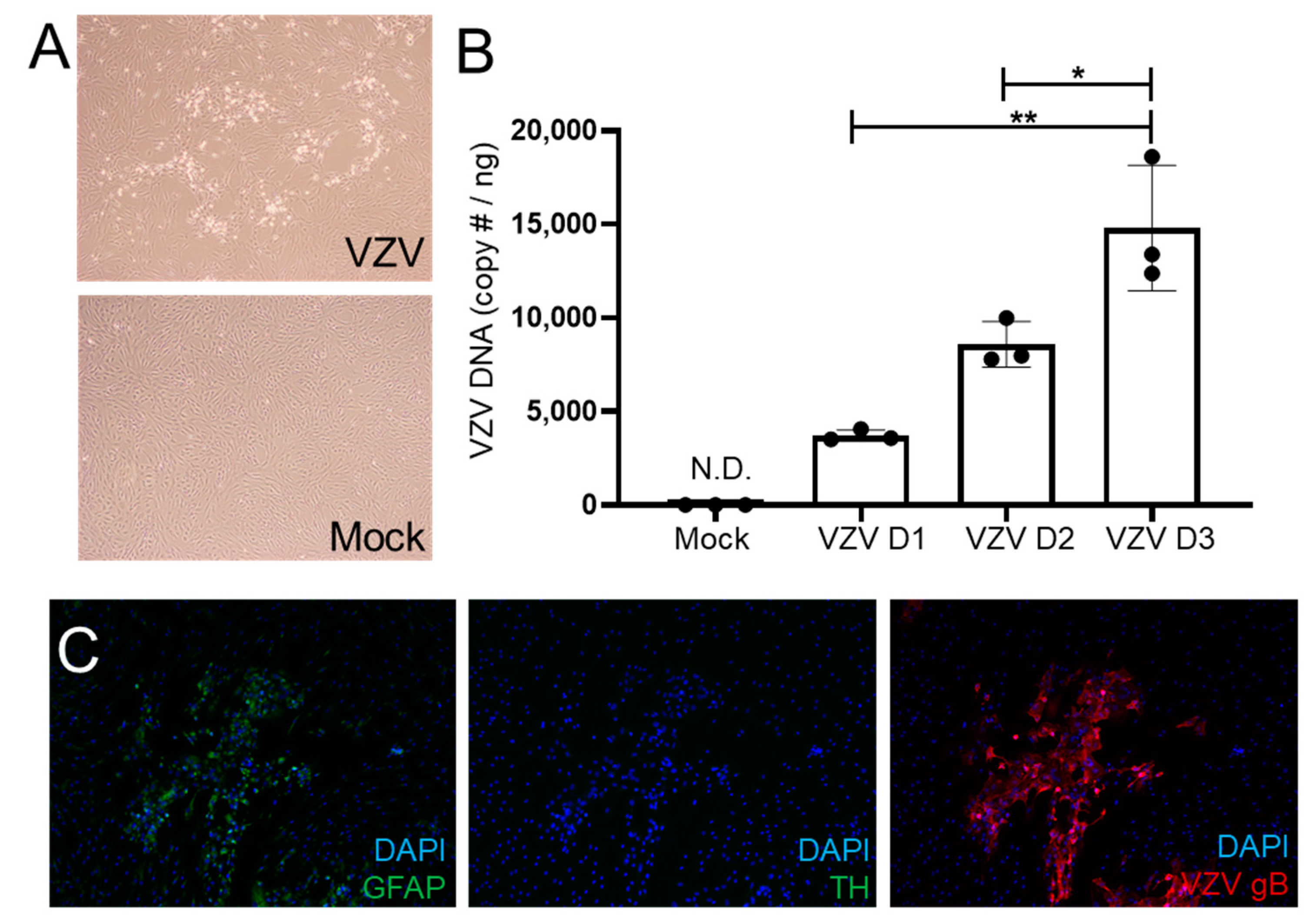
2.2. DNA Extraction and Quantitative PCR (qPCR)
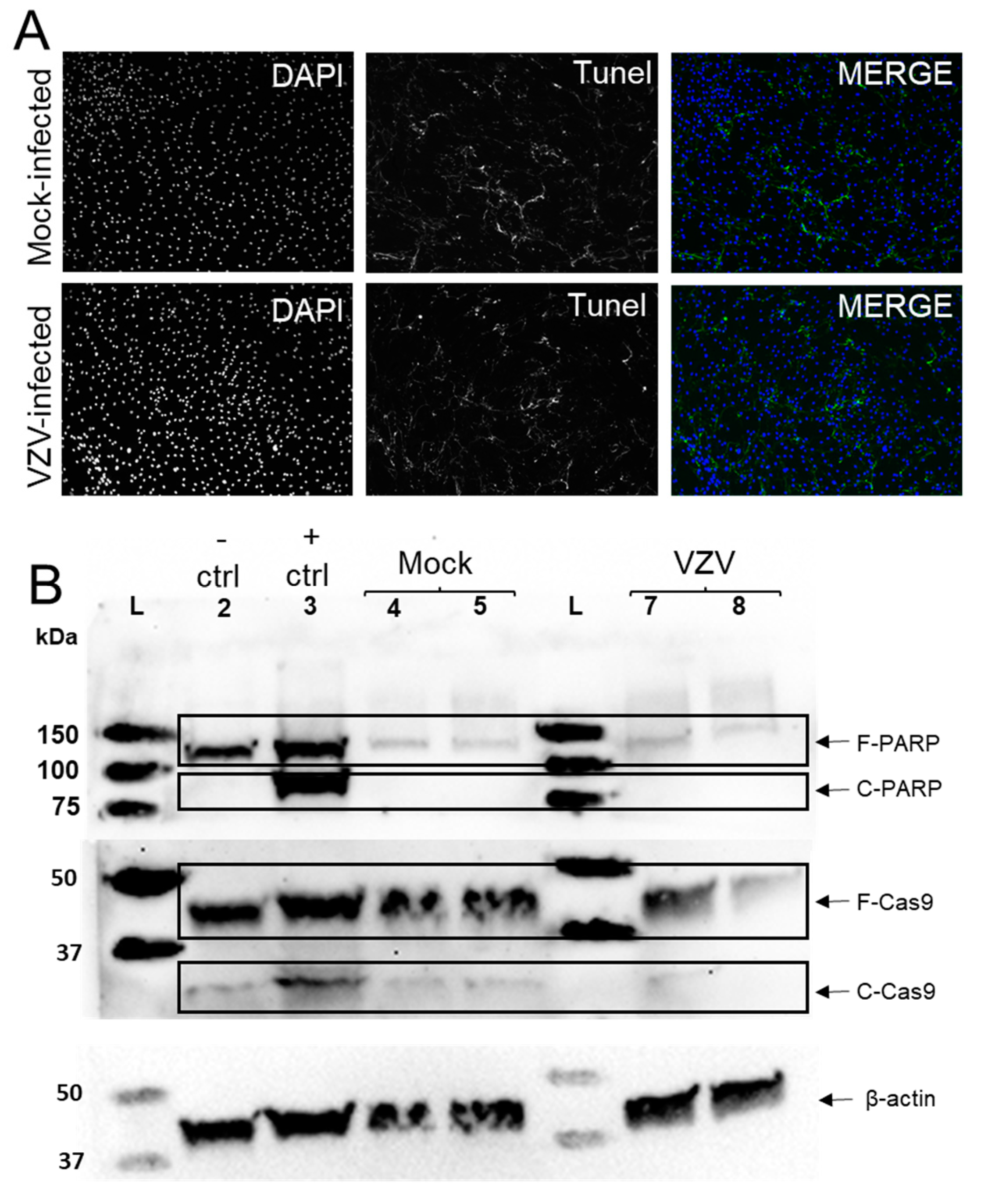
2.3. Immunofluorescent Antibody Assay (IFA)
2.4. Multiplex Electrochemiluminescence Immunoassay
2.5. Western Blot Analysis
2.6. Statistical Analysis
3. Results
3.1. VZV Productively Infects qHAdCCs
3.2. VZV Infection of qHAdCCs Does Not Cause Cell Death
3.3. VZV-Infected qHAdCCs Produce Proinflammatory Cytokines
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Badani, H.; White, T.; Schulick, N.; Raeburn, C.D.; Topkaya, I.; Gilden, D.; Nagel, M.A. Frequency of varicella zoster virus DNA in human adrenal glands. J. Neurovirol. 2016, 22, 400–402. [Google Scholar] [CrossRef] [Green Version]
- Krajewska, B. Two cases of hemorrhage into the suprarerenal glands in the course of varicella. Helv. Paediatr. Acta 1960, 15, 71–73. [Google Scholar]
- Montgomery, R.R.; Ólafsson, M. Waterhouse-Friderichsen syndrome in varicella. Ann. Intern. Med. 1960, 53, 576–579. [Google Scholar] [CrossRef]
- Rudkowski, Z.; Rutowski, R. Zespół Waterhouse-Friderichsena w przebiegu ospy wietrznej u dziecka z toksoplazmoza [Wa-terhouse-Friderichsen syndrome during varicella in a child with toxoplasmosis]. Pediatr. Pol. 1974, 49, 1023–1026. [Google Scholar]
- Heitz, A.F.N.; Hofstee, H.M.A.; Gelinck, L.B.S.; Puylaert, J.B. A rare case of Waterhouse- Friderichsen syndrome during primary varicella zoster. Neth. J. Med. 2017, 75, 351–353. [Google Scholar]
- Niemeyer, C.S.; Mescher, T.; Griggs, R.; Orlicky, D.J.; Wilkerson, G.K.; Bubak, A.N.; Hassell, J.E.J.; Feia, B.; Mahalingam, R.; Traina-Dorge, V.; et al. Histopathological analysis of adrenal glands after simian varicella virus infection. Viruses 2021, 13, 1245. [Google Scholar] [CrossRef]
- Bubak, A.N.; Como, C.N.; Blackmon, A.M.; Jones, D.; Nagel, M.A. Varicella zoster virus differentially alters morphology and suppresses proinflammatory cytokines in primary human spinal cord and hippocampal astrocytes. J. Neuroinflamm. 2018, 15, 318. [Google Scholar] [CrossRef]
- Blackmon, A.M.; Como, C.N.; Bubak, A.N.; Mescher, T.; Jones, D.; Nagel, M.A. Varicella zoster virus alters expression of cell adhesion proteins in human perineurial cells via interleukin 6. J. Infect. Dis. 2019, 220, 1453–1461. [Google Scholar] [CrossRef]
- Bubak, A.N.; Como, C.N.; Blackmon, A.M.; Frietze, S.; Mescher, T.; Jones, D.; Cohrs, R.J.; Paucek, P.; Baird, N.L.; Nagel, M.A. Varicella zoster virus induces nuclear translocation of the neurokinin-1 receptor, promoting lamellipodia formation and viral spread in spinal astrocytes. J. Infect. Dis. 2018, 218, 1324–1335. [Google Scholar] [CrossRef] [Green Version]
- Schindelin, J.; Arganda-Carreras, I.; Frise, E.; Kaynig, V.; Longair, M.; Pietzsch, T.; Preibisch, S.; Rueden, C.; Saalfeld, S.; Schmid, B.; et al. Fiji: An open-source platform for biological-image analysis. Nat. Methods 2012, 9, 676–682. [Google Scholar] [CrossRef] [Green Version]
- Sadzot-Delvaux, C.; Thonard, P.; Schoonbroodt, S.; Piette, J.; Rentierm, B. Varicella-zoster virus induces apoptosis in cell culture. J. Gen. Virol. 1995, 76, 2875–2879. [Google Scholar] [CrossRef] [PubMed]
- Hood, C.; Cunningham, A.L.; Slobedman, B.; Boadle, R.A.; Abendroth, A. Varicella-zoster virus-infected human sensory neurons are resistant to apoptosis, yet human foreskin fibroblasts are susceptible: Evidence for a cell-type-specific apoptotic response. J. Virol. 2003, 77, 12852–12864. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Koenig, A.; Wolff, M.H. Infectibility of separated peripheral blood mononuclear cell subpopulations by varicella-zoster virus (VZV). J. Med. Virol. 2003, 70 (Suppl. 1), S59–S63. [Google Scholar] [CrossRef] [PubMed]
- König, A.; Hömme, C.; Hauröder, B.; Dietrich, A.; Wolff, M.H. The varicella-zoster virus induces apoptosis in vitro in subpopulations of primary human peripheral blood mononuclear cells. Microbes Infect. 2003, 5, 879–889. [Google Scholar] [CrossRef]
- Brazeau, E.; Mahalingam, R.; Gilden, D.; Wellish, M.; Kaufer, B.B.; Osterrieder, N.; Pugazhenthi, S. Varicella-zoster virus–induced apoptosis in MeWo cells is accompanied by down-regulation of Bcl-2 expression. J. Neurovirol. 2010, 16, 133–140. [Google Scholar] [CrossRef]
- Como, C.N.; Bubak, A.N.; Blackmon, A.M.; Jones, D.; Mueller, N.H.; Davidson, R.; Nagel, M.A. Varicella zoster virus induces differential cell-type specific responses in human corneal epithelial cells and keratocytes. Invest. Ophthalmol. Vis. Sci. 2019, 60, 704–711. [Google Scholar] [CrossRef]
- Judd, A.M.; Call, G.B.; Barney, M.; McIlmoil, C.J.; Balls, A.G.; Adams, A.; Oliveira, G.K. Possible function of IL-6 and TNF as intraadrenal factors in the regulation of adrenal steroid secretion. Ann. N. Y. Acad. Sci. 2000, 917, 628–637. [Google Scholar] [CrossRef]
- Steensberg, A.; Fischer, C.P.; Keller, C.; Møller, K.; Pedersen, B.K. IL-6 enhances plasma IL-1ra, IL-10, and cortisol in humans. Am. J. Physiol. Endocrinol. Metab. 2003, 285, E433–E437. [Google Scholar] [CrossRef]
- Jarosinski, K.W.; Carpenter, J.E.; Buckingham, E.M.; Jackson, W.; Knudtson, K.; Moffat, J.F.; Kita, H.; Grose, C. Cellular stress response to varicella-zoster virus infection of human skin includes highly elevated interleukin-6 expression. Infect. Dis. 2018, 5, ofy118. [Google Scholar] [CrossRef] [Green Version]
- Woods, A.M.; Judd, A.M. Interleukin-4 increases cortisol release and decreases adrenal androgen release from bovine adrenal cells. Domest. Anim. Endocrinol. 2008, 34, 372–382. [Google Scholar] [CrossRef]
- Frank, M.; Miguel, Z.; Watkins, L.; Maier, S. Prior exposure to glucocorticoids sensitizes the neuroinflammatory and peripheral inflammatory responses to E. coli lipopolysaccharide. Brain Behav. Immun. 2010, 24, 19–30. [Google Scholar] [CrossRef] [PubMed]
- Derijk, R.; Michelson, D.; Karp, B.; Petrides, J.; Galliven, E.; Deutster, P.; Paciotti, G.; Gold, P.W.; Sternberg, E.M. Exercise and circadian rhythm-induced variations in plasma cortisol differentially regulate interleukin-1 beta (IL-1 beta), IL-6, and tumor necrosis factor-alpha (TNF alpha) production in humans: High sensitivity of TNF alpha and resistance of IL-6. J. Clin. Endocrinol. Metab. 1997, 82, 2182–2191. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ruzek, M.C.; Miller, A.H.; Opal, S.M.; Pearce, B.D.; Biron, C.A. Characterization of early cytokine responses and an interleukin (IL)-6–dependent pathway of endogenous glucocorticoid induction during murine cytomegalovirus infection. J. Exp. Med. 1997, 185, 1185–1192. [Google Scholar] [CrossRef] [PubMed]
- James, S.F.; Mahalingam, R.; Gilden, D. Does apoptosis play a role in varicella zoster virus latency and reactivation? Viruses 2012, 4, 1509–1514. [Google Scholar] [CrossRef]
- Kennedy, P.G.; Graner, M.W.; Gunaydin, D.; Bowlin, J.; Pointon, T.; Yu, X. Varicella-zoster virus infected human neurons are resistant to apoptosis. J. Neurovirol. 2020, 26, 330–337. [Google Scholar] [CrossRef]
- Upadhyay, J.; Sudhindra, P.; Abraham, G.; Trivedi, N. Tuberculosis of the adrenal gland: A case report and review of the literature of infections of the adrenal gland. Int. J. Endocrinol. 2014, 2014, 876037. [Google Scholar] [CrossRef]
- Alevritis, E.M.; Sarubbi, F.A.; Jordan, R.M.; Peiris, A.N. Infectious causes of adrenal insufficiency. South. Med. J. 2003, 96, 96–888. [Google Scholar] [CrossRef]
- Leow, M.K.; Kwek, D.S.; Ng, A.W.; Ong, K.C.; Kawm, G.J.; Leem, L.S. Hypocortisolism in survivors of severe acute respiratory syndrome (SARS). Clin. Endocrinol. 2005, 63, 197–202. [Google Scholar] [CrossRef]
- Paolo, W.F.J.; Nosanchuk, J.D. Adrenal infections. Int. J. Infect. Dis. 2006, 10, 343–353. [Google Scholar] [CrossRef] [Green Version]
- Muehlenbachs, A.; Bhatnagar, J.; Zaki, S.R. Tissue tropism, pathology and pathogenesis of enterovirus infection. J. Pathol. 2015, 235, 217–228. [Google Scholar] [CrossRef] [Green Version]
- Townsend, L.; Dyer, A.H.; Jones, K.; Dunne, J.; Mooney, A.; Gaffney, F.; O’Connor, L.; Leavy, D.; O’Brien, K.; Dowds, J.; et al. Persistent fatigue following SARS-CoV-2 infection is common and independent of severity of initial infection. PLoS ONE 2020, 15, e0240784. [Google Scholar] [CrossRef] [PubMed]
- Moghimi, N.; Di Napoli, M.; Biller, J.; Siegler, J.E.; Shekhar, R.; McCullough, L.D.; Harkins, M.S.; Hong, E.; Alaouieh, D.A.; Mansueto, G.; et al. The neurological manifestations of post-acute sequelae of SARS-CoV-2 infection. Curr. Neurol. Neurosci. Rep. 2021, 21, 44. [Google Scholar] [CrossRef] [PubMed]
- Mao, Y.; Xu, B.; Guan, W.; Xu, D.; Li, F.; Ren, R.; Zhu, X.; Gao, Y.; Jiang, L. The adrenal cortex, an underestimated site of SARS-CoV-2 infection. Front. Endocrinol. 2021, 11, 593179. [Google Scholar] [CrossRef] [PubMed]
- Kanczkowski, W.; Evert, K.; Stadtmüller, M.; Haberecker, M.; Laks, L.; Chen, L.S.; Frontzek, K.; Pablik, J.; Hantel, C.; Beuschlein, F.; et al. COVID-19 targets human adrenal glands. Lancet Diabetes Endocrinol. 2022, 10, 13–16. [Google Scholar] [CrossRef]
- Freire Santana, M.; Borba, M.G.S.; Baía-da-Silva, D.C.; Val, F.; Alexandre, M.A.A.; Brito-Sousa, J.D.; Melo, G.C.; Queiroga, M.V.O.; Leão Farias, M.E.; Camilo, C.C.; et al. Case report: Adrenal pathology findings in severe COVID-19: An autopsy study. Am. J. Trop. Med. Hyg. 2020, 103, 1604–1607. [Google Scholar] [CrossRef]
- Hashim, M.; Athar, S.; Gaba, W.H. New onset adrenal insufficiency in a patient with COVID-19. BMJ Case Rep. 2021, 14, e237690. [Google Scholar] [CrossRef]
- Tsai, S.-Y.; Yang, T.-Y.; Chen, H.-J.; Chen, C.-S.; Lin, W.-M.; Shen, W.-C.; Kuo, C.-N.; Kao, C.-H. Increased risk of chronic fatigue syndrome following herpes zoster: A population-based study. Eur. J. Clin. Microbiol. Infect. Dis. 2014, 33, 1653–1659. [Google Scholar] [CrossRef]
- Shapiro, J.S. Does varicella-zoster virus infection of the peripheral ganglia cause chronic fatigue syndrome? Med. Hypotheses 2009, 73, 728–734. [Google Scholar] [CrossRef]
- Halpin, P.; Williams, M.V.; Klimas, N.G.; Fletcher, M.A.; Barnes, Z.; Ariza, M.E. Myalgic encephalomyelitis/chronic fatigue syndrome and gulf war illness patients exhibit increased humoral responses to the herpesviruses-encoded dUTPase: Implications in disease pathophysiology. J. Med. Virol. 2017, 89, 1636–1645. [Google Scholar] [CrossRef]
- Nagel, M.A.; Gilden, D. The protean neurologic manifestations of varicella-zoster virus infection. Cleve. Clin. J. Med. 2007, 74, 489–494. [Google Scholar] [CrossRef]

| Condition | |||
|---|---|---|---|
| Mock (pg/mL) | VZV (pg/mL) | LPS (pg/mL) | |
| Analyte | M (±SEM) | M (±SEM) | M (±SEM) |
| IFN-γ | N.D. | N.D. | N.D. |
| IL-10 | N.D. | N.D. | 0.061 (0.003) |
| IL-12p70 | N.D. | 0.066 (0.038) | 1.114 (0.04) |
| IL-13 | N.D. | 0.680 (0.389) | 3.733 (0.426) |
| IL-1β | N.D. | N.D. | 0.325 (0.039) |
| IL-2 | N.D. | N.D. | N.D. |
| IL-4 | N.D. | 0.034 (0.019) | 0.477 (0.048) |
| IL-6 | 18.92 (1.09) | 314.51 (28.53) | 1734.03 a (103.6) |
| IL-8 | 5.28 (0.65) | 304.64 (23.07) | 1275.20 a (78.5) |
| TNF-α | N.D. | 0.199 (0.028) | 0.791 (0.06) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Niemeyer, C.S.; Mescher, T.; Bubak, A.N.; Medina, E.M.; Hassell, J.E., Jr.; Nagel, M.A. VZV Infection of Primary Human Adrenal Cortical Cells Produces a Proinflammatory Environment without Cell Death. Viruses 2022, 14, 674. https://doi.org/10.3390/v14040674
Niemeyer CS, Mescher T, Bubak AN, Medina EM, Hassell JE Jr., Nagel MA. VZV Infection of Primary Human Adrenal Cortical Cells Produces a Proinflammatory Environment without Cell Death. Viruses. 2022; 14(4):674. https://doi.org/10.3390/v14040674
Chicago/Turabian StyleNiemeyer, Christy S., Teresa Mescher, Andrew N. Bubak, Eva M. Medina, James E. Hassell, Jr., and Maria A. Nagel. 2022. "VZV Infection of Primary Human Adrenal Cortical Cells Produces a Proinflammatory Environment without Cell Death" Viruses 14, no. 4: 674. https://doi.org/10.3390/v14040674
APA StyleNiemeyer, C. S., Mescher, T., Bubak, A. N., Medina, E. M., Hassell, J. E., Jr., & Nagel, M. A. (2022). VZV Infection of Primary Human Adrenal Cortical Cells Produces a Proinflammatory Environment without Cell Death. Viruses, 14(4), 674. https://doi.org/10.3390/v14040674







